Erlang-Distributed SEIR Epidemic Models with Cross-Diffusion
Abstract
1. Introduction
2. Model Formulation
2.1. Model Assumptions
- A given population is divided into four categories, which are functions of both time and space.
- −
- Susceptible (). Individuals capable of contracting the disease.
- −
- Exposed (). Infected individuals who are not yet infectious.
- −
- Infectious Symptomatic (). Individuals capable of transmitting the disease.
- −
- Recovered (). Individuals from the infectious pool who have recovered.
- The population size is constant throughout the spatial domain but not at individual points in space,
- The disease is not lethal, and birth and death rates are assumed to be equal to .
- The transmission parameter, , is defined as the average number of effective contacts with other individuals per infectious individual per unit time. An effective contact is an encounter in which the infection is transmitted, we assume this has a probability b. Assuming the contacts per unit time is given by k, the transmission parameter is given by
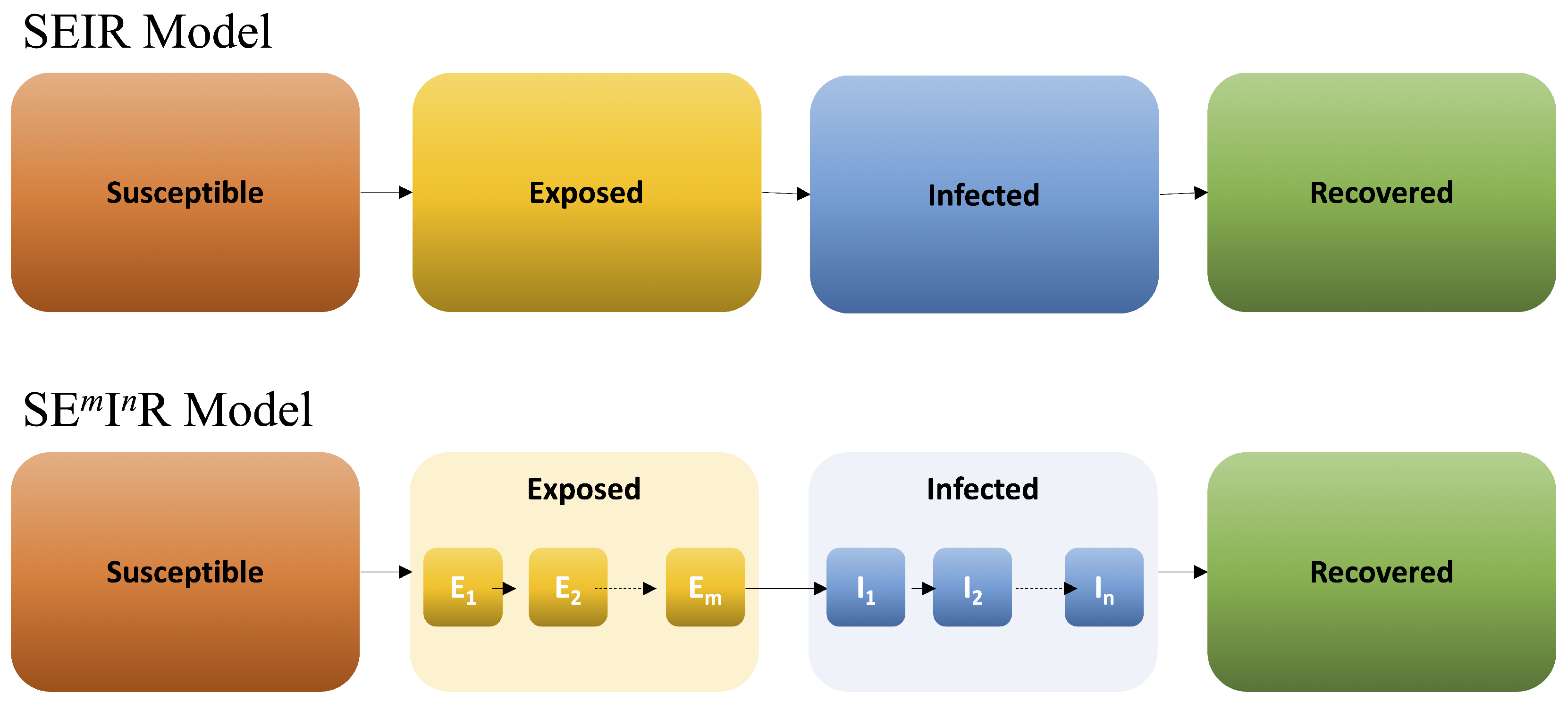
- The exposed class is divided into m subclasses and is the rate of sequential progression through the subclasses, where is the mean latent period. This is a proxy of modeling the latent period as a gamma distribution with shape parameter m and rate parameter , [26].
- The infectious class is divided into n subclasses and is the rate of sequential progression through the subclasses, where is the recovery rate so that is the mean infectious period. As before, this corresponds to a gamma distribution with shape parameter n and rate parameter , [26].
- Recovered individuals are permanently immune.
2.2. System of Equations
2.3. Spatial Structure
2.4. Equilibrium Points
3. Results and Discussion
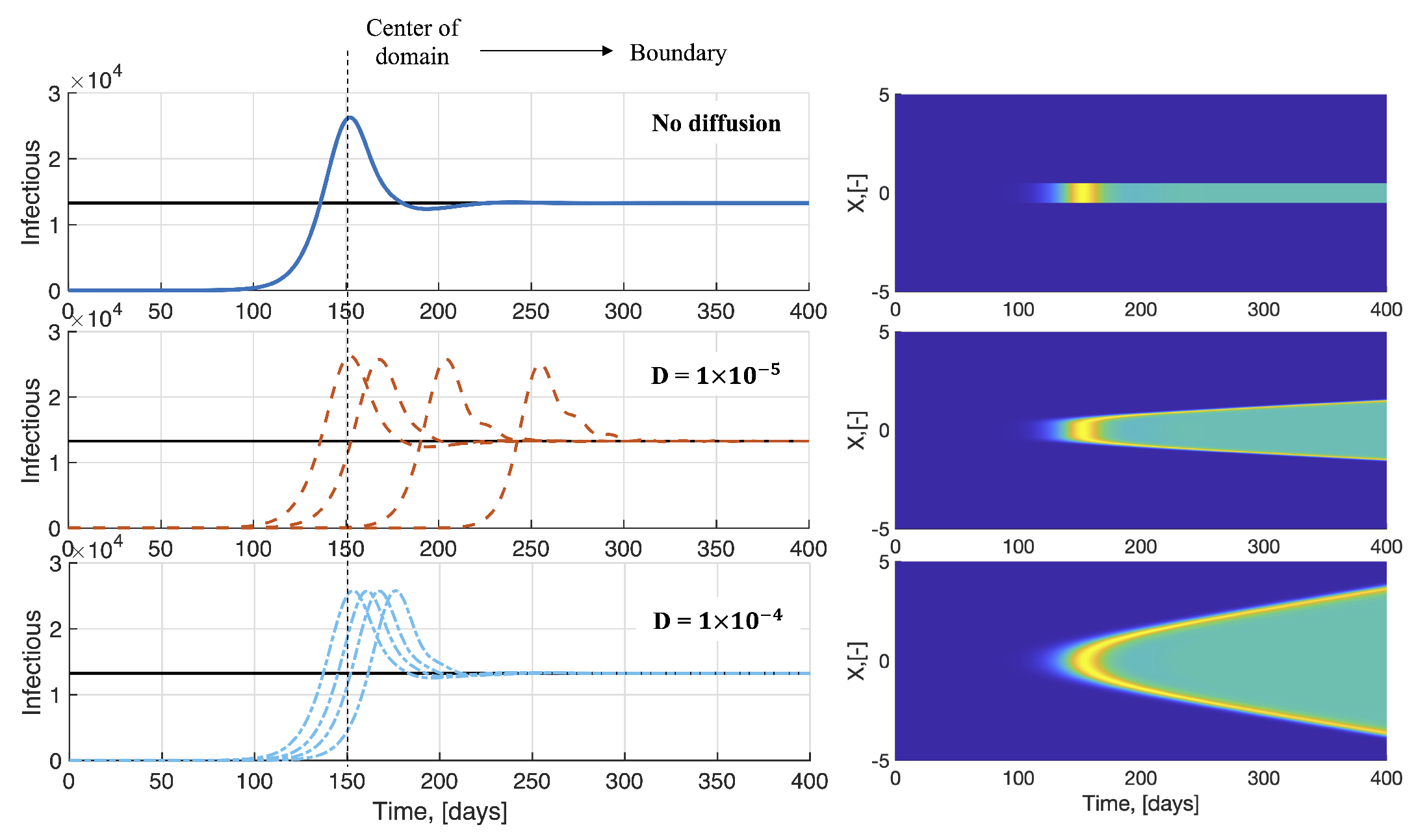
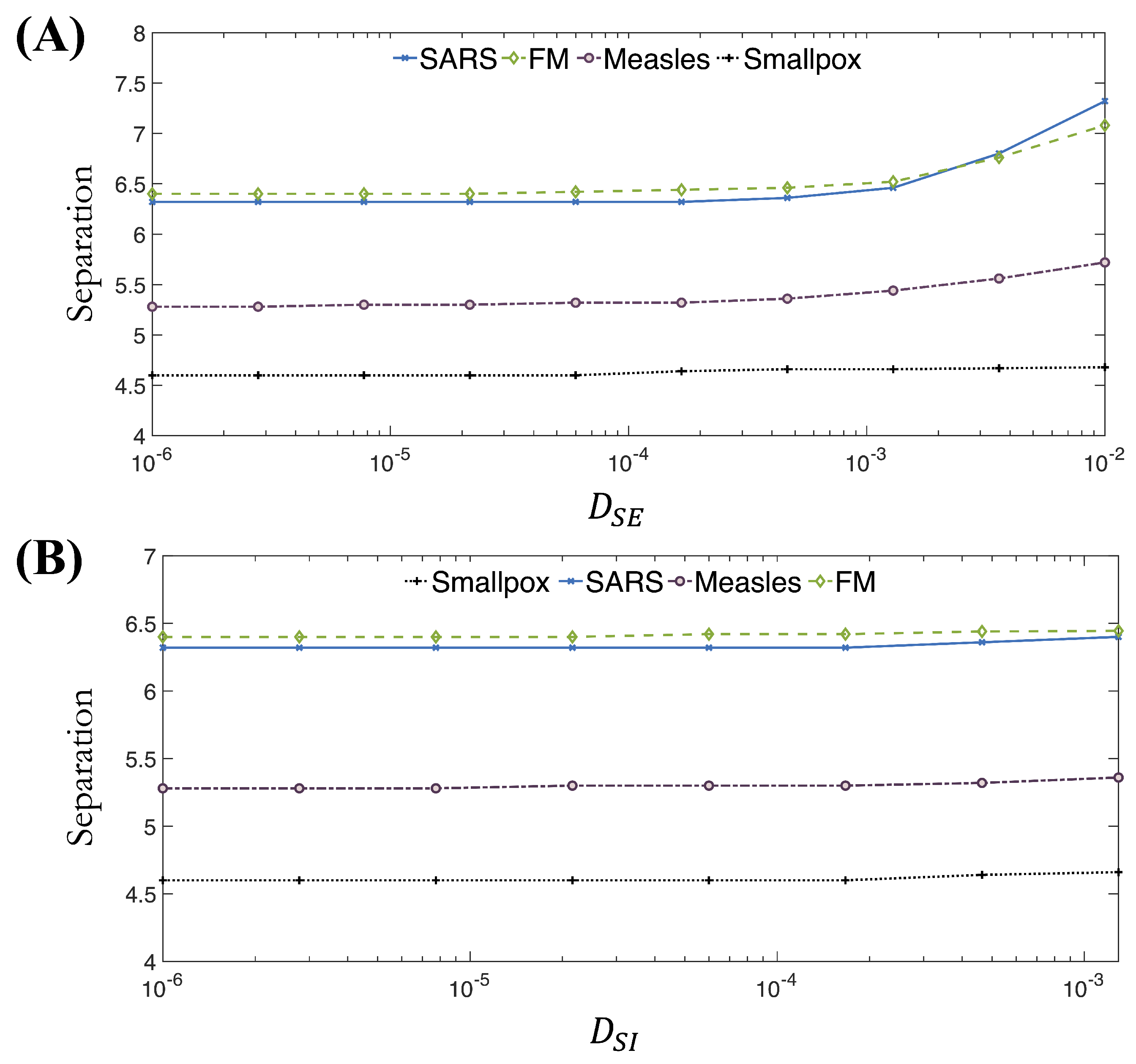
3.1. Effect of Varying the Transmission Parameter When Cross-Diffusion Is Included
3.2. Effect of Variations in the Average Latent Period When Cross-Diffusion Is Included
3.3. Effect of Variations in the Average Infectious Period When Cross-Diffusion Is Included
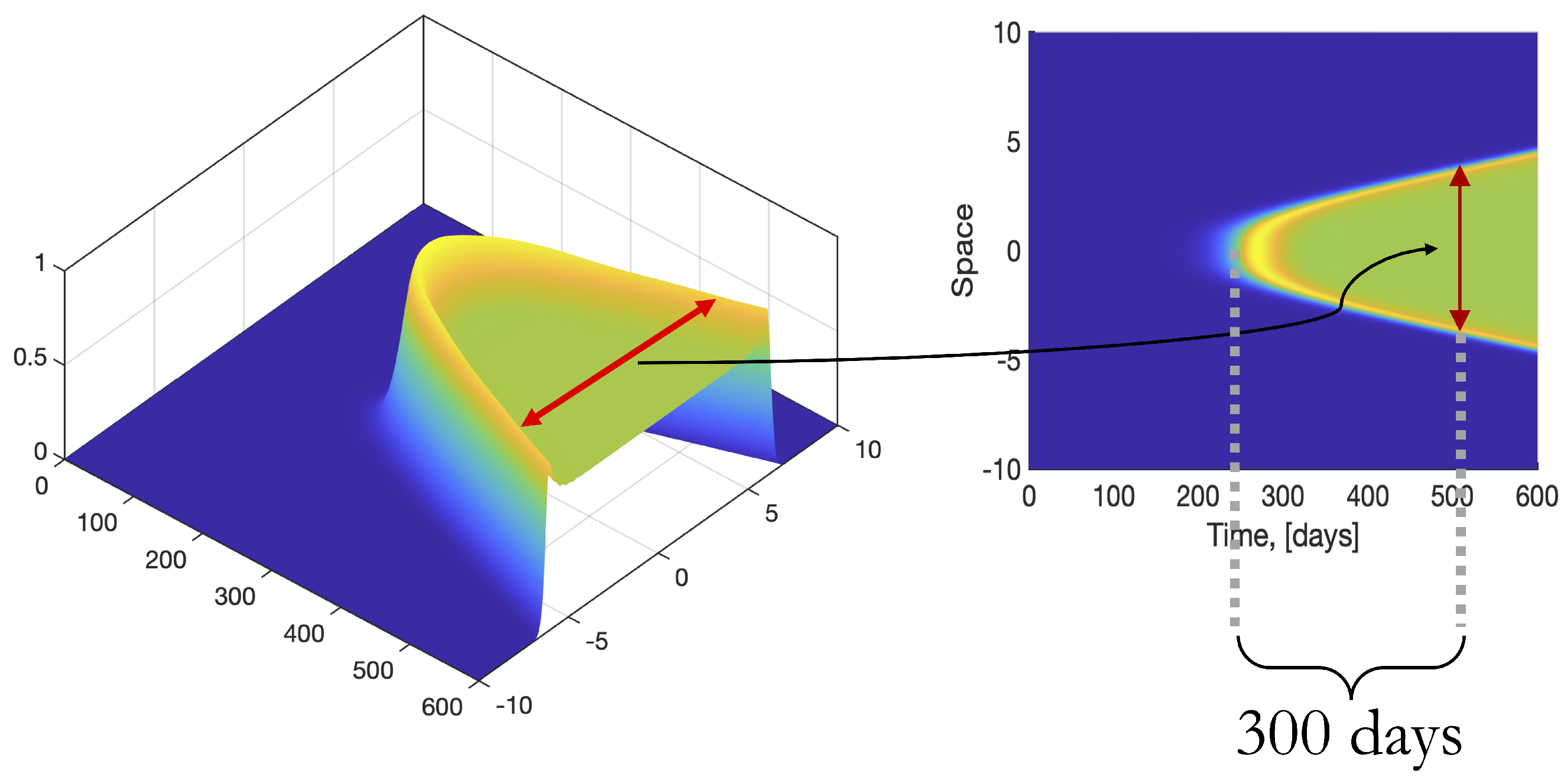
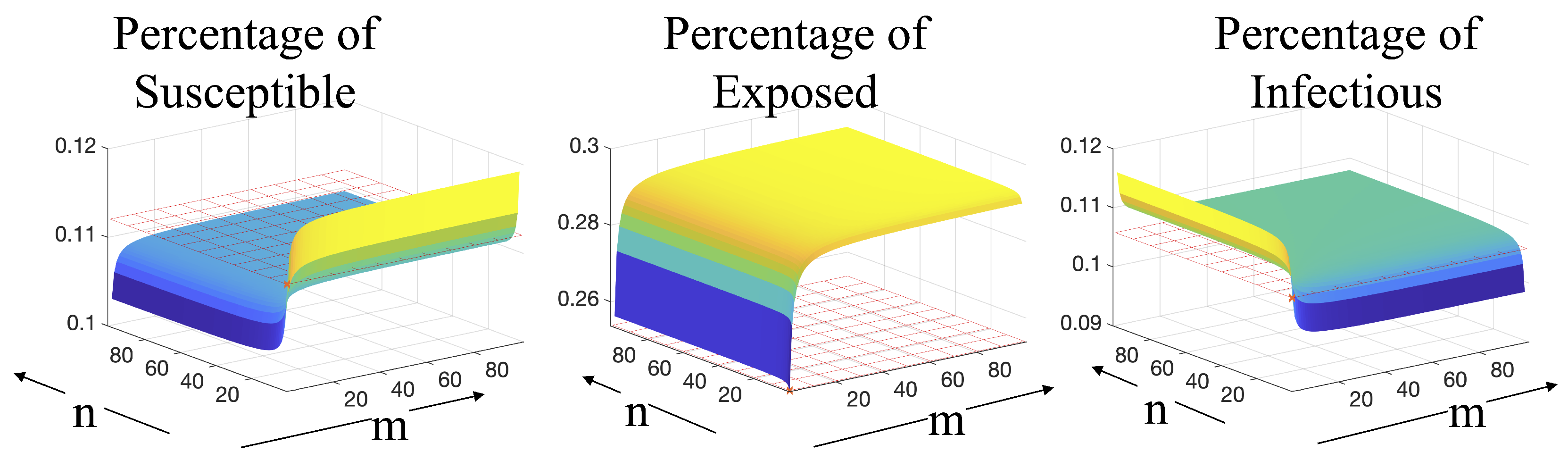
3.4. Effect of Varying Erlang Parameters When Cross-Diffusion Is Included
3.4.1. SARS
3.4.2. Measles
3.4.3. Smallpox
3.4.4. Foot-and-Mouth
3.5. Probability Distribution Functions
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Appendix A. Stability of Equilibrium Points
Appendix A.1. Local Stability
- The zero solution is globally asymptotically stable if for each non-negative integer n the eigenvalue of have negative real parts. Further, there exists positive constants K, ω such that for any ,
- The zero solution is stable if for each non-negative integer n the eigenvalues of have non-positive real parts and those with zero real parts have simple elementary divisors.
- The zero solution is unstable if for some n there exists an eigenvalue of with either positive real part or zero real part with a non-simple elementary divisor.

References
- Koplan, J.P.; Azizullah, M.F.S. Urban hospital and rural village smallpox in Bangladesh. Trop. Georg. Med. 1978, 30, 355–358. [Google Scholar]
- Daszak, P.; Cunningham, A.A.; Hyatt, A.D. Emerging Infectious Diseases of Wildlife—Threats to Biodiversity and Human Health. Science 2000, 287, 443–449. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, C.A.; Ghani, A.C.; Leung, G.M.; Hedley, A.J.; Fraser, C.; Riley, S.; Abu-Raddad, L.J.; Ho, L.M.; Thach, T.Q.; Chau, P.; et al. Epidemiological determinants of spread of causal agent of severe acute respiratory syndrome in Hong Kong. Lancet 2003, 361, 1761–1766. [Google Scholar] [CrossRef]
- Gani, R.L.S. Transmission potential of smallpox in contemporary populations. Nature 2001, 414, 748–751. [Google Scholar] [CrossRef] [PubMed]
- Holmes, E.A.; O’Connor, R.C.; Perry, V.H.; Tracey, I.; Wessely, S.; Arseneault, L.; Ballard, C.; Christensen, H.; Silver, R.C.; Everall, I.; et al. Multidisciplinary research priorities for the COVID-19 pandemic: A call for action for mental health science. Lancet Psychiatry 2020, 7, 547–560. [Google Scholar] [CrossRef] [PubMed]
- Nicola, M.; Alsafi, Z.; Sohrabi, C.; Kerwan, A.; Al-Jabir, A.; Iosifidis, C.; Agha, M.; Agha, R. The socio-economic implications of the coronavirus pandemic (COVID-19): A review. Int. J. Surg. 2020, 78, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Chinazzi, M.; Davis, J.T.; Ajelli, M.; Gioannini, C.; Litvinova, M.; Merler, S.; Pastore y Piontti, A.; Mu, K.; Rossi, L.; Sun, K.; et al. The effect of travel restrictions on the spread of the 2019 novel coronavirus (COVID-19) outbreak. Science 2020, 368, 395–400. [Google Scholar] [CrossRef]
- Kraemer, M.U.; Yang, C.H.; Gutierrez, B.; Wu, C.H.; Klein, B.; Pigott, D.M.; Open COVID-19 Data Working Group; Louis du Plessis 5; Du Plessis, L.; Faria, N.R.; et al. The effect of human mobility and control measures on the COVID-19 epidemic in China. Science 2020, 368, 493–497. [Google Scholar] [CrossRef]
- Keeling, M.J.; Rohani, P. Modeling Infectious Diseases in Humans and Animals; Princeton University Press: Princeton, NJ, USA, 2011. [Google Scholar]
- Keeling, M.J.; Grenfell, B.T. Disease extinction and community size: Modeling the persistence of measles. Science 1997, 275, 65–67. [Google Scholar] [CrossRef]
- Simpson, R.E.H. Infectiousness of communicable diseases in the household (measles, chickenpox, and mumps). Lancet 1952, 263, 549–554. [Google Scholar]
- Nauman, A.; Rafiq, M.; Rehman, M.; Ali, M.; Ahmad, M. Numerical modeling of SEIR measles dynamics with diffusion. Commun. Math. Appl. 2018, 9, 315. [Google Scholar]
- Al-Showaikh, F.N.M.; Twizell, T.E. One-dimensional measles dynamics. Appl. Math. Comput. 2004, 152, 169–194. [Google Scholar] [CrossRef]
- Ferguson, N.M.; Donnelly, C.A.; Anderson, R.M. The foot-and-mouth epidemic in Great Britain: Pattern of spread and impact of interventions. Science 2001, 292, 1155–1160. [Google Scholar] [CrossRef] [PubMed]
- Woolhouse, M.; Chase-Topping, M.; Haydon, D.; Friar, J.; Matthews, L.; Hughes, G.; Shaw, D.; Wilesmith, J.; Donaldson, A.; Cornell, S.; et al. Foot-and-mouth disease under control in the UK. Nature 2001, 411, 258–259. [Google Scholar] [CrossRef]
- Keeling, M.J.; Woolhouse, M.E.; Shaw, D.J.; Matthews, L.; Chase-Topping, M.; Haydon, D.T.; Cornell, S.J.; Kappey, J.; Wilesmith, J.; Grenfell, B.T. Dynamics of the 2001 UK foot and mouth epidemic: Stochastic dispersal in a heterogeneous landscape. Science 2001, 294, 813–817. [Google Scholar] [CrossRef]
- Ferguson, N.M.; Keeling, M.J.; John Edmunds, W.; Gani, R.; Grenfell, B.T.; Anderson, R.M.; Leach, S. Planning for smallpox outbreaks. Nature 2003, 425, 681–685. [Google Scholar] [CrossRef]
- Lipsitch, M.; Cohen, T.; Cooper, B.; Robins, J.M.; Ma, S.; James, L.; Gopalakrishna, G.; Chew, S.K.; Tan, C.C.; Samore, M.H.; et al. Transmission dynamics and control of severe acute respiratory syndrome. Science 2003, 300, 1966–1970. [Google Scholar] [CrossRef]
- Bakare, E.; Adekunle, Y.; Kadiri, K. Modelling and simulation of the dynamics of the transmission of measles. Int. J. Comput. Trends Technol. 2012, 3, 174–178. [Google Scholar]
- Kermack, W.O.; McKendrick, A.G. A contribution to the mathematical theory of epidemics. Proc. R. Soc. A 1927, 115, 700–721. [Google Scholar]
- Cooke, K.L. Stability analysis for a vector disease model. Rocky Mt. J. Math. 1979, 9, 31–42. [Google Scholar] [CrossRef]
- Hethcote, H.W.; Van den Driessche, P. Some epidemiological models with nonlinear incidence. J. Math. Biol. 1991, 29, 271–287. [Google Scholar] [CrossRef] [PubMed]
- Diekmann, O.; Heesterbeek, J.A.P. Mathematical Epidemiology of Infectious Diseases: Model Building, Analysis and Interpretation; John Wiley & Sons: Hoboken, NJ, USA, 2000; Volume 5. [Google Scholar]
- Bratus, S.A.; Novozhilov, A.S.; Platonov, A. Dynamical Systems and Models in Biology; Fizmatlit: Moscow, Russia, 2010. [Google Scholar]
- Hethcote, H.W. The mathematics of infectious diseases. SIAM Rev. 2000, 42, 599–653. [Google Scholar] [CrossRef]
- Wearing, H.J.; Rohani, P.; Keeling, M.J. Appropriate models for the management of infectious diseases. PLoS Med. 2005, 2, e174. [Google Scholar] [CrossRef] [PubMed]
- Blythe, S.; Anderson, R. Distributed incubation and infectious periods in models of the transmission dynamics of the human immunodeficiency virus (HIV). Math. Med. Biol. 1988, 5, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Hethcote, H.W.; Tudor, D.W. Integral equation models for endemic infectious diseases. J. Math. Biol. 1980, 9, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Krylova, O.; Earn, D.J. Effects of the infectious period distribution on predicted transitions in childhood disease dynamics. J. R. Soc. Interface 2013, 10, 20130098. [Google Scholar] [CrossRef]
- Lloyd, A.L. Realistic distributions of infectious periods in epidemic models: Changing patterns of persistence and dynamics. Theor. Popul. Biol. 2001, 60, 59–71. [Google Scholar] [CrossRef]
- Lloyd, A.L. Destabilization of epidemic models with the inclusion of realistic distributions of infectious periods. Proc. R. Soc. London Ser. B 2001, 268, 985–993. [Google Scholar] [CrossRef]
- Champredon, D.; Dushoff, J.; Earn, D.J. Equivalence of the Erlang-distributed SEIR epidemic model and the renewal equation. SIAM J. Appl. Math. 2018, 78, 3258–3278. [Google Scholar] [CrossRef]
- Zhu, H.; Li, Y.; Jin, X.; Huang, J.; Liu, X.; Qian, Y.; Tan, J. Transmission dynamics and control methodology of COVID-19: A modeling study. Appl. Math. Model. 2021, 89, 1983–1998. [Google Scholar] [CrossRef]
- Wayne, M.; Getz, E.R.D. Discrete stochastic analogs of Erlang epidemic models. J. Biol. Dyn. 2018, 12, 16–38. [Google Scholar]
- Leontitsis, A.; Senok, A.; Alsheikh-Ali, A.; Al Nasser, Y.; Loney, T.; Alshamsi, A. Seahir: A specialized compartmental model for COVID-19. Int. J. Environ. Res. Public Health 2021, 18, 2667. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, N.; Elsonbaty, A.; Raza, A.; Rafiq, M.; Adel, W. Numerical simulation and stability analysis of a novel reaction—Diffusion COVID-19 model. Nonlinear Dyn. 2021, 106, 1293–1310. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, N.; Tahira, S.; Rafiq, M.; Rehman, M.; Ali, M.; Ahmad, M. Positivity preserving operator splitting nonstandard finite difference methods for SEIR reaction diffusion model. Open Math. 2019, 17, 313–330. [Google Scholar] [CrossRef]
- Allen, L.J.; Bolker, B.M.; Lou, Y.; Nevai, A.L. Asymptotic profiles of the steady states for an SIS epidemic reaction-diffusion model. Discret. Contin. Dyn. Syst.-A 2008, 21, 1. [Google Scholar] [CrossRef]
- Bendahmane, M.; Langlais, M. A reaction-diffusion system with cross-diffusion modeling the spread of an epidemic disease. J. Evol. Eq. 2010, 10, 883–904. [Google Scholar] [CrossRef]
- Capasso, V.; Di Liddo, A. Asymptotic behaviour of reaction-diffusion systems in population and epidemic models. J. Math. Biol. 1994, 32, 453–463. [Google Scholar] [CrossRef]
- Capasso, V. Mathematical Structures of Epidemic Systems; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2008; Volume 97. [Google Scholar]
- Deng, K.; Wu, Y. Dynamics of a susceptible-infected-susceptible epidemic reaction-diffusion model. Proc. R. Soc. Edinb. Sect. A 2016, 146, 929. [Google Scholar] [CrossRef]
- Liu, P.; Li, H.X. Global behavior of a multi-group SEIR epidemic model with age structure and spatial diffusion. Math. Biosci. Eng. 2020, 17. [Google Scholar] [CrossRef]
- Milner, F.A.; Zhao, R. SIR model with directed spatial diffusion. Math. Popul. Stud. 2008, 15, 160–181. [Google Scholar] [CrossRef]
- Mulone, G.; Straughan, B.; Wang, W. Stability of epidemic models with evolution. Stud. Appl. Math. 2007, 118, 117–132. [Google Scholar] [CrossRef]
- Peng, R. Asymptotic profiles of the positive steady state for an SIS epidemic reaction–diffusion model. Part I. J. Differ. Equ. 2009, 247, 1096–1119. [Google Scholar] [CrossRef]
- Reluga, T. A two-phase epidemic driven by diffusion. J. Theor. Biol. 2004, 229, 249–261. [Google Scholar] [CrossRef]
- Wang, B.G.; Li, W.T.; Wang, Z.C. A reaction–diffusion SIS epidemic model in an almost periodic environment. Z. Angew. Math. Phys. 2015, 66, 3085–3108. [Google Scholar] [CrossRef]
- Wang, W.; Cai, Y.; Wu, M.; Wang, K.; Li, Z. Complex dynamics of a reaction–diffusion epidemic model. Nonlinear Anal. 2012, 13, 2240–2258. [Google Scholar] [CrossRef]
- Hu, Y.; Wang, J. Dynamics of an SIRS epidemic model with cross-diffusion. Commun. Pure Appl. Anakysis 2022, 21. [Google Scholar] [CrossRef]
- Cai, Y.; Chi, D.; Liu, W.; Wang, W. Stationary patterns of a cross-diffusion epidemic model. Abstr. Appl. Anal. 2013, 2013, 852698. [Google Scholar] [CrossRef]
- Riley, S.; Fraser, C.; Donnelly, C.A.; Ghani, A.C.; Abu-Raddad, L.J.; Hedley, A.J.; Leung, G.M.; Ho, L.M.; Lam, T.H.; Thach, T.Q.; et al. Transmission dynamics of the etiological agent of SARS in Hong Kong: Impact of public health interventions. Science 2003, 300, 1961–1966. [Google Scholar] [CrossRef]
- Li, M.; Wang, L. Global Stability in some SEIR epidemic models. In Mathematical Approaches for Emerging and Reemerging Infectious Diseases; Springer: Berlin/Heidelberg, Germany, 2002; Volume 126, pp. 295–311. [Google Scholar]
- Duan, X.; Yuan, S.; Qiu, Z.; Ma, J. Global stability of an SVEIR epidemic model with ages of vaccination and latency. Comput. Math. Appl. 2014, 68, 288–308. [Google Scholar] [CrossRef]
- Available online: https://www.cdc.gov/sars/ (accessed on 6 December 2017).
- Available online: https://www.cdc.gov/measles/ (accessed on 5 November 2020).
- Available online: https://www.cdc.gov/smallpox/ (accessed on 12 July 2017).
- Available online: https://www.cdc.gov/hand-foot-mouth/ (accessed on 2 February 2021).
- dos Santos, J.P.C.; Monteiro, E.; Ferreira, J.C.; Teixeira Lemes, N.H.; Rodrigues, D.S. Well Posedness and Qualitative Analysis of a SEIR Model with Spatial Diffusion for COVID-19 Spread. 2022. Available online: https://ssrn.com/abstract=4208624 (accessed on 3 May 2023).



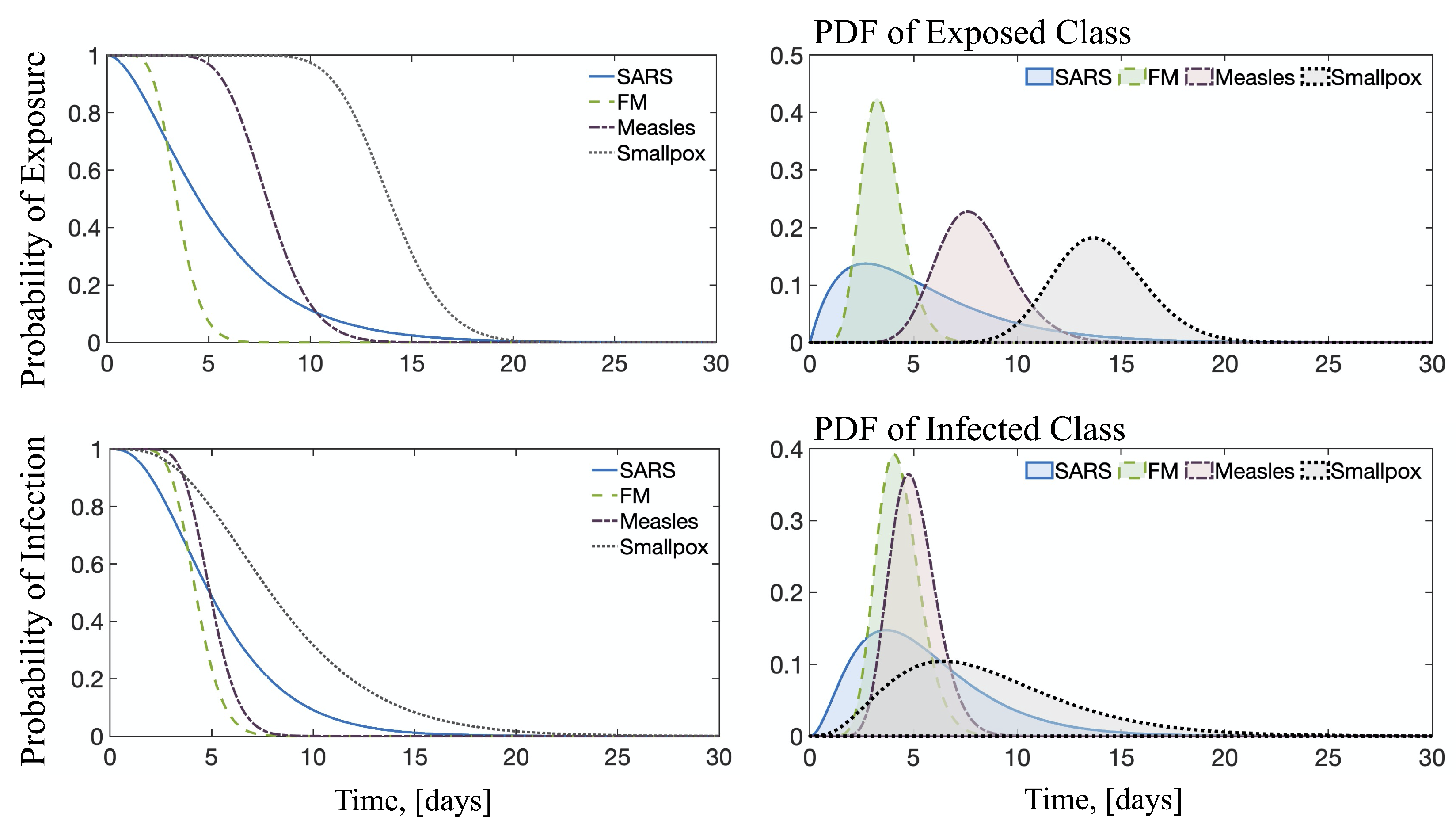
| Disease | m | n | Latent Period , (Days) | Infectious Period , (Days) | Reference | |
|---|---|---|---|---|---|---|
| Measles | 20 | 20 | 8 | 5 | 8.88 | [11] |
| Foot-and-Mouth | 13 | 17 | 3.5 | 4.3 | 9.7 | [14] |
| SARS | 2 | 3 | 5.36 | 5.5 | 10.79 | [52] |
| smallpox | 40 | 4 | 14 | 8.6 | 10.05 | [1] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chebotaeva, V.; Vasquez, P.A. Erlang-Distributed SEIR Epidemic Models with Cross-Diffusion. Mathematics 2023, 11, 2167. https://doi.org/10.3390/math11092167
Chebotaeva V, Vasquez PA. Erlang-Distributed SEIR Epidemic Models with Cross-Diffusion. Mathematics. 2023; 11(9):2167. https://doi.org/10.3390/math11092167
Chicago/Turabian StyleChebotaeva, Victoria, and Paula A. Vasquez. 2023. "Erlang-Distributed SEIR Epidemic Models with Cross-Diffusion" Mathematics 11, no. 9: 2167. https://doi.org/10.3390/math11092167
APA StyleChebotaeva, V., & Vasquez, P. A. (2023). Erlang-Distributed SEIR Epidemic Models with Cross-Diffusion. Mathematics, 11(9), 2167. https://doi.org/10.3390/math11092167






