Using Synchronous Fluorescence to Investigate Chemical Interactions Influencing Foam Characteristics in Sparkling Wines
Abstract
1. Introduction
2. Materials and Methods
2.1. Wine Material
2.2. Basic Wine Composition for the Control Sample
2.3. Wine Samples
2.4. Chemical Additions
2.5. Determination of Foam Parameters
2.6. Fluorescence Spectroscopy
2.7. Parallel Factor Analysis (Parafac)
2.8. Statistical Analysis
3. Results
3.1. Wine Composition
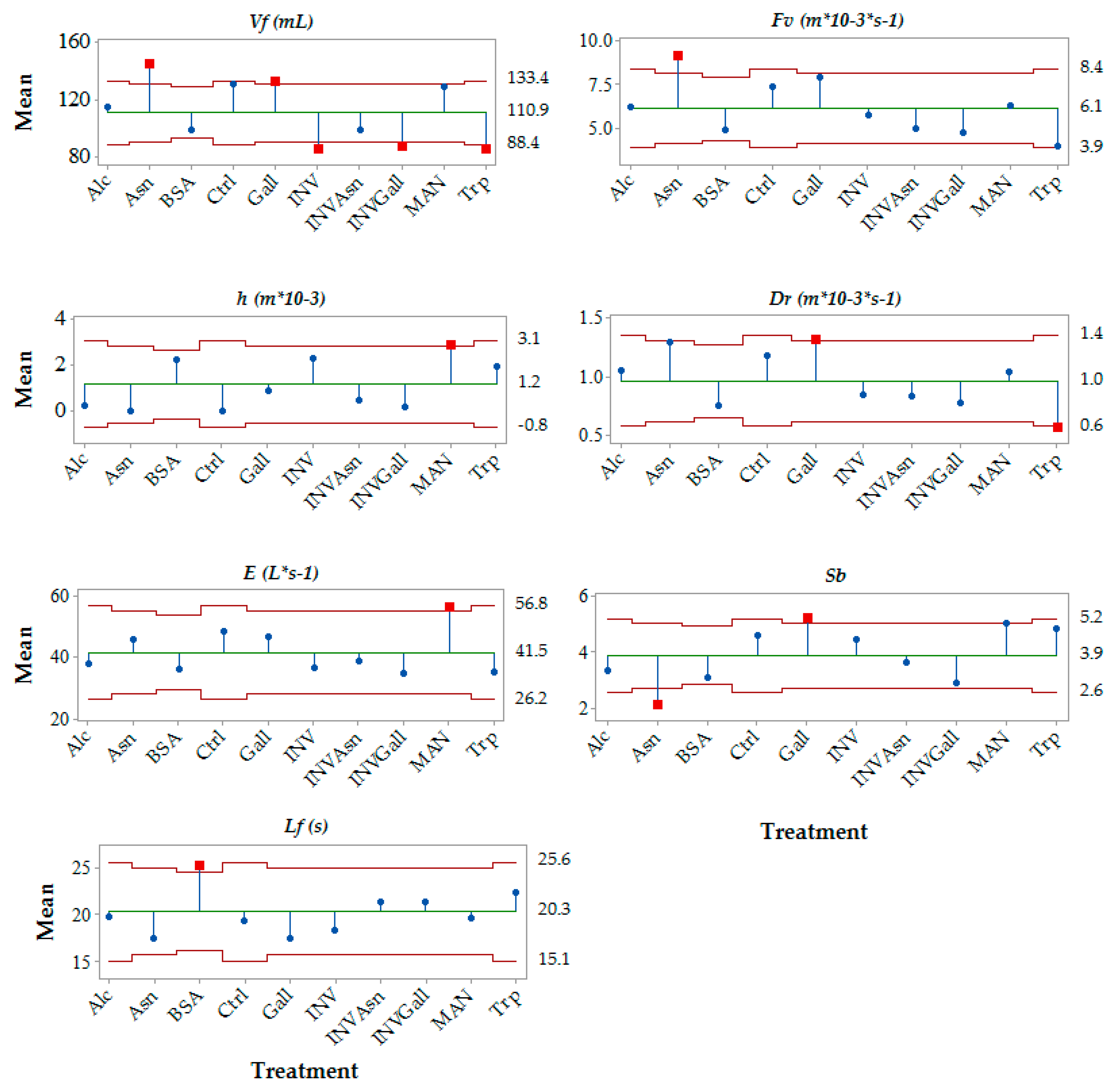
3.2. Effect of the Chemical Additions on Foam Parameters
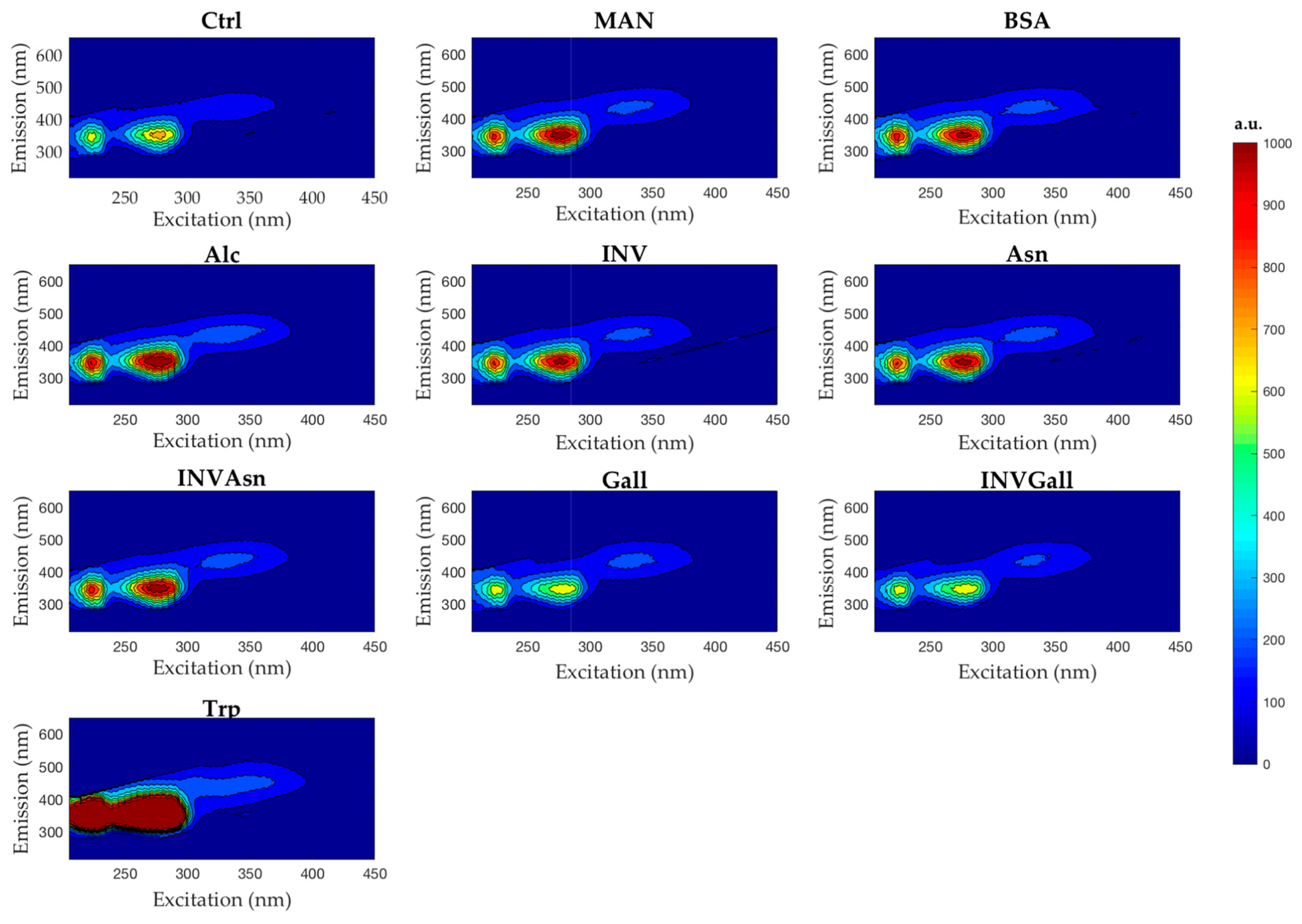
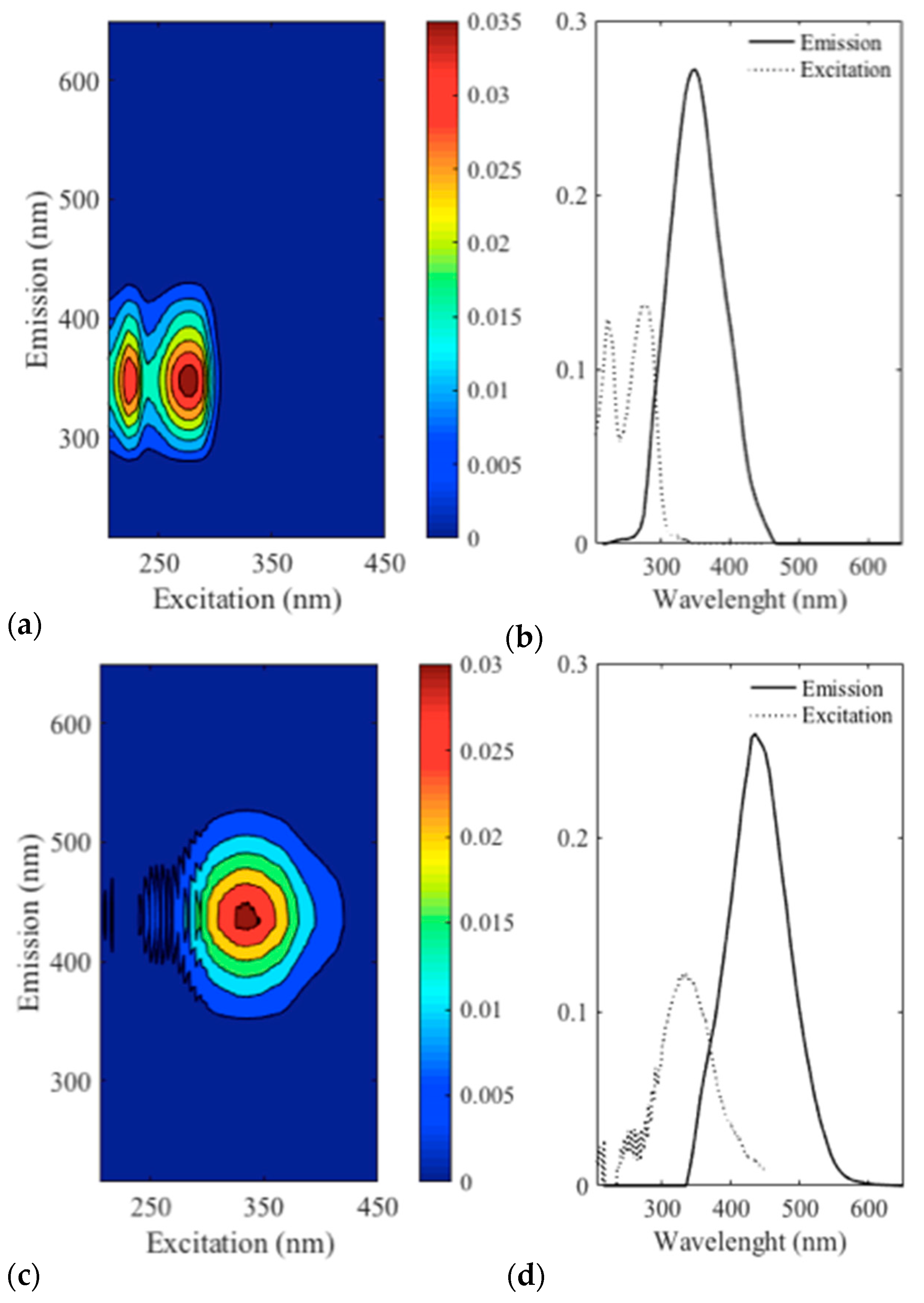
3.3. Spectrofluorescence and Parafac Analysis
4. Discussion
4.1. Identification of Compounds Related to Foam Quality
4.2. Hypotheses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| (Lf) | Average foam lifetime |
| (h) | Collar height |
| (hc) | Collar initial height |
| (Dr) | Drainability |
| λex | Excitation wavelength |
| λem | Emission wavelength |
| (E) | Foam expansion |
| (Ft) | Foam time |
| (Fv) | Foam velocity |
| (Vf) | Foam volume |
| (Wf) | Percentage of wine in the foam |
| (Sb) | Small bubbles |
References
- Spence, C.; Levitan, C.A.; Shankar, M.U.; Zampini, M. Does food color influence taste and flavor perception in humans? Chemosens. Percept. 2010, 3, 68–84. [Google Scholar] [CrossRef]
- Morrot, G.; Brochet, F.; Dubourdieu, D. The color of odors. Brain Lang. 2001, 79, 309–320. [Google Scholar] [CrossRef]
- Bailey, P. Global sparkling wine market trends. In Wine Australia for Australian Wine; Wine Australia: Adelaide, Australia, 2018; p. 28. [Google Scholar]
- Jackson, R.S. Wine Tasting: A Professional Handbook; Academic Press: Cambridge, MA, USA, 2016. [Google Scholar]
- International Organisation of Vine and Wine. Resolution OIV/concours 332a/2009. In OIV Standard for International Wine and Spirituous Beverages of VItivinicultural Origin Competitions; OIV: Paris, France, 2009. [Google Scholar]
- Liger-Belair, G. The physics behind the fizz in champagne and sparkling wines. Eur. Phys. J. Spec. Top. 2012, 201, 1–88. [Google Scholar] [CrossRef]
- Dunkel, A.; Hofmann, T. Carbonic anhydrase iv mediates the fizz of carbonated beverages. Angew. Chem. Int. Ed. Engl. 2010, 49, 2975–2977. [Google Scholar] [CrossRef]
- Kemp, B.; Condé, B.; Jégou, S.; Howell, K.; Vasserot, Y.; Marchal, R. Chemical compounds and mechanisms involved in the formation and stabilization of foam in sparkling wines. Crit. Rev. Food Sci. Nutr. 2018, 59, 2072–2094. [Google Scholar] [CrossRef]
- Alexandre, H.; Guilloux-Benatier, M. Yeast autolysis in sparkling wine: A review. Aust. J. Grape Wine Res. 2006, 12, 119–127. [Google Scholar] [CrossRef]
- Abdallah, Z.; Aguie-Beghin, V.; Abou-Saleh, K.; Douillard, R.; Bliard, C. Isolation and analysis of macromolecular fractions responsible for the surface properties in native champagne wines. Food Res. Int. 2010, 43, 982–987. [Google Scholar] [CrossRef]
- Blasco, L.; Viñas, M.; Villa, T.G. Proteins influencing foam formation in wine and beer: the role of yeast. Int. Microbiol. 2011, 14, 61–71. [Google Scholar]
- Cilindre, C.; Fasoli, E.; D’Amato, A.; Liger-Belair, G.; Righetti, P.G. It’s time to pop a cork on champagne’s proteome! J. Proteom. 2014, 105, 351–362. [Google Scholar] [CrossRef]
- Cilindre, C.; Liger-Belair, G.; Villaume, S.; Jeandet, P.; Marchal, R. Foaming properties of various champagne wines depending on several parameters: Grape variety, aging, protein and CO2 content. Anal. Chim. Acta 2010, 660, 164–170. [Google Scholar] [CrossRef]
- Coelho, E.; Rocha, S.M.; Coimbra, M.A. Foamability and foam stability of molecular reconstituted model sparkling wines. J. Agric. Food Chem. 2011, 59, 8770–8778. [Google Scholar] [CrossRef]
- Marchal, R.; Bouquelet, S.; Maujean, A. Purification and partial biochemical characterization of glycoproteins in a champenois chardonnay wine. J. Agric. Food Chem. 1996, 44, 1716–1722. [Google Scholar] [CrossRef]
- Marchal, R.; Maujean, A. Proprietes effervescentes des vins de champagne [degazage, bulle, vin mousseux]. Bios. Boissons. 1996, 260, 25–29. [Google Scholar]
- Marchal, R.; Warchol, M.; Cilindre, C.; Jeandet, P. Evidence for protein degradation by botrytis cinerea and relationships with alteration of synthetic wine foaming properties. J. Agric. Food Chem. 2006, 54, 5157–5165. [Google Scholar] [CrossRef]
- Maujean, A.; Poinsaut, P.; Dantan, H.; Brissonnet, F.; Cossiez, E. Study of the performance and quality of the foam in sparkling wines. 2. Perfecting of a measuring technique for foaming ability, performance and stability of the foam in sparkling wines [measurement of foam properties, bentonite, vegetable black]. Bull. De L’oiv (Fr.) 1990, 63, 405–427. [Google Scholar]
- Pérez-Magariño, S.; Ortega-Heras, M.; Bueno-Herrera, M.; Martínez-Lapuente, L.; Guadalupe, Z.; Ayestarán, B. Grape variety, aging on lees and aging in bottle after disgorging influence on volatile composition and foamability of sparkling wines. LWT Food Sci. Technol. 2015, 61, 47–55. [Google Scholar] [CrossRef]
- Condé, B.C.; Bouchard, E.; Culbert, J.A.; Wilkinson, K.L.; Fuentes, S.; Howell, K.S. Soluble protein and amino acid content affects the foam quality of sparkling wine. J. Agric. Food Chem. 2017, 65, 9110–9119. [Google Scholar] [CrossRef]
- Dickinson, E. Protein adsorption at liquid interfaces and the relationship to foam stability. In Foams: Physics, Chemistry and Structure; Wilson, A., Ed.; Springer: London, UK, 1989; pp. 39–53. [Google Scholar]
- Dupin, I.V.S.; McKinnon, B.M.; Ryan, C.; Boulay, M.; Markides, A.J.; Jones, G.P.; Williams, P.J.; Waters, E.J. Saccharomycescerevisiae mannoproteins that protect wine from protein haze: Their release during fermentation and lees contact and a proposal for their mechanism of action. J. Agric. Food Chem. 2000, 48, 3098–3105. [Google Scholar] [CrossRef]
- Dambrouck, T.; Marchal, R.; Cilindre, C.; Parmentier, M.; Jeandet, P. Determination of the grape invertase content (using PTA− ELISA) following various fining treatments versus changes in the total protein content of wine. Relationships with wine foamability. J. Agric. Food Chem. 2005, 53, 8782–8789. [Google Scholar] [CrossRef]
- Puff, N.; Marchal, R.; Aguié-Béghin, V.; Douillard, R.J.L. Is grape invertase a major component of the adsorption layer formed at the air/champagne wine interface? Langmuir 2001, 17, 2206–2212. [Google Scholar] [CrossRef]
- Brissonnet, F.; Maujean, A. Characterization of foaming proteins in a champagne base wine. Am. J. Enol. Vitic. 1993, 44, 297–301. [Google Scholar]
- Martínez-Rodríguez, A.J.; Carrascosa, A.V.; Martín-Álvarez, P.J.; Moreno-Arribas, V.; Polo, M.C. Influence of the yeast strain on the changes of the amino acids, peptides and proteins during sparkling wine production by the traditional method. J. Ind. Microbiol. Biotechnol. 2002, 29, 314–322. [Google Scholar] [CrossRef]
- Moreno-Arribas, V.; Pueyo, E.; Nieto, F.J.; Martı́n-Álvarez, P.J.; Polo, M.C. Influence of the polysaccharides and the nitrogen compounds on foaming properties of sparkling wines. Food Chem. 2000, 70, 309–317. [Google Scholar] [CrossRef]
- Martinez-Lapuente, L.; Guadalupe, Z.; Ayestaran, B.; Perez-Magarino, S. Role of major wine constituents in the foam properties of white and rose sparkling wines. Food Chem. 2015, 174, 330–338. [Google Scholar] [CrossRef]
- Medina-Trujillo, L.; Matias-Guiu, P.; López-Bonillo, F.; Canals, J.M.; Zamora, F. Physicochemical characterization of the foam of white and rosé base wines for sparkling wine production (AOC Cava). Am. J. Enol. Vitic. 2017, 68, 485–495. [Google Scholar] [CrossRef]
- Ozdal, T.; Capanoglu, E.; Altay, F.J.F.R.I. A review on protein–phenolic interactions and associated changes. Food Res. Int. 2013, 51, 954–970. [Google Scholar] [CrossRef]
- Prigent, S.; Voragen, A.; Van Koningsveld, G.; Baron, A.; Renard, C.; Gruppen, H. Interactions between globular proteins and procyanidins of different degrees of polymerization. J. Dairy Sci. 2009, 92, 5843–5853. [Google Scholar] [CrossRef]
- Ojha, H.; Mishra, K.; Hassan, M.I.; Chaudhury, N.K. Spectroscopic and isothermal titration calorimetry studies of binding interaction of ferulic acid with bovine serum albumin. Acta 2012, 548, 56–64. [Google Scholar] [CrossRef]
- Wu, X.; Wu, H.; Liu, M.; Liu, Z.; Xu, H.; Lai, F. Analysis of binding interaction between (−)-epigallocatechin (egc) and β-lactoglobulin by multi-spectroscopic method. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2011, 82, 164–168. [Google Scholar] [CrossRef]
- Yuksel, Z.; Avci, E.; Erdem, Y.K. Characterization of binding interactions between green tea flavanoids and milk proteins. Food Chem. 2010, 121, 450–456. [Google Scholar] [CrossRef]
- Labuckas, D.O.; Maestri, D.M.; Perello, M.; Martinez, M.L.; Lamarque, A.L. Phenolics from walnut (juglans regia l.) kernels: Antioxidant activity and interactions with proteins. Food Chem. 2008, 107, 607–612. [Google Scholar] [CrossRef]
- Duodu, K.; Taylor, J.; Belton, P.; Hamaker, B.J. Factors affecting sorghum protein digestibility. J. Cereal Sci. 2003, 38, 117–131. [Google Scholar] [CrossRef]
- Kanakis, C.; Hasni, I.; Bourassa, P.; Tarantilis, P.; Polissiou, M.; Tajmir-Riahi, H.A. Milk β-lactoglobulin complexes with tea polyphenols. Food Chem. 2011, 127, 1046–1055. [Google Scholar] [CrossRef]
- Tantoush, Z.; Stanic, D.; Stojadinovic, M.; Ognjenovic, J.; Mihajlovic, L.; Atanaskovic-Markovic, M.; Velickovic, T.C. Digestibility and allergenicity of β-lactoglobulin following laccase-mediated cross-linking in the presence of sour cherry phenolics. Food Chem. 2011, 125, 84–91. [Google Scholar] [CrossRef]
- Nuthong, P.; Benjakul, S.; Prodpran, T.J. Effect of phenolic compounds on the properties of porcine plasma protein-based film. Food Hydrocoll. 2009, 23, 736–741. [Google Scholar] [CrossRef]
- Spencer, C.M.; Cai, Y.; Martin, R.; Gaffney, S.H.; Goulding, P.N.; Magnolato, D.; Lilley, T.H.; Haslam, E. Polyphenol complexation—Some thoughts and observations. Phytochemistry 1988, 27, 2397–2409. [Google Scholar] [CrossRef]
- Siebert, K.J.; Troukhanova, N.V.; Lynn, P.Y.J. Nature of polyphenol− protein interactions. J. Agric. Food Chem. 1996, 44, 80–85. [Google Scholar] [CrossRef]
- Ryder, A.G. Cell culture media analysis using rapid spectroscopic methods. Curr. Opin. Chem. Eng. 2018, 22, 11–17. [Google Scholar] [CrossRef]
- Dufour, É.; Letort, A.; Laguet, A.; Lebecque, A.; Serra, J.N. Investigation of variety, typicality and vintage of french and german wines using front-face fluorescence spectroscopy. Anal. Chim. Acta 2006, 563, 292–299. [Google Scholar] [CrossRef]
- Binder, B.M.E.; Eric Schaller, G.E.; Kessenbrock, M.; Groth, G.; Walker, J.M.S.E. Circular dichroism and fluorescence spectroscopy to study protein structure and protein–Protein interactions in Ethylene Signaling. Methods Mol. Biol. 2017, 1573, 141–159. [Google Scholar]
- Fatemeh, S.M.S.; Farid, M.; Masoumeh, N. Spectroscopy and molecular dynamics simulation study on the interaction of sunset yellow food additive with pepsin. Int. J. Biol. Macromol. 2018, 115, 273–280. [Google Scholar]
- Xiang, L.; Zhen, Y.; Yu, B. Fluorescence spectroscopic analysis of the interaction of papain and bromelain with l-ascorbic acid, α-tocopherol, β-carotene and astaxanthin. Int. J. Biol. Macromol. 2018, 107, 144–156. [Google Scholar]
- Melo, A.M.; Prieto, M.; Coutinho, A. Quantifying lipid-protein interaction by fluorescence correlation spectroscopy (FCS). Methods Mol. Biol. 2014, 1076, 575–595. [Google Scholar]
- Boothello, R.S.; Al-Horani, R.A.; Desai, U.R. Glycosaminoglycan–protein interaction studies using fluorescence spectroscopy. Methods Mol. Biol. 2014, 1229, 335–353. [Google Scholar]
- Chen, Y.; Barkley, M.D. Toward understanding tryptophan fluorescence in proteins. Biochemistry 1998, 37, 9976–9982. [Google Scholar] [CrossRef]
- Iland, P.N.B.; Edwards, G.S.; Caloghiris, E.W. Chemical Analysis of Grapes and Wine: Techniques and Concepts, 2nd ed.; Patrick iland Wine Promotions Pty Ltd.: Athelstone, Australia, 2013. [Google Scholar]
- Sigma-Aldrich. Product Information—Invertase From Baker’s Yeast. 2019. Available online: https://www.sigmaaldrich.com/Graphics/COfAInfo/SigmaSAPQM/SPEC/I4/I4504/I4504-BULK________SIGMA____.pdf (accessed on 23 August 2019).
- Condé, B.C.; Fuentes, S.; Caron, M.; Xiao, D.; Collmann, R.; Howell, K.S. Development of a robotic and computer vision method to assess foam quality in sparkling wines. Food Control. 2017, 71, 383–392. [Google Scholar] [CrossRef]
- Lakowicz, J.R. Principles of Fluorescence Spectroscopy, 3rd ed.; Springer: New York, NY, USA, 2006. [Google Scholar]
- Hirsch, R.E. Front-face fluorescence spectroscopy of hemoglobins. Methods Enzym. 1994, 232, 231–246. [Google Scholar]
- Zandomeneghi, M.; Carbonaro, L.; Caffarata, C. Fluorescence of vegetable oils: Olive oils. J. Agric. Food Chem. 2005, 53, 759–766. [Google Scholar] [CrossRef]
- Abbas, O.; Rébufa, C.; Dupuy, N.; Permanyer, A.; Kister, J.; Azevedo, D. Application of chemometric methods to synchronous uv fluorescence spectra of petroleum oils. Fuel 2006, 85, 2653–2661. [Google Scholar] [CrossRef]
- Murphy, K.R.; Stedmon, C.A.; Graeber, D.; Bro, R. Fluorescence spectroscopy and multi-way techniques. Parafac. Anal. Methods 2013, 5, 6557–6566. [Google Scholar] [CrossRef]
- Ammor, S.; Yaakoubi, K.; Chevallier, I.; Dufour, E. Identification by fluorescence spectroscopy of lactic acid bacteria isolated from a small-scale facility producing traditional dry sausages. J. Microbiol Methods 2004, 59, 271–281. [Google Scholar] [CrossRef]
- Leblanc, L.; Dufour, É. Monitoring the identity of bacteria using their intrinsic fluorescence. Fems Microbiol. Lett. 2002, 211, 147–153. [Google Scholar] [CrossRef]
- Cantor, C.R.; Schimmel, P.R. Techniques for the Study of Biological Structure and Function; W. H. Freeman: San Francisco, CA, USA, 1980. [Google Scholar]
- Papadopoulou, A.; Green, R.J.; Frazier, R.A. Interaction of flavonoids with bovine serum albumin: A fluorescence quenching study. J. Agric. Food Chem. 2005, 53, 158–163. [Google Scholar] [CrossRef]
- Martin, C.; Bruneel, J.L.; Guyon, F.; Médina, B.; Jourdes, M.; Teissedre, P.L.; Guillaume, F. Raman spectroscopy of white wines. Food Chem. 2015, 181, 235–240. [Google Scholar] [CrossRef]
- El-Seedi, H.R.; Taher, E.A.; Sheikh, B.Y.; Anjum, S.; Saeed, A.; AlAjmi, M.F.; Moustafa, M.S.; Al-Mousawi, S.M.; Farag, M.A.; Hegazy, M.E.F. Chapter 8 hydroxycinnamic acids: Natural sources, biosynthesis, possible biological activities, and roles in islamic medicine. In Studies in Natural Products Chemistry; Atta, R., Ed.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 269–292. [Google Scholar]
- Waterhouse, A.L.; Sacks, G.L.; Jeffery, D.W. Understanding Wine Chemistry; John Wiley Sons, Inc.: Chichester, UK, 2016. [Google Scholar]
- Buitimea-Cantua, N.E.; Gutierrez-Uribe, J.A.; Serna-Saldivar, S.O. Phenolic-protein interactions: Effects on food properties and health benefits. J. Med. Food 2018, 21, 188–198. [Google Scholar] [CrossRef]
- Rodrigues, A.; Ricardo-Da-Silva, J.M.; Lucas, C.; Laureano, O. Effect of commercial mannoproteins on wine colour and tannins stability. Food Chem. 2012, 131, 907–914. [Google Scholar] [CrossRef]
- Ramos-Pineda, A.M.; García-Estévez, I.; Dueñas, M.; Escribano-Bailón, M.T. Effect of the addition of mannoproteins on the interaction between wine flavonols and salivary proteins. Food Chem. 2018, 264, 226–232. [Google Scholar] [CrossRef]
- Strumeyer, D.H.; Malin, M.J. Resistance of extracellular yeast invertase and other glycoproteins to denaturation by tannins. Biochem. J. 1970, 118, 899–900. [Google Scholar] [CrossRef]
- Albani, J.R. Origin of tryptophan fluorescence lifetimes. Part 2: Fluorescence lifetimes origin of tryptophan in proteins. J. Fluoresc. 2014, 24, 105–117. [Google Scholar] [CrossRef]
- Lakowicz, J.R. Topics in Fluorescence Spectroscopy; Protein fluorescence; Springer: Boston, MA, USA, 2002; Volume 6. [Google Scholar]
- Cao, H.; Liu, Q.; Shi, J.; Xiao, J.; Xu, M. Comparing the affinities of flavonoid isomers with protein by fluorescence spectroscopy. Anal. Lett. 2008, 41, 521–532. [Google Scholar] [CrossRef]
- Valeur, B.; Berberan-Santos, M.N. Molecular Fluorescence: Principles and Applications, 2nd ed.; Wiley-VCH: Weinheim, Germany, 2013. [Google Scholar]
| Sample | Concentration Added | Abbreviation |
|---|---|---|
| Control | n.a. | Ctrl |
| Alcohol | 0.1% ABV | Alc |
| Yeast Invertase | 100 mg/L | INV |
| Bovine Serum Albumin | 100 mg/L | BSA |
| Manonoprotein | 100 mg/L | MAN |
| Gallic Acid | 100 mg/L | Gall |
| Asparagine | 100 mg/L | Asn |
| Tryptophan | 100mg/L | Trp |
| Yeast Invertase + Asparagine | 100 mg/L + 100 mg/L | INVAsn |
| Yeast Invertase + Gallic Acid | 100 mg/L + 100 mg/L | INVGall |
| Sample | Foam Volume (Vf) | Foam Velocity (Fv) | Drainability (Dr) | Collar Height (h) | Small Bubbles Ratio (Sb) |
|---|---|---|---|---|---|
| Control | n.a. | n.a. | n.a. | n.a. | n.a. |
| Alcohol | n.s. | n.s. | n.s. | n.s. | n.s. |
| Yeast Invertase | ↓ | n.s. | n.s. | n.s. | n.s. |
| Bovine Serum albumin | ↓ | n.s. | n.s. | n.s. | n.s. |
| Mannoprotein | n.s. | n.s. | n.s. | ↑ | n.s. |
| Gallic Acid | n.s. | n.s. | n.s. | n.s. | n.s. |
| Asparagine | n.s. | n.s. | n.s. | n.s. | ↓ |
| Tryptophan | ↓ | ↓ | ↓ | n.s. | n.s. |
| Yeast Invertase + asparagine | ↓ | n.s. | n.s. | n.s. | n.s. |
| Sample | λem | FI |
|---|---|---|
| Control | 350 | 52,222 |
| Alcohol | 335 | 75,279 |
| Asparagine | 335 | 73,024 |
| Bovine Serum Albumin | 340 | 72,225 |
| Gallic Acid | 340 | 46,462 |
| Yeast Invertase | 340 | 70,930 |
| Yeast Invertase + Asparagine | 335 | 73,377 |
| Yeast Invertase + Gallic Acid | 345 | 45,953 |
| Mannoprotein | 335 | 73,257 |
| Tryptophan | 305 | 86,562 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Condé, B.; Robinson, A.; Bodet, A.; Monteau, A.-C.; Fuentes, S.; Scollary, G.; Smith, T.; Howell, K.S. Using Synchronous Fluorescence to Investigate Chemical Interactions Influencing Foam Characteristics in Sparkling Wines. Beverages 2019, 5, 54. https://doi.org/10.3390/beverages5030054
Condé B, Robinson A, Bodet A, Monteau A-C, Fuentes S, Scollary G, Smith T, Howell KS. Using Synchronous Fluorescence to Investigate Chemical Interactions Influencing Foam Characteristics in Sparkling Wines. Beverages. 2019; 5(3):54. https://doi.org/10.3390/beverages5030054
Chicago/Turabian StyleCondé, Bruna, Alanna Robinson, Amandine Bodet, Anne-Charlotte Monteau, Sigfredo Fuentes, Geoffrey Scollary, Trevor Smith, and Kate S. Howell. 2019. "Using Synchronous Fluorescence to Investigate Chemical Interactions Influencing Foam Characteristics in Sparkling Wines" Beverages 5, no. 3: 54. https://doi.org/10.3390/beverages5030054
APA StyleCondé, B., Robinson, A., Bodet, A., Monteau, A.-C., Fuentes, S., Scollary, G., Smith, T., & Howell, K. S. (2019). Using Synchronous Fluorescence to Investigate Chemical Interactions Influencing Foam Characteristics in Sparkling Wines. Beverages, 5(3), 54. https://doi.org/10.3390/beverages5030054






