Meat Quality Derived from High Inclusion of a Micro-Alga or Insect Meal as an Alternative Protein Source in Poultry Diets: A Pilot Study
Abstract
:1. Introduction
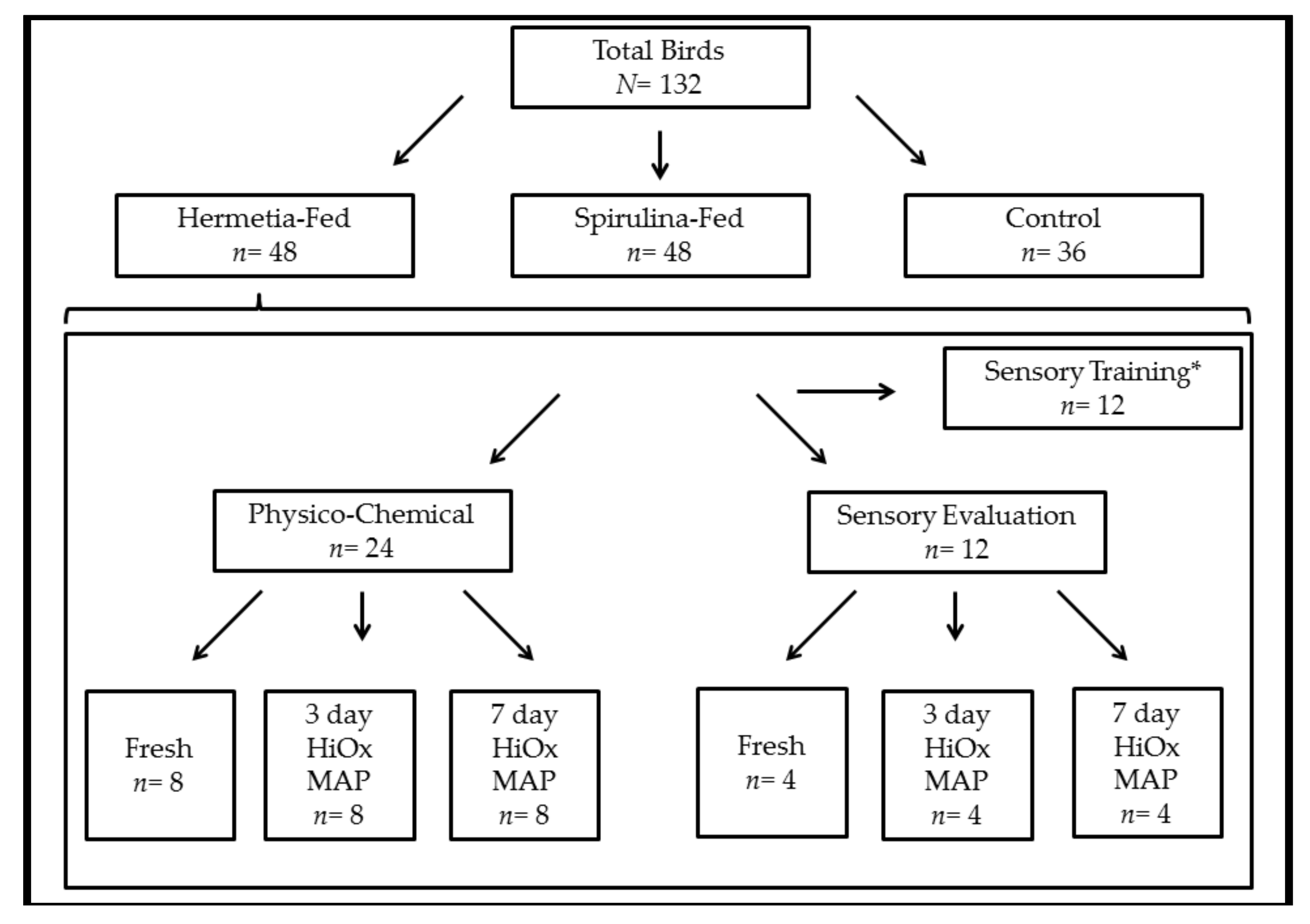
2. Materials and Methods
2.1. Animals and Diets
2.2. Animal and Sample Management
2.3. Physico-Chemical Characteristics
2.4. Lipid Oxidation
2.5. Sensory Analysis
2.6. Statistical Analyses
3. Results
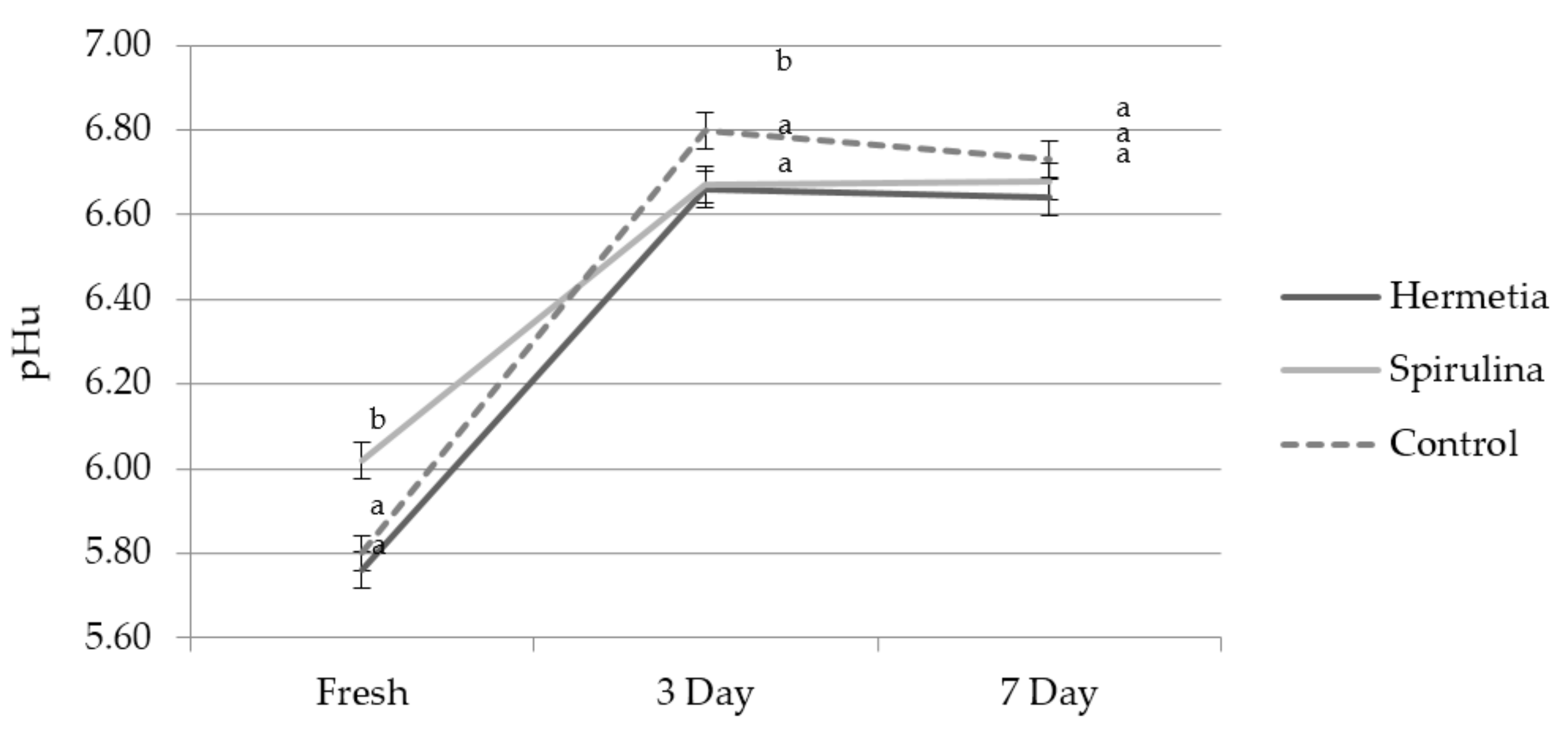
3.1. Physico-Chemical Results
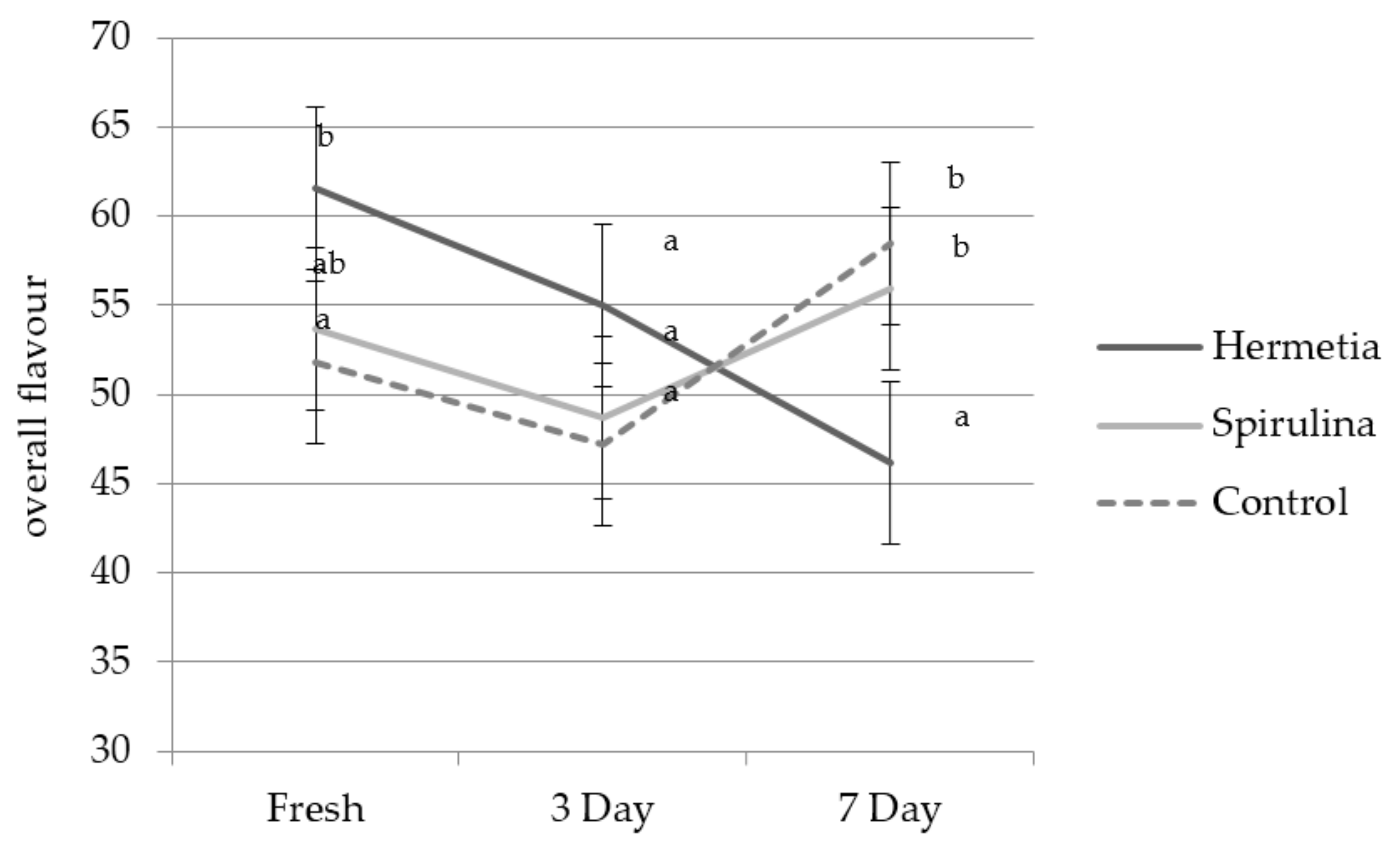
3.2. Sensory Results
4. Discussion
4.1. Spirulina in Poultry Diets
4.2. Hermetia in Poultry Diets
4.3. Impact of Storage Time
4.4. Interaction Effects Feed × Storage
4.5. Looking Forward
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix A
| Attribute | Description | Sense | References * |
|---|---|---|---|
| 1. Colour intensity | The intensity of the colour from white to dark beige | Visual | N/A |
| 2. Elasticity | How far the sample can be pressed and still return to original form | Visual | <30: spreadable cheese |
| 50: white bread | |||
| 100: wine gum | |||
| 3. Fibrousness | Fibre thickness on the product surface | Visual | <30: human hair |
| <70: ca. 2 mm thick | |||
| 4. Overall odour | The intensity of the smell when product held 2 cm from nose | Smell | N/A |
| 5. Animal/Barn odour | Intensity of faecal, barn, or animal odour | Smell | Skatol |
| 6. Metallic odour | Intensity of metal odour | Smell | old oxidized coins |
| 7. Cooked chicken odour | Intensity of the odour from chicken meat in soup | Smell | cooked chicken juice |
| 8. Overall flavour | Intensity of the overall flavour | Taste | N/A |
| 9. Sweet taste | Intensity of sweetness | Taste | sucrose solutions |
| 50: 6 g/L | |||
| 100: 18 g/L | |||
| 10. Sour taste | Intensity of sour taste | Taste | citric acid solutions |
| 50: 0.28 g/L | |||
| 100: 0.4 g/L | |||
| 11. Bitter taste | Intensity of bitterness | Taste | caffeine solution |
| 50: 0.21 g/L | |||
| 100: 0.3 g/L | |||
| 12. Metallic flavour | Intensity of metallic taste, such as serum or old coins | Taste | N/A |
| 13. Chicken flavour | Intensity of taste similar to chicken soup | Taste | chicken soup broth |
| 14. Aftertaste | The intensity of the aftertaste after swallowing | Taste | N/A |
| 15. Hardness | The force needed to bite through the sample | Texture | <30: spreadable cheese |
| 50: gouda cheese | |||
| 100: Werther’s Original | |||
| 16. Juiciness | Amount of fluid released from product upon first bite | Texture | <30: raw carrot |
| 50: cucumber | |||
| 100: orange | |||
| 17. Tenderness | The degree that the sample remains in one piece while chewing | Texture | <30: raw carrot |
| 50: gouda cheese | |||
| 100: hard candy | |||
| 18. Adhesiveness | The degree the product sticks to your teeth; force needed to pull teeth apart | Texture | 50: spreadable cheese |
| 100: peanut butter | |||
| 19. Crumbliness | Number of particles in mouth directly prior to swallowing | Texture | N/A |
References
- OECD/Food and Agriculture Organization of the United Nations. OECD-FAO Agricultural Outlook 2015–2024; OECD/Food and Agriculture Organization of the United Nations: Rome, Italy, 2015. [Google Scholar] [CrossRef]
- Taelman, S.E.; De Meester, S.; Van Dijk, W.; da Silva, V.; Dewulf, J. Environmental sustainability analysis of a protein-rich livestock feed ingredient in The Netherlands: Microalgae production versus soybean import. Resour. Conserv. Recycl. 2015, 101, 61–72. [Google Scholar] [CrossRef]
- Song, B.; Marchant, M.A.; Reed, M.R.; Xu, S. Competitive analysis and market power of China’s soybean import market. Int. Food Agribus. Manag. Rev. 2009, 12, 21–42. [Google Scholar]
- Da Silva, V.P.; van der Werf, H.M.G.; Spies, A.; Soares, S.R. Variability in environmental impacts of Brazilian soybean according to crop production and transport scenarios. J. Environ. Manag. 2010, 91, 1831–1839. [Google Scholar] [CrossRef] [PubMed]
- Tokusoglu, Ö.; Unal, M.K. Biomass nutrient profiles of three microalgae: Spirulina platensis, Chlorella vulgaris and Isochrysis galbana. J. Food Sci. 2003, 68, 1144–1148. [Google Scholar] [CrossRef]
- Habib, M.A.B.; Parvin, M.; Huntington, T.C.; Hasan, M.R. A Review on Culture, Production and Use of Spirulina as Food for Humans and Feeds for Domestic Animals and Fish. In FAO Fisheries and Aquaculture Circular; FAO: Rome, Italy, 2008; No. 1034; ISBN 978–92-5-106106-0. [Google Scholar]
- Spranghers, T.; Ottoboni, M.; Klootwijk, C.; Ovyn, A.; Deboosere, S.; De Meulenaer, B.; Michiels, J.; Eeckhout, M.; De Clercq, P.; De Smet, S. Nutritional composition of black soldier fly (Hermetia illucens) prepupae reared on different organic waste substrates. J. Sci. Food Agric. 2016, 97, 2594–2600. [Google Scholar] [CrossRef] [PubMed]
- Newton, G.L.; Sheppard, D.C.; Burtle, G.J. Research Briefs: Black Soldier Fly Prepupae—A Compelling Alternative to Fish Meal and Fish Oil. Available online: http://www.extension.org/pages/15054/research-summary:-black-soldier-fly-prepupae-a-compelling-alternative-to-fish-meal-and-fish-oil (accessed on 8 August 2017).
- European Union. Commission Regulation (EU) 2017/893. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:32017R0893 (accessed on 6 November 2017).
- Toldra, F.; Flores, M. Analysis of Meat Quality. In Handbook of Food Analysis, 2nd ed.; Nollet, L.M.L., Ed.; CRC Press: Boca Raton, FL, USA, 2004; Volume 3, pp. 1961–1978. ISBN 978-1-4398-3305-6. [Google Scholar]
- Oliver, M.A.; Nute, G.R.; Font-i-Furnols, M.; San Julián, R.; Campo, M.M.; Sañudo, C.; Cañeque, V.; Guerrero, L.; Alvarez, I.; Díaz, M.T.; et al. Eating quality of beef, from different production systems, assessed by German, Spanish and British consumers. Meat Sci. 2006, 74, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Korzen, S.; Lassen, J. Meat in context. On the relation between perceptions and contexts. Appetite 2010, 54, 274–281. [Google Scholar] [CrossRef] [PubMed]
- Font-i-Furnols, M.; Candek-Potokar, M.; Maltin, C.; Prevolnik Povše, M. A Handbook of Reference Methods for Meat Quality Assessment; European Cooperation in Science and Technology (COST): Brussels, Belgium, 2015. [Google Scholar]
- BfR. Fragen und Antworten zu Fleisch, Welches unter Schutzatmosphäre mit Erhöhtem Sauerstoffgehalt Verpackt Wurde. Bundesinstitut für Risikobewertung. Available online: http://www.bfr.bund.de/de/fragen_und_antworten_zu_fleisch__welches_unter_schutzatmosphaere_mit_erhoehtem_sauerstoffgehalt_verpackt_wurde-51981.html#topic_51983 (accessed on 25 July 2017). (In German).
- Wecke, C.; Liebert, F. Improving the reliability of optimal in-feed amino acid ratios based on individual amino acid efficiency data from N balance studies in growing chicken. Animals 2013, 3, 558–573. [Google Scholar] [CrossRef] [PubMed]
- Ross 308 Broiler: Nutrition Specifications (All Plant-Protein Based Feeds). Available online: http://eu.aviagen.com/assets/Tech_Center/Ross_Broiler/Ross-308-Broiler-Nutrition-Specs-plant-2014-EN.pdf (accessed on 22 February 2018).
- Neumann, C.; Velten, S.; Liebert, F. Improving the dietary protein quality by amino acid fortification with a high inclusion level of micro algae (Spirulina platensis) or insect meal (Hermetia illucens) in meat type chicken diets. Open J. Anim. Sci. 2018, 8, 12–26. [Google Scholar] [CrossRef]
- WPSA. The Prediction of Apparent Metabolizable Energy Values for Poultry in Compound Feeds. Worlds Poult. Sci J. 1984, 40, 181–182. [Google Scholar]
- Verordnung zum Schutz Landwirtschaftlicher Nutztiere und Anderer zur Erzeugung Tierischer Produkte Gehaltener Tiere bei ihrer Haltung. Available online: http://www.gesetze-im-internet.de/tierschnutztv/ (accessed on 6 August 2017). (In German).
- European Communities. Regulation (EC) NO 853/2004 of the European Parliament and of the Council. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/?qid=1520433365249&uri=CELEX:32004R0853 (accessed on 6 August 2017).
- Anderson, S. Determination of Fat, Moisture, and Protein in Meat and Meat Products by Using the FOSS FoodScan™ Near-Infrared Spectrophotometer with FOSS Artificial Neural Network Calibration Model and Associated Database: Collaborative Study. J. AOAC Int. 2007, 90, 1073–1083. [Google Scholar] [PubMed]
- Xiong, R.; Cavitt, L.C.; Meullenet, J.F.; Owens, C.M. Comparison of Allo-Kramer, Warner-Bratzler and razor blade shears for predicting sensory tenderness of broiler breast meat. J. Text. Stud. 2006, 37, 179–199. [Google Scholar] [CrossRef]
- Bruna, J.M.; Ordóñez, J.A.; Fernández, M.; Herranz, B.; De La Hoz, L. Microbial and physico-chemical changes during the ripening of dry fermented sausages superficially inoculated with or having added an intracellular cell-free extract of Penicillium aurantiogriseum. Meat Sci. 2001, 59, 87–96. [Google Scholar] [CrossRef]
- International Organization for Standardization. ISO 8589:2007: Sensory Analysis—General Guidance for the Design of Test Rooms. Available online: https://www.iso.org/standard/36385.html (accessed on 20 January 2018).
- International Organization for Standardization. ISO 8586-1:1993: Sensory Analysis—General Guidance for the Selection, Training and Monitoring of Assessors—Part 1: Selected Assessors. Available online: https://www.iso.org/standard/15875.html (assessed on 20 January 2018).
- Cohen, J. The Effect Size Index: D. In Statistical Power Analysis for the Behavioural Sciences, 2nd ed.; Lawrence Erlbaum Associates: Mahwah, NJ, USA, 1988; pp. 20–27. ISBN 0805802835. [Google Scholar]
- Batorek, N.; Čandek-Potokar, M.; Bonneau, M.; Van Milgen, J. Meta-analysis of the effect of immunocastration on production performance, reproductive organs and boar taint compounds in pigs. Animal 2012, 6, 1330–1338. [Google Scholar] [CrossRef] [PubMed]
- Martin, N. What is the way forward for protein supply? The European perspective. Oilseeds Fats Crops Lipids 2014, 21, D403. [Google Scholar] [CrossRef]
- Berri, C.; Besnard, J.; Relandeau, C. Increasing Dietary Lysine Increases Final pH and Decreases Drip Loss of Broiler Breast Meat. Poult. Sci. 2008, 87, 480–484. [Google Scholar] [CrossRef] [PubMed]
- Aaslyng, M.D.; Bejerholm, C.; Ertbjerg, P.; Bertram, H.C.; Andersen, H.J. Cooking loss and juiciness of pork in relation to raw meat quality and cooking procedure. Food Qual. Prefer. 2003, 14, 277–288. [Google Scholar] [CrossRef]
- Liu, Y.; Lyon, B.G.; Windham, W.R.; Lyon, C.E.; Savage, E.M. Principal component analysis of physical, color and sensory characteristics of chicken breasts deboned at two, four, six and twenty-four hours postmortem. Poult. Sci. 2004, 83, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.-S.; Hwang, K.E.; Jeong, T.J.; Kim, Y.B.; Jeon, K.H.; Kim, E.M.; Sung, J.M.; Kim, H.W.; Kim, C.J. Comparative study on the effects of boiling, steaming, grilling, microwaving and superheated steaming on quality characteristics of marinated chicken steak. Korean J. Food Sci. Anim. Resour. 2016, 36, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Neethling, J.; Hoffman, L.C.; Muller, M. Factors influencing the flavour of game meat: A review. Meat Sci. 2016, 113, 139–153. [Google Scholar] [CrossRef] [PubMed]
- Campo, M.M.; Nute, G.R.; Hughes, S.I.; Enser, M.; Wood, J.D.; Richardson, R.I. Flavour perception of oxidation in beef. Meat Sci. 2006, 72, 303–311. [Google Scholar] [CrossRef] [PubMed]
- Brunton, N.P.; Cronin, D.A.; Monahan, F.J.; Durcan, R. A comparison of solid-phase microextraction (SPME) fibres for measurement of hexanal and pentanal in cooked turkey. Food Chem. 2000, 68, 339–345. [Google Scholar] [CrossRef]
- Jayasena, D.D.; Ahn, D.U.; Nam, K.C.; Jo, C. Flavour chemistry of chicken meat: A review. Asian-Australas. J. Anim. Sci. 2013, 26, 732–742. [Google Scholar] [CrossRef] [PubMed]
- Toyomizu, M.; Sato, K.; Taroda, H.; Kato, T.; Akiba, Y. Effects of dietary Spirulina on meat colour in muscle of broiler chickens. Br. Poult. Sci. 2001, 42, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Venkataraman, L.V.; Somasekaran, T.; Becker, E.W. Replacement value of blue-green alga (Spirulina platensis) for fishmeal and a vitamin-mineral premix for broiler chicks. Br. Poult. Sci. 1994, 35, 373–381. [Google Scholar] [CrossRef] [PubMed]
- Holman, B.W.B.; Malau-Aduli, A.E.O. Spirulina as a livestock supplement and animal feed. J. Anim. Physiol. Anim. Nutr. 2013, 97, 615–623. [Google Scholar] [CrossRef] [PubMed]
- Viana, E.S.; Gomide, L.A.M.; Vanetti, M.C.D. Effect of modified atmospheres on microbiological, color and sensory properties of refrigerated pork. Meat Sci. 2005, 71, 696–705. [Google Scholar] [CrossRef] [PubMed]
- McMillin, K.W. Where is MAP Going? A review and future potential of modified atmosphere packaging for meat. Meat Sci. 2008, 80, 43–65. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, C.E.; Cornforth, D.P.; Whittier, D. Consumer preferences for beef color and packaging did not affect eating satisfaction. Meat Sci. 2001, 57, 359–363. [Google Scholar] [CrossRef]
- Gawaad, A.; Brune, H. Insect protein as a possible source of protein to poultry. Z. Tierphysiol. Tierernaehrung Futtermittelkd 1979, 42, 216–222. [Google Scholar] [CrossRef]
- Delles, R.M.; Xiong, Y.L. The effect of protein oxidation on hydration and water-binding in pork packaged in an oxygen-enriched atmosphere. Meat Sci. 2014, 97, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Spanos, D.; Tørngren, M.A.; Christensen, M.; Baron, C.P. Effect of oxygen level on the oxidative stability of two different retail pork products stored using modified atmosphere packaging (MAP). Meat Sci. 2016, 113, 162–169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bao, Y.; Puolanne, E.; Ertbjerg, P. Effect of oxygen concentration in modified atmosphere packaging on color and texture of beef patties cooked to different temperatures. Meat Sci. 2016, 121, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.L.; Park, D.; Ooizumi, T. Variation in the Cross-Linking Pattern of Porcine Myofibrillar Protein Exposed to Three Oxidative Environments. J. Agric. Food Chem. 2008, 57, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Muhlisin, P.; Kim, D.S.; Song, Y.R.; Lee, S.J.; Lee, J.K.; Lee, S.K. Effects of gas composition in the modified atmosphere packaging on the shelf-life of Longissimus dorsi of Korean native black pigs-duroc crossbred during refrigerated storage. Asian-Australas. J. Anim. Sci. 2014, 27, 1157–1163. [Google Scholar] [CrossRef] [PubMed]
- Allen, C.D.; Russell, S.M.; Fletcher, D.L. The relationship of broiler breast meat color and pH to shelf-life and odor development. Poult. Sci. 1997, 76, 1042–1046. [Google Scholar] [CrossRef] [PubMed]
| Ingredients/Diets | Starter Period (1–21 day(s)) | Grower Period (22–34 days) | ||||
|---|---|---|---|---|---|---|
| Hermetia | Spirulina | Control | Hermetia | Spirulina | Control | |
| Wheat | 358.3 | 377.9 | 328.8 | 402.6 | 416.8 | 375.8 |
| Corn | 179.2 | 189.0 | 164.4 | 201.3 | 208.4 | 187.9 |
| Soybean meal | 195.0 | 195.0 | 390.0 | 160.0 | 160.0 | 320.0 |
| Insect meal | 145.4 | - | - | 119.0 | - | - |
| Algae meal | 118.2 | 97.0 | ||||
| Soybean oil | 78.5 | 78.5 | 78.5 | 78.5 | 78.5 | 78.5 |
| Premix * | 10.0 | 10.0 | 10.0 | 10.0 | 10.0 | 10.0 |
| CaHPO4 | 12.0 | 12.0 | 11.0 | 8.0 | 10.0 | 10.0 |
| CaCO3 | 9.9 | 9.1 | 11.0 | 8.0 | 8.0 | 9.0 |
| NaCl | 1.7 | 1.7 | 3.0 | 2.0 | 2.0 | 3.0 |
| Wheat starch | - | - | - | 3.0 | 3.0 | - |
| TiO2 | - | - | - | 3.0 | ||
| l-Lysine∙HCl | 3.2 | 4.4 | 1.3 | 2.4 | 3.5 | 0.8 |
| dl-Methionine | 4.1 | 3.5 | 2.0 | 3.0 | 2.5 | 2.0 |
| l-Threonine | 0.6 | - | - | 0.4 | - | - |
| l-Arginine | 2.2 | 0.7 | - | 1.4 | 0.1 | - |
| l-Valine | - | - | - | 0.5 | 0.2 | - |
| Diets | Starter Period (1–21 day(s)) | Grower Period (22–34 days) | ||||
|---|---|---|---|---|---|---|
| Hermetia | Spirulina | Control | Hermetia | Spirulina | Control | |
| Crude protein | 259.3 | 241.4 | 249.5 | 230.9 | 207.2 | 220.2 |
| Ether extract | 131.1 | 116.6 | 111.6 | 131.4 | 118.4 | 112.8 |
| Crude fibre | 47.1 | 31.1 | 45.2 | 41.7 | 30.4 | 40.4 |
| Crude ash | 60.4 | 59.2 | 65.6 | 56.5 | 53.5 | 61.6 |
| N-free extract | 502.1 | 551.7 | 528.1 | 539.5 | 590.5 | 565 |
| AMEN (MJ/kg DM) * | 15.3 | 15.4 | 14.4 | 15.6 | 15.6 | 14.8 |
| Parameters | Hermetia | SD | Cohen’s d | Spirulina | SD | Cohen’s d | Control | SD |
|---|---|---|---|---|---|---|---|---|
| Live weight (g) | 2329 a (n = 48) | 322 | 0.509 | 2121 b (n = 48) | 218 | 0.181 | 2169 b (n = 36) | 306 |
| Carcass weight (g) | 1789 a (n = 48) | 261 | 0.476 | 1577 b (n = 48) | 175 | 0.465 | 1672 b (n = 36) | 230 |
| Breast filet yield (%) | 20.4 a (n = 36) | 1.9 | 0.205 | 20.0 a (n = 36) | 2.0 | 0.400 | 20.8 a (n = 24) | 2.0 |
| Protein (% breast filet) | 21.07 a (n = 8) | 0.27 | 0.99 | 21.60 a (n = 8) | 0.64 | 1.56 | 20.52 a (n = 8) | 0.74 |
| IMF (% breast filet) | 3.41 a (n = 8) | 0.45 | 0.02 | 3.12 a (n = 8) | 0.50 | 0.47 | 3.40 a (n = 8) | 0.68 |
| Moisture (% breast filet) | 73.88 a (n = 8) | 0.43 | 0.62 | 73.62 a (n = 8) | 0.71 | 0.87 | 74.29 a (n = 8) | 0.83 |
| pH20min | 6.65 b (n = 36) | 0.12 | 0.917 | 6.67 b (n = 36) | 0.12 | 0.750 | 6.76 a (n = 24) | 0.12 |
| pH24h | 5.80 a (n = 20) | 0.18 | 0.00 | 6.06 b (n = 19) | 0.13 | 1.852 | 5.80 a (n = 8) | 0.15 |
| Parameters | Feed Groups | Partial η2 | Storage Time | Partial η2 | SE | ||||
|---|---|---|---|---|---|---|---|---|---|
| Hermetia | Spirulina | Control | Fresh | 3 Day | 7 Day | ||||
| L* | 55.60 a | 53.01 b | 55.40 a | 0.243 | 51.10 f | 54.92 e | 58.00 d | 0.648 | 0.454 |
| a* | 2.00 b | 3.48 a | 2.43 b | 0.212 | 0.85 f | 4.04 d | 3.03 e | 0.554 | 0.261 |
| b* | 11.15 b | 12.31 a | 11.33 b | 0.131 | 8.65 e | 13.15 d | 12.99 d | 0.716 | 0.286 |
| TBARS (μg/g) | 0.106 a | 0.095 a | 0.098 a | 0.006 | 0.081 e | 0.084 e | 0.134 d | 0.150 | 0.013 |
| Storage loss (%) | 3.00 a | 2.48 b | 3.04 a | 0.147 | - | 2.72 d | 2.96 d | 0.035 | 0.134 |
| Cooking loss (%) | 31.80 a | 29.00 b | 33.05 a | 0.169 | 33.29 d | 30.29 e | 30.25 e | 0.125 | 0.821 |
| Shear force (N) | 10.86 a | 11.16 a | 11.80 a | 0.060 | 11.01 d | 11.58 d | 11.21 d | 0.058 | 0.336 |
| Sensory Attribute | Hermetia | Spirulina | Control | Partial η2 | SE |
|---|---|---|---|---|---|
| Metallic flavour (not metallic to strongly metallic) | 20.4 a | 15.8 b | 17.1 a | 0.223 | 3.90 |
| Hardness (soft to hard) | 28.8 b | 25.0 b | 40.8 a | 0.300 | 4.93 |
| Tenderness (tender to tough) | 30.8 b | 24.4 b | 47.1 a | 0.371 | 6.05 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Altmann, B.A.; Neumann, C.; Velten, S.; Liebert, F.; Mörlein, D. Meat Quality Derived from High Inclusion of a Micro-Alga or Insect Meal as an Alternative Protein Source in Poultry Diets: A Pilot Study. Foods 2018, 7, 34. https://doi.org/10.3390/foods7030034
Altmann BA, Neumann C, Velten S, Liebert F, Mörlein D. Meat Quality Derived from High Inclusion of a Micro-Alga or Insect Meal as an Alternative Protein Source in Poultry Diets: A Pilot Study. Foods. 2018; 7(3):34. https://doi.org/10.3390/foods7030034
Chicago/Turabian StyleAltmann, Brianne A., Carmen Neumann, Susanne Velten, Frank Liebert, and Daniel Mörlein. 2018. "Meat Quality Derived from High Inclusion of a Micro-Alga or Insect Meal as an Alternative Protein Source in Poultry Diets: A Pilot Study" Foods 7, no. 3: 34. https://doi.org/10.3390/foods7030034





