Automated Collateral Classification on CT Angiography in Acute Ischemic Stroke: Performance Trends Across Hyperparameter Combinations
Abstract
1. Introduction
2. Materials and Methods
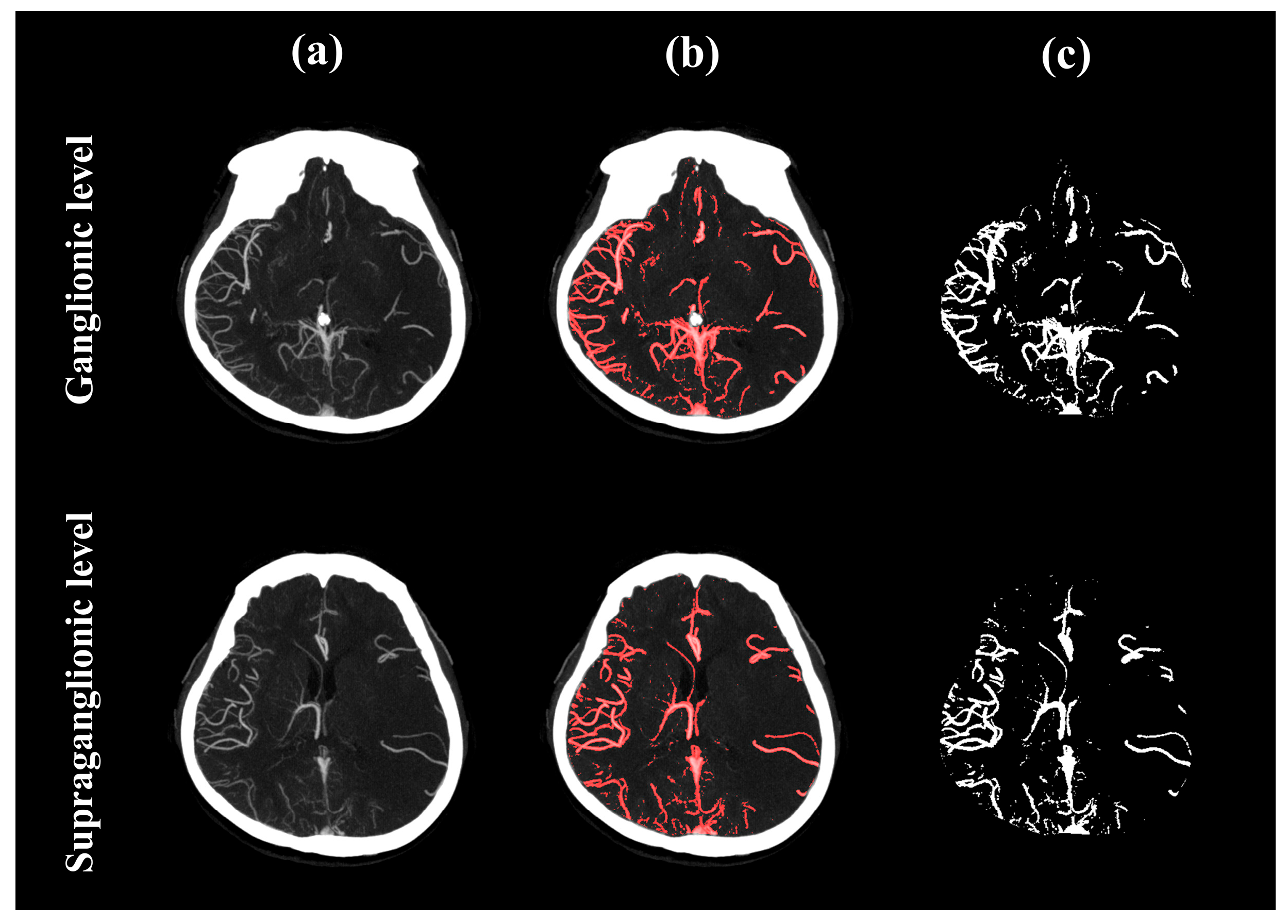
2.1. Data Acquisition
2.2. Image Preprocessing
2.3. Collateral Classification Model
2.4. Performance Metrics
2.5. Statistical Analysis
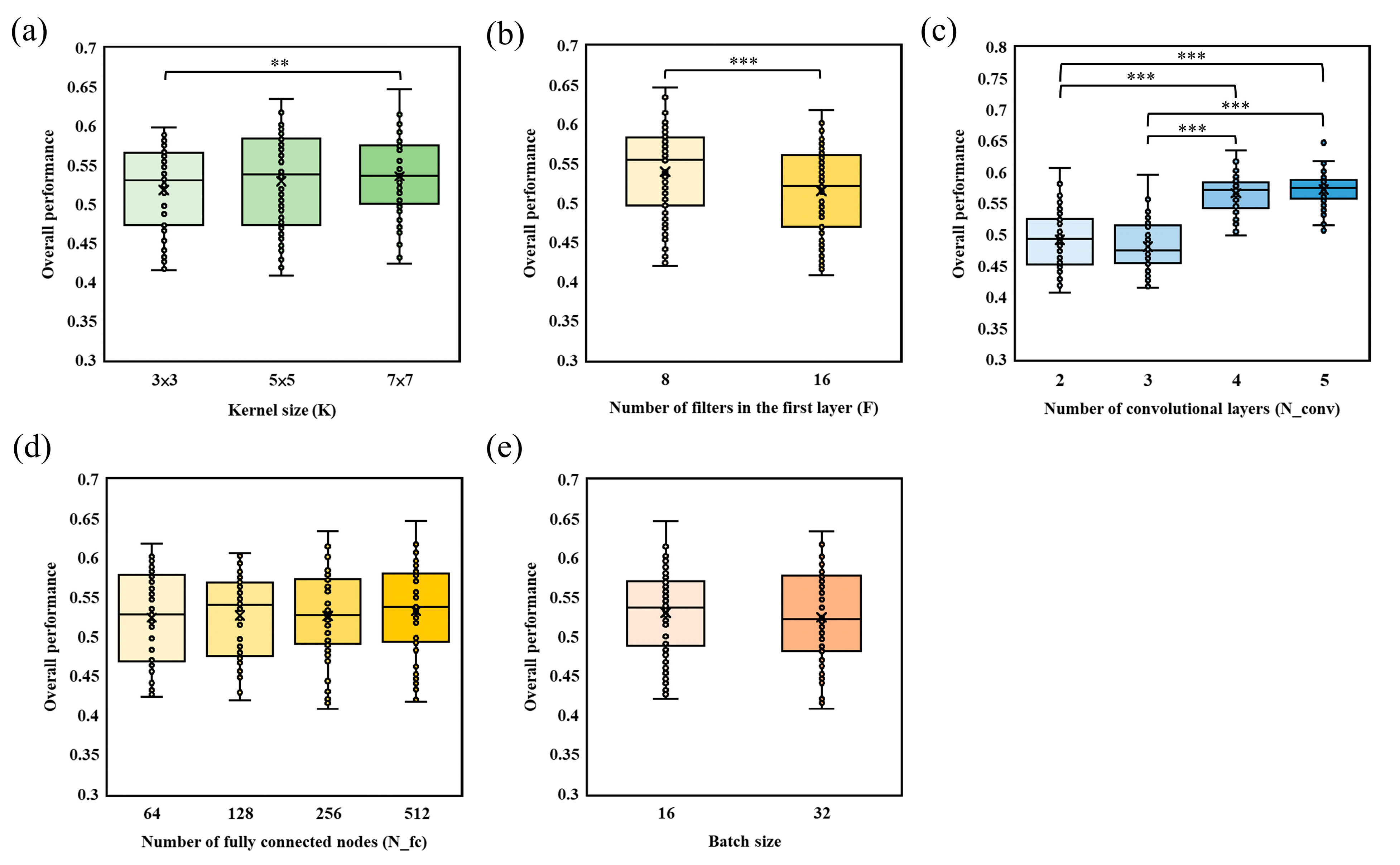
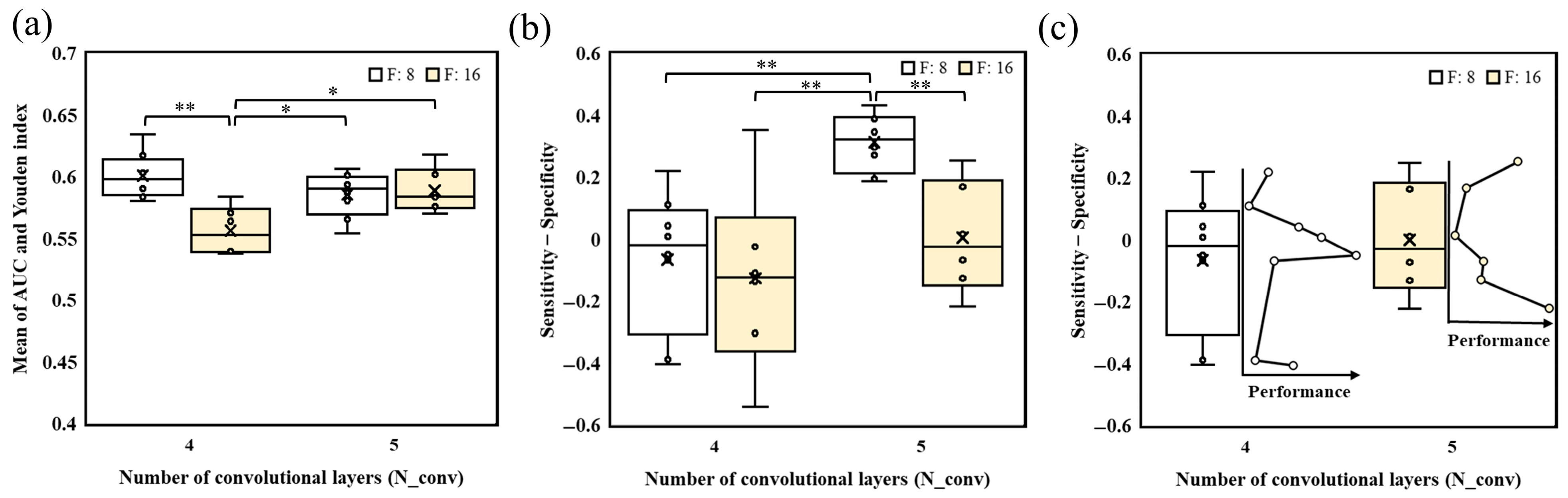
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| EVT | Endovascular thrombectomy |
| AIS | Acute ischemic stroke |
| CTA | Computed tomography angiography |
| CNN | Convolutional neural networks |
| NIHSS | National Institutes of Health Stroke Scale |
| mRS | modified Rankin Scale |
| PACS | Picture Archiving and Communication System |
| IQR | Interquartile range |
| ROC | Receiver operating characteristic |
| AUC | Area under the ROC curve |
| K | Kernel sizes |
| F | Number of filters in the first layer |
| N_conv | Number of convolutional layers |
| N_fc | Number of fully connected nodes |
References
- Goyal, M.; Menon, B.K.; Van Zwam, W.H.; Dippel, D.W.; Mitchell, P.J.; Demchuk, A.M.; Dávalos, A.; Majoie, C.B.; van Der Lugt, A.; De Miquel, M.A. Endovascular thrombectomy after large-vessel ischaemic stroke: A meta-analysis of individual patient data from five randomised trials. Lancet 2016, 387, 1723–1731. [Google Scholar] [CrossRef] [PubMed]
- Berkhemer, O.A.; Fransen, P.S.; Beumer, D.; Van Den Berg, L.A.; Lingsma, H.F.; Yoo, A.J.; Schonewille, W.J.; Vos, J.A.; Nederkoorn, P.J.; Wermer, M.J. A randomized trial of intraarterial treatment for acute ischemic stroke. N. Engl. J. Med. 2015, 372, 11–20. [Google Scholar] [CrossRef]
- Goyal, M.; Demchuk, A.M.; Menon, B.K.; Eesa, M.; Rempel, J.L.; Thornton, J.; Roy, D.; Jovin, T.G.; Willinsky, R.A.; Sapkota, B.L. Randomized assessment of rapid endovascular treatment of ischemic stroke. N. Engl. J. Med. 2015, 372, 1019–1030. [Google Scholar] [CrossRef] [PubMed]
- Campbell, B.C.; Mitchell, P.J.; Kleinig, T.J.; Dewey, H.M.; Churilov, L.; Yassi, N.; Yan, B.; Dowling, R.J.; Parsons, M.W.; Oxley, T.J. Endovascular therapy for ischemic stroke with perfusion-imaging selection. N. Engl. J. Med. 2015, 372, 1009–1018. [Google Scholar] [CrossRef] [PubMed]
- Saver, J.L.; Goyal, M.; Bonafe, A.; Diener, H.-C.; Levy, E.I.; Pereira, V.M.; Albers, G.W.; Cognard, C.; Cohen, D.J.; Hacke, W. Stent-retriever thrombectomy after intravenous t-PA vs. t-PA alone in stroke. N. Engl. J. Med. 2015, 372, 2285–2295. [Google Scholar] [CrossRef]
- Jovin, T.G.; Chamorro, A.; Cobo, E.; de Miquel, M.A.; Molina, C.A.; Rovira, A.; San Román, L.; Serena, J.; Abilleira, S.; Ribó, M. Thrombectomy within 8 hours after symptom onset in ischemic stroke. N. Engl. J. Med. 2015, 372, 2296–2306. [Google Scholar] [CrossRef]
- Bang, O.Y.; Goyal, M.; Liebeskind, D.S. Collateral circulation in ischemic stroke: Assessment tools and therapeutic strategies. Stroke 2015, 46, 3302–3309. [Google Scholar] [CrossRef]
- Berkhemer, O.A.; Jansen, I.G.; Beumer, D.; Fransen, P.S.; Van Den Berg, L.A.; Yoo, A.J.; Lingsma, H.F.; Sprengers, M.E.; Jenniskens, S.F.; Lycklama à Nijeholt, G.J. Collateral status on baseline computed tomographic angiography and intra-arterial treatment effect in patients with proximal anterior circulation stroke. Stroke 2016, 47, 768–776. [Google Scholar] [CrossRef]
- Sinha, A.; Stanwell, P.; Beran, R.G.; Calic, Z.; Killingsworth, M.C.; Bhaskar, S.M. Stroke aetiology and collateral Status in acute ischemic stroke patients receiving reperfusion therapy—A meta-analysis. Neurol. Int. 2021, 13, 608–621. [Google Scholar] [CrossRef]
- de Havenon, A.; Haynor, D.R.; Tirschwell, D.L.; Majersik, J.J.; Smith, G.; Cohen, W.; Andre, J.B. Association of collateral blood vessels detected by arterial spin labeling magnetic resonance imaging with neurological outcome after ischemic stroke. JAMA Neurol. 2017, 74, 453–458. [Google Scholar] [CrossRef]
- Sheth, K.N.; Terry, J.B.; Nogueira, R.G.; Horev, A.; Nguyen, T.N.; Fong, A.K.; Gandhi, D.; Prabhakaran, S.; Wisco, D.; Glenn, B.A. Advanced modality imaging evaluation in acute ischemic stroke may lead to delayed endovascular reperfusion therapy without improvement in clinical outcomes. J. Neurointerv. Surg. 2013, 5, i62–i65. [Google Scholar] [CrossRef] [PubMed]
- McVerry, F.; Liebeskind, D.; Muir, K. Systematic review of methods for assessing leptomeningeal collateral flow. Am. J. Neuroradiol. 2012, 33, 576–582. [Google Scholar] [CrossRef] [PubMed]
- Maas, M.B.; Lev, M.H.; Ay, H.; Singhal, A.B.; Greer, D.M.; Smith, W.S.; Harris, G.J.; Halpern, E.; Kemmling, A.; Koroshetz, W.J. Collateral vessels on CT angiography predict outcome in acute ischemic stroke. Stroke 2009, 40, 3001–3005. [Google Scholar] [CrossRef] [PubMed]
- Miteff, F.; Levi, C.R.; Bateman, G.A.; Spratt, N.; McElduff, P.; Parsons, M.W. The independent predictive utility of computed tomography angiographic collateral status in acute ischaemic stroke. Brain 2009, 132, 2231–2238. [Google Scholar] [CrossRef]
- Tan, I.; Demchuk, A.; Hopyan, J.; Zhang, L.; Gladstone, D.; Wong, K.; Martin, M.; Symons, S.; Fox, A.; Aviv, R. CT angiography clot burden score and collateral score: Correlation with clinical and radiologic outcomes in acute middle cerebral artery infarct. Am. J. Neuroradiol. 2009, 30, 525–531. [Google Scholar] [CrossRef]
- Lu, Q.; Zhang, H.; Cao, X.; Fu, J.; Pan, Y.; Zheng, X.; Wang, J.; Geng, D.; Zhang, J. Quantitative collateral score for the prediction of clinical outcomes in stroke patients: Better than visual grading. Front. Neurosci. 2022, 16, 980135. [Google Scholar] [CrossRef]
- Boers, A.; Barros, R.S.; Jansen, I.; Berkhemer, O.; Beenen, L.; Menon, B.K.; Dippel, D.; van der Lugt, A.; van Zwam, W.; Roos, Y. Value of quantitative collateral scoring on CT angiography in patients with acute ischemic stroke. Am. J. Neuroradiol. 2018, 39, 1074–1082. [Google Scholar] [CrossRef]
- Boers, A.; Berkhemer, O.; Slump, C.; Van Zwam, W.; Roos, Y.; van der Lugt, A.; van Oostenbrugge, R.; Yoo, A.J.; Dippel, D.; Marquering, H. Topographic distribution of cerebral infarct probability in patients with acute ischemic stroke: Mapping of intra-arterial treatment effect. J. Neurointerv. Surg. 2017, 9, 431–436. [Google Scholar] [CrossRef]
- Uniken Venema, S.M.; Wolff, L.; van den Berg, S.A.; Reinink, H.; Luijten, S.P.; Lingsma, H.F.; Marquering, H.A.; Boers, A.M.; Bot, J.; Hammer, S. Time since stroke onset, quantitative collateral score, and functional outcome after endovascular treatment for acute ischemic stroke. Neurology 2022, 99, e1609–e1618. [Google Scholar] [CrossRef]
- Wolff, L.; Uniken Venema, S.M.; Luijten, S.P.; Hofmeijer, J.; Martens, J.M.; Bernsen, M.L.E.; van Es, A.C.; van Doormaal, P.J.; Dippel, D.W.; van Zwam, W. Diagnostic performance of an algorithm for automated collateral scoring on computed tomography angiography. Eur. Radiol. 2022, 32, 5711–5718. [Google Scholar] [CrossRef]
- Grunwald, I.Q.; Kulikovski, J.; Reith, W.; Gerry, S.; Namias, R.; Politi, M.; Papanagiotou, P.; Essig, M.; Mathur, S.; Joly, O. Collateral automation for triage in stroke: Evaluating automated scoring of collaterals in acute stroke on computed tomography scans. Cerebrovasc. Dis. 2019, 47, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Dou, Q.; Chen, H.; Yu, L.; Qin, J.; Heng, P.-A. Multilevel contextual 3-D CNNs for false positive reduction in pulmonary nodule detection. IEEE Trans. Biomed. Eng. 2016, 64, 1558–1567. [Google Scholar] [CrossRef] [PubMed]
- Monkam, P.; Qi, S.; Xu, M.; Han, F.; Zhao, X.; Qian, W. CNN models discriminating between pulmonary micro-nodules and non-nodules from CT images. Biomed. Eng. Online 2018, 17, 96. [Google Scholar] [CrossRef] [PubMed]
- Hafeez, H.A.; Elmagzoub, M.A.; Abdullah, N.A.B.; Al Reshan, M.S.; Gilanie, G.; Alyami, S.; Hassan, M.U.; Shaikh, A. A CNN-model to classify low-grade and high-grade glioma from mri images. IEEE Access 2023, 11, 46283–46296. [Google Scholar] [CrossRef]
- Ali, N.H.; Abdullah, A.R.; Saad, N.M.; Muda, A.S. Collateral circulation classification based on cone beam computed tomography images using ResNet18 convolutional neural network. Int. J. Adv. Comput. Sci. Appl. 2023, 14. [Google Scholar] [CrossRef]
- Su, J.; Wolff, L.; van Es, A.C.M.; Van Zwam, W.; Majoie, C.; Dippel, D.W.; Van Der Lugt, A.; Niessen, W.J.; Van Walsum, T. Automatic collateral scoring from 3D CTA images. IEEE Trans. Med. Imaging 2020, 39, 2190–2200. [Google Scholar] [CrossRef]
- Fortunati, V.; Su, J.; Wolff, L.; van Doormaal, P.-J.; Hofmeijer, J.; Martens, J.; Bokkers, R.P.; van Zwam, W.H.; van der Lugt, A.; van Walsum, T. Siamese model for collateral score prediction from computed tomography angiography images in acute ischemic stroke. Front. Neuroimaging 2024, 2, 1239703. [Google Scholar] [CrossRef]
- Kuang, H.; Wan, W.; Wang, Y.; Wang, J.; Qiu, W. Automated collateral scoring on CT angiography of patients with acute ischemic stroke using hybrid CNN and transformer network. Biomedicines 2023, 11, 243. [Google Scholar] [CrossRef]
- Sahoo, A.; Abdalkader, M.; Yamagami, H.; Huo, X.; Sun, D.; Jia, B.; Weyland, C.S.; Diana, F.; Kaliaev, A.; Klein, P. Endovascular therapy for acute stroke: New evidence and indications. J. Neuroendovasc. Ther. 2023, 17, 232–242. [Google Scholar] [CrossRef]
- Menon, B.K.; d’Esterre, C.D.; Qazi, E.M.; Almekhlafi, M.; Hahn, L.; Demchuk, A.M.; Goyal, M. Multiphase CT angiography: A new tool for the imaging triage of patients with acute ischemic stroke. Radiology 2015, 275, 510–520. [Google Scholar] [CrossRef]
- Chang, C.-H.; Ku, C.-M.; Ger, T.-R.; Lin, W.-P. Fractal-Based Quantitative Collateral Assessment for Thrombectomy Candidate Selection in Acute Ischemic Stroke: A Preliminary Study. Diagnostics 2025, 15, 1590. [Google Scholar] [CrossRef]
- Won, Y.-D.; Kim, J.-M.; Cheong, J.-H.; Ryu, J.-I.; Koh, S.-H.; Han, M.-H. Effect of possible osteoporosis on parenchymal-type hemorrhagic transformation in patients with cardioembolic stroke. J. Clin. Med. 2021, 10, 2526. [Google Scholar] [CrossRef]
- Zack, G.W.; Rogers, W.E.; Latt, S.A. Automatic measurement of sister chromatid exchange frequency. J. Histochem. Cytochem. 1977, 25, 741–753. [Google Scholar] [CrossRef]
- Huang, C.-C.; Chiang, H.-F.; Hsieh, C.-C.; Chou, C.-L.; Jhou, Z.-Y.; Hou, T.-Y.; Shaw, J.-S. Using deep-learning-based artificial intelligence technique to automatically evaluate the collateral status of multiphase cta in acute ischemic stroke. Tomography 2023, 9, 647–656. [Google Scholar] [CrossRef]
- Bang, O.Y.; Saver, J.L.; Buck, B.H.; Alger, J.R.; Starkman, S.; Ovbiagele, B.; Kim, D.; Jahan, R.; Duckwiler, G.R.; Yoon, S.R. Impact of collateral flow on tissue fate in acute ischaemic stroke. J. Neurol. Neurosurg. Psychiatry 2008, 79, 625–629. [Google Scholar] [CrossRef]




| Model (K, F, N_conv) | Overall Performance | AUC | Optimal Youden Index | Sensitivity | Specificity | Accuracy |
|---|---|---|---|---|---|---|
| Sensitivity-oriented model (K = 7 × 7, F = 8, N_conv = 5) | 0.647 | 0.773 | 0.522 | 87.18% | 65.00% | 81.53% |
| Balanced model (K = 5 × 5, F = 8, N_conv = 4) | 0.635 | 0.768 | 0.501 | 72.65% | 77.50% | 73.89% |
| Specificity-oriented model (K = 5 × 5, F = 16, N_conv=5) | 0.618 | 0.753 | 0.482 | 63.25% | 85.00% | 68.79% |
| H. Kuang et al. [12] | 0.649 | 0.766 | 0.532 | 92.56% | 60.61% | 85.71% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Ku, C.-M.; Ger, T.-R. Automated Collateral Classification on CT Angiography in Acute Ischemic Stroke: Performance Trends Across Hyperparameter Combinations. Bioengineering 2026, 13, 124. https://doi.org/10.3390/bioengineering13010124
Ku C-M, Ger T-R. Automated Collateral Classification on CT Angiography in Acute Ischemic Stroke: Performance Trends Across Hyperparameter Combinations. Bioengineering. 2026; 13(1):124. https://doi.org/10.3390/bioengineering13010124
Chicago/Turabian StyleKu, Chi-Ming, and Tzong-Rong Ger. 2026. "Automated Collateral Classification on CT Angiography in Acute Ischemic Stroke: Performance Trends Across Hyperparameter Combinations" Bioengineering 13, no. 1: 124. https://doi.org/10.3390/bioengineering13010124
APA StyleKu, C.-M., & Ger, T.-R. (2026). Automated Collateral Classification on CT Angiography in Acute Ischemic Stroke: Performance Trends Across Hyperparameter Combinations. Bioengineering, 13(1), 124. https://doi.org/10.3390/bioengineering13010124







