Assessing Clinical Outcomes in Colorectal Cancer with Assays for Invasive Circulating Tumor Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient and Clinical Samples
2.2. Clinical Data Collection
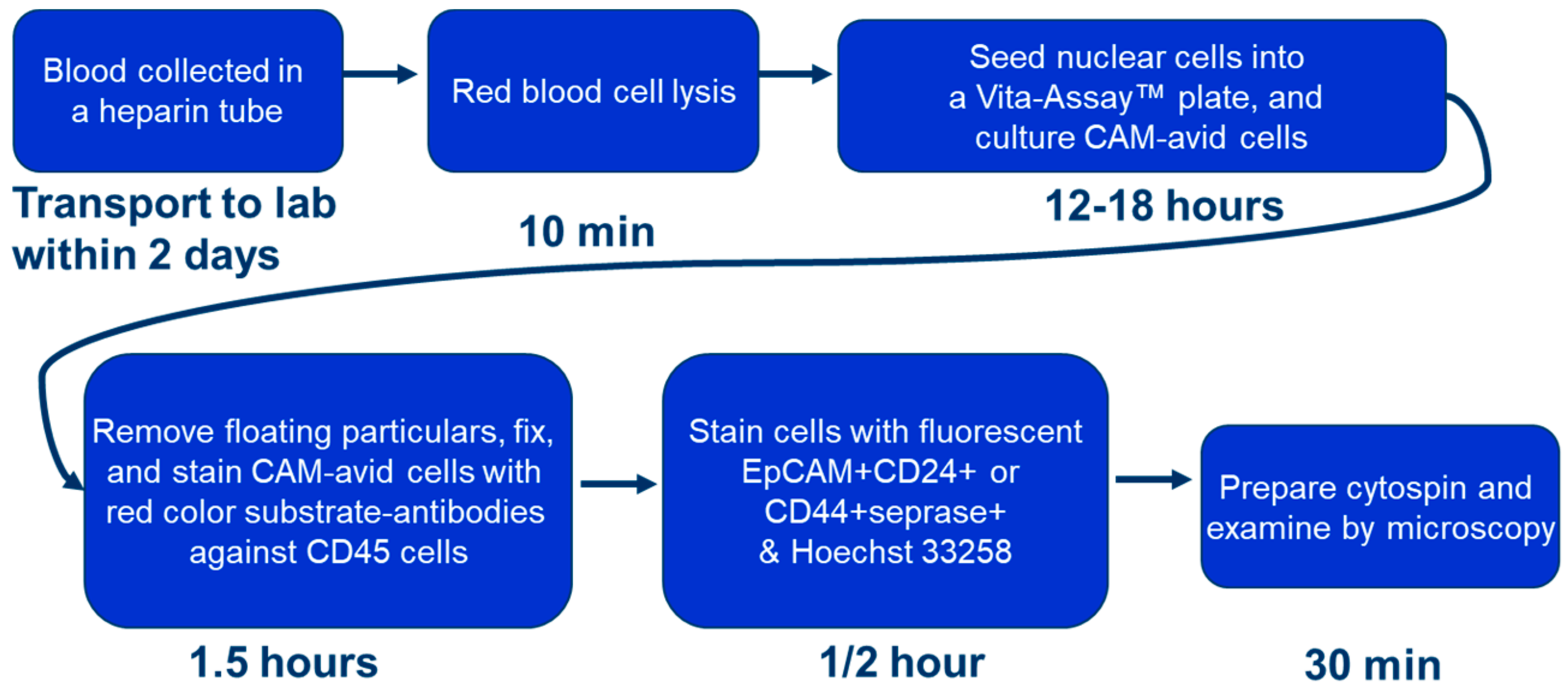
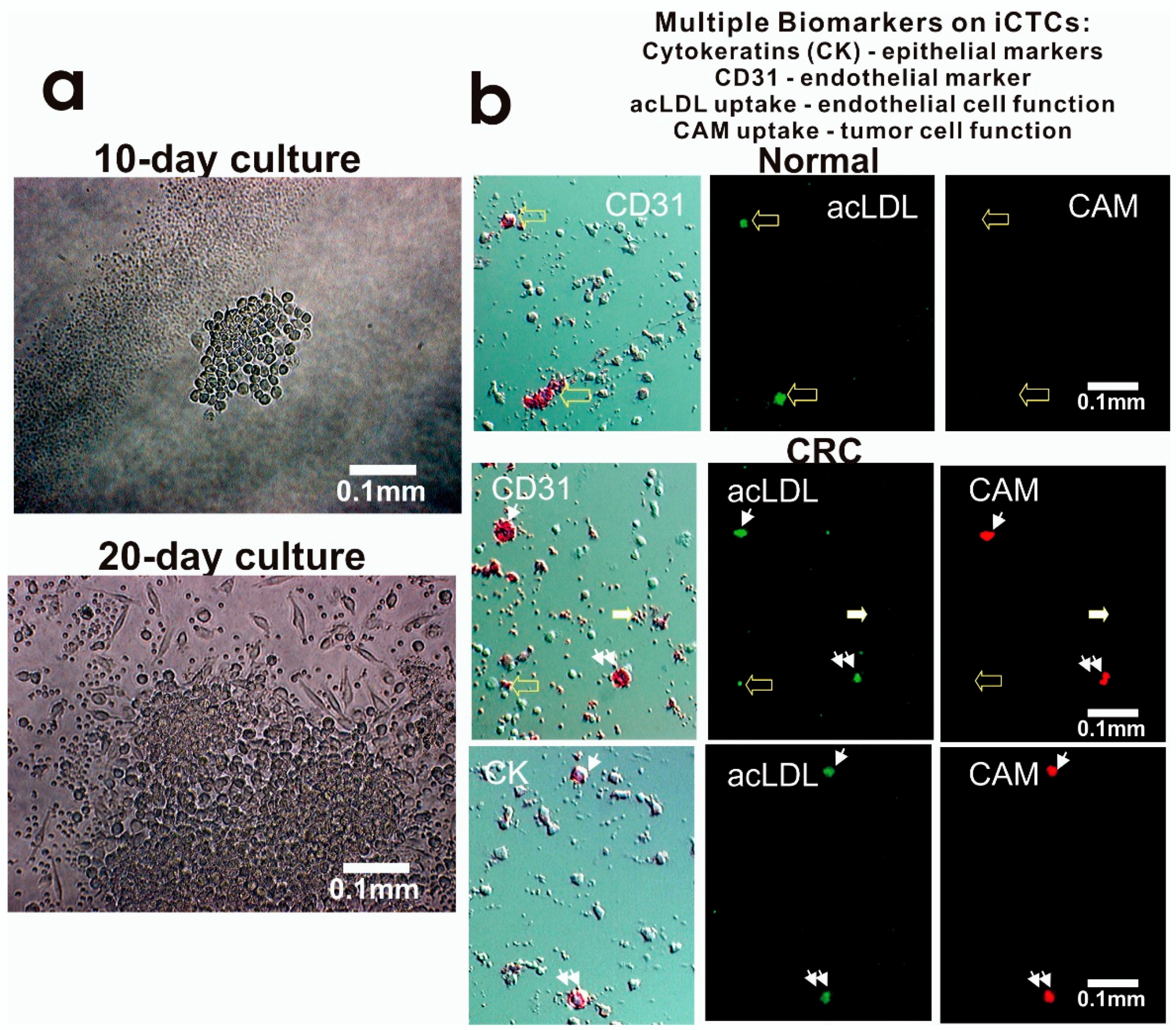
2.3. Cell Culture, Identification of CTCs and iCTCs, and Functional Proliferation/Invasion Assays
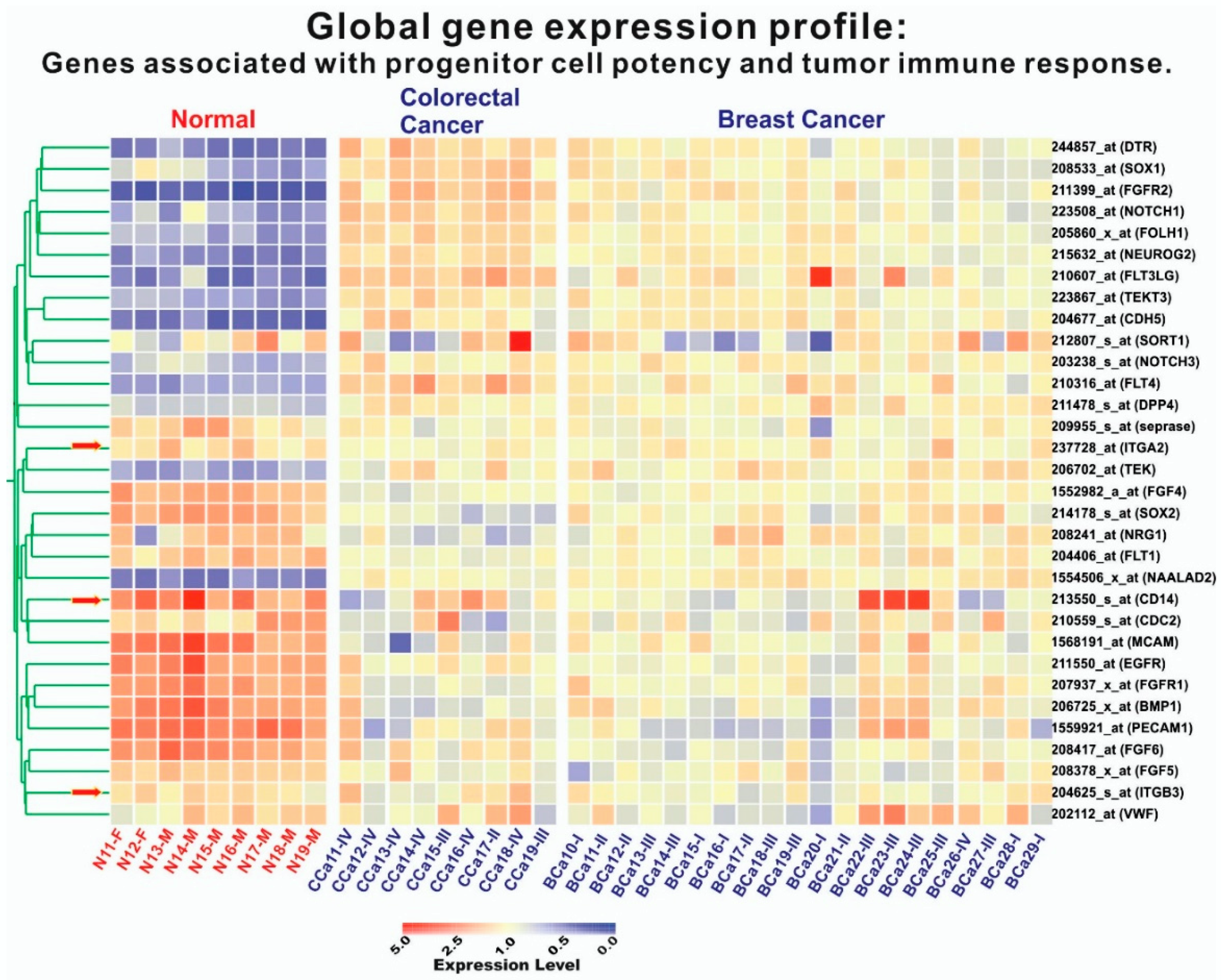
2.4. Microarray Data Analysis
2.5. Statistical Analysis
3. Results
3.1. Patient Characteristics
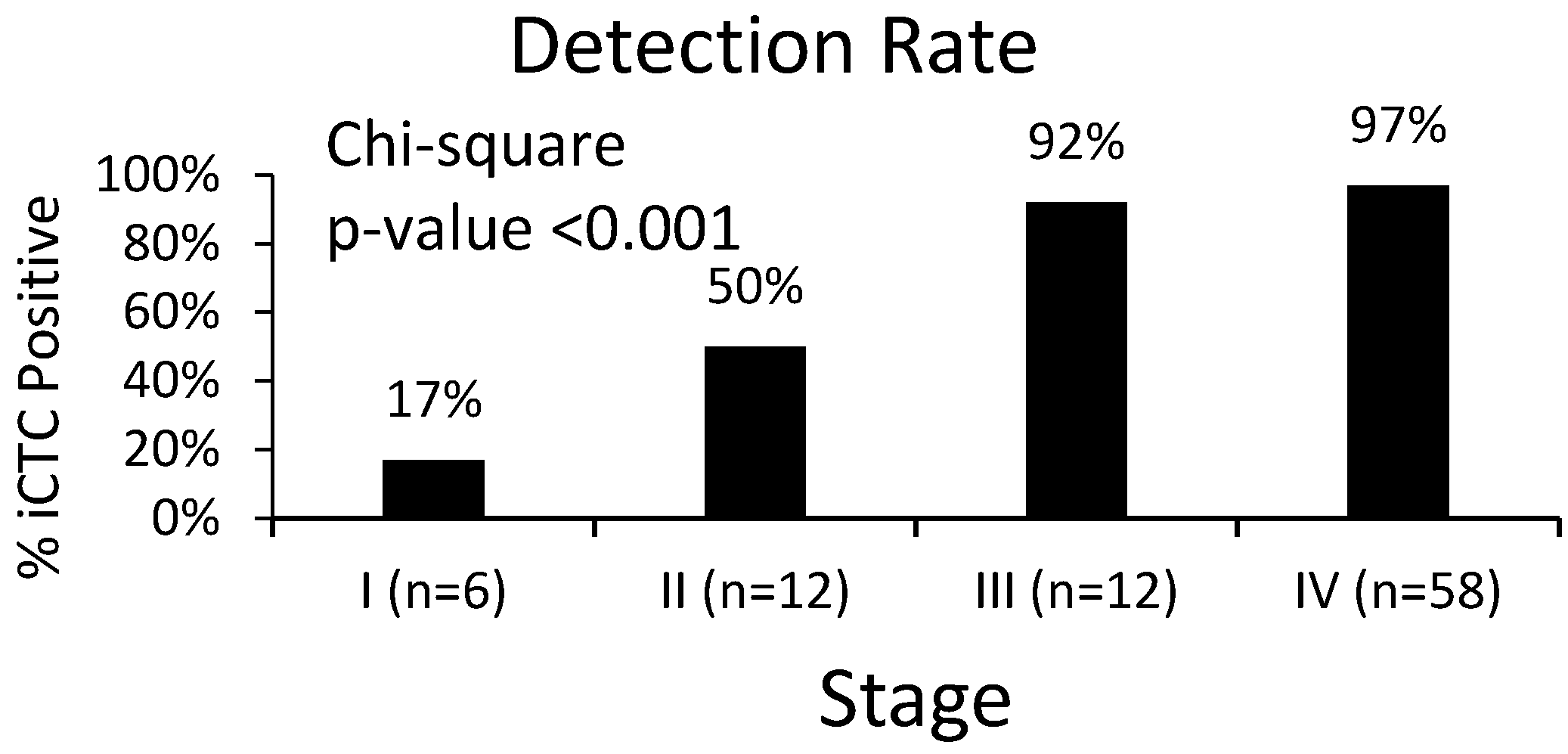
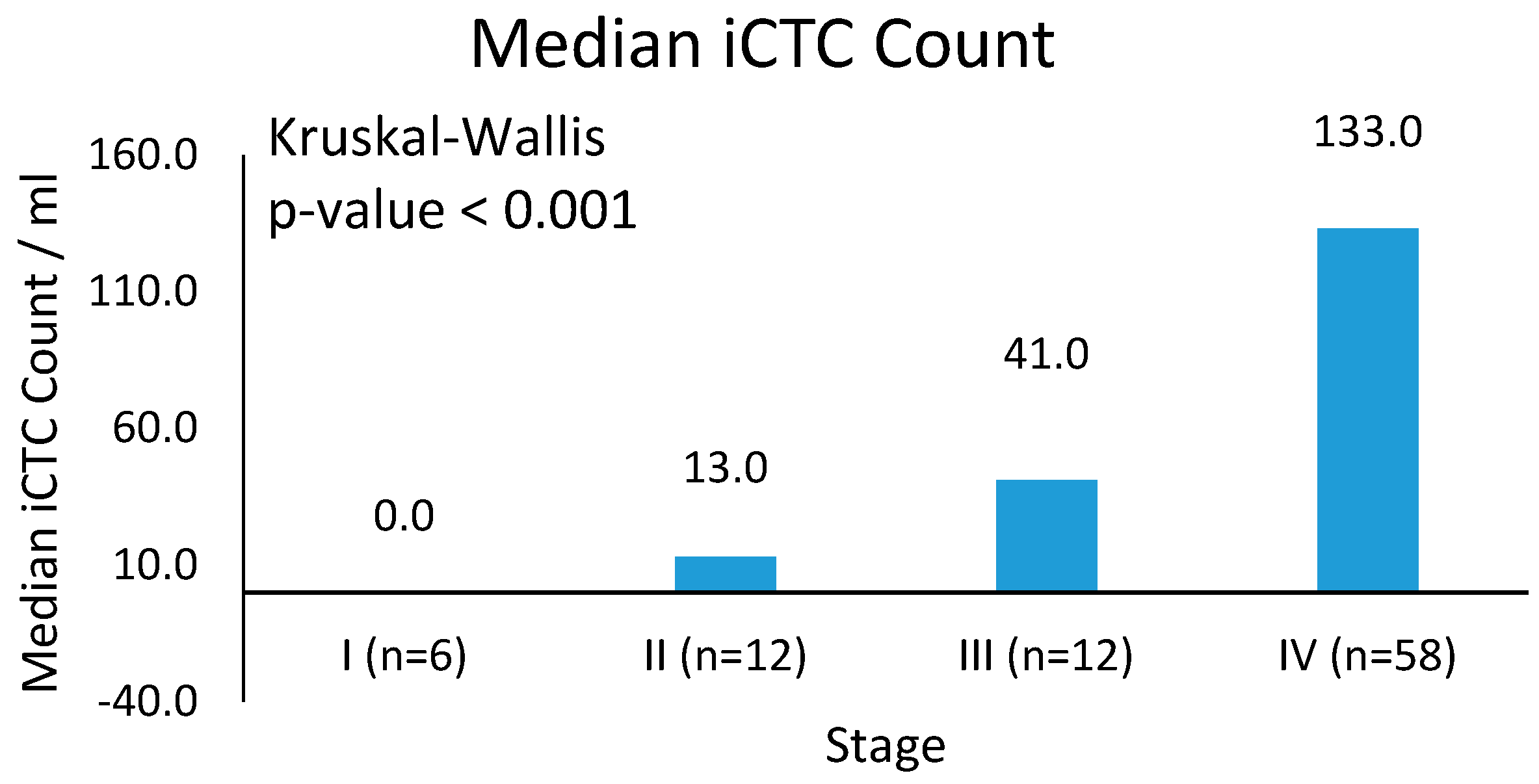
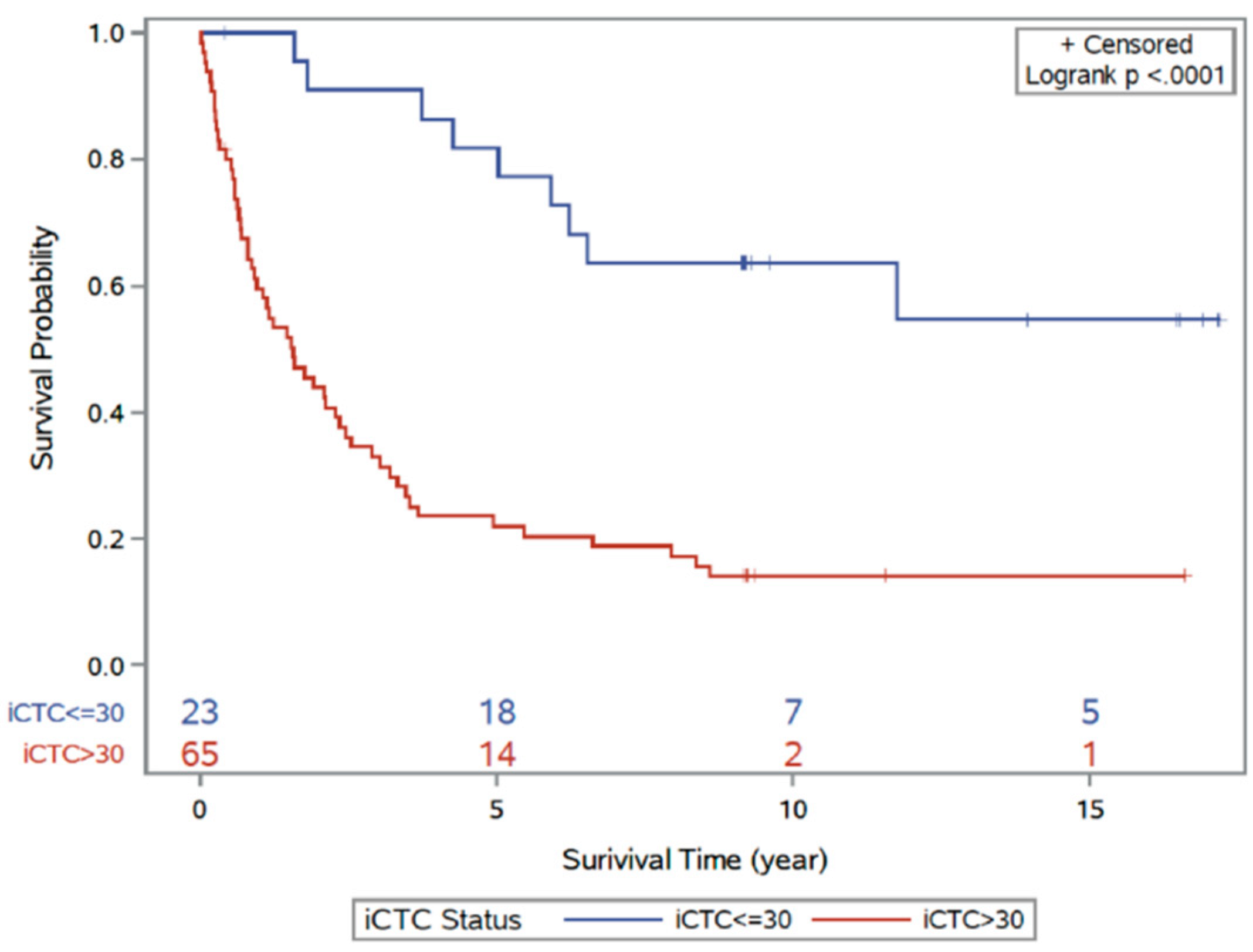
3.2. Clinical Characteristics of iCTCs: Stage and Survival
3.3. Molecular and Functional Phenotyping of iCTCs in CRC Patients
4. Discussion
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| Progression-free survival | PFS |
| Overall survival | OS |
| Colorectal carcinoma | CRC |
| Circulating tumor cells | CTCs |
| Epithelial cell adhesion molecule | EpCAM |
| Collagen adhesion matrix | CAM |
| Epithelial markers | Epi+ |
| Hazard ratio | HR |
| Confidence interval | CI |
| Akaike information criterion | AIC |
| Invasive CTCs | iCTCs |
| Carcinoembryonic antigen | CEA |
References
- American Cancer Society. Cancer Facts & Figures; The Society: Atlanta, GA, USA, 2017. [Google Scholar]
- Brenner, H.; Kloor, M.; Pox, C.P. Colorectal cancer. Lancet 2014, 383, 1490–1502. [Google Scholar] [CrossRef]
- Torino, F.; Bonmassar, E.; Bonmassar, L.; De Vecchis, L.; Barnabei, A.; Zuppi, C.; Capoluongo, E.; Aquino, A. Circulating tumor cells in colorectal cancer patients. Cancer Treat Rev. 2013, 39, 759–772. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, M.; Inoue, Y.; Komeda, K.; Shimizu, T.; Asakuma, M.; Hirokawa, F.; Miyamoto, Y.; Okuda, J.; Takeshita, A.; Shibayama, Y.; et al. Clinicopathological analysis of recurrence patterns and prognostic factors for survival after hepatectomy for colorectal liver metastasis. BMC Surg. 2010, 10, 27. [Google Scholar] [CrossRef] [PubMed]
- Fakih, M.G. Metastatic colorectal cancer: Current state and future directions. J. Clin. Oncol. 2015, 33, 1809–1824. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.M.; Krebs, M.; Ward, T.; Sloane, R.; Priest, L.; Hughes, A.; Clack, G.; Ranson, M.; Blackhall, F.; Dive, C. Circulating tumor cells as a window on metastasis biology in lung cancer. Am. J. Pathol. 2011, 178, 989–996. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.Y.; Tsai, H.L.; Huang, J.J.; Wang, J.Y. Clinical Implications and Future Perspectives of Circulating Tumor Cells and Biomarkers in Clinical Outcomes of Colorectal Cancer. Transl. Oncol. 2016, 9, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Sequist, L.V.; Nagrath, S.; Toner, M.; Haber, D.A.; Lynch, T.J. The CTC-chip: An exciting new tool to detect circulating tumor cells in lung cancer patients. J. Thorac. Oncol. 2009, 4, 281–283. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Fan, T.; Zhao, Q.; Zeng, W.; Zaslavsky, E.; Chen, J.J.; Frohman, M.A.; Golightly, M.G.; Madajewicz, S.; Chen, W.T. Isolation of circulating epithelial and tumor progenitor cells with an invasive phenotype from breast cancer patients. Int. J. Cancer 2010, 126, 669–683. [Google Scholar] [CrossRef] [PubMed]
- Pearl, M.L.; Zhao, Q.; Yang, J.; Dong, H.; Tulley, S.; Zhang, Q.; Golightly, M.; Zucker, S.; Chen, W.T. Prognostic analysis of invasive circulating tumor cells (iCTCs) in epithelial ovarian cancer. Gynecol. Oncol. 2014, 134, 581–590. [Google Scholar] [CrossRef] [PubMed]
- Fan, T.; Zhao, Q.; Chen, J.J.; Chen, W.T.; Pearl, M.L. Clinical significance of circulating tumor cells detected by an invasion assay in peripheral blood of patients with ovarian cancer. Gynecol. Oncol. 2009, 112, 185–191. [Google Scholar] [CrossRef]
- Pearl, M.L.; Dong, H.; Tulley, S.; Zhao, Q.; Golightly, M.; Zucker, S.; Chen, W.T. Treatment monitoring of patients with epithelial ovarian cancer using invasive circulating tumor cells (iCTCs). Gynecol. Oncol. 2015, 137, 229–238. [Google Scholar] [CrossRef] [PubMed]
- Paris, P.L.; Kobayashi, Y.; Zhao, Q.; Zeng, W.; Sridharan, S.; Fan, T.; Adler, H.L.; Yera, E.R.; Zarrabi, M.H.; Zucker, S.; et al. Functional phenotyping and genotyping of circulating tumor cells from patients with castration resistant prostate cancer. Cancer Lett. 2009, 277, 164–173. [Google Scholar] [CrossRef] [PubMed]
- Tulley, S.; Zhao, Q.; Dong, H.; Pearl, M.L.; Chen, W.T. Vita-Assay (TM) Method of Enrichment and Identification of Circulating Cancer Cells/Circulating Tumor Cells (CTCs). Methods Mol. Biol. 2016, 1406, 107–119. [Google Scholar] [PubMed]
- Friedlander, T.W.; Ngo, V.T.; Dong, H.; Premasekharan, G.; Weinberg, V.; Doty, S.; Zhao, Q.; Gilbert, E.G.; Ryan, C.J.; Chen, W.T.; et al. Detection and characterization of invasive circulating tumor cells derived from men with metastatic castration-resistant prostate cancer. Int. J. Cancer 2014, 134, 2284–2293. [Google Scholar] [CrossRef] [PubMed]
- Tsai, W.S.; Chen, J.S.; Shao, H.J.; Wu, J.C.; Lai, J.M.; Lu, S.H.; Hung, T.F.; Chiu, Y.C.; You, J.F.; Hsieh, P.S.; et al. Circulating Tumor Cell Count Correlates with Colorectal Neoplasm Progression and Is a Prognostic Marker for Distant Metastasis in Non-Metastatic Patients. Sci. Rep. 2016, 6, 24517. [Google Scholar] [CrossRef] [PubMed]
- Alix-Panabieres, C.; Bartkowiak, K.; Pantel, K. Functional studies on circulating and disseminated tumor cells in carcinoma patients. Mol. Oncol. 2016, 10, 443–449. [Google Scholar] [CrossRef] [PubMed]
- Asahara, T.; Murohara, T.; Sullivan, A.; Silver, M.; van der Zee, R.; Li, T.; Witzenbichler, B.; Schatteman, G.; Isner, J.M. Isolation of putative progenitor endothelial cells for angiogenesis. Science 1997, 275, 964–967. [Google Scholar] [CrossRef] [PubMed]
- Micalizzi, D.S.; Haber, D.A.; Maheswaran, S. Cancer metastasis through the prism of epithelial to mesenchymal transition in circulating tumor cells. Mol. Oncol. 2017, 11, 770–780. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.J.; Punt, C.J.; Iannotti, N.; Saidman, B.H.; Sabbath, K.D.; Gabrail, N.Y.; Picus, J.; Morse, M.; Mitchell, E.; Miller, M.C.; et al. Relationship of circulating tumor cells to tumor response, progression-free survival, and overall survival in patients with metastatic colorectal cancer. J. Clin. Oncol. 2008, 26, 3213–3221. [Google Scholar] [CrossRef] [PubMed]
- Tol, J.; Koopman, M.; Miller, M.C.; Tibbe, A.; Cats, A.; Creemers, G.J.; Vos, A.H.; Nagtegaal, I.D.; Terstappen, L.W.; Punt, C.J. Circulating tumour cells early predict progression-free and overall survival in advanced colorectal cancer patients treated with chemotherapy and targeted agents. Ann. Oncol. 2010, 21, 1006–1012. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Zhang, Z.; Gao, X.H.; Shen, Z.; Jing, Y.; Lu, H.; Li, H.; Yang, X.; Cui, X.; Li, Y.; et al. Clinical significance of detecting circulating tumor cells in colorectal cancer using subtraction enrichment and immunostaining-fluorescence in situ hybridization (SE-iFISH). Oncotarget 2017, 8, 21639–21649. [Google Scholar] [CrossRef] [PubMed]
- Cabel, L.; Proudhon, C.; Gortais, H.; Loirat, D.; Coussy, F.; Pierga, J.Y.; Bidard, F.C. Circulating tumor cells: Clinical validity and utility. Int. J. Clin. Oncol. 2017. 22, 421–430. [CrossRef]
- León-Mateos, L.; Casas, H.; Abalo, A.; Vieito, M.; Abreu, M.; Anido, U.; Gómez-Tato, A.; López, R.; Abal, M.; Muinelo-Romay, L. Improving circulating tumor cells enumeration and characterization to predict outcome in first line chemotherapy mCRPC patients. Oncotarget 2017, 8, 54708–54721. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.Y.; Yeh, C.S.; Tzou, W.S.; Hsieh, J.S.; Chen, F.M.; Lu, C.Y.; Yu, F.J.; Cheng, T.L.; Huang, T.J.; Lin, S.R. Analysis of progressively overexpressed genes in tumorigenesis of colorectal cancers using cDNA microarray. Oncol. Rep. 2005, 14, 65–72. [Google Scholar] [PubMed]
- Wang, J.Y.; Lin, S.R.; Wu, D.C.; Lu, C.Y.; Yu, F.J.; Hsieh, J.S.; Cheng, T.L.; Koay, L.B.; Uen, Y.H. Multiple molecular markers as predictors of colorectal cancer in patients with normal perioperative serum carcinoembryonic antigen levels. Clin. Cancer Res. 2007, 13, 2406–2413. [Google Scholar] [CrossRef] [PubMed]
- Paterlini-Brechot, P.; Benali, N.L. Circulating tumor cells (CTC) detection: Clinical impact and future directions. Cancer Lett. 2007, 253, 180–204. [Google Scholar] [CrossRef] [PubMed]
| Total (n = 88 *) | ||
|---|---|---|
| Male | 63 (72%) | |
| Race | Caucasian | 70 (80%) |
| African American | 5 (6%) | |
| Asian | 2 (2%) | |
| Hispanic | 3 (3%) | |
| Unknown | 8 (9%) | |
| Stage | 1 | 6 (7%) |
| 2 | 12 (14%) | |
| 3 | 12 (14%) | |
| 4 | 58 (65%) | |
| Chemotherapy | 71 (81%) | |
| Surgery | 74 (84%) | |
| Radiation | 30 (34%) |
| Stage | CTC < 5 | CTC ≥ 5 | CTC ≥ 5% |
|---|---|---|---|
| I (n = 6) | 5 | 1 | 17% |
| II (n = 12) | 6 | 6 | 50% |
| III (n = 12) | 1 | 11 | 92% |
| IV (n = 58) | 2 | 56 | 97% |
| Stage | iCTC Median | iCTC Mean | iCTC Standard Error |
|---|---|---|---|
| I (n = 6) | 0.0 | 8.3 | 8.3 |
| II (n = 12) | 13.0 | 35.8 | 13.5 |
| III (n = 12) | 41.0 | 65.9 | 23.1 |
| IV (n = 58) | 133.0 | 144.8 | 13.8 |
| Factor | Univariable Model | Multivariable Model | ||
|---|---|---|---|---|
| Hazard Ratio (95% CI) | p Value | Hazard Ratio (95% CI) | p Value | |
| iCTCs | 1.05 (1.03–1.07) | <0.0001 | 1.04 (1.01–1.06) | 0.009 |
| Stage | 1.89 (1.37–2.61) | 0.0001 | 1.66 (1.12–2.47) | 0.01 |
| Surgery | 0.17 (0.08–0.36) | <0.0001 | 0.37 (0.15–0.92) | 0.03 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.; Zarrabi, K.; Hou, W.; Madajewicz, S.; Choi, M.; Zucker, S.; Chen, W.-T. Assessing Clinical Outcomes in Colorectal Cancer with Assays for Invasive Circulating Tumor Cells. Biomedicines 2018, 6, 69. https://doi.org/10.3390/biomedicines6020069
Zhang Y, Zarrabi K, Hou W, Madajewicz S, Choi M, Zucker S, Chen W-T. Assessing Clinical Outcomes in Colorectal Cancer with Assays for Invasive Circulating Tumor Cells. Biomedicines. 2018; 6(2):69. https://doi.org/10.3390/biomedicines6020069
Chicago/Turabian StyleZhang, Yue, Kevin Zarrabi, Wei Hou, Stefan Madajewicz, Minsig Choi, Stanley Zucker, and Wen-Tien Chen. 2018. "Assessing Clinical Outcomes in Colorectal Cancer with Assays for Invasive Circulating Tumor Cells" Biomedicines 6, no. 2: 69. https://doi.org/10.3390/biomedicines6020069
APA StyleZhang, Y., Zarrabi, K., Hou, W., Madajewicz, S., Choi, M., Zucker, S., & Chen, W.-T. (2018). Assessing Clinical Outcomes in Colorectal Cancer with Assays for Invasive Circulating Tumor Cells. Biomedicines, 6(2), 69. https://doi.org/10.3390/biomedicines6020069





