Diagnostic Imaging of the Skeletal System: Overview of Applications in Human and Veterinary Medicine
Abstract
1. Introduction
2. Search and Selection Strategy
3. Diagnostic Imaging Modalities
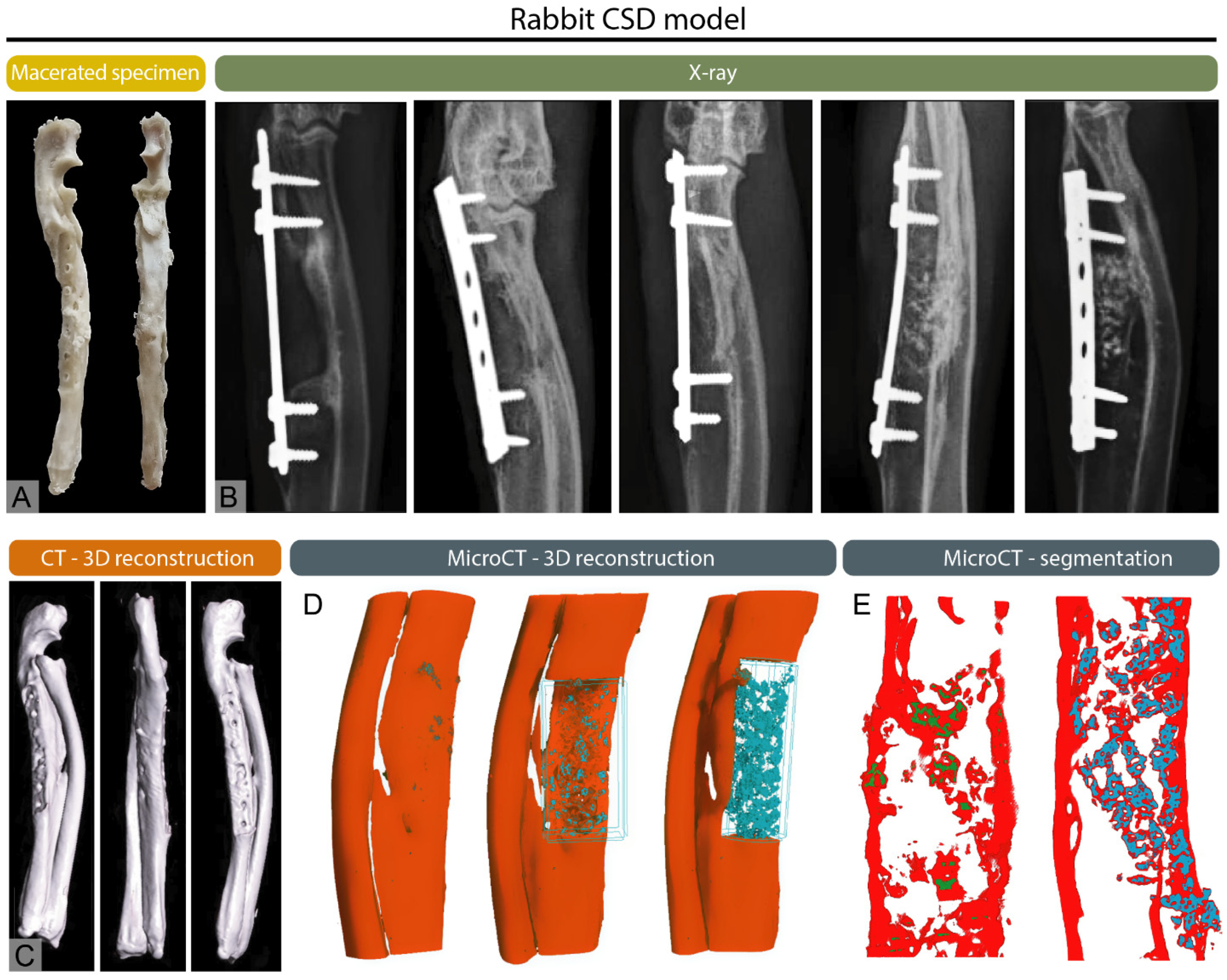
3.1. Conventional Radiography
3.2. Dual-Energy X-Ray Absorptiometry
3.3. Computed Tomography
3.4. Quantitative Computed Tomography
3.5. Micro-Computed Tomography
3.6. Nano-Computed Tomography
3.7. Positron Emission Tomography-Computed Tomography
3.8. Magnetic Resonance Imaging
3.9. Quantitative Magnetic Resonance Imaging
3.10. Ultrasound
3.11. Quantitative Ultrasound
| Imaging Modality | Indications | Contraindications | Advantages | Disadvantages | References |
|---|---|---|---|---|---|
| X-ray imaging | Fracture, joint dislocation, bone deformities, degenerative joint/spine disease, osteoporosis screening, tumors, pre/postoperative monitoring | Pregnancy, contrast allergy, severe obesity | High spatial resolution for bone, fast, accessible, portable | Limited soft tissue contrast, 2D only, ionizing radiation | [1,12,15,16,26,27,28,29,31,32,33,36,37,39,64] |
| Dual-energy X-ray absorptiometry (DXA) | Osteoporosis diagnosis, fracture risk, BMD monitoring, lean mass/fat distribution | Pregnancy, severe obesity, recent contrast, inability to remain still | High precision BMD, low radiation, non-invasive standard | Limited microarchitecture info, artifacts affect accuracy | [1,40,41,44,45,46,47,48,49,50,52,54,55,56,78] |
| Computed Tomography (CT) | Complex fractures, degenerative diseases, tumor and infection assessment, 3D surgical planning | Pregnancy, contrast allergy, severe obesity | Three-dimensional bone imaging, fast acquisition, detailed fracture visualization | Higher radiation dose, limited soft tissue contrast | [1,62,63,64,67,68,69,70,72,73,74,85,117] |
| Quantitative Computed Tomography (QCT) | Volumetric BMD, osteoporosis progression, patients unsuitable for DXA, opportunistic screening | Pregnancy, contrast allergy, severe obesity | Volumetric BMD, distinguishes cortical/trabecular bone, sensitive | Higher radiation, cost, calibration required, limited availability | [1,75,77,78,79] |
| Micro-CT | Preclinical bone microstructure analysis, research | None reported | High-resolution 3D bone microarchitecture, quantitative analysis | Limited to small samples, high radiation, costly | [1,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94,98] |
| Nano-CT | Bone ultrastructure research, biomaterials, tissue engineering | None reported | Nanoscale resolution, quantifies nano-architecture | Small sample size, expensive, technical complexity | [95,96,97,98,99,101] |
| Positron emission tomography-computed tomography (PET-CT) | Cancer detection/staging, neuro/cardiac/inflammatory disease assessment | Pregnancy, radiotracer allergy, metal implants, severe obesity | Detects metabolic changes early, whole-body imaging, quantitative uptake | High cost, ionizing radiation, limited spatial resolution, limited specificity | [102,103,104,105,106,107,108,109,110,111,112,113,114,156] |
| Magnetic resonance imaging (MRI) | Soft tissue injury, bone marrow edema, tumor staging, congenital/inflammatory disorders | Metallic implants, obesity, allergy, pregnancy, claustrophobia | Superior soft tissue contrast, multiplanar imaging, no radiation | High cost, sensitive to motion, limited cortical bone imaging | [115,116,117,118,119,120] |
| Quantitative MRI (QMRI) | Cartilage and trabecular bone quantification, marrow composition, early OA/osteoporosis detection | Same as MRI | Objective tissue quantification, serial monitoring, no radiation | Technical complexity, long scan/analysis time, variable reproducibility | [128,129,130,131,135] |
| Ultrasound (US) | Fractures, periosteal reactions, synovitis, soft tissue, procedural guidance | None major | Real-time, portable, high resolution superficial imaging | Limited penetration, operator dependent, limited bone imaging | [136,137,138,139,140] |
| Quantitative ultrasound (QUS) | Osteoporosis screening, bone quality, peripheral fracture risk | Acute injury/amputation site, obesity, recent surgery | Radiation-free, portable, low cost, good for mass screening | Limited to peripheral sites, less accurate than DXA/QCT | [1,46,54,142,143,144,145,146,147,148,149,150,151,152,153] |
4. Key Differences and Selection of Imaging Methods in Human and Veterinary Medicine
5. Artificial Intelligence in Diagnostic Imaging
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| DXA | Dual-energy X-ray absorptiometry |
| CT | Computed tomography |
| MRI | Magnetic resonance imaging |
| QUS | Quantitative ultrasound |
| PET-CT | Positron emission tomography-computed tomography |
| QCT | Quantitative computed tomography |
| micro-CT | Micro-computed tomography |
| nano-CT | Nano-computed tomography |
| BMD | Bone mineral density |
| MDCT | Multidetector computed tomography |
| ALARA | As Low As Reasonably Achievable |
| FDG | Fluorodeoxyglucose |
| [18F]-FDG | Fluorine-18 fluorodeoxyglucose |
| [18F] | sodium fluoride (NaF) |
| NMR | Nuclear magnetic resonance |
| WORMS | Whole-organ magnetic resonance imaging score |
| SPARCC | Spondyloarthritis Research Consortium of Canada |
| RAMRIS | Rheumatoid Arthritis Magnetic Resonance Imaging Score |
| fMRI | functional MRI |
| QMRI | Quantitative magnetic resonance imaging |
| US | Ultrasound |
| SOS | Speed of sound |
| BUA | Broadband ultrasound attenuation |
| CSD | Critical segmental defect |
| PLF | Postero-lateral fusion |
| 3R | replacement, reduction, refinement |
| AI | artificial intelligence |
References
- Choplin, R.H.; Allen, M.R. Skeletal Imaging. In Basic and Applied Bone Biology; Academic Press: Cambridge, MA, USA, 2019; pp. 103–123. [Google Scholar] [CrossRef]
- Hart, N.H.; Newton, R.U.; Tan, J.; Rantalainen, T.; Chivers, P.; Siafarikas, A.; Nimphius, S. Biological Basis of Bone Strength: Anatomy, Physiology and Measurement. J. Musculoskelet. Neuronal Interact. 2020, 20, 347. [Google Scholar]
- Piccolo, C.L.; Mallio, C.A.; Vaccarino, F.; Grasso, R.F.; Zobel, B.B. Imaging of Knee Osteoarthritis: A Review of Multi-Modal Diagnostic Approach. Quant. Imaging Med. Surg. 2023, 13, 7582. [Google Scholar] [CrossRef]
- Bury, D.C.; Rogers, T.S.; Dickman, M.M. Osteomyelitis: Diagnosis and Treatment. Am. Fam. Physician 2021, 104, 395–402. [Google Scholar]
- Mehta, K.; McBee, M.P.; Mihal, D.C.; England, E.B. Radiographic Analysis of Bone Tumors: A Systematic Approach. Semin. Roentgenol. 2017, 52, 194–208. [Google Scholar] [CrossRef]
- Lange, M.B.; Nielsen, M.L.; Andersen, J.D.; Lilholt, H.J.; Vyberg, M.; Petersen, L.J. Diagnostic accuracy of imaging methods for the diagnosis of skeletal malignancies: A retrospective analysis against a pathology-proven reference. Eur. J. Radiol. 2016, 85, 61–67. [Google Scholar] [CrossRef]
- Cunha, L.; Horvath, I.; Ferreira, S.; Lemos, J.; Costa, P.; Vieira, D.; Veres, D.S.; Szigeti, K.; Summavielle, T.; Máthé, D.; et al. Preclinical Imaging: An Essential Ally in Modern Biosciences. Mol. Diagn. Ther. 2014, 18, 153–173. [Google Scholar] [CrossRef]
- Finnerty, N.M.; Rodriguez, R.M.; Carpenter, C.R.; Sun, B.C.; Theyyunni, N.; Ohle, R.; Dodd, K.W.; Schoenfeld, E.M.; Elm, K.D.; Kline, J.A.; et al. Clinical Decision Rules for Diagnostic Imaging in the Emergency Department: A Research Agenda. Acad. Emerg. Med. 2015, 22, 1406–1416. [Google Scholar] [CrossRef] [PubMed]
- Bonakdarpour, A.; Reinus, W.R.; Khurana, J.S. Diagnostic Imaging of Musculoskeletal Diseases: A Systematic Approach; Springer: Berlin, Germany, 2010. [Google Scholar] [CrossRef]
- Roentgen, W.C. On a New Kind of Rays. Ann. Phys. Chem. 1898, 64, 1–11. [Google Scholar]
- Assmus, A. Early History of X Rays. Beam Line 1995, 25, 10–24. [Google Scholar]
- Shen, R.; Li, Z.; Zhang, L.; Hua, Y.; Mao, M.; Li, Z.; Cai, Z.; Qiu, Y.; Gryak, J.; Najarian, K. Osteosarcoma Patients Classification Using Plain X-Rays and Metabolomic Data. In Proceedings of the 2018 40th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Honolulu, HI, USA, 17–21 July 2018; pp. 690–693. [Google Scholar] [CrossRef]
- Yilmaz, O.; Koca, D.; Sahin, M.E.; Avcilar, T. Sex Determination in Budgerigars Using Radiographic Pelvimetry. Vet. Med. Sci. 2024, 10, e1340. [Google Scholar] [CrossRef] [PubMed]
- Vetere, A.; Capasso, M.; Di Ianni, F. Sex Determination in Reptiles: A Review. Animals 2025, 15, 168. [Google Scholar] [CrossRef]
- Lim, S.J.; Park, Y.S. Plain Radiography of the Hip: A Review of Radiographic Techniques and Image Features. Hip Pelvis 2015, 27, 125. [Google Scholar] [CrossRef] [PubMed]
- Ou, X.; Chen, X.; Xu, X.; Xie, L.; Chen, X.; Hong, Z.; Bai, H.; Liu, X.; Chen, Q.; Li, L.; et al. Recent Development in X-Ray Imaging Technology: Future and Challenges. Research 2021, 2021, 9892152. [Google Scholar] [CrossRef] [PubMed]
- Stokovic, N.; Ivanjko, N.; Javor, A.; Pecin, M.; Muzina, K.; Stepanic, Z.M.; Capak, H.; Vrbanac, Z.; Maticic, D.; Vukicevic, S. Regeneration of a Rabbit Segmental Defect with a New Bone Therapy: Autologous Blood Coagulum with Bone Morphogenetic Protein 6 and Synthetic Ceramics. Biomater. Res. 2025, 29, 12. [Google Scholar] [CrossRef]
- Ivanjko, N.; Stokovic, N.; Pecin, M.; Vnuk, D.; Smajlovic, A.; Ivkic, N.; Capak, H.; Javor, A.; Vrbanac, Z.; Maticic, D.; et al. Calcium Phosphate Ceramics Combined with rhBMP6 within Autologous Blood Coagulum Promote Posterolateral Lumbar Fusion in Sheep. Sci. Rep. 2023, 13, 22079. [Google Scholar] [CrossRef] [PubMed]
- Stokovic, N.; Ivanjko, N.; Pecin, M.; Erjavec, I.; Smajlović, A.; Milesevic, M.; Karlović, S.; Capak, H.; Vrbanac, Z.; Matičić, D.; et al. Long-Term Posterolateral Spinal Fusion in Rabbits Induced by rhBMP6 Applied in Autologous Blood Coagulum with Synthetic Ceramics. Sci. Rep. 2022, 12, 11649. [Google Scholar] [CrossRef]
- Whiley, S.P. Evaluating Fracture Healing Using Digital X-ray Image Analysis: Fracture Healing Is Not Easily Monitored Using Currently Available Techniques. Cont. Med. Educ. 2011, 29, 122–124. [Google Scholar]
- Slongo, T.; Audigé, L.; Clavert, J.M.; Lutz, N.; Frick, S.; Hunter, J. The AO Comprehensive Classification of Pediatric Long-Bone Fractures: A Web-Based Multicenter Agreement Study. J. Pediatr. Orthop. 2007, 27, 171–180. [Google Scholar] [CrossRef]
- Salter, R.B.; Harris, W.R. Injuries Involving the Epiphyseal Plate. J. Bone Jt. Surg. Am. 1963, 45, 587–622. [Google Scholar] [CrossRef]
- Julian, T.H.; Broadbent, R.H.; Ward, A.E. Surgical vs. Non-Surgical Management of Weber B Fractures: A Systematic Review. Foot Ankle Surg. 2020, 26, 494–502. [Google Scholar] [CrossRef]
- Zeltser, D.W.; Leopold, S.S. Classifications in Brief: Schatzker Classification of Tibial Plateau Fractures. Clin. Orthop. Relat. Res. 2013, 471, 371–374. [Google Scholar] [CrossRef] [PubMed]
- Pinna, S.; Vezzoni, A.; Di Benedetto, M.; Lambertini, C.; Tassani, C. Characterization of FCI (Fédération Cynologique Internationale) Grades for Hip Dysplasia in Five Dog Breeds. Animals 2023, 13, 2212. [Google Scholar] [CrossRef] [PubMed]
- Offiah, A.C.; Hall, C.M. The Radiologic Diagnosis of Skeletal Dysplasias: Past, Present and Future. Pediatr. Radiol. 2020, 50, 1650–1657. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, D.; Roemer, F.W.; Jarraya, M.; Guermazi, A. Imaging of Osteoarthritis. J. Clin. Med. 2025, 14, 1122. [Google Scholar]
- Endara-Mina, J.; Kumar, H.; Ghosh, B.; Mehta, A.; Dey, R.C.; Singh, P.; Rai, N.; Mandadi, M.; Opara, O.; Quinonez, J. Comparative use of ultrasound and radiography for the detection of fractures: A systematic review and narrative synthesis. Ann. Med. Surg. 2023, 85, 5085–5095. [Google Scholar] [CrossRef]
- Moza, B.; Mukherjee, D.; Singh, M.; Pahwa, V.; Ujjainia, P.; Pathak, S.; Saha, A.; Srivastava, A. Advancements in the Imaging Techniques for Detection of Skeletal Pathologies: A Comprehensive Review. Tuijin Jishu/J. Propuls. Technol. 2024, 45, 100–110. [Google Scholar]
- Johansson, H.; Azizieh, F.; Al Ali, N.; Alessa, T.; Harvey, N.C.; McCloskey, E.; Kanis, J.A. FRAX-vs. T-Score-Based Intervention Thresholds for Osteoporosis. Osteoporos. Int. 2017, 28, 3099–3105. [Google Scholar] [CrossRef]
- Puthoor, D.; Puthoori, D. Analysis of X-ray Patterns in Infection and Tumor. J. Bone Soft Tissue Tumors 2021, 7, 5–8. [Google Scholar] [CrossRef]
- Gerber, E.; Said-Hartley, Q.; Gamieldien, R.; Hartley, T.; Candy, S. Accuracy of Plain Radiographs in Diagnosing Biopsy-Proven Malignant Bone Lesions. S. Afr. J. Radiol. 2019, 23, 1–7. [Google Scholar] [CrossRef]
- Peng, H.M.; Feng, B.; Chen, X.; Wang, Y.O.; Bian, Y.Y.; Wang, W.; Weng, X.S.; Qian, W.W. Usefulness of a Simple Preoperative Planning Technique Using Plain X-Rays for Direct Anterior Approach for Total Hip Arthroplasty. Orthop. Surg. 2021, 13, 145–152. [Google Scholar] [CrossRef]
- Gómez-Barrena, E.; Padilla-Eguiluz, N.G.; García-Rey, E.; Hernández-Esteban, P.; Cordero-Ampuero, J.; Rubio-Suárez, J.C. Validation of a Long Bone Fracture Non-Union Healing Score After Treatment with Mesenchymal Stromal Cells Combined to Biomaterials. Injury 2020, 51, S55–S62. [Google Scholar] [CrossRef]
- Cook, S.D.; Baffes, G.C.; Wolfe, M.W.; Sampath, T.K.; Rueger, D.C. Recombinant Human Bone Morphogenetic Protein-7 Induces Healing in a Canine Long-Bone Segmental Defect Model. Clin. Orthop. Relat. Res. 1994, 301, 302–312. [Google Scholar] [CrossRef]
- Parks, E.T.; Williamson, G.F. Digital Radiography: An Overview. J. Contemp. Dent. Pract. 2002, 3, 23–39. [Google Scholar] [CrossRef]
- Wang, X.; Oo, W.M.; Linklater, J.M. What Is the Role of Imaging in the Clinical Diagnosis of Osteoarthritis and Disease Management? Rheumatology 2018, 57, iv51–iv60. [Google Scholar] [CrossRef] [PubMed]
- Schreiner, M.; Frühmann, B.; Jembrih-Simbürger, D.; Linke, R. X-rays in Art and Archaeology: An Overview. Powder Diffr. 2004, 19, 3–11. [Google Scholar] [CrossRef]
- Yeung, A.W.K. The “As Low as Reasonably Achievable” (ALARA) Principle: A Brief Historical Overview and a Bibliometric Analysis of the Most Cited Publications. Radioprotection 2019, 54, 103–109. [Google Scholar] [CrossRef]
- Shevroja, E.; Cafarelli, F.P.; Guglielmi, G.; Hans, D. DXA Parameters, Trabecular Bone Score (TBS) and Bone Mineral Density (BMD), in Fracture Risk Prediction in Endocrine-Mediated Secondary Osteoporosis. Endocrine 2021, 74, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Lewiecki, E.M.; Binkley, N. DXA: 30 Years and Counting: Introduction to the 30th Anniversary Issue. Bone 2017, 104, 1–3. [Google Scholar] [CrossRef]
- Messina, C.; Albano, D.; Gitto, S.; Tofanelli, L.; Bazzocchi, A.; Ulivieri, F.M.; Guglielmi, G.; Sconfienza, L.M. Body Composition with Dual Energy X-ray Absorptiometry: From Basics to New Tools. Quant. Imaging Med. Surg. 2020, 10, 1687–1698. [Google Scholar] [CrossRef]
- Bazzocchi, A.; Ponti, F.; Albisinni, U.; Battista, G.; Guglielmi, G. DXA: Technical Aspects and Application. Eur. J. Radiol. 2016, 85, 1481–1492. [Google Scholar] [CrossRef]
- Sheu, A.; Diamond, T. Bone Mineral Density: Testing for Osteoporosis. Aust. Prescr. 2016, 39, 35–39. [Google Scholar] [CrossRef] [PubMed]
- Jelić, Đ.; Stefanović, D.; Petronijević, M.; Anđelić-Jelić, M. Why Dual X-ray Absorptiometry Is the Gold Standard in Diagnosing Osteoporosis. Vojnosanit. Pregl. 2008, 65, 919–922. [Google Scholar] [CrossRef]
- Roldan, E.Q.; Brianese, N.; Raffetti, E.; Focà, E.; Pezzoli, M.C.; Bonito, A.; Castelli, F. Comparison between the Gold Standard DXA with Calcaneal Quantitative Ultrasound Based-Strategy (QUS) to Detect Osteoporosis in an HIV Infected Cohort. Braz. J. Infect. Dis. 2017, 21, 581–586. [Google Scholar] [CrossRef]
- Nowell, J.; Murray, R.S.; Oetgen, M.E.; Martin, B.D. Decreasing Radiation Exposure in the Treatment of Pediatric Long Bone Fractures Using a DXA Scan: A Proof of Concept. J. Pediatr. Orthop. Soc. N. Am. 2024, 5, e230007. [Google Scholar] [CrossRef]
- Dimai, H.P. Use of Dual-Energy X-ray Absorptiometry (DXA) for Diagnosis and Fracture Risk Assessment; WHO-Criteria, T- and Z-Score, and Reference Databases. Bone 2017, 104, 39–43. [Google Scholar] [CrossRef]
- Bao, J.; Zou, D.; Li, W. Characteristics of the DXA Measurements in Patients Undergoing Lumbar Fusion for Lumbar Degenerative Diseases: A Retrospective Analysis of Over 1000 Patients. Clin. Interv. Aging 2021, 16, 1131–1137. [Google Scholar] [CrossRef]
- Stults-Kolehmainen, M.A.; Stanforth, P.R.; Bartholomew, J.B.; Lu, T.; Abolt, C.J.; Sinha, R. DXA Estimates of Fat in Abdominal, Trunk and Hip Regions Vary by Ethnicity in Men. Nutr. Diabetes 2013, 3, e64. [Google Scholar] [CrossRef]
- Miyamura, S.; Kuriyama, K.; Ebina, K.; Oka, K.; Kashii, M.; Shigi, A.; Nakashima, Y.; Murase, T. Utility of Distal Forearm DXA as a Screening Tool for Primary Osteoporotic Fragility Fractures of the Distal Radius: A Case-Control Study. JBJS Open Access 2020, 5, e0036. [Google Scholar] [CrossRef]
- You, H.; Shang, J.; Huang, Z.; He, W.; Zeng, C.; Xu, H.; Gong, J. Research on DXA Bone Density Measurements and Trabecular Bone Scores in Chinese Men and Women with Obesity before and after Bariatric Surgery. Sci. Rep. 2024, 14, 29355. [Google Scholar] [CrossRef] [PubMed]
- Hussain, D.; Han, S.M.; Kim, T.S. Automatic Hip Geometric Feature Extraction in DXA Imaging Using Regional Random Forest. J. X-Ray Sci. Technol. 2019, 27, 207–236. [Google Scholar] [CrossRef] [PubMed]
- Pennington, Z.; Ehresman, J.; Lubelski, D.; Cottrill, E.; Schilling, A.; Ahmed, A.K.; Feghali, J.; Witham, T.F.; Sciubba, D.M. Assessing Underlying Bone Quality in Spine Surgery Patients: A Narrative Review of Dual-Energy X-ray Absorptiometry (DXA) and Alternatives. Spine J. 2021, 21, 321–331. [Google Scholar] [CrossRef]
- Gupta, P.; Cherian, K.E.; Kapoor, N.; Paul, T.V. Aortic Calcification Artifact Causing Spuriously High Bone Mineral Density in the Lumbar Spine. AACE Clin. Case Rep. 2020, 7, 284–286. [Google Scholar] [CrossRef]
- Jacobson, J.A.; Jamadar, D.A.; Hayes, C.W. Dual X-ray Absorptiometry: Recognizing Image Artifacts and Pathology. Am. J. Roentgenol. 2000, 174, 1699–1705. [Google Scholar] [CrossRef]
- Hounsfield, G.N. Computerized Transverse Axial Scanning (Tomography): Part 1. Description of System. Br. J. Radiol. 1973, 46, 1016–1022. [Google Scholar] [CrossRef]
- Seeram, E. Nobel Prize for CT and MRI Pioneers: Historical Article. Radiographer 2006, 53, 4–7. [Google Scholar] [CrossRef]
- Platten, D. Multi-Slice Helical CT Physics and Technology; ImPACT: London, UK, 2003. [Google Scholar]
- Goldman, L.W. Principles of CT and CT Technology. J. Nucl. Med. Technol. 2007, 35, 115–128. [Google Scholar] [CrossRef]
- Koppula, B.R.; Morton, K.A.; Al-Dulaimi, R.; Fine, G.C.; Damme, N.M.; Brown, R.K. SPECT/CT in the Evaluation of Suspected Skeletal Pathology. Tomography 2021, 7, 581–605. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, T.; Yonenaga, T.; Miyasaka, T.; Kimura, T.; Jinzaki, M.; Ojiri, H. CT in Osteoarthritis: Its Clinical Role and Recent Advances. Skelet. Radiol. 2023, 52, 2199–2210. [Google Scholar] [CrossRef] [PubMed]
- Notohamiprodjo, S.; Stahl, R.; Braunagel, M.; Kazmierczak, P.M.; Thierfelder, K.M.; Treitl, K.M.; Wirth, S.; Notohamiprodjo, M. Diagnostic Accuracy of Contemporary Multidetector Computed Tomography (MDCT) for the Detection of Lumbar Disc Herniation. Eur. Radiol. 2017, 27, 3443–3451. [Google Scholar] [CrossRef]
- Bureš, T.; Capak, H.; Škrlin, B.; Stanin, D.; Kraljević, A.; Nappo, D.; Vrbanac, Z. Usporedba metoda slikovne dijagnostike hernijacije intervertebralnog diska u pasa. Vet. Stanica 2022, 53, 475–486. [Google Scholar] [CrossRef]
- Sharma, S.; Boston, S.E.; Skinner, O.T.; Perry, J.A.; Verstraete, F.J.M.; Lee, D.B.; Van Stee, L.L.L.; Thompson, C.; Boylan, M.; McKee, T.; et al. Survival Time of Juvenile Dogs with Oral Squamous Cell Carcinoma Treated with Surgery Alone: A Veterinary Society of Surgical Oncology Retrospective Study. Vet. Surg. 2021, 50, 740–747. [Google Scholar] [CrossRef]
- Artiles, C.A.; Guzman, D.S.M.; Beaufrère, H.; Phillips, K.L. Computed Tomographic Findings of Dental Disease in Domestic Rabbits (Oryctolagus cuniculus): 100 Cases (2009–2017). J. Am. Vet. Med. Assoc. 2020, 257, 313–327. [Google Scholar] [CrossRef]
- Mathee, N.; Robert, M.; Higgerty, S.M.; Fosgate, G.T.; Rogers, A.L.; d’Ablon, X.; Carstens, A. Computed Tomographic Evaluation of the Distal Limb in the Standing Sedated Horse: Technique, Imaging Diagnoses, Feasibility, and Artifacts. Vet. Radiol. Ultrasound 2023, 64, 243–252. [Google Scholar] [CrossRef]
- Grüneboom, A.; Kling, L.; Christiansen, S.; Mill, L.; Maier, A.; Engelke, K.; Quick, H.H.; Schett, G.; Gunzer, M. Next-Generation Imaging of the Skeletal System and Its Blood Supply. Nat. Rev. Rheumatol. 2019, 15, 533–549. [Google Scholar] [CrossRef]
- Bellolio, M.F.; Heien, H.C.; Sangaralingham, L.R.; Jeffery, M.M.; Campbell, R.L.; Cabrera, D.; Shah, N.D.; Hess, E.P. Increased Computed Tomography Utilization in the Emergency Department and Its Association with Hospital Admission. West. J. Emerg. Med. 2017, 18, 835–845. [Google Scholar] [CrossRef]
- Dozeman, E.T.; Prittie, J.E.; Fischetti, A.J. Utilization of whole body computed tomography in polytrauma patients. J. Vet. Emerg. Crit. Care 2020, 30, 28–33. [Google Scholar] [CrossRef]
- Kwee, R.M.; Toxopeus, R.; Kwee, T.C. Imaging Overuse in the Emergency Department: The View of Radiologists and Emergency Physicians. Eur. J. Radiol. 2024, 176, 111536. [Google Scholar] [CrossRef]
- Rehani, M.M.; Yang, K.; Melick, E.R.; Heil, J.; Šalát, D.; Sensakovic, W.F.; Liu, B. Patients Undergoing Recurrent CT Scans: Assessing the Magnitude. Eur. Radiol. 2020, 30, 1828–1836. [Google Scholar] [CrossRef] [PubMed]
- Buckwalter, K.A.; Lin, C.; Ford, J.M. Managing Postoperative Artifacts on Computed Tomography and Magnetic Resonance Imaging. Semin. Musculoskelet. Radiol. 2011, 15, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Winsor, C.; Li, X.; Qasim, M.; Henak, C.R.; Pickhardt, P.J.; Ploeg, H.; Viceconti, M. Evaluation of Patient Tissue Selection Methods for Deriving Equivalent Density Calibration for Femoral Bone Quantitative CT Analyses. Bone 2021, 143, 115759. [Google Scholar] [CrossRef] [PubMed]
- Tsurusaki, K.; Ito, M.; Hayashi, K. Differential Effects of Menopause and Metabolic Disease on Trabecular and Cortical Bone Assessed by Peripheral Quantitative Computed Tomography (pQCT). Br. J. Radiol. 2000, 73, 14–22. [Google Scholar] [CrossRef]
- Engelke, K.; Museyko, O.; Wang, L.; Laredo, J.D. Quantitative Analysis of Skeletal Muscle by Computed Tomography Imaging—State of the Art. J. Orthop. Transl. 2018, 15, 91–103. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Li, J.; Jiao, X.; Jia, X.; Zhang, X.; Fan, G.; Guo, J. The Accuracy of Bone Mineral Density Measurement Using Dual-Energy Spectral CT and Quantitative CT: A Comparative Phantom Study. Clin. Radiol. 2020, 75, 320.e9. [Google Scholar] [CrossRef]
- Genant, H.K.; Engelke, K.; Prevrhal, S. Advanced CT Bone Imaging in Osteoporosis. Rheumatology 2008, 47, iv9–iv16. [Google Scholar] [CrossRef]
- Brett, A.D.; Brown, J.K. Quantitative Computed Tomography and Opportunistic Bone Density Screening by Dual Use of Computed Tomography Scans. J. Orthop. Transl. 2015, 3, 178–184. [Google Scholar] [CrossRef]
- Cengiz, I.F.; Oliveira, J.M.; Reis, R.L. Micro-CT—A Digital 3D Microstructural Voyage into Scaffolds: A Systematic Review of the Reported Methods and Results. Biomater. Res. 2018, 22, 26. [Google Scholar] [CrossRef]
- Gersing, A.S.; Kimm, M.A.; Bollwein, C.; Ilg, P.; Mogler, C.; Gassert, F.G.; Pfeiffer, F.; Feuerriegel, G.C.; Knebel, C.; Wörtler, K.; et al. Chondrosarcoma Evaluation Using Hematein-Based X-Ray Staining and High-Resolution 3D Micro-CT: A Feasibility Study. Eur. Radiol. Exp. 2024, 8, 58. [Google Scholar] [CrossRef]
- Wu, Y.; Adeeb, S.; Doschak, M.R. Using Micro-CT Derived Bone Microarchitecture to Analyze Bone Stiffness—A Case Study on Osteoporosis Rat Bone. Front. Endocrinol. 2015, 6, 80. [Google Scholar] [CrossRef] [PubMed]
- Vande Velde, G.; Poelmans, J.; De Langhe, E.; Hillen, A.; Vanoirbeek, J.; Himmelreich, U.; Lories, R.J. Longitudinal Micro-CT Provides Biomarkers of Lung Disease That Can Be Used to Assess the Effect of Therapy in Preclinical Mouse Models, and Reveal Compensatory Changes in Lung Volume. Dis. Model. Mech. 2016, 9, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Grgurevic, L.; Oppermann, H.; Pecin, M.; Erjavec, I.; Capak, H.; Pauk, M.; Vukicevic, S. Recombinant Human Bone Morphogenetic Protein 6 Delivered within Autologous Blood Coagulum Restores Critical Size Segmental Defects of Ulna in Rabbits. JBMR Plus 2019, 3, e10085. [Google Scholar] [CrossRef]
- Kirschner, S.; Mürle, B.; Felix, M.; Arns, A.; Groden, C.; Wenz, F.; Brockmann, M.A. Imaging of Orthotopic Glioblastoma Xenografts in Mice Using a Clinical CT Scanner: Comparison with Micro-CT and Histology. PLoS ONE 2016, 11, e0165994. [Google Scholar] [CrossRef]
- Stokovic, N.; Ivanjko, N.; Milesevic, M.; Matic Jelic, I.; Bakic, K.; Rumenovic, V.; Oppermann, H.; Shimp, L.; Sampath, T.K.; Pecina, M.; et al. Synthetic Ceramic Macroporous Blocks as a Scaffold in Ectopic Bone Formation Induced by Recombinant Human Bone Morphogenetic Protein 6 within Autologous Blood Coagulum in Rats. Int. Orthop. 2021, 45, 1097–1107. [Google Scholar] [CrossRef]
- Stokovic, N.; Ivanjko, N.; Erjavec, I.; Milosevic, M.; Oppermann, H.; Shimp, L.; Sampath, K.T.; Vukicevic, S. Autologous Bone Graft Substitute Containing rhBMP6 within Autologous Blood Coagulum and Synthetic Ceramics of Different Particle Size Determines the Quantity and Structural Pattern of Bone Formed in a Rat Subcutaneous Assay. Bone 2020, 141, 115654. [Google Scholar] [CrossRef] [PubMed]
- Štoković, N.; Ivanjko, N.; Pećin, M.; Erjavec, I.; Karlović, S.; Smajlović, A.; Capak, H.; Milošević, M.; Bubić Špoljar, J.; Vnuk, D.; et al. Evaluation of Synthetic Ceramics as Compression Resistant Matrix to Promote Osteogenesis of Autologous Blood Coagulum Containing Recombinant Human Bone Morphogenetic Protein 6 in Rabbit Posterolateral Lumbar Fusion Model. Bone 2020, 140, 115544. [Google Scholar] [CrossRef] [PubMed]
- Vukicevic, S.; Grgurevic, L.; Erjavec, I.; Bordukalo-Niksic, T.; Stokovic, N.; Pecin, M.; Lipar, M.; Maticic, D.; Capak, H.; Windhager, R.; et al. Autologous Blood Coagulum Is a Physiological Carrier for BMP6 to Induce New Bone Formation and Promote Posterolateral Lumbar Spine Fusion in Rabbits. J. Tissue Eng. Regen. Med. 2020, 14, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Grgurevic, L.; Erjavec, I.; Gupta, M.; Pecin, M.; Bordukalo Niksic, T.; Stokovic, N.; Vnuk, D.; Farkas, V.; Capak, H.; Milosevic, M.; et al. Autologous Blood Coagulum Containing rhBMP6 Induces New Bone Formation to Promote Anterior Lumbar Interbody Fusion (ALIF) and Posterolateral Lumbar Fusion (PLF) of Spine in Sheep. Bone 2020, 138, 115448. [Google Scholar] [CrossRef]
- Ito, M.; Nishida, A.; Aoyagi, K.; Uetani, M.; Hayashi, K.; Nakamura, T. Analysis of Trabecular Microarchitecture of Human Iliac Bone Using Microcomputed Tomography in Patients with Hip Arthrosis with or without Vertebral Fracture. Bone 1998, 23, 163–169. [Google Scholar] [CrossRef]
- Clark, D.P.; Badea, C.T. Advances in Micro CT Imaging of Small Animals. Phys. Med. 2021, 88, 175–192. [Google Scholar] [CrossRef]
- Stokovic, N.; Ivanjko, N.; Maticic, D.; Luyten, F.P.; Vukicevic, S. Bone Morphogenetic Proteins, Carriers, and Animal Models in the Development of Novel Bone Regenerative Therapies. Materials 2021, 14, 3513. [Google Scholar] [CrossRef]
- Stokovic, N.; Ivanjko, N.; Erjavec, I.; Breski, A.; Peric, M.; Vukicevic, S. Zoledronate Bound to Ceramics Increases Ectopic Bone Volume Induced by rhBMP6 Delivered in Autologous Blood Coagulum in Rats. Biomedicines 2021, 9, 1487. [Google Scholar] [CrossRef]
- Khoury, B.M.; Bigelow, E.M.; Smith, L.M.; Schlecht, S.H.; Scheller, E.L.; Andarawis-Puri, N.; Jepsen, K.J. The Use of Nano-Computed Tomography to Enhance Musculoskeletal Research. Connect. Tissue Res. 2015, 56, 106–119. [Google Scholar] [CrossRef]
- Peyrin, F.; Dong, P.; Pacureanu, A.; Langer, M. Micro- and Nano-CT for the Study of Bone Ultrastructure. Curr. Osteoporos. Rep. 2014, 12, 465–474. [Google Scholar] [CrossRef]
- Salmon, P.L.; Sasov, A.Y. Application of Nano-CT and High-Resolution Micro-CT to Study Bone Quality and Ultrastructure, Scaffold Biomaterials and Vascular Networks. In Advanced Bioimaging Technologies in Assessment of the Quality of Bone and Scaffold Materials: Techniques and Applications; Springer: Berlin/Heidelberg, Germany, 2007; pp. 323–331. [Google Scholar] [CrossRef]
- Wähnert, D.; Greiner, J.; Brianza, S.; Kaltschmidt, C.; Vordemvenne, T.; Kaltschmidt, B. Strategies to Improve Bone Healing: Innovative Surgical Implants Meet Nano-/Micro-Topography of Bone Scaffolds. Biomedicines 2021, 9, 746. [Google Scholar] [CrossRef]
- Hildebrand, T.; Nogueira, L.; Sunde, P.T.; Ørstavik, D.; Glasmacher, B.; Haugen, H.J. Contrast-Enhanced Nano-CT Reveals Soft Dental Tissues and Cellular Layers. Int. Endod. J. 2021, 54, 1275–1288. [Google Scholar] [CrossRef] [PubMed]
- Cuijpers, V.M.; Jaroszewicz, J.; Anil, S.; Al Farraj Aldosari, A.; Walboomers, X.F.; Jansen, J.A. Resolution, Sensitivity, and In Vivo Application of High-Resolution Computed Tomography for Titanium-Coated Polymethyl Methacrylate (PMMA) Dental Implants. Clin. Oral Implant. Res. 2014, 25, 359–365. [Google Scholar] [CrossRef]
- Ahmed, H.M. Nano-Computed Tomography: Current and Future Perspectives. Restor. Dent. Endod. 2016, 41, 236–238. [Google Scholar] [CrossRef] [PubMed]
- Hirata, K.; Tamaki, N. Quantitative FDG PET Assessment for Oncology Therapy. Cancers 2021, 13, 869. [Google Scholar] [CrossRef] [PubMed]
- Krause, B.J.; Schwarzenböck, S.; Souvatzoglou, M. FDG PET and PET/CT. In Molecular Imaging in Oncology; Springer: Berlin/Heidelberg, Germany, 2012; pp. 351–369. [Google Scholar]
- Boellaard, R.; Delgado-Bolton, R.; Oyen, W.J.; Giammarile, F.; Tatsch, K.; Eschner, W.; Verzijlbergen, F.J.; Barrington, S.F.; Pike, L.C.; Weber, W.A.; et al. European Association of Nuclear Medicine (EANM). FDG PET/CT: EANM procedure guidelines for tumour imaging: Version 2.0. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 328–354. [Google Scholar] [CrossRef]
- El-Galaly, T.C.; Gormsen, L.C.; Hutchings, M. PET/CT for Staging; Past, Present, and Future. Semin. Nucl. Med. 2018, 48, 4–16. [Google Scholar] [CrossRef]
- Marcus, C.; Mena, E.; Subramaniam, R.M. Brain PET in the Diagnosis of Alzheimer’s Disease. Clin. Nucl. Med. 2014, 39, e413–e426. [Google Scholar] [CrossRef]
- Gaemperli, O.; Kaufmann, P.A. PET and PET/CT in Cardiovascular Disease. Ann. N. Y. Acad. Sci. 2011, 1228, 109–136. [Google Scholar] [CrossRef]
- Kung, B.T.; Seraj, S.M.; Zadeh, M.Z.; Rojulpote, C.; Kothekar, E.; Ayubcha, C.; Alavi, A. An update on the role of 18F-FDG-PET/CT in major infectious and inflammatory diseases. Am. J. Nucl. Med. Mol. Imaging 2019, 9, 255. [Google Scholar]
- Lu, W.; Duan, Y.; Li, K.; Qiu, J.; Cheng, Z. Glucose uptake and distribution across the human skeleton using state-of-the-art total-body PET/CT. Bone Res. 2023, 11, 36. [Google Scholar] [CrossRef] [PubMed]
- Purbhoo, K.; Vangu, M.D.T. Normal Variants and Pitfalls of 18F-FDG PET/CT Imaging in Pediatric Oncology. Front. Nucl. Med. 2022, 2, 825891. [Google Scholar] [CrossRef]
- Zoch, M.L.; Abou, D.S.; Clemens, T.L.; Thorek, D.L.J.; Riddle, R.C. In Vivo Radiometric Analysis of Glucose Uptake and Distribution in Mouse Bone. Bone Res. 2016, 4, 16004. [Google Scholar] [CrossRef] [PubMed]
- Menendez, M.I.; Moore, R.R.; Abdel-Rasoul, M.; Wright, C.L.; Fernandez, S.; Jackson, R.D.; Knopp, M.V. [18F] Sodium Fluoride Dose Reduction Enabled by Digital Photon Counting PET/CT for Evaluation of Osteoblastic Activity. Front. Med. 2022, 8, 725118. [Google Scholar] [CrossRef]
- Pijl, J.P.; Nienhuis, P.H.; Kwee, T.C.; Glaudemans, A.W.J.M.; Slart, R.H.J.A.; Gormsen, L.C. Limitations and Pitfalls of FDG-PET/CT in Infection and Inflammation. Semin. Nucl. Med. 2021, 51, 633–645. [Google Scholar] [CrossRef]
- Narváez, J.; Estrada, P.; Vidal-Montal, P.; Sánchez-Rodríguez, I.; Sabaté-Llobera, A.; Nolla, J.M.; Cortés-Romera, M. Impact of Previous Glucocorticoid Therapy on Diagnostic Accuracy of [18F] FDG PET-CT in Giant Cell Arteritis. Semin. Arthritis Rheum. 2023, 60, 152183. [Google Scholar] [CrossRef]
- Vlaardingerbroek, M.T.; Boer, J.A. Magnetic Resonance Imaging: Theory and Practice; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2013. [Google Scholar]
- Van Geuns, R.J.M.; Wielopolski, P.A.; de Bruin, H.G.; Rensing, B.J.; van Ooijen, P.M.; Hulshoff, M.; de Feyter, P.J. Basic principles of magnetic resonance imaging. Prog. Cardiovasc. Dis. 1999, 42, 149–156. [Google Scholar] [CrossRef]
- Akhavan, S.; Martinkovich, S.C.; Kasik, C.; DeMeo, P.J. Bone marrow edema, clinical significance, and treatment options: A review. J. Am. Acad. Orthop. Surg. 2020, 28, e888–e899. [Google Scholar] [CrossRef] [PubMed]
- Florkow, M.C.; Willemsen, K.; Mascarenhas, V.V.; Oei, E.H.G.; van Stralen, M.; Seevinck, P.R. Magnetic resonance imaging versus computed tomography for three-dimensional bone imaging of musculoskeletal pathologies: A review. J. Magn. Reson. Imaging 2022, 56, 11–34. [Google Scholar] [CrossRef]
- Sammet, S. Magnetic Resonance Safety. Abdom. Radiol. 2016, 41, 444–451. [Google Scholar] [CrossRef]
- Katti, G.; Ara, S.A.; Shireen, A. Magnetic Resonance Imaging (MRI)–A Review. Int. J. Dent. Clin. 2011, 3, 65–70. [Google Scholar]
- Marty, B.; Coppa, B.; Carlier, P.G. Monitoring Skeletal Muscle Chronic Fatty Degenerations with Fast T1-Mapping. Eur. Radiol. 2018, 28, 4662–4668. [Google Scholar] [CrossRef]
- Weber, M.A. (Ed.) Magnetic Resonance Imaging of the Skeletal Musculature; Springer: Berlin/Heidelberg, Germany, 2014. [Google Scholar] [CrossRef]
- Peterfy, C.G.; Guermazi, A.; Zaim, S.; Tirman, P.F.J.; Miaux, Y.; White, D.; Genant, H.K. Whole-Organ Magnetic Resonance Imaging Score (WORMS) of the Knee in Osteoarthritis. Osteoarthr. Cartil. 2004, 12, 177–190. [Google Scholar] [CrossRef]
- Maksymowych, W.P.; Mallon, C.; Morrow, S.; Shojania, K.; Olszynski, W.P.; Wong, R.L.; Conner-Spady, B. Development and Validation of the Spondyloarthritis Research Consortium of Canada (SPARCC) Enthesitis Index. Ann. Rheum. Dis. 2009, 68, 948–953. [Google Scholar] [CrossRef] [PubMed]
- Østergaard, M.; Peterfy, C.G.; Bird, P.; Gandjbakhch, F.; Glinatsi, D.; Eshed, I.; Conaghan, P.G. The OMERACT Rheumatoid Arthritis Magnetic Resonance Imaging (MRI) Scoring System: Updated Recommendations by the OMERACT MRI in Arthritis Working Group. J. Rheumatol. 2017, 44, 1706–1712. [Google Scholar] [CrossRef] [PubMed]
- Iacobellis, F.; Grassi, R.; Berritto, D.; Mansi, L. Preclinical Techniques in Animals. In Advanced Imaging Techniques in Clinical Pathology; Sacerdoti, F.M., Giordano, A., Cavaliere, C., Eds.; Humana/Springer: New York, NY, USA, 2016; pp. 113–121. [Google Scholar] [CrossRef]
- Niendorf, T.; Pohlmann, A.; Reimann, H.M.; Waiczies, H.; Peper, E.; Huelnhagen, T.; Seeliger, E.; Schreiber, A.; Kettritz, R.; Strobel, K.; et al. Advancing Cardiovascular, Neurovascular, and Renal Magnetic Resonance Imaging in Small Rodents Using Cryogenic Radiofrequency Coil Technology. Front. Pharmacol. 2015, 6, 255. [Google Scholar] [CrossRef] [PubMed]
- Keenan, K.E.; Biller, J.R.; Delfino, J.G.; Boss, M.A.; Does, M.D.; Evelhoch, J.L.; Sullivan, D.C. Recommendations towards Standards for Quantitative MRI (qMRI) and Outstanding Needs. J. Magn. Reson. Imaging 2019, 49, e26–e39. [Google Scholar] [CrossRef]
- Cashmore, M.T.; McCann, A.J.; Wastling, S.J.; McGrath, C.; Thornton, J.; Hall, M.G. Clinical Quantitative MRI and the Need for Metrology. Br. J. Radiol. 2021, 94, 20201215. [Google Scholar] [CrossRef]
- Damon, B.M.; Li, K.; Dortch, R.D.; Welch, E.B.; Park, J.H.; Buck, A.K.; Bryant, N.D. Quantitative Magnetic Resonance Imaging of Skeletal Muscle Disease. J. Vis. Exp. 2016, 118, 52352. [Google Scholar] [CrossRef]
- Manfrini, E.; Smits, M.; Thust, S.; Geiger, S.; Bendella, Z.; Petr, J.; Solymosi, L.; Keil, V.C. From Research to Clinical Practice: A European Neuroradiological Survey on Quantitative Advanced MRI Implementation. Eur. Radiol. 2021, 31, 6334–6341. [Google Scholar] [CrossRef]
- Han, X.; Jovicich, J.; Salat, D.; van der Kouwe, A.; Quinn, B.; Czanner, S.; Fischl, B. Reliability of MRI-derived measurements of human cerebral cortical thickness: The effects of field strength, scanner upgrade and manufacturer. Neuroimage 2006, 32, 180–194. [Google Scholar] [CrossRef]
- Hooijmans, M.T.; Schlaffke, L.; Bolsterlee, B.; Schlaeger, S.; Marty, B.; Mazzoli, V. Compositional and Functional MRI of Skeletal Muscle: A Review. J. Magn. Reson. Imaging 2023, 60, 860–877. [Google Scholar] [CrossRef]
- Güttsches, A.K.; Rehmann, R.; Schreiner, A.; Rohm, M.; Forsting, J.; Froeling, M.; Tegenthoff, M.; Vorgerd, M.; Schlaffke, L. Quantitative Muscle-MRI Correlates with Histopathology in Skeletal Muscle Biopsies. J. Neuromuscul. Dis. 2021, 8, 669–678. [Google Scholar] [CrossRef] [PubMed]
- Schlaffke, L.; Rehmann, R.; Güttsches, A.K.; Vorgerd, M.; Meyer-Frießem, C.H.; Dinse, H.R.; Enax-Krumova, E.; Froeling, M.; Forsting, J. Evaluation of Neuromuscular Diseases and Complaints by Quantitative Muscle MRI. J. Clin. Med. 2024, 13, 1958. [Google Scholar] [CrossRef]
- Adler, R.S. What Is the Place of Ultrasound in MSK Imaging? Skelet. Radiol. 2024, 53, 1699–1709. [Google Scholar] [CrossRef]
- Todd-Donato, A.B.; VanDeventer, G.M.; Porter, I.R.; Krotscheck, U. Ultrasound Is an Accurate Imaging Modality for Diagnosing Hip Luxation in Dogs Presenting with Hind Limb Lameness. J. Am. Vet. Med. Assoc. 2024, 1, 1–9. [Google Scholar] [CrossRef]
- Karatrantos, A.T.; Sideri, A.I.; Gouletsou, P.G.; Bektsi, C.G.; Barbagianni, M.S. Ultrasound Imaging Modalities in the Evaluation of the Dog’s Stifle Joint. Vet. Sci. 2025, 12, 734. [Google Scholar] [CrossRef]
- Ponti, F.; Arioli, A.; Longo, C.; Miceli, M.; Colangeli, M.; Papalexis, N.; Spinnato, P. Ultrasound-Guided Percutaneous Bone Biopsy: Feasibility, Diagnostic Yield and Technical Notes. Diagnostics 2023, 13, 1773. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Zhang, Y.; Wang, X.; Liu, J. Comparative Analysis of Imaging Modalities for Diagnosing Musculoskeletal Disorders. J. Innov. Med. Res. 2024, 3, 45–53. [Google Scholar] [CrossRef]
- Duck, F.A.; Thomas, A.M.K. Paul Langevin (1872–1946): The Father of Ultrasonics. Med. Phys. 2022, 10, 1. [Google Scholar] [CrossRef]
- Yamaguchi, T. Basic Concept and Clinical Applications of Quantitative Ultrasound (QUS) Technologies. J. Med. Ultrason. 2021, 48, 391–402. [Google Scholar] [CrossRef] [PubMed]
- Escobio-Prieto, I.; Blanco-Díaz, M.; Pinero-Pinto, E.; Rodriguez-Rodriguez, A.M.; Ruiz-Dorantes, F.J.; Albornoz-Cabello, M. Quantitative Ultrasound and Bone Health in Elderly People, a Systematic Review. Biomedicines 2023, 11, 1175. [Google Scholar] [CrossRef] [PubMed]
- Cloutier, G.; Destrempes, F.; Yu, F.; Tang, A. Quantitative Ultrasound Imaging of Soft Biological Tissues: A Primer for Radiologists and Medical Physicists. Insights Imaging 2021, 12, 127. [Google Scholar] [CrossRef] [PubMed]
- Buttar, S.; Cooper, D., Jr.; Olivieri, P.; Barca, M.; Drake, A.B.; Ku, M.; Rose, G.; Siadecki, S.D.; Saul, T. Air and Its Sonographic Appearance: Understanding the Artifacts. J. Emerg. Med. 2017, 53, 241–247. [Google Scholar] [CrossRef]
- Oelze, M.L.; Mamou, J. Review of quantitative ultrasound: Envelope statistics and backscatter coefficient imaging and contributions to diagnostic ultrasound. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2016, 63, 336–351. [Google Scholar] [CrossRef]
- Carstanjen, B.; Lepage, O.M.; Detilleux, J.; Duboeuf, F.; Amory, H. Use of Multi-Site Quantitative Ultrasonography for Noninvasive Assessment of Bone in Horses. Am. J. Vet. Res. 2002, 63, 1464–1469. [Google Scholar] [CrossRef]
- Lauer, S.K.; Davies, E.S.; Hosgood, G.; Cremer, J. Quantitative Ultrasonography for Assessment of Bone Mineral Density in the Canine Radius and Tibia. Vet. Radiol. Ultrasound 2009, 50, 97–103. [Google Scholar] [CrossRef]
- Krieg, M.A.; Barkmann, R.; Gonnelli, S.; Stewart, A.; Bauer, D.C.; Barquero, L.D.R.; Hans, D. Quantitative Ultrasound in the Management of Osteoporosis: The 2007 ISCD Official Positions. J. Clin. Densitom. 2008, 11, 163–187. [Google Scholar] [CrossRef]
- Prins, S.H.; Jørgensen, H.L.; Jørgensen, L.V.; Hassager, C. The Role of Quantitative Ultrasound in the Assessment of Bone: A Review. Clin. Physiol. 1998, 18, 3–17. [Google Scholar] [CrossRef] [PubMed]
- To, W.W.K.; Wong, M.W.N. Bone Mineral Density Changes during Pregnancy in Actively Exercising Women as Measured by Quantitative Ultrasound. Arch. Gynecol. Obstet. 2012, 286, 357–363. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hans, D.; Baim, S. Quantitative Ultrasound (QUS) in the Management of Osteoporosis and Assessment of Fracture Risk. J. Clin. Densitom. 2017, 20, 322–333. [Google Scholar] [CrossRef] [PubMed]
- Grimal, Q.; Laugier, P. Quantitative Ultrasound Assessment of Cortical Bone Properties beyond Bone Mineral Density. IRBM 2019, 40, 16–24. [Google Scholar] [CrossRef]
- Goossens, L.; Vanderoost, J.; Jaecques, S.; Boonen, S.; D’hooge, J.; Lauriks, W.; Van der Perre, G. The Correlation between the SOS in Trabecular Bone and Stiffness and Density Studied by Finite-Element Analysis. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2008, 55, 1234–1242. [Google Scholar] [CrossRef]
- Makanjee, C.R.; Bergh, A.M.; Hoffmann, W.A. Distributed Decision Making in Action: Diagnostic Imaging Investigations within the Bigger Picture. J. Med. Radiat. Sci. 2018, 65, 5–12. [Google Scholar] [CrossRef]
- Hussain, S.; Mubeen, I.; Ullah, N.; Shah, S.S.; Khan, B.A.; Zahoor, M.; Ullah, R.; Khan, F.A.; Sultan, M.A. Modern Diagnostic Imaging Technique Applications and Risk Factors in the Medical Field: A Review. Biomed. Res. Int. 2022, 2022, 5164970. [Google Scholar] [CrossRef]
- Garrett, K.S. When Radiography and Ultrasonography Are Not Enough: The Use of Computed Tomography and Magnetic Resonance Imaging for Equine Lameness Cases. J. Am. Vet. Med. Assoc. 2022, 260, 1113–1123. [Google Scholar] [CrossRef]
- Andrade, M.C.; De Rus Aznar, I.; Brunnberg, M.; Slunsky, P. Indications for the Removal of Implants after Fracture Healing: A Comparison between Human and Veterinary Medicine. Vet. Med. 2023, 68, 259–270. [Google Scholar] [CrossRef]
- Laignel, A. Radiographic Examinations in Alert Dogs and Dogs Under Chemical Restraint: Link Between Animal Stress and the Radiographic Procedure. Ph.D. Thesis, Ecole Nationale Vétérinaire d’Alfort-Enva, Maisons-Alfort, France, 2024. [Google Scholar]
- Surjan, Y.; Ostwald, P.; Milross, C.; Warren-Forward, H. Radiation Safety Considerations and Compliance within Equine Veterinary Clinics: Results of an Australian Survey. Radiography 2015, 21, 224–230. [Google Scholar] [CrossRef]
- Gregorich, S.L.; Sutherland-Smith, J.; Sato, A.F.; May-Trifiletti, J.A.; Miller, K.J. Survey of Veterinary Specialists Regarding Their Knowledge of Radiation Safety and the Availability of Radiation Safety Training. J. Am. Vet. Med. Assoc. 2018, 252, 1133–1140. [Google Scholar] [CrossRef]
- Belotta, A.F.; Mayer, M.N.; Koehncke, N.K.; Carmalt, J.; Freitas, F.P.; Waldner, C.L. Survey of Self-Reported Radiation Safety Practices among North American Veterinary Technicians Involved in Equine Radiography Using Portable X-Ray Equipment. J. Am. Vet. Med. Assoc. 2021, 259, 919–926. [Google Scholar] [CrossRef]
- Ndiaye, Y.S.; Cramton, P.; Chernev, C.; Ockenfels, A.; Schwarz, T. Comparison of Radiological Interpretation Made by Veterinary Radiologists and State-of-the-Art Commercial AI Software for Canine and Feline Radiographic Studies. Front. Vet. Sci. 2025, 12, 1502790. [Google Scholar] [CrossRef] [PubMed]
- Gitto, S.; Serpi, F.; Albano, D.; Risoleo, G.; Fusco, S.; Messina, C.; Sconfienza, L.M. AI Applications in Musculoskeletal Imaging: A Narrative Review. Eur. Radiol. Exp. 2024, 8, 22. [Google Scholar] [CrossRef] [PubMed]
- Thrall, D.E.; Robertson, I.D. Atlas of Normal Radiographic Anatomy and Anatomic Variants in the Dog and Cat-E-Book; Elsevier Health Sciences: Amsterdam, The Netherlands, 2022. [Google Scholar]
- Naeem, Z.; Bennion, C. Choosing the Best Modality. Imaging Ther. Pract. 2016, 5, 287–294. [Google Scholar] [CrossRef]
- Sharma, P.; Suehling, M.; Flohr, T.; Comaniciu, D. Artificial Intelligence in Diagnostic Imaging: Status Quo, Challenges, and Future Opportunities. J. Thorac. Imaging 2020, 35, S11–S16. [Google Scholar] [CrossRef]
- Debs, P.; Fayad, L.M. The Promise and Limitations of Artificial Intelligence in Musculoskeletal Imaging. Front. Radiol. 2023, 3, 1242902. [Google Scholar] [CrossRef]
- Burti, S.; Banzato, T.; Coghlan, S.; Wodzinski, M.; Bendazzoli, M.; Zotti, A. Artificial Intelligence in Veterinary Diagnostic Imaging: Perspectives and Limitations. Res. Vet. Sci. 2024, 175, 105317. [Google Scholar] [CrossRef]
- Dixon, J.; Biggi, M.; Weller, R. Common Artefacts and Pitfalls in Equine Computed and Digital Radiography and How to Avoid Them. Equine Vet. Educ. 2018, 30, 326–335. [Google Scholar] [CrossRef]
- Hennessey, E.; DiFazio, M.; Hennessey, R.; Cassel, N. Artificial Intelligence in Veterinary Diagnostic Imaging: A Literature Review. Vet. Radiol. Ultrasound 2022, 63, 851–870. [Google Scholar] [CrossRef]
- Kulak, O.; Gören, H.G.; Supciller, A.A. A New Multi Criteria Decision Making Approach for Medical Imaging Systems Considering Risk Factors. Appl. Soft Comput. 2015, 35, 931–941. [Google Scholar] [CrossRef]
- Castillo, C.; Steffens, T.; Sim, L.; Caffery, L. The Effect of Clinical Information on Radiology Reporting: A Systematic Review. J. Med. Radiat. Sci. 2021, 68, 60–74. [Google Scholar] [CrossRef]
- Demehri, S.; Baffour, F.I.; Klein, J.G.; Ghotbi, E.; Ibad, H.A.; Moradi, K.; Taguchi, K.; Fritz, J.; Carrino, J.A.; Guermazi, A.; et al. Musculoskeletal CT Imaging: State-of-the-Art Advancements and Future Directions. Radiology 2023, 308, e230344. [Google Scholar] [CrossRef]
- Dundamadappa, S.K.; Cauley, K.A. MR Imaging of Acute Cervical Spinal Ligamentous and Soft Tissue Trauma. Emerg. Radiol. 2012, 19, 277–286. [Google Scholar] [CrossRef]
- Ariyachaipanich, A.; Bae, W.C.; Statum, S.; Chung, C.B. Update on MRI Pulse Sequences for the Knee: Imaging of Cartilage, Meniscus, Tendon, and Hardware. Semin. Musculoskelet. Radiol. 2017, 21, 45–62. [Google Scholar] [CrossRef]
- Stouge, A.; Khan, K.S.; Kristensen, A.G.; Tankisi, H.; Schlaffke, L.; Froeling, M.; Væggemose, M.; Andersen, H. MRI of Skeletal Muscles in Participants with Type 2 Diabetes with or without Diabetic Polyneuropathy. Radiology 2020, 297, 608–619. [Google Scholar] [CrossRef] [PubMed]
- Bosma, S.E.; Vriens, D.; Gelderblom, H.; van de Sande, M.A.; Dijkstra, P.D.; Bloem, J.L. 18F-FDG PET-CT versus MRI for Detection of Skeletal Metastasis in Ewing Sarcoma. Skelet. Radiol. 2019, 48, 1735–1746. [Google Scholar] [CrossRef] [PubMed]
- Pecin, M.; Stokovic, N.; Ivanjko, N.; Smajlovic, A.; Kreszinger, M.; Capak, H.; Vrbanac, Z.; Oppermann, H.; Maticic, D.; Vukicevic, S. A Novel Autologous Bone Graft Substitute Containing rhBMP6 in Autologous Blood Coagulum with Synthetic Ceramics for Reconstruction of a Large Humerus Segmental Gunshot Defect in a Dog: The First Veterinary Patient to Receive a Novel Osteoinductive Therapy. Bone Rep. 2021, 14, 100759. [Google Scholar] [CrossRef] [PubMed]
- Stokovic, N.; Ivanjko, N.; Milesevic, M.; Sampath, T.K.; Vukicevic, S. A Simple Rodent Subcutaneous Assay for Identification of New Osteoinductive Molecules: The Key Method for Screening of Novel Bone Regeneration Implants. Rad Hrvat. Akad. Znan. I umjetnosti. Med. Znan. 2022, 553, 40–53. [Google Scholar] [CrossRef]
- Ivanjko, N.; Stokovic, N.; Milesevic, M.; Rumenovic, V.; Windhager, R.; Sampath, K.T.; Kovacic, N.; Grcevic, D.; Vukicevic, S. rhBMP6 in Autologous Blood Coagulum Is a Preferred Osteoinductive Device to rhBMP2 on Bovine Collagen Sponge in the Rat Ectopic Bone Formation Assay. Biomed. Pharmacother. 2023, 169, 115844. [Google Scholar] [CrossRef]
- Stokovic, N.; Ivanjko, N.; Rumenovic, V.; Breski, A.; Sampath, K.T.; Peric, M.; Pecina, M.; Vukicevic, S. Comparison of Synthetic Ceramic Products Formulated with Autologous Blood Coagulum Containing rhBMP6 for Induction of Bone Formation. Int. Orthop. 2022, 46, 2693–2704. [Google Scholar] [CrossRef] [PubMed]
- Machrowska, A.; Karpiński, R.; Maciejewski, M.; Jonak, J.; Krakowski, P.; Syta, A. Application of Recurrence Quantification Analysis in the Detection of Osteoarthritis of the Knee with the Use of Vibroarthrography. Adv. Sci. Technol. Res. J. 2024, 18, 19–31. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Javor, A.; Štoković, N.; Ivanjko, N.; Lukša, I.; Capak, H.; Vrbanac, Z. Diagnostic Imaging of the Skeletal System: Overview of Applications in Human and Veterinary Medicine. Bioengineering 2025, 12, 1358. https://doi.org/10.3390/bioengineering12121358
Javor A, Štoković N, Ivanjko N, Lukša I, Capak H, Vrbanac Z. Diagnostic Imaging of the Skeletal System: Overview of Applications in Human and Veterinary Medicine. Bioengineering. 2025; 12(12):1358. https://doi.org/10.3390/bioengineering12121358
Chicago/Turabian StyleJavor, Ana, Nikola Štoković, Natalia Ivanjko, Iva Lukša, Hrvoje Capak, and Zoran Vrbanac. 2025. "Diagnostic Imaging of the Skeletal System: Overview of Applications in Human and Veterinary Medicine" Bioengineering 12, no. 12: 1358. https://doi.org/10.3390/bioengineering12121358
APA StyleJavor, A., Štoković, N., Ivanjko, N., Lukša, I., Capak, H., & Vrbanac, Z. (2025). Diagnostic Imaging of the Skeletal System: Overview of Applications in Human and Veterinary Medicine. Bioengineering, 12(12), 1358. https://doi.org/10.3390/bioengineering12121358






