Solar Advanced Oxidation Processes Using Parabolic Trough Concentrators: A Mini-Review
Abstract
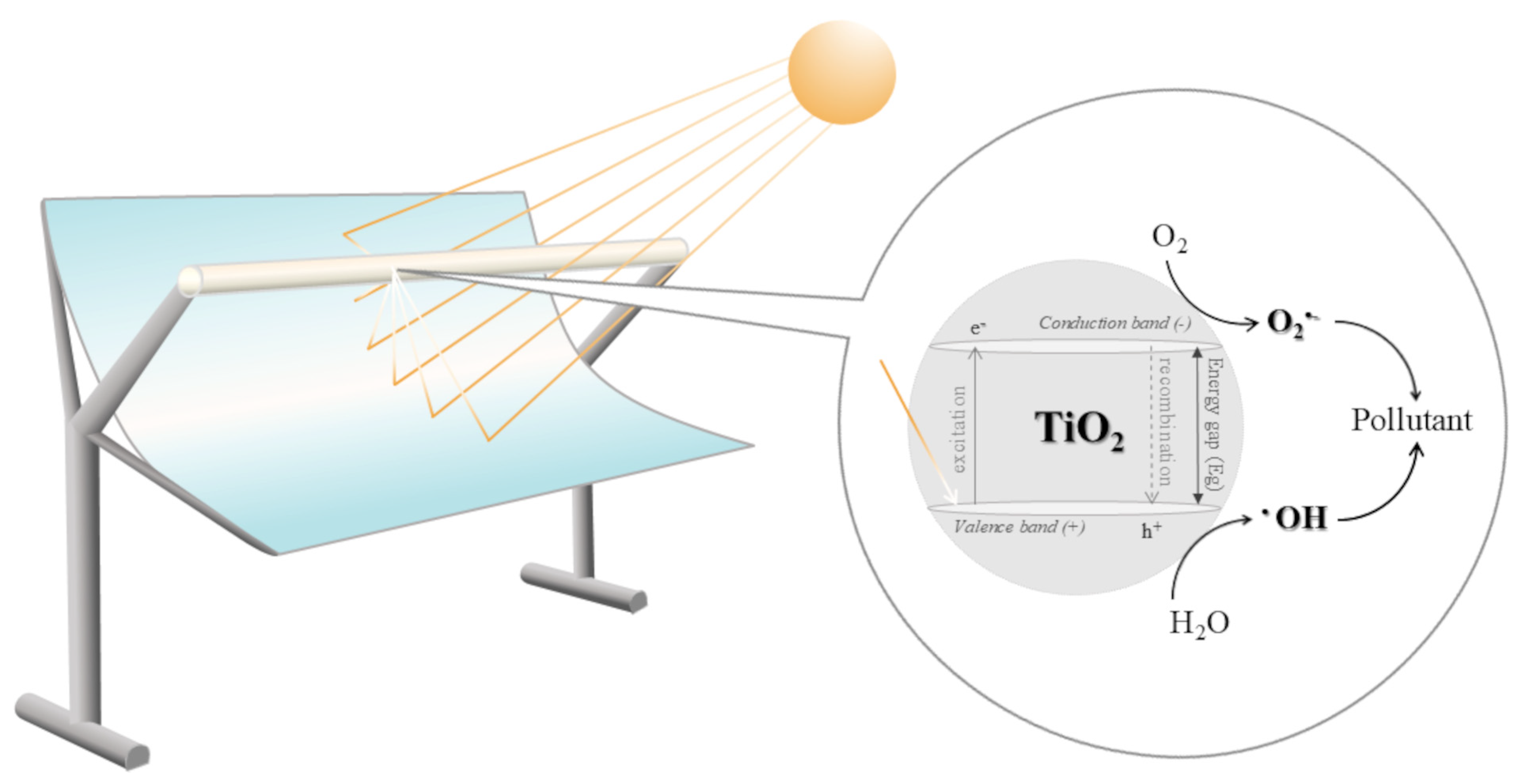
1. Introduction
- Turbidity tolerance—CPCs are often more robust when treating turbid wastewaters or operating on cloudy days, since they capture both direct and diffuse irradiation. In contrast, PTC efficiency depends on direct irradiation, which makes them sensitive to cloud cover and water turbidity, which scatters photons.
- Mass transfer and fluid dynamics—While both systems aim for high optical efficiency, the intense energy concentration in PTC allows for a more compact reactor footprint. Furthermore, PTCs typically maintain turbulent flow within the receiver tube, ensuring mass transfer and efficient mixing of the WW, catalyst and oxidant.
- Operational complexity and cost—PTC requires precise (automated) solar tracking systems to keep the aperture perpendicular to solar rays, which increases the initial capital investment and maintenance requirements. In contrast, the stationary design of the CPC reduces mechanical complexity and costs, though it requires a larger land area to achieve comparable treatment volumes.
2. Methodology
- “wastewater treatment” AND “advanced oxidation process”—this search resulted in highest number of publications, with 8306 in Scopus and 7981 in WoS;
- “wastewater treatment” AND “advanced oxidation process” AND “parabolic trough collector/concentrator”—resulted in 0 publications in Scopus and 84 in WoS;
- “wastewater treatment” AND “parabolic trough collector/concentrator”—resulted in 27 publications in Scopus and 24 in WoS.
Calculations for Performance Review
- PTC receiver volume (or irradiated volume; Virr (L)), assuming cylindrical geometry, where dr is receiver diameter (inner or outer, depending on availability; cm) and lr is receiver length (cm):
- Recirculation number (RN), defined as the ratio between the total treated WW volume (Vtotal; L) and irradiated volume (Virr; L):
- Residence time (tres) represents the time that WW remains under irradiation in the PTC-AOP system, where Q represents flow rate (L/min):
- PTC concentration ratio (CR) was approximated using the PTC parabola width (wp; cm) and receiver’s outer diameter (dr; cm):
- PTC aperture area (A; m2), where lp is parabola length and wp is parabola width:
- Photon utilisation ratio (PU; m2/L) indicates the extent of concentrated solar irradiation (A; m2) applied to the WW in the PTC receiver (Virr; L):
- The pseudo-first-order reaction rate constant (kFO) was calculated, where process efficiency (eff) or mineralisation (min) and reaction time (t; min) were reported:
- Finally, the time required to reduce the pollutant concentration by one order of magnitude (90%; τ90) was determined based on the calculated kFO for process efficiency or mineralisation:
3. Overview of PTC-Driven AOPs for WW Treatment
3.1. Wastewater Treatment in PTC-AOP System
| Pollutant | Concentration | Medium 1 | Process 2 | Ref. | Pollutant | Concentration | Medium 1 | Process 2 | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| Dye | Tetracycline | 10 mg/L | S | PC | [30] | ||||
| Dyes | - | RE | P | [35] | Amoxicillin | 100 mg/L | S | PC | [39] |
| Remazol Brilliant Blue | 60 mg/L | S | P | [17] | Industrial effluents | ||||
| Reactive Red 120 | 100 mg/L | S | P | [31] | Simulated WW | 7500 mg COD/L | S | PC | [40] |
| PF | Olive oil WW | - | S | PF | [41] | ||||
| Methylene Blue | 10 mg/L | S | PC | [33] | Municipal WW | 440 mg COD/L | S | PC | [42] |
| Rhodamine B | 15 mg/L | S | PC | Paper mill WW | 490 mg COD/L | RE | PC | [43] | |
| 2 mg/L | S | PC | [30] | Dairy processing WW | 569 mg COD/L | RE | PC | ||
| Methylene Blue | 5 mg/L | S | PC | Oil-field WW | 80 mg DOC/L | RE | PF | [25] | |
| Methyl Orange | 5 mg/L | S | PC | Tannery WW | 1428 mg COD/L | RE | PC | [44] | |
| Magenta dye | 100 mg/L | RE | PC | [45] | Gold mine WW | 2948 mg CN−/L | RE | PC | [46] |
| Aminosilicone | 50 mg CN−/L | S | PC | ||||||
| Silicone emulsion | 653 mg COD/L | S | PF | [25] | Metal | ||||
| Phenol | Cr | 6.88 mg Cr/L | RE | PCR | [44] | ||||
| Phenol | 100 mg DOC/L | S | PF | Cr(VI) | 20 mg/L | S | PCR | [47] | |
| 550 mg DOC/L | S | PF | Pb | 20 mg/L | S | PCR | [48] | ||
| 100 mg TOC/L | S | PF | [49] | Cu | 20 mg/L | S | PCR | ||
| Natural organic matter | Ni | 20 mg/L | S | PCR | |||||
| Humic acid | 50 mg/L | S | PF | [50] | Zn | 20 mg/L | S | PCR | |
| Organic acid | Microbial | ||||||||
| Oxalic acid | 900 mg/L | S | PC | [51] | E. coli log | 7.55 × 102 MPN/100 mL | RSW | D | [29] |
| Organic alcohol | E. coli | 1 × 106 CFU/mL | S | PC | [52] | ||||
| Methanol | 3204 mg/L | S | PC | [20] | E. coli | 1.2 × 105 CFU/mL | S | PC | [30] |
| Pesticide | Total coliforms log | 2.94 × 103 MPN/100 mL | RSW | D | [29] | ||||
| Clomazone | 97 mg DOC/L | S | PF | [25] | Total coliform | 70,000 MPN/100 mL | DRE | D | [37] |
| Thiophanate-methyl | 1369 mg/L | S | PF | [32] | Total heterotrophic | 1.034 × 104 CFU/mL | RE | D | [29] |
| Isoproturon | 25 mg/L | S | PC | [53] | bacterial count log | ||||
| Pharmaceutical | Total bacterial counts | 300,000 MPN/100 mL | DRE | D | [37] | ||||
| Ciprofloxacin | 10 mg/L | S | P | [17] | Sporeformer | 100,000 MPN/100 mL | DRE | D | |
3.2. PTC Design and Optical Performance
3.2.1. PTC Receiver Design
3.2.2. PTC Parabola Design
3.3. PTC-AOP System Performance
4. Future Perspectives
- Sludge management (homogeneous Fenton process) and catalyst recovery (heterogeneous Fenton process and photocatalysis)—The generation of Fe-rich sludge after Fenton reactions remains a significant operational challenge. Future studies should incorporate sludge management strategies, such as the development of heterogeneous catalysts to minimise secondary-waste generation. Also, greater attention should be given to catalyst recovery, reusability and regeneration.
- Life Cycle Assessment (LCA) and Techno-Economic Analysis (TEA)—Currently, both LCA and TEA are underrepresented in the PTC-AOP literature. The feasibility of PTC systems depends on balancing the high initial capital cost (precision sun-tracking mechanisms and reflective parabola materials) and land footprint requirements with long-term reductions in operational cost through renewable solar energy incorporation. These assessments should quantify the environmental trade-offs between chemical savings and the energy-intensive manufacturing of concentrator components.
- Scaling up of PTC-AOP technology—Most of the existing literature relies on laboratory or pilot conditions; thus, future work should address industrial-scale implementation. Hybrid configurations, where PTC can excel in polishing of recalcitrant pollutants and serve as a tertiary/quaternary stage, could be integrated into existing infrastructure (municipal WW treatment plants) or developed as standalone systems with automated control systems (adjustment of incident angle, flow rate, catalyst/oxidant dosage) to ensure consistent effluent treatment in real time.
5. Conclusions
- Superior optical performance—PTC systems demonstrate a clear advantage over stationary and non-concentrating reactors for the treatment of toxic and persistent pollutants and concentrated effluents. The high concentration ratio can facilitate improved and accelerated reaction kinetics with rapid •OH radical production, leading to a shorter treatment time.
- Necessity of standardised metrics—The systematic analysis revealed that the use of normalised performance indicators, like the time required for a one-order-of-magnitude reduction (τ90; min) and photon utilisation (PU; m2/L), is necessary for assessing treatment efficiency across diverse geographical locations and reactor designs.
- Climate and water resilience—Analytical mapping confirms that PTC-driven technologies are most viable in DNI hotspots. As climate change expands areas affected by severe water scarcity and elevated solar potential, like the Mediterranean and Balkan regions, PTC-AOPs may present a solution for safe WW treatment and reuse, contributing to the circularity and SDG6 targets.
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Kitzmann, N.H.; Caesar, L.; Sakschewski, B.; Rockström, J. Planetary Health Check 2025; Potsdam Institute for Climate Impact Research (PIK): Potsdam, Germany, 2025. [Google Scholar]
- IPCC. Climate Change 2023: Synthesis Report. Contribution of Working Groups I, II and III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change; Arias, P., Bustamante, M., Elgizouli, I., Flato, G., Howden, M., Méndez-Vallejo, C., Pereira, J.J., Pichs-Madruga, R., Rose, S.K., Saheb, Y., et al., Eds.; Intergovernmental Panel on Climate Change (IPCC): Geneva, Switzerland, 2023. [Google Scholar]
- United Nations. The United Nations World Water Development Report 2025—Mountains and Glaciers: Water Towers; UNESCO: Paris, France, 2025; p. 3. [Google Scholar]
- UN-Habitat; WHO. Progress on the Proportion of Domestic and Industrial Wastewater Flows Safely Treated—Mid-Term Status of SDG Indicator 6.3.1 and Acceleration Needs, with a Special Focus on Climate Change, Wastewater Reuse and Health; World Health Organization (WHO): Geneva, Switzerland, 2024.
- Arzate, S.; Pfister, S.; Oberschelp, C.; Sánchez-Pérez, J.A. Environmental Impacts of an Advanced Oxidation Process as Tertiary Treatment in a Wastewater Treatment Plant. Sci. Total Environ. 2019, 694, 133572. [Google Scholar] [CrossRef]
- Garrido-Cardenas, J.A.; Esteban-García, B.; Agüera, A.; Sánchez-Pérez, J.A.; Manzano-Agugliaro, F. Wastewater Treatment by Advanced Oxidation Process and Their Worldwide Research Trends. Int. J. Environ. Res. Public Health 2020, 17, 170. [Google Scholar] [CrossRef]
- Worku, A.K.; Ayele, D.W.; Teshager, M.A.; Omar, M.; Yerkrang, P.P.; Elgaddafi, R.; Alemu, M.A. Recent Advances in Wastewater Treatment Technologies: Innovations and New Insights. Energy Rev. 2025, 4, 100164. [Google Scholar] [CrossRef]
- Machado, F.; Teixeira, A.C.S.C.; Ruotolo, L.A.M. Critical Review of Fenton and Photo-Fenton Wastewater Treatment Processes over the Last Two Decades. Int. J. Environ. Sci. Technol. 2023, 20, 13995–14032. [Google Scholar] [CrossRef]
- Satyam, S.; Patra, S. The Evolving Landscape of Advanced Oxidation Processes in Wastewater Treatment: Challenges and Recent Innovations. Processes 2025, 13, 987. [Google Scholar] [CrossRef]
- Iervolino, G.; Zammit, I.; Vaiano, V.; Rizzo, L. Limitations and Prospects for Wastewater Treatment by UV and Visible-Light-Active Heterogeneous Photocatalysis: A Critical Review. Top. Curr. Chem. 2020, 378, 7. [Google Scholar] [CrossRef]
- Gutierrez-Mata, A.G.; Velazquez-Martínez, S.; Álvarez-Gallegos, A.; Ahmadi, M.; Hernández-Pérez, J.A.; Ghanbari, F.; Silva-Martínez, S. Recent Overview of Solar Photocatalysis and Solar Photo-Fenton Processes for Wastewater Treatment. Int. J. Photoenergy 2017, 2017, 8528063. [Google Scholar] [CrossRef]
- Al-Nuaim, M.A.; Alwasiti, A.A.; Shnain, Z.Y. The Photocatalytic Process in the Treatment of Polluted Water. Chem. Pap. 2023, 77, 677–701. [Google Scholar] [CrossRef]
- Ahtasham Iqbal, M.; Akram, S.; Khalid, S.; Lal, B.; Hassan, S.U.; Ashraf, R.; Kezembayeva, G.; Mushtaq, M.; Chinibayeva, N.; Hosseini-Bandegharaei, A. Advanced Photocatalysis as a Viable and Sustainable Wastewater Treatment Process: A Comprehensive Review. Environ. Res. 2024, 253, 118947. [Google Scholar] [CrossRef]
- Clemente, E.; Domingues, E.; Quinta-Ferreira, R.M.; Leitão, A.; Martins, R.C. Solar Photo-Fenton and Persulphate-Based Processes for Landfill Leachate Treatment: A Critical Review. Sci. Total Environ. 2024, 912, 169471. [Google Scholar] [CrossRef]
- Zhang, C.; Li, N.; An, G. Review of Concentrated Solar Power Technology Applications in Photocatalytic Water Purification and Energy Conversion: Overview, Challenges and Future Directions. Energies 2024, 17, 463. [Google Scholar] [CrossRef]
- Zhang, S.; Xu, L.; Xu, J.; Shen, B. A Mini-Review of Recent Progress in Zeolite-Based Catalysts for Photocatalytic or Photothermal Environmental Pollutant Treatment. Catalysts 2025, 15, 158. [Google Scholar] [CrossRef]
- El Golli, A.; Fendrich, M.; Bajpai, O.P.; Bettonte, M.; Edebali, S.; Orlandi, M.; Miotello, A. Parabolic Trough Concentrator Design, Characterization, and Application: Solar Wastewater Purification Targeting Textile Industry Dyes and Pharmaceuticals—Techno-Economic Study. EuroMediterr. J. Environ. Integr. 2024, 9, 1907–1919. [Google Scholar] [CrossRef]
- Malato, S.; Blanco, J.; Alarcón, D.C.; Maldonado, M.I.; Fernández-Ibáñez, P.; Gernjak, W. Photocatalytic Decontamination and Disinfection of Water with Solar Collectors. Catal. Today 2007, 122, 137–149. [Google Scholar] [CrossRef]
- Zhang, Y.; Sivakumar, M.; Yang, S.; Enever, K.; Ramezanianpour, M. Application of Solar Energy in Water Treatment Processes: A Review. Desalination 2018, 428, 116–145. [Google Scholar] [CrossRef]
- Martín-Sómer, M.; Moreno-SanSegundo, J.; Álvarez-Fernández, C.; van Grieken, R.; Marugán, J. High-Performance Low-Cost Solar Collectors for Water Treatment Fabricated with Recycled Materials, Open-Source Hardware and 3d-Printing Technologies. Sci. Total Environ. 2021, 784, 147119. [Google Scholar] [CrossRef] [PubMed]
- Teixeira Andrade Queiroz, B.; Sabara, G.; Alvim, B.; Queiroz, A.; Leão, D.; Maria, M.; Amorim, C.; Teixeira Andrade Queiroz, M.; Godoy Sabara, M.; Barbosa Alvim, L.; et al. A Brief Review on the Importance Use of Solar Energy in the Treatment of Recalcitrant Effluents Applying Advanced Oxidation Processes. Ciência Nat. 2015, 37, 703–724. [Google Scholar] [CrossRef]
- Fendrich, M.A.; Quaranta, A.; Orlandi, M.; Bettonte, M.; Miotello, A. Solar Concentration for Wastewaters Remediation: A Review of Materials and Technologies. Appl. Sci. 2019, 9, 118. [Google Scholar] [CrossRef]
- Giannakis, S.; López, M.I.P.; Spuhler, D.; Pérez, J.A.S.; Ibáñez, P.F.; Pulgarin, C. Solar Disinfection Is an Augmentable, in Situ-Generated Photo-Fenton Reaction-Part 2: A Review of the Applications for Drinking Water and Wastewater Disinfection. Appl. Catal. B 2016, 198, 431–446. [Google Scholar] [CrossRef]
- Paliyal, P.S.; Mondal, S. Parabolic Solar Collectors for Sustainable Water Treatment: A Review of Applications, Advancements and Future Directions. Curr. Opin. Environ. Sci. Health 2025, 49, 100691. [Google Scholar] [CrossRef]
- Nascimento, C.A.O.; Teixeira, A.C.S.C.; Guardani, R.; Quina, F.H.; Chiavone-Filho, O.; Braun, A.M. Industrial Wastewater Treatment by Photochemical Processes Based on Solar Energy. J. Sol. Energy Eng. Trans. ASME 2007, 129, 45–52. [Google Scholar] [CrossRef]
- ESMAP. Global Solar Atlas 2.0 Technical Report. 170-14/2019; World Bank Group: Washington, DC, USA, 2019. [Google Scholar]
- C3S; WMO. European State of the Climate 2024; World Meteorological Organization (WMO): Geneva, Switzerland, 2025. [Google Scholar]
- Bolton, J.R.; Bircher, K.G.; Tumas, W.; Tolman, C.A. Figures-of-Merit for the Technical Development and Application of Advanced Oxidation Technologies for both Electric-and Solar-Driven Systems † (IUPAC Technical Report). Pure Appl. Chem. 2001, 73, 627–637. [Google Scholar] [CrossRef]
- Sakhrieh, A.H.; Alamir, A.I.; Hindiyeh, M.Y. Water Disinfection Using CSP Technology. 2016. Volume 11. Available online: https://www.ripublication.com/Volume/ijaerv11n15.htm (accessed on 14 August 2024).
- Zhang, C.; An, G.; Zhu, Y.; Sun, X.; Chen, G.; Yang, Y. Development of a Sunlight Adjustable Parabolic Trough Reactor for 24-Hour Efficient Photocatalytic Wastewater Treatment. Chem. Eng. J. 2024, 497, 154582. [Google Scholar] [CrossRef]
- Pucar Milidrag, G.; Prica, M.; Kerkez, D.; Dalmacija, B.; Kulic, A.; Tomasevic Pilipovic, D.; Becelic Tomin, M. A Comparative Study of the Decolorization Capacity of the Solar-Assisted Fenton Process Using Ferrioxalate and Al, Fe-Bentonite Catalysts in a Parabolic Trough Reactor. J. Taiwan Inst. Chem. Eng. 2018, 93, 436–449. [Google Scholar] [CrossRef]
- de Andrade Borges, T.; Chiavone-Filho, O.; Carlos Silva Costa Teixeira, A.; Luiz Foletto, E.; Luiz Dotto, G.; Augusto Oller do Nascimento, C. Degradation of Thiophanate-Methyl Fungicide by Photo-Fenton Process Using Lab-Scale Annular and Solar Tubular Reactors. Int. J. Environ. Technol. Manag. 2019, 22, 128–137. [Google Scholar] [CrossRef]
- Barzegar, M.H.; Sabzehmeidani, M.M.; Ghaedi, M.; Avargani, V.M.; Moradi, Z.; Roy, V.A.L.; Heidari, H. S-Scheme Heterojunction g-C3N4/TiO2 with Enhanced Photocatalytic Activity for Degradation of a Binary Mixture of Cationic Dyes Using Solar Parabolic Trough Reactor. Chem. Eng. Res. Des. 2021, 174, 307–318. [Google Scholar] [CrossRef]
- Khan, S.; Noor, T.; Iqbal, N.; Yaqoob, L. Photocatalytic Dye Degradation from Textile Wastewater: A Review. ACS Omega 2024, 9, 21751–21767. [Google Scholar] [CrossRef] [PubMed]
- Majhi, P.K.; Azam, R.; Kothari, R.; Arora, N.K.; Tyagi, V.V. Impact of Flow Rate in Integration with Solar Radiation on Color and COD Removal in Dye Contaminated Textile Industry Wastewater: Optimization Study. Energy Eng. 2022, 119, 419–427. [Google Scholar] [CrossRef]
- Jorge, A.M.S.; Athira, K.K.; Alves, M.B.; Gardas, R.L.; Pereira, J.F.B. Textile Dyes Effluents: A Current Scenario and the Use of Aqueous Biphasic Systems for the Recovery of Dyes. J. Water Process Eng. 2023, 55, 104125. [Google Scholar] [CrossRef]
- Abdel Dayem, A.M.; El-Ghetany, H.H.; El-Taweel, G.E.; Kamel, M.M. Thermal Performance and Biological Evaluation of Solar Water Disinfection Systems Using Parabolic Trough Collectors. Desalin. Water Treat. 2011, 36, 119–128. [Google Scholar] [CrossRef]
- Chaúque, B.J.M.; Jankoski, P.R.; Doyle, R.L.; Da Motta, A.S.; Benetti, A.D.; Rott, M.B. Pilot Scale Continuous-Flow Solar Water Disinfection System by Heating and Ultraviolet Radiation Inactivating Acanthamoeba Cysts and Bacillus Spores. J. Environ. Chem. Eng. 2023, 11, 110074. [Google Scholar] [CrossRef]
- Dixit, D.; Verma, A.; Gupta, S.; Bansal, P. Assessment of Solar Photocatalytic Degradation and Mineralization of Amoxicillin Trihydrate (AMT) Using Slurry and Fixed-Bed Batch Reactor: Efficacy of Parabolic Trough Collector. RSC Adv. 2016, 6, 36109–36117. [Google Scholar] [CrossRef]
- Singh, C.; Chaudhary, R.; Gandhi, K. Preliminary Study on Optimization of PH, Oxidant and Catalyst Dose for High COD Content: Solar Parabolic Trough Collector. Iran. J. Environ. Health Sci. Eng. 2013, 10, 13. [Google Scholar] [CrossRef]
- Tony, M.A.; Tayeb, A.M. Pre-Treatment of Olive Oil Mill Wastewaters Based on Solar Management Techniques: An Integrated Rational Approach. Eng. Res. J. 2015, 38, 65–71. Available online: https://journals.ekb.eg/article_66778_45166c33b716a0ab2050e5f5493cbd58.pdf (accessed on 22 October 2024). [CrossRef]
- Barwal, A.; Chaudhary, R. Feasibility Study for the Treatment of Municipal Wastewater by Using a Hybrid Bio-Solar Process. J. Environ. Manag. 2016, 177, 271–277. [Google Scholar] [CrossRef] [PubMed]
- El-Awady, M.H.; El-Ghetany, H. Experimental Investigation of An Industrial Wastewater Treatment Unit Using Multi Filter Hybrid with Parabolic Trough Solar Concentrator. Egypt. J. Chem. 2023, 66, 101–108. [Google Scholar] [CrossRef]
- Goutam, S.P.; Saxena, G.; Singh, V.; Yadav, A.K.; Bharagava, R.N.; Thapa, K.B. Green Synthesis of TiO2 Nanoparticles Using Leaf Extract of Jatropha curcas L. for Photocatalytic Degradation of Tannery Wastewater. Chem. Eng. J. 2018, 336, 386–396. [Google Scholar] [CrossRef]
- Pucar Milidrag, G.; Nikić, J.; Gvoić, V.; Kulić Mandić, A.; Agbaba, J.; Bečelić-Tomin, M.; Kerkez, D. Photocatalytic Degradation of Magenta Effluent Using Magnetite Doped TiO2 in Solar Parabolic Trough Concentrator. Catalysts 2022, 12, 986. [Google Scholar] [CrossRef]
- Tirado-Munõz, O.; Tirado-Ballestas, I.; Lopez, A.L.B.; Colina-Marquez, J. Heterogeneous Photocatalytic Pilot Plant for Cyanide Decontamination: A Novel Solar Rotary Photoreactor. J. Sol. Energy Eng. Trans. ASME 2022, 144, 051005. [Google Scholar] [CrossRef]
- Kabra, K.; Chaudhary, R.; Sawhney, R.L. Photocatalytic Reduction of Cr(VI) in Aqueous Titania Suspensions Exposed to Concentrated Solar Radiation. Int. J. Sustain. Energy 2007, 26, 195–207. [Google Scholar] [CrossRef]
- Kabra, K.; Chaudhary, R.; Sawhney, R.L. Effect of PH on Solar Photocatalytic Reduction and Deposition of Cu(II), Ni(II), Pb(II) and Zn(II): Speciation Modeling and Reaction Kinetics. J. Hazard. Mater. 2007, 149, 680–685. [Google Scholar] [CrossRef]
- Will, I.B.S.; Moraes, J.E.F.; Teixeira, A.C.S.C.; Guardani, R.; Nascimento, C.A.O. Photo-Fenton Degradation of Wastewater Containing Organic Compounds in Solar Reactors. Sep. Purif. Technol. 2004, 34, 51–57. [Google Scholar] [CrossRef]
- Sajjadi, S.A.; Afsharnia, M.; Azrah, K.; Javan, N.S.; Biglari, H. Humic Acid Degradation via Solar Photo-Fenton Process in Aqueous Environment. Iran. J. Health 2015, 2, 304–312. Available online: http://www.ijhse.ir/index.php/IJHSE/article/download/88/pdf_38 (accessed on 15 August 2024).
- Bandala, E.R.; Arancibia-Bulnes, C.A.; Orozco, S.L.; Estrada, C.A. Solar Photoreactors Comparison Based on Oxalic Acid Photocatalytic Degradation. Sol. Energy 2004, 77, 503–512. [Google Scholar] [CrossRef]
- Ahmad, N.; Gondal, M.A.; Sheikh, A.K. Comparative Study of Different Solar-Based Photo Catalytic Reactors for Disinfection of Contaminated Water. Desalination Water Treat. 2016, 57, 213–220. [Google Scholar] [CrossRef]
- Verma, A.; Tejo Prakash, N.; Toor, A.P.; Bansal, P.; Sangal, V.K.; Kumar, A. Concentrating and Nonconcentrating Slurry and Fixed-Bed Solar Reactors for the Degradation of Herbicide Isoproturon. J. Sol. Energy Eng. Trans. ASME 2018, 140, 021006. [Google Scholar] [CrossRef]
- Razavi, Z.; Mirghaffari, N.; Alemrajabi, A.A.; Davar, F.; Soleimani, M. Adsorption and Photocatalytic Removal of SO2 Using Natural and Synthetic Zeolites-Supported TiO2 in a Solar Parabolic Trough Collector. J. Clean. Prod. 2021, 310, 127376. [Google Scholar] [CrossRef]
- Abid, M.F.; Ebrahim, M.; Nafi, O.; Hussain, L.; Maneual, N.; Sameer, A. Designing and Operating a Pilot Plant for Purification of Industrial Wastewater from Toxic Organic Compounds by Utilizing Solar Energy. Korean J. Chem. Eng. 2014, 31, 1194–1203. [Google Scholar] [CrossRef]
- Zaruma-Arias, P.E.; Núñez-Núñez, C.M.; González-Burciaga, L.A.; Proal-Nájera, J.B. Solar Heterogenous Photocatalytic Degradation of Methylthionine Chloride on a Flat Plate Reactor: Effect of PH and H2O2 Addition. Catalysts 2022, 12, 132. [Google Scholar] [CrossRef]
- Yu, W.; Chen, J.; Ateia, M.; Cates, E.L.; Johnson, M.S. Do Gas Nanobubbles Enhance Aqueous Photocatalysis? Experiment and Analysis of Mechanism. Catalysts 2021, 11, 511. [Google Scholar] [CrossRef]
- Feng, W.; Deng, Y.; Yang, F.; Miao, Q.; Ngien, S.K. Systematic Review of Contaminants of Emerging Concern (CECs): Distribution, Risks, and Implications for Water Quality and Health. Water 2023, 15, 3922. [Google Scholar] [CrossRef]
- Tagle-Salazar, P.D.; Nigam, K.D.P.; Rivera-Solorio, C.I. Parabolic Trough Solar Collectors: A General Overview of Technology, Industrial Applications, Energy Market, Modeling, and Standards. Green Process. Synth. 2020, 9, 595–649. [Google Scholar] [CrossRef]
- Anisuzzaman, S.M.; Joseph, C.G.; Pang, C.K.; Affandi, N.A.; Maruja, S.N.; Vijayan, V. Current Trends in the Utilization of Photolysis and Photocatalysis Treatment Processes for the Remediation of Dye Wastewater: A Short Review. ChemEngineering 2022, 6, 58. [Google Scholar] [CrossRef]
- Ouaissa, Y.A.; Madi, N.E.H.; Chabani, M.; Bouafia-Chergui, S. Solar Advanced Oxidation Processes for Refinery Wastewater Treatment: Comparative Efficiencies, Modeling, and Feasibility for Cooling Tower Reuse. Water Air Soil Pollut. 2025, 236, 639. [Google Scholar] [CrossRef]


| Reactor Type | Design Principle | Advantages | Disadvantages | Key Performance Metrics |
|---|---|---|---|---|
| Raceway Pond Reactor (RPR) | Non-concentrating and open reactor. Usually, shallow hippodrome-shaped bathtub. Water circulation is maintained by paddle wheel system. | High capacity. Low-cost construction. Flexible design, easy to scale up. | Non-concentrating. Water loss due to evaporation. Effluent overflow in rain conditions. Limited to daylight. | CR: Non-concentrating System type: Open; possible overflow Flow regime: Laminar Irradiance: Direct and diffuse |
| Inclined Plate Collector (IPC) | Flat or corrugated inclined plate over which reactant fluid flows in thin film. Uses supported or immobilised photocatalysts. Uses both direct and diffuse radiation | Simple design and low cost. Large surface area for supported catalyst. Utilises both direct and diffuse radiation. | Low mass transfer rates due to laminar flow. With open and thin film of water stream, high losses of chemicals/water due to evaporation. Requires large surface area. Longer treatment times compared to other systems. | CR: 1 System type: Open Flow regime: Laminar Irradiance: Direct and diffuse |
| Compound Parabolic Collector (CPC) | Stationary collector with reflective parabolic surface to concentrate solar irradiance. Uses both direct and diffuse sunlight. Concentration ratio is between 1 and 15. | Utilises both direct and diffuse radiation. No tracking system, reducing complexity and cost. Homogeneous distribution of radiation onto receiver tube. High optical and quantum efficiency. | High initial investment costs. Requires large land area (for industrial implementation). Potentially excessive light irradiance and overheating. High manufacturing costs due to reflective mirror design. | CR: Low (1–1.5) System type: Closed Flow regime: Turbulent Irradiance: Direct and diffuse |
| Parabolic Trough Collector (PTC) | Uses parabolic reflective mirror to concentrate direct solar radiation onto receiver tube. Requires solar tracking system to keep the aperture perpendicular to the solar rays. | Receives large amount of energy per volume unit. Maintains turbulent conditions. Nearly closed system, volatile compounds do not vaporise. High UV intensity produces more hydroxyl radicals. | Uses direct irradiation, with lower efficiency on cloudy conditions. Requires tracking system. Risk of overheating. Excessive light irradiance can limit efficiency due to high electron/hole recombination. | CR: High (>15) System type: Closed Flow regime: Turbulent Irradiance: Direct |
| Linear Fresnel Reflectors (LFRs) | Line-focus system using array of segmented flat mirrors to track sun and reflect direct irradiation onto fixed receiver. Equipped with PTC or CPC. | Can achieve high concentration ratio with high thermal efficiency. Use small, flat mirrors for low-cost manufacture. Fixed receiver has large collection area, leading to simpler photoreactor design. | Require complex tracking mechanisms. Rely on direct sunlight radiation. Lower efficiency on cloudy days. Lower optical efficiency due to greater distance between mirrors and receiver. In sunny conditions, ultra-high illumination can limit photocatalytic activity. | CR: High (>15) System type: Closed Flow regime: Turbulent Irradiance: Direct |
| Criteria | Inclusion | Exclusion |
|---|---|---|
| Document type | Original peer-reviewed research articles | Non-research documents, books and book chapters, conference papers and review articles |
| Technology | AOPs utilising PTC reactors | AOPs utilising other CSP reactor designs |
| Water matrix | Both synthetic and real WW | Non-WW matrices |
| Light source | Solar; solar with artificial lamp | Only artificial UV systems |
| Scale | All experimental scales |
| Refs. | PTC System | Material | Outer Diameter (cm) | Inner Diameter (cm) | Receiver Length (cm) | Volume Irradiated (L) | Volume Treated (L) | Flow Rate (L/min) | Recirculation Number 1 |
|---|---|---|---|---|---|---|---|---|---|
| [17] | 1 | Quartz | 6.00 | 5.5 | 120 | 2.20 | 5 | 0.17 | 2 |
| [32] | 2 | Quartz | - 2 | 1.6 | 122 | 0.25 1 | 6 | 6.20 | 24 |
| [29] | 3 | Borosilicate | 5.80 | - | 170 | 5.00 | 5 | Batch | 1 |
| [25,49] | 4 | Borosilicate | - | 1.10 | 120 | 0.11 | 2 5 | 1.20 30.0 | 18 45 |
| [39,53] | 5 | Borosilicate | 2.54 | - | 125 | 0.63 | 6 10 | 1.00 | 10 16 |
| [40,47,48] | 6 | Borosilicate | - | 3.80 | 180 | 1.00 | 5 | 0.75 | 5 |
| [20] | 7 | Borosilicate | 2.80 | 2.60 | 38.0 | 0.20 | 2 | 12.3 | 10 |
| [46] | 8 | Borosilicate | 2.54 | 2.24 | 50.0 | 0.55 | 20 | 20.0 | 36 |
| [44] | 9 | Borosilicate | - | 2.5 | 96.0 | 0.47 | 5 | 15.0 | 11 |
| [37] | 10 | Pyrex | 2.54 | - | 200 | 1.01 | 100 | 4.5 | 99 |
| [31,45] | 11 | Pyrex | 1.86 | 1.26 | 130 | 0.18 | 1 | 0.05 | 6 |
| [43] | 12 | Pyrex | 10.7 | - | - | - | - | - | / 3 |
| [35] | 13 | Glass | - | - | 150 | 4.50 | 8 | 1.95 | 2 |
| [41] | 14 | Glass | - | - | 200 | - | - | 1.03 | / |
| [33] | 15 | Glass | 2.00 | - | 50.0 | 0.94 | 7 | - | 7 |
| [51] | 16 | Glass | 2.54 | - | - | - | 10 | - | / |
| [52] | 17 | Glass | 3.40 | 3.15 | - | - | 20 | 1.50 | / |
| [42] | 18 | Glass × 2 | - | 3.80 | 380 | 4.31 | 20 | 0.10 | 5 |
| [50] | 19 | PET | - | - | - | 1.00 | 1 | Batch | 1 |
| [30] | 20 | Non specified × 4 | 2 | - | - | - | 1 | - | / |
| Refs. | PTC System | Material | Length (cm) | Width (cm) | Aperture Area (m2) | Concentration Ratio | Photon Utilisation (m2/L) | Tracking Mechanism | Solar Intensity (W/m2) |
|---|---|---|---|---|---|---|---|---|---|
| [17] | 1 | Anodised aluminium | 110 | 95.0 | 1.05 | 6.00 | 0.48 1 | Yes, 2 axes | 700 2 |
| [32] | 2 | Polished aluminium | - | - | - | - | - | No, fixed | 900 2 |
| [29] | 3 | Galvanised steel | 170 | 18.0 | 0.34 | 1.11 | 0.07 | No, fixed | 650 2 |
| [25,49] | 4 | Polished aluminium | - | - | 0.26 | - | 2.28 | No, fixed | - |
| [39,53] | 5 | Stainless steel | 125 | 80.0 | 1.00 | 19.9 | 1.59 | Yes | 860 2 |
| [40,47,48] | 6 | - | 172 | 57.8 | 0.99 | 4.84 | 0.99 | Yes, 1 axis | 862 2 |
| [20] | 7 | Aluminium foil | 38.0 | 8.17 | 0.04 | 3.10 | 0.18 | Yes, 1 axis | - |
| [46] | 8 | Aluminium sheet | 50.0 | 30.9 | - | 3.87 | - | Yes | 55 2 |
| [44] | 9 | Aluminium chrome-plated | 104 | 84.0 | 0.87 | 10.7 | 1.85 | - | 985 2 |
| [37] | 10 | Aluminium foil | 200 | 27.2 | 0.54 | 3.41 | 0.54 | No, fixed | - |
| [31,45] | 11 | Stainless steel sheet | 130 | 113 | 1.47 | 19.3 | 8.37 | Yes | 1000 2 |
| [43] | 12 | Glass mirror | 300 | 100 | 3.00 | 3.13 | - | - | - |
| [35] | 13 | Aluminium sheet | 200 | - | - | - | - | Manual | 850 2 |
| [41] | 14 | Reflective material | 200 | 86.0 | 1.72 | - | - | - | 1220 2 |
| [33] | 15 | - | 140 | 50.0 | 0.70 | 7.96 | 0.74 | No, fixed | - |
| [51] | 16 | Aluminium sheet | - | - | 0.72 | 13.0 | - | Yes, 2 axes | - |
| [52] | 17 | Steel sheet | - | - | - | - | - | Yes | 750 2 |
| [42] | 18 | Polished aluminium | 172 | 57.8 | 0.99 | 4.84 | - | Manual | 700 2 |
| [50] | 19 | - | 30.0 | 100 | 3.00 | - | 3.00 | No | 2600 3 |
| [30] | 20 | Flexible mirror | - | 5.40–39.6 | - | 0.86– 6.30 | - | Yes | 1500 4 |
| Ref. | PTC System | Pollutant | Catalyst | H2O2 | Efficiency | Mineralisation | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Name | Conc. (mg/L) | Name | Conc. (g/L) | Conc. (mM) | pH | Time (min) | PFO 1 (min−1) | τ90 2 (min) | PFO (min−1) | τ90 (min) | ||
| Photolysis | ||||||||||||
| [35] | 13 | Dyes | RE | - | - | - | 8.2 | 360 | 0.0040 | 574.1 | 0.0043 | 531.1 |
| [31] | 11 | Reactive Red 120 | 100 | - | - | 5.00 | 7 | 120 | 0.0137 | 167.5 | ||
| [17] | 1 | Remazol Brilliant Blue | 60 | - | - | 100 | - | 180 | 0.0045 | 516.3 | 0.0069 | 334.8 |
| Ciprofloxacin | 10 | - | - | 10.0 | - | 180 | 0.0097 | 238.6 | 0.0005 | 4971 | ||
| Photo-Fenton | ||||||||||||
| [31] | 11 | Reactive Red 120 | 100 | CuOFeB | 0.050 | 5.00 | 7 | 120 | 0.0051 | 451.5 | 0.0058 | 398.6 |
| AlFeB | 0.100 | 5.00 | 4 | 120 | 0.0145 | 158.8 | 0.0058 | 398.6 | ||||
| [25] | 4 | Silicone emulsion | 653 | FeSO4 | 0.279 | 163 | 3 | 300 | 0.0074 | 313.0 | ||
| Phenol | 100 | FeSO4 | 0.056 | 100 | 3 | 240 | 0.0096 | 240.0 | ||||
| 550 | FeSO4 | 0.056 | 100 | 3 | 300 | 0.0020 | 1155 | |||||
| [49] | 4 | Phenol | 100 | FeSO4 | 0.028 | 7.00 | 3 | 180 | 0.0128 | 180.0 | ||
| [50] | 19 | Humic acid | 50 | FeSO4 | 0.004 | 0.59 | 4 | 30 | 0.1304 | 17.66 | ||
| [25] | 4 | Clomazone | 97 | FeSO4 | 0.056 | 13.5 | 3 | 180 | 0.0256 | 90.00 | ||
| [32] | 2 | Thiophanate-methyl | 1369 | FeSO4 | 0.034 | 0.04 | 3 | 120 | 0.0134 | 171.7 | ||
| [41] | 14 | Olive oil WW | - | Fe2+ salt | 0.194 | 19.9 | 3 | 40 | 0.0262 | 87.73 | ||
| [25] | 4 | Oil-field WW | 80 | FeSO4 | 0.056 | 200 | 3 | 270 | 0.0089 | 258.2 | ||
| Photocatalysis | ||||||||||||
| [33] | 15 | Methylene Blue | 10 | g-C3N4/TiO2 | 0.020 | - | 10 | 80 | 0.0231 | 99.68 | ||
| [30] | 20 | Methylene Blue | 5 | TiO2-SGB | - | - | - | - | 0.0190 | 121.2 | ||
| [33] | 15 | Rhodamine B | 15 | g-C3N4/TiO2 | 0.020 | - | 10 | 80 | 0.0191 | 120.6 | ||
| [30] | 20 | Rhodamine B | 2 | TiO2-SGB 4 | - | - | - | - | 0.0338 | 68.12 | ||
| Methyl Orange | 5 | TiO2-SGB | - | - | - | - | 0.0094 | 245.0 | ||||
| [53] | 5 | Isoproturon | 25 | TiO2-ICB 5 | 0.750 | 14.7 | 5 | 240 | 0.0080 | 287.8 | 0.0079 | 291.3 |
| [30] | 20 | Tetracycline | 10 | TiO2-SGB | - | - | - | - | 0.0201 | 114.6 | ||
| [39] | 5 | Amoxicillin trihydrate | 100 | TiO2-CCB 3 | 1.000 | 3.53 | 5.8 | 240 | 0.0105 | 218.8 | 0.0074 | 311.9 |
| [40] | 6 | Simulated organic WW | 7500 | TiO2 | 1.000 | 353 | 6.8 | 180 | 0.0109 | 210.8 | ||
| [42] | 18 | Municipal WW | 440 | TiO2 | 1.000 | - | 7.6 | 240 | 0.0049 | 471.8 | ||
| [44] | 9 | Tannery WW | 1428 | TiO2-NP 6 | 1.000 | - | 8.2 | 300 | 0.0318 | 72.43 | ||
| [46] | 8 | Cyanide gold mine WW | 2948 | TiO2 | 0.550 | - | 9.5 | 240 | 0.0048 | 475.8 | ||
| Cyanide | 50 | TiO2 | 0.300 | - | 9.5 | 180 | 0.0115 | 200.1 | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Kulić Mandić, A.; Pucar Milidrag, G.; Bečelić-Tomin, M.; Leovac Maćerak, A.; Slijepčević, N.; Duduković, N.; Kerkez, Đ. Solar Advanced Oxidation Processes Using Parabolic Trough Concentrators: A Mini-Review. Processes 2026, 14, 510. https://doi.org/10.3390/pr14030510
Kulić Mandić A, Pucar Milidrag G, Bečelić-Tomin M, Leovac Maćerak A, Slijepčević N, Duduković N, Kerkez Đ. Solar Advanced Oxidation Processes Using Parabolic Trough Concentrators: A Mini-Review. Processes. 2026; 14(3):510. https://doi.org/10.3390/pr14030510
Chicago/Turabian StyleKulić Mandić, Aleksandra, Gordana Pucar Milidrag, Milena Bečelić-Tomin, Anita Leovac Maćerak, Nataša Slijepčević, Nataša Duduković, and Đurđa Kerkez. 2026. "Solar Advanced Oxidation Processes Using Parabolic Trough Concentrators: A Mini-Review" Processes 14, no. 3: 510. https://doi.org/10.3390/pr14030510
APA StyleKulić Mandić, A., Pucar Milidrag, G., Bečelić-Tomin, M., Leovac Maćerak, A., Slijepčević, N., Duduković, N., & Kerkez, Đ. (2026). Solar Advanced Oxidation Processes Using Parabolic Trough Concentrators: A Mini-Review. Processes, 14(3), 510. https://doi.org/10.3390/pr14030510







