Purification and Characterization of a Novel Factor of Crotoxin Inter-CRO (V-1), a New Phospholipase A2 Isoform from Crotalus durissus collilineatus Snake Venom Using an In Vitro Neuromuscular Preparation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Venoms and Reagents
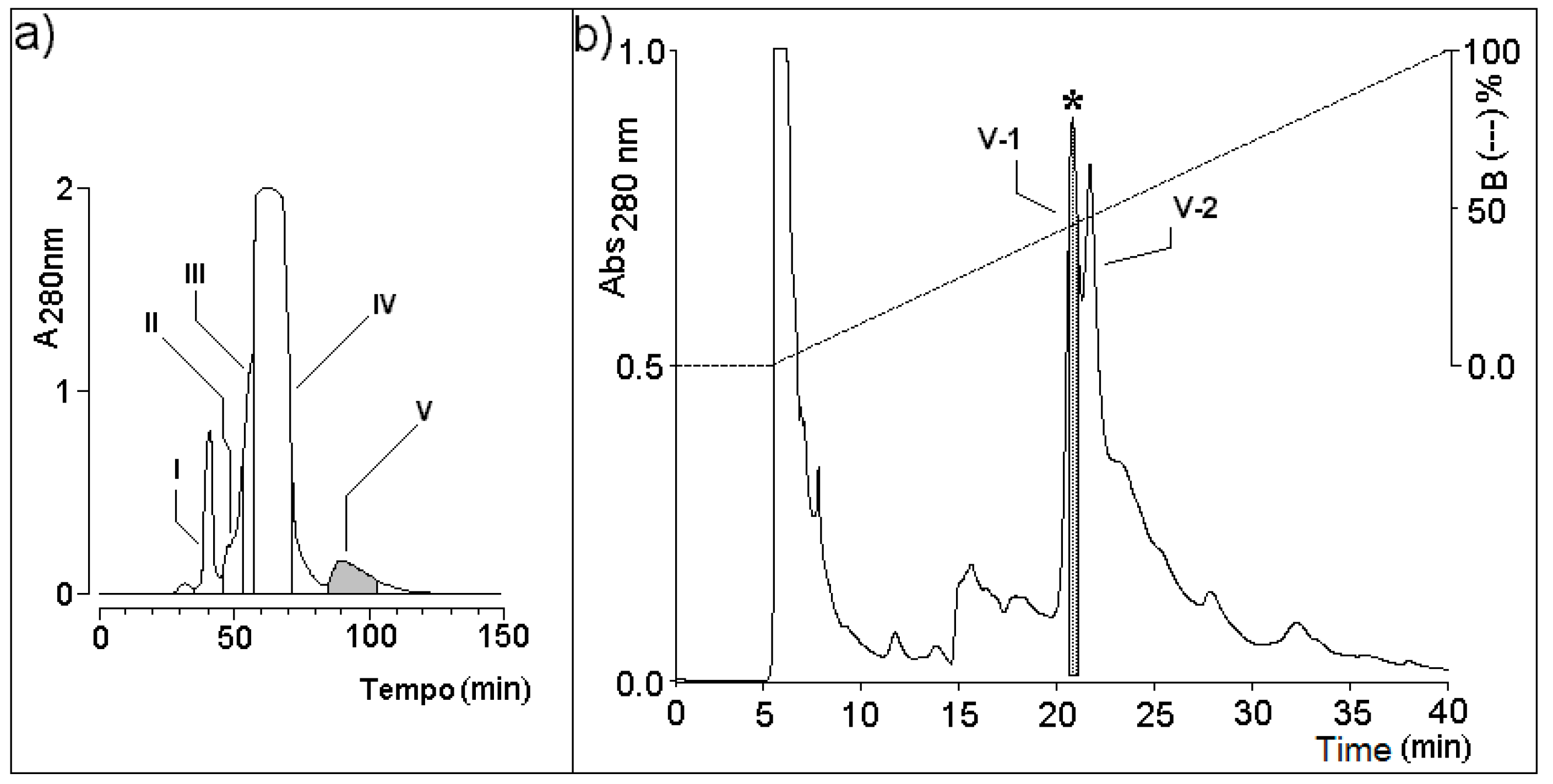
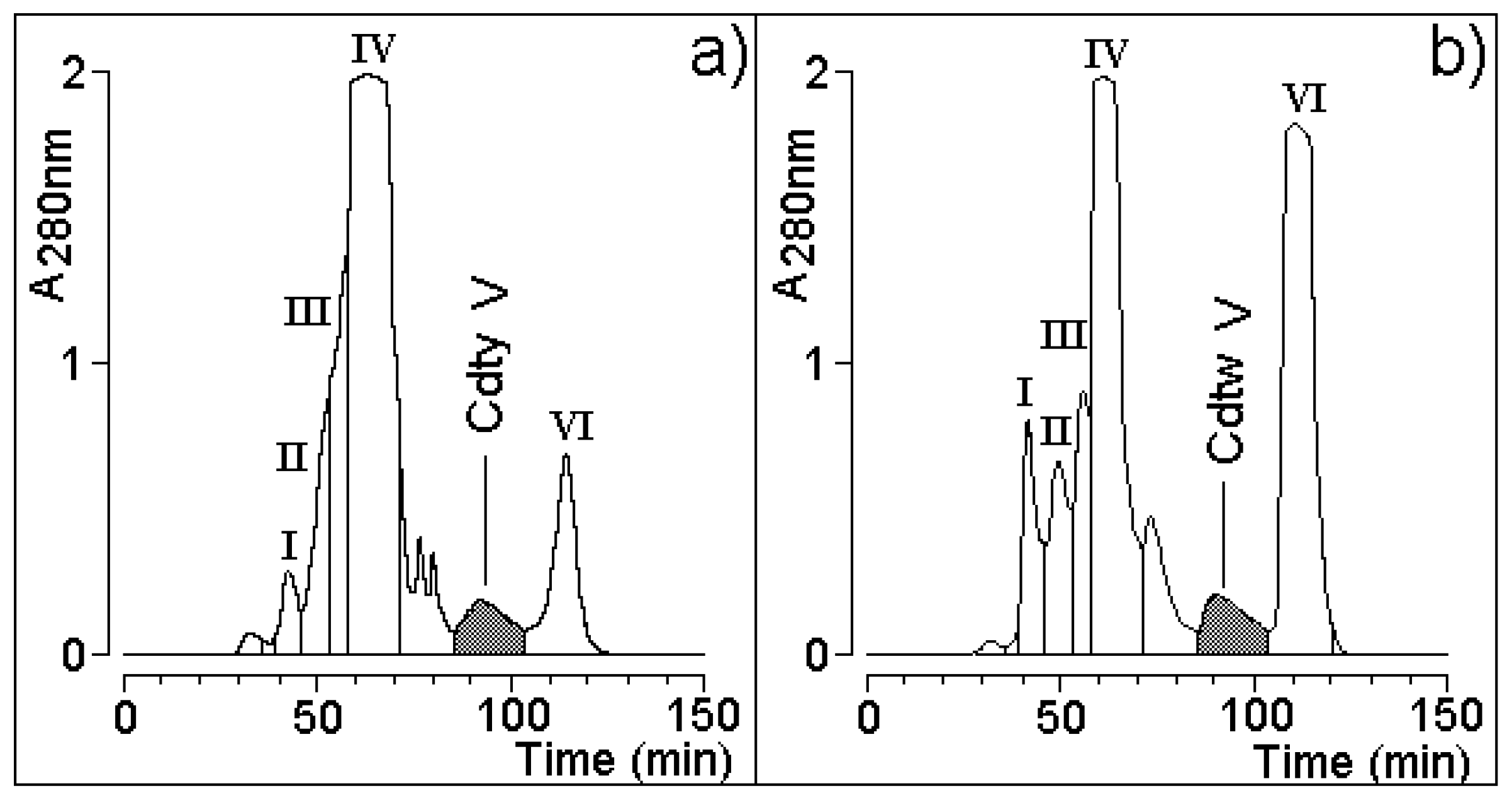
2.2. Molecular Exclusion HPLC
2.3. Reverse Phase HPLC (RP-HPLC)
2.4. Electrophoresis
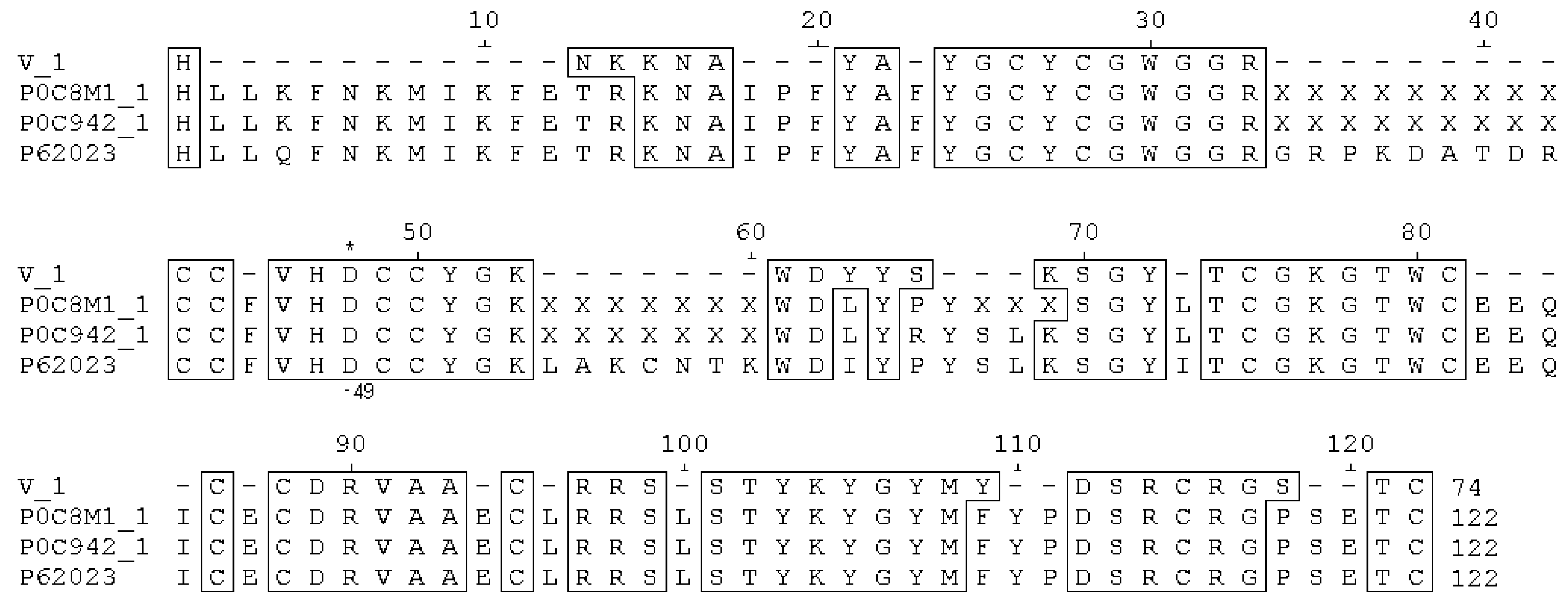
2.5. Sequencing Procedure
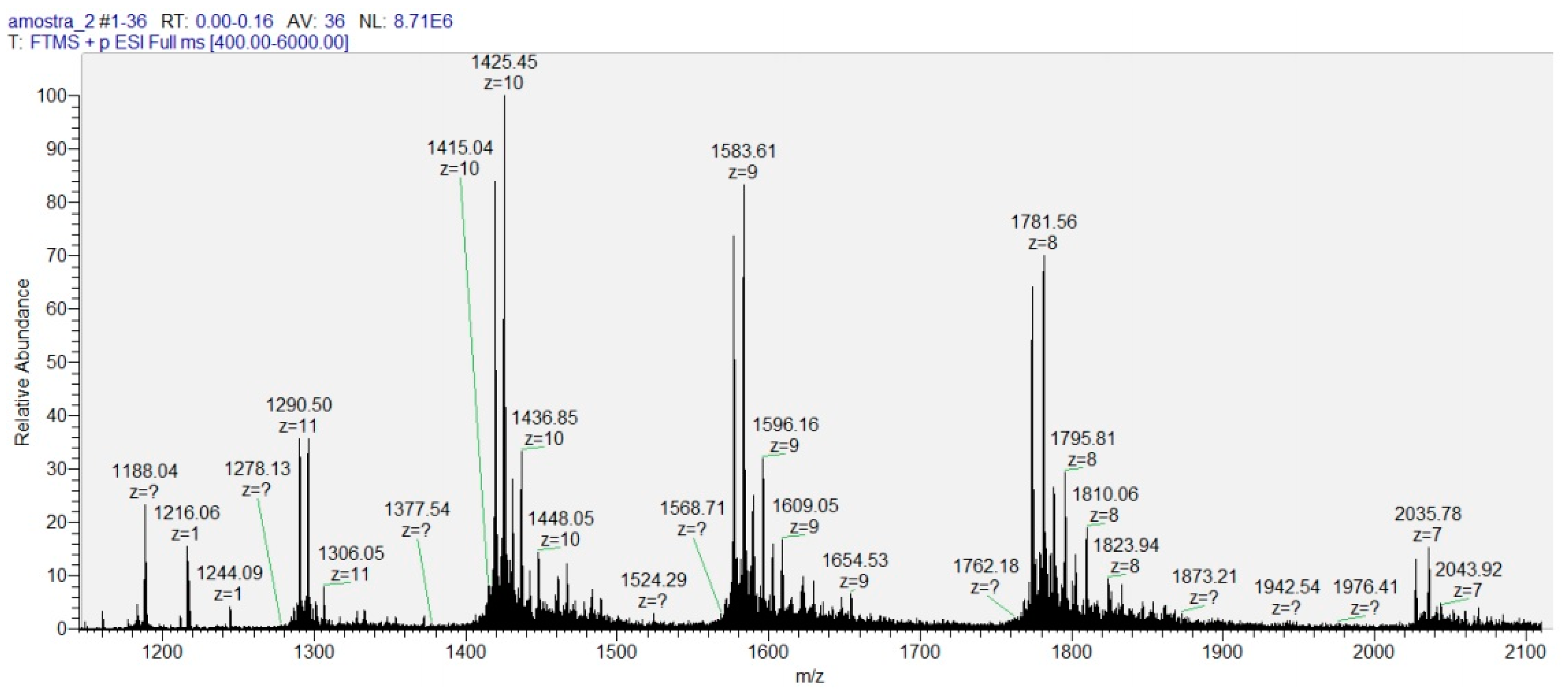
2.6. MALDI-TOF Mass Spectrometric (MS) Analysis
2.7. Molecular Mass Determination
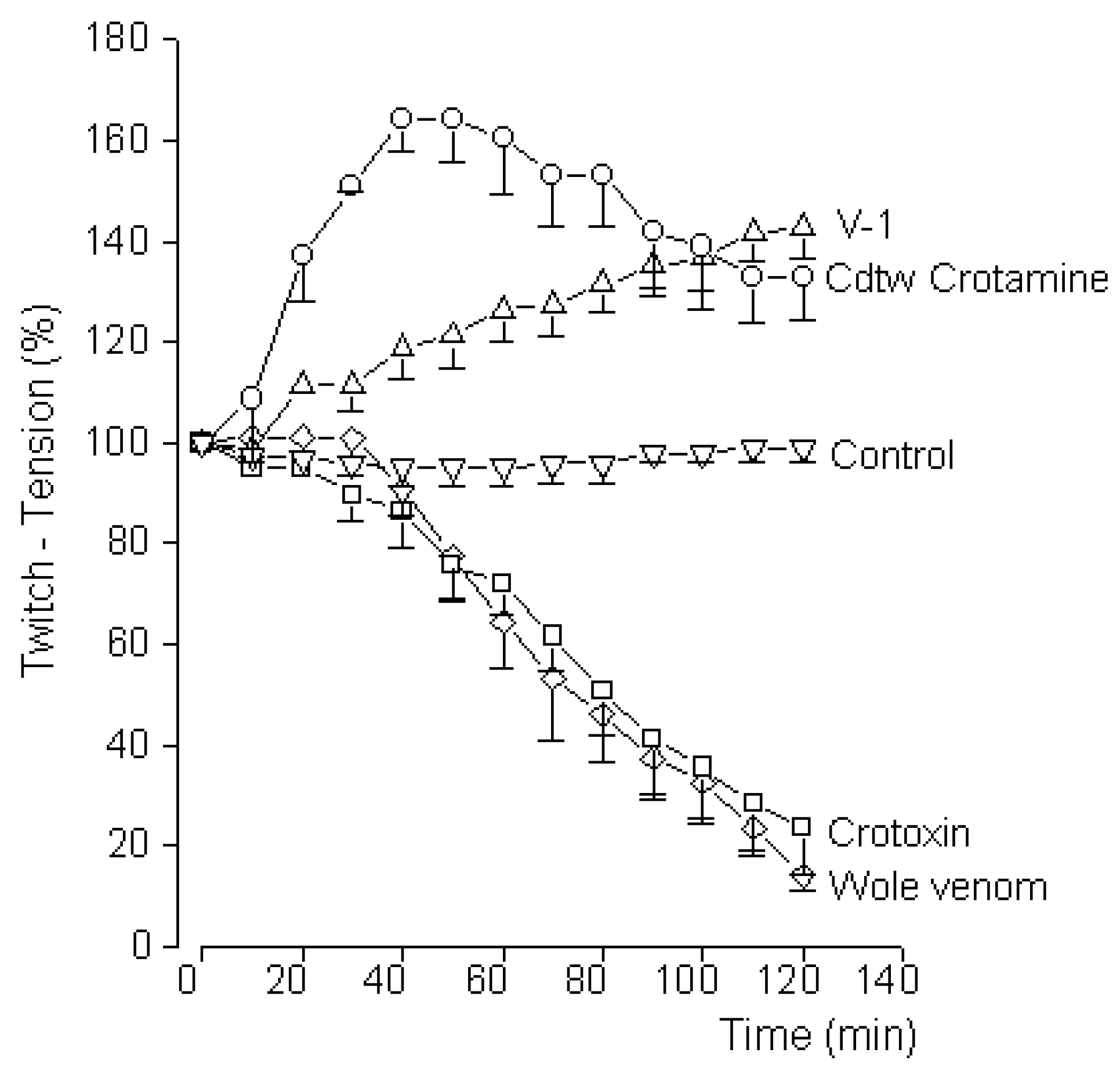
2.8. Mouse Phrenic Nerve-Diaphragm Preparation
2.9. Statistical Analysis
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Križaj, I.; Faure, G.; Gubenšek, F.; Bon, C. Neurotoxic Phospholipases A2 Ammodytoxin and Crotoxin Bind to Distinct High-Affinity Protein Acceptors in Torpedo marmorata Electric Organ. Biochemistry 1997, 36, 2779–2787. [Google Scholar] [CrossRef]
- Valente, R.H.; Novello, J.C.; Marangoni, S.; Oliveira, B.; Pereira-da-Silva, L.; Macedo, D.V. Mitochondrial swelling and oxygen consumption during respiratory state 4 induced by phospholipase A2 isoforms isolated from the South American rattlesnake (Crotalus durissus terrificus) venom. Toxicon 1998, 36, 901–913. [Google Scholar] [CrossRef]
- Faure, G.; Villela, C.; Perales, J.; Bon, C. Interaction of the neurotoxic and nontoxic secretory phospholipases A2 with the crotoxin inhibitor from Crotalus serum. Eur. J. Biochem. 2000, 267, 4799–4808. [Google Scholar] [CrossRef] [Green Version]
- Ponce-Soto, L.A.; Toyama, M.H.; Hyslop, S.; Novello, J.C.; Marangoni, S. Isolation and Preliminary Enzymatic Characterization of a Novel PLA2 from Crotalus durissus collilineatus Venom. J. Protein Chem. 2002, 21, 131–136. [Google Scholar] [CrossRef]
- Rangel-Santos, A.; Dos-Santos, E.C.; Lopes-Ferreira, M.; Lima, C.; Cardoso, D.F.; Mota, I. A comparative study of biological activities of crotoxin and CB fraction of venoms from Crotalus durissus terrificus, Crotalus durissus cascavella and Crotalus durissus collilineatus. Toxicon 2004, 43, 801–810. [Google Scholar] [CrossRef] [PubMed]
- Boldrini-França, J.; Rodrigues, R.S.; Fonseca, F.P.P.; Menaldo, D.L.; Ferreira, F.B.; Henrique-Silva, F.; Soares, A.M.; Hamaguchi, A.; Rodrigues, V.M.; Otaviano, A.R.; et al. Crotalus durissus collilineatus venom gland transcriptome: Analysis of gene expression profile. Biochimie 2009, 91, 586–595. [Google Scholar] [CrossRef] [PubMed]
- Amora, D.N.; Sousa, T.M.; Martins, A.M.C.; Barbosa, P.S.F.; Magalhães, M.R.; Toyama, M.H.; Fonteles, M.C.; de Menezes, D.B.; Monteiro, H.S.A. Effects of Crotalus durissus collilineatus venom in the isolated rat kidney. Toxicon 2006, 47, 260–264. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, I.S.; Cardoso, I.A.; Bordon, K.D.C.F.; Carone, S.E.I.; Boldrini-França, J.; Pucca, M.B.; Zoccal, K.F.; Faccioli, L.H.; Sampaio, S.V.; Rosa, J.C.; et al. Global proteomic and functional analysis of Crotalus durissus collilineatus individual venom variation and its impact on envenoming. J. Proteom. 2019, 191, 153–165. [Google Scholar] [CrossRef]
- Teixeira, N.B.; Sant’Anna, M.B.; Giardini, A.C.; Araujo, L.P.; Fonseca, L.A.; Basso, A.S.; Cury, Y.; Picolo, G. Crotoxin down-modulates pro-inflammatory cells and alleviates pain on the MOG35-55-induced experimental autoimmune encephalomyelitis, an animal model of multiple sclerosis. Brain Behav. Immun. 2020, 84, 253–268. [Google Scholar] [CrossRef]
- Moreira, L.A.; Oliveira, L.P.; Magalhães, M.R.; Oliveira, S.A.M.; Oliveira-Neto, J.R.; Carvalho, P.M.G.; Carvalho, A.A.V.; Fajemiroye, J.O.; Cruz, A.C.; Cunha, L.C. Acute toxicity, antinociceptive, and anti-inflammatory activities of the orally administered crotamine in mice. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2021, 394, 1703–1711. [Google Scholar] [CrossRef]
- Canelli, A.P.; dos Santos Rodrigues, T.F.; de Goes, V.F.; Caetano, G.F.; Mazzi, M.V. Evaluation of the Effectiveness of Crotoxin as an Antiseptic against Candida spp. Biofilms. Toxins 2020, 12, 532. [Google Scholar] [CrossRef]
- Favoretto, B.C.; Ricardi, R.; Silva, S.R.; Jacysyn, J.F.; Fernandes, I.; Takehara, H.A.; Faquim-Mauro, E.L. Immunomodulatory effects of crotoxin isolated from Crotalus durissus terrificus venom in mice immunised with human serum albumin. Toxicon 2011, 57, 600–607. [Google Scholar] [CrossRef]
- Minutti-Zanella, C.; Gil-Leyva, E.J.; Vergara, I. Immunomodulatory properties of molecules from animal venoms. Toxicon 2021, 191, 54–68. [Google Scholar] [CrossRef]
- Almeida, C.F.; Amaral, C.; Augusto, T.V.; Correia-da-Silva, G.; Marques de Andrade, C.; Torqueti, M.R.; Teixeira, N. The anti-cancer potential of crotoxin in estrogen receptor-positive breast cancer: Its effects and mechanism of action. Toxicon 2021, 200, 69–77. [Google Scholar] [CrossRef]
- Muller, S.P.; Silva, V.A.O.; Silvestrini, A.V.P.; de Macedo, L.H.; Caetano, G.F.; Reis, R.M.; Mazzi, M.V. Crotoxin from Crotalus durissus terrificus venom: In vitro cytotoxic activity of a heterodimeric phospholipase A2 on human cancer-derived cell lines. Toxicon 2018, 156, 13–22. [Google Scholar] [CrossRef] [Green Version]
- Sant’Anna, M.B.; Lopes, F.S.; Kimura, L.F.; Giardini, A.C.; Sant’Anna, O.A.; Picolo, G. Crotoxin Conjugated to SBA-15 Nanostructured Mesoporous Silica Induces Long-Last Analgesic Effect in the Neuropathic Pain Model in Mice. Toxins 2019, 11, 679. [Google Scholar] [CrossRef] [Green Version]
- Brazil, O.V. Neurotoxins from the South American rattle snake venom. Taiwan Yi Xue Hui Za Zhi 1972, 71, 394–400. [Google Scholar]
- Prado-Franceschi, J.; Vital Brazil, O. Convulxin, a new toxin from the venom of the South American rattlesnake Crotalus durissus terrificus. Toxicon 1981, 19, 875–887. [Google Scholar] [CrossRef]
- Toyama, M.H.; Carneiro, E.M.; Marangoni, S.; Amaral, M.E.C.; Velloso, L.A.; Boschero, A.C. Isolation and Characterization of a Convulxin-Like Protein from Crotalus durissus collilineatus Venom. J. Protein Chem. 2001, 20, 585–591. [Google Scholar] [CrossRef]
- Eble, J.A. Structurally Robust and Functionally Highly Versatile—C-Type Lectin (-Related) Proteins in Snake Venoms. Toxins 2019, 11, 136. [Google Scholar] [CrossRef] [Green Version]
- Rego, C.M.A.; Francisco, A.F.; Boeno, C.N.; Paloschi, M.V.; Lopes, J.A.; Silva, M.D.S.; Santana, H.M.; Serrath, S.N.; Rodrigues, J.E.; Lemos, C.T.L.; et al. Inflammasome NLRP3 activation induced by Convulxin, a C-type lectin-like isolated from Crotalus durissus terrificus snake venom. Sci. Rep. 2022, 12, 4706. [Google Scholar] [CrossRef]
- Alexander, G.; Grothusen, J.; Zepeda, H.; Schwartzman, R.J. Gyroxin, a toxin from the venom of Crotalus durissus terrificus, is a thrombin-like enzyme. Toxicon 1988, 26, 953–960. [Google Scholar] [CrossRef]
- Aguiar, A.S.; Alves, C.R.; Melgarejo, A.; Giovanni-de-Simone, S. Purification and partial characterization of a thrombin-like/gyroxin enzyme from bushmaster (Lachesis muta rhombeata) venom. Toxicon 1996, 34, 555–565. [Google Scholar] [CrossRef]
- Magalhaes, A.; Da Fonseca, B.C.B.; Diniz, C.R.; Gilroy, J.; Richardson, M. The complete amino acid sequence of a thrombin-like enzyme/gyroxin analogue from venom of the bushmaster snake (Lachesis muta muta). FEBS Lett. 1993, 329, 116–120. [Google Scholar] [CrossRef] [Green Version]
- Torres-Huaco, F.D.; Werneck, C.C.; Vicente, C.P.; Vassequi-Silva, T.; Nery-Diez, A.C.C.; Mendes, C.B.; Antunes, E.; Marangoni, S.; Damico, D.C.S. Rapid Purification and Procoagulant and Platelet Aggregating Activities of Rhombeobin: A Thrombin-Like/Gyroxin-Like Enzyme from Lachesis muta rhombeata Snake Venom. BioMed Res. Int. 2013, 2013, 903292. [Google Scholar] [CrossRef] [Green Version]
- Ullah, A.; Masood, R.; Ali, I.; Ullah, K.; Ali, H.; Akbar, H.; Betzel, C. Thrombin-like enzymes from snake venom: Structural characterization and mechanism of action. Int. J. Biol. Macromol. 2018, 114, 788–811. [Google Scholar] [CrossRef]
- Ferrari, C.Z.; Ribeiro, R.; Lima, A.M.; Soares, A.M.; Cavalcante, W.L.G.; Vieira, L.B. Gyroxin, a toxin from Crotalus durissus terrificus snake venom, induces a calcium dependent increase in glutamate release in mice brain cortical synaptosomes. Neuropeptides 2020, 83, 102081. [Google Scholar] [CrossRef]
- Bourillet, F. Comparative neuromuscular action of crotamine and veratrine. Ann. Pharm. Fr. 1970, 28, 535–544. [Google Scholar] [PubMed]
- Cheymol, J.; Gonçalves, J.M.; Bourillet, F.; Roch-Arveiller, M. A comparison of the neuromuscular action of crotamine and the venom of Crotalus durissus terrificus var. crotaminicus. 2. Isolated preparations. Toxicon Off. J. Int. Soc. Toxinology 1971, 9, 287–289. [Google Scholar] [CrossRef]
- Vital Brazil, O.; Fontana, M.D. Toxins as tools in the study of sodium channel distribution in the muscle fibre membrane. Toxicon 1993, 31, 1085–1098. [Google Scholar] [CrossRef]
- Fletcher, J.E.; Hubert, M.; Wieland, S.J.; Gong, Q.-H.; Jiang, M.-S. Similarities and differences in mechanisms of cardiotoxins, melittin and other myotoxins. Toxicon 1996, 34, 1301–1311. [Google Scholar] [CrossRef]
- Cameron, D.L.; Tu, A.T. Chemical and functional homology of myotoxin a from prairie rattlesnake venom and crotamine from south american rattlesnake venom. Biochim. Biophys. Acta BBA Protein Struct. 1978, 532, 147–154. [Google Scholar] [CrossRef]
- Matavel, A.C.S.; Ferreira-Alves, D.L.; Beirão, P.S.L.; Cruz, J.S. Tension generation and increase in voltage-activated Na+ current by crotamine. Eur. J. Pharmacol. 1998, 348, 167–173. [Google Scholar] [CrossRef]
- Vieira, L.F.; Magro, A.J.; Fernandes, C.A.; de Souza, B.M.; Cavalcante, W.L.; Palma, M.S.; Rosa, J.C.; Fuly, A.L.; Fontes, M.R.; Gallacci, M.; et al. Biochemical, functional, structural and phylogenetic studies on Intercro, a new isoform phospholipase A2 from Crotalus durissus terrificus snake venom. Biochimie 2013, 95, 2365–2375. [Google Scholar] [CrossRef] [Green Version]
- Toyama, M.H.; Carneiro, E.M.; Marangoni, S.; Barbosa, R.L.; Corso, G.; Boschero, A.C. Biochemical characterization of two crotamine isoforms isolated by a single step RP-HPLC from Crotalus durissus terrificus (South American rattlesnake) venom and their action on insulin secretion by pancreatic islets. Biochim. Biophys. Acta BBA Gen. Subj. 2000, 1474, 56–60. [Google Scholar] [CrossRef]
- Toyama, M.H.; de Oliveira, D.G.; Beriam, L.O.S.; Novello, J.C.; Rodrigues-Simioni, L.; Marangoni, S. Structural, enzymatic and biological properties of new PLA2 isoform from Crotalus durissus terrificus venom. Toxicon 2003, 41, 1033–1038. [Google Scholar] [CrossRef]
- Laure, C.J. Die primärstruktur des crotamins. Hoppe Seylers Z. Physiol. Chem 1975, 356, 213–216. [Google Scholar] [CrossRef]
- Smith, L.A.; Schmidt, J.J. Cloning and nucleotide sequences of crotamine genes. Toxicon 1990, 28, 575–585. [Google Scholar] [CrossRef]
- Rádis-Baptista, G.; Oguiura, N.; Hayashi, M.A.F.; Camargo, M.E.; Grego, K.F.; Oliveira, E.B.; Yamane, T. Nucleotide sequence of crotamine isoform precursors from a single South American rattlesnake (Crotalus durissus terrificus). Toxicon 1999, 37, 973–984. [Google Scholar] [CrossRef]
- Kerkis, I.; Hayashi, M.A.F.; Prieto da Silva, A.R.B.; Pereira, A.; De Sá Júnior, P.L.; Zaharenko, A.J.; Rádis-Baptista, G.; Kerkis, A.; Yamane, T. State of the Art in the Studies on Crotamine, a Cell Penetrating Peptide from South American Rattlesnake. BioMed Res. Int. 2014, 2014, 675985. [Google Scholar] [CrossRef] [Green Version]
- Siqueira, A.M.; Martins, N.F.; De Lima, M.E.; Diniz, C.R.; Cartier, A.; Brown, D.; Maigret, B. A proposed 3D structure for crotamine based on homology building, molecular simulations and circular dichroism. J. Mol. Graph. Model. 2002, 20, 389–398. [Google Scholar] [CrossRef]
- Schägger, H.; von Jagow, G. Tricine-sodium dodecyl sulfate-polyacrylamide gel electrophoresis for the separation of proteins in the range from 1 to 100 kDa. Anal. Biochem. 1987, 166, 368–379. [Google Scholar] [CrossRef]
- Dal Belo, C.; Toyama, M.; Toyama, D.; Marangoni, S.; Moreno, F.; Cavada, B.; Fontana, M.; Hyslop, S.; Carneiro, E.; Boschero, A. Determination of the Amino Acid Sequence of a New Phospholipase A2 (MIDCA1) Isolated from Micrurus dumerilii carinicauda Venom. Protein J. 2005, 24, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Ponce-Soto, L.A.; Bonfim, V.; Simioni, L.; Novello, J.; Marangoni, S. Determination of Primary Structure of Two Isoforms 6-1 and 6-2 PLA2 D49 from Bothrops jararacussu Snake Venom and Neurotoxic Characterization Using in vitro Neuromuscular Preparation. Protein J. 2006, 25, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Tabatabai, L.B.; Zehr, E.S. Identification of five outer membrane-associated proteins among cross-protective factor proteins of Pasteurella multocida. Infect Immun. 2004, 72, 1195–1198. [Google Scholar] [CrossRef] [Green Version]
- Da Silva, S.L.; Dias-Junior, C.A.; Baldasso, P.A.; Damico, D.C.S.; Carvalho, B.M.A.; Garanto, A.; Acosta, G.; Oliveira, E.; Albericio, F.; Soares, A.M.; et al. Vascular effects and electrolyte homeostasis of the natriuretic peptide isolated from Crotalus oreganus abyssus (North American Grand Canyon rattlesnake) venom. Peptides 2012, 36, 206–212. [Google Scholar] [CrossRef] [Green Version]
- Calgarotto, A.K.; Damico, D.C.; Ponce-Soto, L.A.; Baldasso, P.A.; Da Silva, S.L.; Souza, G.H.; Eberlin, M.N.; Marangoni, S. Biological and biochemical characterization of new basic phospholipase A(2) BmTX-I isolated from Bothrops moojeni snake venom. Toxicon 2008, 51, 1509–1519. [Google Scholar] [CrossRef]
- Cavada, B.S.; Bari, A.U.; Pinto-Junior, V.R.; Lossio, C.F.; Silva, M.T.L.; Souza, L.A.G.; Oliveira, M.V.; Souza-Filho, C.H.D.; Correia, S.E.G.; Vital, A.P.M.S.; et al. Purification and partial characterization of a new lectin from Parkia panurensis Benth. ex H.C. Hopkins seeds (Leguminosae family; Mimosoideae subfamily) and evaluation of its biological effects. Int. J. Biol. Macromol. 2020, 145, 845–855. [Google Scholar] [CrossRef]
- Guimarães, B.G.; Barbosa, R.L.; Soprano, A.S.; Campos, B.M.; de Souza, T.A.; Tonoli, C.C.C.; Leme, A.F.P.; Murakami, M.T.; Benedetti, C.E. Plant Pathogenic Bacteria Utilize Biofilm Growth-associated Repressor (BigR), a Novel Winged-helix Redox Switch, to Control Hydrogen Sulfide Detoxification under Hypoxia. J. Biol. Chem. 2011, 286, 26148–26157. [Google Scholar] [CrossRef] [Green Version]
- Bülbring, E. Observations on the Isolated Phrenic Nerve Diaphragm Preparation of the Rat. Br. J. Pharmacol. 1997, 120, 3–26. [Google Scholar] [CrossRef] [Green Version]
- Prado-Franceschi, J.; Hyslop, S.; Cogo, J.C.; Andrade, A.L.; Assakura, M.; Cruz-Höfling, M.A.; Rodrigues-Simioni, L. The effects of duvernoy’s gland secretion from the xenodontine colubrid Philodryas olfersii on striated muscle and the neuromuscular junction: Partial characterization of a neuromuscular fraction. Toxicon 1996, 34, 459–466. [Google Scholar] [CrossRef]
- Damico, D.C.S.; Lilla, S.; de Nucci, G.; Ponce-Soto, L.A.; Winck, F.V.; Novello, J.C.; Marangoni, S. Biochemical and enzymatic characterization of two basic Asp49 phospholipase A2 isoforms from Lachesis muta muta (Surucucu) venom. Biochim. Biophys. Acta BBA Gen. Subj. 2005, 1726, 75–86. [Google Scholar] [CrossRef]
- Fagundes, F.H.R.; Oliveira, M.; Huancahuire-Vega, S.; Romero-Vargas, F.F.; Ponce-Soto, L.A.; Marangoni, S. cDNA and deduced primary structure of basic phospholipase A2 with neurotoxic activity from the venom secretion of the Crotalus durissus collilineatus rattlesnake. Braz. J. Med. Biol. Res. 2010, 43, 262–270. [Google Scholar] [CrossRef] [Green Version]
- Habermann, E.; Breithaupt, H. The crotoxin complex—An example of biochemical and pharmacological protein complementation. Toxicon 1978, 16, 19–30. [Google Scholar] [CrossRef]
- Rowan, E.G.; Pemberton, K.E.; Harvey, A.L. On the blockade of acetylcholine release at mouse motor nerve terminals by β-bungarotoxin and crotoxin. Br. J. Pharmacol. 1990, 100, 301–304. [Google Scholar] [CrossRef]
- Križaj, I.; Bdolah, A.; Gubenšek, F.; Benčina, P.; Pungerčar, J. Protein and cDNA Structures of an Acidic Phospholipase A2, the Enzymatic Part of an Unusual, Two-Component Toxin fromVipera palaestinae. Biochem. Biophys. Res. Commun. 1996, 227, 374–379. [Google Scholar] [CrossRef]
- Bon, C.; Bouchier, C.; Choumet, V.; Faure, G.; Jiang, M.S.; Lambezat, M.P.; Radvanyi, F.; Saliou, B. Crotoxin, half-century of investigations on a phospholipase A2 neurotoxin. Acta Physiol Pharm. Lat. 1989, 39, 439–448. [Google Scholar]
- Rodrigues-Simioni, L.; Hawgood, B.J.; Smith, I.C.H. Properties of the early phases of crotoxin poisoning at frog neuromuscular junctions. Toxicon 1990, 28, 1479–1489. [Google Scholar] [CrossRef]
- Ponce-Soto, L.A.; Baldasso, P.A.; Romero-Vargas, F.F.; Winck, F.V.; Novello, J.C.; Marangoni, S. Biochemical, pharmacological and structural characterization of two PLA2 isoforms Cdr-12 and Cdr-13 from Crotalus durissus ruruima snake venom. Protein J. 2007, 26, 39–49. [Google Scholar] [CrossRef]
- Romero-Vargas, F.F.; Ponce-Soto, L.A.; Martins-de-Souza, D.; Marangoni, S. Biological and biochemical characterization of two new PLA2 isoforms Cdc-9 and Cdc-10 from Crotalus durissus cumanensis snake venom. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2010, 151, 66–74. [Google Scholar] [CrossRef]
- Bon, C.; Changeux, J.-P.; Jeng, T.-W.; Fraenkel-Conrat, H. Postsynaptic Effects of Crotoxin and of Its Isolated Subunits. Eur. J. Biochem. 1979, 99, 471–482. [Google Scholar] [CrossRef]
- Six, D.A.; Dennis, E.A. The expanding superfamily of phospholipase A2 enzymes: Classification and characterization. Biochim. Biophys. Acta BBA Mol. Cell Biol. Lipids 2000, 1488, 1–19. [Google Scholar] [CrossRef]
- Faure, G.; Harvey, A.L.; Thomson, E.; Saliou, B.; Radvanyi, F.; Bon, C. Comparison of crotoxin isoforms reveals that stability of the complex plays a major role in its pharmacological action. Eur. J. Biochem. 1993, 214, 491–496. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vera-Gonzales, C.; Arenas-Chávez, C.A.; Ponce-Soto, L.A.; Alvarez-Risco, A.; Del-Aguila-Arcentales, S.; Davies, N.M.; Yáñez, J.A. Purification and Characterization of a Novel Factor of Crotoxin Inter-CRO (V-1), a New Phospholipase A2 Isoform from Crotalus durissus collilineatus Snake Venom Using an In Vitro Neuromuscular Preparation. Processes 2022, 10, 1428. https://doi.org/10.3390/pr10071428
Vera-Gonzales C, Arenas-Chávez CA, Ponce-Soto LA, Alvarez-Risco A, Del-Aguila-Arcentales S, Davies NM, Yáñez JA. Purification and Characterization of a Novel Factor of Crotoxin Inter-CRO (V-1), a New Phospholipase A2 Isoform from Crotalus durissus collilineatus Snake Venom Using an In Vitro Neuromuscular Preparation. Processes. 2022; 10(7):1428. https://doi.org/10.3390/pr10071428
Chicago/Turabian StyleVera-Gonzales, Corina, Carlos Alberto Arenas-Chávez, Luis A. Ponce-Soto, Aldo Alvarez-Risco, Shyla Del-Aguila-Arcentales, Neal M. Davies, and Jaime A. Yáñez. 2022. "Purification and Characterization of a Novel Factor of Crotoxin Inter-CRO (V-1), a New Phospholipase A2 Isoform from Crotalus durissus collilineatus Snake Venom Using an In Vitro Neuromuscular Preparation" Processes 10, no. 7: 1428. https://doi.org/10.3390/pr10071428
APA StyleVera-Gonzales, C., Arenas-Chávez, C. A., Ponce-Soto, L. A., Alvarez-Risco, A., Del-Aguila-Arcentales, S., Davies, N. M., & Yáñez, J. A. (2022). Purification and Characterization of a Novel Factor of Crotoxin Inter-CRO (V-1), a New Phospholipase A2 Isoform from Crotalus durissus collilineatus Snake Venom Using an In Vitro Neuromuscular Preparation. Processes, 10(7), 1428. https://doi.org/10.3390/pr10071428









