Current Role of Lipoprotein Apheresis in the Treatment of High-Risk Patients
Abstract
:1. Introduction
2. Homozygous Familial HCH
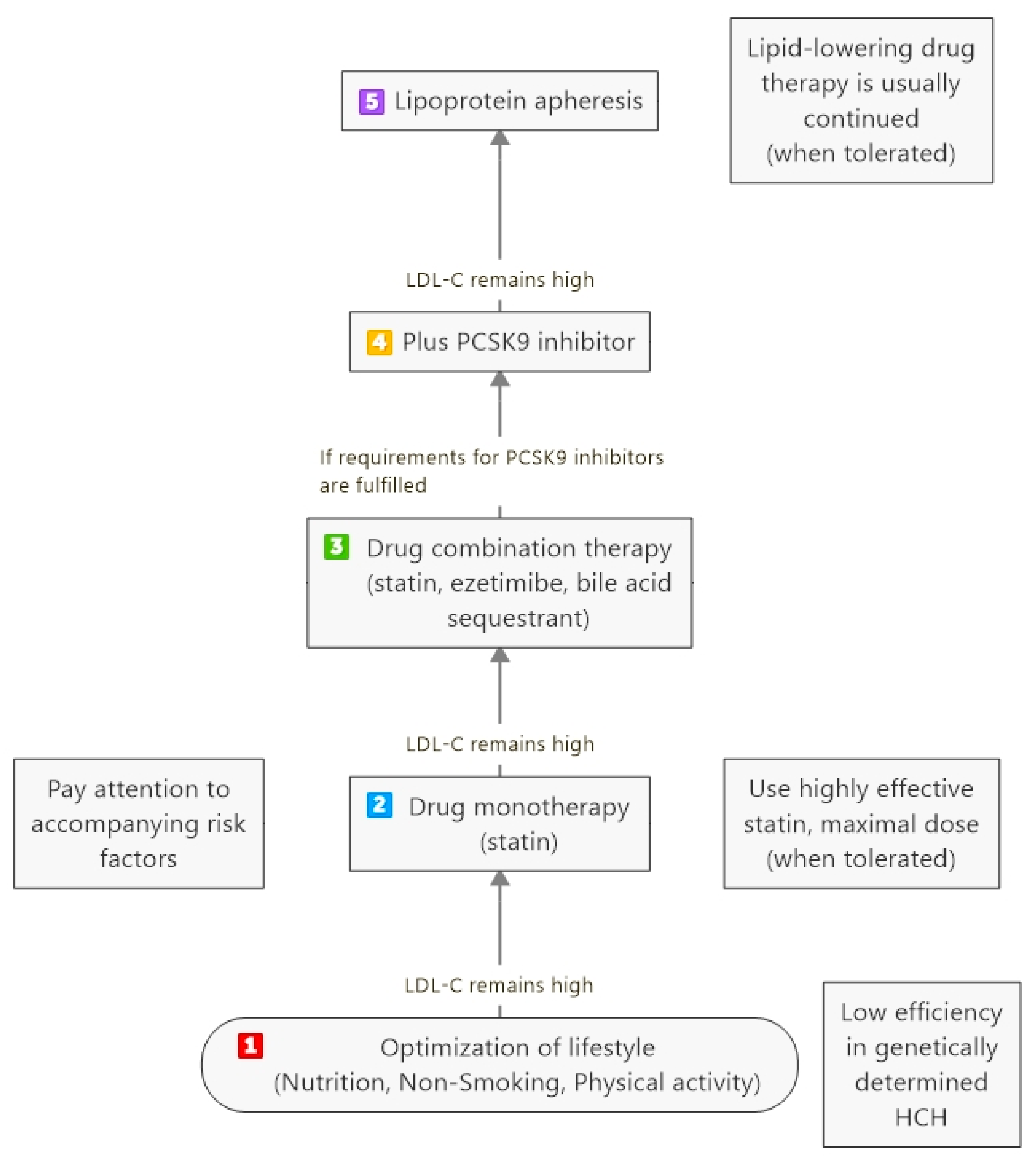
3. Severe Hypercholesterolemia (HCH)
4. Isolated Elevation of Lp(a)
- Lp(a) levels are extremely high.
- A positive family history with early cardiovascular events in first-degree relatives is often seen.
- Patients suffer from a first severe cardiovascular event (reanimation needed after an acute myocardial infarction) already at young age (before 50).
- Many patients underwent several interventions (PTCA, PCI) before the diagnosis of an elevation of Lp(a) is made.
- Very often they were heavy smokers—the combination of smoking and high Lp(a) is especially atherogenic.
- Patients have a concomitant hypertension.
5. Lipoprotein Apheresis—What Is the Evidence
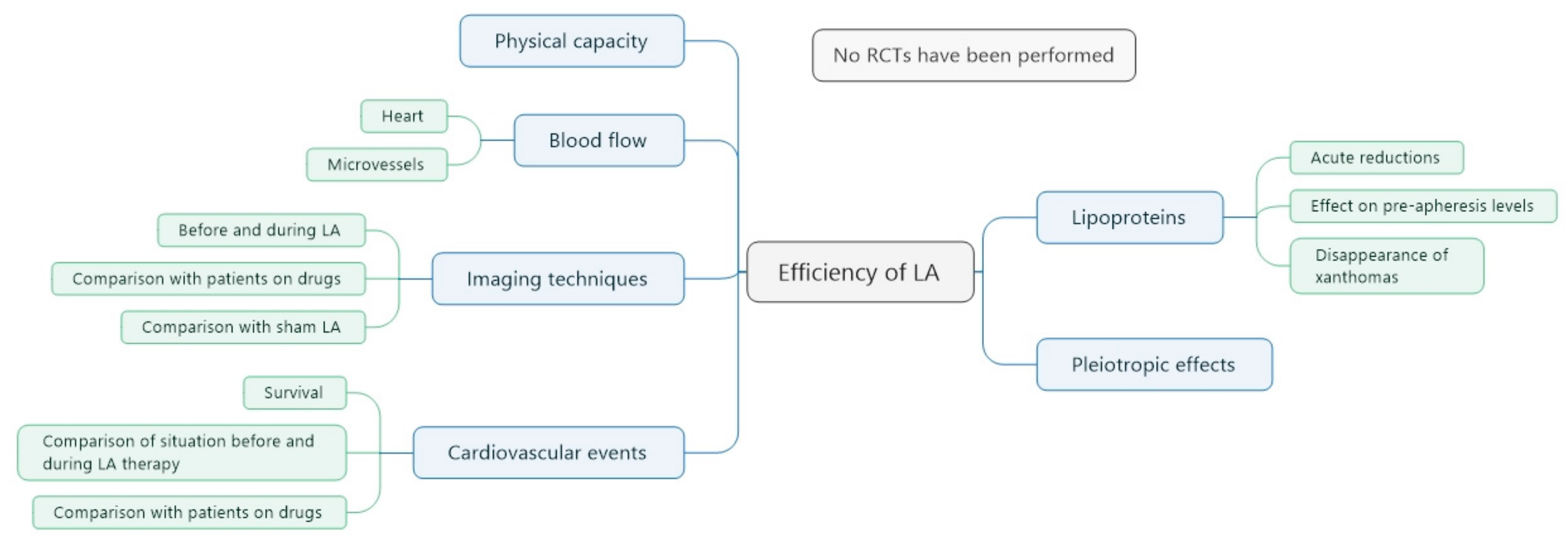
5.1. Evidence for the Effects of LA Treatment
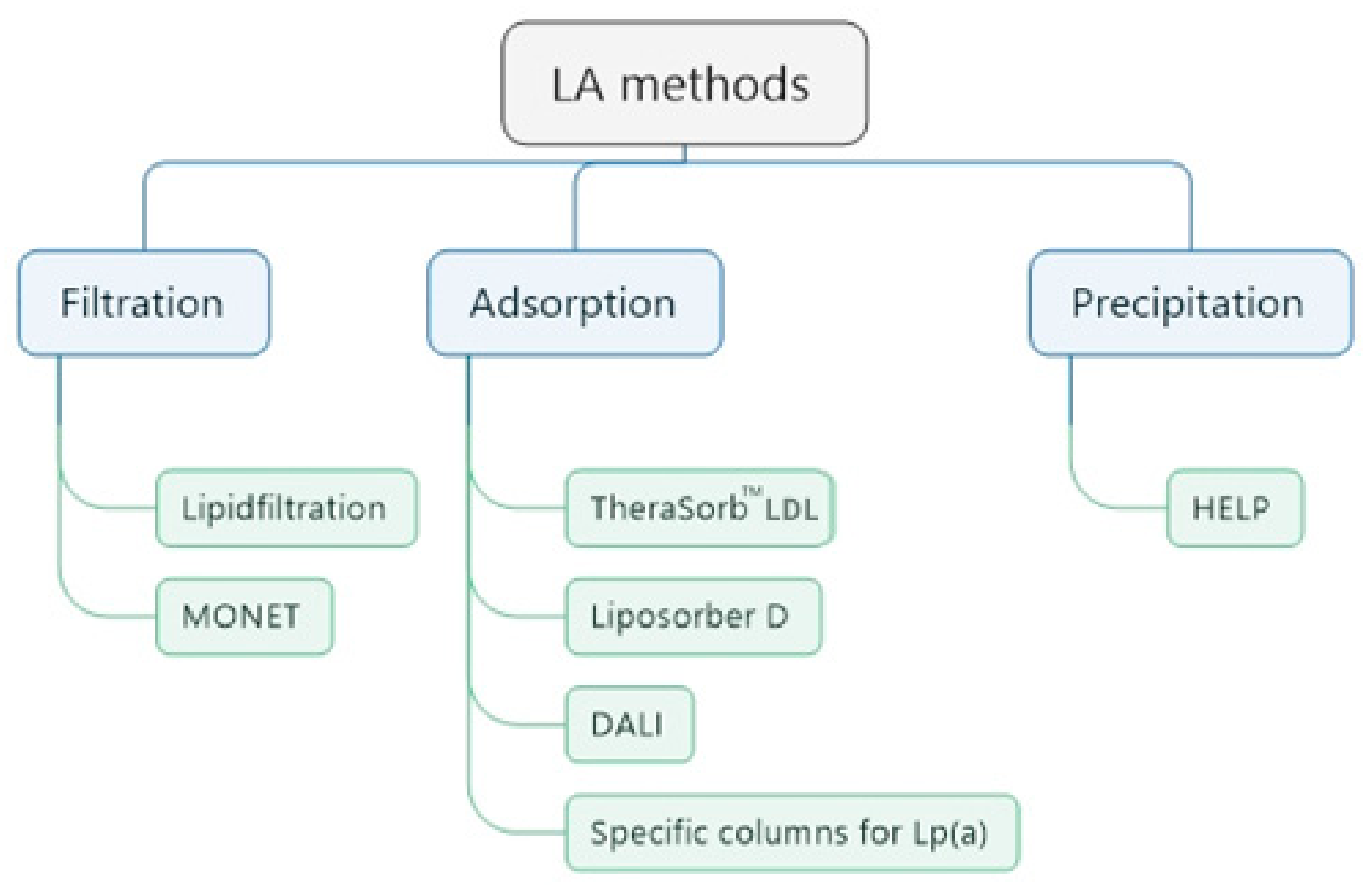
5.2. Comparison of Different LA Methods
5.3. Adverse Effects of LA
6. New Drugs and LA
6.1. PCSK9 Inhibitors
6.2. Inhibitor of Microsomal Transfer Protein (MTP) in Homozygous Familial HCH
6.3. Antisense Oligonucleotide against Apolipoprotein(a)
7. Conclusions
Acknowledgments
Conflicts of Interest
Abbreviations
| HELP | heparin-induced extracorporeal LDL precipitation |
| DALI | direct adsorption of lipoproteins |
| MONET | membrane filtration optimized novel extracorporeal treatment |
| RCT | randomized control trial |
References and Notes
- Julius, U. History of lipidology and lipoprotein apheresis. Atheroscler. Suppl. 2017, 30, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Cannon, C.P.; Blazing, M.A.; Giugliano, R.P.; McCagg, A.; White, J.A.; Theroux, P.; Darius, H.; Lewis, B.S.; Ophuis, T.O.; Jukema, J.W.; et al. Ezetimibe added to statin therapy after acute coronary syndromes. N. Engl. J. Med. 2015, 372, 2387–2397. [Google Scholar] [CrossRef] [PubMed]
- Thompson, G.R.; Catapano, A.; Saheb, S.; Atassi-Dumont, M.; Barbir, M.; Eriksson, M.; Paulweber, B.; Sijbrands, E.; Stalenhoef, A.F.; Parhofer, K.G. Severe hypercholesterolaemia: Therapeutic goals and eligibility criteria for LDL apheresis in Europe. Curr. Opin. Lipidol. 2010, 21, 492–498. [Google Scholar] [CrossRef] [PubMed]
- Thompson, G.R.; Seed, M.; Naoumova, R.P.; Neuwirth, C.; Walji, S.; Aitman, T.J.; Scott, J.; Myant, N.B.; Soutar, A.K. Improved cardiovascular outcomes following temporal advances in lipid-lowering therapy in a genetically-characterised cohort of familial hypercholesterolaemia homozygotes. Atherosclerosis 2015, 243, 328–333. [Google Scholar] [CrossRef] [PubMed]
- Bundesministerium für Gesundheit, Bekanntmachung des Bundesausschusses der Ärzte und Krankenkassen über eine Änderung der Richtlinien über die Bewertung ärztlicher Untersuchungs- und Behandlungsmethoden gemäß § 135 Abs. 1 des Fünften Buches Sozialgesetzbuch (SGB V) (BUB-Richtlinien), BAnz, 2003; 123:14486.
- Bundesministerium für Gesundheit, Bekanntmachung eines Beschlusses des Gemeinsamen Bundesausschusses über eine Änderung der Richtlinie Methoden vertragsärztliche Versorgung: Apherese bei isolierter Lp(a)-Erhöhung, BAnz, 2008; 138:3321.
- Julius, U. Niacin as antidyslipidemic drug. Can. J. Physiol. Pharmacol. 2015, 93, 1043–1054. [Google Scholar] [CrossRef] [PubMed]
- Leebmann, J.; Roeseler, E.; Julius, U.; Heigl, F.; Spitthoever, R.; Heutling, D.; Breitenberger, P.; Maerz, W.; Lehmacher, W.; Heibges, A.; et al. Lipoprotein apheresis in patients with maximally tolerated lipid-lowering therapy, lipoprotein(a)-hyperlipoproteinemia, and progressive cardiovascular disease: Prospective observational multicenter study. Circulation 2013, 128, 2567–2576. [Google Scholar] [CrossRef] [PubMed]
- Gross, E.; Hohenstein, B.; Julius, U. Effects of lipoprotein apheresis on the lipoprotein(a) levels in the long run. Atheroscler. Suppl. 2015, 18, 226–232. [Google Scholar] [CrossRef] [PubMed]
- Mabuchi, H.; Koizumi, J.; Shimizu, M.; Kajinami, K.; Miyamoto, S.; Ueda, K.; Takegoshi, T. Long-term efficacy of low-density lipoprotein apheresis on coronary heart disease in familial hypercholesterolemia. Hokuriku-FH-LDL-apheresis study group. Am. J. Cardiol. 1998, 82, 1489–1495. [Google Scholar] [CrossRef]
- Emmrich, U.; Hohenstein, B.; Julius, U. Actual situation of lipoprotein apheresis in Saxony in 2013. Atheroscler. Suppl. 2015, 18, 215–225. [Google Scholar] [CrossRef] [PubMed]
- Heigl, F.; Hettich, R.; Lotz, N.; Reeg, H.; Pflederer, T.; Osterkorn, D.; Osterkorn, K.; Klingel, R. Efficacy, safety, and tolerability of long-term lipoprotein apheresis in patients with LDL- or Lp(a) hyperlipoproteinemia: Findings gathered from more than 36,000 treatments at one center in Germany. Atheroscler. Suppl. 2015, 18, 154–162. [Google Scholar] [CrossRef] [PubMed]
- Schatz, U.; Tselmin, S.; Muller, G.; Julius, U.; Hohenstein, B.; Fischer, S.; Bornstein, S.R. Most significant reduction of cardiovascular events in patients undergoing lipoproteinapheresis due to raised Lp(a) levels—A multicenter observational study. Atheroscler. Suppl. 2017, 30, 246–252. [Google Scholar] [CrossRef] [PubMed]
- Von Dryander, M.; Fischer, S.; Passauer, J.; Muller, G.; Bornstein, S.R.; Julius, U. Differences in the atherogenic risk of patients treated by lipoprotein apheresis according to their lipid pattern. Atheroscler. Suppl. 2013, 14, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Jaeger, B.R.; Richter, Y.; Nagel, D.; Heigl, F.; Vogt, A.; Roeseler, E.; Parhofer, K.; Ramlow, W.; Koch, M.; Utermann, G.; et al. Longitudinal cohort study on the effectiveness of lipid apheresis treatment to reduce high lipoprotein(a) levels and prevent major adverse coronary events. Nat. Clin. Pract. Cardiovasc. Med. 2009, 6, 229–239. [Google Scholar] [CrossRef] [PubMed]
- Roeseler, E.; Julius, U.; Heigl, F.; Spitthoever, R.; Heutling, D.; Breitenberger, P.; Leebmann, J.; Lehmacher, W.; Kamstrup, P.R.; Nordestgaard, B.G.; et al. Lipoprotein apheresis for lipoprotein(a)-associated cardiovascular disease: Prospective 5 years of follow-up and apo(a) characterization. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 2019–2027. [Google Scholar] [CrossRef] [PubMed]
- Safarova, M.S.; Ezhov, M.V.; Afanasieva, O.I.; Matchin, Y.G.; Atanesyan, R.V.; Adamova, I.Y.; Utkina, E.A.; Konovalov, G.A.; Pokrovsky, S.N. Effect of specific lipoprotein(a) apheresis on coronary atherosclerosis regression assessed by quantitative coronary angiography. Atheroscler. Suppl. 2013, 14, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Khan, T.Z.; Hsu, L.Y.; Arai, A.E.; Rhodes, S.; Pottle, A.; Wage, R.; Banya, W.; Gatehouse, P.D.; Giri, S.; Collins, P.; et al. Apheresis as novel treatment for refractory angina with raised lipoprotein(a): A randomized controlled cross-over trial. Eur. Heart J. 2017, 38, 1561–1569. [Google Scholar] [CrossRef] [PubMed]
- Khan, T.Z. Can lipoprotein apheresis offer a therapeutic role in the management of patients with refractory angina and raised lipoprotein(a)? Ther. Apher. Dial. 2018, 22, 5–7. [Google Scholar] [CrossRef] [PubMed]
- Reimann, M.; Prieur, S.; Lippold, B.; Bornstein, S.R.; Reichmann, H.; Julius, U.; Ziemssen, T. Retinal vessel analysis in hypercholesterolemic patients before and after LDL apheresis. Atheroscler. Suppl. 2009, 10, 39–43. [Google Scholar] [CrossRef]
- Terai, N.; Julius, U.; Haustein, M.; Spoerl, E.; Pillunat, L.E. The effect of low-density lipoprotein apheresis on ocular microcirculation in patients with hypercholesterolaemia: A pilot study. Br. J. Ophthalmol. 2011, 95, 401–404. [Google Scholar] [CrossRef] [PubMed]
- Julius, U. Lipoprotein apheresis in the management of severe hypercholesterolemia and of elevation of lipoprotein(a): Current perspectives and patient selection. Med. Devices 2016, 9, 349–360. [Google Scholar] [CrossRef] [PubMed]
- Julius, U.; Fischer, S.; Schatz, U.; Passauer, J.; Bornstein, S. Why an apheresis center should offer more than one lipoprotein apheresis method. Ther. Apher. Dial. 2013, 17, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Julius, U.; Metzler, W.; Pietzsch, J.; Fassbender, T.; Klingel, R. Intraindividual comparison of two extracorporeal LDL apheresis methods: Lipidfiltration and HELP. Int. J. Artif. Organs 2002, 25, 1180–1188. [Google Scholar] [CrossRef] [PubMed]
- Julius, U.; Siegert, G.; Kostka, H.; Schatz, U.; Hohenstein, B. Effects of different lipoprotein apheresis methods on serum protein levels. Atheroscler. Suppl. 2015, 18, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Julius, U.; Milton, M.; Stoellner, D.; Rader, D.; Gordon, B.; Polk, D.; Waldmann, E.; Parhofer, K.G.; Moriarty, P.M. Effects of lipoprotein apheresis on PCSK9 levels. Atheroscler. Suppl. 2015, 18, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Dittrich-Riediger, J.; Schatz, U.; Hohenstein, B.; Julius, U. Adverse events of lipoprotein apheresis and immunoadsorption at the apheresis center at the University Hospital Dresden. Atheroscler. Suppl. 2015, 18, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Sabatine, M.S.; Leiter, L.A.; Wiviott, S.D.; Giugliano, R.P.; Deedwania, P.; De Ferrari, G.M.; Murphy, S.A.; Kuder, J.F.; Gouni-Berthold, I.; Lewis, B.S.; et al. Cardiovascular safety and efficacy of the PCSK9 inhibitor evolocumab in patients with and without diabetes and the effect of evolocumab on glycaemia and risk of new-onset diabetes: A prespecified analysis of the Fourier randomised controlled trial. Lancet Diabetes Endocrinol. 2017, 5, 941–950. [Google Scholar] [CrossRef]
- Bonaca, M.P.; Nault, P.; Giugliano, R.P.; Keech, A.C.; Pineda, A.L.; Kanevsky, E.; Kuder, J.; Murphy, S.A.; Jukema, J.W.; Lewis, B.S.; et al. Low-density lipoprotein cholesterol lowering with evolocumab and outcomes in patients with peripheral artery disease: Insights from the Fourier trial (further cardiovascular outcomes research with PCSK9 inhibition in subjects with elevated risk). Circulation 2018, 137, 338–350. [Google Scholar] [CrossRef] [PubMed]
- Giugliano, R.P.; Pedersen, T.R.; Park, J.G.; De Ferrari, G.M.; Gaciong, Z.A.; Ceska, R.; Toth, K.; Gouni-Berthold, I.; Lopez-Miranda, J.; Schiele, F.; et al. Clinical efficacy and safety of achieving very low LDL-cholesterol concentrations with the PCSK9 inhibitor evolocumab: A prespecified secondary analysis of the Fourier trial. Lancet 2017, 390, 1962–1971. [Google Scholar] [CrossRef]
- Giugliano, R.P.; Mach, F.; Zavitz, K.; Kurtz, C.; Schneider, J.; Wang, H.; Keech, A.; Pedersen, T.R.; Sabatine, M.S.; Sever, P.S.; et al. Design and rationale of the Ebbinghaus trial: A phase 3, double-blind, placebo-controlled, multicenter study to assess the effect of evolocumab on cognitive function in patients with clinically evident cardiovascular disease and receiving statin background lipid-lowering therapy-a cognitive study of patients enrolled in the Fourier trial. Clin. Cardiol. 2017, 40, 59–65. [Google Scholar] [PubMed]
- Sabatine, M.S.; Giugliano, R.P.; Keech, A.C.; Honarpour, N.; Wiviott, S.D.; Murphy, S.A.; Kuder, J.F.; Wang, H.; Liu, T.; Wasserman, S.M.; et al. Evolocumab and clinical outcomes in patients with cardiovascular disease. N. Engl. J. Med. 2017, 376, 1713–1722. [Google Scholar] [CrossRef] [PubMed]
- Cuchel, M.; Meagher, E.A.; du Toit, T.H.; Blom, D.J.; Marais, A.D.; Hegele, R.A.; Averna, M.R.; Sirtori, C.R.; Shah, P.K.; Gaudet, D.; et al. Efficacy and safety of a microsomal triglyceride transfer protein inhibitor in patients with homozygous familial hypercholesterolaemia: A single-arm, open-label, phase 3 study. Lancet 2013, 381, 40–46. [Google Scholar] [CrossRef]
- D’Erasmo, L.; Cefalu, A.B.; Noto, D.; Giammanco, A.; Averna, M.; Pintus, P.; Medde, P.; Vigna, G.B.; Sirtori, C.; Calabresi, L.; et al. Efficacy of lomitapide in the treatment of familial homozygous hypercholesterolemia: Results of a real-world clinical experience in Italy. Adv. Ther. 2017, 34, 1200–1210. [Google Scholar] [CrossRef] [PubMed]
- Cuchel, M.; Bruckert, E.; Ginsberg, H.N.; Raal, F.J.; Santos, R.D.; Hegele, R.A.; Kuivenhoven, J.A.; Nordestgaard, B.G.; Descamps, O.S.; Steinhagen-Thiessen, E.; et al. Homozygous familial hypercholesterolaemia: New insights and guidance for clinicians to improve detection and clinical management. A position paper from the consensus panel on familial hypercholesterolaemia of the European Atherosclerosis Society. Eur. Heart J. 2014, 35, 2146–2157. [Google Scholar] [CrossRef] [PubMed]
- Blom, D.J.; Averna, M.R.; Meagher, E.A.; du Toit Theron, H.; Sirtori, C.R.; Hegele, R.A.; Shah, P.K.; Gaudet, D.; Stefanutti, C.; Vigna, G.B.; et al. Long-term efficacy and safety of the microsomal triglyceride transfer protein inhibitor lomitapide in patients with homozygous familial hypercholesterolemia. Circulation 2017, 136, 332–335. [Google Scholar] [CrossRef] [PubMed]
- Tsimikas, S.; Viney, N.J.; Hughes, S.G.; Singleton, W.; Graham, M.J.; Baker, B.F.; Burkey, J.L.; Yang, Q.; Marcovina, S.M.; Geary, R.S.; et al. Antisense therapy targeting apolipoprotein(a): A randomised, double-blind, placebo-controlled phase 1 study. Lancet 2015, 386, 1472–1483. [Google Scholar] [CrossRef]
- Viney, N.J.; van Capelleveen, J.C.; Geary, R.S.; Xia, S.; Tami, J.A.; Yu, R.Z.; Marcovina, S.M.; Hughes, S.G.; Graham, M.J.; Crooke, R.M.; et al. Antisense oligonucleotides targeting apolipoprotein(a) in people with raised lipoprotein(a): Two randomised, double-blind, placebo-controlled, dose-ranging trials. Lancet 2016, 388, 2239–2253. [Google Scholar] [CrossRef]

© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Julius, U. Current Role of Lipoprotein Apheresis in the Treatment of High-Risk Patients. J. Cardiovasc. Dev. Dis. 2018, 5, 27. https://doi.org/10.3390/jcdd5020027
Julius U. Current Role of Lipoprotein Apheresis in the Treatment of High-Risk Patients. Journal of Cardiovascular Development and Disease. 2018; 5(2):27. https://doi.org/10.3390/jcdd5020027
Chicago/Turabian StyleJulius, Ulrich. 2018. "Current Role of Lipoprotein Apheresis in the Treatment of High-Risk Patients" Journal of Cardiovascular Development and Disease 5, no. 2: 27. https://doi.org/10.3390/jcdd5020027
APA StyleJulius, U. (2018). Current Role of Lipoprotein Apheresis in the Treatment of High-Risk Patients. Journal of Cardiovascular Development and Disease, 5(2), 27. https://doi.org/10.3390/jcdd5020027



