Health-Promoting Properties of Lacticaseibacillus paracasei: A Focus on Kefir Isolates and Exopolysaccharide-Producing Strains
Abstract
:1. Introduction
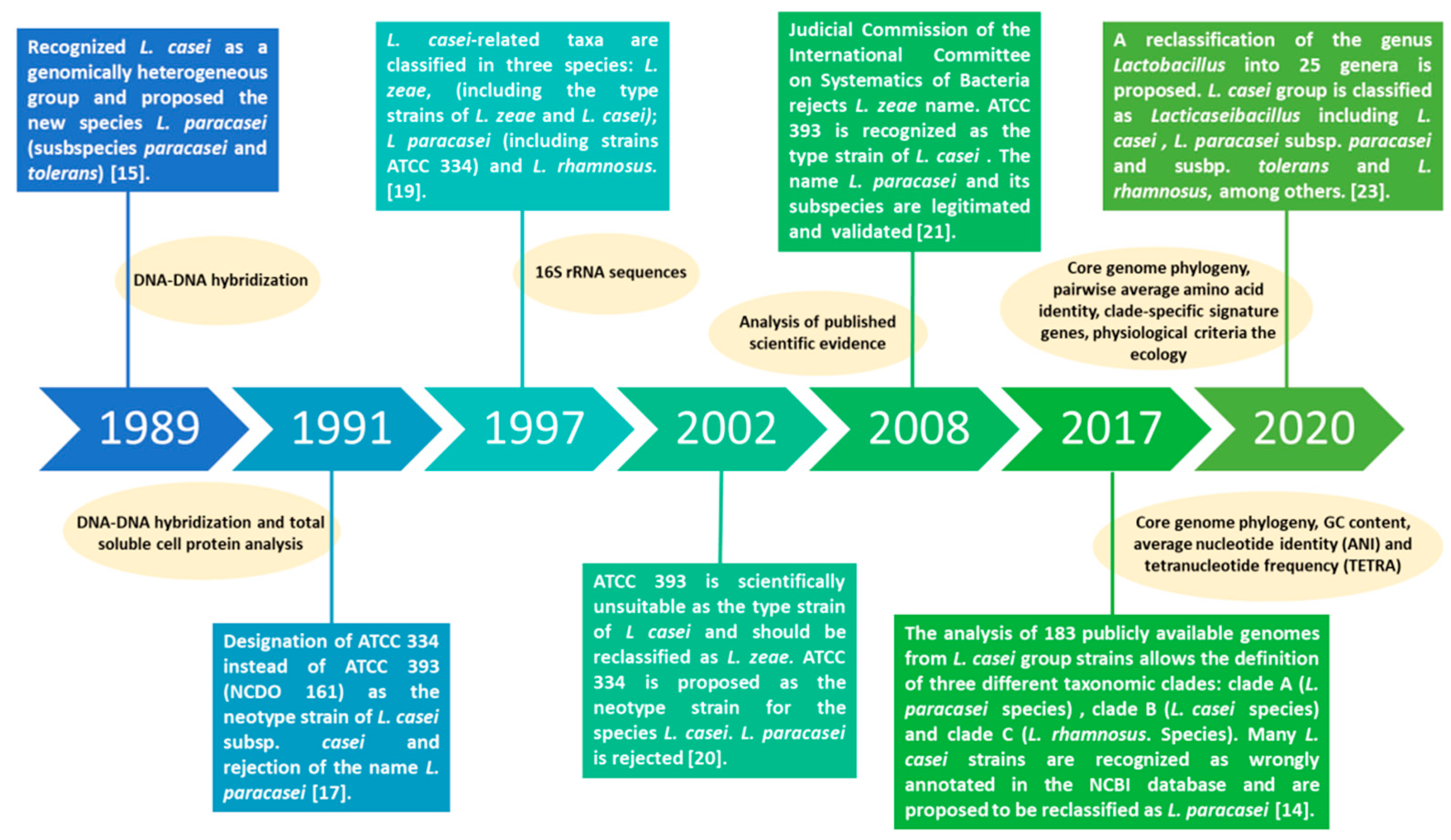
2. Lacticaseibacillus paracasei: A Tour through the Evolution of Taxonomical Classification
3. Identification and Classification of Lacticaseibacillus paracasei
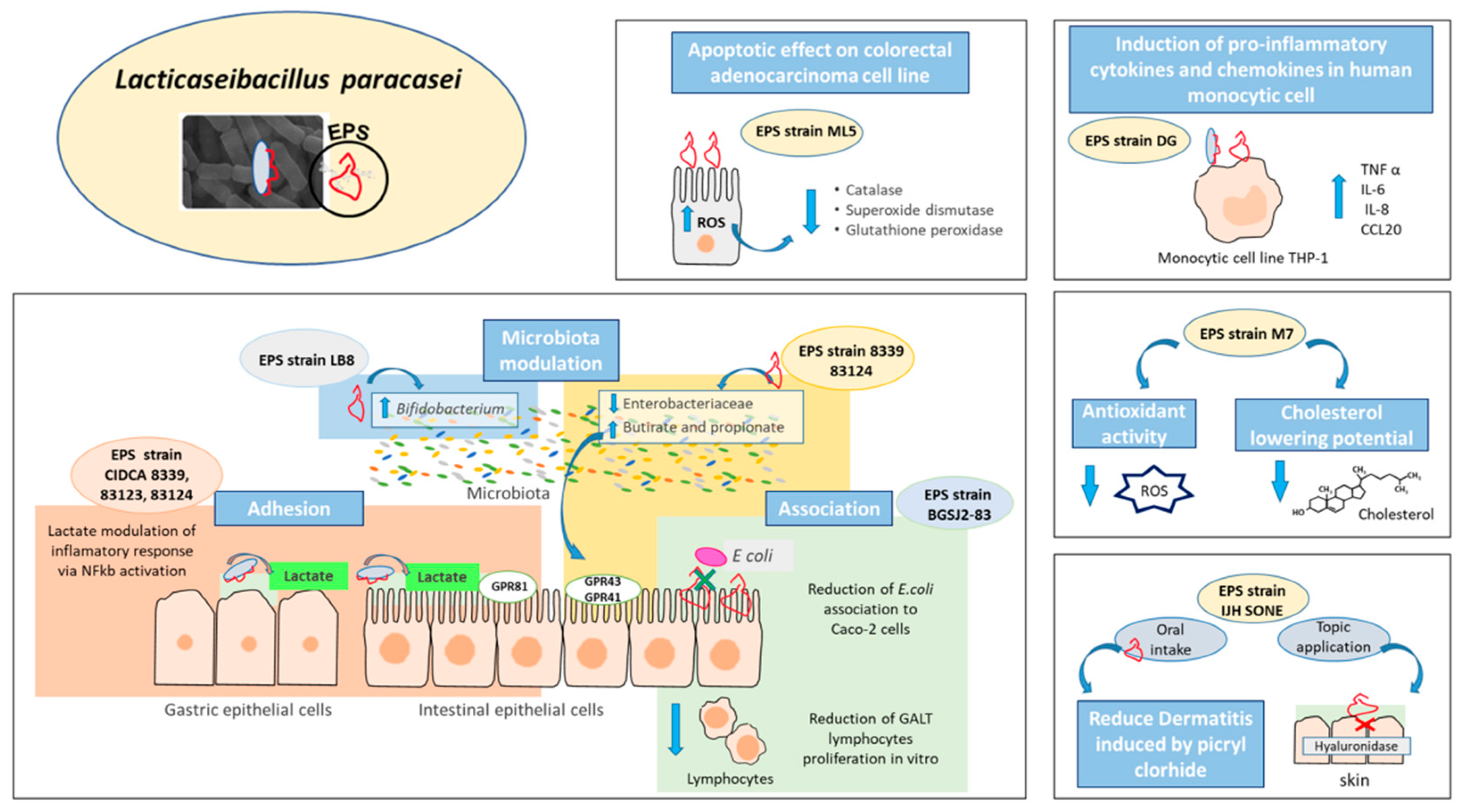
4. Lacticaseibacillus paracasei Health-Promoting Properties and the Effector Molecules Associated
5. Exopolysaccharides as Effector Molecules Associated to Lacticaseibacillus paracasei Health Benefits
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- González, S.; Fernández-Navarro, T.; Arboleya, S.; de los Reyes-Gavilán, C.G.; Salazar, N.; Gueimonde, M. Fermented Dairy Foods: Impact on Intestinal Microbiota and Health-Linked Biomarkers. Front. Microbiol. 2019, 10, 1046. [Google Scholar] [CrossRef] [PubMed]
- Marco, M.L.; Heeney, D.; Binda, S.; Cifelli, C.J.; Cotter, P.D.; Foligné, B.; Gänzle, M.; Kort, R.; Pasin, G.; Pihlanto, A.; et al. Health benefits of fermented foods: Microbiota and beyond. Curr. Opin. Biotechnol. 2017, 44, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Bengoa, A.A.; Iraporda, C.; Garrote, G.L.; Abraham, A.G. Kefir micro-organisms: Their role in grain assembly and health properties of fermented milk. J. Appl. Microbiol. 2019, 126, 686–700. [Google Scholar] [CrossRef] [Green Version]
- Lynch, K.M.; Wilkinson, S.; Daenen, L.; Arendt, E.K. An update on water kefir: Microbiology, composition and production. Int. J. Food Microbiol. 2021, 345, 109128. [Google Scholar] [CrossRef]
- Garofalo, C.; Osimani, A.; Milanović, V.; Aquilanti, L.; De Filippis, F.; Stellato, G.; Di Mauro, S.; Turchetti, B.; Buzzini, P.; Ercolini, D.; et al. Bacteria and yeast microbiota in milk kefir grains from different Italian regions. Food Microbiol. 2015, 49, 123–133. [Google Scholar] [CrossRef]
- Bourrie, B.C.T.; Willing, B.P.; Cotter, P.D. The Microbiota and Health Promoting Characteristics of the Fermented Beverage Kefir. Front. Microbiol. 2016, 7, 647. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marsh, A.J.; O’Sullivan, O.; Hill, C.; Ross, R.P.; Cotter, P.D. Sequence-based analysis of the microbial composition of water kefir from multiple sources. FEMS Microbiol. Lett. 2013, 348, 79–85. [Google Scholar] [CrossRef] [Green Version]
- Azizi, N.F.; Kumar, M.R.; Yeap, S.K.; Abdullah, J.O.; Khalid, M.; Omar, A.R.; Osman, M.A.; Mortadza, S.A.S.; Alitheen, N.B. Kefir and Its Biological Activities. Foods 2021, 10, 1210. [Google Scholar] [CrossRef]
- Peluzio, M.D.C.G.; e Dias, M.D.M.; Martinez, J.A.; Milagro, F.I. Kefir and Intestinal Microbiota Modulation: Implications in Human Health. Front. Nutr. 2021, 8, 638740. [Google Scholar] [CrossRef]
- Slattery, C.; Cotter, P.D.; O’Toole, P.W. Analysis of Health Benefits Conferred by Lactobacillus Species from Kefir. Nutrients 2019, 11, 1252. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hill, D.; Sugrue, I.; Tobin, C.; Hill, C.; Stanton, C.; Ross, R.P. The Lactobacillus casei Group: History and Health Related Applications. Front. Microbiol. 2018, 9, 2107. [Google Scholar] [CrossRef] [Green Version]
- Bengoa, A.A.; Iraporda, C.; Acurcio, L.B.; de Cicco Sandes, S.H.; Costa, K.; Guimarães, G.M.; Arantes, R.M.E.; Neumann, E.; Nunes, Á.C.; Nicoli, J.R.; et al. Physicochemical, immunomodulatory and safety aspects of milks fermented with Lactobacillus paracasei isolated from kefir. Food Res. Int. 2019, 123, 48–55. [Google Scholar] [CrossRef]
- Patterson, E.; Griffin, S.M.; Ibarra, A.; Ellsiepen, E.; Hellhammer, J. Lacticaseibacillus paracasei Lpc-37® improves psychological and physiological markers of stress and anxiety in healthy adults: A randomized, double-blind, placebo-controlled and parallel clinical trial (the Sisu study). Neurobiol. Stress 2020, 13, 100277. [Google Scholar] [CrossRef] [PubMed]
- Wuyts, S.; Wittouck, S.; De Boeck, I.; Allonsius, C.N.; Pasolli, E.; Segata, N.; Lebeer, S. Large-Scale Phylogenomics of the Lactobacillus casei Group Highlights Taxonomic Inconsistencies and Reveals Novel Clade-Associated Features. mSystems 2017, 2, e00061-17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Collins, M.D.; Phillips, B.A.; Zanoni, P. Deoxyribonucleic Acid Homology Studies of Lactobacillus casei, Lactobacillus paracasei sp. nov., subsp. paracasei and subsp. tolerans, and Lactobacillus rhamnosus sp. nov., comb. nov. Int. J. Syst. Evol. Microbiol. 1989, 39, 105–108. [Google Scholar] [CrossRef] [Green Version]
- Wittouck, S.; Wuyts, S.; Lebeer, S. Towards a Genome-Based Reclassification of the Genus Lactobacillus. Appl. Environ. Microbiol. 2019, 85, e02155-18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dellaglio, F.; Dicks, L.M.T.; DU Toit, M.; Torriani, S. Designation of ATCC 334 in Place of ATCC 393 (NCDO 161) as the Neotype Strain of Lactobacillus casei subsp. casei and Rejection of the Name Lactobacillus paracasei (Collins et al., 1989): Request for an Opinion. Int. J. Syst. Evol. Microbiol. 1991, 41, 340–342. [Google Scholar] [CrossRef] [Green Version]
- Dicks, L.M.T.; Du Plessis, E.M.; Dellagl, F.; Lauer, E. Reclassification of Lactobacillus casei subsp. casei ATCC 393 and Lactobacillus rhamnosus ATCC 15820 as Lactobacillus zeae norn. rev., designation of ATCC 334 as the neotype of L. casei subsp. casei, and Rejection of the Name Lactobacillus paracasei. Int. J. Syst. Evol. Microbiol. 1996, 46, 337–340. [Google Scholar] [CrossRef] [Green Version]
- Mori, K.; Yamazaki, K.; Ishiyama, T.; Katsumata, M.; Kobayashi, K.; Kawai, Y.; Inoue, N.; Shinano, H. Comparative Sequence Analyses of the Genes Coding for 16S rRNA of Lactobacillus casei-Related Taxa. Int. J. Syst. Evol. Microbiol. 1997, 47, 54–57. [Google Scholar] [CrossRef] [PubMed]
- Dellaglio, F.; Felis, G.E.; Torriani, S. The status of the species Lactobacillus casei (Orla-Jensen 1916) Hansen and Lessel 1971 and Lactobacillus paracasei Collins et al. 1989. Request for an opinion. Int. J. Syst. Evol. Microbiol. 2002, 52, 285–287. [Google Scholar] [CrossRef] [Green Version]
- Tindall, B.J. The type strain of Lactobacillus casei is ATCC 393, ATCC 334 cannot serve as the type because it represents a different taxon, the name Lactobacillus paracasei and its subspecies names are not rejected and the revival of the name ‘Lactobacillus zeae’ contravenes Rules 51b (1) and (2) of the International Code of Nomenclature of Bacteria. Opinion 82. Int. J. Syst. Evol. Microbiol. 2008, 58, 1764–1765. [Google Scholar] [CrossRef] [Green Version]
- Salvetti, E.; Harris, H.M.B.; Felis, G.E.; O’Toole, P.W. Comparative Genomics of the Genus Lactobacillus Reveals Robust Phylogroups That Provide the Basis for Reclassification. Appl. Environ. Microbiol. 2018, 84, e00993-18. [Google Scholar] [CrossRef] [Green Version]
- Zheng, J.; Wittouck, S.; Salvetti, E.; Franz, C.M.A.P.; Harris, H.M.B.; Mattarelli, P.; O’Toole, P.W.; Pot, B.; Vandamme, P.; Walter, J.; et al. A taxonomic note on the genus Lactobacillus: Description of 23 novel genera, emended description of the genus Lactobacillus Beijerinck 1901, and union of Lactobacillaceae and Leuconostocaceae. Int. J. Syst. Evol. Microbiol. 2020, 70, 2782–2858. [Google Scholar] [CrossRef]
- Fontana, A.; Zacconi, C.; Morelli, L. Genetic Signatures of Dairy Lactobacillus casei Group. Front. Microbiol. 2018, 9, 2611. [Google Scholar] [CrossRef]
- Huang, C.-H.; Li, S.-W.; Huang, L.; Watanabe, K. Identification and Classification for the Lactobacillus casei Group. Front. Microbiol. 2018, 9, 1974. [Google Scholar] [CrossRef]
- Vásquez, A.; Molin, G.; Pettersson, B.; Antonsson, M.; Ahrné, S. DNA-based classification and sequence heterogeneities in the 16S rRNA genes of Lactobacillus casei/paracasei and related species. Syst. Appl. Microbiol. 2005, 28, 430–441. [Google Scholar] [CrossRef] [PubMed]
- Smokvina, T.; Wels, M.; Polka, J.; Chervaux, C.; Brisse, S.; Boekhorst, J.; Van Vlieg, J.E.T.H.; Siezen, R.J. Lactobacillus paracasei Comparative Genomics: Towards Species Pan-Genome Definition and Exploitation of Diversity. PLoS ONE 2013, 8, e68731. [Google Scholar] [CrossRef] [Green Version]
- Stefanovic, E.; McAuliffe, O. Comparative genomic and metabolic analysis of three Lactobacillus paracasei cheese isolates reveals considerable genomic differences in strains from the same niche. BMC Genom. 2018, 19, 1–13. [Google Scholar] [CrossRef]
- Ward, L.J.H.; Timmins, M.J. Differentiation of Lactobacillus casei, Lactobacillus paracasei and Lactobacillus rhamnosus by polymerase chain reaction. Lett. Appl. Microbiol. 1999, 29, 90–92. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iacumin, L.; Ginaldi, F.; Manzano, M.; Anastasi, V.; Reale, A.; Zotta, T.; Rossi, F.; Coppola, R.; Comi, G. High resolution melting analysis (HRM) as a new tool for the identification of species belonging to the Lactobacillus casei group and comparison with species-specific PCRs and multiplex PCR. Food Microbiol. 2015, 46, 357–367. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.-H.; Lee, F.-L. The dnaK gene as a molecular marker for the classification and discrimination of the Lactobacillus casei group. Antonie Leeuwenhoek 2011, 99, 319–327. [Google Scholar] [CrossRef]
- Bottari, B.; Felis, G.E.; Salvetti, E.; Castioni, A.; Campedelli, I.; Torriani, S.; Bernini, V.; Gatti, M. Effective identification of Lactobacillus casei group species: Genome-based selection of the gene mutL as the target of a novel multiplex PCR assay. Microbiology 2017, 163, 950–960. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.-H.; Huang, L. Rapid species- and subspecies-specific level classification and identification of Lactobacillus casei group members using MALDI Biotyper combined with ClinProTools. J. Dairy Sci. 2018, 101, 979–991. [Google Scholar] [CrossRef]
- Naser, S.M.; Dawyndt, P.; Hoste, B.; Gevers, D.; Vandemeulebroecke, K.; Cleenwerck, I.; Vancanneyt, M.; Swings, J. Identification of lactobacilli by pheS and rpoA gene sequence analyses. Int. J. Syst. Evol. Microbiol. 2007, 57, 2777–2789. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, C.-H.; Liou, J.-S.; Lee, A.-Y.; Tseng, M.; Miyashita, M.; Huang, L.; Watanabe, K. Polyphasic characterization of a novel species in the Lactobacillus casei group from cow manure of Taiwan: Description of L. chiayiensis sp. nov. Syst. Appl. Microbiol. 2018, 41, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Silvaraju, S.; Menon, N.; Fan, H.; Lim, K.; Kittelmann, S. Phylotype-Level Characterization of Complex Communities of Lactobacilli Using a High-Throughput, High-Resolution Phenylalanyl-tRNA Synthetase (pheS) Gene Amplicon Sequencing Approach. Appl. Environ. Microbiol. 2020, 87, e02191-20. [Google Scholar] [CrossRef]
- Ghane, M.; Babaeekhou, L.; Ketabi, S.S. Antibiofilm activity of kefir probiotic lactobacilli against uropathogenic Escherichia coli (UPEC). Avicenna J. Med. Biotechnol. 2020, 12, 221–229. [Google Scholar]
- Miao, J.; Guo, H.; Ou, Y.; Liu, G.; Fang, X.; Liao, Z.; Ke, C.; Chen, Y.; Zhao, L.; Cao, Y. Purification and characterization of bacteriocin F1, a novel bacteriocin produced by Lactobacillus paracasei subsp. tolerans FX-6 from Tibetan kefir, a traditional fermented milk from Tibet, China. Food Control 2014, 42, 48–53. [Google Scholar] [CrossRef]
- Romero-Luna, H.E.; Peredo-Lovillo, A.; Hernández-Mendoza, A.; Hernández-Sánchez, H.; Cauich-Sánchez, P.I.; Ribas-Aparicio, R.M.; Dávila-Ortiz, G. Probiotic Potential of Lactobacillus paracasei CT12 Isolated from Water Kefir Grains (Tibicos). Curr. Microbiol. 2020, 77, 2584–2592. [Google Scholar] [CrossRef]
- Mantzourani, I.; Chondrou, P.; Bontsidis, C.; Karolidou, K.; Terpou, A.; Alexopoulos, A.; Bezirtzoglou, E.; Galanis, A.; Plessas, S. Assessment of the probiotic potential of lactic acid bacteria isolated from kefir grains: Evaluation of adhesion and antiproliferative properties in in vitro experimental systems. Ann. Microbiol. 2019, 69, 751–763. [Google Scholar] [CrossRef]
- Plessas, S.; Kiousi, D.E.; Rathosi, M.; Alexopoulos, A.; Kourkoutas, Y.; Mantzourani, I.; Galanis, A.; Bezirtzoglou, E. Isolation of a Lactobacillus paracasei Strain with Probiotic Attributes from Kefir Grains. Biomedicines 2020, 8, 594. [Google Scholar] [CrossRef]
- Leite, A.M.O.; Miguel, M.A.L.; Peixoto, R.S.; Ruas-Madiedo, P.; Paschoalin, V.M.F.; Mayo, B.; Delgado, S. Probiotic potential of selected lactic acid bacteria strains isolated from Brazilian kefir grains. J. Dairy Sci. 2015, 98, 3622–3632. [Google Scholar] [CrossRef] [Green Version]
- Karaffová, V.; Mudroňová, D.; Mad’Ar, M.; Hrčková, G.; Faixová, D.; Gancarčíková, S.; Ševčíková, Z.; Nemcová, R. Differences in Immune Response and Biochemical Parameters of Mice Fed by Kefir Milk and Lacticaseibacillus paracasei Isolated from the Kefir Grains. Microorganisms 2021, 9, 831. [Google Scholar] [CrossRef] [PubMed]
- Hamet, M.F.; Londero, A.; Medrano, M.; Vercammen, E.; Van Hoorde, K.; Garrote, G.L.; Huys, G.; Vandamme, P.; Abraham, A.G. Application of culture-dependent and culture-independent methods for the identification of Lactobacillus kefiranofaciens in microbial consortia present in kefir grains. Food Microbiol. 2013, 36, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.; Yang, S.-M.; Cho, E.-J.; Kim, H.-Y. Novel real-time PCR assay for Lactobacillus casei group species using comparative genomics. Food Microbiol. 2020, 90, 103485. [Google Scholar] [CrossRef]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. Expert Consensus Document: The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef] [Green Version]
- Jones, R.M. The Use of Lactobacillus casei and Lactobacillus paracasei in Clinical Trials for the Improvement of Human Health. In The Microbiota in Gastrointestinal Pathophysiology: Implications for Human Health, Prebiotics, Probiotics, and Dysbiosis; Academic Press: Cambridge, MA, USA, 2017; pp. 99–108. ISBN 9780128040249. [Google Scholar]
- Azais-Braesco, V.; Bresson, J.L.; Guarner, F.; Corthier, G. Not all lactic acid bacteria are probiotics, …but some are. Br. J. Nutr. 2010, 103, 1079–1081. [Google Scholar] [CrossRef] [Green Version]
- Balzaretti, S.; Taverniti, V.; Guglielmetti, S.; Fiore, W.; Minuzzo, M.; Ngo, H.N.; Ngere, J.B.; Sadiq, S.; Humphreys, P.N.; Laws, A.P. A Novel Rhamnose-Rich Hetero-exopolysaccharide Isolated from Lactobacillus paracasei DG Activates THP-1 Human Monocytic Cells. Appl. Environ. Microbiol. 2017, 83, 2702–2718. [Google Scholar] [CrossRef] [Green Version]
- Lin, S.-W.; Tsai, Y.-S.; Chen, Y.-L.; Wang, M.-F.; Chen, C.-C.; Lin, W.-H.; Fang, T.J. An Examination of Lactobacillus paracasei GKS6 and Bifidobacterium lactis GKK2 Isolated from Infant Feces in an Aged Mouse Model. Evid.-Based Complement. Altern. Med. 2021, 2021, 1–9. [Google Scholar] [CrossRef]
- Kim, W.-K.; Jang, Y.J.; Seo, B.; Han, D.H.; Park, S.J.; Ko, G.P. Administration of Lactobacillus paracasei strains improves immunomodulation and changes the composition of gut microbiota leading to improvement of colitis in mice. J. Funct. Foods 2019, 52, 565–575. [Google Scholar] [CrossRef]
- Kim, W.-K.; Jang, Y.J.; Han, D.H.; Jeon, K.; Lee, C.; Han, H.S.; Ko, G.P. Lactobacillus paracasei KBL382 administration attenuates atopic dermatitis by modulating immune response and gut microbiota. Gut Microbes 2020, 12, 1819156. [Google Scholar] [CrossRef]
- Rossoni, R.D.; De Barros, P.P.; do Carmo Mendonça, I.; Medina, R.P.; Silva, D.H.S.; Fuchs, B.B.; Junqueira, J.C.; Mylonakis, E. The Postbiotic Activity of Lactobacillus paracasei 28.4 Against Candida auris. Front. Cell. Infect. Microbiol. 2020, 10, 397. [Google Scholar] [CrossRef]
- Xu, Y.; Tian, Y.; Cao, Y.; Li, J.; Guo, H.; Su, Y.; Tian, Y.; Wang, C.; Wang, T.; Zhang, L. Probiotic Properties of Lactobacillus paracasei subsp. paracasei L1 and Its Growth Performance-Promotion in Chicken by Improving the Intestinal Microflora. Front. Physiol. 2019, 10, 937. [Google Scholar] [CrossRef] [PubMed]
- Lv, X.-C.; Chen, M.; Huang, Z.-R.; Guo, W.-L.; Ai, L.-Z.; Bai, W.-D.; Yu, X.-D.; Liu, Y.-L.; Rao, P.-F.; Ni, L. Potential mechanisms underlying the ameliorative effect of Lactobacillus paracasei FZU103 on the lipid metabolism in hyperlipidemic mice fed a high-fat diet. Food Res. Int. 2021, 139, 109956. [Google Scholar] [CrossRef] [PubMed]
- Jam, S.A.M.; Talebi, M.; Alipour, B.; Khosroushahi, A.Y. The therapeutic effect of potentially probiotic Lactobacillus paracasei on dimethylhydrazine induced colorectal cancer in rats. Food Biosci. 2021, 41, 101097. [Google Scholar] [CrossRef]
- Ye, P.; Wang, J.; Liu, M.; Li, P.; Gu, Q. Purification and characterization of a novel bacteriocin from Lactobacillus paracasei ZFM54. LWT 2021, 143, 111125. [Google Scholar] [CrossRef]
- Bhat, B.; Bajaj, B.K. Hypocholesterolemic potential and bioactivity spectrum of an exopolysaccharide from a probiotic isolate Lactobacillus paracasei M7. Bioact. Carbohydr. Diet. Fibre 2019, 19, 100191. [Google Scholar] [CrossRef]
- Liu, C.-F.; Tseng, K.-C.; Chiang, S.-S.; Lee, B.-H.; Hsu, W.-H.; Pan, T.-M. Immunomodulatory and antioxidant potential of Lactobacillus exopolysaccharides. J. Sci. Food Agric. 2011, 91, 2284–2291. [Google Scholar] [CrossRef]
- Song, W.; Hu, P.; Guo, S.; Hu, J.; Song, C.; Wang, T.; Gao, Z.; Yue, T. Oxidative stress and endoplasmic reticulum stress contribute to L. paracasei subsp. paracasei M5L exopolysaccharide-induced apoptosis in HT-29 cells. Food Sci. Nutr. 2021, 9, 1676–1687. [Google Scholar] [CrossRef]
- Noda, M.; Sugimoto, S.; Hayashi, I.; Danshiitsoodol, N.; Fukamachi, M.; Sugiyama, M. A novel structure of exopolysaccharide produced by a plant-derived lactic acid bacterium Lactobacillus paracasei IJH-SONE68. J. Biochem. 2018, 164, 87–92. [Google Scholar] [CrossRef]
- Noda, M.; Sultana, N.; Hayashi, I.; Fukamachi, M.; Sugiyama, M. Exopolysaccharide Produced by Lactobacillus paracasei IJH-SONE68 Prevents and Improves the Picryl Chloride-Induced Contact Dermatitis. Molecules 2019, 24, 2970. [Google Scholar] [CrossRef] [Green Version]
- Sarikaya, H.; Aslim, B.; Yuksekdag, Z. Assessment of anti-biofilm activity and bifidogenic growth stimulator (BGS) effect of lyophilized exopolysaccharides (l-EPSs) from Lactobacilli strains. Int. J. Food Prop. 2017, 20, 362–371. [Google Scholar] [CrossRef] [Green Version]
- Zavala, L.; Golowczyc, M.A.; Van Hoorde, K.; Medrano, M.; Huys, G.; Vandamme, P.; Abraham, A.G. Selected Lactobacillus strains isolated from sugary and milk kefir reduce Salmonella infection of epithelial cells in vitro. Benef. Microbes 2016, 7, 585–595. [Google Scholar] [CrossRef] [PubMed]
- Bengoa, A.A.; Llamas, M.G.; Iraporda, C.; Dueñas, M.T.; Abraham, A.G.; Garrote, G.L. Impact of growth temperature on exopolysaccharide production and probiotic properties of Lactobacillus paracasei strains isolated from kefir grains. Food Microbiol. 2018, 69, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Bengoa, A.A.; Errea, A.J.; Rumbo, M.; Abraham, A.G.; Garrote, G.L. Modulatory properties of Lactobacillus paracasei fermented milks on gastric inflammatory conditions. Int. Dairy J. 2020, 111, 104839. [Google Scholar] [CrossRef]
- Bengoa, A.A.; Dardis, C.; Gagliarini, N.; Garrote, G.L.; Abraham, A.G. Exopolysaccharides from Lactobacillus paracasei Isolated from Kefir as Potential Bioactive Compounds for Microbiota Modulation. Front. Microbiol. 2020, 11, 583254. [Google Scholar] [CrossRef]
- Chen, C.; Li, J.; Zhang, H.; Xie, Y.; Xiong, L.; Liu, H.; Wang, F. Effects of a probiotic on the growth performance, intestinal flora, and immune function of chicks infected with Salmonella pullorum. Poult. Sci. 2020, 99, 5316–5323. [Google Scholar] [CrossRef] [PubMed]
- Duan, X.; Chen, S.; Duan, S.; Lan, C.; Yang, Z.; Cao, Y.; Miao, J. Antibiotic activities of the natural antimicrobial substance produced by Lactobacillus paracasei FX-6 against Pseudomonas putida. LWT 2020, 123, 109096. [Google Scholar] [CrossRef]
- Duan, X.; Duan, S.; Wang, Q.; Ji, R.; Cao, Y.; Miao, J. Effects of the natural antimicrobial substance from Lactobacillus paracasei FX-6 on shelf life and microbial composition in chicken breast during refrigerated storage. Food Control 2020, 109, 106906. [Google Scholar] [CrossRef]
- Bontsidis, C.; Mallouchos, A.; Terpou, A.; Nikolaou, A.; Batra, G.; Mantzourani, I.; Alexopoulos, A.; Plessas, S. Microbiological and Chemical Properties of Chokeberry Juice Fermented by Novel Lactic Acid Bacteria with Potential Probiotic Properties during Fermentation at 4 °C for 4 Weeks. Foods 2021, 10, 768. [Google Scholar] [CrossRef]
- Maehata, H.; Arai, S.; Iwabuchi, N.; Abe, F. Immuno-modulation by heat-killed Lacticaseibacillus paracasei MCC1849 and its application to food products. Int. J. Immunopathol. Pharmacol. 2021, 35, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Salminen, S.; Collado, M.C.; Endo, A.; Hill, C.; Lebeer, S.; Quigley, E.M.M.; Sanders, M.E.; Shamir, R.; Swann, J.R.; Szajewska, H.; et al. The International Scientific Association of Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of postbiotics. Nat. Rev. Gastroenterol. Hepatol. 2021, 18, 649–667. [Google Scholar] [CrossRef] [PubMed]
- Bengoa, A.A.; Zavala, L.; Carasi, P.; Trejo, S.A.; Bronsoms, S.; de los Ángeles Serradell, M.; Garrote, G.L.; Abraham, A.G. Simulated gastrointestinal conditions increase adhesion ability of Lactobacillus paracasei strains isolated from kefir to Caco-2 cells and mucin. Food Res. Int. 2018, 103, 462–467. [Google Scholar] [CrossRef] [PubMed]
- Garrote, G.L.; Abraham, A.G.; Rumbo, M. Is lactate an undervalued functional component of fermented food products? Front. Microbiol. 2015, 6, 629. [Google Scholar] [CrossRef] [PubMed]
- Iraporda, C.; Romanin, D.E.; Rumbo, M.; Garrote, G.L.; Abraham, A.G. The role of lactate on the immunomodulatory properties of the nonbacterial fraction of kefir. Food Res. Int. 2014, 62, 247–253. [Google Scholar] [CrossRef]
- Iraporda, C.; Errea, A.; Romanin, D.E.; Cayet, D.; Pereyra, E.; Pignataro, O.; Sirard, J.-C.; Garrote, G.L.; Abraham, A.G.; Rumbo, M. Lactate and short chain fatty acids produced by microbial fermentation downregulate proinflammatory responses in intestinal epithelial cells and myeloid cells. Immunobiology 2015, 220, 1161–1169. [Google Scholar] [CrossRef]
- Oleksy, M.; Klewicka, E. Exopolysaccharides produced by Lactobacillus sp.: Biosynthesis and applications. Crit. Rev. Food Sci. Nutr. 2018, 58, 450–462. [Google Scholar] [CrossRef] [PubMed]
- Rajoka, M.S.R.; Wu, Y.; Mehwish, H.M.; Bansal, M.; Zhao, L. Lactobacillus exopolysaccharides: New perspectives on engineering strategies, physiochemical functions, and immunomodulatory effects on host health. Trends Food Sci. Technol. 2020, 103, 36–48. [Google Scholar] [CrossRef]
- Ruas-Madiedo, P.; Abraham, A.G.; Mozzi, F.; de los Reyes-Gavilán, C.G. Functionality of exopolysaccharides produced by lactic acid bacteria in an in vitro gastric system. In Molecular Aspects of Lactic Acid Bacteria for Traditional and New Applications; Research Signpost: Kerala, India, 2008; pp. 137–166. ISBN 9788130802503. [Google Scholar]
- Ryan, P.M.; Ross, R.P.; Fitzgerald, G.F.; Caplice, N.M.; Stanton, C. Sugar-coated: Exopolysaccharide producing lactic acid bacteria for food and human health applications. Food Funct. 2015, 6, 679–693. [Google Scholar] [CrossRef]
- Korcz, E.; Kerényi, Z.; Varga, L. Dietary fibers, prebiotics, and exopolysaccharides produced by lactic acid bacteria: Potential health benefits with special regard to cholesterol-lowering effects. Food Funct. 2018, 9, 3057–3068. [Google Scholar] [CrossRef]
- Zeidan, A.A.; Poulsen, V.K.; Janzen, T.; Buldo, P.; Derkx, P.M.F.; Øregaard, G.; Neves, A.R. Polysaccharide production by lactic acid bacteria: From genes to industrial applications. FEMS Microbiol. Rev. 2017, 41, S168–S200. [Google Scholar] [CrossRef] [Green Version]
- Laws, A.; Gu, Y.; Marshall, V. Biosynthesis, characterisation, and design of bacterial exopolysaccharides from lactic acid bacteria. Biotechnol. Adv. 2001, 19, 597–625. [Google Scholar] [CrossRef]
- Yasuda, E.; Serata, M.; Sako, T. Suppressive Effect on Activation of Macrophages by Lactobacillus casei Strain Shirota Genes Determining the Synthesis of Cell Wall-Associated Polysaccharides. Appl. Environ. Microbiol. 2008, 74, 4746–4755. [Google Scholar] [CrossRef] [Green Version]
- Song, X.; Xiong, Z.-Q.; Kong, L.; Wang, G.; Ai, L. Relationship between Putative eps Genes and Production of Exopolysaccharide in Lactobacillus casei LC2W. Front. Microbiol. 2018, 9, 1882. [Google Scholar] [CrossRef] [Green Version]
- Stack, H.M.; Kearney, N.; Stanton, C.; Fitzgerald, G.F.; Ross, R.P. Association of Beta-Glucan Endogenous Production with Increased Stress Tolerance of Intestinal Lactobacilli. Appl. Environ. Microbiol. 2010, 76, 500–507. [Google Scholar] [CrossRef] [Green Version]
- Živković, M.; Miljković, M.S.; Ruas-Madiedo, P.; Markelić, M.B.; Veljović, K.; Tolinački, M.; Soković, S.; Korać, A.; Golić, N. EPS-SJ Exopolisaccharide Produced by the Strain Lactobacillus paracasei subsp. paracasei BGSJ2-8 Is Involved in Adhesion to Epithelial Intestinal Cells and Decrease on E. coli Association to Caco-2 Cells. Front. Microbiol. 2016, 7, 286. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Castro-Bravo, N.; Wells, J.M.; Margolles, A.; Ruas-Madiedo, P. Interactions of Surface Exopolysaccharides from Bifidobacterium and Lactobacillus Within the Intestinal Environment. Front. Microbiol. 2018, 9, 2426. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Y.; Cui, Y.; Qu, X. Exopolysaccharides of lactic acid bacteria: Structure, bioactivity and associations: A review. Carbohydr. Polym. 2019, 207, 317–332. [Google Scholar] [CrossRef]
- Angelin, J.; Kavitha, M. Exopolysaccharides from probiotic bacteria and their health potential. Int. J. Biol. Macromol. 2020, 162, 853–865. [Google Scholar] [CrossRef]
- Gibson, G.R.; Hutkins, R.; Sanders, M.E.; Prescott, S.L.; Reimer, R.A.; Salminen, S.J.; Scott, K.; Stanton, C.; Swanson, K.S.; Cani, P.D.; et al. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 491–502. [Google Scholar] [CrossRef] [Green Version]
- Salazar, N.; Gueimonde, M.; de los Reyes-Gavilán, C.; Ruas-Madiedo, P. Exopolysaccharides Produced by Lactic Acid Bacteria and Bifidobacteria as Fermentable Substrates by the Intestinal Microbiota. Crit. Rev. Food Sci. Nutr. 2016, 56, 1440–1453. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Xie, Y.H.; Han, T.; Zhang, H.X. Purification and Structure Study on Exopolysaccharides Produced by Lactobacillus paracasei KL1-Liu from Tibetan Kefir. Adv. Mater. Res. 2013, 781–784, 1513–1518. [Google Scholar] [CrossRef]
| L. paracasei Strain | Origin | Health Functional Properties | Biological Activity * | Reference |
|---|---|---|---|---|
| Lpc-37® | DuPont de Nemours, Inc. trademark | Stress, mood and well-being modulation | Reduction of perceived stress and improvement of biomarkers related to stress in a clinical trial. | [13] |
| GKS6 | Healthy infant feces | Antioxidant activity | Delay in the aging process in mice by enhancement of antioxidants activity, resulting in lower oxidative damage. | [50] |
| KBL 382 | Korean healthy feces | Anti-inflammatory activity—Microbiota modulation | Reduction of INF-γ, IL-4, IL-6, TNF and IL-17A levels and increase of anti-inflammatory cytokine IL-10 and CD4+CD25+Foxp3+ T regulatory cells in mesenteric lymph nodes levels. Improvement of cell tight junction and mucus thickness. Increase in bacterial diversity of fecal microbiota. | [51] |
| Ameliorates atopic dermatitis—Immunomodulatory activity—Microbiota modulation | Decrease in T helper cytokines and increase IL-10 and TGF-β production in skin tissue. Increase in the proportion of CD4+ CD25+ Foxp3+ T regulatory cells in mesenteric lymph nodes and changes in the composition of gut microbiota of oral treated mice. | [52] | ||
| 28.4 | Oral cavity of a caries-free individual | Immunomodulatory activity | Bacteria cells have antifungal activity against planktonic cells, biofilms and persisted cells of Candida auris. | [53] |
| Postbiotic elements (free-cell supernatant) inhibit C. auris in vitro and protect Galleria mellonella infected with C. auris enhancing its immune status. | ||||
| L1 | Sweet potato sour liquid | Microbiota modulation | Increase in the abundance of functions related to carbohydrate and protein metabolism and fatty acid biosynthesis in the intestinal microbiota. Improvement in the growth performance of chicken. | [54] |
| FZU103 | Traditional Hongqu rice wine | Improvement of lipid metabolism | Regulation of lipid metabolic pathways of pathogen free mice feed with high fat diet (fatty acid degradation, fatty acid elongation, unsaturated fatty acids biosynthesis, glycerolipid, glycerophospholipid and arachidonic acid metabolism, primary bile acid biosynthesis and riboflavin metabolism). Regulation of the expression of hepatic genes involved in lipid metabolism and bile acid homeostasis and promotion of fecal excretion of bile acids. | [55] |
| B-14 | Traditional fermented dairy product | Antiproliferative—Proapoptotic effects | Downregulation or upregulation of key genes in the cell proliferation, cell survival and intrinsic and extrinsic apoptosis pathways. | [56] |
| ZFM54 | New-born infant’s feces | Protection against foodborne pathogens | In vitro inhibition of Salmonella typhimurium, Micrococcus luteus and Listeria monocytogenes by production of a pore forming bacteriocin ZFM54. | [57] |
| M7 | Human breast milk | Hypocholesterolemic—Antioxidant activity—Protection against pathogens | Antibiofilm potential of EPS against several human pathogens. | [58] |
| NTU 101 | Human feces | Immunomodulatory—Antioxidant activity | Induction of pro-inflammatory molecules (nitric oxide, IL-6, TNFα and IL-1β) and phagocytic activity in murine macrophages Raw 264.7. Antioxidant activity (scavenging of 1,1-Diphenyl-2-picrylhydrazyl radicals, inhibition of linoleic acid peroxidation, reducing power, chelating ability on ferrous ions). | [59] |
| M5L | Kumiss | Antiproliferative activity | Apoptotic effect on human colorectal adenocarcinoma cell line HT-29 mediated by induction of oxidative stress and endoplasmic reticulum stress. | [60] |
| DG | Commercial product | Immunomodulatory activity | Induction of pro-inflammatory cytokines TNF α, IL-6 and the chemokines IL-8 and CCL20 in human monocytic cell line THP-1. | [49] |
| IJH SONE | Fig leaf | Anti-inflammatory activity | Anti-inflammatory effect mediated by inhibition of hyaluronidase activity. | [61] |
| Antiallergenic activity | Antiallergenic effect evidenced by oral and topic administration against contact dermatitis in mice. | [62] | ||
| LB-8 | Feces | Modulation of intestinal microbiota | Bifidogenic effect in vitro. | [63] |
| CIDCA 8339, 83120, 83121, 83123, 83124 | Kefir grains (Argentine) | Protection against pathogens | Adhesion to Caco-2 cells and prevention of Salmonella association/invasion in vitro depending on surface properties of the strain. | [64] |
| CIDCA83123, 83124, 8339 | Kefir grains (Argentine) | Immunomodulatory activity | Modulation of the intestinal epithelial innate immune response by viable whole cell. | [65] |
| Immunomodulatory activity | Fermented milk supernatants downregulate the induced innate immune response in intestinal and gastric cells, with lactate as the metabolite responsible of this effect. | [12,66] | ||
| CIDCA 83124, 8339 | Kefir grains (Argentine) | Microbiota modulation and changes in SCFA profile | EPS 8339 and EPS 83124 modify the microbiota by reducing the enterobacteria and increasing the production of propionic and butyric acid. | [67] |
| CIDCA 8339 | Kefir grains (Argentine) | Gastroprotection | Adhesion to AGS gastric cell line. The strain consumption shows a gastroprotective effect in an acute gastritis murine model. | [66] |
| Ž2 | Kefir grains (Tibet) | Immunomodulatory activity | Increase in the proportion of all T cells (CD3+), CD4+ lymphocytes and the ratio of CD4+:CD8+ cells in vivo and increase in the gene expression for mucins (MUC-1 and MUC-2) and IgA at intestinal level. | [43] |
| KL1-Liu | Kefir grains (Tibet) | Protection against pathogens | Mixed probiotic of L. paracasei KL1-Liu (EPS producer) and L. plantarum Zhang-LL reduces the mortality of pullorosis in chicks. | [68] |
| MRS59 | Kefir grains (Brazil) | Adhesion to intestinal epithelial cells | Adhesion to human Caco-2 epithelial cells, bacteriocin production. | [42] |
| Antimicrobial-Antioxidant activity | Antagonistic activity against food pathogens by bacteriocin-like inhibitory substance and antioxidative activity of cell extract. | |||
| FX6 | Kefir grains (Tibet) | Antimicrobial activity | Bacteriostatic effect on Pseudomonas putida due to the increase of bacterial membrane permeability and ability of the antimicrobial substance to affect the synthesis of protein and bind to genomic DNA. | [69] |
| Antimicrobial activity | Bacteriocin F1 with a wide antimicrobial spectrum. | [38] | ||
| Antimicrobial activity | Antibacterial effect on Serratia in chicken breast during refrigerated storage. | [70] | ||
| LAB2, LAB4 | Kefir grains (Iran) | Protection against pathogens | Neutralized cell-free supernatant inhibits the growth and the biofilm formation by uropathogenic E. coli. | [37] |
| SP5 | Kefir grains (Russia) | Antiproliferative activity | Reduction of cancer cell proliferation in vitro in a time- and concentration-dependent manner. | [40] |
| SP5 | Antioxidant activity | Fermentation metabolites produced by the breakdown of anthocyanins and other larger-in-size phenolic compounds present in chokeberry juice, leading to increased levels of total phenol content. | [71] | |
| AGR4 | Kefir grains (Greece) | Antiproliferative activity | Time- and dose-dependent antiproliferative activity of HT-29 cells and human melanoma cell line A375. | [41] |
| LMG R40086, LMG R39998, LMG R40122, LMG R40006 | Water kefir grains(Belgium) | Adhesion to intestinal epithelial cells | Adhesion ability to Caco-2 cells. | [64] |
| CT12 | Water kefir grains (Mexico) | Antimicrobial-Antioxidative activity | Antimicrobial, antifungal and antioxidant capacity of cell-free supernatant. | [39] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bengoa, A.A.; Dardis, C.; Garrote, G.L.; Abraham, A.G. Health-Promoting Properties of Lacticaseibacillus paracasei: A Focus on Kefir Isolates and Exopolysaccharide-Producing Strains. Foods 2021, 10, 2239. https://doi.org/10.3390/foods10102239
Bengoa AA, Dardis C, Garrote GL, Abraham AG. Health-Promoting Properties of Lacticaseibacillus paracasei: A Focus on Kefir Isolates and Exopolysaccharide-Producing Strains. Foods. 2021; 10(10):2239. https://doi.org/10.3390/foods10102239
Chicago/Turabian StyleBengoa, Ana Agustina, Carolina Dardis, Graciela L. Garrote, and Analía G. Abraham. 2021. "Health-Promoting Properties of Lacticaseibacillus paracasei: A Focus on Kefir Isolates and Exopolysaccharide-Producing Strains" Foods 10, no. 10: 2239. https://doi.org/10.3390/foods10102239
APA StyleBengoa, A. A., Dardis, C., Garrote, G. L., & Abraham, A. G. (2021). Health-Promoting Properties of Lacticaseibacillus paracasei: A Focus on Kefir Isolates and Exopolysaccharide-Producing Strains. Foods, 10(10), 2239. https://doi.org/10.3390/foods10102239






