Abstract
Background: Surgical treatments for snoring and sleep-related symptoms are invasive, may have complications, have inconstant success rates, and may have a significant relapse rate. Methods: We evaluated the effectiveness of a non-surgical, non-invasive outpatient 808 nm diode laser treatment. Forty-five patients with snoring and sleep disorders were treated with an Elexxion-Claros® 50W Diode Laser 808-nm using the specific SNORE3 application. The possible presence of obstructive sleep apnea syndrome in patients was diagnosed with Polysomnography. The Epworth sleepiness scale was reported pre- and post-laser treatments. Patients were classified according to both Friedman tongue position and Mallampati classification. During treatment, the pain reported by patients was measured on a visual analogue scale. Results: The following data improved significantly: visual analogue scale for the loudness of snoring, Epworth sleepiness scale, waking up during sleep because of snoring, dreaming during the night, dry mouth on awakening, daytime sleepiness, tiredness on awakening, Mallampati score, Friedman tongue position and degree of oropharynx at nose, oropharynx, hypopharynx, and larynx classification (p < 0.001). A cessation of choking in 89% of the patients and of restless legs syndrome in 83% of the patients (p ≤ 0.005 and p ≤ 0.025) was also noted after the laser treatments. All of the patients who reported suffering from headaches upon waking (10 subjects) reported the complete cessation of this phenomenon after laser therapy. Conclusions: diode laser treatment is a promising procedure not only in reducing the loudness of snoring, but also in raising the global quality of sleep, a statistically significant reduction of AHI, and a cessation of bothersome phenomenon related to apnea. Results were sustainable at one year post-treatment.
1. Introduction
Sleep is mandatory for human life and optimum functioning of the human body and its organ systems [1,2].
Snoring can be dangerous for patients because it can generate an obstructive sleep apnea syndrome (OSAS), or mask an existing OSAS, which is correlated with several cardiovascular [3], neurologic [4], psychiatric [5], and metabolic disorders [6]. OSAS affects 1–5% of children 2–8 years of age [7,8]. In adults, moderate–severe OSAS affects 23.4% of female and 49% of male subjects, and these data may increase with aging and weight gain [9,10]. OSAS in adults is also a major risk factor for car accidents through excessive sleepiness because of poor sleep quality [11].
Snoring is caused by the vibration of the oropharyngeal soft tissues, as a consequence of a partial obstruction of the upper airway [12]. The main sources of vibration in adults are represented by the soft palate and lateral pharyngeal walls, which may cause snoring when redundant tissue is present. Furthermore, hypertrophic tonsils, an enlarged base of the tongue or/and, in a minority of cases, a collapsible epiglottis that blocks the airway (floppy epiglottis) [12,13] can also cause snoring.
In adults, simple snoring or light OSAS often requires behavioural changes and may be treated through various surgical procedures (palatal implants, snoreplasty, radiofrequency assisted uvuloplasty, radiofrequency ablation, uvulopalatopharyngoplasty) or by wearing a mandibular advancement device (MAD) at night. [14,15,16,17,18]. The gold standard for moderate or severe OSAS is c-PAP, or alternatively several surgical procedures: uvulopalatopharyngoplasty (UPPP), palatal implants, genioglossus advancement, hyoid suspension, radiofrequency tongue ablation, reduction glossectomy, transoral robotic surgery base-of-tongue reduction (TORS BOT), maxillomandibular osteotomy (MMO), and advancement (MMA).
In general, surgical treatments may have complications, low success rates, significant relapse rates, and are invasive [19,20,21]. The peer-reviewed literature lists many morbidities post-UPPP surgery, including haemorrhage wound dehiscence, velopharyngeal insufficiency, difficulty swallowing, taste disturbances, voice changes, globus sensation, and dry pharynx [21]. Currently, people are less willing to undergo invasive procedures and if possible tend to avoid surgery or wearing a MAD [22]. These are all reason why our group is trying to develop new treatments for sleep disorder Recently, we demonstrated [23] in a prospective series of 40 patients that a non-surgical, minimally invasive outpatient Er: YAG laser treatment was effective in reducing snoring and improving the quality of sleep; these data are supported by the literature [24]. Diode lasers have been used for surgical treatment of sleep apnea [25,26]. However, to date, no study has been performed using an 808 nm diode laser device for non-ablative treatment of snoring and sleep disorders. The combination of high laser energy (50 W maximum) with a short pulse duration (9 μm as a minimum) with a corresponding frequency of 20,000 Hz as a maximum was used to achieve a tightening of the slackened oropharyngeal tissues with shrinkage of soft tissue collagen fibres due to the affinity of this wavelength for haemoglobin, creating a painless, non-surgcal photothermal effect.
The purpose of this study was to evaluate the effect of diode laser light on sleep-related symptoms. The specific aim of our pilot-study was to detect the effectiveness of an 808 nm laser (Elexxion Claros® 50W SNORE 3 application) on snoring on 45 patients (23 out of 45 with OSAS).
2. Materials and Methods
2.1. Ethical Considerations
This study followed the Declaration of Helsinki on medical protocol and ethics. The Regional Ethical Review Board of Department of Surgical Sciences and Integrated Diagnostics (University of Genoa) approved the study (Unige-Disc-Prot./9619). A written informed consent with clear information about the clinical study, including the research procedure and purpose as well as risks and benefits, was obtained from all patients, who voluntarily agreed to undergo the procedure and declared their willingness to return at regular intervals for evaluation.
2.2. Compliance with Ethical Standards and Study Population
The study was carried out according to the STROBE statement.
Forty-five Caucasian patients (62.2% males and 37.8% females, average age 55 years) who presented for snoring-cessation therapy to the private practice of one of the authors were selected for this study. The patients were treated and followed-up at the Department of Surgical Sciences and Integrated Diagnostics (University of Genoa), from January 2017 to October 2019.
The presence of OSAS in patients was diagnosed with polysomnography (CareFusion Noxturnal) performed at the homes of the patients themselves and analyzed by the same operator pre- and post-laser treatments.
In accordance with the previous study of our group [23], at the first visit all patients underwent an ear, nose, and throat (ENT) examination by fiberoptic nasopharyngoscopy (Xion, Germany). A Muller maneuver (FNMM) was performed [27] in the supine position without any pillow. According to the literature for surgical planning, “identifying the site and the dynamic pattern of obstruction is mandatory in therapeutic decision-making, and in particular if a surgical therapy option is taken into account”. [28]. Therefore, to make precise planning, even for a non-surgical approach such as ours, a Muller test with nasopharynx cavity and walls, oropharynx, hypopharynx, and larynx (NOHL) classification [28] was performed in order to evaluate the sites of obstruction and eliminate cases with non-appropriate situations, such as a floppy epiglottis causing the laryngeal obstruction. Unfortunately, this site is not suitable for laser treatment; therefore, these patients were excluded from this cohort, as well as patients with a collapse of more than 50% of epiglottis at the NOHL classification (L3 at NOHL), due to the inability to check a floppy epiglottis during a DISE, which was refused by all of the patients. The presence of indirect signs of laryngo-pharyngeal reflux (LPR) was detected with video-fibroscopy (Karl Storz, Germany) by Reflux Finding Score (RFS) [29], and the patients self-scored discomfort by filling in the Reflux Symptom Index (RSI) [30,31].
The patients with OSAS were offered other evidence-based treatments such as wearing positive airway pressure (c-PAP) devices in moderate or severe OSAS, surgery, or wearing mandibular advancement device (MAD), but they all refused to accept treatments considered invasive or uncomfortable. The patients with OSAS were advised that in case of failure of treatment in reducing apneas with diode laser sessions, they could eventually have a multi-step strategy of treatment using the conventional treatments with an increasing level of invasiveness, assessing results step by step with polysomnography.
Before initiating treatment, informed consent was obtained, and patients with moderate or severe OSAS stated their refusal of conventional therapies. The study’s exclusion criteria were pediatric patients, pregnant women (for medico-legal reasons), patients with central apneas, Reinke’s chronic vocal fold oedema, FNMM: N > or = of 3, H > or = of 3, L > or = of 3 at NOHL, tonsil size > 50% of obstruction of the oropharynx (as noticed during the previous study of our group [23] the results were better in patients with tonsils obstructing less than 50% of the oropharynx), nasal polyposis, or other obstructive masses of the nose, rhynopharynx, or hypopharynx. Conversely, the study’s inclusion criteria were simple snoring and OSAS (any degree of severity), only if patients refused any other kind of conventional treatment that was accurately described.
2.3. Laser Treatment Description (The Fini Storchi–Unige Protocol)
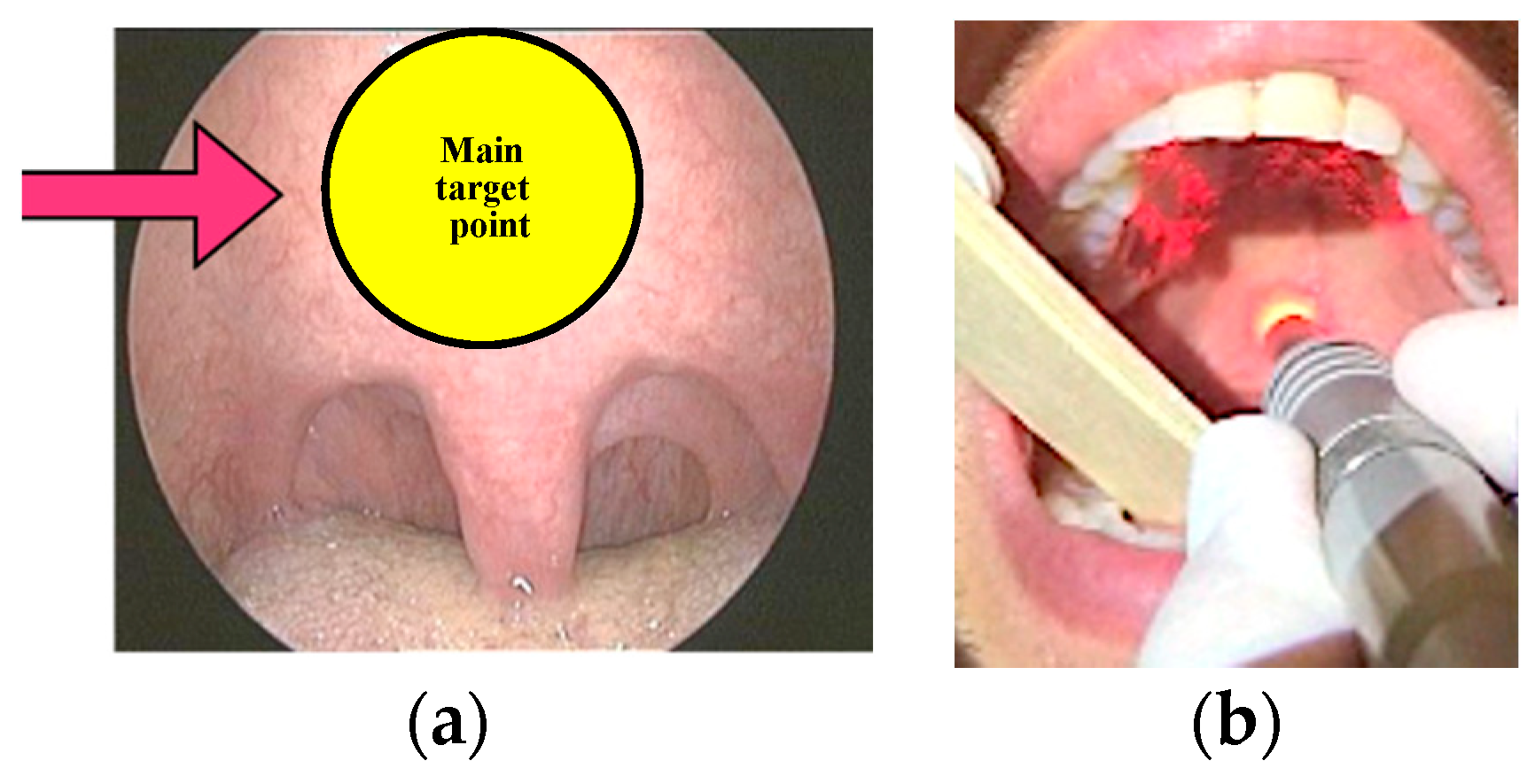
The patients were all treated in an outpatient setting with the Diode 808 nm (Claros® 50 W Elexxion-Germany) using the specific SNORE 3 application (Elexxion has not disclosed details on the actual laser settings). During these kinds of treatments, in the personal experience of the authors, there is usually no need for topical anaesthetics. The sessions were performed with no anaesthetics, and all the patients were informed that they could stop the procedure and ask at any moment for a topical anaesthetic. In fact, what patients perceived and reported was the heat, which could be overcome by moving the laser handpiece when the temperature was no longer tolerable for the patient or using topical anaesthetics if requested. Any gag reflexes were overcome by using relaxing breathing techniques, acupuncture point stimulations, or in the most severely symptomatic patients, a topical lidocaine spray (Lidocain topical spray 10 mg/100 mL—Ecocain Molteni Dental). The procedure began with irradiation of at least eight passes of all regions with an overlap covering the entire mucosal surface of the soft palate and uvula; tonsillar regions including the anterior and posterior pillars, using the straight glass rod (Figure 1); and the base of the tongue behind the circumvallate papillae with the curved glass rod as far as the anatomy of the patient and their tolerance permitted (Figure 2). The irradiation was performed in non-contact and a non-standard mode for the absence of a millimiter spacer by keeping the hand-piece at 3–5 mm of distance from the target tissue. At the end of the session, an “over boost” of 10 min with the same settings previously used was performed at the centre of the soft palate, beginning from the junction of the hard and soft palate, on an area of about 3 × 3 cm, where the vibration of the soft palate itself initiates; this area is called “the main target point” by the authors (Figure 3a,b). After the 10 min overboost, three additional minutes of laser irradiation on the main target point were given with the following parameters: 20 W of laser energy with a mean output of 4 W, frequency of 20,000 Hz, and a 10-microsecond pulse. The amount of treatment per region and patient was based on the severity of the Muller test score, the anatomy of the patient, the presence of OSAS, and the severity of symptoms. Therefore, the treatment time varied from a minimum of 40 min to a maximum of 65 min.
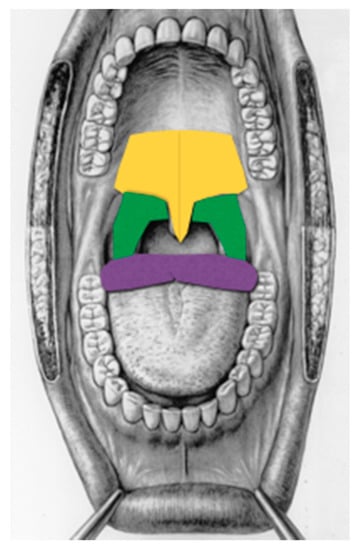
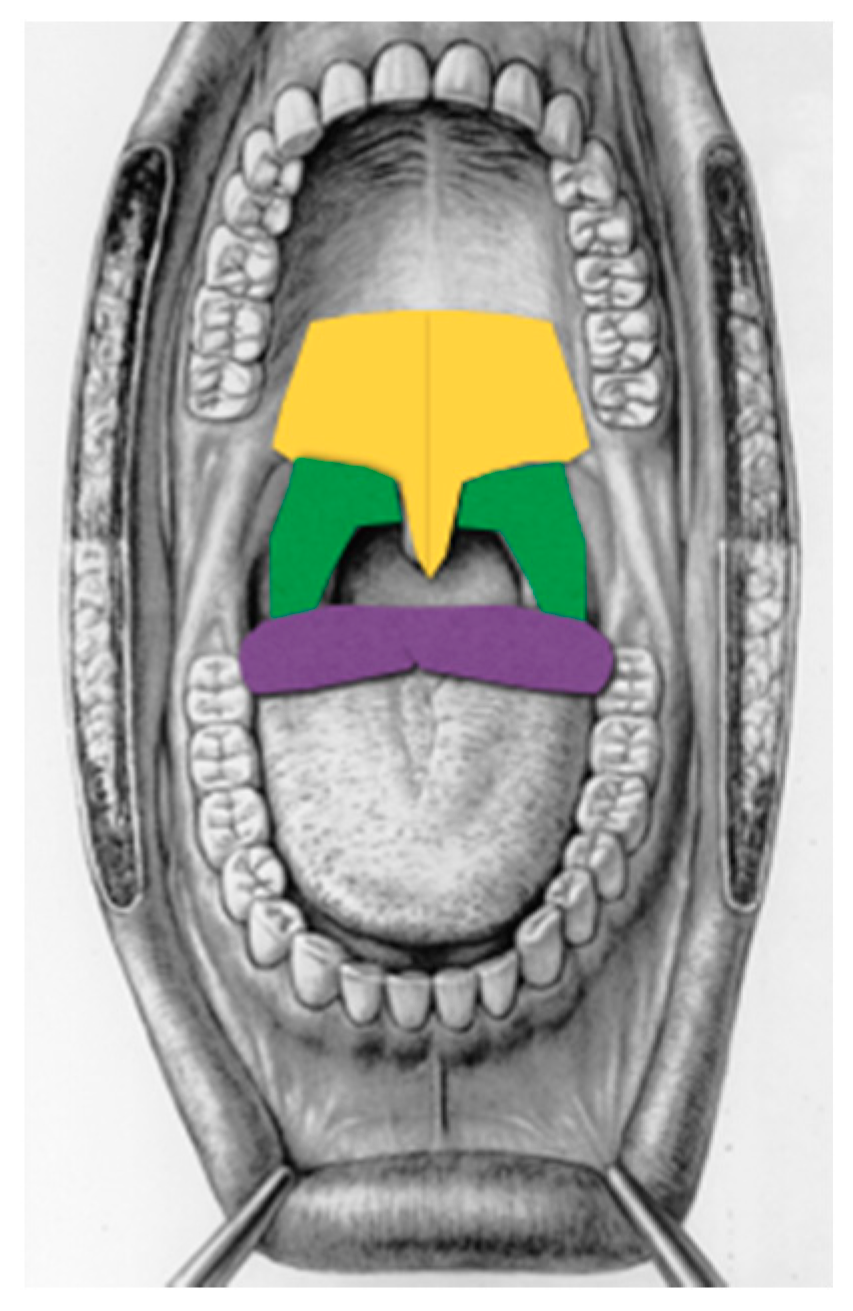
Figure 1.
Treated regions (Fini Storchi et al., 2018, modified). Yellow = soft palate, green = tonsil region, purple= base of the tongue.
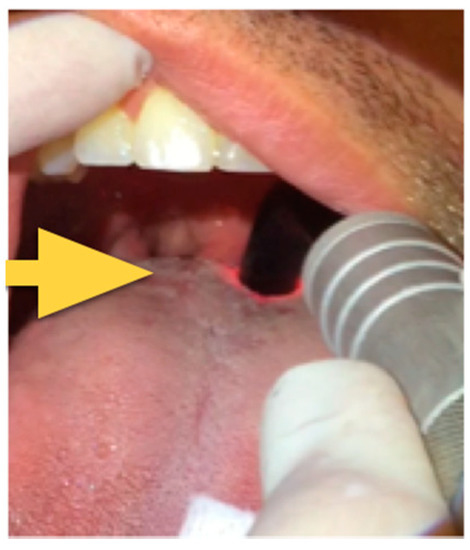
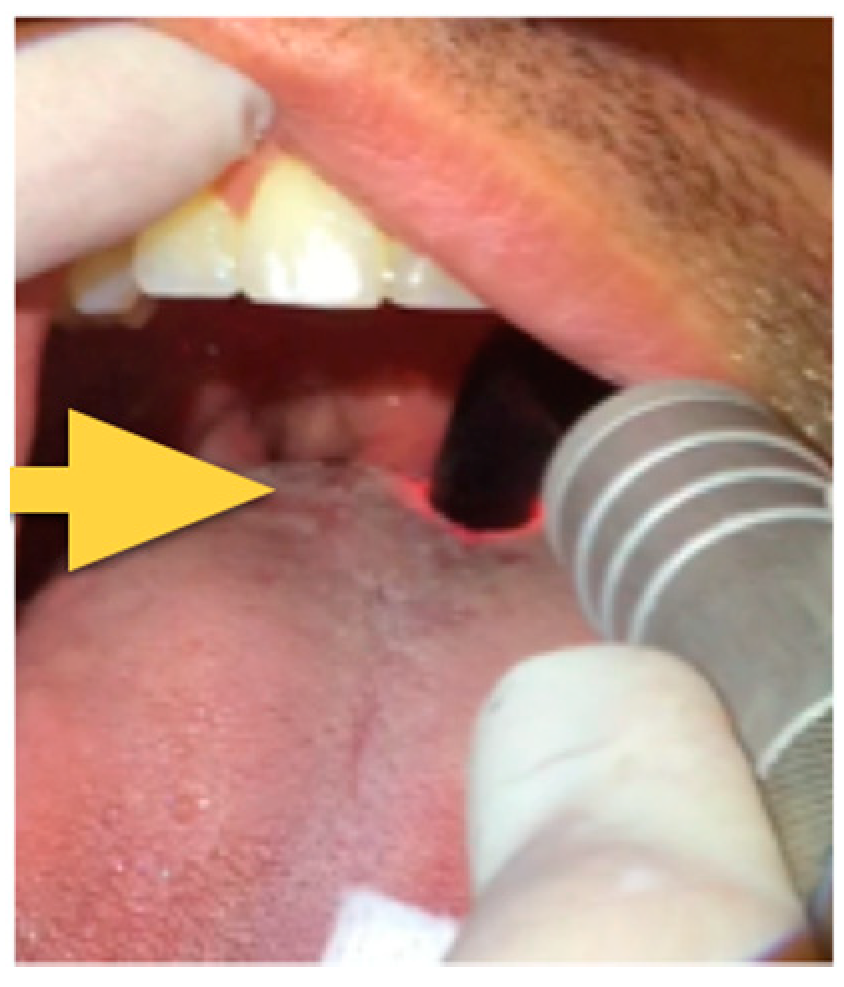
Figure 2.
Base of tongue treatment using the curved glass rod.
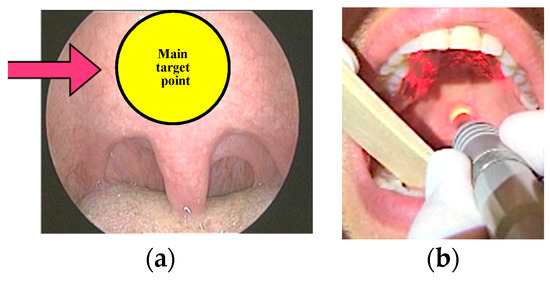
Figure 3.
(a) = The “main target point” (Fini Storchi et al., 2018); (b) = Over boost in the “main target point” (Fini Storchi et al., 2018).
Table 1 reports the Fini Storchi–Unige Protocol selection criteria, and Table 2 reports the Fini Storchi–Unige Protocol laser treatment description.

Table 1.
The Fini Storchi–Unige Protocol. Selection criteria.

Table 2.
The Fini Storchi–Unige Protocol. Treatment.
All patients received at least three laser sessions, and nine out of 12 patients with severe OSAS received four sessions that were performed at 7–10 days apart.
2.4. Outcome Measures
Results were examined by submitting a questionnaire with the same questions to the patient and his/her bed partner pre- and post-laser treatment session. To reduce bias, all questionnaires were submitted to the patient and his/her partner by the same researcher, before and after treatment. In order to quantify the efficacy of the laser treatment, Table 3 reports subjective outcomes that were scored pre- and post-laser treatment by each patient and his/her bed partner using a visual analogue scale (VAS) from 1 to 10 (Table 2) and the examination parameters.

Table 3.
Outcome measures of snoring loudness, quality of sleep questionnaire, and examination parameters.
The Epworth sleepiness scale [31] (ESS) and patient reports of headache, choking, daytime sleepiness, and restless legs were reported pre- and post-laser treatment.
Patients were classified according to Friedman tongue position (FTP) [32] and to Mallampati classification as reported in Table 3. All questionnaires, evaluation of Mallampati, FTP, and Muller test with NOHL were performed before and after laser treatments by the same author to reduce bias.
The efficacy of the treatment on the OSAS patients was tested with polysomnography by using the same polygraph (CareFusion Noxturnal) and by the same referent.
During treatment, the discomfort reported by the patients due to heat of the laser was measured on a VAS from 0 (not bothersome) to 5 (severely bothersome), and at the end of the treatment, the patient valued his/her immediate perception of a wider throat from a range from 0 (no wider throat) to 5 (much wider throat).
2.5. Statistical Analysis
In accordance with the previous study of our group [23], “continuous variables were summarized as median (1st–3rd quartiles) and categorical variables as frequencies and percentages. Comparison of quantitative variables between pre- and post-treatment was made by Wilcoxon matched-pairs signed-ranks test, and comparisons of frequencies were made by McNemar test. Values of p < 0.05 were considered statistically significant. All analyses were performed using SAS version 9.3 (SAS Institute, Cary, NC, USA).
3. Results
3.1. Overall Population
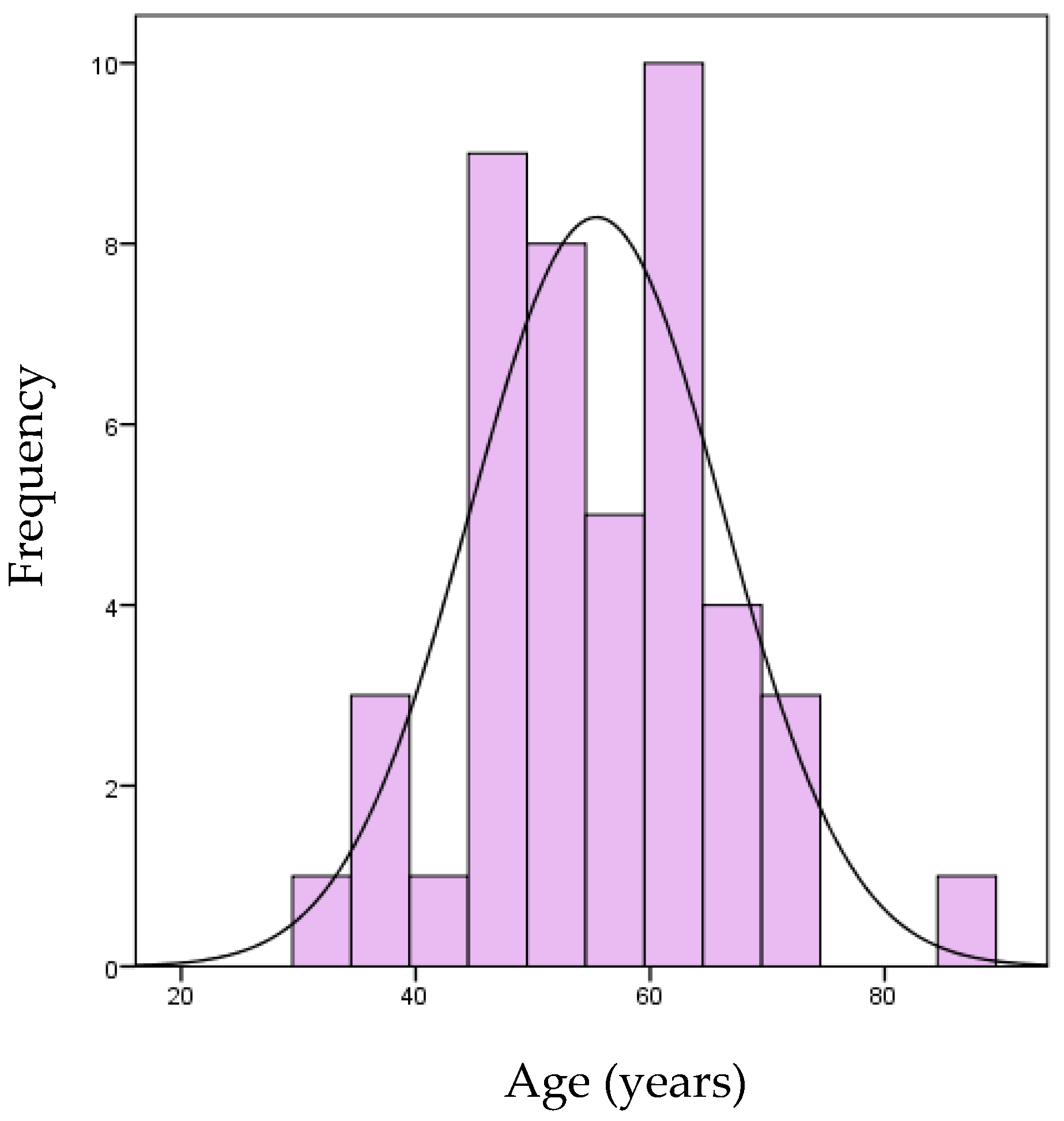
Demographic and clinical characteristics of patients are reported in Figure 4 and Table 4. Twenty-two subjects (48.9%) had a BMI (Body Mass Index) lower than 25, 19 subjects (42.2%) were overweight, and four subjects (8.9%) were obese.
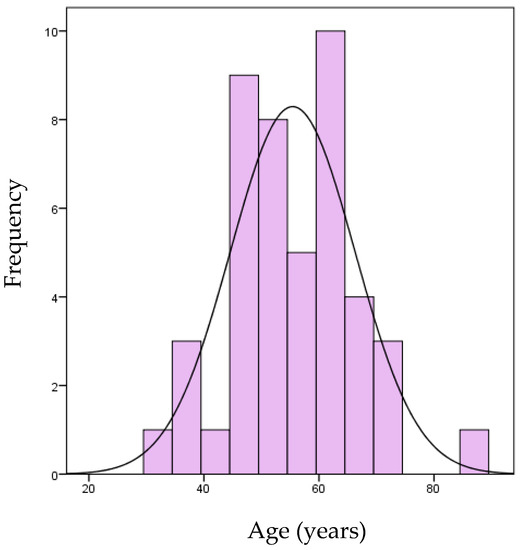
Figure 4.
Age of patients of the case series.

Table 4.
Demographic and clinical characteristics of the 45 patients enrolled at baseline. Data are expressed as medians (1st–3rd quartiles) or absolute frequencies (percentage) unless otherwise indicated.
According to the Mallampati classification, twenty-six patients (57.8%) were classified at baseline as class 4, 15 patients as class 3 (33.3%), and four patients as class 2 (8.9%).
OSAS cases were diagnosed with polysomnography in 23 out of 45 patients, which all showed suspected altered ESS symptoms or apnea noted by the partner, or choking.
Of the 23 OSAS patients (51.1%), one patient had an Apnea–Hypopnea Index (AHI) score from 5 to 15 (light OSAS), 10 patients had an AHI score ranging from 15 to 30 (moderate OSAS), and 12 patients an AHI score greater than 30 (severe OSAS); nine patients reported frequent choking during sleep. All refused all of the conventional treatments that were described (c-PAP, surgery, or MAD for moderate or severe OSAS, depending on the site of collapse). Nine of the 12 with severe OSAS underwent four laser sessions. Indirect signs of LPR were present in the great majority of the sample population (86.7%).
The median discomfort reported by patients after every session was 1 point (0–2) on the 0–5 bother scale.
Only 4/45 patients (8.8%) received topical lidocaine spray at the first session for intolerance or gag reflexes. Only one patient asked for nitrous oxide to cope with anxiety and gag reflexes in all the three sessions.
Side effects were noticed only on three patients showing minor transitory complications. One patient had moderate oedema of the uvula that required a 4 mg Betametasone injection for two days, one patient that used warfarin had a small (4 millimetres) palatal ecchymosis that reabsorbed spontaneously, and one patient (2.2%) reported a transitory mild sore throat that did not require painkillers and resolved spontaneously.
3.2. Laser Treatments Results
Of the 26 patients in class 4 according to the Mallampati classification, 22 (84.6%) were reclassified to class 3 after the therapy, while only four patients did not change their classification (p < 0.0001). Of 36 subjects classified as class 4 by the FTP scale, ~70% were in class 3 at the end of the therapy (p < 0.0001); the oropharynx retropalatal collapse grade of obstruction at the NOHL (“O”) classifications was also significantly improved after the diode laser treatment (p < 0.0001).
All the patients’ assessments about snoring reduction after laser treatment were presented in Table 5.

Table 5.
Comparisons of patient’s general assessment of snoring reduction variables before and after laser treatment.
The median snoring VAS scores of daytime sleepiness, dryness of mouth at wake-up time, difficulty in waking in the morning, feeling of tiredness on awakening, and waking up during sleep because of snoring were all significantly decreased after the laser treatments.
The quality of sleep and the intensity of dreaming perceived by the patients both showed a significant increase (p < 0.0001 for both).
Notably, seven patients who reported that they never used to dream in the five years before the therapy started to dream again from the day after the first session. Eighty-nine per cent of patients who reported choking and 83% of patients who reported restless leg syndrome before the treatment reported a total cessation of these symptoms (p = 0.005 and p = 0.025, respectively) after the laser treatments. Patients were questioned about their own satisfaction after the three laser therapy sessions. Of the 44/45 treated patients, 27 (61.3%) were very satisfied, 15 patients (34.09%) reported being fairly satisfied with the therapy, and only two subjects (4.4%) were not satisfied. After the laser treatments, 30/38 (78.9%) patients experienced an immediate sensation of breathing improvement, and 37 patients (82.2%) reported to have a sensation of wider throat.
All patients suffering from headaches upon waking (10 cases) and all patients with some difficulty in equalizing ear pressure during landing when flying (13 cases) reported a complete cessation of these phenomena following laser therapy.
The reduction of daily sleepiness measured with ESS in patients with an altered ESS before treatments was statistically significant (p < 0.0001).
The reduction of AHI measured by polysomnography was also significant (see Table 5) from X= 29 (23.9–42.2) to X = 16.8 (11.5–31) after the treatment.
Of note, of the 24 sexually active males in our sample, five (20.8%) reported an improvement in their sex life after the laser treatment.
An exclusion criterion of the study was patient significant weight loss (more than 2 kg). No patients were excluded for weight loss.
3.3. Subgroup Analysis
Some subgroup analyses were performed to evaluate the laser treatment effect in a subgroup of patients defined by baseline characteristics. Regarding BMI, patients were classified into two groups: normal weight and overweight or obese patients. The change pre- and post-laser treatment in the quality of sleep, the feeling of restlessness upon awakening, and daytime sleepiness was significantly different in the two groups (p = 0.03, p = 0.05, and p = 0.009, respectively) with overweight/obese patients seeming to benefit the most from the laser treatment. The change pre- and post-laser treatment in the rating of snoring intensity (p = 0.03), quality of sleep (p = 0.001), feeling of restlessness upon awakening (p = 0.0005), and daytime sleepiness (p < 0.0001) was significantly different in patients with and without OSAS. In our sample, patients with OSAS appeared to benefit the most from the laser treatment.
3.4. Follow-Up
The follow-up was made on 37 of 45 patients. Eight were excluded, for the following reasons:
(a) Three patients with severe OSAS cases decided to use c-PAP despite the improvements obtained with laser because their AHI was still over 15 and they were strongly encouraged to use c-PAP.
(b) Three were lost to follow up.
(c) Two did not have any improvement with laser sessions.
After a median of 12 months of follow-up (range 7–26 months), the 37 patients were asked to assess the sustainability of the treatment effect, especially regarding the snoring reduction and the quality of sleep. The majority of patients (28/37, 75.6%) stated that the improvement they obtained with the treatment was significant and sustainable, 5/37 (13.5%) stated that there was a partial relapse, and only four (10.8%) patients had relapsed by follow-up time.
The patients who had the partial loss of results underwent a new laser session and reported that the sleep parameters improved and were invited to perform a sleep study in order to see if there was the need to do further treatment planning. An interesting anecdotal finding during the study was that seven out of seven patients who suffered from recurrent tonsillitis (more than three episodes per year) never had another similar episode during the period of follow up.
4. Discussion
To date, no study has been performed using an 808 nm diode laser device for non-ablative treatment of snoring and sleep disorders. The specific aim of our pilot-study was to treat patients with snoring and apneas in a non-invasive way with a diode 808-nm laser. In fact, currently people tend to avoid invasive treatments for snoring and sleep disorders. Many patients avoid facing their sleeping problems due to of the invasiveness of surgery and of wearing an unpleasant mask for the rest of their life. All patients of our study actually refused any other type of invasive or uncomfortable solution for their snoring or apneas, even if they were informed about the risks that could occur when significative apneas were present.
The role of lasers has been expanded in otolaryngology according to the specific laser wavelength and dosimetry parameters. Cetinkaya et al. [24] showed that the NightLase TM Er:YAG laser (Fotona, Slovenia) treatment for snoring was useful in the treatment of simple snorers in an outpatient setting, and we recently demonstrated [23] that the Er: YAG laser was significantly effective in reducing the loudness of snoring and the fatigue that occurs when the quality of sleep is not restful; the patients were all treated non-contact mode with irradiation of at least 7–8 pass using a PS04 handpiece and collimated beam, in LP mode and 10 Hz, 1.6 J/cm2.
The mechanism of action of the laser on snoring is not clear, but in vitro studies pointed out that the Er: YAG laser affects tissue through a photo-thermal effect, which causes acute shrinkage due to coagulation of the collagen fibres [33].
The same animal study [34] demonstrated histologically a noticeable contraction of the soft palate, showing an acute shrinkage of the mucosa, that occurred immediately after laser irradiation for 2 min with the Er:YAG laser (LightWalker AT; Fotona) with the specific snoring device handpiece (PS04) in a noncontact mode (1.15 W, 2 Hz, 1.5 J/cm2), in rats that were sacrificed after treatments.
The amount of energy absorbed during laser therapy is related to the specific wavelength and the biological tissue characteristics. Diode lasers have an affinity for haemoglobin and oxyhemoglobin, and their use on soft tissues is well documented; in contrast, Er:YAG lasers have an affinity for water and are significantly more superficial in penetration [35].
Further studies will demonstrate if diode lasers will have a role in reducing oedema in tissues by shrinkage of the mucosa and denaturation of collagen, as it was demonstrated for the Nd:YAG wavelength in a histological mouse study [36] (Q-switched of 5 ns, and long-pulsed mode of 0.3 ms), for the Er:YAG wavelength in a pilot study on the soft palate in snorers [24], and in another prospective pilot study performed on OSAS patients treated with a non-ablative Er:YAG laser (Variable Pulse Laser Mode at 500 mJ; 6 Hz; 3 W, Spot size 10.0 mm at 20.0 to 25.0 mm distance), which demonstrated via preoperative and postoperative cone-beam computed tomography (CBCT), a statistically significant increase of the mean total oropharyngeal airway volume (particularly at the most constricted point) [37].
It is known that snoring occurs when a partial obstruction of the upper airways occurs, and apneas occur when the obstruction is complete. The cause of obstruction is mostly through a narrowness of the upper airways that can be caused by, or associated with, the thickening and slackening of the oro-hypopharyngeal submucosal layers due to accumulation in the tissues of fat or oedema that may be caused by ageing, weight gain, and by chronic inflammation, for instance by LPR or smoking, although further studies are needed to better define these factors [9,38,39].
Indeed, in our work, overweight and obesity were present in more than half of cases, and LPR in a very large percentage of cases, which could suggest that, apart from fat in the submucosal layers, which is a well-known factor, LPR may also have a role in the multifactorial genesis of snoring or OSAS in this series, by slackening the mucosa and rendering tissues more edematous.
In this study, the patients that were overweight or obese seemed to be the ones that benefitted more from laser treatment compared to normoweight patients. In a minority of cases, the obstruction in snoring and apneas may be caused by a floppy epiglottis [13] that collapses during inspiration. Those cases where a floppy epiglottis was suspected were excluded (L > 3 at NOHL), but a DISE, if not refused by all patients who were searching for a non-invasive approach, would be the optimal selection. As the sites of obstruction can be multiple, it was also noticed that the results were better if the main site of obstruction was retropalatal, as the soft palate is easily reachable with the diode laser outpatient treatment. With this treatment, apart from the epiglottis, which is non-reachable as mentioned before, the base of the tongue may not always be optimally reachable with the laser, depending on patient’s anatomy and compliance to treatment.
The significant reduction in the VAS of snoring loudness and the better quality of rest may be explained by the reduction of soft palate thickness and by the major tightness of tissues that tend to collapse and vibrate less. In fact, the majority of patients reported a sensation of a wider throat after laser treatments. The significant decrease of dry mouth sensation in the morning may be due to the superior breathing that was reported by patients (probably due to an increase of retropalatal airway volume) and to the minor friction of tissues that underwent minor vibration during sleep compared to before treatments. The increase of dreaming reported by patients might be due to an increase in REM sleep, which is diminished or absent in apneas. The improvement in sexual activity that was reported by patients in our study has been documented previously in the published literature with Er: YAG outpatient laser treatment of snoring [23]. Choking, restless legs syndrome, and headache upon waking were significantly reduced or eliminated entirely. All of these symptoms are OSAS-related symptoms, and their disappearance may be explained by the significative lowering of AHI. The cessation of tonsillitis in the follow-up time after laser treatment may be explained by the well-known decontaminating effect of lasers, with a reduction or rupture of bacterial biofilm of the tonsillar crypts.
These above-mentioned subjective findings, along with the significant reduction of ESS scores, were further verified by an objective polysomnographic AHI decrease that was statistically significant. In our study, we noticed that patients with OSAS seemed to gain the most benefit from the treatment.
This treatment can be repeated if the effect diminishes. In this study, 37/45 subjects were followed up at 7–26 months with a median of 12 months. The results were sustainable at 12 months in 75.6% of cases and partially sustainable in 13.5% of cases. An accurate follow up is needed to prevent relapses and to treat patients if a relapse is noticed. A yearly treatment could be proposed to patients, but this needs further investigations and new long-term follow-up studies. Non-invasive outpatient diode laser treatments for snoring and OSAS will need further double-blind studies on large groups of patients with similar characteristics, such as groups with similar BMI and degree of severity of OSAS, but this requires a large number of patients.
5. Conclusions
Some weaknesses in our study, such as the limited number of patients and any kind of OSAS severity, should be taken into account, and they suggest further investigations to support our data. However, the evidence of our results allows concluding, for the first time in literature, that the treatment of snoring and sleep-related symptoms with an 808-nm Elexxion Claros® 50W Diode Laser by the specific SNORE 3 application could be effective in treating snoring and poor quality of sleep by widening the upper airways. This diode laser treatment might, therefore, represent a non-invasive alternative procedure for both simple snoring and OSAS patients that decline conventional therapies.
It is important to stress that our diode laser treatment had no major side-effects and the results were sustainable at 12 months.
Author Contributions
Conceptualization: I.F.S., A.A., and F.B.; data collection: I.F.S., F.B., and A.A.; investigation: I.F.S.; methodology: I.F.S.; software: F.B.; supervision: S.B. and A.A.; writing—original draft: I.F.S. and A.A.; writing—review and editing: M.F., A.Z., S.B., A.A., and R.A.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Regional Ethical Review Board of Department of Surgical Sciences and Integrated Diagnostics (University of Genoa), Unige-Disc-Prot./9619.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
Data available on request from the authors.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Ellenbogen, J.M. Cognitive benefits of sleep and their loss due to sleep deprivation. Neurology 2005, 64, E25–E27. [Google Scholar] [CrossRef] [PubMed]
- Valentino, D.J., III; O’Donnell, A.E. Reasons and referrals for sleep medicine consultation in obstructive sleep apnea. Chest 2006, 130, 264S. [Google Scholar] [CrossRef]
- Zamarrón, C.; Valdés Cuadrado, L.; Alvarez-Sala, R. Pathophysiologic mechanisms of cardiovascular disease in obstructive sleep apnea syndrome. Pulm. Med. 2013, 2013, 521087. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Ma, Y.; Ouyang, R.; Zeng, Z.; Zhan, Z.; Lu, H.; Cui, Y.; Dai, Z.; Luo, L.; He, C.; et al. The relationship between inflammation and neurocognitive dysfunction in obstructive sleep apnea syndrome. J. Neuroinflamm. 2020, 17, 229. [Google Scholar] [CrossRef]
- Gałecki, P.; Florkowski, A.; Zboralski, K.; Pietras, T.; Szemraj, J.; Talarowska, M. Psychiatric and psychological complications in obstructive sleep apnea syndrome. Pneumonol. Alergol. Pol. 2011, 79, 26–31. [Google Scholar]
- Mannarino, M.R.; di Filippo, F.; Pirro, M. Obstructive sleep apnea syndrome. Eur. J. Intern. Med. 2012, 23, 586–593. [Google Scholar] [CrossRef]
- Di Carlo, G.; Zara, F.; Rocchetti, M.; Venturini, A.; Ortiz-Ruiz, A.J.; Luzzi, V.; Cattaneo, P.M.; Polimeni, A.; Vozza, I. Prevalence of sleep-disordered breathing in children referring for first dental examination. A multicenter cross-sectional study using pediatric sleep questionnaire. Int. J. Environ. Res. Public Health 2020, 17, 8460. [Google Scholar]
- Li, Z.; Celestin, J.; Lockey, R.F. Pediatric Sleep Apnea Syndrome: An Update. J. Allergy Clin. Immunol. Pract. 2016, 4, 852–861. [Google Scholar] [CrossRef]
- Garvey, J.F.; Pengo, M.F.; Drakatos, P.; Kent, B.D. Epidemiological aspects of obstructive sleep apnea. J. Thorac. Dis. 2015, 7, 920–929. [Google Scholar]
- Senaratna, C.V.; Perret, J.L.; Lodge, C.J.; Lowe, A.J.; Campbell, B.E.; Matheson, M.C.; Hamilton, G.S.; Dharmage, S.C. Prevalence of obstructive sleep apnea in the general population: A systematic review. Sleep Med. Rev. 2017, 34, 70–81. [Google Scholar] [CrossRef]
- Stigter, H.; Lammers, G.J.; Rooyackers, J.M. Werkgroep Rijgeschiktheid. Sleep disorders and the ability to drive, revision of legislation. Ned. Tijdschr. Geneeskd. 2009, 153, A690. [Google Scholar]
- Stuck, B.A.; Dreher, A.; Heiser, C.; Herzog, M.; Kuhnel, T.; Maurer, J.T.; Pistner, H.; Sitter, H.; Steffen, A.; Verse, T. Diagnosis and treatment of snoring in adults-S2k Guideline of the German Society of Otorhinolaryngology, Head and Neck Surgery. Sleep Breath. 2015, 19, 135–148. [Google Scholar] [CrossRef]
- Sanches, I.; Martins, V.; dos Santos, J.M. Obstructive sleep apnea refractory to treatment due to floppy epiglottis. Arch Bronconeumol 2015, 51, 94. [Google Scholar] [CrossRef] [PubMed]
- Server, E.A.; Alkan, Z.; Yiğit, Ö.; Yasak, A.G. Long-term results of pillar implant procedure. Kulak Burun Bogaz Ihtis Derg 2016, 26, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Olszewska, E.; Panek, J.; O’Day, J.; Rogowski, M. Usefulness of snoreplasty in the treatment of simple snoring and mild obstructive sleep apnea/hypopnea syndrome—Preliminary report. Otolaryngol. Pol. 2014, 68, 184–188. [Google Scholar] [CrossRef]
- Chiesa Estomba, C.M.; Rivera Schmitz, T.; Ossa Echeverri, C.C.; Betances Reinoso, F.A.; Fariña Conde, J.; Alonso Parraga, D. The treatment of snoring by radiofrequency-assisted uvulopalatoplasty and results after one-session protocol: A prospective, longitudinal, non-randomized study. Eur. Arch Otorhinolaryngol. 2015, 272, 3059–3063. [Google Scholar] [CrossRef]
- Pazos, G.; Mair, E.A. Complications of radiofrequency ablation in the treatment of sleep-disordered breathing. Otolaryngol. Head Neck Surg. 2001, 125, 462–466. [Google Scholar] [CrossRef] [PubMed]
- Mezzofranco, L.; Luigi, A.; Gracco, T.; Milano, F.; Di-Bernardo, G.A.; Vezzali, L.; Giovannini, D. Personality traits and oral device: A new challenge to increase compliance with MAD therapy for OSAS and snoring. Sleep Sci. 2020, 13, 10–17. [Google Scholar]
- Li, S.; Wu, D.; Shi, H. Reoperation on patients with obstructive sleep apnea-hypopnea syndrome after failed uvulopalatopharyngoplasty. Eur. Arch Otorhinolaryngol. 2015, 272, 407–412. [Google Scholar] [CrossRef]
- Rombaux, P.; Hamoir, M.; Bertrand, B.; Aubert, G.; Liistro, G.; Rodenstein, D. Postoperative pain and side effects after uvulopalatopharyngoplasty, laser-assisted uvulopalatoplasty, and radiofrequency tissue volume reduction in primary snoring. Laryngoscope 2003, 113, 2169–2173. [Google Scholar] [CrossRef]
- Tang, J.A.; Salapatas, A.M.; Bonzelaar, L.B.; Friedman, M. Long-Term Incidence of Velopharyngeal Insufficiency and Other Sequelae following Uvulopalatopharyngoplasty. Otolaryngol. Head Neck Surg. 2017, 156, 606–610. [Google Scholar] [CrossRef] [PubMed]
- Nishigawa, K.; Hayama, R.; Matsuka, Y. Complications causing patients to discontinue using oral appliances for treatment of obstructive sleep apnea. J. Prosthodont Res. 2017, 61, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Fini Storchi, I.; Parker, S.; Bovis, F.; Benedicenti, S.; Amaroli, A. Outpatient erbium: YAG (2940 nm) laser treatment for snoring: A prospective study on 40 patients. Lasers Med. Sci. 2018, 33, 399–406. [Google Scholar] [CrossRef]
- Cetinkaya, E.A.; Turker, M.; Kiraz, K.; Gulkesen, H.K. Er: YAG laser treatment of simple snorers in an outpatient setting. ORL J. Otorhinolaryngol. Relat. Spec. 2016, 78, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Mure, C.; Blumen, M.; Alali, A.; Page, L.; Chabolle, F. Surgical ablation of lingual tonsils in the treatment of obstructive sleep apnea. Eur. Ann. Otorhinolaryngol. Head Neck Dis. 2019, 136, 19–23. [Google Scholar] [CrossRef]
- Pavelec, V.; Bohmanova, J. A comparison of postoperative recovery from laser-assisted uvulopalatoplasty using different laser systems. Otolaryngol. Head Neck Surg. 2008, 138, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Sher, A.E.; Thorpy, M.J.; Shprintzen, R.J.; Spielman, A.J.; Burack, B.; McGregor, P.A. Predictive value of Muller maneuver in selection of patients for uvulopalatopharyngoplasty. Laryngoscope 1985, 95, 1483–1487. [Google Scholar] [CrossRef]
- Vicini, C.; de Vito, A.; Benazzo, M.; Frassineti, S.; Campanini, A.; Frasconi, P.; Mira, E. The nose oropharynx hypopharynx and larynx (NOHL) classification: A new system of diagnostic standardized examination for OSAHS patients. Eur. Arch Otorhinolaryngol. 2012, 269, 1297–1300. [Google Scholar] [CrossRef]
- Belafsky, P.C.; Postma, G.N.; Koufman, J.A. The validity and reliability of the reflux finding score (RFS). Laryngoscope 2001, 111, 1313–1317. [Google Scholar] [CrossRef]
- Belafsky, P.C.; Postma, G.N.; Koufman, J.A. Validity and reliability of the reflux symptom index (RSI). J. Voice 2002, 16, 274–277. [Google Scholar] [CrossRef]
- Smith, S.S.; Oei, T.P.; Douglas, J.A.; Brown, I.; Jorgensen, G.; Andrews, J. Confirmatory factor analysis of the Epworth Sleepiness Scale (ESS) in patients with obstructive sleep apnoea. Sleep Med. 2008, 9, 739–744. [Google Scholar] [CrossRef] [PubMed]
- Friedman, M.; Hamilton, C.; Samuelson, C.G.; Lundgren, M.E.; Pott, T. Diagnostic value of the Friedman tongue position and Mallampati classification for obstructive sleep apnea: A meta-analysis. Otolaryngol. Head Neck Surg. 2013, 148, 540–547. [Google Scholar] [CrossRef] [PubMed]
- Dams, S.D.; de Liefde-van Beest, M.; Nuijs, A.M.; Oomens, C.W.; Baaijens, F.P. Pulsed heat shocks enhance procollagen type I and procollagen type III expression in human dermal fibroblasts. Skin Res. Technol. 2010, 16, 354–364. [Google Scholar] [CrossRef]
- Unver, T.; Aytugar, E.; Ozturan, O.; Kıran, T.; Ademci, E.; Usumez, A. Histological effects of Er:YAG laser irradiation with snoring handpiece in the rat soft palate. Photomed Laser Surg. 2016, 34, 321–325. [Google Scholar] [CrossRef]
- Parker, S.; Cronshaw, M.; Anagnostaki, E.; Mylona, V.; Lynch, E.; Grootveld, M. Current Concepts of Laser Oral Tissue interaction. Dent. J. 2020, 8, 61. [Google Scholar] [CrossRef]
- Liu, H.; Dang, Y.; Wang, Z.; Chai, X.; Ren, Q. Laser induced collagen remodeling: A comparative study in vivo on mouse model. Lasers Surg. Med. 2008, 40, 13–19. [Google Scholar] [CrossRef]
- Lee, Y.S.C.; Lee, C.Y.C. Evaluation of a non-ablative Er: YAG laser procedure to increase the oropharyngeal airway volume: A pilot study. Dent. Oral Craniofac. Res. 2015, 1, 56–59. [Google Scholar]
- Kim, K.S.; Kim, J.H.; Park, S.Y.; Won, H.R.; Lee, H.J.; Yang, H.S.; Kim, H.J. Smoking induces oropharyngeal narrowing and increases the severity of obstructive sleep apnea syndrome. J. Clin. Sleep Med. 2012, 8, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, M.M.; Dibbern, R.S.; Santos, V.J.; Passeri, L.A. Influence of obesity on the correlation between laryngopharyngeal reflux and obstructive sleep apnea. Braz. J. Otorhinolaryngol. 2014, 80, 5–10. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).