Preclinical Mechanistic Evaluation of Hyaluronan/Niacinamide (Vitamin B3) Hydrogels: Toward an Enhanced Viscosupplement System with Ancillary Anti-Arthritic Attributes
Abstract
1. Introduction
2. Materials and Methods
2.1. Investigational Sample Preparation
2.2. Cell Lines and Primary Chondrocyte Cell Type
2.3. MTS Cellular Viability Assays
2.4. Modulation of Protein Synthesis in Cell Lines
2.5. Modulation of Signaling Pathways in Cell Lines
2.6. ELISA Dosing of PGE2, TNF-α, IL-1β, and MMP-13 in Cell Lines
2.7. Dosing of Nitric Oxide
2.8. Western Blot Detection of COX-2, iNOS, P38, NF-κB, and ERK1/2
2.9. Evaluation of Anti-Hypertrophic Effects on Rat Chondrocytes
2.9.1. In Vitro Assay Design
- Untreated cells (i.e., cultures in proliferation medium);
- IL-1β-treated cells;
- IL-1β-treated cells + 1:10 dilution of HA sample without niacinamide;
- IL-1β-treated cells + 1:10 dilution of HA sample with niacinamide;
- IL-1β-treated cells + 1:100 dilution of HA sample with niacinamide.
2.9.2. Preparation of Test Compounds
2.9.3. qPCR Analyses
2.9.4. ELISA Analyses
2.10. Statistical Analyses and Data Presentation
3. Results and Discussion
3.1. Cellular Viability Evaluation by MTS Assays
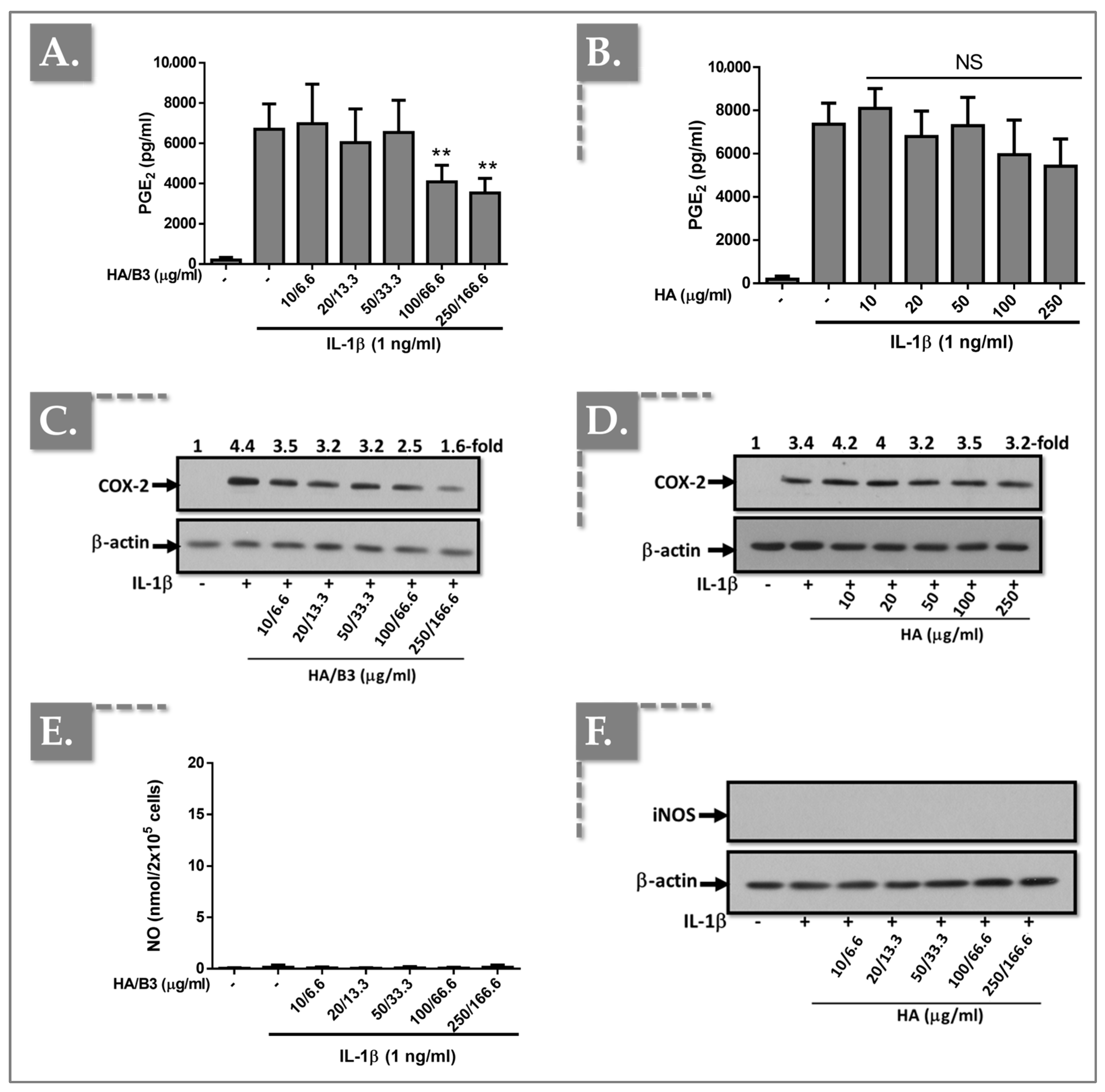
3.2. The HA/B3 Combination Inhibits NO and PGE2 Production by Reducing iNOS and COX-2 Expression
3.3. HA/B3 Combinations Inhibit the Production of TNF-α in LPS-Induced Macrophages
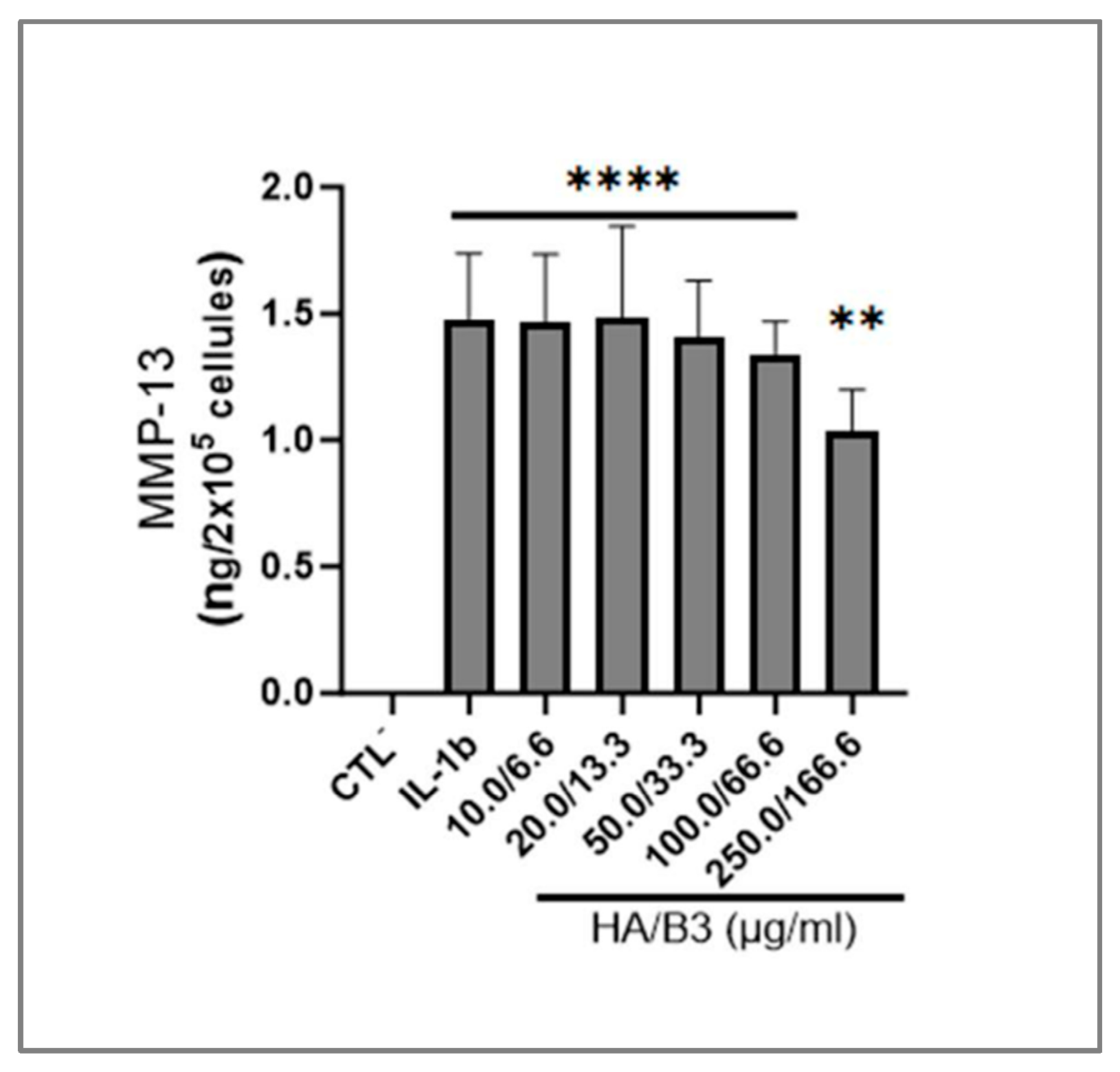
3.4. HA/B3 Combinations Inhibit the MAP Kinase Signaling Pathway and Downregulate the Production of MMP-13
3.5. HA/B3 Combinations Display Anti-Hypertrophic Effects in Rat Primary Chondrocytes
3.6. General Discussion
3.7. Study Limitations and Perspectives
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| DMEM | Dulbecco’s modified Eagle medium |
| ECM | extracellular matrix |
| ELISA | enzyme-linked immunosorbent assay |
| FBS | fetal bovine serum |
| FDA | US Food and Drug Administration |
| H2O2 | hydrogen peroxide |
| HA | hyaluronic acid |
| HYAL | hyaluronidases |
| IL | interleukins |
| LPS | lipopolysaccharide |
| MDa | megadalton |
| min | minute |
| NA | non-applicable |
| NO | nitric oxide |
| OA | osteoarthritis |
| OD | optic density |
| PBS | phosphate-buffered saline |
| PRP | platelet-rich plasma |
| RNA | ribonucleic acid |
| ROS | reactive oxygen species |
| TNF | tumor necrosis factor |
| USA | United States of America |
References
- Sacitharan, P.K. Ageing and osteoarthritis. Sub-Cell. Biochem. 2019, 91, 123–159. [Google Scholar] [CrossRef]
- Lee, Y.T.; Mohd Yunus, M.H.; Yazid, M.D.; Ugusman, A. Unraveling the path to osteoarthritis management: Targeting chondrocyte apoptosis for therapeutic intervention. Front. Cell Develop. Biol. 2024, 12, 1347126. [Google Scholar] [CrossRef] [PubMed]
- Heinegård, D.; Saxne, T. The role of the cartilage matrix in osteoarthritis. Nat. Rev. Rheumatol. 2011, 7, 50–56. [Google Scholar] [CrossRef]
- Song, J.; Zeng, X.; Li, C.; Yin, H.; Mao, S.; Ren, D. Alteration in cartilage matrix stiffness as an indicator and modulator of osteoarthritis. Biosci. Rep. 2024, 44, BSR20231730. [Google Scholar] [CrossRef]
- Rim, Y.A.; Nam, Y.; Ju, J.H. The role of chondrocyte hypertrophy and senescence in osteoarthritis initiation and progression. Int. J. Mol. Sci. 2020, 21, 2358. [Google Scholar] [CrossRef] [PubMed]
- Pujol, J.P.; Brisset, M.; Jourdan, C.; Bocquet, J.; Jouis, V.; Béliard, R.; Loyau, G. Effect of a monocyte cell factor (MCF) on collagen production in cultured articular chondrocytes: Role of prostaglandin E2. Biochem. Biophys. Res. Comm. 1984, 119, 499–508. [Google Scholar] [CrossRef]
- Gowen, M.; Wood, D.D.; Ihrie, E.J.; Meats, J.E.; Russell, R.G. Stimulation by human interleukin 1 of cartilage breakdown and production of collagenase and proteoglycanase by human chondrocytes but not by human osteoblasts in vitro. Biochim. Biophys. Acta 1984, 797, 186–193. [Google Scholar] [CrossRef]
- Zayed, N.; Afif, H.; Chabane, N.; Mfuna-Endam, L.; Benderdour, M.; Martel-Pelletier, J.; Pelletier, J.P.; Motiani, R.K.; Trebak, M.; Duval, N.; et al. Inhibition of Interleukin-1β-Induced Matrix Metalloproteinases 1 and 13 Production in Human Osteoarthritic Chondrocytes by Prostaglandin D2. Arthritis Rheum. 2008, 58, 3530–3540. [Google Scholar] [CrossRef]
- Benderdour, M.; Tardif, G.; Pelletier, J.P.; Di Battista, J.A.; Reboul, P.; Ranger, P.; Martel-Pelletier, J. Interleukin 17 (IL-17) induces collagenase-3 production in human osteoarthritic chondrocytes via AP-1 dependent activation: Differential activation of AP-1 members by IL-17 and IL-1β. J. Rheumatol. 2002, 29, 1262–1272. [Google Scholar]
- Abusarah, J.; Bentz, M.; Benabdoune, H.; Rondon, P.E.; Shi, Q.; Fernandes, J.C.; Fahmi, H.; Benderdour, M. An Overview of the Role of Lipid Peroxidation-Derived 4-Hydroxynonenal in Osteoarthritis. Inflamm. Res. 2017, 66, 637–651. [Google Scholar] [CrossRef] [PubMed]
- Shi, Q.; Abusarah, J.; Zaouter, C.; Moldovan, F.; Fernandes, J.C.; Fahmi, H.; Benderdour, M. New Evidence Implicating 4-Hydroxynonenal in the Pathogenesis of Osteoarthritis In Vivo. Arthritis Rheumatol. 2014, 66, 2461–2471. [Google Scholar] [CrossRef]
- Akkiraju, H.; Nohe, A. Role of chondrocytes in cartilage formation, progression of osteoarthritis and cartilage regeneration. J. Dev. Biol. 2015, 3, 177–192. [Google Scholar] [CrossRef] [PubMed]
- Taruc-Uy, R.L.; Lynch, S.A. Diagnosis and treatment of osteoarthritis. Prim. Care 2013, 40, 821–836. [Google Scholar] [CrossRef]
- Abramoff, B.; Caldera, F.E. Osteoarthritis: Pathology, diagnosis, and treatment options. Med. Clin. N. Am. 2020, 104, 293–311. [Google Scholar] [CrossRef]
- Molliard, S.G.; Bétemps, J.B.; Hadjab, B.; Topchian, D.; Micheels, P.; Salomon, D. Key rheological properties of hyaluronic acid fillers: From tissue integration to product degradation. Plast. Aesthetic Res. 2018, 5, 17. [Google Scholar] [CrossRef]
- Schanté, C.; Zubera, G.; Herlinb, C.; Vandamme, T.F. Synthesis of N-alanyl-hyaluronamide with high degree of substitution for enhanced resistance to hyaluronidase-mediated digestion. Carbohydr. Polym. 2011, 86, 747–752. [Google Scholar] [CrossRef]
- Žádníková, P.; Šínová, R.; Pavlík, V.; Šimek, M.; Šafránková, B.; Hermannová, M.; Nešporová, K.; Velebný, V. The degradation of hyaluronan in the skin. Biomolecules 2022, 12, 251. [Google Scholar] [CrossRef] [PubMed]
- Mongkhon, J.M.; Thach, M.; Shi, Q.; Fernandes, J.C.; Fahmi, H.; Benderdour, M. Sorbitol-modified hyaluronic acid reduces oxidative stress, apoptosis and mediators of inflammation and catabolism in human osteoarthritic chondrocytes. Infl. Res. 2014, 63, 691–701. [Google Scholar] [CrossRef]
- Conrozier, T.; Mathieu, P.; Rinaudo, M. Mannitol preserves the viscoelastic properties of hyaluronic acid in an in vitro model of oxidative stress. Rheumatol. Ther. 2014, 1, 45–54. [Google Scholar] [CrossRef]
- Hintze, V.; Schnabelrauch, M.; Rother, S. Chemical modification of hyaluronan and their biomedical applications. Front. Chem. 2022, 10, 830671. [Google Scholar] [CrossRef] [PubMed]
- Raeissadat, S.A.; Ghazi Hosseini, P.; Bahrami, M.H.; Salman Roghani, R.; Fathi, M.; Gharooee Ahangar, A.; Darvish, M. The comparison effects of intra-articular injection of Platelet Rich Plasma (PRP), Plasma Rich in Growth Factor (PRGF), Hyaluronic Acid (HA), and ozone in knee osteoarthritis; A one year randomized clinical trial. BMC Musculoskel. Dis. 2021, 22, 134. [Google Scholar] [CrossRef]
- Tang, J.Z.; Nie, M.J.; Zhao, J.Z.; Zhang, G.C.; Zhang, Q.; Wang, B. Platelet-rich plasma versus hyaluronic acid in the treatment of knee osteoarthritis: A meta-analysis. J. Orthop. Surg. Res. 2020, 15, 403. [Google Scholar] [CrossRef]
- Pratt, R.L. Hyaluronan and the fascial frontier. Int. J. Mol. Sci. 2021, 22, 6845. [Google Scholar] [CrossRef]
- Hardingham, T.; Asar, A.; Longas, M.O. Chemistry and Biology of Hyaluronan, 1st ed.; Garg, H.G., Hales, C.A., Eds.; Elsevier Science: Danvers, MA, USA, 2004. [Google Scholar]
- Bagga, H.; Burkhardt, D.; Sambrook, P.; March, L. Longterm effects of intraarticular hyaluronan on synovial fluid in osteoarthritis of the knee. J. Rheum. 2006, 33, 946–950. [Google Scholar]
- Campo, G.M.; Avenoso, A.; Nastasi, G.; Micali, A.; Prestipino, V.; Vaccaro, M.; D’Ascola, A.; Calatroni, A.; Campo, S. Hyaluronan reduces inflammation in experimental arthritis by modulating TLR-2 and TLR-4 cartilage expression. Biochim. Biophys. Acta 2011, 1812, 1170–1181. [Google Scholar] [CrossRef]
- Ghosh, P.; Guidolin, D. Potential mechanism of action of intra-articular hyaluronan therapy in osteoarthritis: Are the effects molecular weight dependent? Sem. Arthritis Rheum. 2002, 32, 10–37. [Google Scholar] [CrossRef]
- Cyphert, J.M.; Trempus, C.S.; Garantziotis, S. Size Matters: Molecular Weight Specificity of Hyaluronan Effects in Cell Biology. Int. J. Cell Biol. 2015, 2015, 563818. [Google Scholar] [CrossRef] [PubMed]
- Zhai, P.; Peng, X.; Li, B.; Liu, Y.; Sun, H.; Li, X. The application of hyaluronic acid in bone regeneration. Int. J. Biol. Macromol. 2020, 151, 1224–1239. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.C.; Lall, R.; Srivastava, A.; Sinha, A. Hyaluronic acid: Molecular mechanisms and therapeutic trajectory. Front. Vet. Sci. 2019, 6, 192. [Google Scholar] [CrossRef]
- Entwistle, J.; Hall, C.L.; Turley, E.A. HA receptors: Regulators of signalling to the cytoskeleton. J. Cell. Biochem. 1996, 61, 569–577. [Google Scholar] [CrossRef]
- Bano, F.; Tammi, M.I.; Kang, D.W.; Harris, E.N.; Richter, R.P. Single-molecule unbinding forces between the polysaccharide hyaluronan and its binding proteins. Biophys. J. 2018, 114, 2910–2922. [Google Scholar] [CrossRef]
- Kobayashi, T.; Chanmee, T.; Itano, N. Hyaluronan: Metabolism and function. Biomolecules 2020, 10, 1525. [Google Scholar] [CrossRef]
- Karna, E.; Miltyk, W.; Pałka, J.A.; Jarzabek, K.; Wołczyński, S. Hyaluronic acid counteracts interleukin-1-induced inhibition of collagen biosynthesis in cultured human chondrocytes. Pharmacol. Res. 2006, 54, 275–281. [Google Scholar] [CrossRef]
- Misra, S.; Hascall, V.C.; Markwald, R.R.; Ghatak, S. Interactions between hyaluronan and its receptors (CD44, RHAMM) regulate the activities of inflammation and cancer. Front. Immunol. 2015, 6, 201. [Google Scholar] [CrossRef]
- Turley, E.A.; Noble, P.W.; Bourguignon, L.Y. Signaling properties of hyaluronan receptors. J. Biol. Chem. 2002, 277, 4589–4592. [Google Scholar] [CrossRef] [PubMed]
- Skandalis, S.S.; Karalis, T.; Heldin, P. Intracellular hyaluronan: Importance for cellular functions. Sem. Cancer Biol. 2020, 62, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Porcello, A.; Hadjab, F.; Ajouaou, M.; Philippe, V.; Martin, R.; Abdel-Sayed, P.; Hirt-Burri, N.; Scaletta, C.; Raffoul, W.; Applegate, L.A.; et al. Ex vivo functional benchmarking of hyaluronan-based osteoarthritis viscosupplement products: Comprehensive assessment of rheological, lubricative, adhesive, and stability attributes. Gels 2023, 9, 808. [Google Scholar] [CrossRef] [PubMed]
- McCarty, M.F.; Russell, A.L. Niacinamide therapy for osteoarthritis—Does it inhibit nitric oxide synthase induction by interleukin 1 in chondrocytes? Med. Hyp. 1999, 53, 350–360. [Google Scholar] [CrossRef]
- ISO 10993-5; Biological Evaluation of Medical Devices—Part 5: Tests for In Vitro Cytotoxicity. International Organization for Standardization: Geneva, Switzerland, 2009.
- NCL Method ITA-7 Detection of Nitric Oxide Production by the Macrophage Cell Line RAW264.7. Nanotechnology Characterization Laboratory: Frederick, MD, USA. Available online: https://www.cancer.gov/nano/research/ncl/protocols-capabilities/ncl-method-ita-7.pdf (accessed on 3 March 2025).
- Jazrawi, L.M.; Rosen, J. Intra-articular hyaluronic acid: Potential treatment of younger patients with knee injury and/or post-traumatic arthritis. Physician Sportsmed. 2011, 39, 107–113. [Google Scholar] [CrossRef]
- Kang, H.T.; Hwang, E.S. Nicotinamide Enhances Mitochondria Quality through Autophagy Activation in Human Cells. Aging Cell 2009, 8, 426–438. [Google Scholar] [CrossRef]
- Mitsui, Y.; Gotoh, M.; Nakama, K.; Yamada, T.; Higuchi, F.; Nagata, K. Hyaluronic acid inhibits mRNA expression of proinflammatory cytokines and cyclooxygenase-2/prostaglandin E(2) production via CD44 in interleukin-1-stimulated subacromial synovial fibroblasts from patients with rotator cuff disease. J. Orthop. Res. 2008, 26, 1032–1037. [Google Scholar] [CrossRef]
- Chang, C.C.; Hsieh, M.S.; Liao, S.T.; Chen, Y.H.; Cheng, C.W.; Huang, P.T.; Lin, Y.F.; Chen, C.H. Hyaluronan regulates PPARγ and inflammatory responses in IL-1β-stimulated human chondrosarcoma cells, a model for osteoarthritis. Carbohydr. Polym. 2012, 90, 1168–1175. [Google Scholar] [CrossRef] [PubMed]
- Duan, D.; Yang, S.; Shao, Z.; Wang, H.; Xiong, X. Protective effect of niacinamide on interleukin-1β-induced annulus fibrosus type II collagen degeneration in vitro. J. Huazhong Univ. Sci. Technol. Med. Sci. 2007, 27, 68–71. [Google Scholar] [CrossRef] [PubMed]
- Su, C.-F.; Liu, D.D.; Kao, S.J.; Chen, H.I. Nicotinamide abrogates acute lung injury caused by ischaemia/reperfusion. Eur. Respir. J. 2007, 30, 199–204. [Google Scholar] [CrossRef] [PubMed]
- Kulesza, A.; Paczek, L.; Burdzinska, A. The role of COX-2 and PGE2 in the regulation of immunomodulation and other functions of mesenchymal stromal cells. Biomedicines 2023, 11, 445. [Google Scholar] [CrossRef]
- Cheng, H.; Huang, H.; Guo, Z.; Chang, Y.; Li, Z. Role of prostaglandin E2 in tissue repair and regeneration. Theranostics 2021, 11, 8836–8854. [Google Scholar] [CrossRef]
- Nakata, K.; Hanai, T.; Take, Y.; Osada, T.; Tsuchiya, T.; Shima, D.; Fujimoto, Y. Disease-modifying effects of COX-2 selective inhibitors and non-selective NSAIDs in osteoarthritis: A systematic review. Osteoarthr. Cart. 2018, 26, 1263–1273. [Google Scholar] [CrossRef]
- Marques, C.; Hadjab, F.; Porcello, A.; Lourenço, K.; Scaletta, C.; Abdel-Sayed, P.; Hirt-Burri, N.; Applegate, L.A.; Laurent, A. Mechanistic insights into the multiple functions of niacinamide: Therapeutic implications and cosmeceutical applications in functional skincare products. Antioxidants 2024, 13, 425. [Google Scholar] [CrossRef]
- Andrade, J.; Ramírez, R.; Conde, M.; Sobrino, F.; Bedoya, F.J. Nicotinamide inhibits inducible nitric oxide synthase enzyme activity in macrophages by allowing nitric oxide to inhibit its own formation. Life Sci. 1997, 61, 1843–1850. [Google Scholar] [CrossRef]
- Feng, Y.; Wang, Y.; Jiang, C.; Fang, Z.; Zhang, Z.; Lin, X.; Sun, L.; Jiang, W. Nicotinamide Induces Mitochondrial-Mediated Apoptosis through Oxidative Stress in Human Cervical Cancer HeLa Cells. Life Sci. 2017, 181, 62–69. [Google Scholar] [CrossRef]
- Li, Z.; Dai, A.; Yang, M.; Chen, S.; Deng, Z.; Li, L. p38MAPK signaling pathway in osteoarthritis: Pathological and therapeutic aspects. J. Infl. Res. 2022, 15, 723–734. [Google Scholar] [CrossRef]
- Li, H.; Wang, D.; Yuan, Y.; Min, J. New insights on the MMP-13 regulatory network in the pathogenesis of early osteoarthritis. Arthritis Res. Ther. 2017, 19, 248. [Google Scholar] [CrossRef]
- Knäuper, V.; López-Otin, C.; Smith, B.; Knight, G.; Murphy, G. Biochemical characterization of human collagenase-3. J. Biol. Chem. 1996, 271, 1544–1550. [Google Scholar] [CrossRef] [PubMed]
- Knäuper, V.; Cowell, S.; Smith, B.; López-Otin, C.; O’Shea, M.; Morris, H.; Zardi, L.; Murphy, G. The role of the C-terminal domain of human collagenase-3 (MMP-13) in the activation of procollagenase-3, substrate specificity, and tissue inhibitor of metalloproteinase interaction. J. Biol. Chem. 1997, 272, 7608–7616. [Google Scholar] [CrossRef] [PubMed]
- Chou, L.W.; Wang, J.; Chang, P.L.; Hsieh, Y.L. Hyaluronan modulates accumulation of hypoxia-inducible factor-1 alpha, inducible nitric oxide synthase, and matrix metalloproteinase-3 in the synovium of rat adjuvant-induced arthritis model. Arthritis Res. Ther. 2011, 13, R90. [Google Scholar] [CrossRef]
- Porcello, A.; Chemali, M.; Marques, C.; Scaletta, C.; Lourenço, K.; Abdel-Sayed, P.; Raffoul, W.; Hirt-Burri, N.; Applegate, L.A.; Laurent, A. Dual Functionalization of Hyaluronan Dermal Fillers with Vitamin B3: Efficient Combination of Bio-Stimulation Properties with Hydrogel System Resilience Enhancement. Gels 2024, 10, 361. [Google Scholar] [CrossRef]
- Gosset, M.; Berenbaum, F.; Levy, A.; Pigenet, A.; Thirion, S.; Saffar, J.L.; Jacques, C. Prostaglandin E2 synthesis in cartilage explants under compression: mPGES-1 is a mechanosensitive gene. Arthritis Res. Ther. 2006, 8, R135. [Google Scholar] [CrossRef]
- Chisari, E.; Yaghmour, K.M.; Khan, W.S. The effects of TNF-alpha inhibition on cartilage: A systematic review of preclinical studies. Osteoarthr. Cartil. 2020, 28, 708–718. [Google Scholar] [CrossRef] [PubMed]
- Hu, Q.; Ecker, M. Overview of MMP-13 as a promising target for the treatment of osteoarthritis. Int. J. Mol. Sci. 2021, 22, 1742. [Google Scholar] [CrossRef]
- Samavedi, S.; Diaz-Rodriguez, P.; Erndt-Marino, J.D.; Hahn, M.S. A Three-Dimensional Chondrocyte–Macrophage Coculture System to Probe Inflammation in Experimental Osteoarthritis. Tissue Eng. Part A 2017, 23, 101–114. [Google Scholar] [CrossRef]






| Gene | Forward Primer Sequence | Reverse Primer Sequence |
|---|---|---|
| Type II collagen | TCCCTCTGGTTCTGATGGTC | CTCTGTCTCCAGATGCACCA |
| Aggrecan | ACACCCCTACCCTTGCTTCT | AAAGTGTCCAAGGCATCCAC |
| MMP-13 | CTGACCTGGGATTTCCAAAA | ACACGTGGTTCCCTGAGAAG |
| RPL19 | TGCCGGAAGAACACCTTG | GCAGGATCCTCATCCTTCG |
| β-actin | CCAACCGTGAAAAGATGACC | ACCAGAGGCATACAGGGACA |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hadjab, F.; Antoine, S.; Hamel, B.; Benderdour, M.; Fahmi, H.; Porcello, A.; Philippe, V.; Martin, R.; Marques, C.; Lourenço, K.; et al. Preclinical Mechanistic Evaluation of Hyaluronan/Niacinamide (Vitamin B3) Hydrogels: Toward an Enhanced Viscosupplement System with Ancillary Anti-Arthritic Attributes. Bioengineering 2025, 12, 1246. https://doi.org/10.3390/bioengineering12111246
Hadjab F, Antoine S, Hamel B, Benderdour M, Fahmi H, Porcello A, Philippe V, Martin R, Marques C, Lourenço K, et al. Preclinical Mechanistic Evaluation of Hyaluronan/Niacinamide (Vitamin B3) Hydrogels: Toward an Enhanced Viscosupplement System with Ancillary Anti-Arthritic Attributes. Bioengineering. 2025; 12(11):1246. https://doi.org/10.3390/bioengineering12111246
Chicago/Turabian StyleHadjab, Farid, Stivens Antoine, Béatrice Hamel, Mohamed Benderdour, Hassan Fahmi, Alexandre Porcello, Virginie Philippe, Robin Martin, Cíntia Marques, Kelly Lourenço, and et al. 2025. "Preclinical Mechanistic Evaluation of Hyaluronan/Niacinamide (Vitamin B3) Hydrogels: Toward an Enhanced Viscosupplement System with Ancillary Anti-Arthritic Attributes" Bioengineering 12, no. 11: 1246. https://doi.org/10.3390/bioengineering12111246
APA StyleHadjab, F., Antoine, S., Hamel, B., Benderdour, M., Fahmi, H., Porcello, A., Philippe, V., Martin, R., Marques, C., Lourenço, K., Scaletta, C., Hirt-Burri, N., Abdel-Sayed, P., Applegate, L. A., & Laurent, A. E. (2025). Preclinical Mechanistic Evaluation of Hyaluronan/Niacinamide (Vitamin B3) Hydrogels: Toward an Enhanced Viscosupplement System with Ancillary Anti-Arthritic Attributes. Bioengineering, 12(11), 1246. https://doi.org/10.3390/bioengineering12111246










