Potential Applications of High Pressure Homogenization in Winemaking: A Review
Abstract
1. Introduction
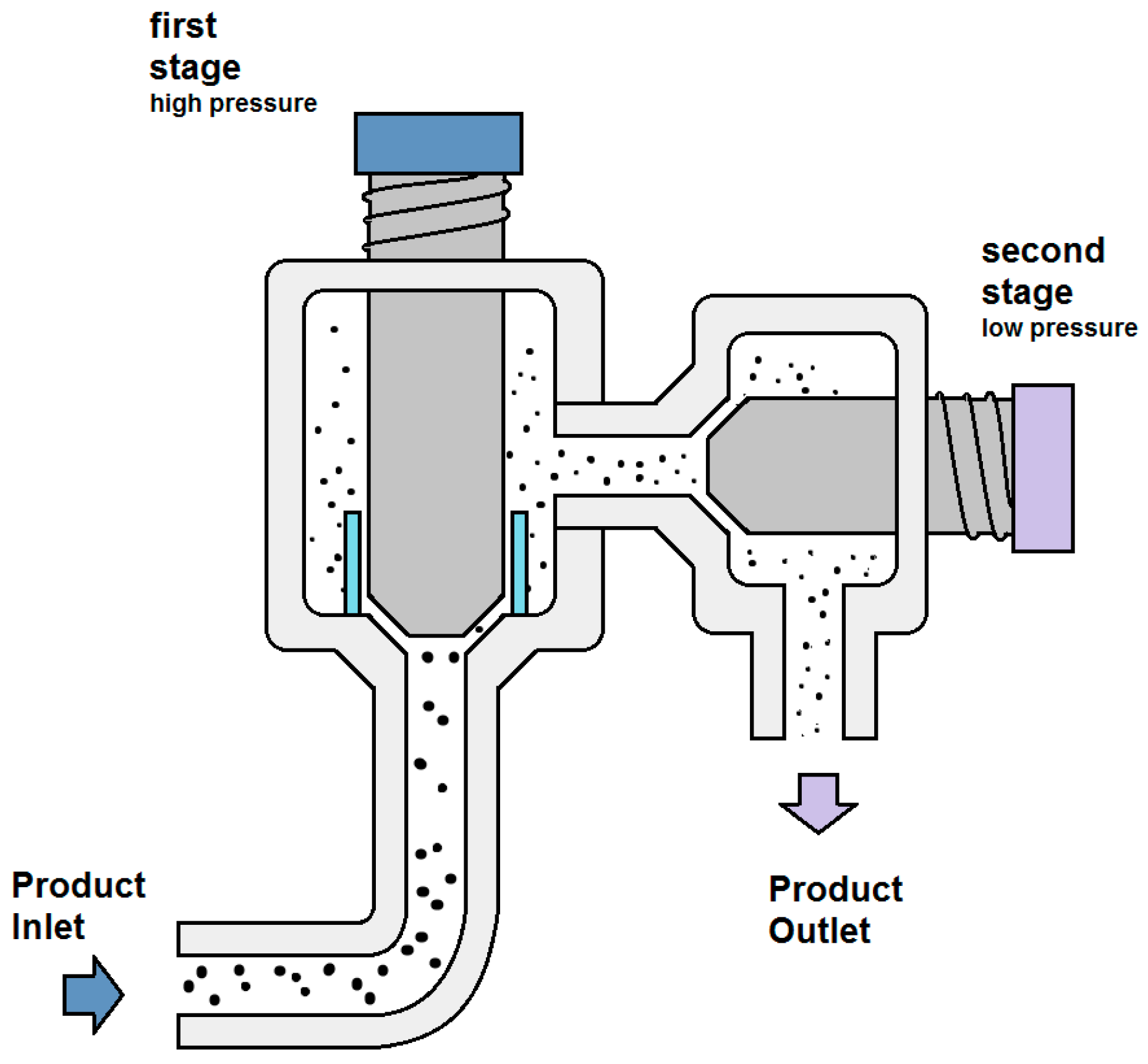
2. Basic Principles of High Pressure Homogenization and Process Design
3. Possible Applications of HPH in the Food Sector
3.1. Emulsification and Nanoemulsification
3.2. Microbial Inactivation
3.3. Cell Disruption and Recovery of Intracellular Components
3.4. Physical and Physical-Chemical Modifications of Food Biopolymers
3.5. Enzyme Inactivation
4. Potential Use of HPH in Winemaking
4.1. Control of Microbial Populations in Grape Juice and Wine
4.2. Acceleration of Yeast Autolysis and Ageing on Lees
4.3. Production of Yeast Derivative Preparations for Enological Use
4.4. Modifications Induced on Wine
4.5. Other Potential Applications and Perspectives of HPH in Wine
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Corrales, M.; Toepfl, S.; Butz, P.; Knorr, D.; Tauscher, B. Extraction of anthocyanins from grape by-products assisted by ultrasonics, high hydrostatic pressure or pulsed electric fields: A comparison. Innov. Food Sci. Emerg. Technol. 2008, 9, 85–91. [Google Scholar] [CrossRef]
- Morata, A.; Loira, I.; Vejarano, R.; Bañuelos, M.A.; Sanz, P.D.; Otero, L.; Suárez-Lepe, J.A. Grape Processing by High Hydrostatic Pressure: Effect on Microbial Populations, Phenol Extraction and Wine Quality. Food Bioprocess Technol. 2015, 8, 277–286. [Google Scholar] [CrossRef]
- Santos, M.C.; Nunes, C.; Saraiva, J.A.; Coimbra, M.A. Chemical and physical methodologies for the replacement/reduction of sulfur dioxide use during winemaking: Review of their potentialities and limitations. Eur. Food Res. Technol. 2012, 234, 1–12. [Google Scholar] [CrossRef]
- Ribéreau-Gayon, P.; Dubourdieu, D.; Donèche, B.; Lonvaud, A. Handbook of Enology Vol. 1 The Microbiology of Wine and Vinifications, 2nd ed.; John Wiley & Sons Ltd.: West Sussex, UK, 2006. [Google Scholar]
- Commission Regulation (EC) No. 606 Laying down certain detailed rules for implementing Council Regulation (EC) No 479/2008 as regards the categories of grapevine products, oenological practices and the applicable restrictions. J. Eur. Commun. 2009, 193, 1–59.
- International Organization of Vine and Wine, O.I.V. International Code of Oenological Practices; O.I.V.: Paris, France, 2018. [Google Scholar]
- Walstra, P. Principles of emulsion formation. Chem. Eng. Sci. 1993, 48, 333–349. [Google Scholar] [CrossRef]
- Dumay, E.; Chevalier-Lucia, D.; Picart-Palmade, L.; Benzaria, A.; Gràcia-Julià, A.; Blayo, C. Technological aspects and potential applications of (ultra) high-pressure homogenisation. Trends Food Sci. Technol. 2013, 31, 13–26. [Google Scholar] [CrossRef]
- Floury, J.; Bellettre, J.; Legrand, J.; Desrumaux, A. Analysis of a new type of high pressure homogeniser. A study of the flow pattern. Chem. Eng. Sci. 2004, 59, 843–853. [Google Scholar] [CrossRef]
- Martínez-Monteagudo, S.I.; Yan, B.; Balasubramaniam, V.M. Engineering Process Characterization of High-Pressure Homogenization—from Laboratory to Industrial Scale. Food Eng. Rev. 2017, 9, 143–169. [Google Scholar] [CrossRef]
- Stang, M.; Schauschmann, H.; Schubert, H. Emulsification in high-pressure homogenizers. Eng. Life Sci. 2001, 1, 151–157. [Google Scholar] [CrossRef]
- Calligaris, S.; Plazzotta, S.; Bot, F.; Grasselli, S.; Malchiodi, A.; Anese, M. Nanoemulsion preparation by combining high pressure homogenization and high power ultrasound at low energy densities. Food Res. Int. 2016, 83, 25–30. [Google Scholar] [CrossRef]
- Plazzotta, S.; Manzocco, L. Effect of ultrasounds and high pressure homogenization on the extraction of antioxidant polyphenols from lettuce waste. Innov. Food Sci. Emerg. Technol. 2018, 50, 11–19. [Google Scholar] [CrossRef]
- Balasubramaniam, V.M.B.; Martínez-Monteagudo, S.I.; Gupta, R. Principles and application of high pressure-based technologies in the food industry. Annu. Rev. Food Sci. Technol. 2015, 6, 435–462. [Google Scholar] [CrossRef] [PubMed]
- Zamora, A.; Guamis, B. Opportunities for Ultra-High-Pressure Homogenisation (UHPH) for the Food Industry. Food Eng. Rev. 2015, 7, 130–142. [Google Scholar] [CrossRef]
- McClements, D.J. Edible nanoemulsions: Fabrication, properties, and functional performance. Soft Matter 2011, 7, 2297–2316. [Google Scholar] [CrossRef]
- Silva, H.D.; Cerqueira, M.A.; Vicente, A.A. Nanoemulsions for Food Applications: Development and Characterization. Food Bioprocess Technol. 2012, 5, 854–867. [Google Scholar] [CrossRef]
- Håkansson, A. Emulsion Formation by Homogenization: Current Understanding and Future Perspectives. Annu. Rev. Food Sci. Technol. 2019, 10, 239–258. [Google Scholar] [CrossRef] [PubMed]
- Patrignani, F.; Lanciotti, R. Applications of high and ultra high pressure homogenization for food safety. Front. Microbiol. 2016, 7, 1132. [Google Scholar] [CrossRef]
- Sevenich, R.; Mathys, A. Continuous Versus Discontinuous Ultra-High-Pressure Systems for Food Sterilization with Focus on Ultra-High-Pressure Homogenization and High-Pressure Thermal Sterilization: A Review. Compr. Rev. Food Sci. Food Saf. 2018, 17, 646–662. [Google Scholar] [CrossRef]
- Barba, F.J.; Grimi, N.; Vorobiev, E. New Approaches for the Use of Non-conventional Cell Disruption Technologies to Extract Potential Food Additives and Nutraceuticals from Microalgae. Food Eng. Rev. 2014, 7, 45–62. [Google Scholar] [CrossRef]
- Liu, D.; Ding, L.; Sun, J.; Boussetta, N.; Vorobiev, E. Yeast cell disruption strategies for recovery of intracellular bio-active compounds—A review. Innov. Food Sci. Emerg. Technol. 2016, 36, 181–192. [Google Scholar] [CrossRef]
- Belmiro, R.H.; Tribst, A.A.L.; Cristianini, M. Application of high-pressure homogenization on gums. J. Sci. Food Agric. 2018, 98, 2060–2069. [Google Scholar] [CrossRef] [PubMed]
- dos Santos Aguilar, J.; Cristianini, M.; Sato, H. Modification of enzymes by use of high-pressure homogenization. Food Res. Int. 2018, 109, 120–125. [Google Scholar] [CrossRef] [PubMed]
- Floury, J.; Desrumaux, A.; Lardières, J. Effect of high-pressure homogenization on droplet size distributions and rheological properties of model oil-in-water emulsions. Innov. Food Sci. Emerg. Technol. 2000, 1, 127–134. [Google Scholar] [CrossRef]
- Qian, C.; McClements, D.J. Formation of nanoemulsions stabilized by model food-grade emulsifiers using high-pressure homogenization: Factors affecting particle size. Food Hydrocoll. 2011, 25, 1000–1008. [Google Scholar] [CrossRef]
- Calligaris, S.; Plazzotta, S.; Valoppi, F.; Anese, M. Combined high-power ultrasound and high-pressure homogenization nanoemulsification: The effect of energy density, oil content and emulsifier type and content. Food Res. Int. 2018, 107, 700–707. [Google Scholar] [CrossRef]
- Donsì, F.; Ferrari, G.; Lenza, E.; Maresca, P. Main factors regulating microbial inactivation by high-pressure homogenization: Operating parameters and scale of operation. Chem. Eng. Sci. 2009, 64, 520–532. [Google Scholar] [CrossRef]
- Innocente, N.; Marino, M.; Calligaris, S. Recovery of brines from cheesemaking using High-Pressure Homogenization treatments. J. Food Eng. 2019, 247, 188–194. [Google Scholar] [CrossRef]
- Thiebaud, M.; Dumay, E.; Picart, L.; Guiraud, J.P.; Cheftel, J.C. High-pressure homogenisation of raw bovine milk. Effects on fat globule size distribution and microbial inactivation. Int. Dairy J. 2003, 13, 427–439. [Google Scholar] [CrossRef]
- Wuytack, E.Y.; Diels, A.M.J.; Michiels, C.W. Bacterial inactivation by high-pressure homogenisation and high hydrostatic pressure. Int. J. Food Microbiol. 2002, 77, 205–212. [Google Scholar] [CrossRef]
- Vachon, J.F.; Kheadr, E.E.; Giasson, J.; Paquin, P.; Fliss, I. Inactivation of foodborne pathogens in milk using dynamic high pressure. J. Food Prot. 2002, 65, 345–352. [Google Scholar] [CrossRef]
- Lanciotti, R.; Vannini, L.; Patrignani, F.; Iucci, L.; Vallicelli, M.; Ndagijimana, M.; Guerzoni, M.E. Effect of high pressure homogenisation of milk on cheese yield and microbiology, lipolysis and proteolysis during ripening of Caciotta cheese. J. Dairy Res. 2006, 73, 216–226. [Google Scholar] [CrossRef] [PubMed]
- Tahiri, I.; Makhlouf, J.; Paquin, P.; Fliss, I. Inactivation of food spoilage bacteria and Escherichia coli O157:H7 in phosphate buffer and orange juice using dynamic high pressure. Food Res. Int. 2006, 39, 98–105. [Google Scholar] [CrossRef]
- Chaves-López, C.; Lanciotti, R.; Serio, A.; Paparella, A.; Guerzoni, E.; Suzzi, G. Effect of high pressure homogenization applied individually or in combination with other mild physical or chemical stresses on Bacillus cereus and Bacillus subtilis spore viability. Food Control 2009, 20, 691–695. [Google Scholar] [CrossRef]
- Chen, W.; Harte, F.M.; Davidson, P.M.; Golden, D.A. Inactivation of Alicyclobacillus acidoterrestris using high pressure homogenization and dimethyl dicarbonate. J. Food Prot. 2013, 76, 1041–1045. [Google Scholar] [CrossRef] [PubMed]
- Bevilacqua, A.; Corbo, M.R.; Sinigaglia, M. Use of natural antimicrobials and high pressure homogenization to control the growth of Saccharomyces bayanus in apple juice. Food Control 2012, 24, 109–115. [Google Scholar] [CrossRef]
- Poliseli-Scopel, F.H.; Hernández-Herrero, M.; Guamis, B.; Ferragut, V. Sterilization and aseptic packaging of soymilk treated by ultra high pressure homogenization. Innov. Food Sci. Emerg. Technol. 2014, 22, 81–88. [Google Scholar] [CrossRef]
- Calligaris, S.; Foschia, M.; Bartolomeoli, I.; Maifreni, M.; Manzocco, L. Study on the applicability of high-pressure homogenization for the production of banana juices. LWT Food Sci. Technol. 2012, 45, 117–121. [Google Scholar] [CrossRef]
- Maresca, P.; Donsì, F.; Ferrari, G. Application of a multi-pass high-pressure homogenization treatment for the pasteurization of fruit juices. J. Food Eng. 2011, 104, 364–372. [Google Scholar] [CrossRef]
- Dong, X.; Zhao, M.; Shi, J.; Yang, B.; Li, J.; Luo, D.; Jiang, G.; Jiang, Y. Effects of combined high-pressure homogenization and enzymatic treatment on extraction yield, hydrolysis and function properties of peanut proteins. Innov. Food Sci. Emerg. Technol. 2011, 12, 478–483. [Google Scholar] [CrossRef]
- Tao, X.; Cai, Y.; Liu, T.; Long, Z.; Huang, L.; Deng, X.; Zhao, Q.; Zhao, M. Effects of pretreatments on the structure and functional properties of okara protein. Food Hydrocoll. 2019, 90, 394–402. [Google Scholar] [CrossRef]
- Cho, S.-C.; Choi, W.-Y.; Oh, S.-H.; Lee, C.-G.; Seo, Y.-C.; Kim, J.-S.; Song, C.-H.; Kim, G.-V.; Lee, S.-Y.; Kang, D.-H.; et al. Enhancement of lipid extraction from marine microalga, Scenedesmus associated with high-pressure homogenization process. J. Biomed. Biotechnol. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Augusto, P.E.D.; Ibarz, A.; Cristianini, M. Effect of high pressure homogenization (HPH) on the rheological properties of a fruit juice serum model. J. Food Eng. 2012, 111, 474–477. [Google Scholar] [CrossRef]
- Bot, F.; Calligaris, S.; Cortella, G.; Nocera, F.; Peressini, D.; Anese, M. Effect of high pressure homogenization and high power ultrasound on some physical properties of tomato juices with different concentration levels. J. Food Eng. 2017, 213, 10–17. [Google Scholar] [CrossRef]
- Venir, E.; Marchesini, G.; Biasutti, M.; Innocente, N. Dynamic high pressure-induced gelation in milk protein model systems. J. Dairy Sci. 2010, 93, 483–494. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Zhou, C.; Fu, F.; Chen, Z.; Wu, Q. Effect of high-pressure homogenization on particle size and film properties of soy protein isolate. Ind. Crops Prod. 2013, 43, 538–544. [Google Scholar] [CrossRef]
- Liu, H.-H.; Kuo, M.-I. Ultra high pressure homogenization effect on the proteins in soy flour. Food Hydrocoll. 2016, 52, 741–748. [Google Scholar] [CrossRef]
- Chen, X.; Zhou, R.; Xu, X.; Zhou, G.; Liu, D. Structural modification by high-pressure homogenization for improved functional properties of freeze-dried myofibrillar proteins powder. Food Res. Int. 2017, 100, 193–200. [Google Scholar] [CrossRef]
- Yu, C.; Cha, Y.; Wu, F.; Xu, X.; Qin, Y.; Li, X.; Du, M. Effects of high-pressure homogenisation on structural and functional properties of mussel (Mytilus edulis) protein isolate. Int. J. Food Sci. Technol. 2018, 53, 1157–1165. [Google Scholar] [CrossRef]
- Panozzo, A.; Manzocco, L.; Calligaris, S.; Bartolomeoli, I.; Maifreni, M.; Lippe, G.; Nicoli, M.C. Effect of high pressure homogenisation on microbial inactivation, protein structure and functionality of egg white. Food Res. Int. 2014, 62, 718–725. [Google Scholar] [CrossRef]
- Villay, A.; de Filippis, F.; Picton, L.; Le Cerf, D.; Vial, C.; Michaud, P. Comparison of polysaccharide degradations by dynamic high-pressure homogenization. Food Hydrocoll. 2012, 27, 278–286. [Google Scholar] [CrossRef]
- Le Thanh-Blicharz, J.; Lewandowicz, G.; Błaszczak, W.; Prochaska, K. Starch modified by high-pressure homogenisation of the pastes - Some structural and physico-chemical aspects. Food Hydrocoll. 2012, 27, 347–354. [Google Scholar] [CrossRef]
- Floury, J.; Desrumaux, A.; Axelos, M.A.V.; Legrand, J. Degradation of methylcellulose during ultra-high pressure homogenisation. Food Hydrocoll. 2002, 16, 47–53. [Google Scholar] [CrossRef]
- Alvarez-Sabatel, S.; Marañón, I.M.D.; Arboleya, J.-C. Impact of high pressure homogenisation (HPH) on inulin gelling properties, stability and development during storage. Food Hydrocoll. 2015, 44, 333–344. [Google Scholar] [CrossRef]
- Bot, F.; Calligaris, S.; Cortella, G.; Plazzotta, S.; Nocera, F.; Anese, M. Study on high pressure homogenization and high power ultrasound effectiveness in inhibiting polyphenoloxidase activity in apple juice. J. Food Eng. 2018, 221, 70–76. [Google Scholar] [CrossRef]
- Delfini, C.; Conterno, L.; Carpi, G.; Rovere, P.; Tabusso, A.; Cocito, C.; Amati, A. Microbiological stabilisation of grape musts and wines by high hydrostatic pressures. J. Wine Res. 1995, 6, 143–151. [Google Scholar] [CrossRef]
- Corrales, M.; García, A.F.; Butz, P.; Tauscher, B. Extraction of anthocyanins from grape skins assisted by high hydrostatic pressure. J. Food Eng. 2009, 90, 415–421. [Google Scholar] [CrossRef]
- Morata, A.; Loira, I.; Vejarano, R.; González, C.; Callejo, M.J.; Suárez-Lepe, J.A. Emerging preservation technologies in grapes for winemaking. Trends Food Sci. Technol. 2017, 67, 36–43. [Google Scholar] [CrossRef]
- Bañuelos, M.A.; Loira, I.; Escott, C.; Del Fresno, J.M.; Morata, A.; Sanz, P.D.; Otero, L.; Suárez-Lepe, J.A. Grape Processing by High Hydrostatic Pressure: Effect on Use of Non-Saccharomyces in Must Fermentation. Food Bioprocess Technol. 2016, 9, 1769–1778. [Google Scholar] [CrossRef]
- Mok, C.; Song, K.T.; Park, Y.S.; Lim, S.; Ruan, R.; Chen, P. High hydrostatic pressure pasteurization of red wine. J. Food Sci. 2006, 71, M265–M269. [Google Scholar] [CrossRef]
- Santos, L.M.R.; Oliveira, F.A.; Ferreira, E.H.R.; Rosenthal, A. Application and possible benefits of high hydrostatic pressure or high-pressure homogenization on beer processing: A review. Food Sci. Technol. Int. 2017, 23, 561–581. [Google Scholar] [CrossRef]
- Puig, A.; Vilavella, M.; Daoudi, L.; Guamis, B.; Minguez, S. Microbiological and biochemical stabilization of wines using the high pressure technique. Bull. OIV 2003, 869, 569–617. [Google Scholar]
- Loira, I.; Morata, A.; Bañuelos, M.A.; Puig-Pujol, A.; Guamis, B.; González, C.; Suárez-Lepe, J.A. Use of Ultra-High Pressure Homogenization processing in winemaking: Control of microbial populations in grape musts and effects in sensory quality. Innov. Food Sci. Emerg. Technol. 2018, 50, 50–56. [Google Scholar] [CrossRef]
- Puig, A.; Olmos, P.; Quevedo, J.M.; Guamis, B.; Mínguez, S. Microbiological and Sensory Effects of Musts Treated by High-pressure Homogenization. Food Sci. Technol. Int. 2008, 14, 5–11. [Google Scholar] [CrossRef]
- Comuzzo, P.; Calligaris, S.; Iacumin, L.; Ginaldi, F.; Voce, S.; Zironi, R. Application of multi-pass high pressure homogenization under variable temperature regimes to induce autolysis of wine yeasts. Food Chem. 2017, 224, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Comuzzo, P.; Calligaris, S.; Iacumin, L.; Ginaldi, F.; Palacios Paz, A.E.; Zironi, R. Potential of high pressure homogenization to induce autolysis of wine yeasts. Food Chem. 2015, 185, 340–348. [Google Scholar] [CrossRef]
- Campos, F.P.; Cristianini, M. Inactivation of Saccharomyces cerevisiae and Lactobacillus plantarum in orange juice using ultra high-pressure homogenisation. Innov. Food Sci. Emerg. Technol. 2007, 8, 226–229. [Google Scholar] [CrossRef]
- Franchi, M.A.; Tribst, A.A.L.; Cristianini, M. High-pressure homogenization: A non-thermal process applied for inactivation of spoilage microorganisms in beer. J. Inst. Brew. 2013, 119, 237–241. [Google Scholar] [CrossRef]
- Lee, M.G.; Ham, T.H.; Song, S.H.; Chung, D.H.; Yoon, W.B. Effects of the high pressure homogenization on the viability of yeast cell and volatile components in non-pasteurized rice wine. Food Sci. Biotechnol. 2016, 25, 1073–1080. [Google Scholar] [CrossRef]
- Charpentier, C.; Feuillat, M. Yeast autolysis. In Wine Microbiology and Biotechnology; Fleet, G.H., Ed.; Taylor and Francis: NY, New York, USA, 1993; pp. 225–242. [Google Scholar]
- Siddiqi, S.F.; Titchener-Hooker, N.J.; Shamlou, P.A. High pressure disruption of yeast cells: The use of scale down operations for the prediction of protein release and cell debris size distribution. Biotechnol. Bioeng. 1997, 55, 642–649. [Google Scholar] [CrossRef]
- Hetherington, P.J.; Follows, M.; Dunhill, P.; Lilly, M.D. Release of protein from baker’s yeast (Saccharomyces cerevisiae) by disruption in an industrial homogeniser. Trans. Inst. Chem. Eng. 1971, 49, 142–148. [Google Scholar]
- Follows, M.; Hetherington, P.J.; Dunnill, P.; Lilly, M.D. Release of enzymes from bakers’ yeast by disruption in an industrial homogenizer. Biotechnol. Bioeng. 1971, 13, 549–560. [Google Scholar] [CrossRef] [PubMed]
- Patrignani, F.; Ndagijimana, M.; Vernocchi, P.; Gianotti, A.; Riponi, C.; Gardini, F.; Lanciotti, R. High-Pressure homogenization to modify yeast performance for sparkling wine production according to traditional methods. Am. J. Enol. Vitic. 2013, 64, 258–267. [Google Scholar] [CrossRef]
- Shynkaryk, M.V.; Lebovka, N.I.; Lanoisellé, J.L.; Nonus, M.; Bedel-Clotour, C.; Vorobiev, E. Electrically-assisted extraction of bio-products using high pressure disruption of yeast cells (Saccharomyces cerevisiae). J. Food Eng. 2009, 92, 189–195. [Google Scholar] [CrossRef]
- González-Marco, A.; Ancín-Azpilicueta, C. Influence of lees contact on evolution of amines in Chardonnay wine. J. Food Sci. 2006, 71, 544–548. [Google Scholar] [CrossRef]
- Carrano, G. Gestione Dell’elevage Sur Lies Mediante Applicazione di Tecnologie Basate Sull’alta Pressione; University of Udine: Udine, Italy, 2016. [Google Scholar]
- Pozo-Bayón, M.Á.; Andújar-Ortiz, I.; Moreno-Arribas, M.V. Scientific evidences beyond the application of inactive dry yeast preparations in winemaking. Food Res. Int. 2009, 42, 754–761. [Google Scholar] [CrossRef]
- International Organization of Vine and Wine, O.I.V. International Oenological Codex; O.I.V.: Paris, France, 2018. [Google Scholar]
- Comuzzo, P.; Tat, L.; Tonizzo, A.; Battistutta, F. Yeast derivatives (extracts and autolysates) in winemaking: Release of volatile compounds and effects on wine aroma volatility. Food Chem. 2006, 99, 217–230. [Google Scholar] [CrossRef]
- Pozo-Bayón, M.Á.; Andújar-Ortiz, I.; Moreno-Arribas, M.V. Volatile profile and potential of inactive dry yeast-based winemaking additives to modify the volatile composition of wines. J. Sci. Food Agric. 2009, 89, 1665–1673. [Google Scholar] [CrossRef]
- Santos, M.C.; Nunes, C.; Ferreira, A.S.; Jourdes, M.; Teissedre, P.-L.; Rodrigues, A.; Amado, O.; Saraiva, J.A.; Coimbra, M.A. Comparison of high pressure treatment with conventional red wine aging processes: Impact on phenolic composition. Food Res. Int. 2019, 116, 223–231. [Google Scholar] [CrossRef]
- Talcott, S.T.; Brenes, C.H.; Pires, D.M.; Del Pozo-Insfran, D. Phytochemical stability and color retention of copigmented and processed muscadine grape juice. J. Agric. Food Chem. 2003, 51, 957–963. [Google Scholar] [CrossRef]
- Santos, M.C.; Nunes, C.; Rocha, M.A.M.; Rodrigues, A.; Rocha, S.M.; Saraiva, J.A.; Coimbra, M.A. High pressure treatments accelerate changes in volatile composition of sulphur dioxide-free wine during bottle storage. Food Chem. 2015, 188, 406–414. [Google Scholar] [CrossRef]
- Santos, M.C.; Nunes, C.; Rocha, M.A.M.; Rodrigues, A.; Rocha, S.M.; Saraiva, J.A.; Coimbra, M.A. Impact of high pressure treatments on the physicochemical properties of a sulphur dioxide-free white wine during bottle storage: Evidence for Maillard reaction acceleration. Innov. Food Sci. Emerg. Technol. 2013, 20, 51–58. [Google Scholar] [CrossRef]
- Serrazanetti, D.I.; Patrignani, F.; Russo, A.; Vannini, L.; Siroli, L.; Gardini, F.; Lanciotti, R. Cell membrane fatty acid changes and desaturase expression of Saccharomyces bayanus exposed to high pressure homogenization in relation to the supplementation of exogenous unsaturated fatty acids. Front. Microbiol. 2015, 6, 1–10. [Google Scholar] [CrossRef] [PubMed]
| Application | Literature Reviews |
|---|---|
| Emulsification—Nanoemulsification | [16,17,18] |
| Microbial inactivation | [19,20] |
| Cell disruption and recovery of intracellular components | [21,22] |
| Physical and physical–chemical modifications of food biopolymers | [23] |
| Enzyme inactivation | [24] |
| Sample | Total Yeasts (Log CFU/g) |
|---|---|
| ADY 1 | 10.8 |
| 50 MPa 2 | 9.9 |
| 100 MPa 2 | 9.2 |
| 150 MPa 2 | 8.6 |
| Sample | Saccharomyces spp. (Log CFU/mL) | NON Saccharomyces spp. (Log CFU/mL) | LAB (Log CFU/mL) | Temperature (°C) 2 |
|---|---|---|---|---|
| Untreated | 1.9 | 1.3 | 3.9 | - |
| 60 MPa | 1.4 | 0.7 | 3.1 | 40 |
| 150 MPa | n.d.1 | n.d. | n.d. | 50 |
| Code | Soluble Proteins 2 (mg/g) | Total Colloids 2 (mg/g) |
|---|---|---|
| ADY 1 | 14 | 43 |
| 50 MPa | 36 | 101 |
| 100 MPa | 47 | 128 |
| 150 MPa | 51 | 165 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Comuzzo, P.; Calligaris, S. Potential Applications of High Pressure Homogenization in Winemaking: A Review. Beverages 2019, 5, 56. https://doi.org/10.3390/beverages5030056
Comuzzo P, Calligaris S. Potential Applications of High Pressure Homogenization in Winemaking: A Review. Beverages. 2019; 5(3):56. https://doi.org/10.3390/beverages5030056
Chicago/Turabian StyleComuzzo, Piergiorgio, and Sonia Calligaris. 2019. "Potential Applications of High Pressure Homogenization in Winemaking: A Review" Beverages 5, no. 3: 56. https://doi.org/10.3390/beverages5030056
APA StyleComuzzo, P., & Calligaris, S. (2019). Potential Applications of High Pressure Homogenization in Winemaking: A Review. Beverages, 5(3), 56. https://doi.org/10.3390/beverages5030056






