Design and Fabrication of Flexible Copper Sensor Decorated with Bismuth Micro/Nanodentrites to Detect Lead and Cadmium in Noninvasive Samples of Sweat
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents and Solutions
2.2. Instruments
2.3. Preparation of Sweat Samples
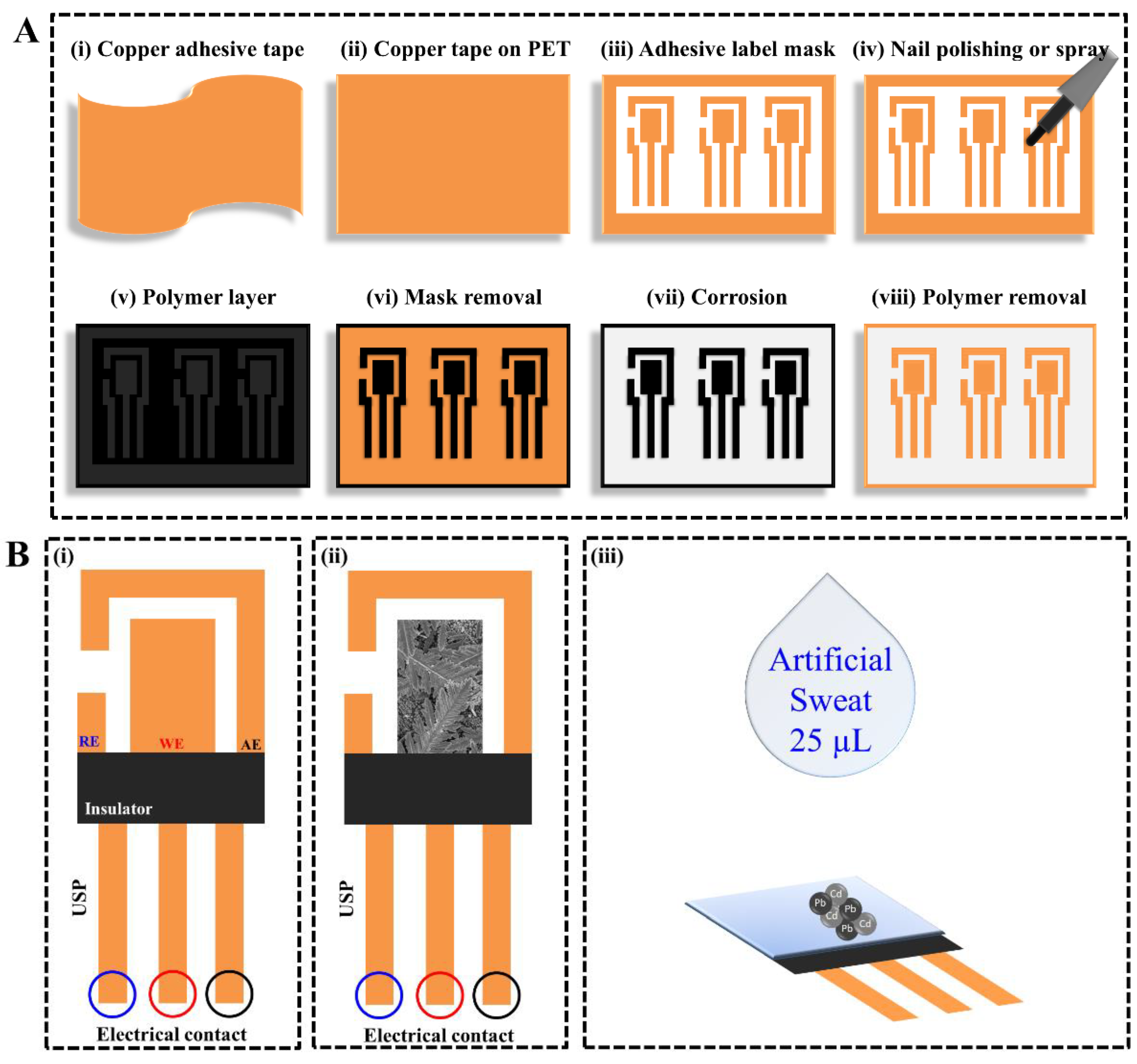
3. Results
3.1. Design and Fabrication of Flexible Copper Sensor Decorated with Bismuth Micro/Nanodentrites
3.2. Analytical Performance
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Anastasova, S.; Crewther, B.; Bembnowicz, P.; Curto, V.; Ip, H.M.; Rosa, B.; Yang, G.-Z. A wearable multisensing patch for continuous sweat monitoring. Biosens. Bioelectron. 2017, 93, 139–145. [Google Scholar] [CrossRef]
- Bennet, D.; Khorsandian, Y.; Pelusi, J.; Mirabella, A.; Pirrotte, P.; Zenhausern, F. Molecular and physical technologies for monitoring fluid and electrolyte imbalance: A focus on cancer population. Clin. Transl. Med. 2021, 11, e461. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Fang, Y.; Chen, J. Wearable Biosensors for Non-Invasive Sweat Diagnostics. Biosensors 2021, 11, 245. [Google Scholar] [CrossRef]
- Xuan, X.; Hui, X.; Yoon, H.; Yoon, S.; Park, J.Y. A rime ice-inspired bismuth-based flexible sensor for zinc ion detection in human perspiration. Mikrochim. Acta 2021, 188, 97. [Google Scholar] [CrossRef] [PubMed]
- Bandodkar, A.J.; Jia, W.; Wang, J. Tattoo-Based Wearable Electrochemical Devices: A Review. Electroanalysis 2015, 27, 562–572. [Google Scholar] [CrossRef]
- Qiao, L.; Benzigar, M.R.; Subramony, J.A.; Lovell, N.H.; Liu, G. Advances in Sweat Wearables: Sample Extraction, Real-Time Biosensing, and Flexible Platforms. ACS Appl. Mater. Interfaces 2020, 12, 34337–34361. [Google Scholar] [CrossRef]
- Ray, T.R.; Choi, J.; Bandodkar, A.J.; Krishnan, S.; Gutruf, P.; Tian, L.; Ghaffari, R.; Rogers, J.A. Bio-Integrated Wearable Systems: A Comprehensive Review. Chem. Rev. 2019, 119, 5461–5533. [Google Scholar] [CrossRef]
- Gao, W.; Nyein, H.Y.Y.; Shahpar, Z.; Fahad, H.M.; Chen, K.; Emaminejad, S.; Gao, Y.; Tai, L.-C.; Ota, H.; Wu, E.; et al. Wearable Microsensor Array for Multiplexed Heavy Metal Monitoring of Body Fluids. ACS Sens. 2016, 1, 866–874. [Google Scholar] [CrossRef]
- Ferreira-Ceccato, A.D.; Ramos, E.M.C.; de Carvalho, L.C.S.; Xavier, R.F.; Teixeira, M.F.D.S.; Raymundo-Pereira, P.A.; Proença, C.D.A.; de Toledo, A.C.; Ramos, D. Short terms effects of air pollution from biomass burning in mucociliary clearance of Brazilian sugarcane cutters. Respir. Med. 2011, 105, 1766–1768. [Google Scholar] [CrossRef]
- Landrigan, P.J.; Lucchini, R.; Kotelchuck, D.; Grandjean, P. Principles for Prevention of the Toxic Effects of Metals. In Handbook on the Toxicology of Metals, 4th ed.; Academic Press: Cambridge, MA, USA, 2015; pp. 507–528. [Google Scholar] [CrossRef]
- Wani, A.L.; Ara, A.; Usmani, J.A. Lead toxicity: A review. Interdiscip. Toxicol. 2015, 8, 55–64. [Google Scholar] [CrossRef]
- Munro, S.; Ebdon, L.; McWeeny, D.J. Application of inductively coupled plasma mass spectrometry (ICP-MS) for trace metal determination in foods. J. Anal. At. Spectrom. 1986, 1, 211–219. [Google Scholar] [CrossRef]
- Hohnadel, D.C.; Sunderman, F.W.; Nechay, M.W.; McNeely, M.D. Atomic Absorption Spectrometry of Nickel, Copper, Zinc, and Lead in Sweat Collected from Healthy Subjects during Sauna Bathing. Clin. Chem. 1973, 19, 1288–1292. [Google Scholar] [CrossRef]
- Montiel, V.R.-V.; Sempionatto, J.R.; Vargas, E.; Bailey, E.; May, J.; Bulbarello, A.; Düsterloh, A.; Matusheski, N.; Wang, J. Decentralized vitamin C & D dual biosensor chip: Toward personalized immune system support. Biosens. Bioelectron. 2021, 194, 113590. [Google Scholar] [CrossRef]
- March, G.; Nguyen, T.D.; Piro, B. Modified Electrodes Used for Electrochemical Detection of Metal Ions in Environmental Analysis. Biosensors 2015, 5, 241–275. [Google Scholar] [CrossRef] [PubMed]
- Koudelkova, Z.; Syrovy, T.; Ambrozova, P.; Moravec, Z.; Kubac, L.; Hynek, D.; Richtera, L.; Adam, V. Determination of Zinc, Cadmium, Lead, Copper and Silver Using a Carbon Paste Electrode and a Screen Printed Electrode Modified with Chromium(III) Oxide. Sensors 2017, 17, 1832. [Google Scholar] [CrossRef]
- Lee, S.; Park, S.-K.; Choi, E.; Piao, Y. Voltammetric determination of trace heavy metals using an electrochemically deposited graphene/bismuth nanocomposite film-modified glassy carbon electrode. J. Electroanal. Chem. 2016, 766, 120–127. [Google Scholar] [CrossRef]
- Hassan, K.M.; Gaber, S.E.; Altahan, M.F.; Azzem, M.A. Single and simultaneous voltammetric sensing of lead(II), cadmium(II) and zinc(II) using a bimetallic Hg-Bi supported on poly(1,2-diaminoanthraquinone)/glassy carbon modified electrode. Sens. Bio-Sensing Res. 2020, 29, 100369. [Google Scholar] [CrossRef]
- Peshoria, S.; Narula, A.K. Bare indium tin oxide electrode for electrochemical sensing of toxic metal ion. J. Mater. Sci. Mater. Electron. 2018, 29, 13858–13863. [Google Scholar] [CrossRef]
- Bohari, N.A.; Siddiquee, S.; Saallah, S.; Misson, M.; Arshad, S.E. Optimization and Analytical Behavior of Electrochemical Sensors Based on the Modification of Indium Tin Oxide (ITO) Using PANI/MWCNTs/AuNPs for Mercury Detection. Sensors 2020, 20, 6502. [Google Scholar] [CrossRef] [PubMed]
- Khoshroo, A.; Sadrjavadi, K.; Taran, M.; Fattahi, A. Electrochemical system designed on a copper tape platform as a nonenzymatic glucose sensor. Sensors Actuators B Chem. 2020, 325, 128778. [Google Scholar] [CrossRef]
- Soares, J.C.; Soares, A.C.; Rodrigues, V.C.; Oiticica, P.R.A.; Raymundo-Pereira, P.A.; Bott-Neto, J.L.; Buscaglia, L.A.; de Castro, L.D.C.; Ribas, L.C.; Scabini, L.; et al. Detection of a SARS-CoV-2 sequence with genosensors using data analysis based on information visualization and machine learning techniques. Mater. Chem. Front. 2021, 5, 5658–5670. [Google Scholar] [CrossRef]
- Raymundo-Pereira, P.A.; Shimizu, F.M.; Coelho, D.; Piazzeta, M.H.; Gobbi, A.L.; Machado, S.A.; Oliveira, O.N. A Nanostructured Bifunctional platform for Sensing of Glucose Biomarker in Artificial Saliva: Synergy in hybrid Pt/Au surfaces. Biosens. Bioelectron. 2016, 86, 369–376. [Google Scholar] [CrossRef]
- Nasraoui, S.; Ameur, S.; Al-Hamry, A.; Ben Ali, M.; Kanoun, O. Development of an Efficient Voltammetric Sensor for the Monitoring of 4-Aminophenol Based on Flexible Laser Induced Graphene Electrodes Modified with MWCNT-PANI. Sensors 2022, 22, 833. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.W.; White, I.M. Inkjet-printed paper-based SERS dipsticks and swabs for trace chemical detection. Analyst 2012, 138, 1020–1025. [Google Scholar] [CrossRef]
- Mannoor, M.S.; Tao, H.; Clayton, J.D.; Sengupta, A.; Kaplan, D.L.; Naik, R.R.; Verma, N.; Omenetto, F.G.; McAlpine, M. Graphene-based wireless bacteria detection on tooth enamel. Nat. Commun. 2012, 3, 763. [Google Scholar] [CrossRef]
- Scordo, G.; Moscone, D.; Palleschi, G.; Arduini, F. A reagent-free paper-based sensor embedded in a 3D printing device for cholinesterase activity measurement in serum. Sensors Actuators B Chem. 2018, 258, 1015–1021. [Google Scholar] [CrossRef]
- Honda, W.; Harada, S.; Arie, T.; Akita, S.; Takei, K. Wearable, Human-Interactive, Health-Monitoring, Wireless Devices Fabricated by Macroscale Printing Techniques. Adv. Funct. Mater. 2014, 24, 3299–3304. [Google Scholar] [CrossRef]
- Li, X.; Tian, J.; Nguyen, T.; Shen, W. Paper-Based Microfluidic Devices by Plasma Treatment. Anal. Chem. 2008, 80, 9131–9134. [Google Scholar] [CrossRef] [PubMed]
- Brazaca, L.C.; Imamura, A.H.; Gomes, N.O.; Almeida, M.B.; Scheidt, D.T.; Raymundo-Pereira, P.A.; Oliveira, O.N.; Janegitz, B.C.; Machado, S.A.S.; Carrilho, E. Electrochemical immunosensors using electrodeposited gold nanostructures for detecting the S proteins from SARS-CoV and SARS-CoV-2. Anal. Bioanal. Chem. 2022, 414, 5507–5517. [Google Scholar] [CrossRef] [PubMed]
- Wang, J. Stripping Analysis at Bismuth Electrodes: A Review. Electroanalysis 2005, 17, 1341–1346. [Google Scholar] [CrossRef]
- Mathew, M.; Ariza, E.; Rocha, L.; Fernandes, A.; Vaz, F. TiCxOy thin films for decorative applications: Tribocorrosion mechanisms and synergism. Tribol. Int. 2008, 41, 603–615. [Google Scholar] [CrossRef]
- de Figueiredo-Filho, L.C.; Baccarin, M.; Janegitz, B.C.; Fatibello-Filho, O. A disposable and inexpensive bismuth film minisensor for a voltammetric determination of diquat and paraquat pesticides in natural water samples. Sensors Actuators B Chem. 2017, 240, 749–756. [Google Scholar] [CrossRef]
- Salazar-Pérez, A.J.; Camacho-López, M.; Morales-Luckie, R.A.; Sánchez-Mendieta, V. Structural evolution of Bi2O3 prepared by thermal oxidation of bismuth nanoparticles. Soc. Mex. Cienc. Tecnol. Superf. Mater. 2005, 18, 4–8. [Google Scholar]
- Meng, L.; Xu, W.; Zhang, Q.; Yang, T.; Shi, S. Study of nanostructural bismuth oxide films prepared by radio frequency reactive magnetron sputtering. Appl. Surf. Sci. 2019, 472, 165–171. [Google Scholar] [CrossRef]
- Zhong, H.; Qiu, Y.; Zhang, T.; Li, X.; Zhang, H.; Chen, X. Bismuth nanodendrites as a high performance electrocatalyst for selective conversion of CO2to formate. J. Mater. Chem. A 2016, 4, 13746–13753. [Google Scholar] [CrossRef]
- Torma, F.; Kádár, M.; Tóth, K.; Tatár, E. Nafion®/2,2′-bipyridyl-modified bismuth film electrode for anodic stripping voltammetry. Anal. Chim. Acta 2008, 619, 173–182. [Google Scholar] [CrossRef]
- Chaiyo, S.; Apiluk, A.; Siangproh, W.; Chailapakul, O. High sensitivity and specificity simultaneous determination of lead, cadmium and copper using μPAD with dual electrochemical and colorimetric detection. Sensors Actuators B Chem. 2016, 233, 540–549. [Google Scholar] [CrossRef]
- Zhao, G.; Liu, G. Synthesis of a three-dimensional (BiO)2CO3@single-walled carbon nanotube nanocomposite and its application for ultrasensitive detection of trace Pb(II) and Cd(II) by incorporating Nafion. Sensors Actuators B Chem. 2019, 288, 71–79. [Google Scholar] [CrossRef]
- Legeai, S.; Vittori, O. A Cu/Nafion/Bi electrode for on-site monitoring of trace heavy metals in natural waters using anodic stripping voltammetry: An alternative to mercury-based electrodes. Anal. Chim. Acta 2006, 560, 184–190. [Google Scholar] [CrossRef]
- Figueiredo-Filho, L.C.S.; Janegitz, B.C.; Fatibelilo-Filho, O.; Marcolino-Junior, L.H.; Banks, C.E. Inexpensive and disposable copper mini-sensor modified with bismuth for lead and cadmium determination using square-wave anodic stripping voltammetry. Anal. Methods 2012, 5, 202–207. [Google Scholar] [CrossRef]
- Kadara, R.O.; Jenkinson, N.; Banks, C.E. Disposable Bismuth Oxide Screen Printed Electrodes for the High Throughput Screening of Heavy Metals. Electroanalysis 2009, 21, 2410–2414. [Google Scholar] [CrossRef]
- Peña, R.C.; Cornejo, L.; Bertotti, M.; Brett, C.M.A. Electrochemical determination of Cd(ii) and Pb(ii) in mining effluents using a bismuth-coated carbon fiber microelectrode. Anal. Methods 2018, 10, 3624–3630. [Google Scholar] [CrossRef]
- Rosolina, S.M.; Chambers, J.Q.; Lee, C.W.; Xue, Z.-L. Direct determination of cadmium and lead in pharmaceutical ingredients using anodic stripping voltammetry in aqueous and DMSO/water solutions. Anal. Chim. Acta 2015, 893, 25–33. [Google Scholar] [CrossRef]
- Riman, D.; Jirovsky, D.; Hrbac, J.; Prodromidis, M.I. Green and facile electrode modification by spark discharge: Bismuth oxide-screen printed electrodes for the screening of ultra-trace Cd(II) and Pb(II). Electrochem. Commun. 2015, 50, 20–23. [Google Scholar] [CrossRef]
- Niu, P.; Fernández-Sánchez, C.; Gich, M.; Navarro-Hernández, C.; Fanjul-Bolado, P.; Roig, A. Screen-printed electrodes made of a bismuth nanoparticle porous carbon nanocomposite applied to the determination of heavy metal ions. Mikrochim. Acta 2015, 183, 617–623. [Google Scholar] [CrossRef]
- Malakhova, N.A.; Mysik, A.A.; Saraeva, S.Y.; Stozhko, N.; Uimin, M.A.; Ermakov, A.E.; Brainina, K.Z. A voltammetric sensor on the basis of bismuth nanoparticles prepared by the method of gas condensation. J. Anal. Chem. 2010, 65, 640–647. [Google Scholar] [CrossRef]
- Promphet, N.; Rattanarat, P.; Rangkupan, R.; Chailapakul, O.; Rodthongkum, N. An electrochemical sensor based on graphene/polyaniline/polystyrene nanoporous fibers modified electrode for simultaneous determination of lead and cadmium. Sensors Actuators B Chem. 2015, 207, 526–534. [Google Scholar] [CrossRef]
- Wu, H.; Qiao, S.; Zhang, N.; Zhang, Y. Preparation of an Electrochemical Sensor for Rapid Detection of Lead(II) in Blueberries. Int. J. Electrochem. Sci. 2021, 16, 1–10. [Google Scholar] [CrossRef]




| Modification | Pb2+ | Cd2+ | Sample | Ref. | ||
|---|---|---|---|---|---|---|
| LOD(µM) | Linear Range (µM) | LOD(µM) | Linear Range (µM) | |||
| Bi/Nafion/Cu | 4.3 × 10−3 | 9.6 × 10−3–0.058 | 9.8 × 10−3 | 0.018–0.11 | Pb2+: ground water Cd2+: aquatic plant extracts | [40] |
| Bi/CuSPE | 0.83 | 1.3–13 | 0.53 | 1.0–12 | River water | [41] |
| Bismuth oxide SPE | 0.048 | 0.048–0.72 | 0.045 | 0.089–1.3 | River water | [42] |
| BiFME | 0.010 | 0.05–0.35 | 9.2 × 10−3 | 0.050–0.35 | Mine effluents | [43] |
| Bi/GCE | 9.2 × 10−3 | 0.024–0.72 | 0.028 | 0.044–1.3 | Representative pharmaceutical matrices | [44] |
| Bi2O3 SPCE | 9.6 × 10−4 | 2.4 × 10−3–0.058 | 1.8 × 10−3 | 4.4 × 10−3–0.11 | Drinking water | [45] |
| BiNP bulk-modified SPCPE | 0.019 | 8.9 × 10−3–0.44 | 0.019 | 4.8 × 10−3–0.24 | Urban wastewater | [46] |
| Binano/TCE | 2.6 × 10−3 | 0.048–0.24 | 3.6 × 10−3 | 0.089–0.44 | - | [47] |
| G/PANI/PS/SPCE | 0.016 | 0.048–2.4 | 0.039 | 0.089–4.4 | River water | [48] |
| NC/GCE | 1.0 × 10−3 | 0.010–4.0 | - | - | Blueberry extract | [49] |
| Binanodendrites/CuF | 0.76 | 2.0–50 | 5.36 | 2.0–50 | Artificial sweat | This work |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Campos, A.M.; Silva, R.R.; Calegaro, M.L.; Raymundo-Pereira, P.A. Design and Fabrication of Flexible Copper Sensor Decorated with Bismuth Micro/Nanodentrites to Detect Lead and Cadmium in Noninvasive Samples of Sweat. Chemosensors 2022, 10, 446. https://doi.org/10.3390/chemosensors10110446
de Campos AM, Silva RR, Calegaro ML, Raymundo-Pereira PA. Design and Fabrication of Flexible Copper Sensor Decorated with Bismuth Micro/Nanodentrites to Detect Lead and Cadmium in Noninvasive Samples of Sweat. Chemosensors. 2022; 10(11):446. https://doi.org/10.3390/chemosensors10110446
Chicago/Turabian Stylede Campos, Anderson M., Robson R. Silva, Marcelo L. Calegaro, and Paulo A. Raymundo-Pereira. 2022. "Design and Fabrication of Flexible Copper Sensor Decorated with Bismuth Micro/Nanodentrites to Detect Lead and Cadmium in Noninvasive Samples of Sweat" Chemosensors 10, no. 11: 446. https://doi.org/10.3390/chemosensors10110446
APA Stylede Campos, A. M., Silva, R. R., Calegaro, M. L., & Raymundo-Pereira, P. A. (2022). Design and Fabrication of Flexible Copper Sensor Decorated with Bismuth Micro/Nanodentrites to Detect Lead and Cadmium in Noninvasive Samples of Sweat. Chemosensors, 10(11), 446. https://doi.org/10.3390/chemosensors10110446








