The In Vivo Antioxidant and Hepatoprotective Actions of Selected Chinese Teas
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Preparation of Tea Extracts
2.3. Animal Study and Sample Preparation
2.4. Measurement of Hepatic Injury Biochemical Markers in the Serum
2.5. Measurement of Antioxidant Biochemical Markers in the Liver
2.6. Measurement of Lipid Peroxidation Levels in the Liver
2.7. Histopathologic Assessment of Liver
2.8. Measurement of Antioxidant Capacity and Total Phenolic Content
2.9. Measurement of Phytochemicals in Teas
2.10. Statistical Analysis
3. Results and Discussion
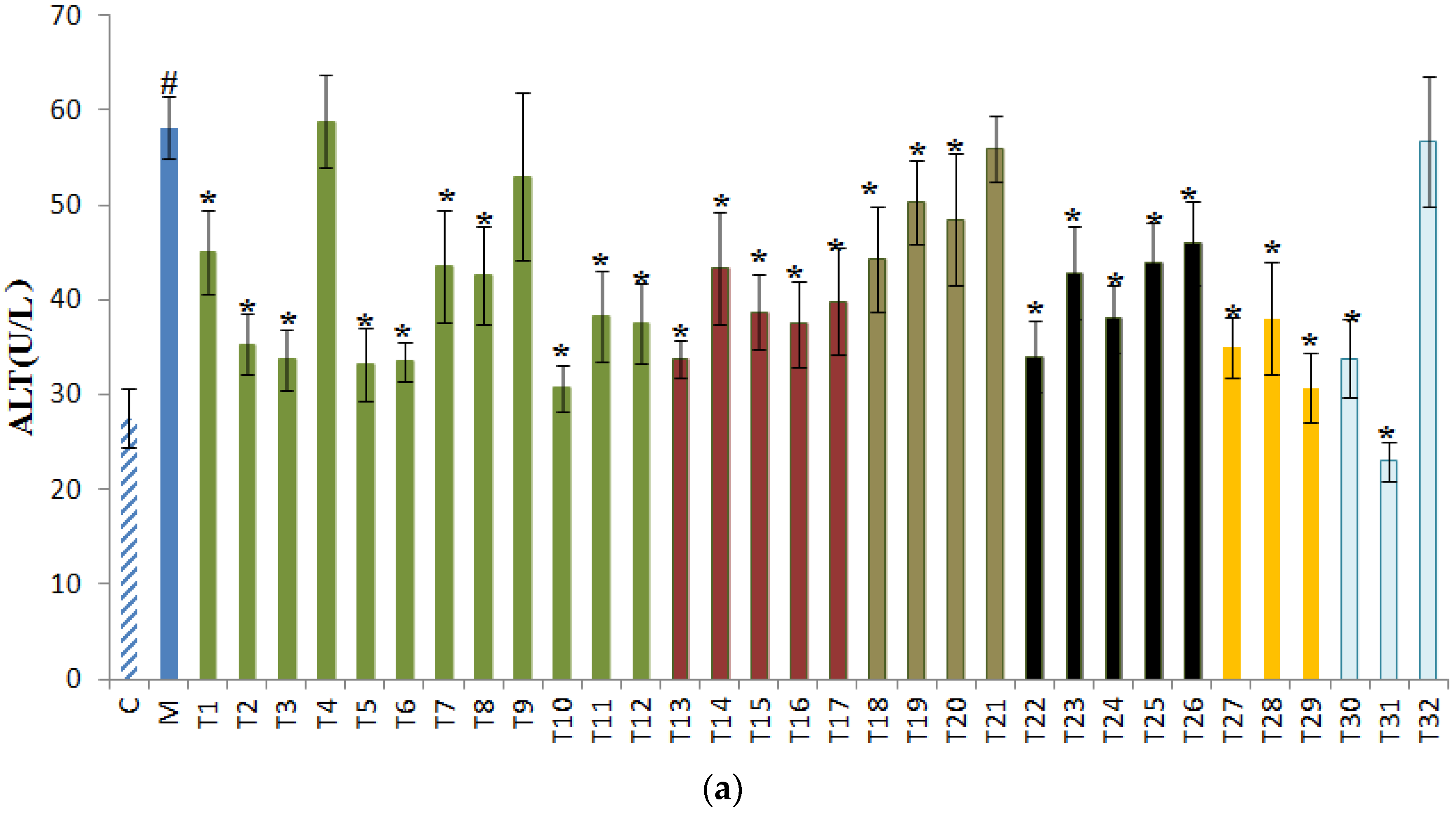
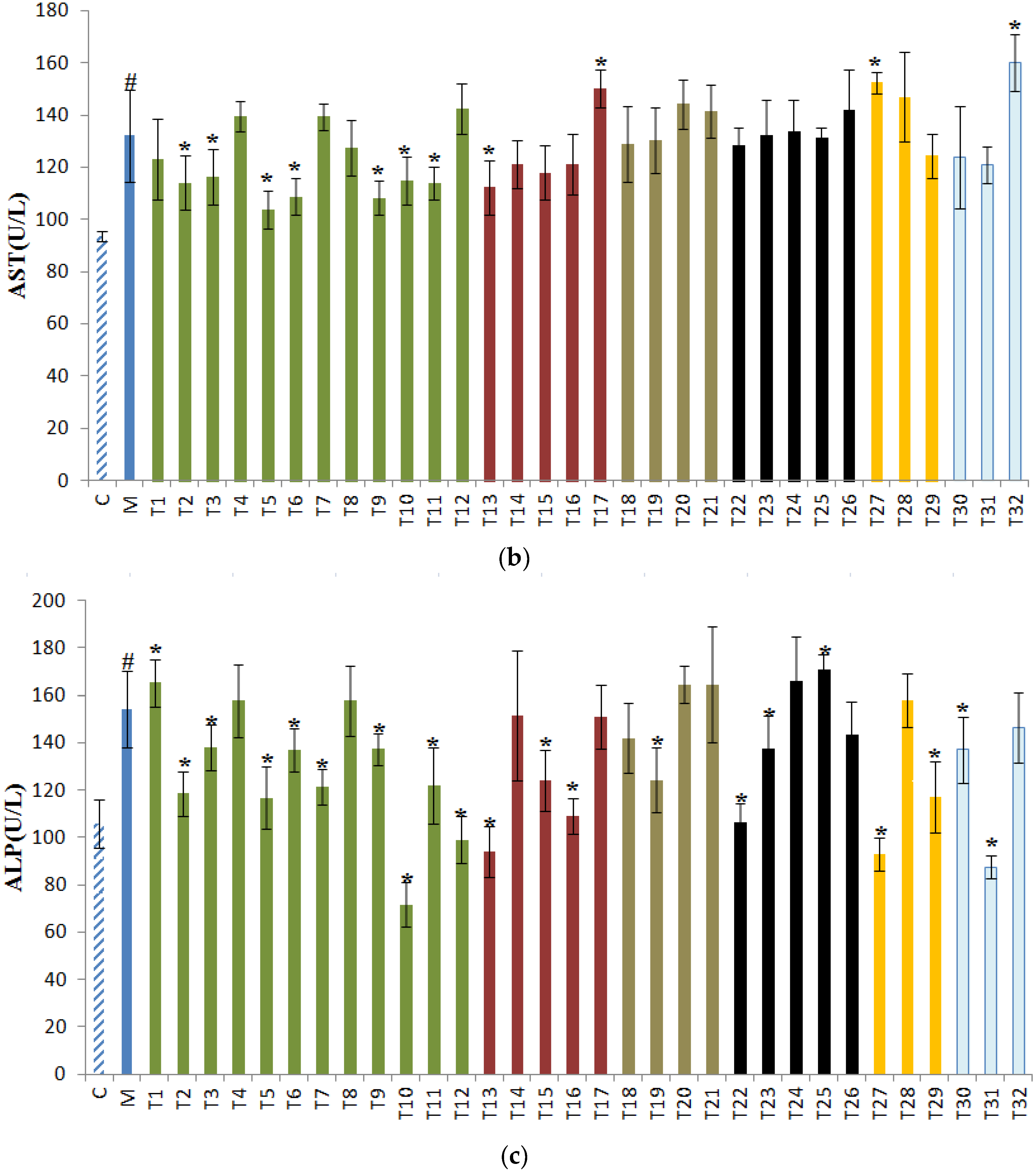
3.1. Effects of Tea on ALT, AST, and ALP in Sera
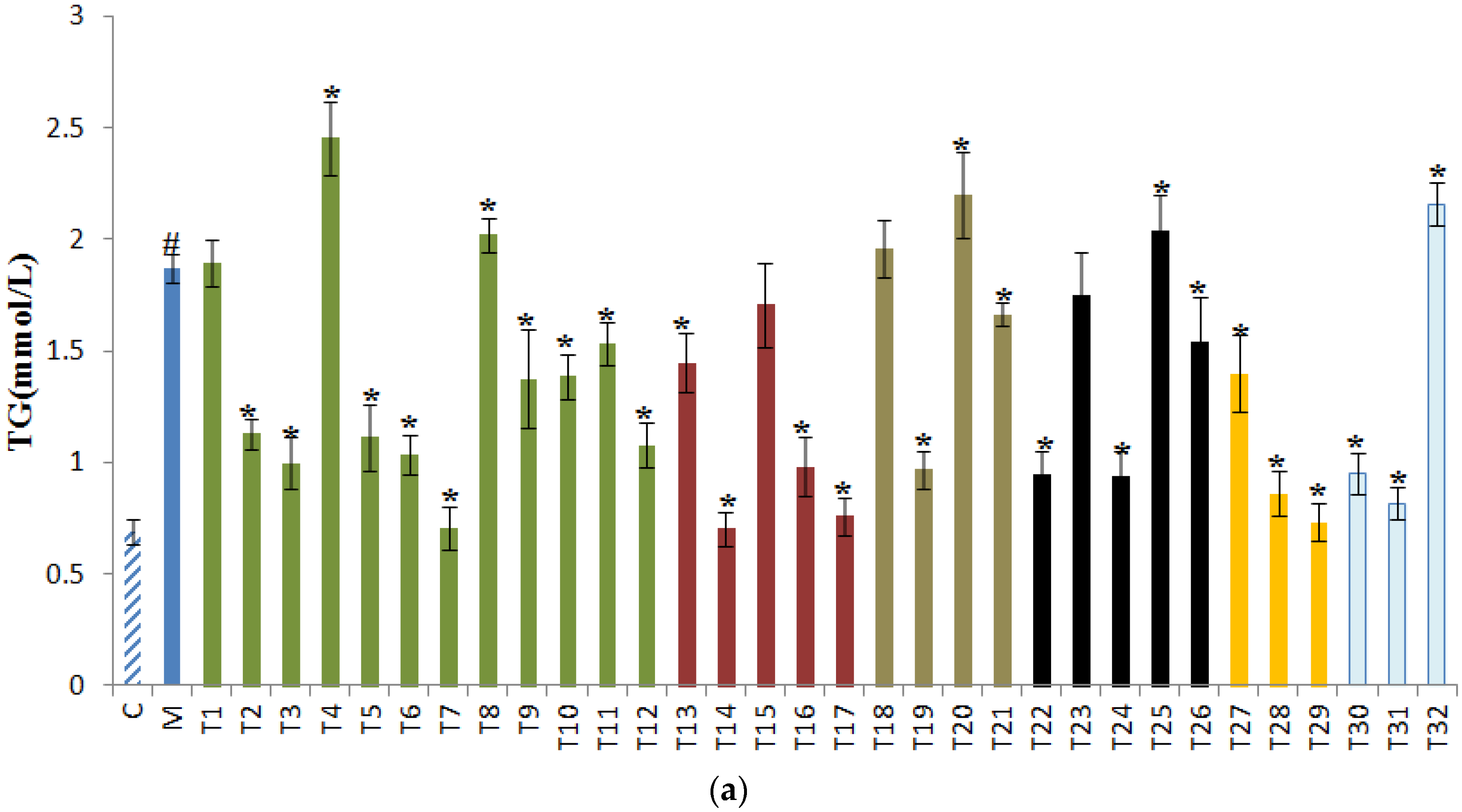
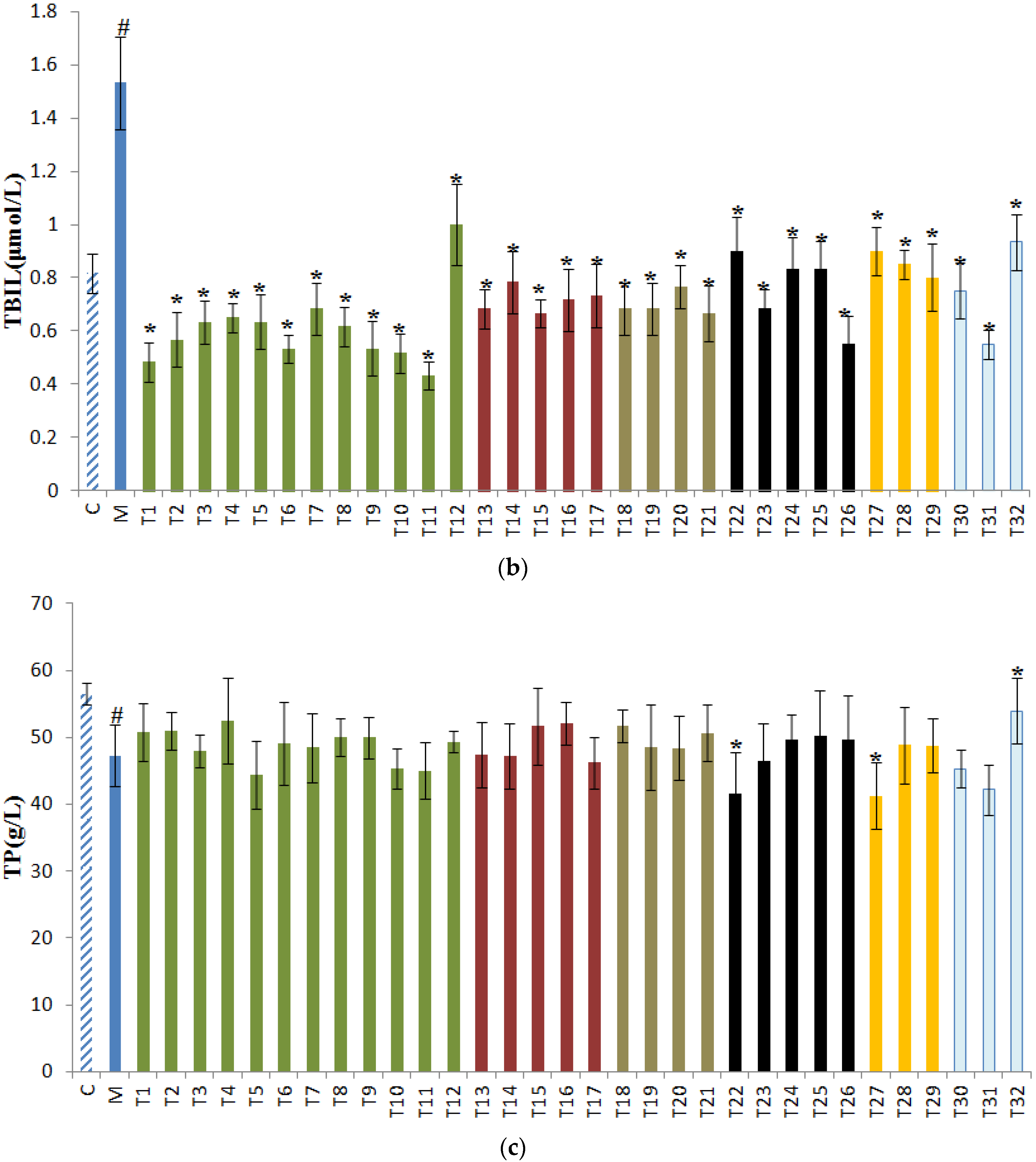
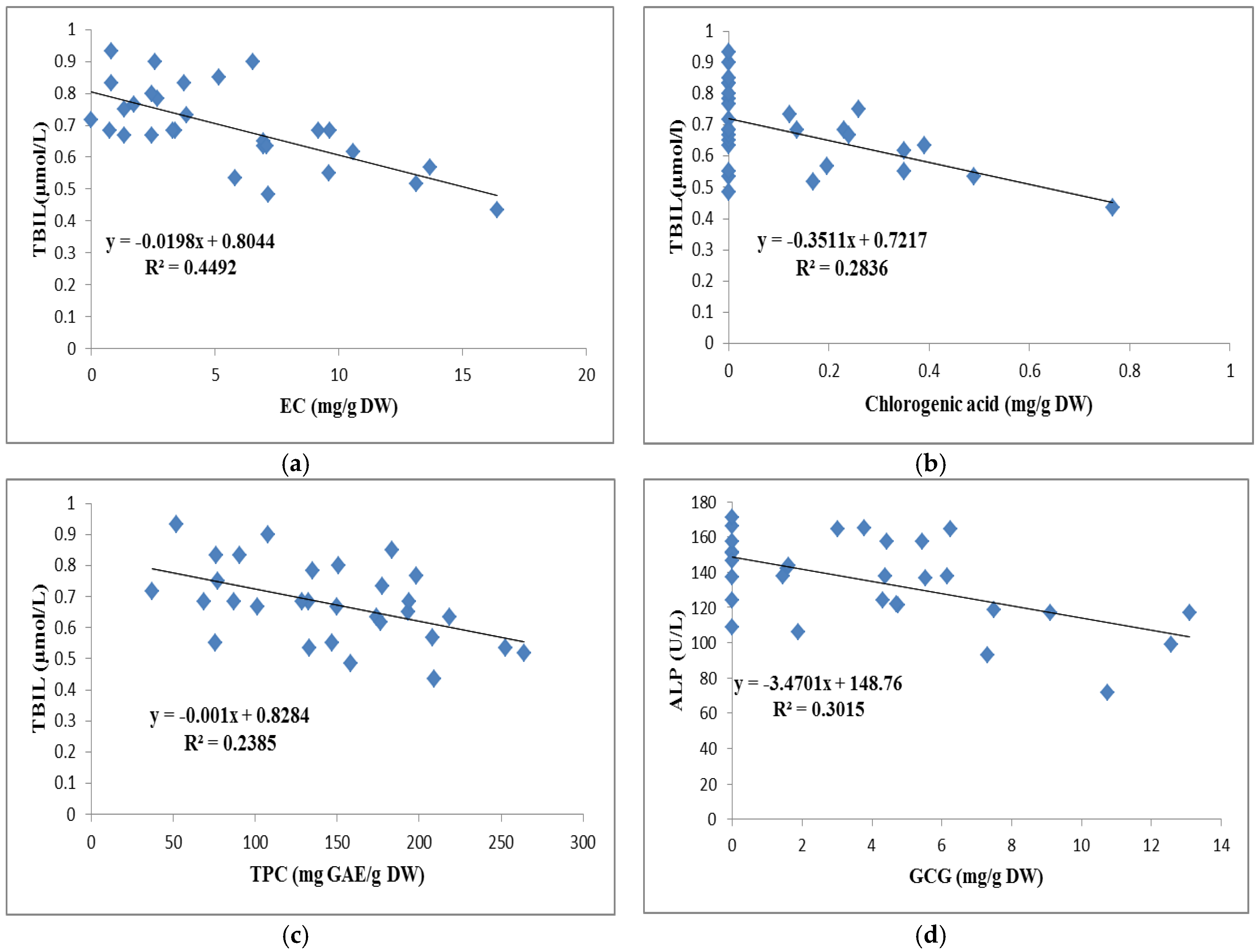
3.2. Effects of Tea on TG, TBIL, and TP in Sera
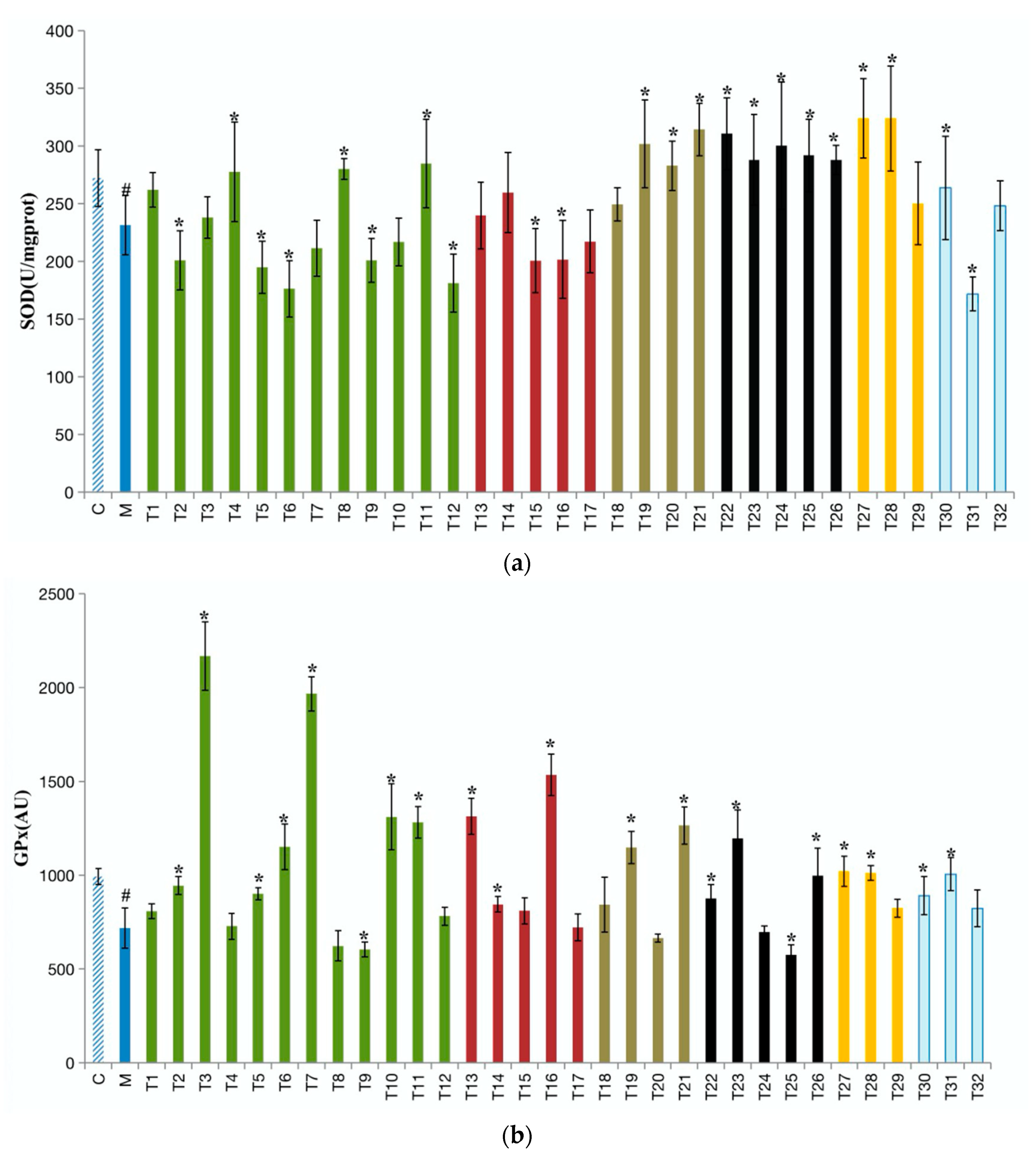
3.3. Effects of 32 Teas on Antioxidant Biochemical Markers in the Liver
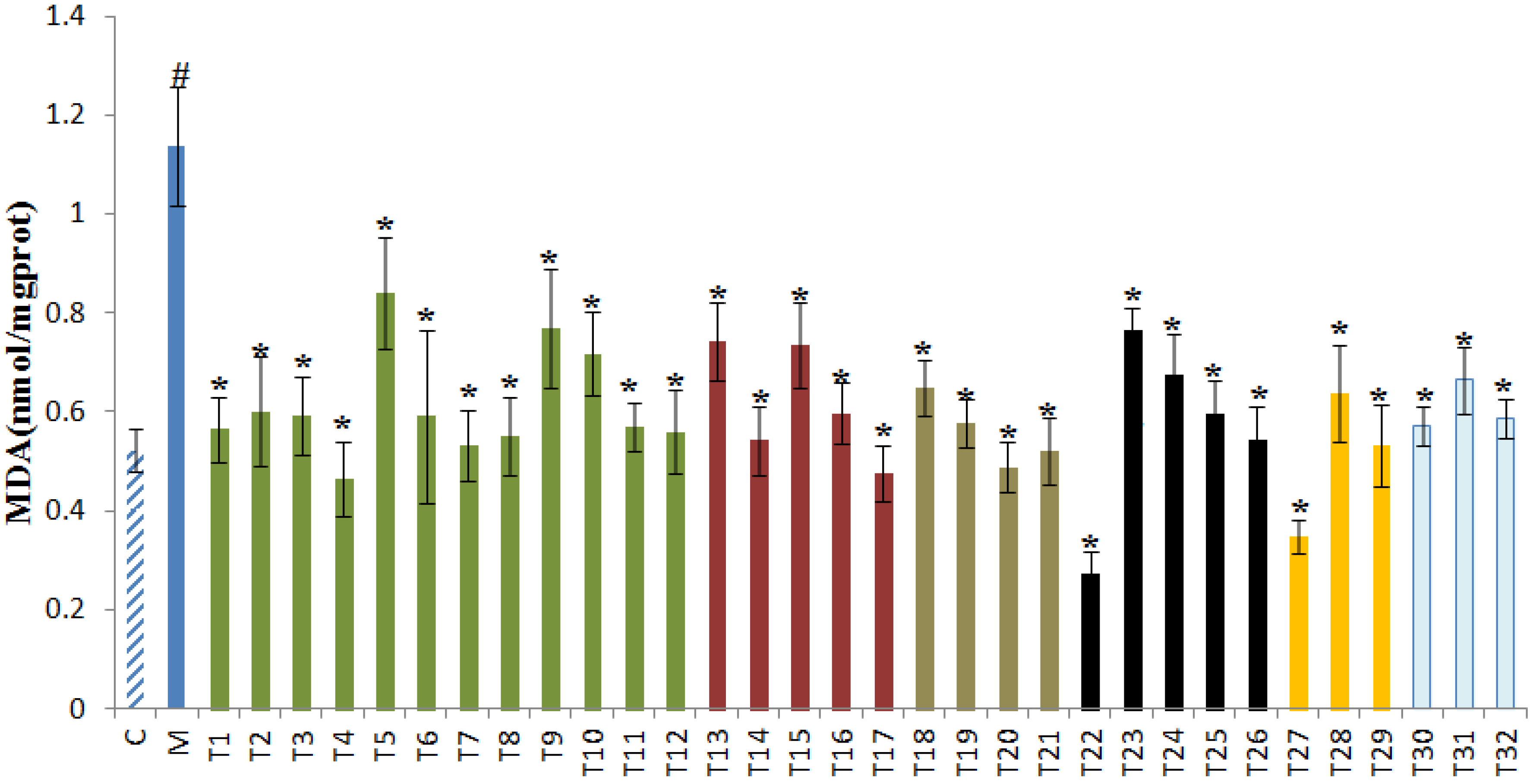
3.4. Effects of 32 Teas on Lipid Peroxidation Levels in the Liver
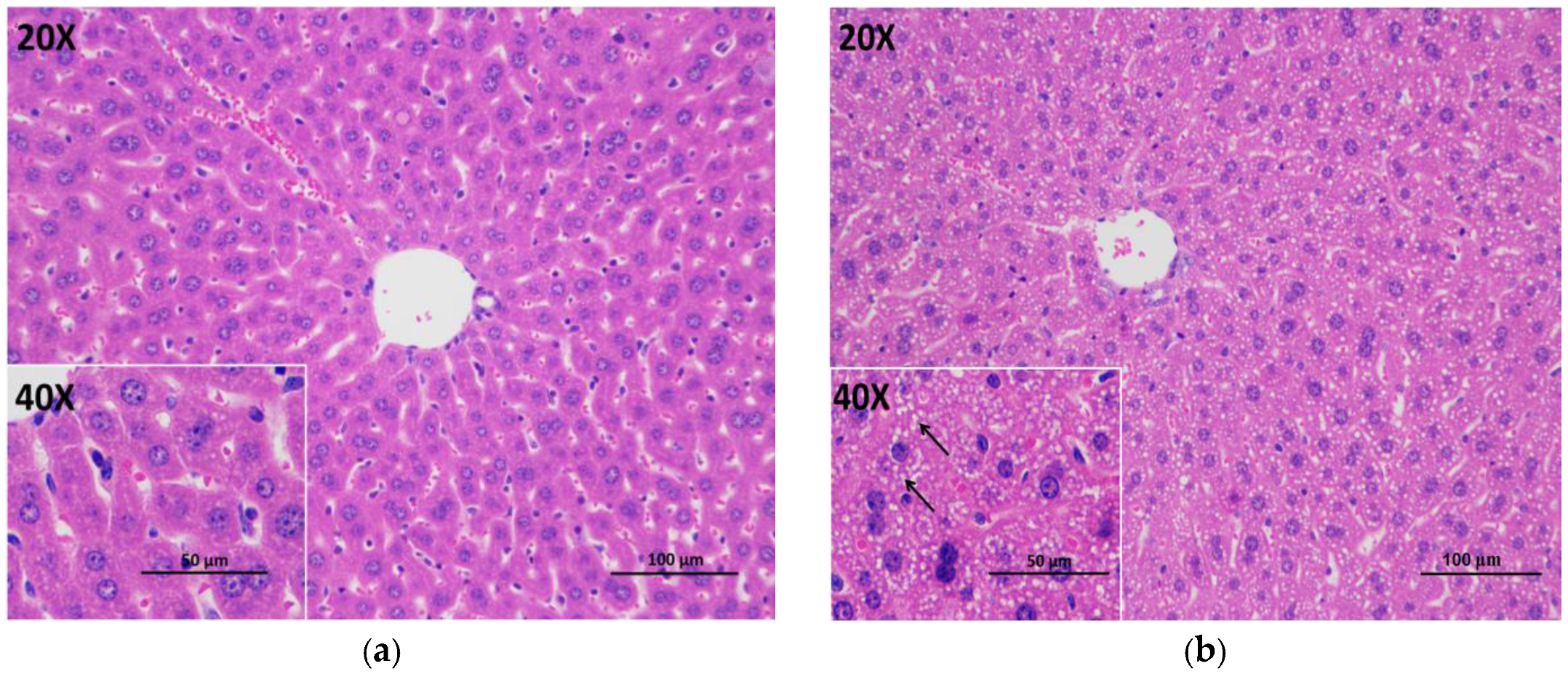
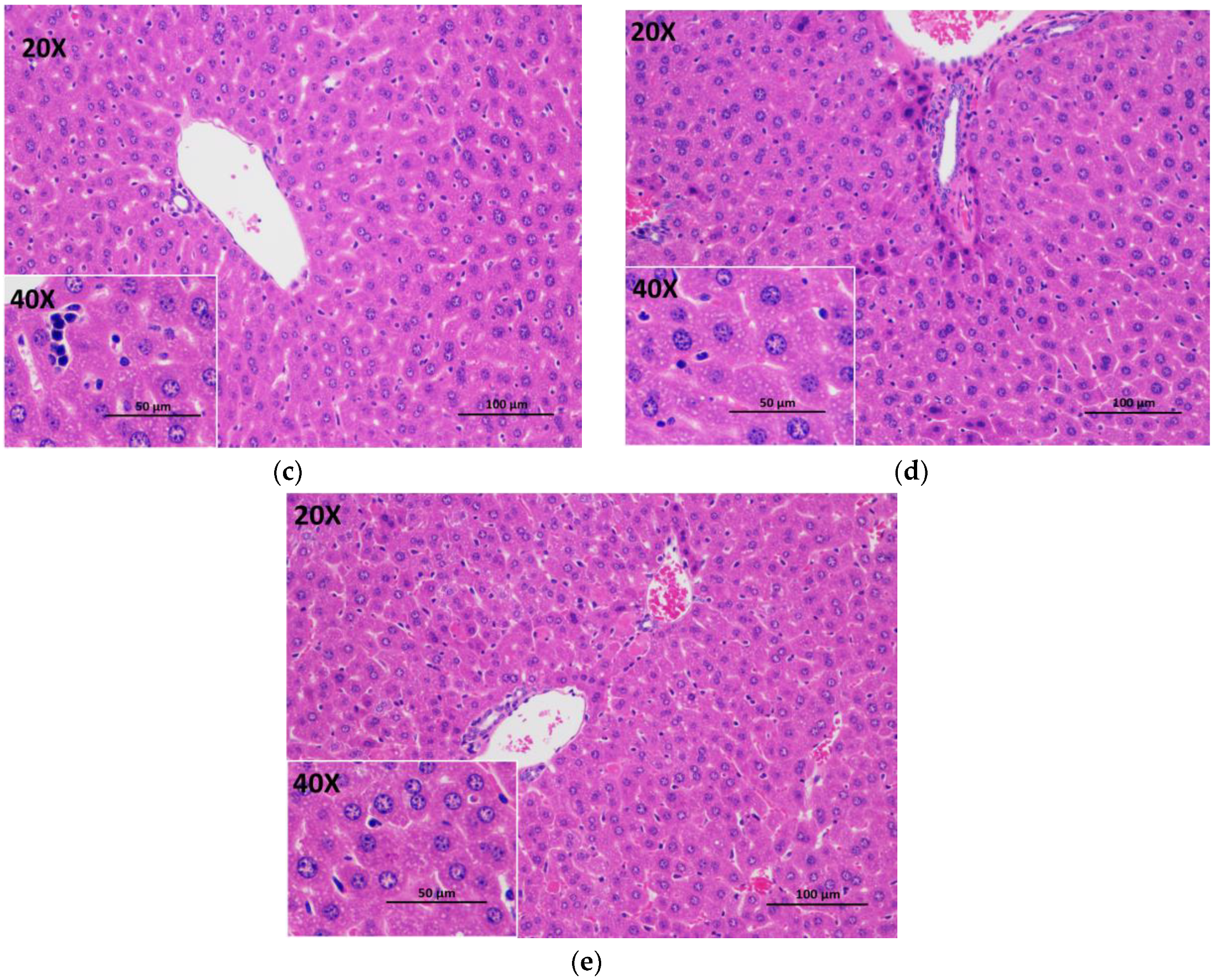
3.5. Histopathological Evaluation
3.6. Systematic Cluster of SOD and MDA Values of 32 Teas
3.7. Phenolic Compounds in Teas
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Finkel, T.; Holbrook, N.J. Oxidants, oxidative stress and the biology of ageing. Nature 2000, 9, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Gan, L.Q.; Li, S.K.; Zheng, J.C.; Xu, D.P.; Li, H.B. Effects of herbal infusions, tea and carbonated beverages on alcohol dehydrogenase and aldehyde dehydrogenase activity. Food Funct. 2014, 5, 42–49. [Google Scholar] [CrossRef] [PubMed]
- WHO/Health Topics/Alcohol. Available online: https://www.who.int/health-topics/alcohol#tab=tab_1 (accessed on 23 February 2020).
- Zhang, Y.J.; Wang, F.; Zhou, Y.; Li, Y.; Zhou, T.; Zheng, J.; Zhang, J.J.; Li, S.; Xu, D.P.; Li, H.B. Effects of 20 selected fruits on ethanol metabolism: Potential health benefits and harmful impacts. Int. J. Environ. Res. Public. Health 2016, 13, 399. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.W.; Jiang, Y.; Zhang, D.Y.; Wang, M.; Chen, W.S.; Su, H.X.; Wang, Y.T.; Wan, J.B. Protective effects of Penthorum chinense Pursh against chronic ethanol-induced liver injury in mice. J. Ethnopharmacol. 2015, 161, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Tan, H.Y.; Wang, N.; Zhang, Z.J.; Lao, L.X.; Wong, C.W.; Feng, Y.B. The role of oxidative stress and antioxidants in liver diseases. Int. J. Mol. Sci. 2015, 16, 26087–26124. [Google Scholar] [CrossRef]
- Li, A.N.; Li, S.; Zhang, Y.J.; Xu, X.R.; Chen, Y.M.; Li, H.B. Resources and biological activities of natural polyphenols. Nutrients 2014, 6, 6020–6047. [Google Scholar] [CrossRef]
- Liu, J.X.; Liu, S.W.; Zhou, H.M.; Hanson, T.; Yang, L.; Chen, Z.M.; Zhou, M.G. Association of green tea consumption with mortality from all-cause, cardiovascular disease and cancer in a Chinese cohort of 165,000 adult men. Eur. J. Epidemiol. 2016, 31, 853–865. [Google Scholar] [CrossRef]
- Meng, X.; Li, Y.; Li, S.; Gan, R.Y.; Li, H.B. Natural products for prevention and treatment of chemical-induced liver injuries. Compr. Rev. Food Sci. Food Saf. 2018, 17, 472–495. [Google Scholar] [CrossRef]
- Acquaviva, R.; Sorrenti, V.; Santangelo, R.; Cardile, V.; Tomasello, B.; Malfa, G.; Vanella, L.; Amodeo, A.; Genovese, C.; Mastrojeni, S.; et al. Effects of an extract of Celtis aetnensis (Tornab.) Strobl twigs on human colon cancer cell cultures. Oncol. Rep. 2016, 36, 2298–2304. [Google Scholar] [CrossRef]
- Tomasello, B.; Antonio, G.; La Mantia, A.; Miceli, N.; Sferrazzo, G.; Fernanda, T.M.; Di Giacomo, C.; Renis, M.; Acquaviva, R. Anti-adipogenic and anti-oxidant effects of a standardised extract of Moro blood oranges (Citrus sinensis (L.) Osbeck) during adipocyte differentiation of 3T3-L1 preadipocytes. Nat. Prod. Res. 2019, in press. [Google Scholar] [CrossRef]
- Cao, S.Y.; Zhao, C.N.; Gan, R.Y.; Xu, X.Y.; Wei, X.L.; Corke, H.; Atanasov, A.G.; Li, H.B. Effects and mechanisms of tea and its bioactive compounds for the prevention and treatment of cardiovascular diseases: An updated review. Antioxidants 2019, 8, 166. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.Y.; Zhao, C.N.; Cao, S.Y.; Tang, G.Y.; Gan, R.Y.; Li, H.B. Effects and mechanisms of tea for the prevention and management of cancers: An updated review. Crit. Rev. Food Sci. Nutr. 2020, in press. [Google Scholar] [CrossRef] [PubMed]
- Meng, J.M.; Cao, S.Y.; Wei, X.L.; Gan, R.Y.; Wang, Y.F.; Cai, S.X.; Xu, X.Y.; Zhang, P.Z.; Li, H.B. Effects and mechanisms of tea for the prevention and management of diabetes mellitus and diabetic complications: An updated review. Antioxidants 2019, 8, 170. [Google Scholar] [CrossRef]
- Tang, G.Y.; Zhao, C.N.; Xu, X.Y.; Gan, R.Y.; Cao, S.Y.; Liu, Q.; Shang, A.; Mao, Q.Q.; Li, H.B. Phytochemical composition and antioxidant capacity of 30 Chinese teas. Antioxidants 2019, 8, 180. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.N.; Tang, G.Y.; Cao, S.Y.; Xu, X.Y.; Gan, R.Y.; Liu, Q.; Mao, Q.Q.; Shang, A.; Li, H.B. Phenolic profiles and antioxidant activities of 30 tea infusions from green, black, oolong, white, yellow and dark teas. Antioxidants 2019, 8, 215. [Google Scholar] [CrossRef]
- Augustyniak, A.; Waszkiewicz, E.; Skrzydlewska, E. Preventive action of green tea from changes in the liver antioxidant abilities of different aged rats intoxicated with ethanol. Nutrition 2005, 21, 925–932. [Google Scholar] [CrossRef]
- Chen, K.H.; Li, P.C.; Lin, W.H.; Chien, C.T.; Low, B.H. Depression by a green tea extract of alcohol-induced oxidative stress and lipogenesis in rat liver. Biosci. Biotechnol. Biochem. 2011, 75, 1668–1676. [Google Scholar] [CrossRef]
- Kaviarasan, S.; Sundarapandiyan, R.; Anuradha, C.V. Epigallocatechin gallate, a green tea phytochemical, attenuates alcohol-induced hepatic protein and lipid damage. Toxicol. Mech. Methods 2008, 18, 645–652. [Google Scholar] [CrossRef]
- Luczaj, W.; Skrzydlewska, E. Antioxidant properties of black tea in alcohol intoxication. Food Chem. Toxicol. 2004, 42, 2045–2051. [Google Scholar] [CrossRef]
- Ghiselli, A.; Serafini, M.; Natella, F.; Scaccini, C. Total antioxidant capacity as a tool to assess redox status: Critical view and experimental data. Free Radic. Biol. Med. 2000, 29, 1106–1114. [Google Scholar] [CrossRef]
- Xu, X.Y.; Zheng, J.; Meng, J.M.; Gan, R.Y.; Mao, Q.Q.; Shang, A.; Li, B.Y.; Wei, X.L.; Li, H.B. Effects of food processing on in vivo antioxidant and hepatoprotective properties of green tea extracts. Antioxidants 2019, 8, 572. [Google Scholar] [CrossRef] [PubMed]
- Al-Basher, G.I. Green tea activity and iron overload induced molecular fibrogenesis of rat liver. Saudi. J. Biol. Sci. 2019, 26, 531–540. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.; Schimidt, H.L.; Garcia, A.; Altermann, C.D.C.; Santos, F.W.; Carpes, F.P.; Da Silva, W.C.; MelloCarpes, P.B. Supplementation with different teas from Camellia sinensis prevents memory deficits and hippocampus oxidative stress in ischemia-reperfusion. Neurochem. Int. 2017, 108, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Seo, D.B.; Jeong, H.W.; Kim, Y.J.; Kim, S.; Kim, J.; Lee, J.H.; Joo, K.; Choi, J.K.; Shin, S.S.; Lee, S.J. Fermented green tea extract exhibits hypolipidaemic effects through the inhibition of pancreatic lipase and promotion of energy expenditure. Brit. J. Nutr. 2017, 117, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.; Zhang, Y.J.; Xu, D.P.; Wang, F.; Zhou, Y.; Zheng, J.; Li, Y.; Zhang, J.J.; Li, H.B. Protective effects of lemon juice on alcohol-induced liver injury in mice. Biomed. Res. Int. 2017, 2017, 7463571. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “Antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radi. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventos, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin–Ciocalteu reagent. In Oxidants and Antioxidants; Packer, L., Ed.; Elsevier Academic Press Inc.: San Diego, CA, USA, 1999; Volume 299, pp. 152–178. [Google Scholar]
- Cai, Z.Y.; Li, X.M.; Liang, J.P.; Xiang, L.P.; Wang, K.R.; Shi, Y.L.; Yang, R.; Shi, M.; Ye, J.H.; Lu, J.L.; et al. Bioavailability of tea catechins and its improvement. Molecules 2018, 23, 2346. [Google Scholar] [CrossRef]
- Wang, F.; Zhang, Y.J.; Zhou, Y.; Li, Y.; Zhou, T.; Zheng, J.; Zhang, J.J.; Li, S.; Xu, D.P.; Li, H.B. Effects of beverages on alcohol metabolism: Potential health benefits and harmful impacts. Int. J. Mol. Sci. 2016, 17, 354. [Google Scholar] [CrossRef]
- Yu, F.; Li, H.; Meng, Y.; Yang, D. Extraction optimization of Angelica sinensis polysaccharides and its antioxidant activity in vivo. Carbohydr. Polym. 2013, 94, 114–119. [Google Scholar] [CrossRef]
- Ali, N.M.; Yusof, H.M.; Long, K.; Yeap, S.K.; Ho, W.Y.; Beh, B.K.; Koh, S.P.; Abdullah, M.P.; Alitheen, N.B. Antioxidant and hepatoprotective effect of aqueous extract of germinated and fermented mung bean on ethanol-mediated liver damage. Biomed. Res. Int. 2013, 2013, 693613. [Google Scholar]
- Xu, Q.; Lu, Z.H.; Zhang, X.M. A novel role of alkaline phosphatase in protection from immunological liver injury in mice. Liver 2002, 22, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Zhang, N.; Shen, X.; Mei, W.; He, Y.; Ge, W. Preparation of total flavonoids from loquat flower and its protective effect on acute alcohol-induced liver injury in mice. J. Food Drug Anal. 2015, 23, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Granato, A.; Gores, G.; Vilei, M.T.; Tolando, R.; Ferraresso, C.; Muraca, M. Bilirubin inhibits bile acid induced apoptosis in rat hepatocytes. Gut 2003, 52, 1774–1778. [Google Scholar] [CrossRef] [PubMed]
- Karmakar, S.; Das, D.; Maiti, A.; Majumdar, S.; Mukherjee, P.; Das, A.S.; Mitra, C. Black tea prevents high fat diet-induced non-alcoholic steatohepatitis. Phytother. Res. 2011, 25, 1073–1081. [Google Scholar] [CrossRef] [PubMed]
- Nwokocha, C.R.; Nwokocha, M.; Mounmbegna, P.; Orhue, J.; Onyezuligbo, O.; Olu-Osifo, E.H.; Okojie, E.; Asuquo, E.; Ejimofor, T.; Ikenna, N.; et al. Proteins and liver function changes in rats following cumulative total body irradiations. West Indian Med. J. 2012, 61, 773–777. [Google Scholar]
- Jurczuk, M.; Brzoska, M.M.; Moniuszko-Jakoniuk, J.; Galazyn-Sidorczuk, M.; Kulikowska-Karpinska, E. Antioxidant enzymes activity and lipid peroxidation in liver and kidney of rats exposed to cadmium and ethanol. Food Chem. Toxicol. 2004, 42, 429–438. [Google Scholar] [CrossRef]
- Wang, M.; Zhu, P.; Jiang, C.; Ma, L.; Zhang, Z.; Zeng, X. Preliminary characterization, antioxidant activity in vitro and hepatoprotective effect on acute alcohol-induced liver injury in mice of polysaccharides from the peduncles of Hovenia dulcis. Food Chem. Toxicol. 2012, 50, 2964–2970. [Google Scholar] [CrossRef]
- Reddy, A.C.; Lokesh, B.R. Studies on spice principles as antioxidants in the inhibition of lipid peroxidation of rat liver microsomes. Mol. Cell Biochem. 1992, 111, 117–124. [Google Scholar]
- Zhang, Y.J.; Zhou, T.; Wang, F.; Zhou, Y.; Li, Y.; Zhang, J.J.; Zheng, J.; Xu, D.P.; Li, H.B. The effects of Syzygium samarangense, Passiflora edulis and Solanum muricatum on alcohol-induced liver injury. Int. J. Mol. Sci. 2016, 17, 1616. [Google Scholar] [CrossRef]
- Jiang, H.; Yu, F.; Qin, L.; Zhang, N.; Cao, Q.; Schwab, W.; Li, D.X.; Song, C.K. Dynamic change in amino acids, catechins, alkaloids, and gallic acid in six types of tea processed from the same batch of fresh tea (Camellia sinensis L.) leaves. J. Food Compos. Anal. 2019, 77, 28–38. [Google Scholar] [CrossRef]
- Zhao, M.; Du, Y.Q.; Yuan, L.; Wang, N.N. Protective effect of puerarin on acute alcoholic liver injury. Am. J. Chin. Med. 2010, 38, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.Y.; Hung, T.W.; Wang, C.J.; Tseng, T.H. Inhibitory effect of Nelumbo nucifera leaf extract on 2-acetylaminofluorene-induced hepatocarcinogenesis through enhancing antioxidative potential and alleviating inflammation in rats. Antioxidants 2019, 8, 329. [Google Scholar] [CrossRef] [PubMed]
- Kalender, Y.; Kaya, S.; Durak, D.; Uzun, F.G.; Demir, F. Protective effects of catechin and quercetin on antioxidant status, lipid peroxidation and testis-histoarchitecture induced by chlorpyrifos in male rats. Environ. Toxicol. Pharmacol. 2012, 33, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Surapaneni, K.M.; Jainu, M. Comparative effect of pioglitazone, quercetin and hydroxy citric acid on the status of lipid peroxidation and antioxidants in experimental non-alcoholic steatohepatitis. J. Physiol. Pharmacol. 2014, 65, 67–74. [Google Scholar] [PubMed]
- Baltaziak, M.; Skrzydlewska, E.; Sulik, A.; Famulski, W.; Koda, M. Green tea as an antioxidant which protects against alcohol induced injury in rats—A histopathological examination. Folia Morphol. (Warsz) 2004, 63, 123–126. [Google Scholar]
- Gong, J.; Peng, C.; Chen, T.; Gao, B.; Zhou, H. Effects of theabrownin from Pu-erh tea on the metabolism of serum lipids in rats: Mechanism of action. J. Food. Sci. 2010, 75, 182–189. [Google Scholar] [CrossRef]
- Friedman, M. Overview of antibacterial, antitoxin, antiviral, and antifungal activities of tea flavonoids and teas. Mol. Nutr. Food Res. 2007, 51, 116–134. [Google Scholar] [CrossRef]


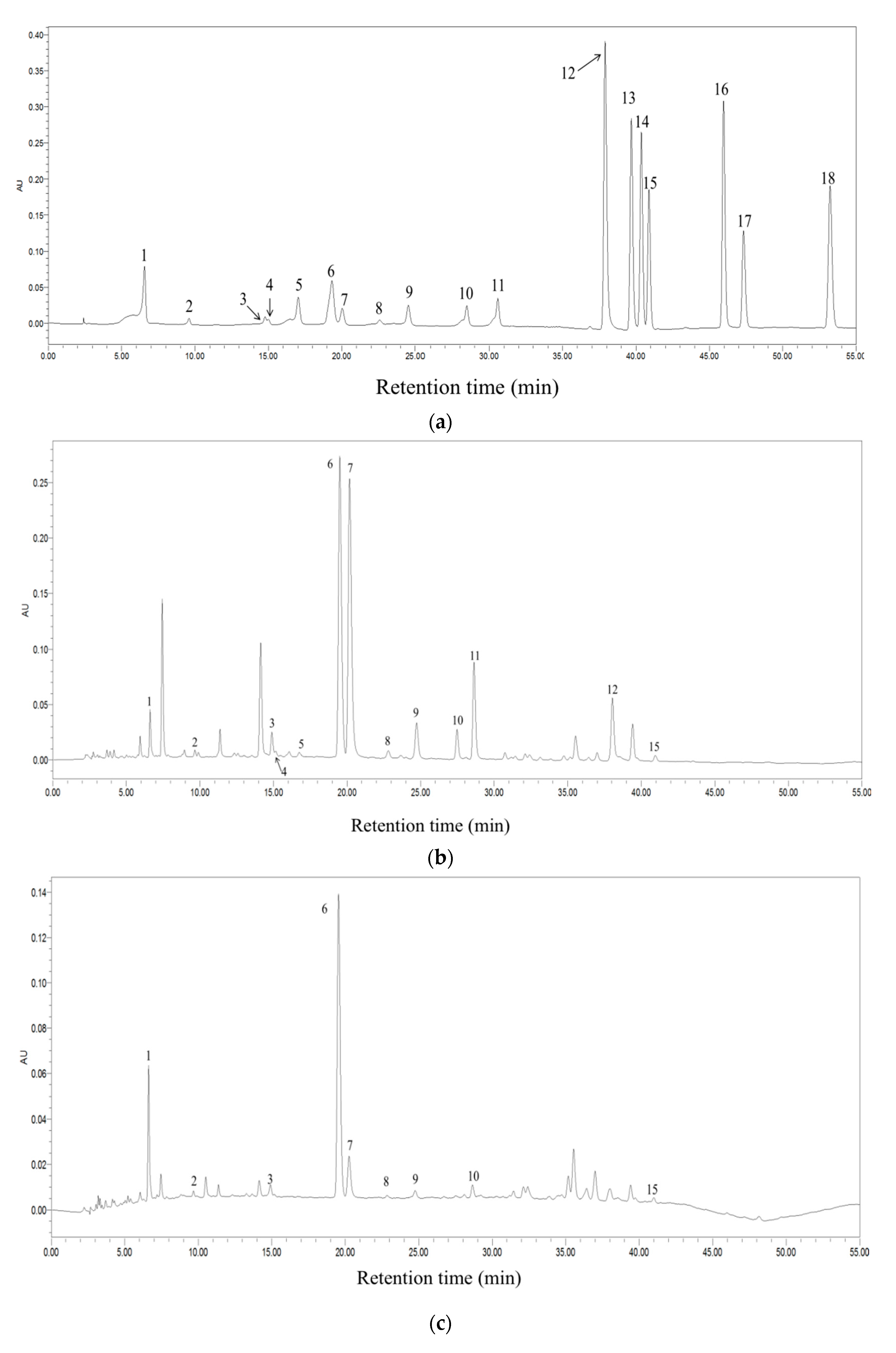
| No. | Name | Category | Fermentation Degree | Production Place |
|---|---|---|---|---|
| T1 | Chaoqing Green Tea | Green Tea | Unfermented | Yichang, Hubei |
| T2 | Selenium-Enriched Chaoqing Green Tea | Green Tea | Unfermented | Enshi, Hubei |
| T3 | Enshi Yulu Tea | Green Tea | Unfermented | Enshi, Hubei |
| T4 | Selenium-Enriched Matcha | Green Tea | Unfermented | Enshi, Hubei |
| T5 | Xihu Longjing Tea | Green Tea | Unfermented | Hangzhou, Zhejiang |
| T6 | Matcha | Green Tea | Unfermented | Shaoxing, Zhejiang |
| T7 | Fried Green Tea | Green Tea | Unfermented | Shaoxing, Zhejiang |
| T8 | Taiping Houkui Tea | Green Tea | Unfermented | Huangshan, Anhui |
| T9 | Dianqing Tea | Green Tea | Unfermented | Kunming, Yunnan |
| T10 | Liping Xiang Tea | Green Tea | Unfermented | Liping, Guizhou |
| T11 | Jieyang Chaoqing Tea | Green Tea | Unfermented | Jieyang, Guangdong |
| T12 | Fenggang Zinc-Selenium-enriched Tea | Green Tea | Unfermented | Guiyang, Guizhou |
| T13 | Yihong Tea | Black Tea | Deep-fermented | Yichang, Hubei |
| T14 | Selenium-Enriched Black Tea | Black Tea | Deep-fermented | Enshi, Hubei |
| T15 | Dianhong Tea | Black Tea | Deep-fermented | Xishuangbanna, Yunnan |
| T16 | Lapsang Souchong Tea | Black Tea | Deep-fermented | Xiamen, Fujian |
| T17 | Yingde Black Tea | Black Tea | Deep-fermented | Yingde, Guangdong |
| T18 | Tieguanyin Tea | Oolong Tea | Semi-fermented | Xiamen, Fujian |
| T19 | Wuyi Narcissus Tea | Oolong Tea | Semi-fermented | Wuyishan, Fujian |
| T20 | Fenghuang Danzong Tea | Oolong Tea | Semi-fermented | Shantou, Guangdong |
| T21 | Fenghuang Narcissus Tea | Oolong Tea | Semi-fermented | Shantou, Guangdong |
| T22 | Fu Brick Tea | Dark Tea | Post-fermented | Changsha, Hunan |
| T23 | Qing Brick Tea | Dark Tea | Post-fermented | Yichang, Hubei |
| T24 | Pu-erh Tea | Dark Tea | Post-fermented | Pu’er, Yunnan |
| T25 | Liupao Tea | Dark Tea | Post-fermented | Wuzhou, Guangxi |
| T26 | Black Fu Brick Tea | Dark Tea | Post-fermented | Enshi, Hubei |
| T27 | Yuan’an Luyuan Tea | Yellow Tea | Light-fermented | Yichang, Hubei |
| T28 | Mengding Huangya Tea | Yellow Tea | Light-fermented | Mengdingshan, Sichuan |
| T29 | Huoshan Yellow Tea | Yellow Tea | Light-fermented | Pu’er, Yunnan |
| T30 | White Peony Tea | White Tea | Mild-fermented | Fuzhou, Fujian |
| T31 | Gongmei White Tea | White Tea | Mild-fermented | Fuzhou, Fujian |
| T32 | Shoumei White Tea | White Tea | Mild-fermented | Fuzhou, Fujian |
| Ward Method | SOD | MDA | |
|---|---|---|---|
| 1 | Sum | 2010.79 | 4.78 |
| N | 8 | 8 | |
| Mean | 251.35 | 0.60 | |
| SD | 9.71 | 0.07 | |
| % of Total Sum | 25.0% | 25.3% | |
| % of Total N | 25.0% | 25.0% | |
| 2 | Sum | 2173.29 | 7.08 |
| N | 11 | 11 | |
| Mean | 197.57 | 0.64 | |
| SD | 15.5 | 0.11 | |
| % of Total Sum | 27.0% | 37.5% | |
| % of Total N | 34.4% | 34.4% | |
| 3 | Sum | 3867.98 | 7.01 |
| N | 13 | 13 | |
| Mean | 297.54 | 0.54 | |
| SD | 16.24 | 0.13 | |
| % of Total Sum | 48.0% | 37.1% | |
| % of Total N | 40.6% | 40.6% | |
| Total | Sum | 8052.07 | 18.87 |
| N | 32 | 32 | |
| Mean | 251.63 | 0.59 | |
| SD | 46.06 | 0.12 | |
| % of Total Sum | 100.0% | 100.0% | |
| % of Total N | 100.0% | 100.0% | |
| Name | Category | Gallocatechin | Epigallocatechin | Catechin | Epigallocatechin Gallate | Epicatechin | Gallocatechin Gallate | Epicatechin Gallate | Catechin Gallate |
| Chaoqing Green Tea | Green tea | 3.26 ± 0.3 | 8.02 ± 0.48 | - | 23.72 ± 1.91 | 7.17 ± 0.44 | 3.78 ± 0.27 | 2.64 ± 0.23 | 0.43 ± 0.03 |
| Selenium-Enriched Chaoqing Green Tea | Green tea | 5.24 ± 0.42 | 5.13 ± 0.37 | 3.52 ± 0.25 | 29.86 ± 2.46 | 13.70 ± 1.1 | 7.50 ± 0.62 | 24.99 ± 2.10 | - |
| Selenium-Enriched Matcha | Green tea | 3.85 ± 0.29 | 24.93 ± 0.51 | - | 27.56 ± 2.34 | 6.96 ± 0.46 | 4.41 ± 0.18 | 4.23 ± 0.33 | 0.51 ± 0.02 |
| Matcha | Green tea | 3.88 ± 0.19 | 11.07 ± 0.61 | - | 11.55 ± 1.04 | 3.60 ± 0.23 | 5.52 ± 0.25 | 7.64 ± 0.61 | 0.95 ± 0.07 |
| Fried Green Tea | Green tea | 7.22 ± 0.55 | 41.18 ± 2.58 | 1.01 ± 0.08 | 30.97 ± 2.93 | 9.64 ± 0.42 | 4.74 ± 0.41 | 16.92 ± 1.20 | - |
| Liping Xiang Tea | Green tea | 6.30 ± 0.62 | 29.58 ± 2.4 | 2.86 ± 0.26 | 35.80 ± 3.26 | 13.13 ± 1.02 | 10.75 ± 0.96 | 30.47 ± 2.89 | 1.51 ± 0.08 |
| Jieyang Chaoqing Tea | Green tea | 4.86 ± 0.37 | 19.77 ± 1.29 | 7.05 ± 0.35 | 20.86 ± 1.86 | 16.40 ± 1.34 | 4.70 ± 0.37 | 34.02 ± 2.46 | 1.46 ± 0.09 |
| Fenggang Zinc-Selenium-Enriched Tea | Green tea | 5.18 ± 0.48 | 26.03 ± 2.1 | 5.64 ± 0.22 | 56.30 ± 4.59 | 14.04 ± 1.24 | 12.56 ± 0.92 | 47.78 ± 3.39 | 3.01 ± 0.19 |
| Yihong Tea | Black tea | - | - | - | 1.17 ± 0.08 | 0.77 ± 0.02 | - | 4.57 ± 0.22 | - |
| Selenium-Enriched Black Tea | Black tea | - | - | - | 1.37 ± 0.1 | 2.67 ± 0.12 | - | - | |
| Yingde Black Tea | Black tea | - | - | 3.59 ± 0.24 | 1.02 ± 0.07 | 3.87 ± 0.23 | - | 13.03 ± 0.82 | - |
| Wuyi Narcissus Tea | Oolong tea | 9.84 ± 0.87 | 13.66 ± 1.12 | 4.11 ± 0.20 | 11.05 ± 1.07 | 3.39 ± 0.19 | 4.31 ± 00.39 | 7.12 ± 0.34 | - |
| Fenghuang Danzong Tea | Oolong tea | 3.72 ± 0.26 | 12.10 ± 1.01 | - | 41.24 ± 3.45 | 1.75 ± 0.1 | 6.26 ± 0.42 | 17.14 ± 0.94 | 2.44 ± 0.16 |
| Black Fu Brick Tea | Dark tea | 2.21 ± 0.12 | 6.56 ± 0.35 | - | 4.35 ± 0.34 | 2.58 ± 0.09 | 1.89 ± 0.1 | 3.18 ± 0.29 | - |
| Name | Category | Gallic Acid | Chlorogenic Acid | Caffeine | Ellagic Acid | Astragalin | Quercetin | Theaflavin | |
| Chaoqing Green Tea | Green tea | 0.75 ± 0.06 | - | 27.25 ± 1.69 | - | 0.38 ± 0.02 | - | - | |
| Selenium-Enriched Chaoqing Green Tea | Green tea | 1.26 ± 0.08 | 0.20 ± 0.01 | 24.82 ± 0.95 | 1.54 ± 0.14 | 0.72 ± 0.05 | - | - | |
| Selenium-Enriched Matcha | Green tea | 0.90 ± 0.07 | - | 28.51 ± 2.19 | - | 0.41 ± 0.02 | - | - | |
| Matcha | Green tea | 1.26 ± 0.11 | - | 26.16 ± 1.72 | - | - | - | - | |
| Fried Green Tea | Green tea | 0.87 ± 0.07 | 0.14 ± 0.01 | 22.83 ± 0.93 | - | 1.71 ± 0.14 | - | - | |
| Liping Xiang Tea | Green tea | 1.75 ± 0.1 | 0.17 ± 0.01 | 29.68 ± 1.45 | 2.08 ± 0.11 | 0.30 ± 0.01 | - | - | |
| Jieyang Chaoqing Tea | Green tea | 1.44 ± 0.06 | 0.77 ± 0.03 | 29.46 ± 1.82 | - | 1.21 ± 0.09 | - | - | |
| Fenggang Zinc-Selenium-Enriched Tea | Green tea | 2.27 ± 0.18 | 0.22 ± 0.02 | 35.99 ± 2.22 | 2.78 ± 0.17 | 2.11 ± 0.13 | - | - | |
| Yihong Tea | Black tea | 4.22 ± 0.28 | - | 31.68 ± 2.17 | 1.92 ± 0.13 | 0.98 ± 0.07 | - | 0.34 ± 0.02 | |
| Selenium-Enriched Black Tea | Black tea | 4.44 ± 0.21 | - | 29.42 ± 2.1 | 1.78 ± 0.16 | 1.39 ± 0.11 | - | 0.88 ± 0.07 | |
| Yingde Black Tea | Black tea | 2.92 ± 0.15 | 0.12 ± 0.01 | 31.17 ± 1.79 | - | - | 0.63 ± 0.05 | 0.42 ± 0.02 | |
| Wuyi Narcissus Tea | Oolong tea | 2.38 ± 0.13 | - | 21.18 ± 1.33 | 1.26 ± 0.09 | 0.53 ± 0.03 | - | - | |
| Fenghuang Danzong Tea | Oolong tea | 4.02 ± 0.38 | - | 37.95 ± 2.84 | 1.50 ± 0.11 | 0.59 ± 0.04 | - | - | |
| Black Fu Brick Tea | Dark tea | 3.15 ± 0.2 | - | 18.01 ± 0.92 | - | 0.55 ± 0.03 | 0.21 ± 0.01 | - | |
| Name | Category | FRAP (µmol Fe2+/g DW) | TEAC (µmol Trolox/g DW) | TPC (mg GAE/g DW) |
|---|---|---|---|---|
| Chaoqing Green Tea | Green tea | 1530.51 ± 22.40 | 1419.00 ± 3.14 | 132.07 ± 0.79 |
| Selenium-enriched Chaoqing Green Tea | Green tea | 2819.20 ± 22.63 | 1960.46 ± 23.69 | 173.46 ± 1.18 |
| Selenium-Enriched Matcha | Green tea | 2691.66 ± 12.93 | 1567.68 ± 18.83 | 160.96 ± 3.50 |
| Matcha | Green tea | 1315.20 ± 16.80 | 733.30 ± 19.09 | 110.82 ± 1.09 |
| Fried Green Tea | Green tea | 2736.91 ± 52.92 | 1421.22 ± 5.44 | 161.65 ± 0.39 |
| Liping Xiang Tea | Green tea | 3891.20 ± 75.60 | 1589.87 ± 23.23 | 219.99 ± 1.42 |
| Jieyang Chaoqing Tea | Green tea | 2535.77 ± 16.80 | 1432.31 ± 20.58 | 174.43 ± 0.79 |
| Fenggang Zinc-Selenium-Enriched Tea | Green tea | 3891.20 ± 64.65 | 2162.40 ± 24.51 | 251.65 ± 4.43 |
| Yihong Tea | Black tea | 1095.77± 36.99 | 849.18 ± 9.19 | 110.41 ± 0.07 |
| Selenium-Enriched Black Tea | Black tea | 1060.63 ± 17.10 | 872.48 ± 10.79 | 112.65 ± 0.73 |
| Yingde Black Tea | Black tea | 1377.77 ± 32.32 | 1003.96 ± 5.88 | 148.02 ± 1.50 |
| Wuyi Narcissus Tea | Oolong tea | 1460.91 ± 55.16 | 899.94 ± 14.12 | 114.56 ± 1.60 |
| Fenghuang Danzong Tea | Oolong tea | 2657.60 ± 40.41 | 1358.59 ± 9.54 | 165.20 ± 1.88 |
| Black Fu Brick Tea | Dark tea | 1248.34 ± 12.12 | 736.84 ± 3.11 | 90.05 ± 1.40 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cao, S.-Y.; Li, B.-Y.; Gan, R.-Y.; Mao, Q.-Q.; Wang, Y.-F.; Shang, A.; Meng, J.-M.; Xu, X.-Y.; Wei, X.-L.; Li, H.-B. The In Vivo Antioxidant and Hepatoprotective Actions of Selected Chinese Teas. Foods 2020, 9, 262. https://doi.org/10.3390/foods9030262
Cao S-Y, Li B-Y, Gan R-Y, Mao Q-Q, Wang Y-F, Shang A, Meng J-M, Xu X-Y, Wei X-L, Li H-B. The In Vivo Antioxidant and Hepatoprotective Actions of Selected Chinese Teas. Foods. 2020; 9(3):262. https://doi.org/10.3390/foods9030262
Chicago/Turabian StyleCao, Shi-Yu, Bang-Yan Li, Ren-You Gan, Qian-Qian Mao, Yuan-Feng Wang, Ao Shang, Jin-Ming Meng, Xiao-Yu Xu, Xin-Lin Wei, and Hua-Bin Li. 2020. "The In Vivo Antioxidant and Hepatoprotective Actions of Selected Chinese Teas" Foods 9, no. 3: 262. https://doi.org/10.3390/foods9030262
APA StyleCao, S.-Y., Li, B.-Y., Gan, R.-Y., Mao, Q.-Q., Wang, Y.-F., Shang, A., Meng, J.-M., Xu, X.-Y., Wei, X.-L., & Li, H.-B. (2020). The In Vivo Antioxidant and Hepatoprotective Actions of Selected Chinese Teas. Foods, 9(3), 262. https://doi.org/10.3390/foods9030262






