An optimized apparatus design for continuous ATPF ensures spatial separation of the feed phases as well as the processed phases. In addition, the geometry at the inlet of the apparatus must be optimized for a uniform inflow of the bottom phase. Crucial for a good separation process is a suitable and controllable gas input. This requires consideration of the phase mixing effect caused by the rising gas bubbles, as well as the effects of gas input and phase mixing on continuous ATPF.
4.1. Design of the Continuous ATPF Apparatus
When ATPF is operated in batch mode, a vertical flotation cell as known from conventional froth flotation is usually used. This allows integrating only one single gassing unit and disables a spatial separation of unloaded and loaded top phase in a continuous ATPF [
23]. To overcome these disadvantages, an alternative apparatus design is needed. For continuous ATPF, a horizontal flotation tank has the advantage of separating the feed and processed phases across the longitudinal section of the apparatus. In
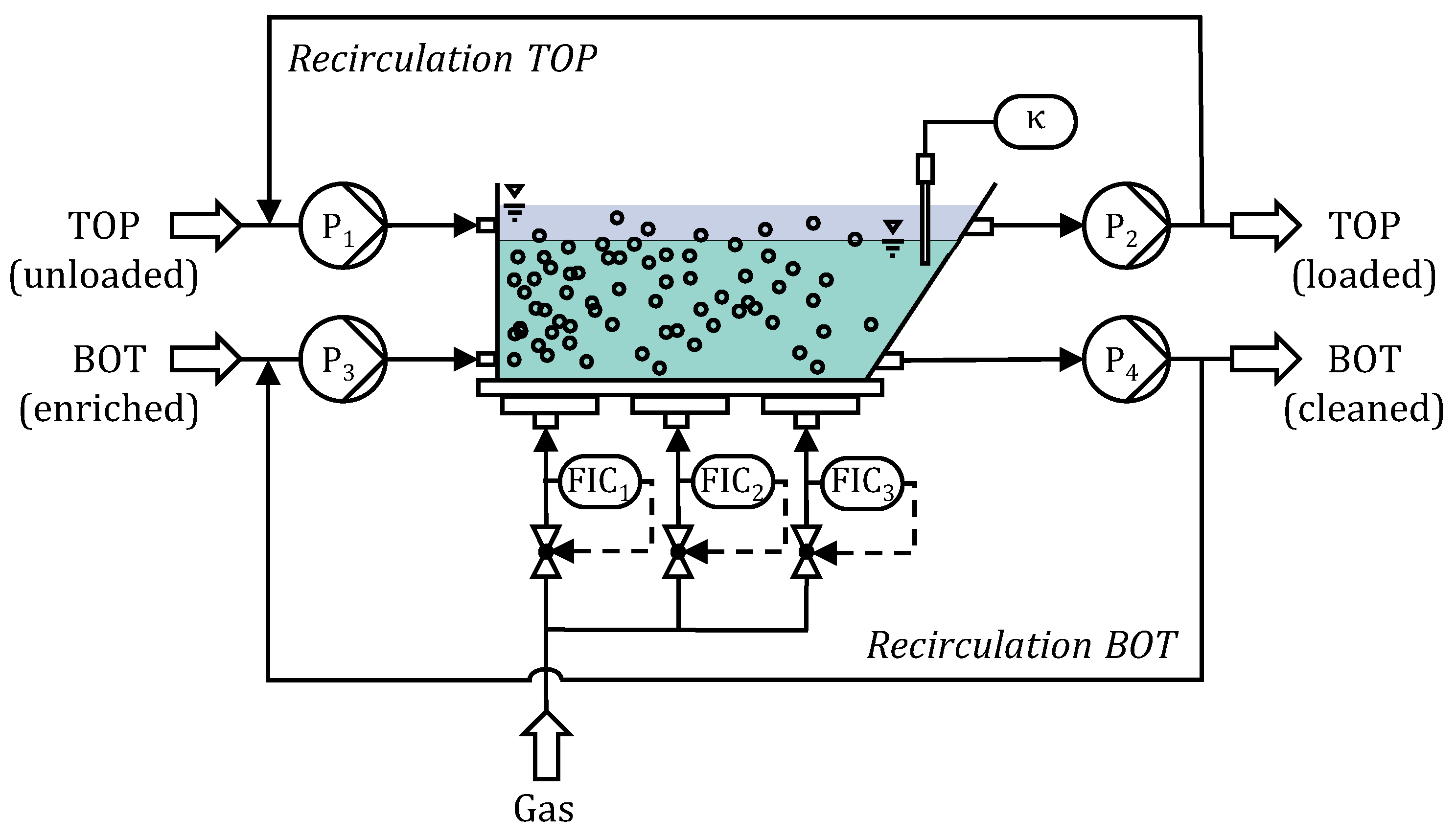
Figure 1, a 3D-printed prototype of the labor-sized horizontal flotation tank is shown and in
Figure 3, the apparatus is schematically illustrated for the case of co-current flow. The enzymes enriched bottom phase (BOT) is insert via the peristaltic pump P
3 at the bottom of the flotation tank on the left side. The unloaded top phase (TOP) entries the flotation tank right above, conveyed by pump P
1. Two outlets at the right side, connected with pump P
2 and P
4, allow exhausting the loaded top and cleaned bottom phase, respectively. While both phases move through the apparatus, gas bubbles are introduced via three identical gassing units. The gas flow rates of each unit can be adjusted separately by the mass flow controllers 1, 2 and 3. Additionally a conductivity measurement is implemented in the bottom phase near the outlet 2 cm below the aqueous phase boundary. For operating continuous ATPF efficiently, the handling of the throughput of bottom and top phase as well as the gas flows is essential. To keep a constant level of the two aqueous phases, the corresponding pumps for bottom phase (P
3 and P
4) can adjust the same flow rate at the inlet and outlet. The two pumps exchanging the top phase (P
1 and P
2) enable a constant volume of top phase in the same manner. By increasing the pumping rates, the phase exchange rate can be increased, if rapid mass transfer ensures high separation efficiency during continuous ATPF.
To achieve a high bubble surface area flux and hence a high flotation rate a high gas flow rate can be adjusted on the entry of the flotation tank. To avoid high penetration of the aqueous phase boundary between bottom and top phase, small bubbles and moderate gas flow rates are needed [
22]. Hence, lower flow rates near the outlet of the apparatus prevent phase mixing, which would lead to back-diffusion of the enzymes from the enriched top into the cleaned bottom phase as well as a drainage of mixed phases. By measuring the electrical conductivity (κ) the phase separation can be characterized and the phase mixing controlled by adjusting the gas input. Since ATPF is most efficient, when there is a high concentration gradient of the enzymes between bottom and top phase [
23], the co-current flow of the two aqueous phases provides a maximal mass transfer near the inlet of the horizontal flotation tank. Due to the spatial separation of the inlet and outlet, the gas input can be adjusted as needed for high amount of bubble introduction in the inlet as well as reduced phase mixing at the outlet. Consequently, the optimized design of the introduced apparatus assurances an efficient continuous ATPF process.
4.2. Geometry Optimization of the Inlet
To ensure a uniform flow through the flotation tank, there must be no stall in the inlet area. Otherwise, dead zones or backflow zones might occur. The Reynolds number at which stall occurs was determined by successively varying the volume flow rate or flow velocity at the inlet, starting at 1.6 mL/min up to 83 mL/min (16 mL/min corresponds to a phase exchange rate of 2/h). To ensure that the flow is stationary, the residence time of the flow domain was estimated.
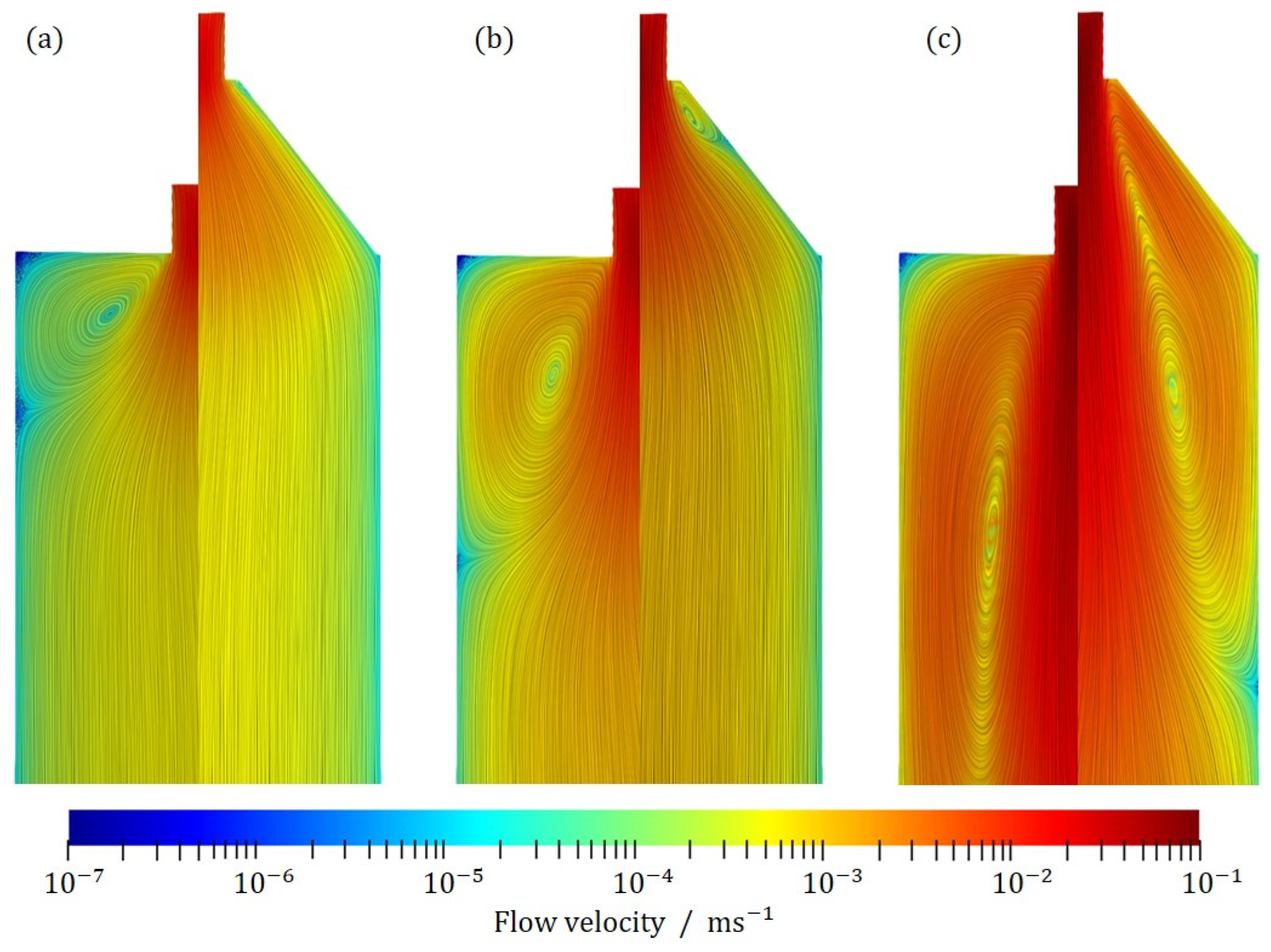
Figure 4 shows exemplary the inlet zone for three different inlet velocities,
,
and
. In each case, the abrupt transition between the inlet pipe and the flotation tank is shown on the left, while a continuous expansion of the cross-section with an angle of 45° is shown on the right. For better visibility, a logarithmic representation of the speed was chosen. In addition, the streamlines are shown.
As expected, an abrupt transition causes an immediate separation of the flow as it enters the flotation tank. A backflow occurs, which may be recognized by the vortex formed by the streamlines. The continuous expansion of the inlet pipe allows a uniform flow through the entire cross-section of the flotation tank. At a Reynolds number of , it may be seen that flow separation occurs immediately at the inlet, where a small step exists. Due to the length of the inlet area, however, the flow stabilizes, so that no backflow occurs in the flotation tank itself and the flow through the tank is uniform. At higher Reynolds numbers, the flow is completely detached and a reverse flow is formed, just as in the case of the abrupt transition. In summary, it may be said that up to a Reynolds number of 25, the flow through the cross-section flotation cell is uniform. At higher values, flow separation and the formation of vortices and dead zones may occur. Thus, a uniform inflow is guaranteed approximately up to Re = 25.
For verification, the final geometry was simulated and compared with experiments. For this purpose, the bottom phase was dyed with red food coloring.
Figure 5 shows the temporal progress of the red colored bottom phase in the simulation (top) and in the experiment (bottom) for the time points 6, 20 and 40 s as well as 80 s at a flow rate of 16 mL/min. Both the simulation and the experiment show the uniform flow of the bottom phase into the flotation tank. At the beginning, the flow pattern indicates the two inlets (6s). After some time, the two inlet flows merge into one uniform flow over the entire width of the flotation tank. In the experiment, the flow through the tank lasts 100 s, in the simulation 106 s. The simulations indicate a steeper parabolic flow profile, while the experiment shows an almost uniform flow front. The different flow profiles may possibly be explained by diffusion, which may have an influence at low flow velocities occurring close to the wall due to the no-slip boundary condition (0 m/s). Experimental data for the diffusion coefficient was not available. In the simulation, the diffusion coefficient was estimated with 10
−9 m
2/s. Apparently, the simulation reproduces the experiment well. For the ATPF process the uniform over the cross-section of the flotation cell guarantees, that the enriched bottom phase comes into contact with gassing area and hence that the enzyme molecules can attach to the gas bubbles.
4.3. Characterization of the Phase Mixing Effect by Measuring the Electrical Conductivity
To characterize the phase mixing effect induced by gas input by measuring the electrical conductivity in the bottom phase, calibration is needed.
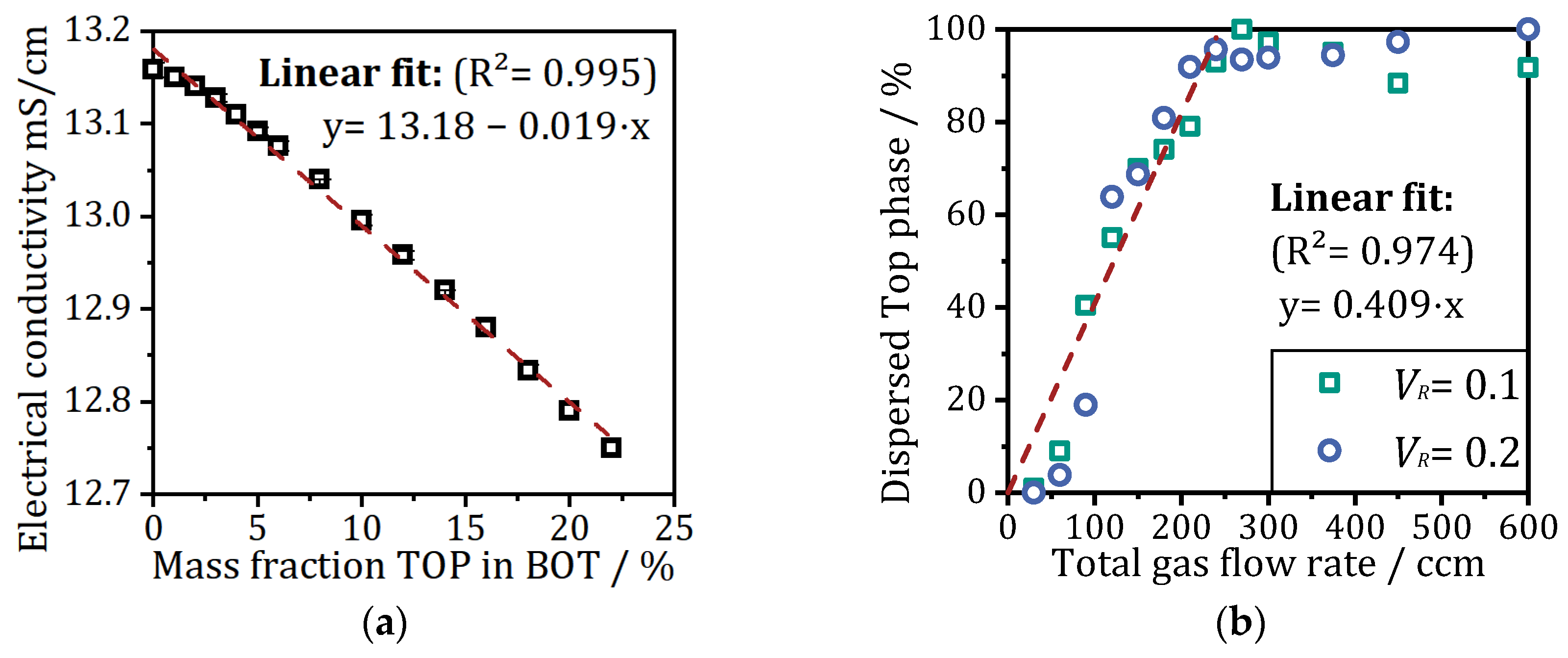
Figure 6a shows the electrical conductivity as a function of the mass fraction of the top phase dispersed in the bottom phase obtained from mixing both phases inside a laboratory beaker. In pure bottom phase a high conductivity of about 13.16 mS/cm is measured. With increasing amount of top phase dispersed, the conductivity decreases linearly down to 12.78 mS/cm at 20% (
w/
w). Since the phase-forming components (citrate and PEG1000) are distributed differently within the two-phase system, the electrical conductivity of the salt-rich bottom phase is very high due to the dissolved ions. The measured values can be classified with the comparison to sea water where the electrical conductivity ranges between 21–63 mS/cm and fresh water in the µS/cm range [
29]. When the polymer-rich top phase gets dispersed in the bottom phase, the polymer molecules inhibit the movement of ions and thus reduce the conductivity. Due to the density difference between the top and bottom phases, a mass fraction of about 11% corresponds to 100% dispersed top phase in ATPF when a volume ratio (
VR) of 0.1 is used and 22% when
VR is 0.2. The linear dependency can be used to calculate the amount of dispersed top phase caused by the rising bubbles during ATPF experiments with integrated conductivity measurements inside the flotation tank.
Figure 6b shows the correlation between the total gas flow rates (all three gassing units with uniform flow rates) and the percentage of dispersed top phase for two different volume ratios of 0.1 and 0.2. For total gas flow rates lower than 60 ccm, less than 10% of the top phase is dispersed. Doubling the flow rates to 120 ccm total gas flow rate increases the amount to more than 55%. At 240 ccm total gas flow rate, more than 90% of the top phase is dispersed. Up to this point, a linear trend of the gassing rate and phase mixing effect can be observed independent of the volume ratio used. Higher flow rates lead to unpredictable high phase mixing effects with about 90–100% dispersed top phase. As described in previous work [
22], rising bubbles can lead to phase mixing effects as they rise from the bottom to the top phase. Crucial to this effect is the bubble size, with larger bubbles causing more microswirls and thus leading to higher dispersion of the top phase. In the previously noted study, it was also observed that an increase in gas flow rate increased not only the number but also the size of the gas bubbles. Therefore, when setting a higher flow rate at all three gassing units, the stress on the aqueous phase boundary between the bottom and top phases increases.
For the ATPF process the phase mixing effect caused by to high gassing rates is crucial. On the one hand, the mixing of the unloaded top and enriched bottom phases near the inlet of the flotation tank increases the contact area and thus the diffusion of the enzyme from bottom into the top phase; on the other hand, near the outlet, it leads to back-diffusion from the loaded top phase into the cleaned bottom phase. In addition to avoiding the undesirable back-diffusion, strong phase mixing near the outlet must also be prevented, so that no mixed phases exit. If there is almost no phase separation, as observed at total flow rates higher than 240 ccm (see
Figure 6b), no enzyme-laden top phase can be collected. To ensure high separation and concentration of enzymes, minimal phase mixing should be ensured at the outlet.
The total gas flow rate is the sum of the individual flow rates of the three gassing units. Since the phase mixing effect is most important to consider close the outlet, lower flow rates should be set at gassing unit 3, and gassing units 2 and 1 can achieve equal or higher flow rates.
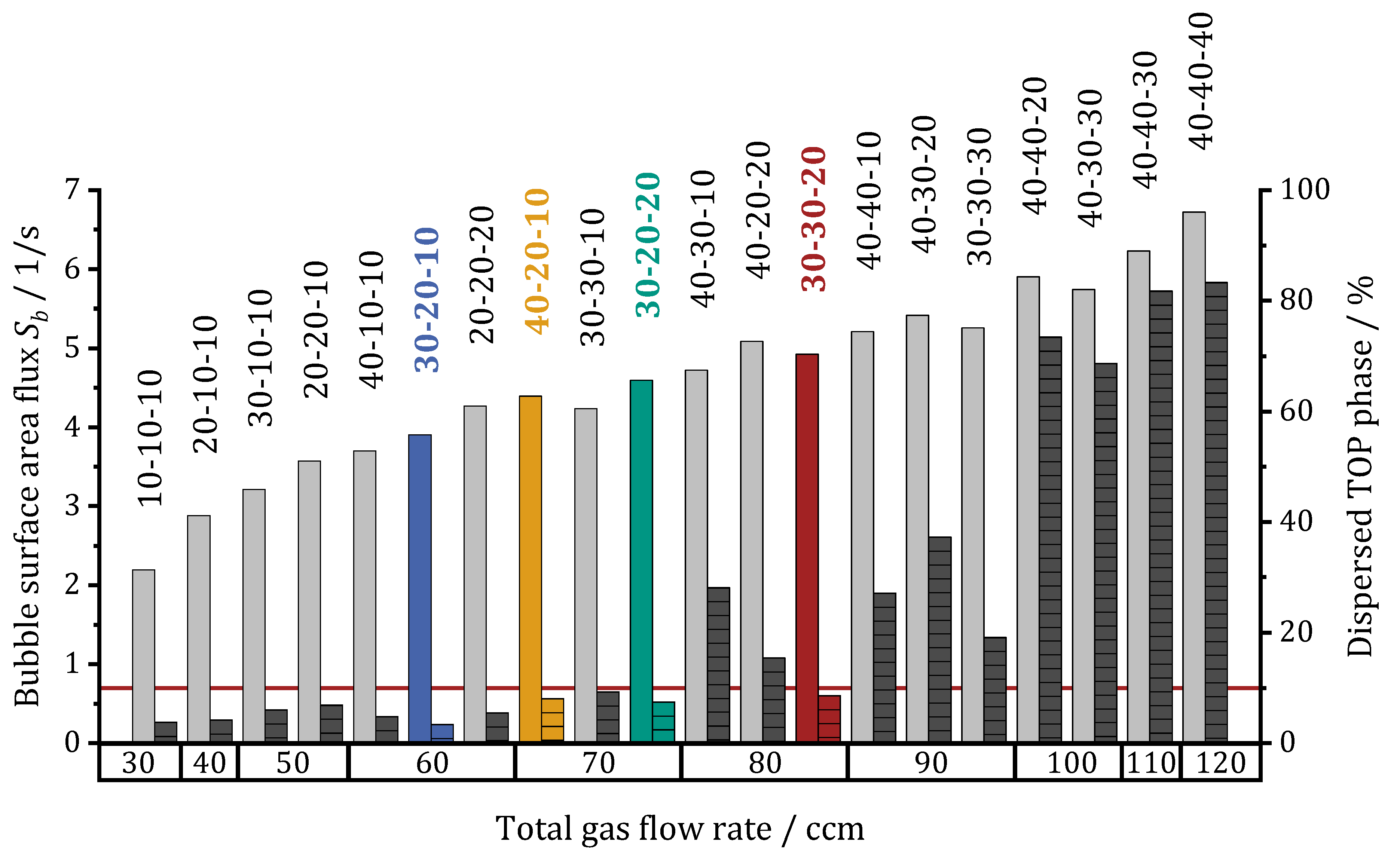
Figure 7 shows the effect of different gassing combinations on the percentage of dispersed top phase (shaded, dark bars) and the bubble surface area flux (left, gray bars). The latter was calculated according to [
22] considering a changing mean Sauter diameter for different gas flow rates. The individual flow rates are written above the bars (FIC
1–FIC
2–FIC
3) and sum up to the total gas flow rate (x-axis labels). The colored bars indicate the flow rate combinations chosen for flotation experiments described later, and the red horizontal line marks 10% dispersed top phase.
Overall, the bubble surface area flux increases at higher total gas flow rates. For one total gas flow rate, the
Sb value can vary for different combinations of the individual flow rates, as can be seen for the total gas flow rate of 70 ccm. Here, the highest
Sb value is about 4.6 (1/s) and results for the 30-20-20 ccm combination. At 30 ccm total gas flow rate, the
Sb value is about 2.2 (1/s), while tripling the total gas flow rate up to 90 ccm increases the
Sb value non-linearly up to a maximum of 5.4 (1/s). This is due to the larger bubble diameter caused by higher individual flow rates [
22]. Nevertheless, the individual flow rates dominate the increased bubble sizes when considering
Sb. Looking at the amount of dispersed top phase, there is no general trend up to a total gas flow rate of 70 ccm. Further increasing of the total gas flow rates leads to higher amounts of dispersed top phase. At 80 ccm only one combination (30-30-20) does not exceed 10% (red horizontal line). At 100 ccm total gas flow rate or more, most of the top phase is dispersed. With respect to the 10% as the maximum amount of accepted top phase dispersion, the various combinations at 70 and 80 ccm are of major interest, as here some gassing combinations result in higher and some in lower percentages. At 30-20-20 ccm, the amount of top phase dispersed is lowest, while
Sb is highest in this group. Increasing the gas flow rate in gassing unit 1 and decreasing it in unit 3 leads to similar values for the 40-20-10 ccm combination, while increasing it in unit 2 and decreasing it in unit 3 causes a higher dispersion amount and a lower
Sb value. The 30-30-20 ccm combination (80 ccm total gas flow rate) shows highest
Sb for all combination avoiding more than 10% dispersed top phase. High individual flow rates of 40 ccm lead to a strong phase mixing effect. When all three gassing units aerate at this setting, most of the top phase is dispersed, as
Figure 6b already suggested. Low individual flow rates of 10 ccm result in only small amounts of dispersed top phase, but also in a small
Sb value when set the same for all three gassing units.
An efficient ATPF process requires both, low phase mixing at the end of the flotation tank and a high Sb value for fast mass transfer of the enzymes into the top phase. Since many combinations of individual flow rates are possible, four combinations were selected for comparison in ATPF-experiments: The 30-30-20 ccm combination (red) shows the highest Sb value targeting the selected limit of 10% dispersed top phase. The second and third highest values are obtained from the combinations 30-20-20 ccm (green) and 30-20-10 (yellow). The 30-20-10 ccm combination (blue) leads to a minimal phase mixing and deviates only in the third or first gassing unit, compared to the two combinations chosen at 70 ccm total gas flow rate.
4.4. Effect of Gas Input and Phase Mixing Effect on Continuous ATPF
To characterize the effects of gas input and phase mixing on continuous ATPF, flotation experiments were performed using the four previously selected gassing combinations and the phospholipase model enzyme. The separation efficiency (E) describes the amount of enzyme molecules separated from the bottom phase and concentrated in the top phase.
Figure 8a shows the separation efficiency during flotation time for different gassing combinations. During the first 60 min, complete recirculation of the bottom and top phase is applied. After that, a continuous operation with complete discharge of the phases starts. At the beginning of the ATPF experiments, all curves of the separation efficiency show a steep increase, with the curve of the 30-20-10 ccm combination (blue circles) showing the flattest increase compared to the others. After 15 min of flotation, the slope of the separation efficiency curve slowly decreases and reaches a constant level after 30 min (
E ≈ 60%). The fastest mass transfer occurs during gassing with the 30-20-20 ccm combination, resulting in a steep increase up to 10 min flotation time, followed by a short period of converging to a separation efficiency of about 70%, which is reached after 15 min and remains constant during the next 45 min of ATPF with recirculation of the phases. The other two gassing combinations range between the two mentioned above. While they have similar slopes in the beginning, the curves flatten out differently. The curve of the 30-30-20 ccm combination (red triangles pointing up) flattens out just above 60%, while that of the 40-20-10 ccm combination (yellow triangles pointing down) levels off above 68%. The rapid separation of the enzymes during aerating with the 30-20-20 ccm combination is caused, on the one hand, by the high bubble surface are flux and, on the other hand, by favorable phase mixing conditions, which led to the highest constant level of the separation efficiency during the recirculation operation.
The fast mass transfer of this gassing combination is also reflected in form of a high flotation rate constant (
k), which is shown in
Figure 8b. As can be seen, higher bubble surface area fluxes lead to higher flotation rate constants expect for the 30-30-20 ccm combination, where
k is only the second highest despite the highest
Sb. One reason for this could be that the high aeration of 30 ccm at gassing unit 2 leads to phase mixing causing enhanced back-diffusion effects, which reduce both flotation rate and the maximum separation efficiency. The latter is the result of an equilibrium between the uptake of enzymes in the top phase by flotation and diffusion and the return by back-diffusion [
23]. After 60 min of flotation, continuous discharge of the phases begins when the equilibrium state is reached for each ATPF experiment shown. While a slight decrease in the separation efficiency curves can be seen for both the 30-30-20 ccm and 30-20-10 ccm combinations, the 40-20-10 ccm combination leads to a constant level and the 30-20-20 ccm combination shows a small increase in separation efficiency. A decrease during continuous operation occurs when not all enzyme molecules in the enriched bottom phase entering the flotation tank can be transferred to the top phase before passing the apparatus and being removed in the cleaned bottom phase. The small increase during gassing with 30-20-20 ccm supports the previously hypothesized favorable phase mixing conditions, which provide high diffusion into the top phase at the inlet, but reduced back-diffusion near the outlet.
Consequently, the ATPF experiments with different gassing combinations show the influence of the local aeration on overall performance, including a fast separation during recirculation to reach the equilibrium as quickly as possible. In case of the 30-20-20 ccm combination, continuous operation could have been started after 15 min, which is half the time of the 30-20-10 ccm combination. As can be seen, high bubble surface area flux is necessary for efficient operation of the ATPF, but the choice of optimal gassing must take into account the phase mixing that causes both the desired diffusion to the top phase at the inlet and the undesired back-diffusion to the bottom phase close the outlet.
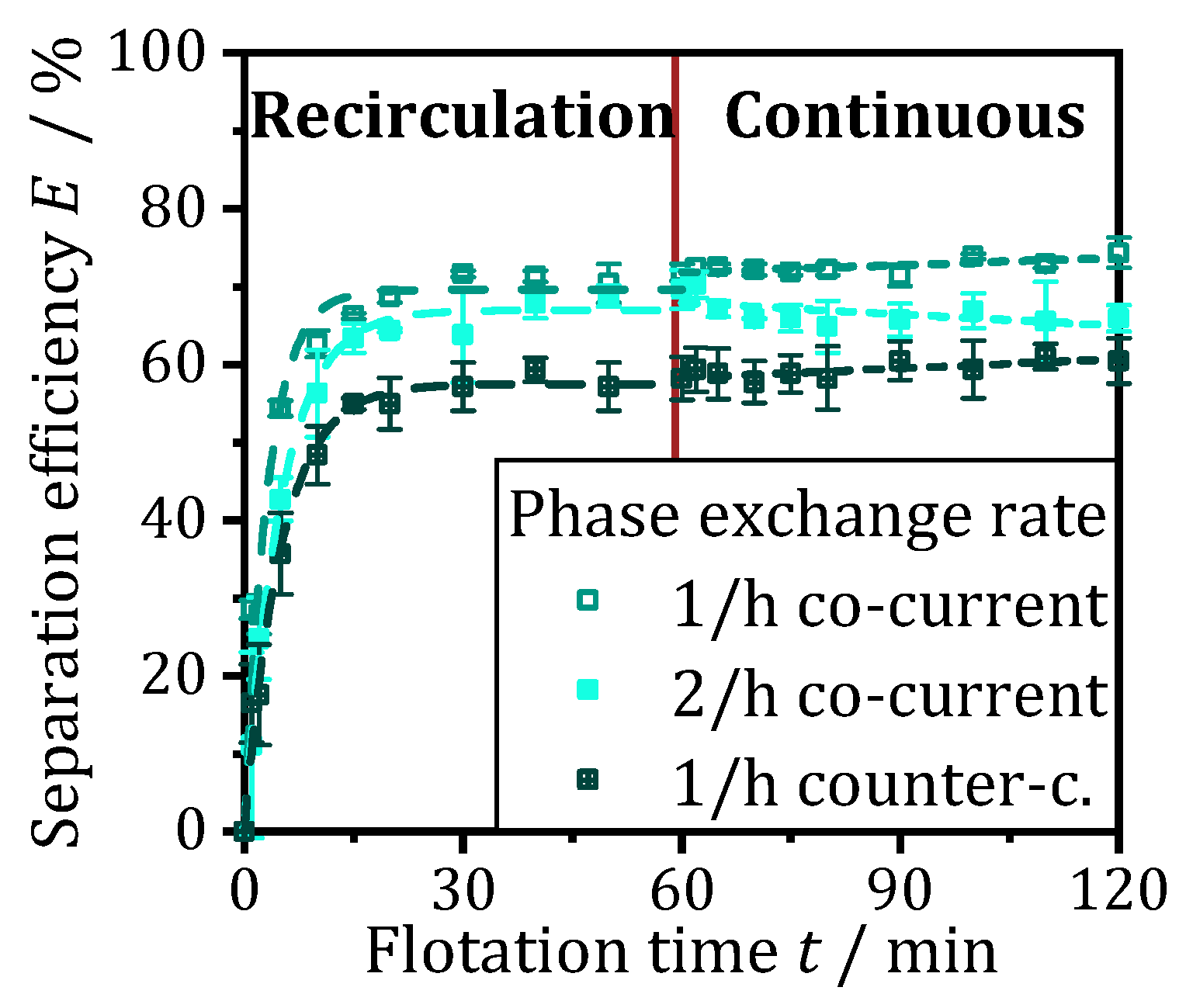
In addition to an ideal gas input, optimal process control is crucial for efficient continuous operation of the ATPF. During the floatation time, as much loaded bottom phase as possible is purified and all enzyme molecules that enter the ATPF apparatus are floated into the top phase. Whether a higher phase throughput is possible while maintaining the separation efficiency constant is shown by an experiment with a higher phase exchange rate with the previously determined preferred gassing combination (30-20-20 ccm).
Figure 9 shows the separation efficiency curve for the gassing combination 30-20-20 ccm and a phase exchange rate of 1/h (already shown in
Figure 8a), compared with the curve for an increased throughput with a phase exchange rate of 2/h (filled squares) with the same gassing. During recirculation in the first 60 min of flotation time, the separation efficiency at increased throughput shows a similar trend as at a phase exchange rate of 1/h, but the curve rises slightly less steeply and flattens out earlier at already approx. 68% instead of 70%. However, a greater difference becomes visible during continuous operation. Here, the separation efficiency decreases slightly and amounts to approx. 65% after a total of 120 min flotation time compared to 74% with slower exchange. The phase exchange rate, which is doubled from 1/h to 2/h, reduces the residence time and thus the possibility of an enzyme molecule being transported by a rising gas bubble into the top phase. This results in both a weaker rise of the separation efficiency and a reduction in the equilibrium between the transport into the top phase and back-diffusion. If the phases are no longer recirculated but exchanged continuously, not all the enzyme molecules introduced into the tank can be purified before they are discharged again, and the separation efficiency decreases over time.
The results suggest that an ideal phase exchange rate lies between 1/h and 2/h. However, further experiments to determine this in more detail do not appear useful, as the ideal setting would only be valid for a defined feed. However, in an industrial application, for example in the purification of a fermentation broth from a continuous fermentation, the enzyme concentration in the loaded bottom phase would be subject to fluctuations. These could be countered with online measurement of the enzyme concentration and a suitable control strategy for both, phase throughput and gassing rates. Therefore, it seems more purposeful to validate the hypothesis that phases fed in co-current lead to an increased diffusion into the top phase due to the higher concentration gradient (enriched bottom phase and unloaded top phase) at the inlet of the tank. Conversely, it can be hypothesized that if the bottom phase and top phase are pumped through the tank in a counter-current flow, the undesired back-diffusion at the outlet of the top phase will decrease due to a reduced concentration gradient when enriched bottom phase meets loaded top phase.
The crossed squares in
Figure 9 show the course of an ATPF with counter-current flowing phases at an exchange rate of 1/h and the gassing combination 30-20-20 ccm. After a steep increase until 15 min, the separation efficiency flattens out at already 58%. Although an increase in separation efficiency to 60% is observed during continuous operation, the maximum purification is about 14% lower than while the same test in co-current operation. The reduced separation efficiency in both recirculation and continuous discharge indicates reduced diffusion into the top phase, which seems to dominate a reduced back-difussion from the loaded top phase into the enriched bottom phase. The hypothesis to be tested, that co-current pumped phases lead to an increased transfer of the enzymes into the top phase, can thus be confirmed. Therefore, the best results were obtained with a gassing rate of 30-20-20 ccm and a phase exchange rate of 1/h in co-current flow.