Influence of Humidity on Performance of Single Chamber Air-Cathode Microbial Fuel Cells with Different Separators
Abstract
1. Introduction
2. Materials and Methods
2.1. Construction of Single Chamber Air-Cathode Microbial Fuel Cell
2.2. Biofilm Formation and Operation
2.3. Measurement and Analysis
3. Results and Discussion
3.1. MFC Power Generation with Different Separators and Relative Humidity Conditions
3.2. Polarization Performance of Air-Cathode MFCs
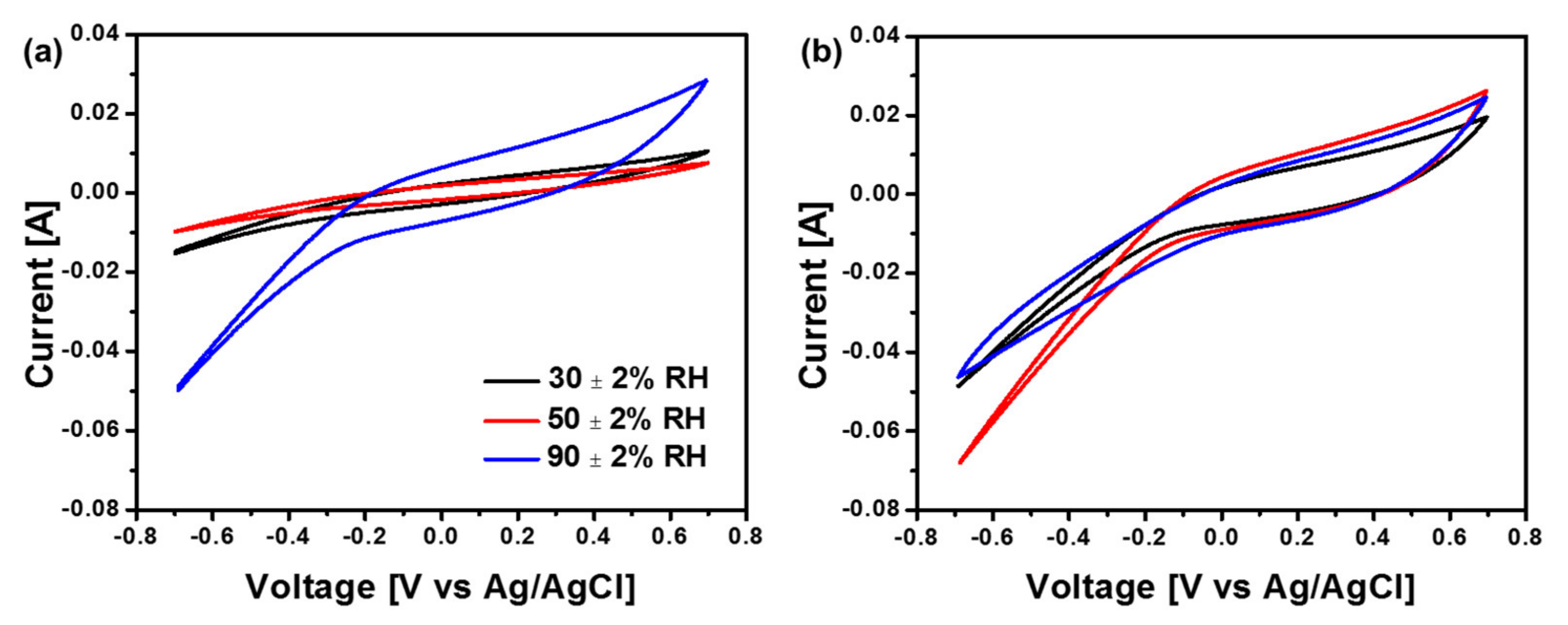
3.3. Cyclic Voltammogram Analysis of Cathode Electrode
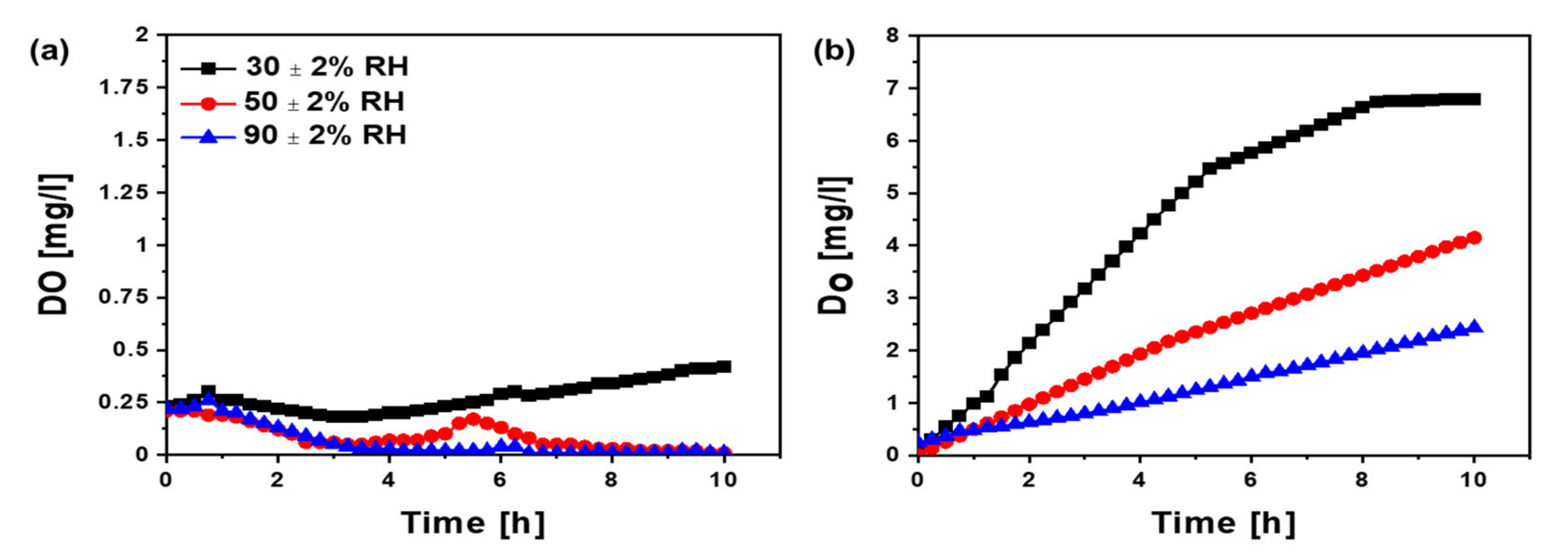
3.4. Oxygen Transport from Cathode to Anode at Varied Relative Humidity Conditions with Different Separators
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lee, M.; Reddy, C.N.; Min, B. In situ integration of microbial electrochemical systems into anaerobic digestion to improve methane fermentation at different substrate concentrations. Int. J. Hydrog. Energy 2019, 44, 2380–2389. [Google Scholar] [CrossRef]
- Vu, H.T.; Min, B. Integration of submersible microbial fuel cell in anaerobic digestion for enhanced production of methane and current at varying glucose levels. Int. J. Hydrog. Energy 2019, 44, 7574–7582. [Google Scholar] [CrossRef]
- Capodaglio, A.G.; Cecconet, D.; Molognoni, D. An integrated mathematical model of microbial fuel cell processes: bioelectrochemical and microbiologic aspects. Processes 2017, 5, 73. [Google Scholar] [CrossRef]
- Muddemann, T.; Haupt, D.; Jiang, B.; Sievers, M.; Kunz, U. Investigation and Improvement of Scalable Oxygen Reducing Cathodes for Microbial Fuel Cells by Spray Coating. Processes 2020, 8, 11. [Google Scholar] [CrossRef]
- Hong, S.W.; Chang, I.S.; Choi, Y.S.; Chung, T.H. Experimental evaluation of influential factors for electricity harvesting from sediment using microbial fuel cell. Bioresour.Technol. 2009, 100, 3029–3035. [Google Scholar] [CrossRef]
- Jayashree, C.; Sweta, S.; Arulazhagan, P.; Yeom, I.; Iqbal, M.; Banu, J.R. Electricity generation from retting wastewater consisting of recalcitrant compounds using continuous upflow microbial fuel cell. Biotechnol. Bioprocess Eng. 2015, 20, 753–759. [Google Scholar] [CrossRef]
- Kondaveeti, S.; Lee, J.; Kakarla, R.; Kim, H.S.; Min, B. Low-cost separators for enhanced power production and field application of microbial fuel cells (MFCs). Electrochim. Acta 2014, 132, 434–440. [Google Scholar] [CrossRef]
- Moon, J.M.; Kondaveeti, S.; Lee, T.H.; Song, Y.C.; Min, B. Minimum interspatial electrode spacing to optimize air-cathode microbial fuel cell operation with a membrane electrode assembly. Bioelectrochemistry 2015, 106, 263–267. [Google Scholar] [CrossRef]
- Lee, M.; Kakarla, R.; Min, B. Performance of an air-cathode microbial fuel cell under varied relative humidity conditions in the cathode chamber. Bioprocess Biosyst. Eng. 2019, 42, 1247–1254. [Google Scholar] [CrossRef]
- Ahn, Y.; Zhang, F.; Logan, B.E. Air humidity and water pressure effects on the performance of air-cathode microbial fuel cell cathodes. J. Power Sources 2014, 247, 655–659. [Google Scholar] [CrossRef]
- Jeon, D.H.; Kim, K.N.; Baek, S.M.; Nam, J.H. The effect of relative humidity of the cathode on the performance and the uniformity of PEM fuel cells. Int. J. Hydrog. Energy 2011, 36, 12499–12511. [Google Scholar] [CrossRef]
- Liu, H.; Cheng, S.; Logan, B.E. Production of electricity from acetate or butyrate using a single-chamber microbial fuel cell. Environ. Sci. Technol. 2005, 39, 658–662. [Google Scholar] [CrossRef] [PubMed]
- Kakarla, R.; Kim, J.R.; Jeon, B.-H.; Min, B. Enhanced performance of an air–cathode microbial fuel cell with oxygen supply from an externally connected algal bioreactor. Bioresour. Technol 2015, 195, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Kakarla, R.; Min, B. Photoautotrophic microalgae Scenedesmus obliquus attached on a cathode as oxygen producers for microbial fuel cell (MFC) operation. Int. J. Hydrog. Energy 2014, 39, 10275–10283. [Google Scholar] [CrossRef]
- Liu, T.; Yu, Y.-y.; Li, D.; Song, H.; Yan, X.; Chen, W.N. The effect of external resistance on biofilm formation and internal resistance in Shewanella inoculated microbial fuel cells. RSC Advances 2016, 6, 20317–20323. [Google Scholar] [CrossRef]
- Kakarla, R.; Min, B. Evaluation of microbial fuel cell operation using algae as an oxygen supplier: carbon paper cathode vs. carbon brush cathode. Bioprocess Biosyst. Eng. 2014, 37, 2453–2461. [Google Scholar] [CrossRef]
- Bi, W.; Sun, Q.; Deng, Y.; Fuller, T.F. The effect of humidity and oxygen partial pressure on degradation of Pt/C catalyst in PEM fuel cell. Electrochim. Acta 2009, 54, 1826–1833. [Google Scholar] [CrossRef]
- Natarajan, D.; Van Nguyen, T. Three-dimensional effects of liquid water flooding in the cathode of a PEM fuel cell. J. Power Sources 2003, 115, 66–80. [Google Scholar] [CrossRef]
- Xu, H.; Kunz, H.R.; Fenton, J.M. Analysis of proton exchange membrane fuel cell polarization losses at elevated temperature 120 C and reduced relative humidity. Electrochim. Acta 2007, 52, 3525–3533. [Google Scholar] [CrossRef]
- Sevda, S.; Dominguez-Benetton, X.; Vanbroekhoven, K.; Sreekrishnan, T.; Pant, D. Characterization and comparison of the performance of two different separator types in air–cathode microbial fuel cell treating synthetic wastewater. Chem. Eng. J. 2013, 228, 1–11. [Google Scholar] [CrossRef]
- Xu, L.; Zhang, G.; Chen, J.; Yuan, G.E.; Fu, L.; Yang, F. Prussian blue/graphene-modified electrode used as a novel oxygen reduction cathode in microbial fuel cell. J. Taiwan Inst. Chem. Eng. 2016, 58, 374–380. [Google Scholar] [CrossRef]
- Zhang, X.; He, W.; Ren, L.; Stager, J.; Evans, P.J.; Logan, B.E. COD removal characteristics in air-cathode microbial fuel cells. Bioresour. Bechnol. 2015, 176, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Mateo, S.; Rodrigo, M.; Fonseca, L.P.; Cañizares, P.; Fernandez-Morales, F.J. Oxygen availability effect on the performance of air-breathing cathode microbial fuel cell. Biotechnol. Prog. 2015, 31, 900–907. [Google Scholar] [CrossRef] [PubMed]
- Khalili, H.-B.; Mohebbi-Kalhori, D.; Afarani, M.S. Microbial fuel cell (MFC) using commercially available unglazed ceramic wares: low-cost ceramic separators suitable for scale-up. Int. J. Hydrog. Energy 2017, 42, 8233–8241. [Google Scholar] [CrossRef]
- Oliot, M.; Etcheverry, L.; Mosdale, A.; Basséguy, R.; Delia, M.-L.; Bergel, A. Separator electrode assembly (SEA) with 3-dimensional bioanode and removable air-cathode boosts microbial fuel cell performance. J. Power Sources 2017, 356, 389–399. [Google Scholar] [CrossRef]




| Relative Humidity (%) | Nafion | PP80 | ||
|---|---|---|---|---|
| Tafel Slope (mV/decade) | Exchange Current Density (mA/cm2) | Tafel Slope (mV/decade) | Exchange Current Density (mA/cm2) | |
| 30 ± 2 | 0.455 | 0.006 | 0.516 | 0.156 |
| 50 ± 2 | 0.448 | 0.002 | 0.501 | 0.323 |
| 90 ± 2 | 0.424 | 0.077 | 0.704 | 0.290 |
| Relative Humidity (%) | Voltage (V) | Power Density (mW/m2) | a Do (cm2/s) | b CE (%) | ||||
|---|---|---|---|---|---|---|---|---|
| Nafion | PP80 | Nafion | PP80 | Nafion | PP80 | Nafion | PP80 | |
| 30 ± 2 | 0.28 | 0.53 | 105 | 381 | 0.70 × 10−6 | 5.70 × 10−5 | 3.2 | 5.09 |
| 50 ± 2 | 0.26 | 0.50 | 76 | 352 | - | 3.30 × 10−5 | 2.6 | 2.87 |
| 90 ± 2 | 0.35 | 0.48 | 116 | 284 | - | 1.70 × 10−5 | 3.2 | 2.72 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, M.; Kondaveeti, S.; Jeon, T.; Kim, I.; Min, B. Influence of Humidity on Performance of Single Chamber Air-Cathode Microbial Fuel Cells with Different Separators. Processes 2020, 8, 861. https://doi.org/10.3390/pr8070861
Lee M, Kondaveeti S, Jeon T, Kim I, Min B. Influence of Humidity on Performance of Single Chamber Air-Cathode Microbial Fuel Cells with Different Separators. Processes. 2020; 8(7):861. https://doi.org/10.3390/pr8070861
Chicago/Turabian StyleLee, Mungyu, Sanath Kondaveeti, Taeyeon Jeon, Inhae Kim, and Booki Min. 2020. "Influence of Humidity on Performance of Single Chamber Air-Cathode Microbial Fuel Cells with Different Separators" Processes 8, no. 7: 861. https://doi.org/10.3390/pr8070861
APA StyleLee, M., Kondaveeti, S., Jeon, T., Kim, I., & Min, B. (2020). Influence of Humidity on Performance of Single Chamber Air-Cathode Microbial Fuel Cells with Different Separators. Processes, 8(7), 861. https://doi.org/10.3390/pr8070861





