Effect of the Atmospheric Pressure Cold Plasma Treatment on Tempranillo Red Wine Quality in Batch and Flow Systems
Abstract
:1. Introduction
2. Materials and Methods
2.1. Wine and APCP Treatments
2.2. Analysis of Physical and Color Parameters
2.3. Analysis of Anthocyanins and Vitisins by HPLC
2.4. Statistical Analysis
3. Results and Discussion
3.1. Impact of APCP Treatments on Physical and Color Parameters
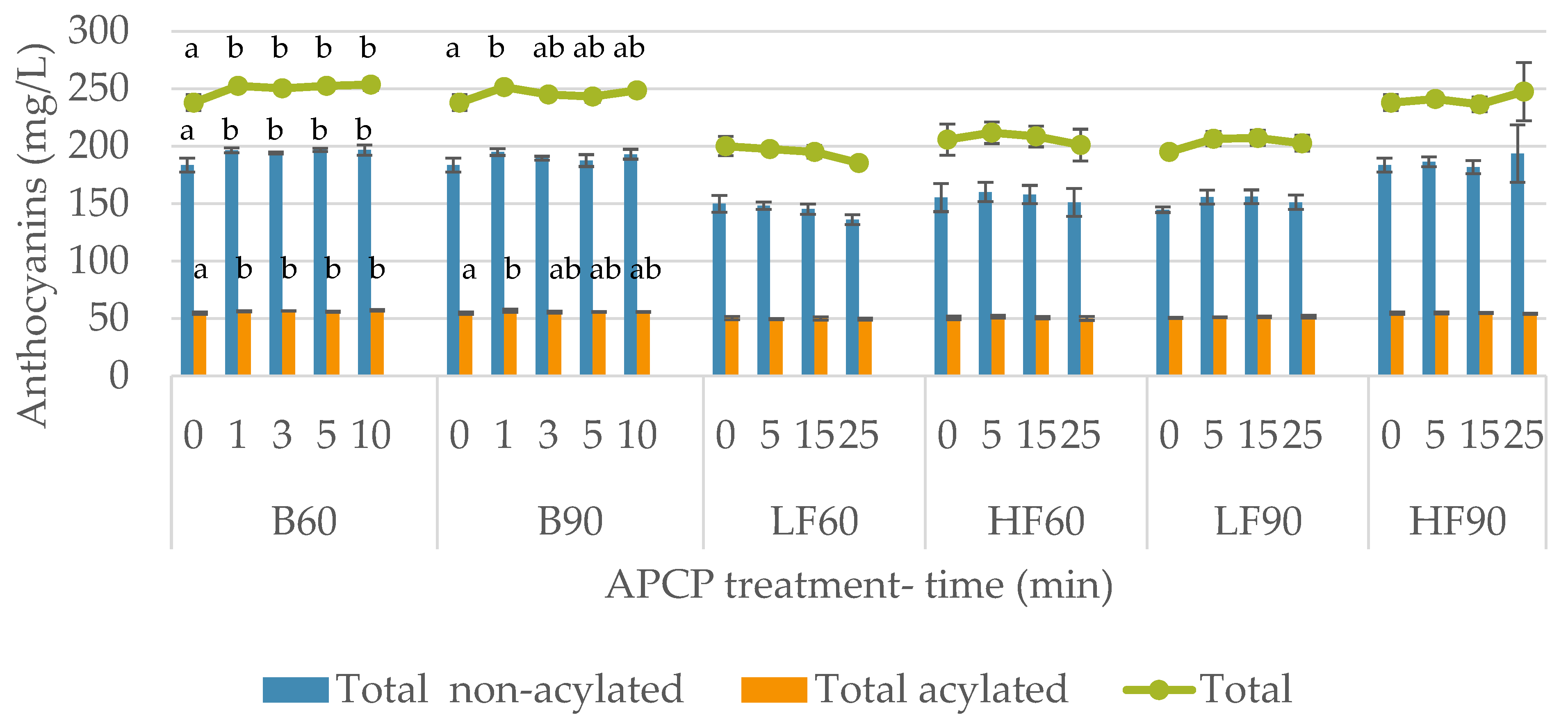
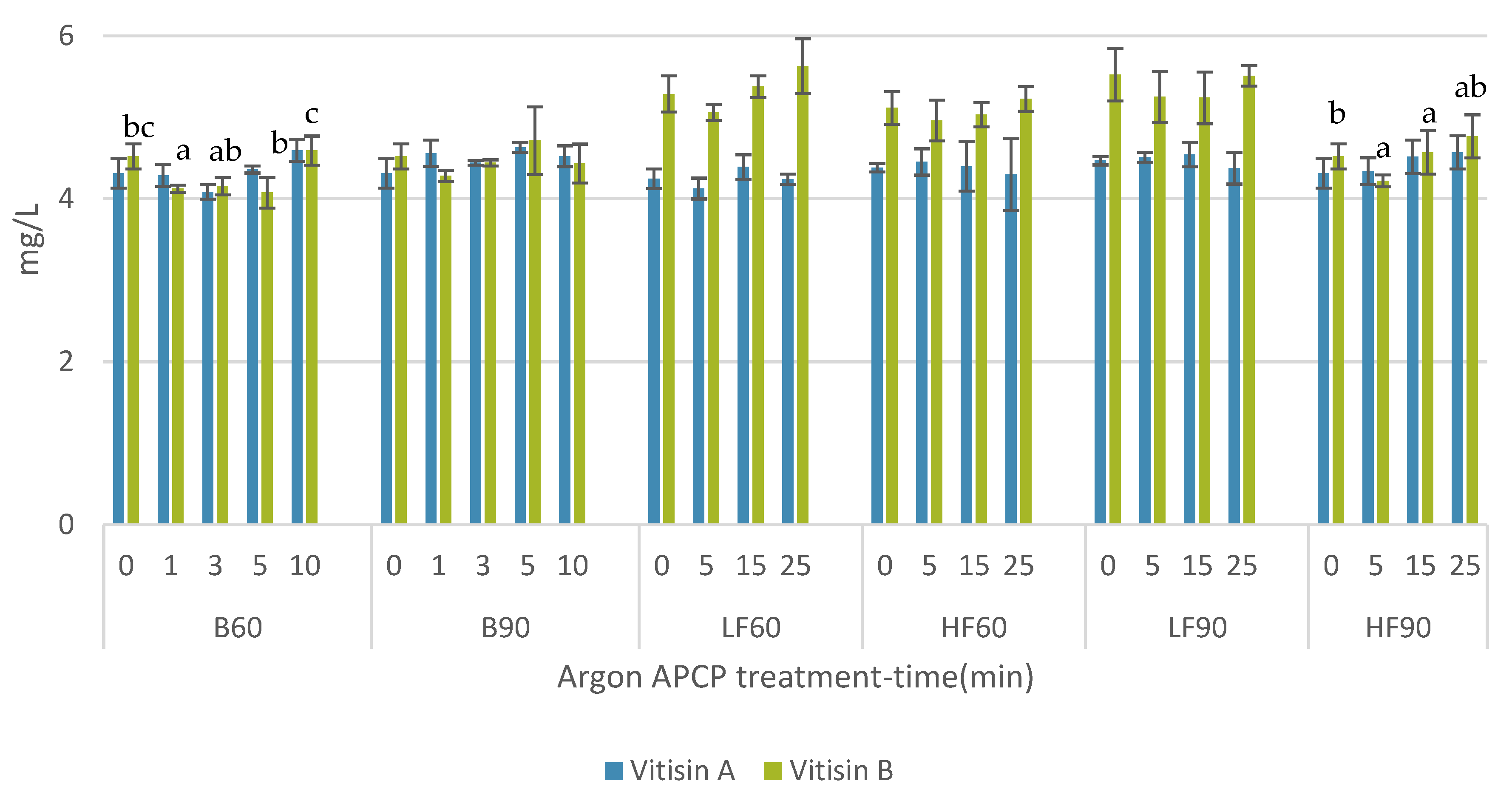
3.2. Impact of APCP Treatments on Anthocyanin and Vitisins Contents
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Costanigro, M.; Appleby, C.; Menke, S.D. The Wine Headache: Consumer Perceptions of Sulfites and Willingness to Pay for Non-Sulfited Wines. Food Qual. Prefer. 2014, 31, 81–89. [Google Scholar] [CrossRef]
- D’Amico, M.; Di Vita, G.; Monaco, L. Exploring Environmental Consciousness and Consumer Preferences for Organic Wines without Sulfites. J. Clean. Prod. 2016, 120, 64–71. [Google Scholar] [CrossRef]
- Arnous, A.; Makris, D.P.; Kefalas, P. Effect of Principal Polyphenolic Components in Relation to Antioxidant Characteristics of Aged Red Wines. J. Agric. Food Chem. 2001, 49, 5736–5742. [Google Scholar] [CrossRef] [PubMed]
- Ziuzina, D.; Boehm, D.; Patil, S.; Cullen, P.J.; Bourke, P. Cold Plasma Inactivation of Bacterial Biofilms and Reduction of Quorum Sensing Regulated Virulence Factors. PLoS ONE 2015, 10, e0138209. [Google Scholar] [CrossRef]
- Misra, N.; Schlüter, O.; Cullen, P. Cold Plasma in Food and Agriculture: Fundamentals and Applications; Academic Press Elsevier: Amsterdam, The Nederland, 2016. [Google Scholar]
- Kim, H.-J.; Yong, H.I.; Park, S.; Kim, K.; Choe, W.; Jo, C. Microbial Safety and Quality Attributes of Milk Following Treatment with Atmospheric Pressure Encapsulated Dielectric Barrier Discharge Plasma. Food Control 2015, 47, 451–456. [Google Scholar] [CrossRef]
- Parpinello, G.P.; Versari, A.; Chinnici, F.; Galassi, S. Relationship among Sensory Descriptors, Consumer Preference and Color Parameters of Italian Novello Red Wines. Food Res. Int. 2009, 42, 1389–1395. [Google Scholar] [CrossRef]
- Barsotti, L.; Dumay, E.; Mu, T.H.; Fernandez Diaz, M.D.; Cheftel, J.C. Effects of High Voltage Electric Pulses on Protein-Based Food Constituents and Structures. Trends Food Sci. Technol. 2001, 12, 136–144. [Google Scholar] [CrossRef]
- García-Beneytez, E.; Revilla, E.; Cabello, F. Anthocyanin Pattern of Several Red Grape Cultivars and Wines Made from Them. Eur. Food Res. Technol. 2002, 215, 32–37. [Google Scholar] [CrossRef]
- Sacchi, K.L.; Bisson, L.F.; Adams, D.O. A Review of the Effect of Winemaking Techniques on Phenolic Extraction in Red Wines. Am. J. Enol. Vitic. 2005, 56, 197–206. [Google Scholar]
- Minussi, R.C.; Rossi, M.; Bologna, L.; Cordi, L.; Rotilio, D.; Pastore, G.M.; Durán, N. Phenolic Compounds and Total Antioxidant Potential of Commercial Wines. Food Chem. 2003, 82, 409–416. [Google Scholar] [CrossRef]
- Briviba, K.; Abrahamse, S.L.; Pool-Zobel, B.L.; Rechkemmer, G. Neurotensin-and EGF-Induced Metabolic Activation of Colon Carcinoma Cells Is Diminished by Dietary Flavonoid Cyanidin but Not by Its Glycosides. Nutr. Cancer 2001, 41, 172–179. [Google Scholar] [CrossRef]
- Negro, C.; Tommasi, L.; Miceli, A. Phenolic Compounds and Antioxidant Activity from Red Grape Marc Extracts. Bioresour. Technol. 2003, 87, 41–44. [Google Scholar] [CrossRef]
- Pérez-Magariño, S.; González-San José, M.L. Polyphenols and Colour Variability of Red Wines Made from Grapes Harvested at Different Ripeness Grade. Food Chem. 2006, 96, 197–208. [Google Scholar] [CrossRef]
- Pankaj, S.K.; Wan, Z.; Colonna, W.; Keener, K.M. Effect of High Voltage Atmospheric Cold Plasma on White Grape Juice Quality. J. Sci. Food Agric. 2017, 97, 4016–4021. [Google Scholar] [CrossRef] [PubMed]
- Lukić, K.; Vukušić, T.; Tomašević, M.; Ćurko, N.; Gracin, L.; Kovačević Ganić, K. The Impact of High Voltage Electrical Discharge Plasma on the Chromatic Characteristics and Phenolic Composition of Red and White Wines. Innov. Food Sci. Emerg. Technol. 2017, 53, 70–77. [Google Scholar] [CrossRef]
- Bursać Kovačević, D.; Putnik, P.; Dragović-Uzelac, V.; Pedisić, S.; Režek Jambrak, A.; Herceg, Z. Effects of Cold Atmospheric Gas Phase Plasma on Anthocyanins and Color in Pomegranate Juice. Food Chem. 2016, 190, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Commission Regulation. Commission Regulation (EC) No 606/2009 of 10 July 2009 Laying down Certain Detailed Rules for Implementing Council Regulation (EC) No 479/2008 as Regards the Categories of Grapevine Products, Oenological Practices and the Applicable Restrictions. Off. J. Eur. Union L 2009, 193, 1–59. [Google Scholar]
- Portu, J.; López, R.; Baroja, E.; Santamaría, P.; Garde-Cerdán, T. Improvement of Grape and Wine Phenolic Content by Foliar Application to Grapevine of Three Different Elicitors: Methyl Jasmonate, Chitosan, and Yeast Extract. Food Chem. 2016, 201, 213–221. [Google Scholar] [CrossRef]
- Mandal, R.; Singh, A.; Singh, A.P. Recent Developments in Cold Plasma Decontamination Technology in the Food Industry. Trends Food Sci. Technol. 2018, 80, 93–103. [Google Scholar] [CrossRef]
- Thirumdas, R.; Kothakota, A.; Annapure, U.; Siliveru, K.; Blundell, R.; Gatt, R.; Valdramidis, V.P. Plasma Activated Water (PAW): Chemistry, Physico-Chemical Properties, Applications in Food and Agriculture. Trends Food Sci. Technol. 2018, 77, 21–31. [Google Scholar] [CrossRef]
- Coutinho, N.M.; Silveira, M.R.; Rocha, R.S.; Moraes, J.; Ferreira, M.V.S.; Pimentel, T.C.; Freitas, M.Q.; Silva, M.C.; Raices, R.S.L.; Ranadheera, C.S.; et al. Cold Plasma Processing of Milk and Dairy Products. Trends Food Sci. Technol. 2018, 74, 56–68. [Google Scholar] [CrossRef]
- Bosso, A.; Motta, S.; Petrozziello, M.; Guaita, M.; Asproudi, A.; Panero, L. Validation of a Rapid Conductimetric Test for the Measurement of Wine Tartaric Stability. Food Chem. 2016, 212, 821–827. [Google Scholar] [CrossRef]
- Herceg, Z.; Kovačević, D.B.; Kljusurić, J.G.; Jambrak, A.R.; Zorić, Z.; Dragović-Uzelac, V. Gas Phase Plasma Impact on Phenolic Compounds in Pomegranate Juice. Food Chem. 2016, 190, 665–672. [Google Scholar] [CrossRef]
- Portu, J.; López-Alfaro, I.; Gómez-Alonso, S.; López, R.; Garde-Cerdán, T. Changes on Grape Phenolic Composition Induced by Grapevine Foliar Applications of Phenylalanine and Urea. Food Chem. 2015, 180, 171–180. [Google Scholar] [CrossRef]
- Elez Garofulić, I.; Režek Jambrak, A.; Milošević, S.; Dragović-Uzelac, V.; Zorić, Z.; Herceg, Z. The Effect of Gas Phase Plasma Treatment on the Anthocyanin and Phenolic Acid Content of Sour Cherry Marasca (Prunus Cerasus Var. Marasca) Juice. LWT-Food Sci. Technol. 2015, 62, 894–900. [Google Scholar] [CrossRef]
- Morata, A.; Calderón, F.; González, M.C.; Gómez-Cordovés, M.C.; Suárez, J.A. Formation of the Highly Stable Pyranoanthocyanins (Vitisins A and B) in Red Wines by the Addition of Pyruvic Acid and Acetaldehyde. Food Chem. 2007, 100, 1144–1152. [Google Scholar] [CrossRef]
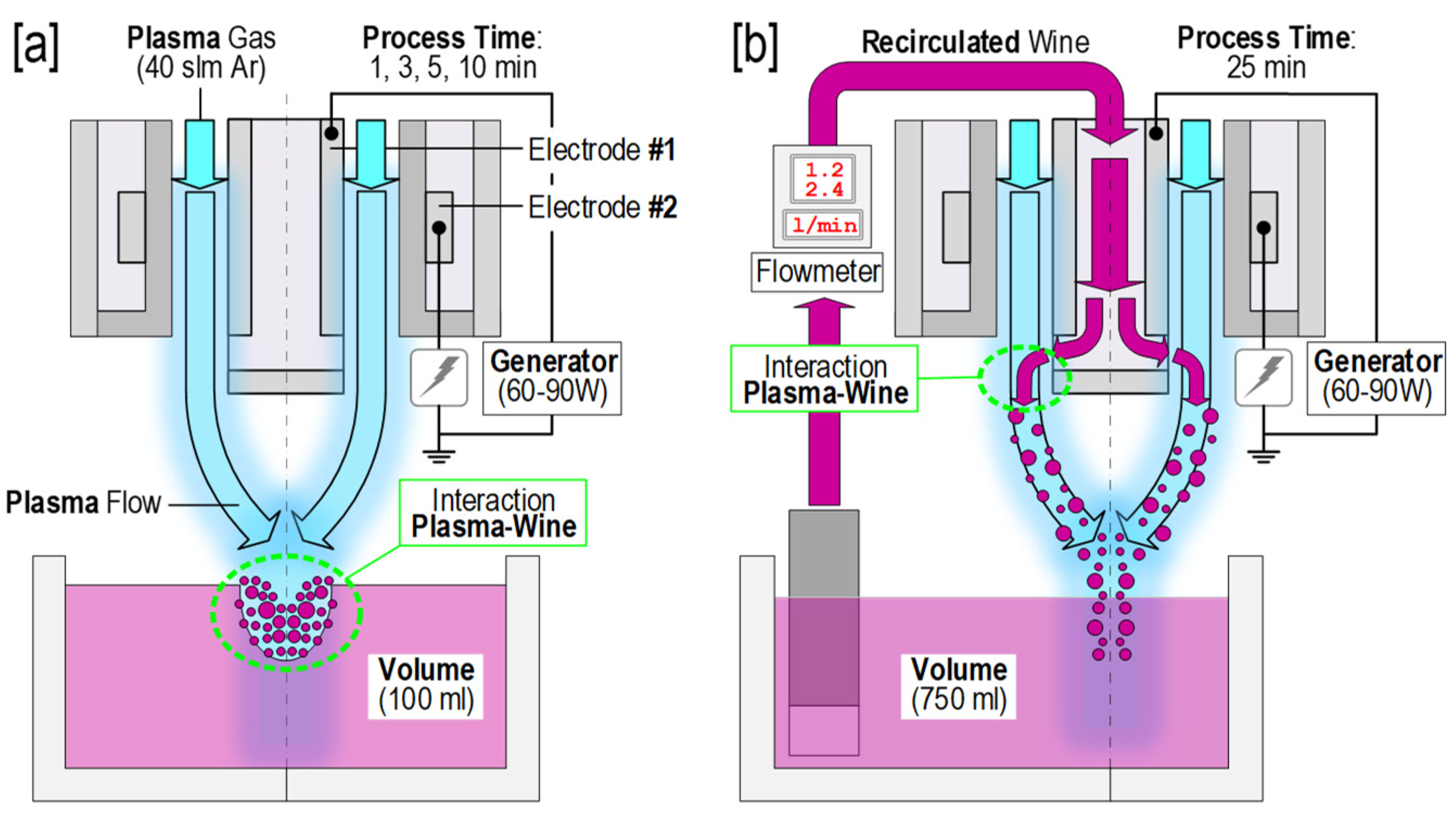
| Treatment | Power (W) | Batch | Wine Flow (mL/h) | Volume (mL) | Time (min) |
|---|---|---|---|---|---|
| B60 | 60 | Yes | No | 100 | 0,1,3,5,10 |
| B90 | 90 | Yes | No | 100 | 0,1,3,5,10 |
| LF60 | 60 | No | 1.2 | 750 | 0,10,15,20,25 |
| HF60 | 60 | No | 2.4 | 750 | 0,10,15,20,25 |
| LF90 | 90 | No | 1.2 | 750 | 0,10,15,20,25 |
| HF90 | 90 | No | 2.4 | 750 | 0,10,15,20,25 |
| Treatment | Time (min) | T (°C) | pH | C (µS) |
|---|---|---|---|---|
| B60 | 0 | 21.4 ± 0.0 | 3.77 ± 0.00 ab | 1750 ± 0 a |
| 1 | 21.3 ± 0.0 | 3.93 ± 0.19 b | 1760 ± 17 a | |
| 3 | 21.1 ± 0.0 | 3.93 ± 0.10 b | 1787 ± 6 ab | |
| 5 | 21.0 ± 0.0 | 3.89 ± 0.04 b | 1830 ± 30 b | |
| 10 | 19.9 ± 0.0 | 3.61 ± 0.00 a | 1903 ± 25 c | |
| B90 | 0 | 21.4 ± 0.0 | 3.77 ± 0.00 | 1750 ± 0 b |
| 1 | 21.3 ± 0.0 | 3.85 ± 0.27 | 1730 ± 0 a | |
| 3 | 21.2 ± 0.0 | 3.60 ± 0.03 | 1770 ± 10 c | |
| 5 | 21.0 ± 0.0 | 3.66 ± 0.07 | 1847 ± 12 d | |
| 10 | 22.0 ± 0.0 | 3.69 ± 0.15 | 1923 ± 6 e | |
| LF60 | 0 | 20.0 ± 0.1 | 3.69 ± 0.02 | 1873 ± 15 |
| 5 | 19.9 ± 0.1 | 3.70 ± 0.02 | 1867 ± 12 | |
| 10 | 19.8 ± 0.1 | 3.71 ± 0.03 | 1850 ± 10 | |
| 15 | 19.7 ± 0.1 | 3.62 ± 0.02 | 1793 ± 57 | |
| 20 | 19.6 ± 0.2 | 3.69 ± 0.09 | 1553 ± 453 | |
| 25 | 19.7 ± 0.4 | 3.72 ± 0.07 | 1460 ± 624 | |
| HF60 | 0 | 19.7 ± 0.3 ab | 3.47 ± 0.30 | 1590 ± 243 |
| 5 | 19.5 ± 0.2 a | 3.38 ± 0.30 | 1583 ± 240 | |
| 10 | 19.7 ± 0.1 a | 3.47 ± 0.23 | 1560 ± 217 | |
| 15 | 19.9 ± 0.0 ab | 3.49 ± 0.25 | 1517 ± 168 | |
| 20 | 20.2 ± 0.1 bc | 3.53 ± 0.20 | 1450 ± 280 | |
| 25 | 20.4 ± 0.1 c | 3.58 ± 0.13 | 1213 ± 68 | |
| LF90 | 0 | 20.0 ± 0.3 ab | 3.60 ± 0.06 | 1460 ± 52 |
| 5 | 19.7 ± 0.5 a | 3.55 ± 0.11 | 1460 ± 10 | |
| 10 | 20.1 ± 0.6 ab | 3.56 ± 0.07 | 1457 ± 12 | |
| 15 | 20.2 ± 0.3 ab | 3.46 ± 0.13 | 1447 ± 6 | |
| 20 | 20.7 ± 0.3 ab | 3.47 ± 0.14 | 1367 ± 42 | |
| 25 | 20.9 ± 0.3 b | 3.52 ± 0.09 | 1343 ± 93 | |
| HF90 | 0 | 20.7 ± 0.2 b | 3.70 ± 0.05 | 1755 ± 5 b |
| 5 | 20.3 ± 0.2 a | 3.70 ± 0.07 | 1760 ± 10 b | |
| 10 | 20.7 ± 0.2 b | 3.74 ± 0.08 | 1740 ± 20 b | |
| 15 | 21.1 ± 0.1 c | 3.79 ± 0.13 | 1737 ± 15 b | |
| 20 | 21.5 ± 0.1 d | 3.81 ± 0.14 | 1617 ± 47 a | |
| 25 | 21.8 ± 0.1 d | 3.81 ± 0.15 | 1560 ± 20 a |
| Treatment | Time (min) | CI | Hue | TP (mg/L Gallic Acid) |
|---|---|---|---|---|
| B60 | 0 | 6.50 ± 0.00 a | 0.599 ± 0.00 d | 1067 ± 71 a |
| 1 | 8.30 ± 0.05 ab | 0.579 ± 0.00 c | 1120 ± 8 ab | |
| 3 | 8.37 ± 0.01 ab | 0.571 ± 0.00 b | 1132 ± 14 ab | |
| 5 | 8.42 ± 0.05 ab | 0.567 ± 0.00 a | 1140 ± 11 ab | |
| 10 | 8.47 ± 0.08 b | 0.563 ± 0.00 a | 1170 ± 24 b | |
| B90 | 0 | 6.50 ± 0.00 a | 0.599 ± 0.00 d | 1067 ± 71 |
| 1 | 7.36 ± 0.02 b | 0.592 ± 0.00 cd | 1132 ± 26 | |
| 3 | 7.66 ± 0.04 c | 0.587 ± 0.00 bc | 1145 ± 30 | |
| 5 | 7.94 ± 0.02 d | 0.581 ± 0.00 b | 1155 ± 22 | |
| 10 | 8.24 ± 0.17 e | 0.564 ± 0.01a | 1194 ± 26 | |
| LF60 | 0 | 6.72 ± 0.15 a | 0.590 ± 0.01 | 1044 ± 29 |
| 5 | 6.94 ± 0.05 ab | 0.592 ± 0.01 | 1049 ± 55 | |
| 10 | 6.98 ± 0.16 ab | 0.587 ± 0.01 | 1063 ± 33 | |
| 15 | 7.05 ± 0.12 ab | 0.585 ± 0.01 | 1081 ± 45 | |
| 20 | 7.08 ± 0.10 ab | 0.581 ± 0.01 | 1077 ± 16 | |
| 25 | 7.19 ± 0.15 b | 0.581 ± 0.01 | 1066 ± 12 | |
| HF60 | 0 | 7.47 ± 0.56 | 0.593 ± 0.01 | 1062 ± 59 |
| 5 | 7.54 ± 0.54 | 0.592 ± 0.01 | 1058 ± 66 | |
| 10 | 7.56 ± 0.51 | 0.589 ± 0.01 | 1062 ± 56 | |
| 15 | 7.64 ± 0.54 | 0.589 ± 0.01 | 1084 ± 60 | |
| 20 | 7.72 ± 0.51 | 0.586 ± 0.00 | 1067 ± 13 | |
| 25 | 7.78 ± 0.55 | 0.583 ± 0.00 | 1105 ± 41 | |
| LF90 | 0 | 7.82 ± 0.06 | 0.588 ± 0.00 | 1087 ± 81 |
| 5 | 7.89 ± 0.10 | 0.590 ± 0.00 | 1070 ± 70 | |
| 10 | 7.91 ± 0.12 | 0.593 ± 0.00 | 1078 ± 71 | |
| 15 | 7.97 ± 0.02 | 0.587 ± 0.00 | 1089 ± 62 | |
| 20 | 8.00 ± 0.06 | 0.587 ± 0.00 | 1041 ± 8 | |
| 25 | 8.02 ± 0.06 | 0.585 ± 0.00 | 1052 ± 19 | |
| HF90 | 0 | 6.49 ± 0.03 a | 0.608 ± 0.00 c | 1087 ± 81 |
| 5 | 6.66 ± 0.05 b | 0.604 ± 0.01 bc | 1097 ± 48 | |
| 10 | 6.75 ± 0.02 bc | 0.599 ± 0.00 abc | 1103 ± 51 | |
| 15 | 6.84 ± 0.06 cd | 0.599 ± 0.00 abc | 1108 ± 57 | |
| 20 | 6.95 ± 0.04 d | 0.595 ± 0.00 ab | 1087 ± 54 | |
| 25 | 7.09 ± 0.04 e | 0.593 ± 0.00 a | 1101 ± 42 |
| Treatment | Time (min) | Dp-3-glc | Cn-3-glc | Pt-3-glc | Pn-3-glc | Mv-3-glc |
|---|---|---|---|---|---|---|
| B60 | 0 | 13.2 ± 0.5 a | 2.97 ± 0.03 b | 20.3 ± 0.5 a | 7.64 ± 0.32 | 139 ± 5 a |
| 1 | 14.1 ± 0.1 b | 2.89 ± 0.02 ab | 22.3 ± 0.2 b | 7.90 ± 0.13 | 149 ± 2 b | |
| 3 | 14.0 ± 0.2 ab | 2.91 ± 0.05 ab | 21.8 ± 0.1 b | 7.82 ± 0.06 | 148 ± 1 b | |
| 5 | 14.2 ± 0.2 b | 2.86 ± 0.02 a | 22.1 ± 0.3 b | 8.05 ± 0.05 | 150 ± 1 b | |
| 10 | 14.2 ± 0.4 b | 2.92 ± 0.05 ab | 22.2 ± 0.4 b | 7.94 ± 0.26 | 149 ± 3 b | |
| B90 | 0 | 13.2 ± 0.5 a | 2.97 ± 0.03 | 20.3 ± 0.5 a | 7.64 ± 0.32 | 139 ± 5 |
| 1 | 14.3 ± 0.4 b | 2.98 ± 0.07 | 22.2 ± 0.5 c | 8.09 ± 0.34 | 147 ± 2 | |
| 3 | 13.7 ± 0.1 ab | 2.94 ± 0.04 | 21.3 ± 0.2 abc | 7.60 ± 0.09 | 144 ± 1 | |
| 5 | 13.4 ± 0.5 ab | 3.01 ± 0.02 | 20.9 ± 0.5 bc | 7.53 ± 0.20 | 142 ± 4 | |
| 10 | 13.9 ± 0.3 ab | 2.96 ± 0.04 | 21.8 ± 0.4 ab | 7.83 ± 0.19 | 146 ± 3 | |
| LF60 | 0 | 10.4 ± 0.6 | 2.90 ± 0.08 | 16.8 ± 0.8 | 6.55 ± 0.33 b | 113 ± 6 b |
| 5 | 10.3 ± 0.5 | 2.90 ± 0.05 | 16.5 ± 0.4 | 6.47 ± 0.10 ab | 112 ± 2 ab | |
| 15 | 10.1 ± 0.5 | 2.96 ± 0.11 | 16.0 ± 0.5 | 6.34 ± 0.19 ab | 110 ± 3 ab | |
| 25 | 9.5 ± 0.4 | 2.96 ± 0.08 | 15.4 ± 0.5 | 5.95 ± 0.21 a | 102 ± 4 a | |
| HF60 | 0 | 10.8 ± 1.0 | 2.91 ± 0.08 | 17.3 ± 1.3 | 6.63 ± 0.39 | 118 ± 9 |
| 5 | 11.2 ± 0.5 | 2.90 ± 0.11 | 17.9 ± 1.0 | 6.85 ± 0.40 | 121 ± 6 | |
| 15 | 11.2 ± 0.5 | 2.97 ± 0.07 | 17.6 ± 0.9 | 6.68 ± 0.36 | 119 ± 6 | |
| 25 | 10.6 ± 0.8 | 3.00 ± 0.10 | 17.2 ± 1.5 | 6.32 ± 0.44 | 114 ± 10 | |
| LF90 | 0 | 10.1 ± 0.1 | 2.84 ± 0.05 | 16.2 ± 0.2 | 6.31 ± 0.16 | 109 ± 2 |
| 5 | 10.9 ± 0.6 | 2.87 ± 0.04 | 17.6 ± 0.9 | 6.61 ± 0.19 | 118 ± 5 | |
| 15 | 10.9 ± 0.6 | 2.91 ± 0.04 | 17.5 ± 0.4 | 6.62 ± 0.15 | 118 ± 5 | |
| 25 | 10.6 ± 0.6 | 2.89 ± 0.11 | 16.9 ± 0.8 | 6.42 ± 0.16 | 114 ± 5 | |
| LF90 | 0 | 13.2 ± 0.5 | 2.97 ± 0.03 | 20.3 ± 0.5 | 7.64 ± 0.32 | 139 ± 5 |
| 5 | 13.3 ± 0.3 | 3.00 ± 0.01 | 20.9 ± 0.5 | 7.70 ± 0.15 | 142 ± 3 | |
| 15 | 12.9 ± 0.3 | 3.04 ± 0.08 | 20.5 ± 0.8 | 7.50 ± 0.27 | 138 ± 5 | |
| 25 | 12.6 ± 0.3 | 2.99 ± 0.02 | 20.0 ± 0.5 | 7.27 ± 0.18 | 151 ± 25 |
| Treatment | Time (min) | Acetylated | Coumaroylated | Caffeoylated | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Dp-3-acglc | Pt-3-acglc | Pn-3-acglc | Mv-3-acglc | Cn-3-cmglc | Dp-3-cmglc | Pt-3-cmglc | Pn-3-cmglc | Mv-3-Cis-cmglc | Mv-3-Trans-cmglc | Mv-3-cfGlc | ||
| B60 | 0 | 2.64 ± 0.05 | 3.28 ± 0.05 a | 2.34 ± 0.02 | 7.87 ± 0.25 a | 2.32 ± 0.02 | 3.32 ± 0.05 a | 3.26 ± 0.10 a | 3.73 ± 0.09 a | 3.57 ± 0.11 ab | 10.54 ± 0.64 a | 2.69 ± 0.03 a |
| 1 | 2.61 ± 0.04 | 3.36 ± 0.03 ab | 2.35 ± 0.01 | 8.47 ± 0.09 b | 2.37 ± 0.06 | 3.64 ± 0.14 b | 3.46 ± 0.10 b | 3.75 ± 0.10 ab | 3.60 ± 0.06 ab | 11.45 ± 0.17 ab | 2.71 ± 0.01 ab | |
| 3 | 2.62 ± 0.01 | 3.40 ± 0.01 b | 2.33 ± 0.02 | 8.37 ± 0.06 b | 2.35 ± 0.02 | 3.48 ± 0.03 ab | 3.47 ± 0.04 b | 3.97 ± 0.09 b | 3.57 ± 0.03 ab | 11.80 ± 0.17 b | 2.73 ± 0.03 ab | |
| 5 | 2.63 ± 0.05 | 3.46 ± 0.04 b | 2.35 ± 0.01 | 8.38 ± 0.06 b | 2.35 ± 0.03 | 3.54 ± 0.08 ab | 3.36 ± 0.07 ab | 3.76 ± 0.09 ab | 3.46 ± 0.14 a | 11.23 ± 0.08 ab | 2.75 ± 0.06 ab | |
| 10 | 2.67 ± 0.02 | 3.44 ± 0.07 b | 2.33 ± 0.02 | 8.27 ± 0.12 b | 2.32 ± 0.00 | 3.52 ± 0.12 ab | 3.38 ± 0.02 ab | 3.86 ± 0.04 ab | 3.72 ± 0.07 b | 11.50 ± 0.40 b | 2.77 ± 0.02 b | |
| B90 | 0 | 2.64 ± 0.05 | 3.28 ± 0.05 a | 2.34 ± 0.02 | 7.87 ± 0.25 a | 2.32 ± 0.02 | 3.32 ± 0.05 a | 3.26 ± 0.10 | 3.73 ± 0.09 | 3.57 ± 0.11 | 10.54 ± 0.64 a | 2.69 ± 0.03 |
| 1 | 2.63 ± 0.03 | 3.47 ± 0.07 b | 2.34 ± 0.03 | 8.56 ± 0.18 b | 2.30 ± 0.01 | 3.54 ± 0.10 b | 3.41 ± 0.05 | 3.87 ± 0.15 | 3.61 ± 0.09 | 11.45 ± 0.73 b | 2.71 ± 0.06 | |
| 3 | 2.65 ± 0.03 | 3.34 ± 0.03 ab | 2.32 ± 0.02 | 8.19 ± 0.01 ab | 2.31 ± 0.02 | 3.42 ± 0.06 ab | 3.25 ± 0.04 | 3.80 ± 0.16 | 3.63 ± 0.03 | 10.91 ± 0.46 ab | 2.69 ± 0.01 | |
| 5 | 2.71 ± 0.07 | 3.28 ± 0.05 ab | 2.31 ± 0.03 | 8.11 ± 0.22 ab | 2.33 ± 0.01 | 3.40 ± 0.07 ab | 3.25 ± 0.04 | 3.72 ± 0.09 | 3.68 ± 0.14 | 10.74 ± 0.23 ab | 2.71 ± 0.01 | |
| 10 | 2.66 ± 0.04 | 3.43 ± 0.09 b | 2.32 ± 0.01 | 8.21 ± 0.07 ab | 2.32 ± 0.01 | 3.43 ± 0.05 ab | 3.30 ± 0.07 | 3.72 ± 0.04 | 3.60 ± 0.10 | 10.81 ± 0.26 ab | 2.71 ± 0.01 | |
| LF60 | 0 | 2.66 ± 0.08 | 3.11 ± 0.17 b | 2.30 ± 0.06 | 6.71 ± 0.28 | 2.30 ± 0.01 | 2.92 ± 0.04 | 2.92 ± 0.06 | 3.28 ± 0.21 | 3.61 ± 0.17 | 8.30 ± 0.73 | 2.55 ± 0.06 |
| 5 | 2.61 ± 0.02 | 2.97 ± 0.14 ab | 2.24 ± 0.02 | 6.58 ± 0.13 | 2.26 ± 0.01 | 2.96 ± 0.02 | 2.90 ± 0.06 | 3.28 ± 0.05 | 3.62 ± 0.09 | 8.13 ± 0.24 | 2.57 ± 0.04 | |
| 15 | 2.64 ± 0.09 | 2.98 ± 0.03 ab | 2.31 ± 0.06 | 6.64 ± 0.19 | 2.25 ± 0.04 | 2.90 ± 0.07 | 2.90 ± 0.04 | 3.25 ± 0.21 | 3.65 ± 0.06 | 8.13 ± 0.69 | 2.51 ± 0.02 | |
| 25 | 2.63 ± 0.05 | 2.93 ± 0.02 a | 2.28 ± 0.03 | 6.41 ± 0.15 | 2.26 ± 0.03 | 2.94 ± 0.02 | 2.79 ± 0.02 | 3.27 ± 0.20 | 3.74 ± 0.10 | 7.72 ± 0.51 | 2.52 ± 0.05 | |
| HF60 | 0 | 2.63 ± 0.03 | 3.05 ± 0.10 | 2.29 ± 0.01 | 6.82 ± 0.46 | 2.24 ± 0.01 | 3.03 ± 0.08 | 2.93 ± 0.08 | 3.26 ± 0.09 | 3.65 ± 0.02 | 8.48 ± 0.72 | 2.52 ± 0.04 |
| 5 | 2.65 ± 0.03 | 3.11 ± 0.09 | 2.28 ± 0.02 | 7.04 ± 0.19 | 2.26 ± 0.01 | 3.04 ± 0.10 | 3.00 ± 0.10 | 3.45 ± 0.11 | 3.62 ± 0.10 | 9.12 ± 0.81 | 2.55 ± 0.04 | |
| 15 | 2.63 ± 0.06 | 3.04 ± 0.07 | 2.30 ± 0.01 | 6.95 ± 0.19 | 2.26 ± 0.01 | 3.13 ± 0.06 | 2.95 ± 0.07 | 3.21 ± 0.09 | 3.61 ± 0.12 | 8.44 ± 0.40 | 2.60 ± 0.06 | |
| 25 | 2.63 ± 0.03 | 3.03 ± 0.13 | 2.27 ± 0.01 | 6.73 ± 0.42 | 2.24 ± 0.02 | 2.97 ± 0.07 | 2.90 ± 0.07 | 3.27 ± 0.18 | 3.68 ± 0.06 | 8.21 ± 0.96 | 2.53 ± 0.03 | |
| LF90 | 0 | 2.65 ± 0.05 | 3.03 ± 0.10 | 2.27 ± 0.03 | 6.65 ± 0.22 | 2.26 ± 0.03 | 2.94 ± 0.02 | 2.81 ± 0.04 | 3.39 ± 0.14 | 3.66 ± 0.19 | 8.22 ± 0.34 | 2.50 ± 0.02 |
| 5 | 2.66 ± 0.07 | 3.09 ± 0.09 | 2.30 ± 0.01 | 6.71 ± 0.15 | 2.27 ± 0.03 | 3.04 ± 0.02 | 2.91 ± 0.10 | 3.36 ± 0.04 | 3.53 ± 0.14 | 8.68 ± 0.23 | 2.52 ± 0.02 | |
| 15 | 2.63 ± 0.05 | 3.08 ± 0.04 | 2.28 ± 0.05 | 6.79 ± 0.24 | 2.29 ± 0.04 | 3.10 ± 0.15 | 2.91 ± 0.07 | 3.44 ± 0.10 | 3.67 ± 0.14 | 8.81 ± 0.26 | 2.58 ± 0.08 | |
| 25 | 2.66 ± 0.04 | 3.06 ± 0.10 | 2.29 ± 0.03 | 6.93 ± 0.24 | 2.26 ± 0.01 | 3.07 ± 0.07 | 2.95 ± 0.07 | 3.37 ± 0.17 | 3.61 ± 0.14 | 8.63 ± 0.54 | 2.60 ± 0.11 | |
| LF90 | 0 | 2.64 ± 0.05 | 3.28 ± 0.05 | 2.34 ± 0.02 | 7.87 ± 0.25 | 2.32 ± 0.02 | 3.32 ± 0.05 | 3.26 ± 0.10 | 3.73 ± 0.09 | 3.57 ± 0.11 | 10.54 ± 0.64 | 2.69 ± 0.03 |
| 5 | 2.63 ± 0.05 | 3.37 ± 0.07 | 2.34 ± 0.03 | 8.24 ± 0.25 | 2.32 ± 0.02 | 3.36 ± 0.05 | 3.26 ± 0.12 | 3.69 ± 0.11 | 3.61 ± 0.10 | 10.55 ± 0.37 | 2.70 ± 0.03 | |
| 15 | 2.62 ± 0.03 | 3.31 ± 0.07 | 2.32 ± 0.02 | 8.03 ± 0.35 | 2.31 ± 0.01 | 3.36 ± 0.04 | 3.20 ± 0.07 | 3.60 ± 0.05 | 3.68 ± 0.08 | 10.36 ± 0.4 | 2.70 ± 0.05 | |
| 25 | 2.61 ± 0.01 | 3.23 ± 0.08 | 2.33 ± 0.01 | 7.92 ± 0.17 | 2.32 ± 0.02 | 3.28 ± 0.07 | 3.13 ± 0.03 | 3.60 ± 0.08 | 3.70 ± 0.06 | 9.89 ± 0.37 | 2.64 ± 0.04 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sainz-García, E.; López-Alfaro, I.; Múgica-Vidal, R.; López, R.; Escribano-Viana, R.; Portu, J.; Alba-Elías, F.; González-Arenzana, L. Effect of the Atmospheric Pressure Cold Plasma Treatment on Tempranillo Red Wine Quality in Batch and Flow Systems. Beverages 2019, 5, 50. https://doi.org/10.3390/beverages5030050
Sainz-García E, López-Alfaro I, Múgica-Vidal R, López R, Escribano-Viana R, Portu J, Alba-Elías F, González-Arenzana L. Effect of the Atmospheric Pressure Cold Plasma Treatment on Tempranillo Red Wine Quality in Batch and Flow Systems. Beverages. 2019; 5(3):50. https://doi.org/10.3390/beverages5030050
Chicago/Turabian StyleSainz-García, Elisa, Isabel López-Alfaro, Rodolfo Múgica-Vidal, Rosa López, Rocío Escribano-Viana, Javier Portu, Fernando Alba-Elías, and Lucía González-Arenzana. 2019. "Effect of the Atmospheric Pressure Cold Plasma Treatment on Tempranillo Red Wine Quality in Batch and Flow Systems" Beverages 5, no. 3: 50. https://doi.org/10.3390/beverages5030050
APA StyleSainz-García, E., López-Alfaro, I., Múgica-Vidal, R., López, R., Escribano-Viana, R., Portu, J., Alba-Elías, F., & González-Arenzana, L. (2019). Effect of the Atmospheric Pressure Cold Plasma Treatment on Tempranillo Red Wine Quality in Batch and Flow Systems. Beverages, 5(3), 50. https://doi.org/10.3390/beverages5030050






