Effects of High Voltage Electrical Discharge (HVED) on Endogenous Hormone and Polyphenol Profile in Wheat
Abstract
1. Introduction
2. Results
2.1. Effect of HVED Treatment on Germination and Growth
2.2. HVED Impact on Hormonal and Metabolite Profile
2.3. Comparative Statistical Analyses
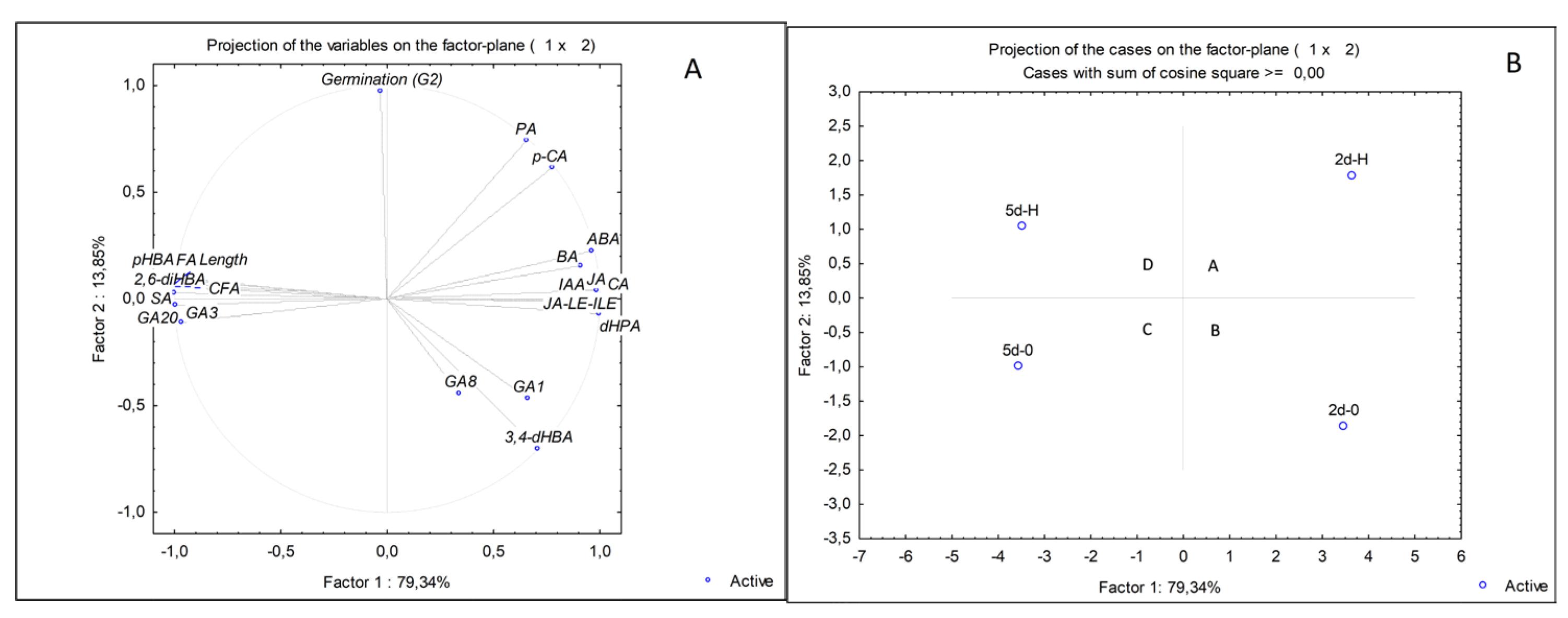
2.4. PCA Analyses
3. Discussion
3.1. HVED Impact on Germination and Growth
3.2. Root Hormonal and Polyphenol Response
3.3. Shoot Hormonal and Polyphenol Response
4. Materials and Methods
4.1. Plant Material and Treatment
4.2. Hormone and Metabolite Measurements
4.3. Data Analysis
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Pretty, J. Agricultural sustainability: Concepts, principles and evidence. Philos. Trans. R. Soc. B Biol. Sci. 2008, 363, 447–465. [Google Scholar] [CrossRef] [PubMed]
- Baby, S.M.; Narayanaswamy, G.K.; Anand, A. Superoxide radical production and performance index of Photosystem II in leaves from magnetoprimed soybean seeds. Plant Signal. Behav. 2011, 6, 1635–1637. [Google Scholar] [CrossRef] [PubMed]
- Maffei, M.E. Magnetic field effects on plant growth, development, and evolution. Front. Plant Sci. 2014, 5, 445. [Google Scholar] [CrossRef] [PubMed]
- Marcu, D.; Damian, G.; Cosma, C.; Cristea, V. Gamma radiation effects on seed germination, growth and pigment content, and ESR study of induced free radicals in maize (Zea mays L.). J. Biol. Phys. 2013, 39, 625–634. [Google Scholar] [CrossRef]
- Qi, W.; Zhang, L.; Wang, L.; Xu, H.; Jin, Q.; Jiao, Z. Pretreatment with low-dose gamma irradiation enhances tolerance to the stress of cadmium and lead in Arabidopsis thaliana seedlings. Ecotoxicol. Environ. Saf. 2015, 115, 243–249. [Google Scholar] [CrossRef]
- Feng, J.; Wang, D.; Shao, C.; Zhang, L.; Tang, X. Effects of cold plasma treatment on alfalfa seed growth under simulated drought stress. Plasma Sci. Technol. 2018, 20, 035505. [Google Scholar] [CrossRef]
- Sadhu, S.; Thirumdas, R.; Deshmukh, R.R.; Annapure, U.S. Influence of cold plasma on the enzymatic activity in germinating mung beans (Vigna radiate). LWT Food Sci. Technol. 2017, 78, 97–104. [Google Scholar] [CrossRef]
- Park, Y.; Oh, K.S.; Oh, J.; Seok, D.C.; Kim, S.B.; Yoo, S.J.; Lee, M.-J. The biological effects of surface dielectric barrier discharge on seed germination and plant growth with barley. Plasma Process. Polym. 2016, 15, e1600056. [Google Scholar] [CrossRef]
- Boussetta, N.; Turk, M.; De Taeye, C.; Larondelle, Y.; Lanoisellé, J.; Vorobiev, E. Effect of high voltage electrical discharges, heating and ethanol concentration on the extraction of total polyphenols and lignans from flaxseed cake. Ind. Crop. Prod. 2013, 49, 690–696. [Google Scholar] [CrossRef]
- Thirumdas, R.; Kothakota, A.; Annapure, U.; Siliveru, K.; Blundell, R.; Gatt, R.; Valdramidis, V.P. Plasma activated water (PAW): Chemistry, physico-chemical properties, applications in food and agriculture. Trends Food Sci. Technol. 2018, 77, 21–31. [Google Scholar] [CrossRef]
- Jirešová, J.; Scholtz, V.; Julák, J.; Šerá, B. Comparison of the Effect of Plasma-Activated Water and Artificially Prepared Plasma-Activated Water on Wheat Grain Properties. Plants 2022, 11, 1471. [Google Scholar] [CrossRef] [PubMed]
- Marček, T.; Kovač, T.; Jukić, K.; Lončarić, A.; Ižaković, M. Application of High Voltage Electrical Discharge Treatment to Improve Wheat Germination and Early Growth under Drought and Salinity Conditions. Plants 2021, 10, 2137. [Google Scholar] [CrossRef]
- Bormashenko, E.; Grynyov, R.; Bormashenko, Y.; Drori, E. Cold Radiofrequency Plasma Treatment Modifies Wettability and Germination Speed of Plant Seeds. Sci. Rep. 2012, 2, 741. [Google Scholar] [CrossRef] [PubMed]
- Filatova, I.; Filatova, I.; Azharonok, V.; Kadyrov, M.; Beljavsky, V.; Gvozdov, A.; Shik, A.; Antonuk, A. The Effect of Plasma Treatment of Seeds of Some Grain and Legumes on Their Sowing Quality and Productivity Enhancement. Rom. J. Phys. 2011, 56, 139–143. [Google Scholar]
- Rathjen, J.R.; Strounina, E.V.; Mares, D.J. Water movement into dormant and non-dormant wheat (Triticum aestivum L.) grains. J. Exp. Bot. 2009, 60, 1619–1631. [Google Scholar] [CrossRef]
- Marthandan, V.; Geetha, R.; Kumutha, K.; Renganathan, V.; Karthikeyan, A.; Ramalingam, J. Seed Priming: A Feasible Strategy to Enhance Drought Tolerance in Crop Plants. Int. J. Mol. Sci. 2020, 21, 8258. [Google Scholar] [CrossRef]
- Qi, P.-F.; Jiang, Y.-F.; Guo, Z.-R.; Chen, Q.; Ouellet, T.; Zong, L.-J.; Wei, Z.-Z.; Wang, Y.; Zhang, Y.-Z.; Xu, B.-J.; et al. Transcriptional reference map of hormone responses in wheat spikes. BMC Genom. 2019, 20, 390. [Google Scholar] [CrossRef]
- Ali, A.S.; Elozeiri, A.A. Metabolic Processes During Seed Germination. In Seed Biology, 6th ed.; Jimenez-Lopez, J.C., Ed.; InTech: Rang-Du-Fliers, France, 2017; pp. 141–166. [Google Scholar]
- Nonogaki, H. Repression of Transcription Factors by MicroRNA during Seed Germination and Postgermination. Plant Signal. Behav. 2008, 3, 65–67. [Google Scholar] [CrossRef]
- Soliman, A.S. Plant Growth Hormones. In Cell Growth, 1st ed.; Vikas, B., Fasullo, M., Eds.; IntechOpen: Rang-Du-Fliers, France, 2000. [Google Scholar] [CrossRef]
- Hedden, P. The Current Status of Research on Gibberellin Biosynthesis. Plant Cell Physiol. 2020, 61, 1832–1849. [Google Scholar] [CrossRef]
- Salazar-Cerezo, S.; Martínez-Montiel, N.; García-Sánchez, J.; Pérez-Y-Terrón, R.; Martínez-Contreras, R.D. Gibberellin biosynthesis and metabolism: A convergent route for plants, fungi and bacteria. Microbiol. Res. 2018, 208, 85–98. [Google Scholar] [CrossRef]
- Cui, D.; Yin, Y.; Li, H.; Hu, X.; Zhuang, J.; Ma, R.; Jiao, Z. Comparative transcriptome analysis of atmospheric pressure cold plasma enhanced early seedling growth in Arabidopsis thaliana. Plasma Sci. Technol. 2021, 23, 085502. [Google Scholar] [CrossRef]
- Zaharia, L.I.; Walker-Simmon, M.K.; Rodríguez, C.N.; Abrams, S.R. Chemistry of Abscisic Acid, Abscisic Acid Catabolites and Analogs. J. Plant Growth Regul. 2005, 24, 274–284. [Google Scholar] [CrossRef]
- Attri, P.; Ishikawa, K.; Okumura, T.; Koga, K.; Shiratani, M.; Mildaziene, V. Impact of seed color and storage time on the radish seed germination and sprout growth in plasma agriculture. Sci. Rep. 2021, 11, 2539. [Google Scholar] [CrossRef] [PubMed]
- Suriyasak, C.; Hatanaka, K.; Tanaka, H.; Okumura, T.; Yamashita, D.; Attri, P.; Koga, K.; Shiratani, M.; Hamaoka, N.; Ishibashi, Y. Alterations of DNA Methylation Caused by Cold Plasma Treatment Restore Delayed Germination of Heat-Stressed Rice (Oryza sativa L.) Seeds. ACS Agric. Sci. Technol. 2021, 1, 5–10. [Google Scholar] [CrossRef]
- Belin, C.; Megies, C.; Hauserová, E.; Lopez-Molina, L. Abscisic Acid Represses Growth of the Arabidopsis Embryonic Axis after Germination by Enhancing Auxin Signaling. Plant Cell 2009, 21, 2253–2268. [Google Scholar] [CrossRef]
- Liu, X.D.; Zhang, H.; Zhao, Y.; Feng, Z.Y.; Li, Q.; Yang, H.-Q.; Luan, S.; Li, J.M.; He, Z.-H. Auxin controls seed dormancy through stimulation of abscisic acid signaling by inducing ARF-mediated ABI3 activation in Arabidopsis. Proc. Natl. Acad. Sci. USA 2013, 110, 15485–15490. [Google Scholar] [CrossRef]
- Shu, K.; Liu, X.-D.; Xie, Q.; He, Z.-H. Two Faces of One Seed: Hormonal Regulation of Dormancy and Germination. Mol. Plant 2016, 9, 34–45. [Google Scholar] [CrossRef]
- Wang, L.; Hua, D.P.; He, J.; Duan, Y.; Chen, Z.Z.; Hong, X.H.; Gong, Z.Z. Auxin Response Factor2 (ARF2) and Its Regulated Homeodomain Gene HB33 Mediate Abscisic Acid Response in Arabidopsis. PLoS Genet. 2011, 7, e1002172. [Google Scholar] [CrossRef]
- Watanabe, S.; Ono, R.; Hayashi, N.; Shiratani, M.; Tashiro, K.; Kuhara, S.; Inoue, A.; Yasuda, K.; Hagiwara, H. Growth enhancement and gene expression of Arabidopsis thaliana irradiated with active oxygen species. Jpn. J. Appl. Phys. 2016, 55, 07LG10. [Google Scholar] [CrossRef]
- Takahashi, K.; Hayashi, K.-I.; Kinoshita, T. Auxin Activates the Plasma Membrane H+-ATPase by Phosphorylation during Hypocotyl Elongation in Arabidopsis. Plant Physiol. 2012, 159, 632–641. [Google Scholar] [CrossRef]
- Wasternack, C. Action of jasmonates in plant stress responses and development—Applied aspects. Biotechnol. Adv. 2014, 32, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Ruan, J.; Zhou, Y.; Zhou, M.; Yan, J.; Khurshid, M.; Weng, W.; Cheng, J.; Zhang, K. Jasmonic Acid Signaling Pathway in Plants. Int. J. Mol. Sci. 2019, 20, 2479. [Google Scholar] [CrossRef] [PubMed]
- Llanes, A.; Andrade, A.; Alemano, S.; Luna, V. Alterations of Endogenous Hormonal Levels in Plants under Drought and Salinity. Am. J. Plant Sci. 2016, 7, 1357–1371. [Google Scholar] [CrossRef]
- Wang, J.; Song, L.; Gong, X.; Xu, J.; Li, M. Functions of Jasmonic Acid in Plant Regulation and Response to Abiotic Stress. Int. J. Mol. Sci. 2020, 21, 1446. [Google Scholar] [CrossRef]
- Adhikari, B.; Adhikari, M.; Ghimire, B.; Park, G.; Choi, E.H. Cold Atmospheric Plasma-Activated Water Irrigation Induces Defense Hormone and Gene expression in Tomato seedlings. Sci. Rep. 2019, 9, 16080. [Google Scholar] [CrossRef] [PubMed]
- Koo, Y.M.; Heo, A.Y.; Choi, H.W. Salicylic Acid as a Safe Plant Protector and Growth Regulator. Plant Pathol. J. 2020, 36, 1. [Google Scholar] [CrossRef]
- Widhalm, J.R.; Dudareva, N. A Familiar Ring to It: Biosynthesis of Plant Benzoic Acids. Mol. Plant 2015, 8, 83–97. [Google Scholar] [CrossRef]
- Iriti, M.; Faoro, F. Bioactive Chemicals and Health Benefits of Grapevine Products. In Bioactive Foods in Promoting Health Fruits and Vegetables; Watson, R.R., Preedy, V.R., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2010; pp. 581–620. [Google Scholar] [CrossRef]
- Dong, N.Q.; Lin, H.X. Contribution of phenylpropanoid metabolism to plant development and plant–environment interactions. J. Integr. Plant Biol. 2021, 63, 180–209. [Google Scholar] [CrossRef]
- Jasim, S.F.; Al-Zubaidi, A.; Abdulbaqi, N.J. The Effect of Cold Plasma on Gene Expression of Major Genes in the Biosynthesis of Phenylpropanoids and Essential Oil Contents in Ocimum basilicum L. Ann. Rom. Soc. Cell Biol. 2021, 25, 14996–15010. [Google Scholar]
- Los, A.; Ziuzina, D.; Boehm, D.; Cullen, P.J.; Bourke, P. Investigation of mechanisms involved in germination enhancement of wheat (Triticum aestivum L.) by cold plasma: Effects on seed surface chemistry and characteristics. Plasma Process. Polym. 2019, 16, 1800148. [Google Scholar] [CrossRef]
- Meng, Y.R.; Qu, G.Z.; Wang, T.C.; Sun, Q.H.; Liang, D.L.; Hu, S.B. Enhancement of Germination and Seedling Growth of Wheat Seed Using Dielectric Barrier Discharge Plasma with Various Gas Sources. Plasma Chem. Plasma Process. 2017, 37, 1105–1119. [Google Scholar] [CrossRef]
- Molina, R.; López-Santos, C.; Gómez-Ramírez, A.; Vílchez, A.; Espinós, J.P.; González-Elipe, A.R. Influence of irrigation conditions in the germination of plasma treated Nasturtium seeds. Sci. Rep. 2018, 8, 16442. [Google Scholar] [CrossRef] [PubMed]
- Ling, L.; Jiangang, L.; Minchong, S.; Chunlei, Z.; Yuanhua, D. Cold plasma treatment enhances oilseed rape seed germination under drought stress. Sci. Rep. 2015, 5, 13033. [Google Scholar] [CrossRef]
- De Sousa Araújo, S.; Paparella, S.; Dondi, D.; Bentivoglio, A.; Carbonera, D.; Balestrazzi, A. Physical Methods for Seed Invigoration: Advantages and Challenges in Seed Technology. Front. Plant Sci. 2016, 7, 646. [Google Scholar] [CrossRef]
- Lukacova, Z.; Svubova, R.; Selvekova, P.; Hensel, K. The Effect of Plasma Activated Water on Maize (Zea mays L.) under Arsenic Stress. Plants 2021, 10, 1899. [Google Scholar] [CrossRef] [PubMed]
- Mildaziene, V.; Ivankov, A.; Sera, B.; Baniulis, D. Biochemical and Physiological Plant Processes Affected by Seed Treatment with Non-Thermal Plasma. Plants 2022, 11, 856. [Google Scholar] [CrossRef]
- Mildaziene, V.; Sera, B. Effects of Non-Thermal Plasma Treatment on Plant Physiological and Biochemical Processes. Plants 2022, 11, 1018. [Google Scholar] [CrossRef] [PubMed]
- Priatama, R.A.; Pervitasari, A.N.; Park, S.; Park, S.J.; Lee, Y.K. Current Advancements in the Molecular Mechanism of Plasma Treatment for Seed Germination and Plant Growth. Int. J. Mol. Sci. 2022, 23, 4609. [Google Scholar] [CrossRef]
- Ji, S.-H.; Choi, K.-H.; Pengkit, A.; Im, J.S.; Kim, J.S.; Kim, Y.H.; Park, Y.; Hong, E.J.; Jung, S.K.; Choi, E.-H.; et al. Effects of high voltage nanosecond pulsed plasma and micro DBD plasma on seed germination, growth development and physiological activities in spinach. Arch. Biochem. Biophys. 2016, 605, 117–128. [Google Scholar] [CrossRef]
- Alonso-Ramírez, A.; Rodríguez, D.; Reyes, D.; Jiménez, J.A.; Nicolás, G.; López-Climent, M.; Gómez-Cadenas, A.; Nicolás, C. Cross-Talk between Gibberellins and Salicylic Acid in Early Stress Responses in Arabidopsis thaliana Seeds. Plant Signal. Behav. 2009, 4, 750–751. [Google Scholar] [CrossRef]
- Pei, Z.-M.; Murata, Y.; Gregor, B.; Sebastien, T.; Birgit, K.; Allen, G.J.; Grill, E.; Julian, S. Calcium Channels Activated by Hydrogen Peroxide Mediate Abscisic Acid Signalling in Guard Cells. Nature 2000, 406, 731–734. [Google Scholar] [CrossRef] [PubMed]
- Brookbank, B.P.; Patel, J.; Gazzarrini, S.; Nambara, E. Role of Basal ABA in Plant Growth and Development. Genes 2021, 12, 1936. [Google Scholar] [CrossRef] [PubMed]
- Vishal, B.; Kumar, P.P. Regulation of Seed Germination and Abiotic Stresses by Gibberellins and Abscisic Acid. Front. Plant Sci. 2018, 9, 838. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, J.V.; Pearce, D.W.; Poole, A.T.; Pharis, R.P.; Mander, L.N. Abscisic acid, phaseic acid and gibberellin contents associated with dormancy and germination in barley. Physiol. Plant. 2002, 115, 428–441. [Google Scholar] [CrossRef]
- Miransari, M.; Smith, D.L. Plant hormones and seed germination. Environ. Exp. Bot. 2014, 99, 110–121. [Google Scholar] [CrossRef]
- Guo, Q.; Wang, Y.; Zhang, H.; Qu, G.; Wang, T.; Sun, Q.; Liang, D. Alleviation of adverse effects of drought stress on wheat seed germination using atmospheric dielectric barrier discharge plasma treatment. Sci. Rep. 2017, 7, 16680. [Google Scholar] [CrossRef]
- Du, L.; Yu, P. Effect of barley variety and growth year on ferulic and para-coumaric acids, and their ratio in the seed and hull. Cereal Res. Commun. 2010, 38, 521–532. [Google Scholar] [CrossRef]
- Cortese, E.; Settimi, A.G.; Pettenuzzo, S.; Cappellin, L.; Galenda, A.; Famengo, A.; Dabalà, M.; Antoni, V.; Navazio, L. Plasma-Activated Water Triggers Rapid and Sustained Cytosolic Ca2+ Elevations in Arabidopsis thaliana. Plants 2021, 10, 2516. [Google Scholar] [CrossRef]
- Riveras, E.; Alvarez, J.M.; Vidal, E.A.; Oses, C.; Vega, A.; Gutiérrez, R.A. The Calcium Ion Is a Second Messenger in the Nitrate Signaling Pathway of Arabidopsis. Plant Physiol. 2015, 169, 1397–1404. [Google Scholar] [CrossRef]
- Wang, X.; Feng, C.; Tian, L.; Hou, C.; Tian, W.; Hu, B.; Zhang, Q.; Ren, Z.; Niu, Q.; Song, J.; et al. A transceptor–channel complex couples nitrate sensing to calcium signaling in Arabidopsis. Mol. Plant 2021, 14, 774–786. [Google Scholar] [CrossRef]
- Yukiko, I.; Yasuo, K.; Minoru, T. Effects of Phenolic Compounds on Seed Germination of Shirakamba Birch, Betula platyphylla var. japonica. Eurasian J. For. Res. 2001, 2, 17–25. [Google Scholar]
- Marchiosi, R.; Dos Santos, W.D.; Constantin, R.P.; De Lima, R.B.; Soares, A.R.; Finger-Teixeira, A.; Mota, T.R.; de Oliveira, D.M.; Foletto-Felipe, M.D.P.; Abrahão, J.; et al. Biosynthesis and metabolic actions of simple phenolic acids in plants. Phytochem. Rev. 2020, 19, 865–906. [Google Scholar] [CrossRef]
- Chen, Z.; Zheng, Z.; Huang, J.; Lai, Z.; Fan, B. Biosynthesis of Salicylic Acid 494 Plant Signaling. Plant Signal. Behav. 2009, 4, 6. [Google Scholar] [CrossRef] [PubMed]
- Hasan, M.; Sohan, S.R.; Sajib, S.A.; Hossain, F.; Miah, M.; Maruf, M.H.; Khalid-Bin-Ferdaus, K.M.; Kabir, A.H.; Talukder, M.R.; Rashid, M.; et al. The Effect of Low-Pressure Dielectric Barrier Discharge (LPDBD) Plasma in Boosting Germination, Growth, and Nutritional Properties in Wheat. Plasma Chem. Plasma Process. 2021, 42, 339–362. [Google Scholar] [CrossRef]
- Berim, A.; Gang, D.R. Accumulation of Salicylic Acid and Related Metabolites in Selaginella moellendorffii. Plants 2022, 11, 461. [Google Scholar] [CrossRef]
- Lee, S.A.; Jang, S.; Yoon, E.K.; Heo, J.-O.; Chang, K.S.; Choi, J.W.; Dhar, S.; Kim, G.; Choe, J.-E.; Heo, J.B.; et al. Interplay between ABA and GA Modulates the Timing of Asymmetric Cell Divisions in the Arabidopsis Root Ground Tissue. Mol. Plant 2016, 9, 870–884. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Torres-Garcia, J.; Latrasse, D.; Benhamed, M.; Schilderink, S.; Zhou, W.; Kulikova, O.; Hirt, H.; Bisseling, T. Plant-Specific Histone Deacetylases HDT1/2 Regulate GIBBERELLIN 2-OXIDASE2 Expression to Control Arabidopsis Root Meristem Cell Number. Plant Cell 2017, 29, 2183–2196. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, Y.; Huffaker, A.; Bryan, A.C.; Tax, F.E.; Ryan, C.A. PEPR2 Is a Second Receptor for the Pep1 and Pep2 Peptides and Contributes to Defense Responses in Arabidopsis. Plant Cell 2010, 22, 508–522. [Google Scholar] [CrossRef]
- Avramova, Z. The jasmonic acid-signalling and abscisic acid-signalling pathways cross talk during one, but not repeated, dehydration stress: A non-specific ‘panicky’ or a meaningful response? Plant Cell Environ. 2017, 40, 1704–1710. [Google Scholar] [CrossRef]
- Moghanloo, M.; Iranbakhsh, A.; Ebadi, M.; Ardebili, Z.O. Differential physiology and expression of phenylalanine ammonia lyase (PAL) and universal stress protein (USP) in the endangered species Astragalus fridae following seed priming with cold plasma and manipulation of culture medium with silica nanoparticles. 3 Biotech 2019, 9, 288. [Google Scholar] [CrossRef]
- Sheteiwy, M.S.; An, J.; Yin, M.; Jia, X.; Guan, Y.; He, F.; Hu, J. Cold plasma treatment and exogenous salicylic acid priming enhances salinity tolerance of Oryza sativa seedlings. Protoplasma 2018, 256, 79–99. [Google Scholar] [CrossRef] [PubMed]
- Ji, S.H.; Yoo, S.; Choi, E.H.; Kim, S.B.; Oh, J. Biochemical and molecular characterization of enhanced growth of Panax ginseng C. A. Meyer treated with atmospheric pressure plasma. J. Phys. D Appl. Phys. 2020, 53, 494001. [Google Scholar] [CrossRef]
- Marcato, D.C.; Spagnol, C.M.; Salgado, H.R.N.; Isaac, V.L.B.; Corrêa, M.A. New and potential properties, characteristics, and analytical methods of ferulic acid: A review. Braz. J. Pharm. Sci. 2022, 58, e18747. [Google Scholar] [CrossRef]
- Gupta, N.; Prasad, V.B.R.; Chattopadhyay, S. LeMYC2 acts as a negative regulator of blue light mediated photomorphogenic growth, and promotes the growth of adult tomato plants. BMC Plant Biol. 2014, 14, 38. [Google Scholar] [CrossRef] [PubMed]
- Mewis, I.; Schreiner, M.; Nguyen, C.N.; Krumbein, A.; Ulrichs, C.; Lohse, M.; Zrenner, R. UV-B Irradiation Changes Specifically the Secondary Metabolite Profile in Broccoli Sprouts: Induced Signaling Overlaps with Defense Response to Biotic Stressors. Plant Cell Physiol. 2012, 53, 1546–1560. [Google Scholar] [CrossRef] [PubMed]
- Koo, A.J.; Gao, X.; Jones, A.D.; Howe, G.A. A rapid wound signal activates the systemic synthesis of bioactive jasmonates in Arabidopsis. Plant J. 2009, 59, 974–986. [Google Scholar] [CrossRef]
- Larrieu, A.; Vernoux, T. Q&A: How does jasmonate signaling enable plants to adapt and survive? BMC Biol. 2016, 14, 79. [Google Scholar] [CrossRef]
- Thabet, S.G.; Moursi, Y.S.; Karam, M.A.; Graner, A.; Alqudah, A.M. Genetic basis of drought tolerance during seed germination in barley. PLoS ONE 2018, 13, e0206682. [Google Scholar] [CrossRef]
- Pituch, K.A.; Stevens, J.P. Applied Multivariate Statistics for the Social Sciences, 6th ed.; Riegert, D., Ed.; Taylor and Francis: New York, NY, USA, 2016. [Google Scholar]





| Variable | Treatment | S2 | S3 | S4 | S5 | R2 | R3 | R4 | R5 | G1 | G2 | G3 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Treatment | ||||||||||||
| S2 | 0.91 *** | |||||||||||
| S3 | 0.90 *** | 0.82 *** | ||||||||||
| S4 | 0.89 *** | 0.86 *** | 0.82 *** | |||||||||
| S5 | 0.89 *** | 0.75 *** | 0.80 *** | 0.68 * | ||||||||
| R2 | 0.90 *** | 0.83 *** | 0.76 *** | 0.85 *** | 0.74 *** | |||||||
| R3 | 0.89 *** | 0.84 *** | 0.70 *** | 0.77 *** | 0.69 *** | 0.82 *** | ||||||
| R4 | 0.88 *** | 0.81 *** | 0.75 *** | 0.78 *** | 0.66 *** | 0.83 *** | 0.86 *** | |||||
| R5 | 0.77 *** | 0.78 *** | 0.59 * | 0.57 * | 0.68 *** | 0.75 *** | 0.78 *** | 0.74 *** | ||||
| G1 | 0.88 *** | 0.78 *** | 0.84 *** | 0.81 *** | 0.77 *** | 0.80 *** | 0.74 *** | 0.75 *** | 0.65 * | |||
| G2 | 0.81 *** | 0.70 *** | 0.78 *** | 0.78 *** | 0.62 * | 0.72 *** | 0.74 *** | 0.77 *** | 0.58 * | 0.85 *** | ||
| G3 | 0.80 *** | 0.70 *** | 0.79 *** | 0.77 *** | 0.64 * | 0.73 *** | 0.71 *** | 0.75 *** | 0.53 ** | 0.86 *** | 0.97 *** |
| 5 DAY | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Variable | Treatment | CA | IAA | BA | pHBA | SA | 3,4-dHBA | 2,6-dHBA | p-CA | CFA | FA | JA | ABA | PA | dHPA | JA-Le-Ile | GA 4 | GA 20 | GA 3 | GA 1 | GA 8 | G% | ShL | |
| 2 DAY | Treatment | −0.58 ns | 0.06 ns | −0.87 * | 0.28 | 0.17 ns | −0.73 * | −0.39 ns | 0.51 ns | 0.28 ns | 0.68 ns | 0.88 * | −0.06 ns | 0.51 ns | n.d. | 0.85 ** | n.d. | −0.28 ns | −0.51 ns | 0.88 * | −0.28 ns | 0.80 *** | 0.88 *** | |
| CA | 0.87 ** | −0.14 ns | 0.36 ns | 0.57 ns | 0.21 ns | 0.43 ns | −0.21 ns | 0.46 ns | 0.11 ns | −0.20 ns | −0.60 ns | −0.05 ns | −0.25 ns | −0.57 ns | 0.04 ns | 0.32 ns | −0.37 ns | 0.39 ns | −0.44 ns | −0.47 ns | ||||
| IAA | −0.25 ns | −0.27 ns | −0.61 ns | −0.33 ns | 0.02 ns | −0.19 ns | 0.21 ns | −0.12 ns | −0.26 ns | 0.71 ns | 0.89 * | 0.20 ns | 0.40 ns | −0.29 ns | −0.26 ns | −0.21 ns | −0.03 ns | 0.05 ns | −0.14 ns | 0.01 ns | ||||
| BA | 0.87 ** | 0.95 *** | 0.00 ns | −0.32 ns | −0.54 ns | 0.93 ** | 0.21 ns | −0.46 ns | −0.46 ns | −0.94 ** | −0.77 ns | 0.23 ns | −0.54 ns | −0.61 ns | 0.57 ns | 0.57 ns | −0.83 * | 0.43 ns | −0.65 ns | −0.67 ns | ||||
| pHBA | 0.14 ns | 0.10 ns | −0.14 ns | 0.04 ns | 0.45 ns | −0.45 ns | −0.45 ns | 0.74 * | 0.43 ns | 0.60 ns | −0.03 ns | −0.61 ns | 0.00 ns | 0.48 ns | −0.45 ns | −0.14 ns | 0.37 ns | −0.36 ns | 0.17 ns | 0.06 ns | ||||
| SA | 0.87 ** | 0.69 ns | −0.22 ns | 0.60 ns | 0.23 ns | −0.64 ns | 0.26 ns | 0.24 ns | 0.90 ** | 0.71 ns | −0.03 ns | −0.60 ns | −0.52 ns | −0.02 ns | −0.69 ns | −0.48 ns | 0.20 ns | −0.62 ns | 0.10 ns | 0.08 ns | ||||
| 3,4-dHBA | −0.03 ns | 0.22 ns | −0.70 * | 0.12 ns | 0.18 ns | −0.19 ns | −0.07 ns | −0.38 ns | −0.55 ns | −0.8 * | −0.71 ns | 0.42 ns | −0.02 ns | −0.55 ns | 0.71 * | 0.55 ns | −0.60 ns | 0.71 * | −0.45 ns | −0.50 ns | ||||
| 2,6-dHBA | 0.76 * | 0.67 ns | 0.02 ns | 0.66 ns | 0.05 ns | 0.71 ns | −0.31 ns | −0.76 * | 0.14 ns | −0.09 ns | −0.49 ns | −0.28 ns | −0.55 ns | −0.52 ns | −0.14 ns | 0.17 ns | −0.49 ns | −0.45 ns | −0.41 ns | −0.26 ns | ||||
| p-CA | 0.69 * | 0.70 * | 0.29 ns | 0.86 * | 0.50 ns | 0.71 ns | −0.10 ns | 0.52 ns | 0.31 ns | 0.43 ns | 0.26 ns | −0.01 ns | 0.36 ns | 0.45 ns | −0.10 ns | −0.12 ns | 0.49 ns | 0.17 ns | 0.49 ns | 0.44 ns | ||||
| CFA | 0.66 * | 0.64 * | −0.21 ns | 0.57 ns | 0.78 ** | 0.71 * | 0.13 ns | 0.60 ns | 0.70 * | 0.54 ns | −0.09 ns | −0.54 ns | −0.48 ns | 0.10 ns | −0.40 ns | −0.33 ns | 0.49 ns | −0.48 ns | 0.38 ns | 0.34 ns | ||||
| FA | 0.87 * | 0.89 * | −0.11 ns | 0.94 ** | 0.14 ns | 0.60 ns | 0.00 ns | 0.49 ns | 0.94 * | 0.57 ns | 0.60 ns | −0.54 ns | 0.54 ns | 0.54 ns | −0.89 * | −0.37 ns | 0.90 * | −0.66 ns | 0.62 ns | 0.60 ns | ||||
| JA | 0.87 * | 0.99 *** | −0.11 ns | 0.93 ** | 0.20 ns | 0.66 ns | 0.36 ns | 0.70 ns | 0.77 ns | 0.82 * | 0.90 * | 0.09 ns | 0.94 ** | 0.94 * | −0.60 ns | −0.77 ns | 0.70 ns | −0.26 ns | 0.62 ns | 0.54 ns | ||||
| ABA | 0.00 ns | 0.30 ns | −0.13 ns | 0.14 ns | 0.33 ns | 0.21 ns | 0.10 ns | 0.36 ns | 0.05 ns | 0.42 ns | 0.20 ns | 0.43 ns | 0.29 ns | −0.22 ns | 0.59 ns | 0.14 ns | −0.26 ns | 0.78 * | 0.00 ns | 0.09 ns | ||||
| PA | −0.24 ns | −0.08 ns | 0.25 ns | −0.02 ns | −0.04 ns | 0.05 ns | 0.14 ns | 0.05 ns | −0.05 ns | −0.02 ns | −0.36 ns | 0.14 ns | 0.20 ns | 0.43 ns | 0.24 ns | 0.05 ns | 0.71 ns | 0.38 ns | 0.49 ns | 0.50 ns | ||||
| dHPA | −0.35 ns | −0.43 ns | 0.37 ns | −0.39 ns | −0.17 ns | −0.32 ns | −0.65 ns | −0.12 ns | −0.10 ns | −0.30 ns | −0.14 ns | −0.54 ns | −0.33 ns | −0.62 ns | n.d. | n.d. | ||||||||
| JA-Le_Ile | 0.59 ns | 0.73 * | −0.35 ns | 0.71 * | −0.18 ns | 0.74 * | 0.03 ns | 0.86 ** | 0.37 ns | 0.28 ns | 0.54 ns | 0.82 * | 0.33 ns | −0.09 ns | −0.02 ns | −0.29 ns | −0.43 ns | 0.77 ns | −0.36 ns | 0.68 ns | 0.56 ns | |||
| GA 4 | 0.38 ns | 0.13 ns | −0.52 ns | −0.07 ns | −0.35 ns | 0.17 ns | −0.01 ns | 0.19 ns | −0.35 ns | −0.09 ns | −0.07 ns | −0.18 ns | −0.33 ns | −0.55 ns | 0.17 ns | 0.19 ns | n.d. | |||||||
| GA 20 | −0.48 ns | −0.47 ns | 0.46 ns | −0.71 ns | −0.35 ns | −0.36 ns | −0.38 ns | −0.02 ns | −0.22 ns | −0.48 ns | −0.43 ns | −0.71 ns | −0.54 ns | 0.36 ns | 0.34 ns | −0.06 ns | −0.21 ns | 0.76 * | 0.03 ns | 0.81 * | 0.10 ns | 0.14 ns | ||
| GA 3 | 0.10 ns | 0.02 ns | −0.29 ns | −0.05 ns | 0.35 ns | −0.02 ns | −0.04 ns | 0.29 ns | 0.13 ns | 0.16 ns | 0.32 ns | −0.07 ns | 0.30 ns | −0.42 ns | 0.10 ns | 0.18 ns | −0.02 ns | −0.13 ns | −0.37 ns | 0.48 ns | −0.22 ns | −0.12 ns | ||
| GA 1 | 0.30 ns | 0.37 ns | −0.40 ns | 0.67 ns | 0.22 ns | −0.45 ns | 0.89 ** | −0.30 ns | 0.37 ns | 0.11 ns | 0.67 ns | 0.63 ns | −0.41 ns | −0.19 ns | −0.48 ns | −0.37 ns | 0.07 ns | −0.39 ns | −0.11 ns | −0.09 ns | 0.93 ** | 0.84 * | ||
| GA 8 | 0.24 ns | 0.33 ns | −0.13 ns | 0.17 ns | 0.81 ** | 0.02 ns | 0.35 ns | 0.43 ns | 0.60 ns | 0.76 * | 0.32 ns | 0.43 ns | 0.38 ns | 0.10 ns | −0.45 ns | −0.08 ns | −0.38 ns | −0.34 ns | 0.38 ns | 0.48 ns | −0.01 ns | 0.01 ns | ||
| G% | 0.81 *** | 0.71 * | −0.17 ns | 0.75 * | 0.20 ns | 0.82 * | −0.08 ns | 0.53 ns | 0.70 * | 0.61 ns | 0.73 ns | 0.65 ns | −0.14 ns | −0.23 ns | −0.34 ns | 0.41 ns | 0.32 ns | −0.64 ns | −0.06 ns | 0.32 ns | 0.09 ns | 0.77 ** | ||
| ShL | 0.92 *** | 0.85 ** | −0.15 ns | 0.91 ** | 0.32 ns | 0.70 ns | 0.01 ns | 0.60 ns | 0.83 ** | 0.69 * | 0.95 *** | 0.92 ** | 0.19 ns | −0.34 ns | −0.27 ns | 0.47 ns | 0.13 ns | −0.65 ns | 0.24 ns | 0.39 ns | 0.40 ns | 0.83 *** | ||
 | ||||||||||||||||||||||||
| 5 DAY | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Variable | Treatment | CA | IAA | BA | pHBA | SA | 3,4-dHBA | 2,6-dHBA | p-CA | CFA | FA | JA | ABA | PA | dHPA | JA-Le_Ile | GA 1 | GA 8 | G% | RhL | |
| 2 DAY | Treatment | 0.87 ** | 0.52 ns | 0.87 ** | 0.52 ns | 0.69 * | 0.43 ns | 0.78 * | 0.61 | −0.17 ns | 0.22 ns | 0.65 ns | −0.26 ns | n.d. | −0.90 *** | 0.43 ns | 0.87 * | n.d. | 0.81 ** | 0.18 ns | |
| CA | 0.10 ns | 0.41 ns | 0.78 * | 0.53 ns | 0.68 * | 0.30 ns | 0.64 ns | 0.72 * | −0.04 ns | 0.36 ns | 0.63 ns | −0.46 ns | −0.89 ** | 0.09 ns | 0.56 ns | 0.62 ns | −0.08 ns | ||||
| IAA | 0.09 ns | −0.32 ns | 0.60 ns | 0.07 ns | 0.47 ns | 0.90 ** | 0.48 ns | 0.22 ns | −0.12 ns | 0.29 ns | 0.33 ns | −0.30 ns | −0.52 ns | 0.35 ns | 0.25 ns | 0.47 ns | 0.62 ns | ||||
| BA | 0.58 ns | 0.40 ns | 0.50 ns | 0.62 ns | 0.74 * | 0.36 ns | 0.76 * | 0.60 ns | −0.24 ns | 0.25 ns | 0.11 ns | 0.12 ns | −0.81 * | 0.31 ns | 0.75 ns | 0.69 ns | 0.00 ns | ||||
| pHBA | 0.87 ** | 0.26 ns | −0.22 ns | 0.36 ns | 0.30 ns | −0.14 ns | 0.27 ns | 0.83 ** | 0.18 ns | 0.57 ns | 0.24 ns | −0.28 ns | −0.30 ns | −0.08 ns | 0.71 ns | 0.50 ns | −0.52 ns | ||||
| SA | 0.17 ns | −0.67 ns | 0.11 ns | 0.58 ns | 0.20 ns | 0.55 ns | 0.83 ** | 0.50 ns | 0.00 ns | 0.40 ns | 0.26 ns | 0.03 ns | −0.64 ns | 0.23 ns | 0.21 ns | 0.45 ns | 0.28 ns | ||||
| 3,4-dHBA | −0.88 ** | −0.41 ns | −0.01 ns | −0.31 ns | −0.78 * | 0.04 ns | 0.54 ns | 0.08 ns | −0.13 ns | 0.14 ns | 0.40 ns | −0.28 ns | −0.40 ns | 0.44 ns | 0.14 ns | 0.41 ns | 0.81 ** | ||||
| 2,6-dHBA | 0.26 ns | 0.03 ns | 0.42 ns | 0.64 ns | 0.08 ns | 0.30 ns | −0.20 ns | 0.47 ns | −0.28 ns | 0.14 ns | 0.38 ns | 0.02 ns | −0.71 * | 0.58 ns | 0.64 ns | 0.57 ns | 0.44 ns | ||||
| p-CA | 0.49 ns | 0.40 ns | 0.77 ns | 0.54 ns | 0.31 ns | −0.40 ns | −0.70 ns | 0.31 ns | 0.42 ns | 0.38 ns | 0.55 ns | −0.52 ns | −0.41 ns | 0.15 ns | 0.54 ns | 0.47 ns | −0.22 ns | ||||
| CFA | 0.35 ns | 0.88 * | −0.43 ns | −0.05 ns | 0.54 ns | −0.46 ns | −0.59 ns | −0.11 ns | 0.37 ns | −0.33 ns | 0.14 ns | −0.30 ns | 0.37 ns | 0.02 ns | −0.29 ns | −0.10 ns | −0.21 ns | ||||
| FA | 0.88 * | −0.30 ns | 0.83 * | 0.49 ns | 0.66 ns | 0.20 ns | −0.60 ns | 0.60 ns | 0.8 * | 0.06 ns | −0.11 ns | −0.05 ns | −0.43 ns | −0.62 ns | −0.25 ns | 0.01 ns | −0.23 ns | ||||
| JA | 0.00 ns | 0.20 ns | −0.86 * | −0.43 ns | 0.21 ns | 0.43 ns | −0.31 ns | −0.36 ns | −0.89 * | 0.36 ns | −0.90 * | −0.83 * | −0.46 ns | 0.48 ns | 0.37 ns | 0.70 ns | 0.06 ns | ||||
| ABA | 0.88 * | 0.50 ns | 0.09 ns | 0.70 ns | 0.77 ns | 0.10 ns | −0.80 ns | 0.71 ns | 0.20 ns | 0.37 ns | 0.8 * | 0.14 ns | 0.18 ns | −0.05 ns | −0.07 ns | −0.28 ns | −0.05 ns | ||||
| PA | 0.87 ** | 0.26 ns | 0.02 ns | 0.32 ns | 0.72 * | 0.30 ns | −0.92 ** | 0.28 ns | 0.31 ns | 0.47 ns | 0.83 * | 0.36 ns | 0.83 * | n.d. | n.d. | ||||||
| dHPA | −0.49 ns | 0.40 ns | −0.89 * | −0.60 ns | −0.20 ns | 0.20 ns | 0.52 ns | −0.37 ns | −0.90 * | 0.20 ns | −0.98 ** | 0.90 * | −0.20 ns | −0.37 ns | −0.22 ns | −0.79 * | −0.68 * | −0.08 ns | |||
| JA-LE_Ile | −0.22 ns | −0.26 ns | 0.12 ns | 0.22 ns | −0.25 ns | 0.61 ns | 0.57 ns | 0.51 ns | −0.55 ns | −0.47 ns | 0.35 | 0.05 ns | 0.03 ns | −0.04 ns | 0.38 ns | 0.68 ns | 0.53 ns | 0.56 ns | |||
| GA 1 | −0.88 * | 0.50 ns | −0.60 ns | −0.20 ns | −0.66 ns | 0.00 ns | 0.60 ns | −0.60 ns | −0.8 * | −0.06 ns | −0.83 * | 0.90 * | −0.60 ns | −0.66 ns | 0.99 *** | 0.03 ns | 0.89** | −0.09 ns | |||
| GA 8 | 0.26 ns | 0.32 ns | −0.56 ns | −0.07 ns | 0.38 ns | 0.31 ns | −0.36 ns | 0.27 ns | −0.60 ns | 0.45 ns | −0.14 ns | 0.86 * | 0.31 ns | 0.57 ns | 0.70 ns | 0.36 ns | 0.20 ns | n.d. | |||
| G% | 0.8 ** | 0.31 ns | −0.01 ns | 0.64 ns | 0.78 * | −0.01 ns | −0.74 * | 0.42 ns | 0.27 ns | 0.33 ns | 0.74 ns | −0.04 ns | 0.99 *** | 0.71 * | −0.21 ns | −0.11 ns | −0.74 ns | 0.25 ns | 0.30 ns | ||
| RhL | 0.90 *** | 0.19 ns | −0.20 ns | 0.16 ns | 0.82 ** | −0.16 ns | −0.85 ** | −0.04 ns | 0.38 ns | 0.57 ns | 0.79 * | 0.24 ns | 0.79 * | 0.85 ** | −0.26 ns | −0.45 ns | −0.88 * | 0.33 ns | 0.69 * | ||
 | |||||||||||||||||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marček, T.; Hamow, K.Á.; Janda, T.; Darko, E. Effects of High Voltage Electrical Discharge (HVED) on Endogenous Hormone and Polyphenol Profile in Wheat. Plants 2023, 12, 1235. https://doi.org/10.3390/plants12061235
Marček T, Hamow KÁ, Janda T, Darko E. Effects of High Voltage Electrical Discharge (HVED) on Endogenous Hormone and Polyphenol Profile in Wheat. Plants. 2023; 12(6):1235. https://doi.org/10.3390/plants12061235
Chicago/Turabian StyleMarček, Tihana, Kamirán Áron Hamow, Tibor Janda, and Eva Darko. 2023. "Effects of High Voltage Electrical Discharge (HVED) on Endogenous Hormone and Polyphenol Profile in Wheat" Plants 12, no. 6: 1235. https://doi.org/10.3390/plants12061235
APA StyleMarček, T., Hamow, K. Á., Janda, T., & Darko, E. (2023). Effects of High Voltage Electrical Discharge (HVED) on Endogenous Hormone and Polyphenol Profile in Wheat. Plants, 12(6), 1235. https://doi.org/10.3390/plants12061235









