Is Sellaphora the New Navicula? Cymbosellaphora (Cymbellales), a New Genus Based on Taxa Previously Assigned to Sellaphora
Abstract
:1. Introduction
- (1)
- Striae that are uniseriate or biseriate, radiate, or parallel;
- (2)
- A nonporous conopeum that can be present adjacent to the axial area;
- (3)
- A flat valve face that curves fairly gently into shallow or moderately deep mantles;
- (4)
- Striae that contain small round poroids, which are occluded near their internal apertures by hymenes;
- (5)
- A central and straight raphe system, with distal raphe ends deflected or hooked;
- (6)
- A central external raphe endings that are expanded and slightly deflected towards the primary side;
- (7)
- A central internal raphe endings turned or deflected towards the primary side;
- (8)
- The presence of helictoglossae.
2. Results and Discussion
3. Conclusions
4. Materials and Methods
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Wetzel, C.E.; Ector, L.; Van de Vijver, B.; Compère, P.; Mann, D.G. Morphology, typification and critical analysis of some ecologically important small naviculoid species (Bacillariophyta). Fottea 2015, 15, 203–234. [Google Scholar] [CrossRef]
- Mann, D.G.; Thomas, S.J.; Evans, K.M. Revision of the diatom genus Sellaphora: A first account of the larger species in the British Isles. Fottea 2008, 8, 15–78. [Google Scholar] [CrossRef]
- Liu, Y.; Kociolek, J.P.; Lu, X.; Fan, Y. A new Sellaphora Mereschkowsky species (Bacillariophyceae) from Hainan Island, China, with comments on the current state of the taxonomy and morphology of the genus. Diatom Res. 2020, 35, 85–98. [Google Scholar] [CrossRef]
- Mereschkowsky, C. On Sellaphora, a new genus of diatoms. Ann. Mag. Nat. Hist. 1902, 9, 185–195. [Google Scholar] [CrossRef]
- Mann, D.G.; Chepurnov, V.A.; Droop, S.J.M. Sexuality, incompatibility, size variation, and preferential polyandry in natural populations and clones of Sellaphora pupula (Bacillariophyceae). J. Phycol. 1999, 35, 152–170. [Google Scholar] [CrossRef]
- Mann, D.G. The systematics of the Sellaphora pupula complex: Typification of S. pupula. In Studies on Diatoms. Lange-Bertalot Festschrift; Jahn, R., Kociolek, J.P., Witkowski, A., Compere, P., Eds.; Koeltz: Koenigstein, Germany, 2001. [Google Scholar]
- Mann, D.G.; McDonald, S.M.; Bayer, M.M.; Droop, S.J.M.; Chepurnov, V.A.; Loke, R.E.; Ciobanu, A.; du Buf, J.M.H. The Sellaphora pupula species complex (Bacillariophyceae): Morphometric analysis, ultrastructure and mating data provide evidence for five new species. Phycologia 2004, 43, 459–482. [Google Scholar] [CrossRef]
- Mann, D.G.; Evans, K.M.; Chepurnov, V.A.; Nagai, S. Morphology and formal description of Sellaphora bisexualis sp. nov. (Bacillariophyta). Fottea 2009, 9, 199–209. [Google Scholar] [CrossRef]
- Mann, D.G.; Stickle, A.S. Cytological characteristics of the Sellaphoraceae. Acta Bot. Croat. 2009, 68, 239–250. [Google Scholar]
- Vanormelingen, P.; Evans, K.M.; Chepurnov, V.A.; Vyverman, W.; Mann, D.G. Molecular species discovery in the diatom Sellaphora and its congruence with mating trials. Fottea 2013, 13, 133–148. [Google Scholar] [CrossRef]
- Round, F.E.; Crawford, R.M.; Mann, D.G. The Diatoms. Biology and Morphology of the Genera; Cambridge University Press: Cambridge, UK, 1990; 747p. [Google Scholar]
- Kulikovskiy, M.S.; Glushchenko, A.M.; Genkal, S.I.; Kuznetsova, I.V. Identification Book of Diatoms from Russia; Filigran: Yaroslavl, Russia, 2016; 804p. [Google Scholar]
- Evans, K.M.; Wortley, A.H.; Mann, D.G. An assessment of potential diatom “barcode” genes (cox1, rbcL, 18S and ITS rDNA) and their effectiveness in determining relationships in Sellaphora (Bacillariophyta). Protist 2007, 158, 349–364. [Google Scholar] [CrossRef]
- Evans, K.M.; Wortley, A.H.; Simpson, G.E.; Chepurnov, V.A.; Mann, D.G. A molecular systematic approach to explore diversity within the Sellaphora pupula species complex (Bacillariophyta). J. Phycol. 2008, 44, 215–231. [Google Scholar] [CrossRef] [PubMed]
- Evans, K.M.; Chepurnov, V.A.; Sluiman, H.J.; Thomas, S.J.; Spears, B.M.; Mann, D.G. Highly differentiated populations of the freshwater diatom Sellaphora capitata suggest limited dispersal and opportunities for allopatric speciation. Protist 2009, 160, 386–396. [Google Scholar] [CrossRef]
- Behnke, A.; Friedl, T.; Chepurnov, V.A.; Mann, D.G. Reproductive compatibility and rDNA sequence analyses in the Sellaphora pupula species complex (Bacillariophyta). J. Phycol. 2004, 40, 193–208. [Google Scholar] [CrossRef]
- Mann, D.G.; Poulíčková, A. Homothallism, morphology and phylogenetic position of a new species of Sellaphora (Bacillariophyta), S. pausariae. Plant Ecol. Evol. 2019, 152, 203–218. [Google Scholar] [CrossRef]
- Schiller, W.; Lange-Bertalot, H. Eolimna martinii n. gen., n. sp. (Bacillariophyceae) aus dem Unter-Oligozän von Sieblos/Rhön im Vergleich mit ähnlichen rezenten Taxa. Paläontol. Z. 1997, 71, 163–172. [Google Scholar] [CrossRef]
- Kociolek, J.P.; Stepanek, J.G.; Lowe, R.L.; Johansen, J.R.; Sherwood, A.R. Molecular data show the enigmatic cave-dwelling diatom Diprora (Bacillariophyceae) to be a raphid diatom. Eur. J. Phycol. 2013, 48, 474–484. [Google Scholar] [CrossRef]
- Glushchenko, A.M.; Maltsev, Y.I.; Kociolek, J.P.; Kuznetsova, I.V.; Kulikovskiy, M.S. Molecular and Morphological Investigations of Two Giant Diatom Cymbella Species from the Transbaikal Area (Russia, Siberia) with Comments on Their Distributions. Plants 2022, 11, 2445. [Google Scholar] [CrossRef]
- Kulikovskiy, M.S.; Andreeva, S.A.; Gusev, E.S.; Kuznetsova, I.V.; Annenkova, N.V. Molecular phylogeny of monoraphid diatoms and raphe significance in evolution and taxonomy. Biol. Bull. 2016, 43, 398–407. [Google Scholar] [CrossRef]
- Mann, D.G. Sieves and flaps: Siliceous minutae in the pores of raphid diatoms. In Proceedings of the 6th Symposium on Reecent and Fossil Diatoms, Budapest, Hungary, 1–5 September 1980; Ross, R., Ed.; O. Koeltz, Koenigstein: Koenigstein-Taunus, Germany, 1981; pp. 279–300. [Google Scholar]
- Cox, E.J. Pore occlusions in raphid diatoms—A reassessment of their structure and terminology, with particular reference to members of the Cymbellales. Diatom 2004, 20, 33–46. [Google Scholar]
- Ross, R.; Sims, P. The fine structure of the frustule in centric diatoms: A suggested terminology. Br. Phycol. J. 1972, 7, 139–163. [Google Scholar] [CrossRef]
- Jahn, R. Navicula geisslerae sp. nov.—A small species from the River Spree (Berlin, Germany). Diatom Res. 1992, 7, 69–75. [Google Scholar] [CrossRef]
- Cox, E.J. Placoneis Mereschkowsky: The re-evaluation of a diatom genus originally characterized by its chloroplast type. Diatom Res. 1987, 2, 145–157. [Google Scholar] [CrossRef]
- Kulikovskiy, M.S.; Lange-Bertalot, H.; Metzeltin, D.; Witkowski, A. Lake Baikal: Hotspot of endemic diatoms I. Iconogr. Diatomol. 2012, 23, 7–608. [Google Scholar]
- Lange-Bertalot, H. Navicula sensu stricto, 10 genera separated from Navicula sensu lato, Frustulia. Diatoms Eur. 2001, 2, 1–526. [Google Scholar]
- Kociolek, J.P.; Thomas, E.W. Taxonomy and ultrastructure of five naviculoid diatoms (class Bacillariophyceae) from the Rocky Mountains of Colorado (USA), with the description of a new genus and four new species. Nova Hedwig. 2010, 90, 195–214. [Google Scholar] [CrossRef]
- Kociolek, J.P.; Kulikovskiy, M.; Genkal, S.; Kuznetsova, I. Morphological investigation and transfer of Naviculadicta parasemen Lange-Bertalot to the genus Rexlowea Kociolek & Thomas. Diatom Res. 2017, 32, 477–481. [Google Scholar] [CrossRef]
- Kulikovskiy, M.S.; Kuznetsova, I.V. Morphology, taxonomical position, and distribution of the genera of diatoms Ochigma and Khursevichia from Lake Baikal. Inland Water Biol. 2016, 9, 226–233. [Google Scholar] [CrossRef]
- Metzeltin, D.; Lange-Bertalot, H.; Soninkhishig, N. Diatoms in Mongolia. Iconogr. Diatomol. 2009, 20, 1–691. [Google Scholar]
- Glushchenko, A.; Kuznetsova, I.; Kociolek, J.P.; Kulikoviskiy, M. Karthickia verestigmata gen. et sp. nov.—An interesting diatom with frustular morphology similar to several different cymbelloid diatom genera. Phycologia 2019, 58, 605–613. [Google Scholar] [CrossRef]
- Yana, E.; Nakkaew, S.; Pekkoh, S.; Peerapornpisal, Y.; Tuji, A.; Davis, M.P.; Julius, M.L.; Mayama, S. Valve and ‘Stigma’ Structure and Phylogeny of an Enigmatic Cymbelloid Diatom Karthickia verestigmata Glushchenko, Kulikovskiy & Kociolek. Diatom Res. 2022, 37, 293–305. [Google Scholar] [CrossRef]
- Glushchenko, A.; Gusev, E.; Maltsev, Y.; Kociolek, J.P.; Kuznetsova, I.; Kulikovskiy, M. Cymbopleura natellia—A new species from Transbaikal area (Russia, Siberia) described on the basis of molecular and morphological investigation. PhytoKeys 2021, 183, 95–105. [Google Scholar] [CrossRef]
- Hustedt, F. Die Diatomeenflora norddeutscher Seen mit besonderer Berücksichtigung des holsteinischen Seengebiets. V.-VII. Seen in Mecklenburg, Lauenburg und Nordostdeutschland. Arch. fur Hydrobiol. 1950, 43, 329–458. [Google Scholar]
- Simonsen, R. Atlas and Catalogue of the Diatom Types of Friedrich Hustedt; J. Cramer in der Gebrüder Borntraeger Velagsbuchhandlung: Berlin/Stuttgart, Germany, 1987; Vol. 1. Catalogue. Vol. 2. pls 1–395. Vol. 3. pls. 396–772. pp. 1–525, 772 pls. [Google Scholar]
- Dorofeyuk, N.I.; Kulikovskiy, M.S. Diatoms of Mongolia. Proc. Jt. Russ. Mong. Complex Biol. Exped. 2012, 59, 1–367. [Google Scholar]
- Lange-Bertalot, H.; Hofmann, G.; Werum, M.; Cantonati, M. Freshwater Benthic Diatoms of Central Europe: Over 800 Common Species Used in Ecological Assessment. English Edition with Updated Taxonomy and Added Species; Koeltz Botanical Books: Schmitten-Oberreifenberg, Germany, 2017; 942p. [Google Scholar]
- Lange-Bertalot, H.; Moser, G. Brachysira. Monographie der Gattung und Naviculadicta nov. gen. Bibl. Diatomol. 1994, 29, 1–212. [Google Scholar]
- Chudaev, D.A.; Gololobova, M.A. Diatoms of the Glubokoe Lake (Moscow Region); KMK: Moscow, Russia, 2016; pp. 1–446, 266 pls. (In Russian) [Google Scholar]
- Hustedt, F. Bacillariales aus Schlesien. II. Nachtrag. Int. Rev. Gesamten Hydrobiol. Hydrogr. 1925, 13, 345–357. [Google Scholar] [CrossRef]
- Sumita, M.; Watanabe, T. Epilithic freshwater diatoms in Jakarta, Surabaya and Singapore. Jpn. J. Phycol. 1979, 27, 1–6. [Google Scholar]
- Watanabe, T.; Usman, R. Epilithic freshwater diatoms in Central Sumatra. Diatom 1987, 3, 33–87. [Google Scholar] [CrossRef]

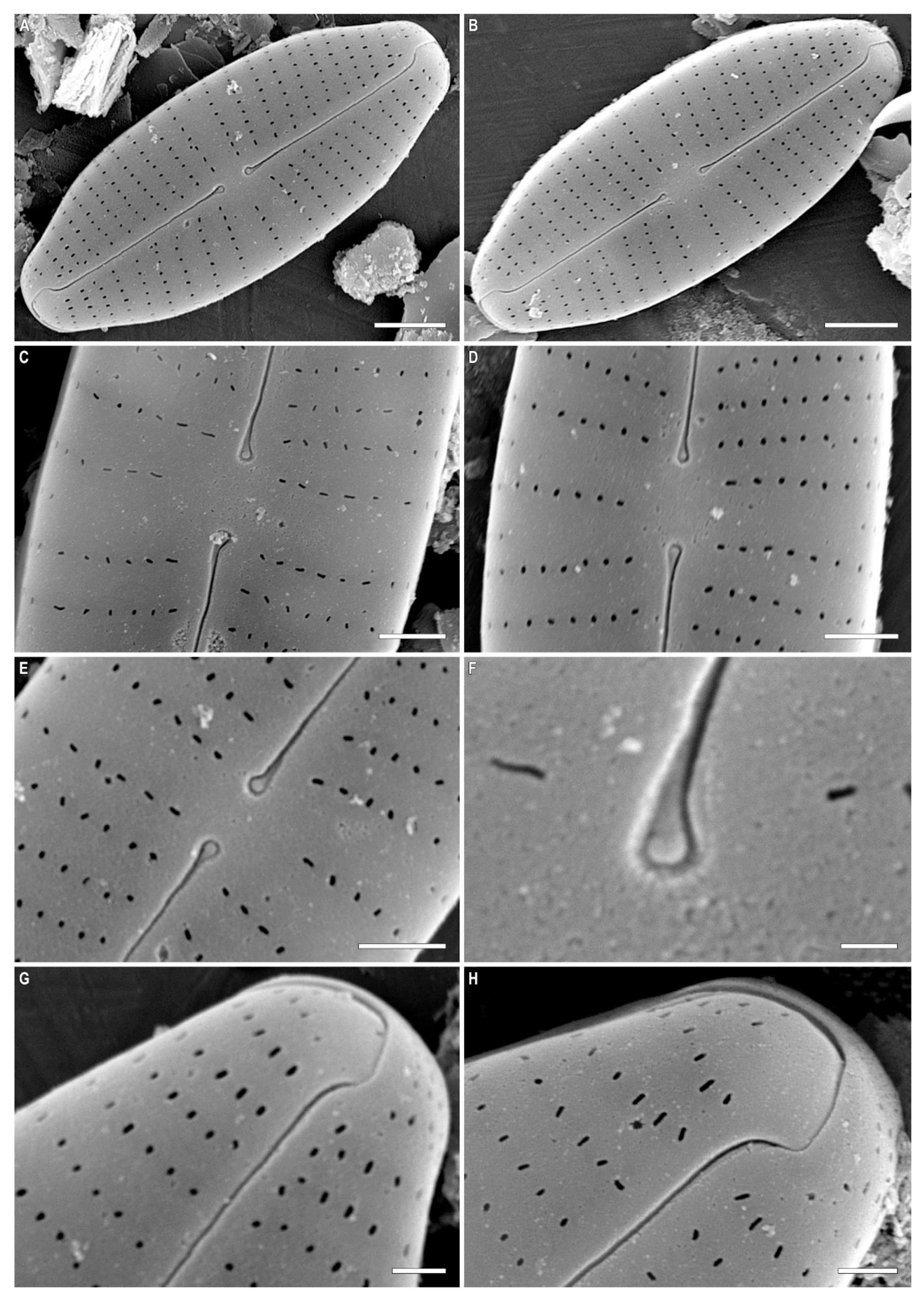

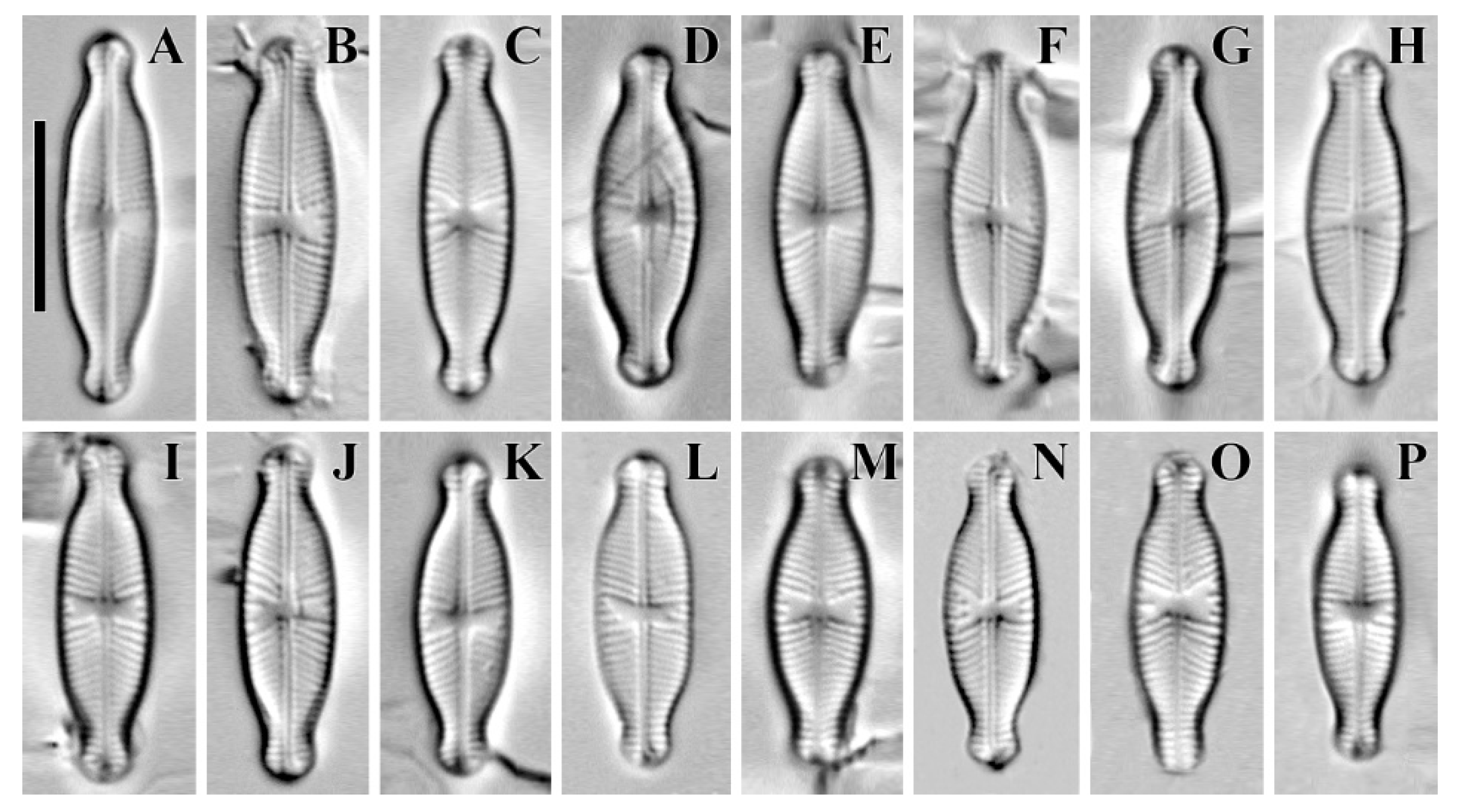



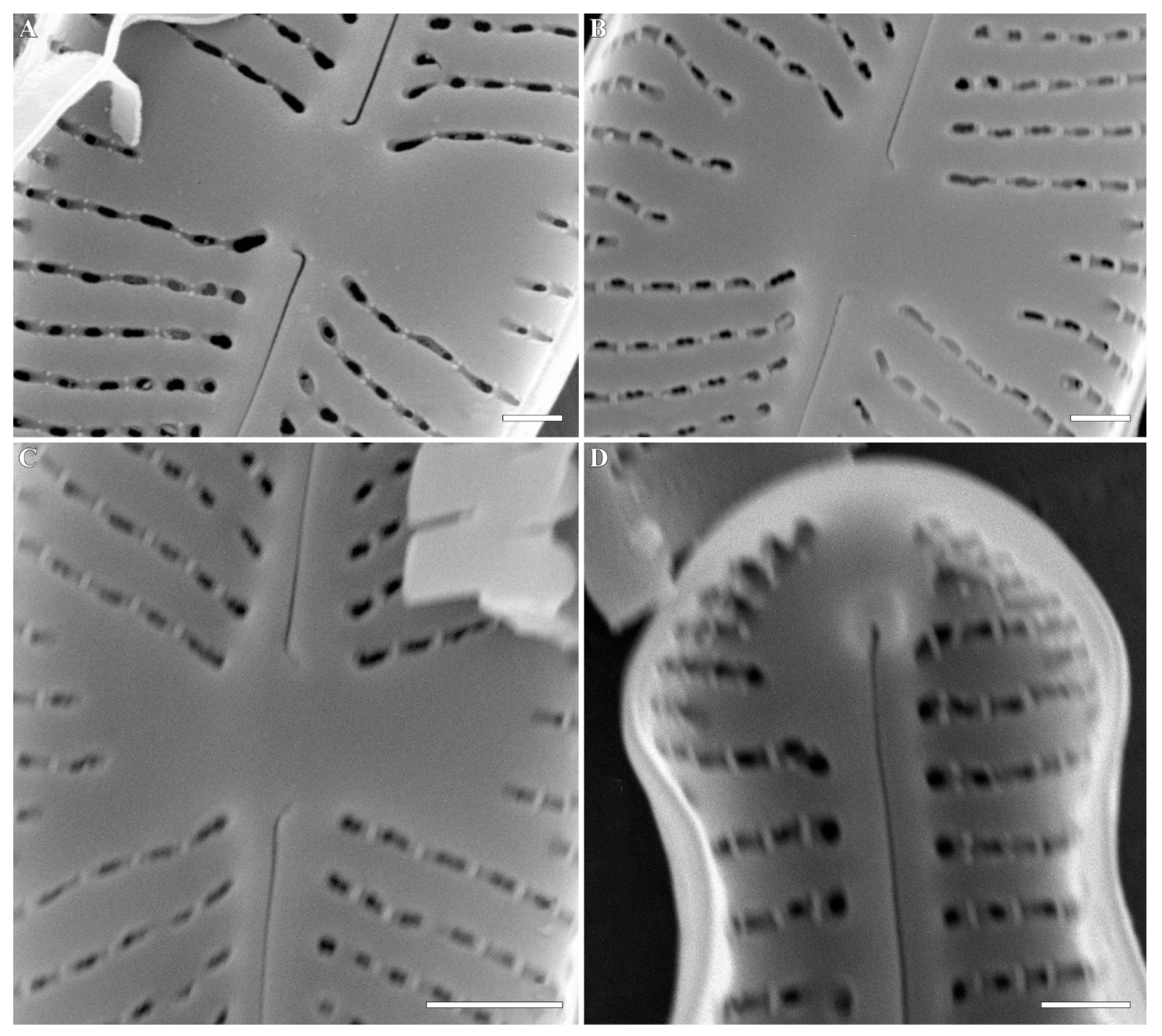

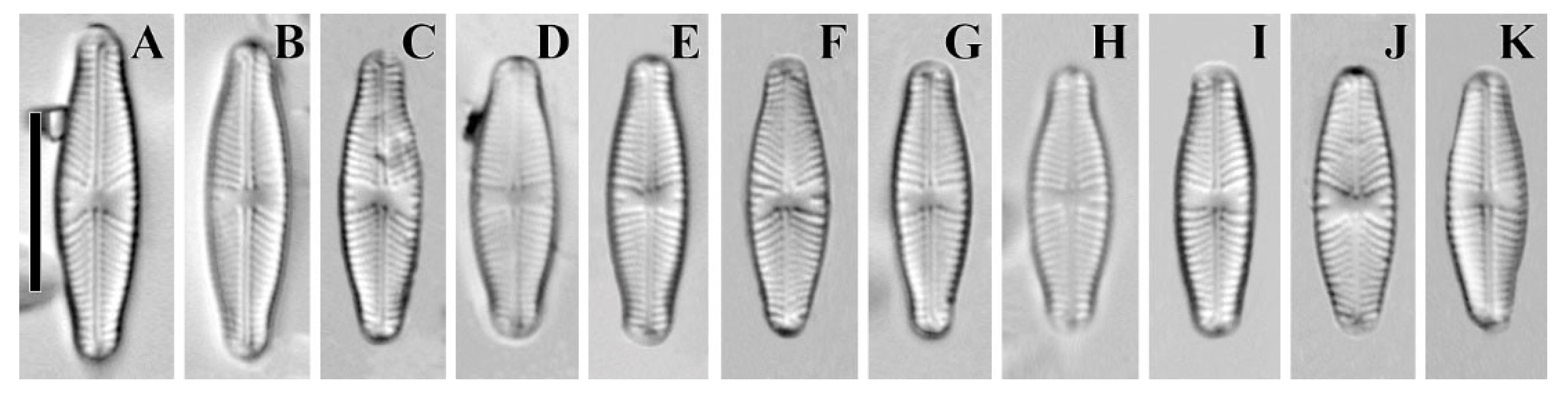

| Cymbosellaphora vietnamensis sp. nov. | Cymbosellaphora absoluta comb. nov. | Cymbosellaphora circumborealis comb. nov. | Cymbosellaphora geisslerae comb. nov. | Cymbosellaphora laterostrata comb. nov. | |
|---|---|---|---|---|---|
| References | This study | [36,37,38,39]; This study | [40]; This study | [25,40,41]; This study | [37,38,40,41,42] |
| Distribution | Vietnam; Ba Bể Lake | Holarctic; Central Europe; Mongolia | Holarctic: Europe; North America; Canada; Mongolia | Europe; Canada; Russia | Europe; Russia; Mongolia |
| Outline | Almost liner to linear–elliptic, naviculoid, slightly dorsiventral in longer cells with broadly protracted ends | Elliptic–lanceolate with drawn out, obtusely rounded rostrate to subcapitate ends | Linear–lanceolate or elliptical–lanceolate or lanceolate | Narrow–elliptical with protracted and broadly rounded poles | Linear–elliptic with subcapitate ends |
| Axial area | Narrow, straight | Narrow | Very narrow | Narrow | Narrow |
| Central area | Variable, small to medium-size, transversely widened and irregularly delimited | Variable, small-to-medium size, transversely widened, and irregularly delimited | Transversely expanded and bordered by 1, 2, and rarely 3 very short striae | Transversely expanded and bordered by 1–4 very short striae | Variable, formed by short striae |
| Valve length (μm) | 10.7–31.4 | 10–20 | 9–18 | 8.9–27.6 | 22.9–31.0 |
| Valve breadth (μm) | 5.0–6.7 | 4.5–6.0 | 4.2–5.5 | 4.0–6.6 | 7.1–8.8 |
| Striae in 10 μm | 14–16 | 25–28 | 17–20 | 18–25 | 18.5–20.4 |
| Areolae in 10 μm | 35–40 | 25–30 | 35–40 | 30.7–38.7 | 24.8–31.1 |
| Sample No | Slide No | Locality | Coordinates | Substratum | Cond., µS/cm | pH | t, °C | Collection of Date |
|---|---|---|---|---|---|---|---|---|
| Vietnam | ||||||||
| BB9 | 02151 | Ba Bể Lake, Bắc Kạn Province | 22°23.605′ N; 105°36.856′ E | benthos a depth of 3 m | 26 | 8.5 | 174 | 29 April 2015 |
| Mongolia | ||||||||
| Mn079 | 02431 | Davaa Lake | 48°10.803′ N; 98°46.107′ E | benthos | 20 | 8.1 | 13 | 8 July 2015 |
| Mn088 | 02440 | Davaa Lake | 48°10.827′ N; 98°45.828′ E | sand | 20 | 6.9 | 13 | 8 July 2015 |
| Mn089 | 02441 | Davaa Lake | 48°10.827′ N; 98°45.828′ E | sand | 20 | 6.9 | 13 | 8 July 2015 |
| Mn090 | 02442 | Davaa Lake | 48°10.827′ N; 98°45.828′ E | stone and sand | 20 | 6.9 | 13 | 8 July 2015 |
| Mn093 | 02445 | Davaa Lake | 48°10.827′ N; 98°45.828′ E | periphyton | 20 | 6.9 | 13 | 8 July 2015 |
| Mn094 | 02446 | Davaa Lake | 48°11.145′ N; 98°45.746′ E | bottom sediment a depth of 2.5 m | 38 | 7.7 | 15 | 8 July 2015 |
| Mn095.1 | 02447 | Davaa Lake | 48°11.145′ N; 98°45.746′ E | bottom sediment a depth of 4 m | 38 | 7.7 | 15 | 8 July 2015 |
| Mn095.5 | 02451 | Davaa Lake | 48°11.145′ N; 98°45.746′ E | bottom sediment a depth of 4 m | 38 | 7.7 | 15 | 8 July 2015 |
| Mn104 | 02457 | Unnamed river flowing out of the Davaa Lake | 48°10.791′ N; 98°46.272′ E | periphyton | 38 | 7.7 | 15 | 8 July 2015 |
| Mn289.1 | 02642 | Unnamed Lake near the Khövsgöl Lake, separated by a sandbar | 50°59.165′ N; 100°42.514′ E | bottom sediment a depth of 5 m | 100 | 8.9 | 18 | 28 July 2015 |
| Mn312 | 02663 | Khövsgöl Lake | 50°47.215′ N; 100°31.837′ E | periphyton | 243 | 8.6 | 12 | 28 July 2015 |
| Mn335.8 | 02684 | Khövsgöl Lake, Heeguer Bay | 50°38.641′ N; 100°31.397′ E | benthos | 304 | 10.6 | 22 | 22 July 2015 |
| Mn308 | 02695 | Khövsgöl Lake | 50°47.215′ N; 100°31.837′ E | periphyton | 243 | 8.6 | 12 | 28 July 2015 |
| Mn283.2 | 03009 | Khövsgöl Lake, Boreug Bay | 50°59.380′ N; 100°42.507′ E | benthos | 236 | 8.7 | 11.5 | 21 July 2015 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kulikovskiy, M.S.; Glushchenko, A.M.; Genkal, S.I.; Kuznetsova, I.V.; Maltsev, Y.I.; Kociolek, J.P. Is Sellaphora the New Navicula? Cymbosellaphora (Cymbellales), a New Genus Based on Taxa Previously Assigned to Sellaphora. Plants 2023, 12, 3890. https://doi.org/10.3390/plants12223890
Kulikovskiy MS, Glushchenko AM, Genkal SI, Kuznetsova IV, Maltsev YI, Kociolek JP. Is Sellaphora the New Navicula? Cymbosellaphora (Cymbellales), a New Genus Based on Taxa Previously Assigned to Sellaphora. Plants. 2023; 12(22):3890. https://doi.org/10.3390/plants12223890
Chicago/Turabian StyleKulikovskiy, Maxim S., Anton M. Glushchenko, Sergei I. Genkal, Irina V. Kuznetsova, Yevhen I. Maltsev, and John Patrick Kociolek. 2023. "Is Sellaphora the New Navicula? Cymbosellaphora (Cymbellales), a New Genus Based on Taxa Previously Assigned to Sellaphora" Plants 12, no. 22: 3890. https://doi.org/10.3390/plants12223890
APA StyleKulikovskiy, M. S., Glushchenko, A. M., Genkal, S. I., Kuznetsova, I. V., Maltsev, Y. I., & Kociolek, J. P. (2023). Is Sellaphora the New Navicula? Cymbosellaphora (Cymbellales), a New Genus Based on Taxa Previously Assigned to Sellaphora. Plants, 12(22), 3890. https://doi.org/10.3390/plants12223890







