Challenges of the Non-Conventional Yeast Wickerhamomyces anomalus in Winemaking
Abstract
1. Introduction
2. W. anomalus Is a Ubiquitous Yeast Generally Associated with Winemaking
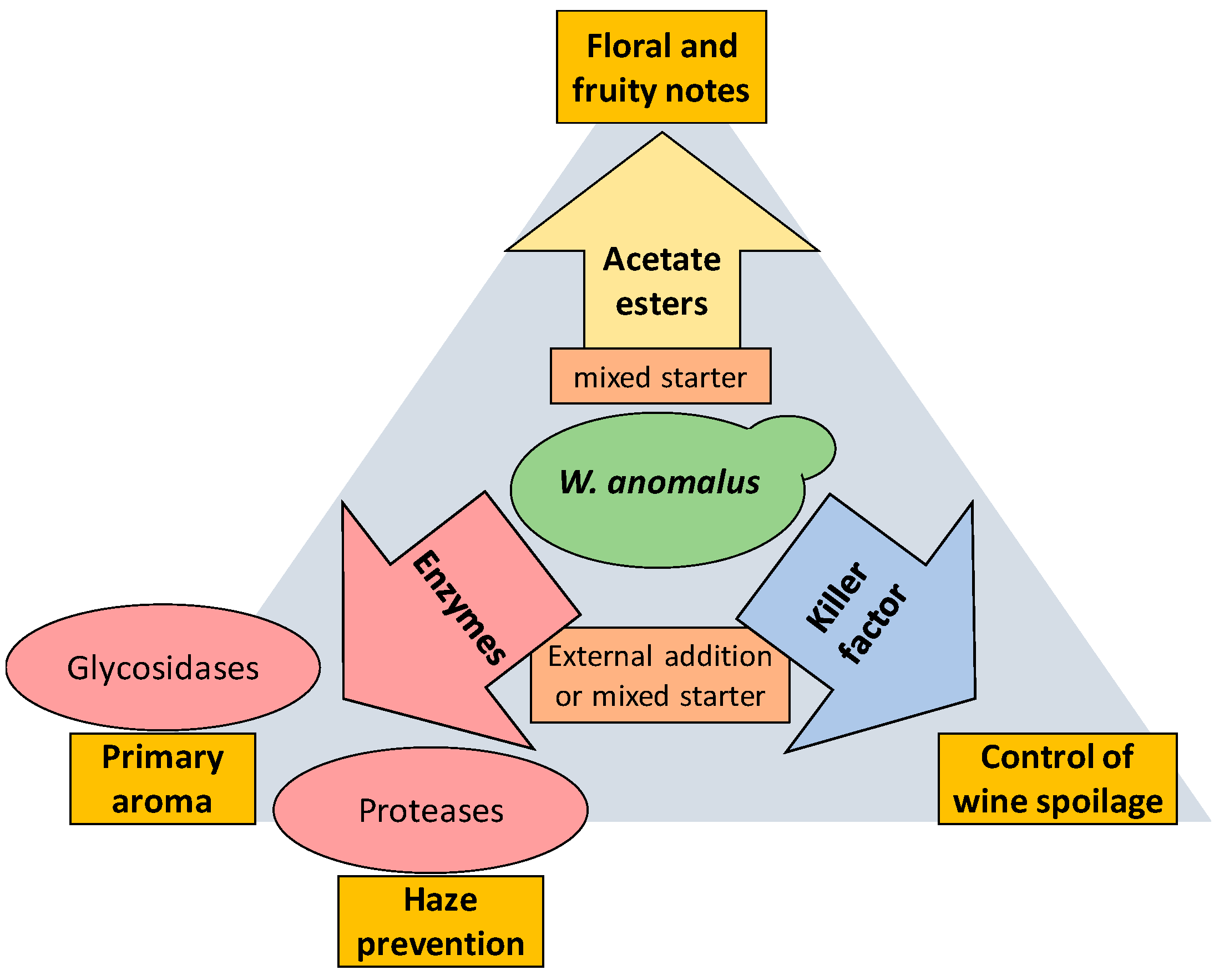
3. W. anomalus Is a Good Producer of Relevant Enzymes for Winemaking
4. W. anomalus Is a Good Producer of Acetate Esters
5. W. anomalus Produces Killer Toxins of Broad Spectrum
6. W. anomalus in Mixed Starters with S. cerevisiae
7. Applications of W. anomalus beyond Wine Fermentation
8. Final Considerations
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fleet, G.H. Wine yeasts for the future. FEMS Yeast Res. 2008, 8, 979–995. [Google Scholar] [CrossRef] [PubMed]
- Comitini, F.; Capece, A.; Ciani, M.; Romano, P. New insights on the use of wine yeasts. Curr. Opin. Food Sci. 2017, 13, 44–49. [Google Scholar] [CrossRef]
- Padilla, B.; Gil, J.V.; Manzanares, P. Past and future of non-Saccharomyces yeasts: From spoilage microorganisms to biotechnological tools for improving wine aroma complexity. Front. Microbiol. 2016, 7, 411. [Google Scholar] [CrossRef] [PubMed]
- Ciani, M.; Morales, P.; Comitini, F.; Tronchoni, J.; Canonico, L.; Curiel, J.A.; Oro, L.; Rodrigues, A.J.; Gonzalez, R. Non-conventional yeast species for lowering ethanol content of wines. Front. Microbiol. 2016, 7, 642. [Google Scholar] [CrossRef] [PubMed]
- Contreras, A.; Hidalgo, C.; Schmidt, S.; Henschke, P.A.; Curtin, C.; Varela, C. The application of non-Saccharomyces yeast in fermentations with limited aeration as a strategy for the production of wine with reduced alcohol content. Int. J. Food Microbiol. 2015, 205, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Englezos, V.; Rantsiou, K.; Cravero, F.; Torchio, F.; Ortiz-Julien, A.; Gerbi, V.; Rolle, L.; Cocolin, L. Starmerella bacillaris and Saccharomyces cerevisiae mixed fermentations to reduce ethanol content in wine. Appl. Microbiol. Biotechnol. 2016, 100, 5515–5526. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, R.; Quiros, M.; Morales, P. Yeast respiration of sugars by non-Saccharomyces yeast species: A promising and barely explored approach to lowering alcohol content of wines. Trends Food Sci. Technol. 2013, 29, 55–61. [Google Scholar] [CrossRef]
- Berbegal, C.; Spano, G.; Fragasso, M.; Grieco, F.; Russo, P.; Capozzi, V. Starter cultures as biocontrol strategy to prevent Brettanomyces bruxellensis proliferation in wine. Appl. Microbiol. Biotechnol. 2018, 102, 569–576. [Google Scholar] [CrossRef] [PubMed]
- Oro, L.; Ciani, M.; Comitini, F. Antimicrobial activity of Metschnikowia pulcherrima on wine yeasts. J. Appl. Microbiol. 2014, 116, 1209–1217. [Google Scholar] [CrossRef] [PubMed]
- Fleet, G.H.; Heard, G.M. Yeast-growth during winemaking. In Wine Microbiology and Biotechnology; Fleet, G.H., Ed.; Harwood Academic Publishers: Chur, Switzerland, 1993; pp. 27–54. [Google Scholar]
- Petruzzi, L.; Capozzi, V.; Berbegal, C.; Corbo, M.R.; Bevilacqua, A.; Spano, G.; Sinigaglia, M. Microbial resources and enological significance: Opportunities and benefits. Front. Microbiol. 2017, 8, 13. [Google Scholar] [CrossRef] [PubMed]
- Kurtzman, C.P. Phylogeny of the ascomycetous yeasts and the renaming of Pichia anomala to Wickerhamomyces anomalus. Antonie van Leeuwenhoek 2011, 99, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Kurtzman, C.P. Pichia e.C. Hansen emend. Kurtzman. In The Yeasts, 4th ed.; Kurtzman, C.P., Fell, J.W., Eds.; Elsevier: Amsterdam, The Netherlands, 1998; pp. 273–352. [Google Scholar]
- Passoth, V.; Fredlund, E.; Druvefors, U.A.; Schnurer, J. Biotechnology, physiology and genetics of the yeast Pichia anomala. FEMS Yeast Res. 2006, 6, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Walker, G.M. Pichia anomala: Cell physiology and biotechnology relative to other yeasts. Antonie van Leeuwenhoek 2011, 99, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Lanciotti, R.; Sinigaglia, M.; Gardini, F.; Elisabetta Guerzoni, M. Hansenula anomala as spoilage agent of cream-filled cakes. Microbiol. Res. 1998, 153, 145–148. [Google Scholar] [CrossRef]
- Tokuoka, K.; Ishitani, T.; Goto, S.; Komagata, K. Identification of yeasts isolated from high-sugar foods. J. Gen. Appl. Microbiol. 1985, 31, 411–427. [Google Scholar] [CrossRef]
- Jonsson, A.; Pahlow, G. Systematic classification and biochemical characterization of yeasts growing in grass silage inoculated with Lactobacillus cultures. Anim. Res. Dev. 1984, 20, 7–22. [Google Scholar]
- Fletcher, E.; Feizi, A.; Kim, S.; Siewers, V.; Nielsen, J. RNA-seq analysis of Pichia anomala reveals important mechanisms required for survival at low pH. Microb. Cell. Fact. 2015, 14, 11. [Google Scholar] [CrossRef] [PubMed]
- Schneider, J.; Rupp, O.; Trost, E.; Jaenicke, S.; Passoth, V.; Goesmann, A.; Tauch, A.; Brinkrolf, K. Genome sequence of Wickerhamomyces anomalus DSM 6766 reveals genetic basis of biotechnologically important antimicrobial activities. FEMS Yeast Res. 2012, 12, 382–386. [Google Scholar] [CrossRef] [PubMed]
- Heard, G.M.; Fleet, G.H. Growth of natural yeast flora during the fermentation of inoculated wines. Appl. Environ. Microbiol. 1985, 50, 727–728. [Google Scholar] [PubMed]
- Díaz, C.; Molina, A.M.; Nähring, J.; Fischer, R. Characterization and dynamic behavior of wild yeast during spontaneous wine fermentation in steel tanks and amphorae. BioMed Res. Int. 2013, 2013, 540465. [Google Scholar] [CrossRef] [PubMed]
- Renouf, V.; Claisse, O.; Lonvaud-Funel, A. Inventory and monitoring of wine microbial consortia. Appl. Microbiol. Biotechnol. 2007, 75, 149–164. [Google Scholar] [CrossRef] [PubMed]
- Sabel, A.; Martens, S.; Petri, A.; Konig, H.; Claus, H. Wickerhamomyces anomalus AS1: A new strain with potential to improve wine aroma. Ann. Microbiol. 2014, 64, 483–491. [Google Scholar] [CrossRef]
- Du Toit, M.; Pretorius, I.S. Microbial spoilage and preservation of wine: Using weapons from nature’s own arsenal—A review. S. Afr. J. Enol. Vitic. 2000, 21, 74–92. [Google Scholar]
- Barata, A.; Malfeito-Ferreira, M.; Loureiro, V. The microbial ecology of wine grape berries. Int. J. Food Microbiol. 2012, 153, 243–259. [Google Scholar] [CrossRef] [PubMed]
- Cordero-Bueso, G.; Arroyo, T.; Serrano, A.; Tello, J.; Aporta, I.; Vélez, M.D.; Valero, E. Influence of the farming system and vine variety on yeast communities associated with grape berries. Int. J. Food Microbiol. 2011, 145, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Bagheri, B.; Bauer, F.F.; Setati, M.E. The diversity and dynamics of indigenous yeast communities in grape must from vineyards employing different agronomic practices and their influence on wine fermentation. S. Afr. J. Enol. Vitic. 2015, 36, 243–251. [Google Scholar] [CrossRef]
- Cordero-Bueso, G.; Esteve-Zarzoso, B.; Cabellos, J.M.; Gil-Diaz, M.; Arroyo, T. Biotechnological potential of non-Saccharomyces yeasts isolated during spontaneous fermentations of Malvar (Vitis vinifera cv. L.). Eur. Food Res. Technol. 2013, 236, 193–207. [Google Scholar] [CrossRef]
- Bagheri, B.; Bauer, F.F.; Setati, M.E. The impact of Saccharomyces cerevisiae on a wine yeast consortium in natural and inoculated fermentations. Front. Microbiol. 2017, 8, 13. [Google Scholar] [CrossRef] [PubMed]
- Ye, M.; Yue, T.; Yuan, Y. Effects of sequential mixed cultures of Wickerhamomyces anomalus and Saccharomyces cerevisiae on apple cider fermentation. FEMS Yeast Res. 2014, 14, 873–882. [Google Scholar] [CrossRef] [PubMed]
- Ocón, E.; Gutiérrez, A.R.; Garijo, P.; López, R.; Santamaría, P. Presence of non-Saccharomyces yeasts in cellar equipment and grape juice during harvest time. Food Microbiol. 2010, 27, 1023–1027. [Google Scholar] [CrossRef] [PubMed]
- Martini, A. Origin and domestication of the wine yeast Saccharomyces cerevisiae. J. Wine Res. 1993, 4, 165–176. [Google Scholar] [CrossRef]
- Ciani, M.; Mannazzu, I.; Marinangeli, P.; Clementi, F.; Martini, A. Contribution of winery-resident Saccharomyces cerevisiae strains to spontaneous grape must fermentation. Antonie van Leeuwenhoek 2004, 85, 159–164. [Google Scholar] [CrossRef] [PubMed]
- Fleet, G.H.; Lafon-Lafourcade, S.; Ribéreau-Gayon, P. Evolution of yeasts and lactic acid bacteria during fermentation and storage of Bordeaux wines. Appl. Environ. Microbiol. 1984, 48, 1034–1038. [Google Scholar] [PubMed]
- Fernández, M.T.; Ubeda, J.F.; Briones, A.I. Comparative study of non-Saccharomyces microflora of musts in fermentation by physiological and molecular methods. FEMS Microbiol. Lett. 1999, 173, 223–229. [Google Scholar] [CrossRef]
- Chavan, P.; Mane, S.; Kulkarni, G.; Shaikh, S.; Ghormade, V.; Nerkar, D.P.; Shouche, Y.; Deshpande, M.V. Natural yeast flora of different varieties of grapes used for wine making in India. Food Microbiol. 2009, 26, 801–808. [Google Scholar] [CrossRef] [PubMed]
- Combina, M.; Elia, A.; Mercado, L.; Catania, C.; Ganga, A.; Martinez, C. Dynamics of indigenous yeast populations during spontaneous fermentation of wines from Mendoza, Argentina. Int. J. Food Microbiol. 2005, 99, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Li, S.-S.; Cheng, C.; Li, Z.; Chen, J.-Y.; Yan, B.; Han, B.-Z.; Reeves, M. Yeast species associated with wine grapes in China. Int. J. Food Microbiol. 2010, 138, 85–90. [Google Scholar] [CrossRef] [PubMed]
- Garofalo, C.; Tristezza, M.; Grieco, F.; Spano, G.; Capozzi, V. From grape berries to wine: Population dynamics of cultivable yeasts associated to “Nero di Troia” autochthonous grape cultivar. World J. Microbiol. Biotechnol. 2016, 32, 10. [Google Scholar] [CrossRef] [PubMed]
- Jolly, N.P.; Pretorius, I.S. The effect of non-Saccharomyces yeasts on fermentation and wine quality. S. Afr. J. Enol. Vitic. 2003, 24, 55–62. [Google Scholar] [CrossRef]
- Nemcová, K.; Breierová, E.; Vadkertiová, R.; Molnarová, J. The diversity of yeasts associated with grapes and musts of the Strekov winegrowing region, Slovakia. Folia Microbiol. 2015, 60, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Mills, D.A.; Johannsen, E.A.; Cocolin, L. Yeast diversity and persistence in Botrytis-affected wine fermentations. Appl. Environ. Microbiol. 2002, 68, 4884–4893. [Google Scholar] [CrossRef] [PubMed]
- Grangeteau, C.; Gerhards, D.; Terrat, S.; Dequiedt, S.; Alexandre, H.; Guilloux-Benatier, M.; von Wallbrunn, C.; Rousseaux, S. FT-IR spectroscopy: A powerful tool for studying the inter- and intraspecific biodiversity of cultivable non-Saccharomyces yeasts isolated from grape must. J. Microbiol. Methods 2016, 121, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Madrigal, T.; Maicas, S.; Tolosa, J.J.M. Glucose and ethanol tolerant enzymes produced by Pichia (Wickerhamomyces) isolates from enological ecosystems. Am. J. Enol. Vitic. 2013, 64, 126–133. [Google Scholar] [CrossRef]
- Rosi, I.; Vinella, M.; Domizio, P. Characterization of β-glucosidase activity in yeasts of oenological origin. J. Appl. Microbiol. 1994, 77, 519–527. [Google Scholar] [CrossRef]
- Charoenchai, C.; Fleet, G.H.; Henschke, P.A.; Todd, B.E.N. Screening of non-Saccharomyces wine yeasts for the presence of extracellular hydrolytic enzymes. Aust. J. Grape Wine Res. 1997, 3, 2–8. [Google Scholar] [CrossRef]
- Manzanares, P.; Rojas, V.; Genovés, S.; Vallés, S. A preliminary search for anthocyanin-β-d-glucosidase activity in non-Saccharomyces wine yeasts. Int. J. Food Sci. Tech. 2000, 35, 95–103. [Google Scholar] [CrossRef]
- Lopez, M.C.; Mateo, J.J.; Maicas, S. Screening of β-glucosidase and β-xylosidase activities in four non-Saccharomyces yeast isolates. J. Food Sci. 2015, 80, C1696–C1704. [Google Scholar] [CrossRef] [PubMed]
- Spagna, G.; Barbagallo, R.N.; Palmeri, R.; Restuccia, C.; Giudici, P. Properties of endogenous β-glucosidase of a Pichia anomala strain isolated from Sicilian musts and wines. Enzym. Microb. Technol. 2002, 31, 1036–1041. [Google Scholar] [CrossRef]
- Manzanares, P.; Ramón, D.; Querol, A. Screening of non-Saccharomyces wine yeasts for the production of β-d-xylosidase activity. Int. J. Food Microbiol. 1999, 46, 105–112. [Google Scholar] [CrossRef]
- Mateo, J.J.; Peris, L.; Ibañez, C.; Maicas, S. Characterization of glycolytic activities from non-Saccharomyces yeasts isolated from Bobal musts. J. Ind. Microbiol. Biotechnol. 2011, 38, 347–354. [Google Scholar] [CrossRef] [PubMed]
- Swangkeaw, J.; Vichitphan, S.; Butzke, C.E.; Vichitphan, K. The characterisation of a novel Pichia anomala β-glucosidase with potentially aroma-enhancing capabilities in wine. Ann. Microbiol. 2009, 59, 335. [Google Scholar] [CrossRef]
- Swangkeaw, J.; Vichitphan, S.; Butzke, C.E.; Vichitphan, K. Characterization of β-glucosidases from Hanseniaspora sp. and Pichia anomala with potentially aroma-enhancing capabilities in juice and wine. World J. Microbiol. Biotechnol. 2011, 27, 423–430. [Google Scholar] [CrossRef]
- Schwentke, J.; Sabel, A.; Petri, A.; Konig, H.; Claus, H. The yeast Wickerhamomyces anomalus AS1 secretes a multifunctional exo-β-1,3-glucanase with implications for winemaking. Yeast 2014, 31, 349–359. [Google Scholar] [CrossRef] [PubMed]
- Gil, J.V.; Manzanares, P.; Genovés, S.; Vallés, S.; González-Candelas, L. Over-production of the major exoglucanase of Saccharomyces cerevisiae leads to an increase in the aroma of wine. Int. J. Food Microbiol. 2005, 103, 57–68. [Google Scholar] [CrossRef] [PubMed]
- Van Rensburg, P.; Pretorius, I.S. Enzymes in winemaking: Harnessing natural catalysts for efficient biotransformations—A review. S. Afr. J. Enol. Vitic. 2000, 21, 52–73. [Google Scholar]
- Schlander, M.; Distler, U.; Tenzer, S.; Thines, E.; Claus, H. Purification and properties of yeast proteases secreted by Wickerhamomyces anomalus 227 and Metschnikovia pulcherrima 446 during growth in a white grape juice. Fermentation 2017, 3, 2. [Google Scholar] [CrossRef]
- Viana, F.; Gil, J.V.; Genoves, S.; Valles, S.; Manzanares, P. Rational selection of non-Saccharomyces wine yeasts for mixed starters based on ester formation and enological traits. Food Microbiol. 2008, 25, 778–785. [Google Scholar] [CrossRef] [PubMed]
- Lambretchts, M.G.; Pretorius, I.S. Yeast and its importance to wine aroma—A review. S. Afr. J. Enol. Vitic. 2000, 21, 97–129. [Google Scholar]
- Domizio, P.; Romani, C.; Lencioni, L.; Comitini, F.; Gobbi, M.; Mannazzu, I.; Ciani, M. Outlining a future for non-Saccharomyces yeasts: Selection of putative spoilage wine strains to be used in association with Saccharomyces cerevisiae for grape juice fermentation. Int. J. Food Microbiol. 2011, 147, 170–180. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.L.; Chi, Z.; Wang, G.Y.; Wang, Z.P.; Li, Y.; Chi, Z.M. Yeast killer toxins, molecular mechanisms of their action and their applications. Crit. Rev. Biotechnol. 2015, 35, 222–234. [Google Scholar] [CrossRef] [PubMed]
- Mehlomakulu, N.N.; Setati, M.E.; Divol, B. Non-Saccharomyces killer toxins: Possible biocontrol agents against Brettanomyces in wine? S. Afr. J. Enol. Vitic. 2015, 36, 94–104. [Google Scholar] [CrossRef]
- Passoth, V.; Olstorpe, M.; Schnürer, J. Past, present and future research directions with Pichia anomala. Antonie van Leeuwenhoek 2011, 99, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Ciani, M.; Comitini, F. Non-Saccharomyces wine yeasts have a promising role in biotechnological approaches to winemaking. Ann. Microbiol. 2011, 61, 25–32. [Google Scholar] [CrossRef]
- Fernández de Ullivarri, M.; Mendoza, L.M.; Raya, R.R. Characterization of the killer toxin KTCf20 from Wickerhamomyces anomalus, a potential biocontrol agent against wine spoilage yeasts. Biol. Control 2018, 121, 223–228. [Google Scholar] [CrossRef]
- Lopes, C.A.; Sáez, J.S.; Sangorrín, M.P. Differential response of Pichia guilliermondii spoilage isolates to biological and physico-chemical factors prevailing in Patagonian wine fermentations. Can. J. Microbiol. 2009, 55, 801–809. [Google Scholar] [CrossRef] [PubMed]
- Sangorrín, M.P.; Lopes, C.A.; Jofré, V.; Querol, A.; Caballero, A.C. Spoilage yeasts from Patagonian cellars: Characterization and potential biocontrol based on killer interactions. World J. Microbiol. Biotechnol. 2008, 24, 945–953. [Google Scholar] [CrossRef]
- Comitini, F.; De, J.I.; Pepe, L.; Mannazzu, I.; Ciani, M. Pichia anomala and Kluyveromyces wickerhamii killer toxins as new tools against Dekkera/Brettanomyces spoilage yeasts. FEMS Microbiol. Lett. 2004, 238, 235–240. [Google Scholar] [CrossRef] [PubMed]
- De Ingeniis, J.; Raffaelli, N.; Ciani, M.; Mannazzu, I. Pichia anomala DBVPG 3003 secretes a ubiquitin-like protein that has antimicrobial activity. Appl. Environ. Microbiol. 2009, 75, 1129–1134. [Google Scholar] [CrossRef] [PubMed]
- Oro, L.; Ciani, M.; Bizzaro, D.; Comitini, F. Evaluation of damage induced by Kwkt and Pikt zymocins against Brettanomyces/Dekkera spoilage yeast, as compared to sulphur dioxide. J. Appl. Microbiol. 2016, 121, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Csutak, O.; Vassu, T.; Corbu, V.; Cirpici, I.; Ionescu, R. Killer activity of Pichia anomala CMGB 88. Biointerface Res. Appl. Chem. 2017, 7, 2085–2089. [Google Scholar]
- Andorrà, I.; Berradre, M.; Mas, A.; Esteve-Zarzoso, B.; Guillamón, J.M. Effect of mixed culture fermentations on yeast populations and aroma profile. LWT-Food Sci. Technol. 2012, 49, 8–13. [Google Scholar] [CrossRef]
- Gil, J.V.; Mateo, J.J.; Jiménez, M.; Pastor, A.; Huerta, T. Aroma compounds in wine as influenced by apiculate yeasts. J. Food Sci. 1996, 61, 1247–1249. [Google Scholar] [CrossRef]
- Gobbi, M.; Comitini, F.; Domizio, P.; Romani, C.; Lencioni, L.; Mannazzu, I.; Ciani, M. Lachancea thermotolerans and Saccharomyces cerevisiae in simultaneous and sequential co-fermentation: A strategy to enhance acidity and improve the overall quality of wine. Food Microbiol. 2013, 33, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Quirós, M.; Rojas, V.; Gonzalez, R.; Morales, P. Selection of non-Saccharomyces yeast strains for reducing alcohol levels in wine by sugar respiration. Int. J. Food Microbiol. 2014, 181, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Canonico, L.; Agarbatu, A.; Comitini, F.; Ciani, M. Torulaspora delbrueckii in the brewing process: A new approach to enhance bioflavour and to reduce ethanol content. Food Microbiol. 2016, 56, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Contreras, A.; Hidalgo, C.; Henschke, P.A.; Chambers, P.J.; Curtin, C.; Varela, C. Evaluation of non-Saccharomyces yeasts for the reduction of alcohol content in wine. Appl. Environ. Microbiol. 2011, 80, 1670–1678. [Google Scholar] [CrossRef] [PubMed]
- Rojas, V.; Gil, J.V.; Piñaga, F.; Manzanares, P. Acetate ester formation in wine by mixed cultures in laboratory fermentations. Int. J. Food Microbiol. 2003, 86, 181–188. [Google Scholar] [CrossRef]
- Bely, M.; Stoeckle, P.; Masneuf-Pomarede, I.; Dubourdieu, D. Impact of mixed Torulaspora delbrueckii-Saccharomyces cerevisiae culture on high-sugar fermentation. Int. J. Food Microbiol. 2008, 122, 312–320. [Google Scholar] [CrossRef] [PubMed]
- Anfang, N.; Brajkovich, M.; Goddard, M.R. Co-fermentation with Pichia kluyveri increases varietal thiol concentrations in Sauvignon blanc. Aust. J. Grape Wine Res. 2009, 15, 1–8. [Google Scholar] [CrossRef]
- Viana, F.; Gil, J.V.; Valles, S.; Manzanares, P. Increasing the levels of 2-phenylethyl acetate in wine through the use of a mixed culture of Hanseniaspora osmophila and Saccharomyces cerevisiae. Int. J. Food Microbiol. 2009, 135, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Izquierdo Cañas, P.; García-Romero, E.; Heras Manso, J.; Fernández-González, M. Influence of sequential inoculation of Wickerhamomyces anomalus and Saccharomyces cerevisiae in the quality of red wines. Eur. Food Res. Technol. 2014, 239, 279–286. [Google Scholar] [CrossRef]
- Izquierdo Cañas, P.M.I.; Garcia, A.T.P.; Romero, E.G. Enhancement of flavour properties in wines using sequential inoculations of non-Saccharomyces (Hansenula and Torulaspora) and Saccharomyces yeast starter. Vitis 2011, 50, 177–182. [Google Scholar]
- Kurita, O. Increase of acetate ester-hydrolysing esterase activity in mixed cultures of Saccharomyces cerevisiae and Pichia anomala. J. Appl. Microbiol. 2008, 104, 1051–1058. [Google Scholar] [CrossRef] [PubMed]
- Luan, Y.; Zhang, B.Q.; Duan, C.Q.; Yan, G.L. Effects of different pre-fermentation cold maceration time on aroma compounds of Saccharomyces cerevisiae co-fermentation with Hanseniaspora opuntiae or Pichia kudriavzevii. LWT-Food Sci. Technol. 2018, 92, 177–186. [Google Scholar] [CrossRef]
- Gobert, A.; Tourdot-Marechal, R.; Morge, C.; Sparrow, C.; Liu, Y.Z.; Quintanilla-Casas, B.; Vichi, S.; Alexandre, H. Non-Saccharomyces yeasts nitrogen source preferences: Impact on sequential fermentation and wine volatile compounds profile. Front. Microbiol. 2017, 8, 2175. [Google Scholar] [CrossRef] [PubMed]
- Rollero, S.; Bloem, A.; Ortiz-Julien, A.; Camarasa, C.; Divol, B. Altered fermentation performances, growth, and metabolic footprints reveal competition for nutrients between yeast species inoculated in synthetic grape juice-like medium. Front. Microbiol. 2018, 9, 196. [Google Scholar] [CrossRef] [PubMed]
- Chasseriaud, L.; Coulon, J.; Marullo, P.; Albertin, W.; Bely, M. New oenological practice to promote non-Saccharomyces species of interest: Saturating grape juice with carbon dioxide. Appl. Microbiol. Biotechnol. 2018, 102, 3779–3791. [Google Scholar] [CrossRef] [PubMed]
- Beckner Whitener, M.E.; Stanstrup, J.; Panzeri, V.; Carlin, S.; Divol, B.; Du Toit, M.; Vrhovsek, U. Untangling the wine metabolome by combining untargeted SPME–GCxGC-TOF-MS and sensory analysis to profile Sauvignon blanc co-fermented with seven different yeasts. Metabolomics 2016, 12, 53. [Google Scholar] [CrossRef]
- Domizio, P.; Romani, C.; Comitini, F.; Gobi, M.; Lencioni, L.; Mannazzu, I.; Ciani, M. Potential spoilage non-Saccharomyces yeasts in mixed cultures with Saccharomyces cerevisiae. Ann. Microbiol. 2011, 61, 137–144. [Google Scholar] [CrossRef]
- Basso, R.F.; Alcarde, A.R.; Portugal, C.B. Could non-Saccharomyces yeasts contribute on innovative brewing fermentations? Food Res. Int. 2016, 86, 112–120. [Google Scholar] [CrossRef]
- Duarte, W.F.; Amorim, J.C.; Schwan, R.F. The effects of co-culturing non-Saccharomyces yeasts with S. cerevisiae on the sugar cane spirit (cachaça) fermentation process. Antonie van Leeuwenhoek 2013, 103, 175–194. [Google Scholar] [CrossRef] [PubMed]
- Zha, M.; Sun, B.; Wu, Y.; Yin, S.; Wang, C. Improving flavor metabolism of Saccharomyces cerevisiae by mixed culture with Wickerhamomyces anomalus for Chinese Baijiu making. J. Biosci. Bioeng. 2018, 126, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Wahyono, A.; Lee, S.B.; Kang, W.W.; Park, H.D. Improving bread quality using co-cultures of Saccharomyces cerevisiae, Torulaspora delbrueckii JK08, and Pichia anomala JK04. Ital. J. Food Sci. 2016, 28, 298–313. [Google Scholar]
- Huyben, D.; Nyman, A.; Vidakovic, A.; Passoth, V.; Moccia, R.; Kiessling, A.; Dicksved, J.; Lundh, T. Effects of dietary inclusion of the yeasts Saccharomyces cerevisiae and Wickerhamomyces anomalus on gut microbiota of rainbow trout. Aquaculture 2017, 473, 528–537. [Google Scholar] [CrossRef]
- Olstorpe, M.; Passoth, V. Pichia anomala in grain biopreservation. Antonie van Leeuwenhoek 2011, 99, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Olstorpe, M.; Borling, J.; Schnürer, J.; Passoth, V. Pichia anomala yeast improves feed hygiene during storage of moist crimped barley grain under swedish farm conditions. Anim. Feed Sci. Technol. 2010, 156, 47–56. [Google Scholar] [CrossRef]
- Buzzini, P.; Martini, A. Large-scale screening of selected Candida maltosa, Debaryomyces hansenii and Pichia anomala killer toxin activity against pathogenic yeasts. Med. Mycol. 2001, 39, 479–482. [Google Scholar] [CrossRef] [PubMed]
- Zha, Y.; Hossain, A.H.; Tobola, F.; Sedee, N.; Havekes, M.; Punt, P.J. Pichia anomala 29X: A resistant strain for lignocellulosic biomass hydrolysate fermentation. FEMS Yeast Res. 2013, 13, 609–617. [Google Scholar] [CrossRef] [PubMed]
- Ruyters, S.; Mukherjee, V.; Verstrepen, K.J.; Thevelein, J.M.; Willems, K.A.; Lievens, B. Assessing the potential of wild yeasts for bioethanol production. J. Ind. Microbiol. Biotechnol. 2015, 42, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Elliston, A.; Le Gall, G.; Colquhoun, I.J.; Collins, S.R.A.; Dicks, J.; Roberts, I.N.; Waldron, K.W. Yeast diversity in relation to the production of fuels and chemicals. Sci. Rep. 2017, 7, 11. [Google Scholar] [CrossRef] [PubMed]
- Kruis, A.J.; Levisson, M.; Mars, A.E.; van der Ploeg, M.; Daza, F.G.; Ellena, V.; Kengen, S.W.M.; van der Oost, J.; Weusthuis, R.A. Ethyl acetate production by the elusive alcohol acetyltransferase from yeast. Metab. Eng. 2017, 41, 92–101. [Google Scholar] [CrossRef] [PubMed]
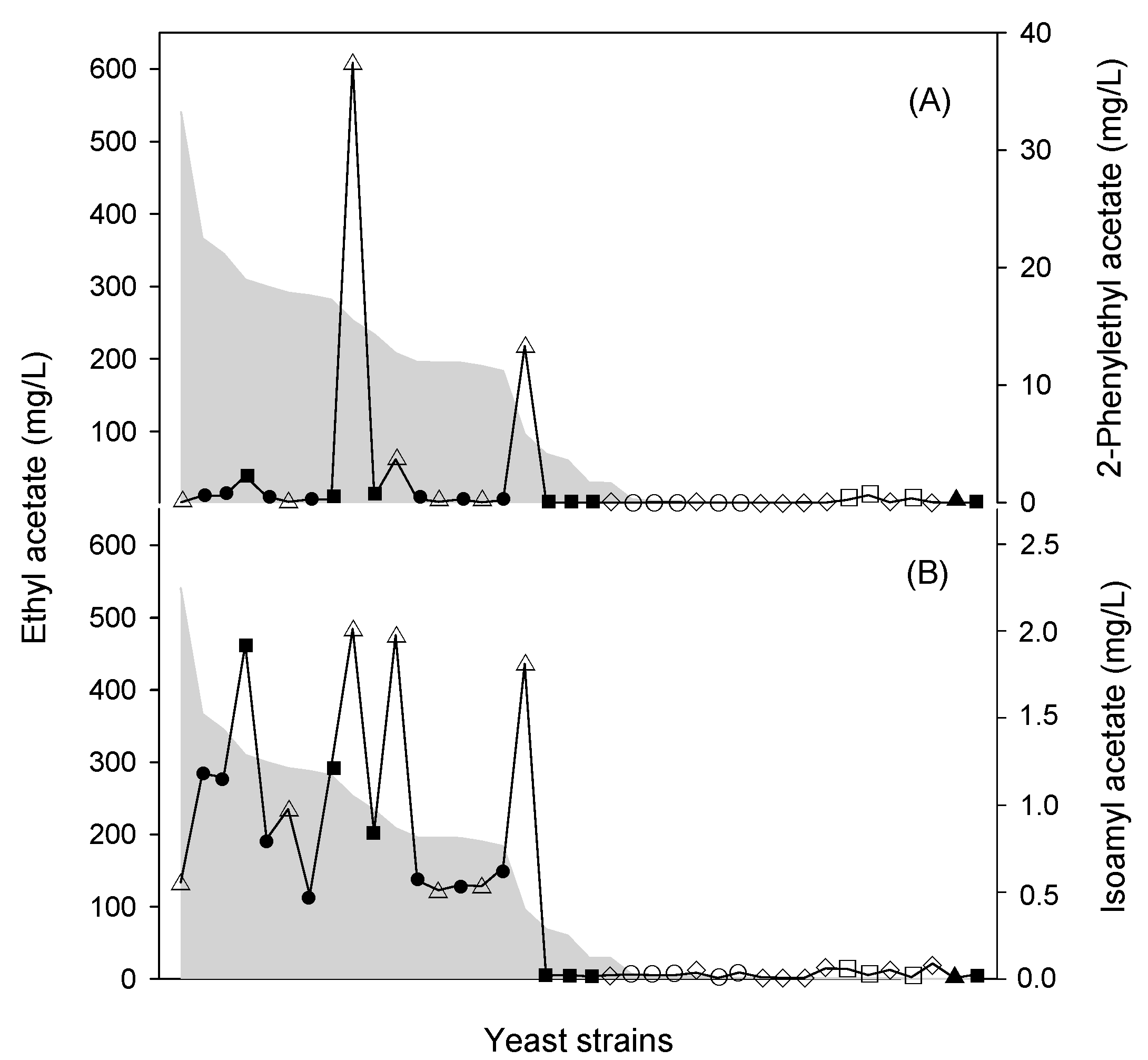
| Mixed Starter | Impact on Wine | Inoculation | Must | Ref. |
|---|---|---|---|---|
| P. anomala/S. cerevisiae | Isoamyl acetate increase | Co-inoculation | Bobal | [79] |
| Isoamyl acetate increase | Co-inoculation | Commercial | [61] | |
| Acetate ester increase | Co-inoculation | Synthetic | [85] | |
| Acetate ester increase and alcohol decrease | Sequential | Airén | [84] | |
| Acetate and ethyl ester increase | Sequential | Mazuela | [83] | |
| P. kudriavzevii/S. cerevisiae | Isoamyl acetate increase | Co-inoculation | Cabernet | [86] |
| Sauvignon | ||||
| P. membranifaciens/S. cerevisiae | Isoamyl and 2-phenetyl acetate | Sequential | Muscat | [87] |
| P. burtonii/S. cerevisiae | Ethyl ester increase | Sequential | Synthetic | [88] |
| P. kluyveri/S. cerevisiae | 3-Mercaptohexyl acetate increase | Co-inoculation | Sauvignon Blanc | [81] |
| 3-Sulfanylhexan-1-ol increase | Co-inoculation | Sauvignon Blanc | [89] | |
| Off-flavor formation | Sequential | Sauvignon Blanc | [90] | |
| P. fermentans/S. cerevisiae | Polysaccharide increase | Co-inoculation | Commercial | [91] |
| Application | Yeast Strain | Reference |
|---|---|---|
| Food and beverage production | ||
| beer | W. anomalus1 | [92] |
| cider | W. anomalus YN6 | [31] |
| cachaça | P. anomala UFLA CAF70 and CAF119 | [93] |
| Chinese Baijiu | W. anomalus GZ3 | [94] |
| bread | P. anomala JK04 | [95] |
| Aquaculture | W. anomalus1 | [96] |
| Biocontrol | ||
| cereal grain preservation | P. anomala J121 | [97,98] |
| antimycotic agent | P. anomala C33, C85, Di8, Di28, DBVPG3649 | [99] |
| P. anomala CMGB88 | [72] | |
| Production of fuels and chemicals | ||
| bioethanol | P. anomala CBS132101 | [100] |
| W. anomalus1 | [101] | |
| ethyl acetate | W. anomalus NCYC16 | [102] |
| W. anomalus DSM 6766 | [103] | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Padilla, B.; Gil, J.V.; Manzanares, P. Challenges of the Non-Conventional Yeast Wickerhamomyces anomalus in Winemaking. Fermentation 2018, 4, 68. https://doi.org/10.3390/fermentation4030068
Padilla B, Gil JV, Manzanares P. Challenges of the Non-Conventional Yeast Wickerhamomyces anomalus in Winemaking. Fermentation. 2018; 4(3):68. https://doi.org/10.3390/fermentation4030068
Chicago/Turabian StylePadilla, Beatriz, Jose V. Gil, and Paloma Manzanares. 2018. "Challenges of the Non-Conventional Yeast Wickerhamomyces anomalus in Winemaking" Fermentation 4, no. 3: 68. https://doi.org/10.3390/fermentation4030068
APA StylePadilla, B., Gil, J. V., & Manzanares, P. (2018). Challenges of the Non-Conventional Yeast Wickerhamomyces anomalus in Winemaking. Fermentation, 4(3), 68. https://doi.org/10.3390/fermentation4030068





