Abstract
Oxygenated polyunsaturated fatty acids (oxylipins) are bioactive molecules established as important mediators during inflammation. Different classes of oxylipins have been found to have opposite effects, e.g., pro-inflammatory prostaglandins and anti-inflammatory resolvins. Production of the different classes of oxylipins occurs during distinct stages of development and resolution of inflammation. Chronic inflammation is involved in the progression of many pathophysiological conditions and diseases such as non-alcoholic fatty liver disease, insulin resistance, diabetes, and obesity. Determining oxylipin profiles before, during, and after inflammatory-related diseases could provide clues to the onset, development, and prevention of detrimental conditions. This review focusses on recent developments in our understanding of the role of oxylipins in inflammatory disease, and outlines novel technological advancements and approaches to study their action.
1. Introduction
Oxylipins are bioactive lipid mediators derived from the enzymatic or non-enzymatic oxidation of polyunsaturated fatty acids (PUFAs). The enzymatic oxidation of PUFAs is carried out by cyclooxygenases (COX), lipoxygenases (LOX) [,,,] or cytochrome P450s (CYP) [,,,,,]. PUFAs are those fatty acids that have more than one double bond in their backbone. When the final double bond is at the carbon–carbon double bond in the n-6 position (the sixth bond counting from the methyl end), they are referred to as omega-6 fatty acids. When that bond is in the n-3 position, these are known as omega-3 fatty acids []. There are two PUFAs in the human body that cannot be produced endogenously, linoleic and alpha-linolenic acid (LA and ALA), and accordingly they are defined as essential fatty acids. LA is an omega-6 fatty acid that is metabolised to arachidonic acid (AA) and dihomo-gamma-linolenic acid (DGLA). ALA is an omega-3 fatty acid which is converted to eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA). Therefore, based on research discoveries so far AA, DGLA, EPA and DHA are the non-essential fatty acids which give rise to numerous oxylipin families (Figure 1). Importantly, mammals including humans cannot convert omega-6 to omega-3 PUFAs. Hence, the levels of these oxylipins reflect the consumption of omega-3 or omega-6 PUFAs. Specific omega-3 and omega-6 metabolites have been implicated to have opposing beneficial or detrimental effects in human health and disease, with omega-3-derived compounds found to be generally anti-inflammatory while oxylipins originating from omega-6 fatty acids have predominantly pro-inflammatory effect [,]. The pleiotropic effects of these compounds are increasingly appreciated with different oxylipins having dissimilar roles in physiological and pathophysiological processes (reviewed excellently in [,,,,]). Importantly, the superclass of oxylipins includes hundreds of structurally and stereochemically distinct species []. Thus, profiling of oxylipins can be an important tool in investigating their role in pathophysiological processes.
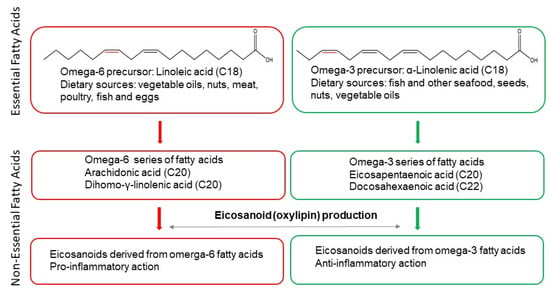
Figure 1.
General overview of essential and non-essential polyunsaturated fatty acids. LA is a non-essential fatty acid and precursor of the omega-6 series of fatty acids and eicosanoids which have mostly pro-inflammatory action. ALA is a non-essential fatty acid and precursor of the omega-3 series of fatty acids and eicosanoids which have predominantly anti-inflammatory effect. Omega-6 and omega-3 double bonds are marked in red. Chemical formulas were created using ChemDraw®. More detailed description of the role of PUFAs in inflammatory processes, including exceptions, is outlined in the main text.
Although all non-essential fatty acids can serve as precursors for the synthesis of oxylipins, the eicosanoids identified to date predominantly originate from AA due to enzymatic activity (Figure 2). In addition, some eicosanoids can form as a result of non-enzymatic oxidation, although those serve mostly as markers for oxidative stress in vivo (eicosanoid biology reviewed in []). However, the majority of the oxylipins mentioned in this review are products of the enzymes LOX, COX, or CYP. Thus, research interest focuses mostly on the enzymatically produced oxylipins.
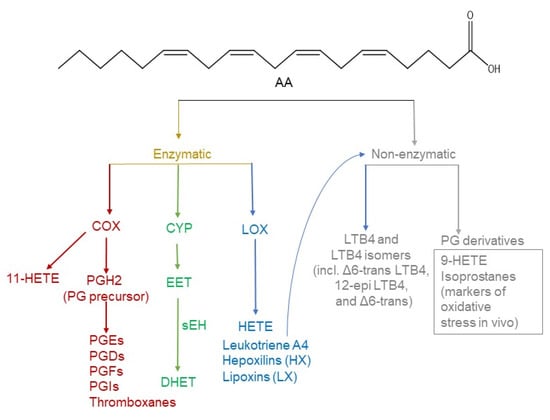
Figure 2.
Arachidonic acid is the main precursor of eicosanoids. Eicosanoids can be produced enzymatically by COX, LOX, and CYP, and non-enzymatically. COX-derived eicosanoids include prostaglandins as well as 11-hydroxyeicosatetraenoic acid (HETE). CYP enzymatic activity produces EETs that are further hydrolysed to DHET by soluble epoxide hydrolase (sEH). HETEs, leukotrienes, hepoxilins, and lipoxins are produced by LOX. Leukotriene A4 is subjected to further non-enzymatic hydrolysis to form Leukotriene B4 LTB4 and its derivatives. Non-enzymatic oxidation of AA results in the formation of eicosanoids used as markers of oxidative stress in vivo. Chemical formula for AA was created using ChemDraw®.
Prostaglandins (PG) are derived from AA by COX, and were the first oxylipins to be identified []. Their importance in disease has been investigated ever since it was reported that aspirin inhibits COX enzymes and the formation of their metabolites (prostaglandins) [,]. Since then, oxylipins have been implicated in inflammation, cardiovascular disease, atherosclerosis, and multiple other physiological and pathophysiological processes [,,,,,,,,,,,]. In September 2022, searching PubMed with ‘oxylipin’ as the keyword returned 6848 results, while ‘eicosanoids’ gave 154,900 results. Although technically eicosanoids refer to lipids containing 20 carbon atoms, with time the term ‘eicosanoids’ has expanded to also include similar metabolites of other PUFAs and therefore is also used as a synonym for ‘oxylipin’. Thus, in this publication the two terms, oxylipin and eicosanoids, will be used interchangeably.
The impressive number of entries for ‘eicosanoid’ in PubMed underscores the efforts put into determining the role of those bioactive molecules in health and disease. The sheer volume of published articles presents a substantial obstacle in reviewing all publications. Therefore, this review focusses on research articles published in the last three years (2019–2022) with the aim of highlighting the latest developments in several key areas. Exceptions to this are articles published prior to 2017 but considered to present results of major importance to the research area. The main areas covered in this review are: (1) non-alcoholic fatty liver disease (NAFLD), (2) obesity, diabetes, and autoimmune diseases, and (3) technological advancements and future directions.
2. Non-Alcoholic Fatty Liver Disease (NAFLD)
NAFLD is a chronic liver disease that results in an excessive increase in fat (steatosis) in the liver without evidence of other causes of liver disease (such as alcohol). There are two progressive subtypes of NAFLD: non-alcoholic fatty liver (NAFL) and non-alcoholic steatohepatosis (NASH) which is progression of NAFLD beyond NAFL alone []. The main difference between the two types is the presence of inflammation. NAFL is characterised by little or no inflammation or liver damage. NAFL usually does not cause complications, though it can lead to pain from liver enlargement [,]. In comparison, NASH is categorised by inflammation and liver damage in addition to the build-up of fat. NASH can result in the development of cirrhosis which in turn can lead to liver cancer [,,,]. Approximately 25% of the general population will develop NAFLD and, of those, 1.5–6.45% will progress to NASH [,]. Thus, detection, prevention, and treatment of NAFLD is a major challenge for modern medicine.
Although the pathogenesis of NAFLD is still not completely understood, its connection to fat build-up is clear. Bodyweight reduction among NAFLD patients showed lower levels of triglycerides, cholesterol, and the eicosanoids 5-Oxo-eicosatetraenoic acid (5-oxo-ETE) and lipoxinA4 (LXA4), and was linked to a significant improvement in steatosis []. Analysis of liver tissue samples from obese patients has indicated a complicated relationship between eicosanoid levels and paediatric NAFLD. CYP-produced eicosanoids have been shown to be downregulated during fibrosis, but increased in the steatosis stage, while the levels of precursor PUFAs and the production of hydroxyeicosatetraenoic acids (HETEs), hydroxyeicosapentaenoic acids (HEPEs), and hydroxydocosahexaenoic acids (HDHAs) remained unchanged []. CYP-derived oxylipins are now investigated for their potential hepatoprotective effects. Quantification of circulating epoxyeicosatrienoic acids (EETs) has indicated that when compared with controls, total EET; 11,12-EET; 14,15-EET, total dihydroxyeicosatrienoic acids (DHET); 11,12-DHET; and 14,15-DHET were significantly lower in NAFLD and, furthermore, total EET and DHET were lower in NASH compared with steatosis (i.e., NAFL, but not NASH) []. EETs are derived from AA, therefore they are omega-6 fatty acids, yet they appear to be potential targets for prevention of NASH development. Indeed, in an animal study it was reported that receiving a diet enriched in the omega-6 precursor LA during pregnancy, lactation and growth did not induce obesity in offspring as previous reports had suggested. The authors reported that high LA intake leads to the simultaneous synthesis of both pro-inflammatory (5-HETE, 12-HETE, Leukotriene B4 (LTB4)) and anti-inflammatory (8,9-EET) and pro-resolving (LXA4) bioactive molecules, thus highlighting the need for a detailed assessment of individual eicosanoids in the development of hepatic steatosis [].
NAFLD is usually asymptomatic and diagnosed after the incidental finding of abnormal liver disease or steatosis during liver imaging, and after other causes of liver disease have been excluded []. Liver biopsy is the gold standard for diagnosing and staging NAFLD (reviewed in more detail in [,]). Therefore, there is significant interest in developing non-invasive and accurate methods for NAFLD diagnosis and assessing its progression. Measuring oxylipin levels presents one possible approach. Evaluation of the levels of eicosanoid concentration in serum and liver tissue in rats during steatosis indicated a strong positive correlation between 9-HODE and 13-HODE and NAFLD progression. In addition, the study reported a moderate correlation between the levels of the specialised pro-resolving mediator (SPM) resolvin E1 in liver and serum. The levels of HETE also showed association with the NAFLD phenotype, but the link was weak []. Thus, there is potential for eicosanoid profiles to be used as biomarkers for tissue remodelling that leads to NAFLD.
Considering the negative impact of NAFLD on human health, much research has also focused on effective treatment approaches in addition to disease diagnosis and progression. One study evaluated the possible use of fibrates (a class of amphipathic carboxylic acids) and omega-3 PUFAs, typically used to combat hypertriglyceridemia. The results indicated a reduction in plasma concentration of the pro-inflammatory prostaglandin E2 (PGE2), as well as prostaglandin E1 (PGE1), prostaglandin D1 (PGD1) and thromboxane B2 (TXB2) (omega-3 PUFAs) and reduced CYP-derived DHETs (fibrate), but increased prostacyclin, 17-HDHA, 18-HEPE, dihydroxydocosapentaenoic acids (19,20-DiHDPA)(omega-3 PUFAs). Thus, omega-3 PUFAs and fibrate have some effect on the formation of lipid mediators with potential effects on chronic inflammation, but their use for treating NAFLD needs further investigation [].
Lipidomics analysis of blood and liver mouse samples indicated that Sagittaria sagittifolia (SSG), a flowering plant in the family Alismataceae, has protective effects against high-fat diet-induced NAFLD. SSG interfered with AA metabolism via the Nrf/HO-1 signalling pathway during oxidative stress in the liver []. Another study indicated that the sodium–glucose cotransporter 2 inhibitor canagliflozin mitigates fatty liver and hyperglycemia without affecting body weight in mice. Lipidomics analysis showed that canagliflozin increased the levels of PGE2 and resolvin E3 in the mouse liver. Interestingly, PGE2 reduced fat depositions in mouse primary hepatocytes exposed to palmitic acid []. Thus, certain eicosanoids might be used as potential treatment options to tackle NAFLD progression.
3. Obesity and Diabetes
Apart from NAFLD, chronic inflammation is associated with obesity which in turn contributes to the development of insulin resistance and type 2 diabetes (T2D). The plasma level of three eicosanoids (unknown eicosanoid (EIC 62), 8-iso-prostaglandin A1 (8-iso-PGA1), and 12-hydroxy-5,8,10-heptadecatrienoic acid (12-HHTrE) was shown to predict incident T2D [] and the resolvin D2/LTB4 ratio, and may serve as a biomarker of prognosis for ischemic stroke []. An analysis of clinical samples revealed significant differences in the levels of four oxylipins (PGF2α, PGE2, 15-keto-PGE2, and 13,14-dihydro-15-keto-PGE2) between T2D patients and the corresponding lean and obese control subjects, with the combination of PGF2α and 15-keto-PGE2 having the most predictive value []. Targeted lipidomics analysis of human urine samples indicated that metabolite products of PGD and PGE are associated with low-grade chronic inflammation in obesity []. Upregulated PGE2 production by β-cells may have a role in the β-cells’ adaptation response to obesity and insulin resistance in T2D when PGE2 and its receptor EP3 are highly expressed []. Further supporting the connection between increased oxylipin levels, insulin resistance and T2D, is how weight-loss intervention in obese, insulin-resistant, sedentary women was accompanied by decreased concentrations of 9,10-DiHODE, 12,13-DiHODE and 9,10-DiHOME [], while 5-LOX leukotrienes were increased []. Thus, eicosanoids have shown potential as predictors of obesity and T2D.
However, the levels of other oxylipins such as the SPMs and hydroxy-DHA metabolites were shown to be lower in obesity and white adipose tissue inflammation []. Animal studies revealed that increasing the bioavailability of SPMs such as Maresin-1 minimises inflammation and mediates therapeutic actions []. It is not surprising therefore that eicosanoids are seen as potential targets to combat obesity. One such approach suggests seeking ways to increase the energy expenditure of thermogenic tissues such as brown and brite adipose tissue. Both 5-HETE and 5,6-EET have been reported to be consistently associated with the abundance of those tissues, though further studies are needed to determine whether those eicosanoids are candidates that affect thermogenic capacity []. In addition, data has suggested that PGE2 metabolites could be an indicator of the efficacy of mesenchymal stem cell treatment in systemic lupus erythematosus. However, further studies and clinical trials are needed []. Thus, eicosanoids also present viable therapeutic targets in a range of pathophysiological conditions.
Another potential approach to influence eicosanoid levels could be through diet and its effect on gut microbiota composition. Recently, the role of eicosanoids as modulators of inflammation in the gastrointestinal tract has been reviewed [] as well as changes in gut microbiota composition caused by dietary and endogenous lipids, including eicosanoids []. These reviews describe very well the link between diet, gut microbiota, and eicosanoids. Therefore, here we will mention only a few examples of that association. Animal studies have demonstrated that gut microbiota dysbiosis modifies the oxylipin profile in healthy and obese rats, with several significant correlations between different bacteria taxa and eicosanoids. Among those, the positive association between Proteobacteria and LTB4 was found to be especially strong []. In addition, gut bacteria have been proposed to be responsible for the post-prandial decrease in soluble epoxide hydrolase, an enzyme responsible for metabolising EETs to the less active diols []. Moreover, prostaglandin E2 (PGE2), a well-known mediator of inflammation, was found to inhibit mucosal regulatory T cells in a manner regulated by gut microbiota []. Thus, evidence suggests that modifying nutritional intake could present a possible non-invasive approach to affect inflammation-related diseases by modulating eicosanoid production.
4. Technological Advancements and Future Directions
The importance of oxylipins in physiological and pathophysiological processes is underscored by their role in the ability of cells to acquire different functional phenotypes depending on the microenvironment. Omega-3 and omega-6 PUFA-derived oxylipins can modulate the inflammatory phenotype of cells [] by acting as ligands for receptors such as the peroxisome proliferator-activated receptor PPAR [] and prostaglandin E2 (PGE2) receptor PTGER4 []. It is not surprising then that there are continuous efforts to improve their detection and quantitation. In this section, we will outline current and new technical approaches to determine oxylipin profiles.
The eicosanoid class comprises hundreds of structurally and stereochemically distinct species derived either enzymatically or non-enzymatically from a handful of precursors. It is therefore not surprising that within the group of eicosanoids can be found isomers (e.g., PGE2 and PGF2α) with different biological functions []. In addition to their diversity, eicosanoids occupy a small mass range and are found in low nanomolar concentrations in biological samples (e.g., human plasma and murine bone marrow-derived macrophages []). Thus, their detection and evaluation require methods that are sensitive, selective, and reproducible.
Currently, mass spectrometry (MS) is the main technique used to interrogate the lipidome as demonstrated by the methods used in the experimental articles on NAFLD, obesity and diabetes reviewed here (Table 1). Mass spectrometry is a powerful technique for identifying and quantifying known and unknown analytes, and offers high specificity and selectivity. Technical advances have given mass spectrometry a range of tools to obtain information at the molecular level from samples, such as determining the molecular weight of an analyte, its separation from isomeric and isobaric species, its chemical formula, its molecular structure, and structural information [,,]. Mass spectrometry can be used to investigate a wide range of classes of molecules, including those with small and large molecular weights, volatile and non-volatile, polar and non-polar [,,].

Table 1.
Methods of eicosanoid detection.
Applications using mass spectrometry are either targeted or untargeted. Untargeted approaches attempt to measure as many compounds as possible in a sample, and can be used to discover unanticipated changes between experimental groups. This information can then be used to generate and test hypotheses, for example by applying targeted approaches which offer a selection of the best possible conditions for the detection, identification, and (absolute or relative) quantitation of particular analytes of choice, and is usually used in follow-up experiments. Targeted approaches may use multiple reaction monitoring (MRM) methods which use the knowledge of the mass of the ionised analyte and its corresponding fragments formed during the mass spectrometry analysis [,]. This provides a high level of selectivity and specificity, ensuring the correct analyte is targeted. It is important to note that targeted approaches do not provide global coverage, and that compounds present in the sample that are not the targets of interest will not be detected.
For lipidomics, liquid chromatography-mass spectrometry (LC-MS) is commonly used. LC separates molecules depending on their hydrophobicity, molecular size, and polarity, and covers a broad range of non-polar and weakly polar analytes [,]. Another separation method that is gaining popularity in metabolomics is ion mobility coupled with mass spectrometry (IMS-MS; IM-MS) (IMS) [,,,,,,]. Coupling IM to MS offers several benefits such as improved selectivity and therefore increased capability to analyse complex mixtures, and the resolving of isomeric [] and isobaric [] compounds. Furthermore, IMS provides additional information about the shape and conformation of the ions under defined experimental conditions [,], thus adding molecular, structural, and conformational information which further improves the accurate determination of metabolites in complex structures [,,]. For example, IM-MS has been used to identify unknown oxylipins in different biologically relevant matrices []. However, like the other methods, IMS has its disadvantages such as the need for improved ion mobility resolutions.
Recently, supercritical fluid chromatography (SFC) has gained traction as an alternative technique to LC in lipidomics due to its high efficiency [,]. Briefly, SFC uses supercritical fluid such as CO2 as a mobile phase [], which results in high separation efficiency and short separation time [,]. SFC-MS/MS methods have been reported to detect inflammation-related lipids including oxylipins in rats [], as well as for the simultaneous measurement of five AA-derived metabolites (PGD2, PGE2, PGF2α, 6KetoPGF1α and LTB4) in biological samples []. Thus, SFC coupled to MS is a promising approach for the interrogation of the lipidome.
Novel ionisation approaches have appeared with the development of ambient mass spectrometry, characterised by direct sampling and ionisation of the analytes with no or minimal sample preparation []. Ambient ionisation MS has been successfully employed to rapidly differentiate bacterial species based on their lipid profiles []. Mass spectrometry imaging (MSI) developments allow more detailed investigations of biological questions such as the biochemical origin of lipid spatial distribution (reviewed in []). Examples of MSI techniques include secondary ion mass spectrometry (SIMS), Desorption electrospray ionisation (DESI) and matrix-assisted desorption/ionisation (MALDI). Lipid characterisation is now possible due to technological advances in SIMS (reviewed in []). DESI-MSI can record 2D distributions of polar lipids in tissue slices at ambient conditions (at atmospheric pressure) [], and has been developed for the simultaneous imaging of polar and non-polar lipids in mouse brain tissue []. Improvements in MALDI resolution resulted in the identification and localisation of lipids within the kidney, as well as the localisation of lipid droplets with lesion-specific macrophages []. Thus, further method developments could provide the means to image eicosanoids in a variety of biological samples.
5. Conclusions
In recent years there has been substantial research focused on eicosanoids, which started with the discovery of prostaglandins and elucidating their role in inflammation. Continuous efforts have resulted in the identification of other oxylipin classes, with resolvins being the latest addition to that list. Eicosanoids have been found to have both pro- and anti-inflammatory roles, and have been shown to be associated with specific disease manifestations and, therefore, could potentially serve as biomarkers [,]. In addition, eicosanoids have been found to have a role in the host–pathogen interactions, thus presenting a prospective therapeutic target in bacterial infections [,,]. It is not surprising then that the field of lipidomics keeps expanding with the application of novel techniques and approaches such as SFC, ambient MS, and ion mobility increasing not only our knowledge of known eicosanoids but also leading to the identification and characterisation of novel lipids (including oxylipins) and other lipid classes.
Author Contributions
Author Contributions: Writing—original draft preparation, M.M., J.M.; writing—review and editing, M.M., J.J., J.M. All authors have read and agreed to the published version of the manuscript.
Funding
This review received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| PUFA | Polyunsaturated fatty acid | COX | Cyclooxygenase |
| LOX | Lipoxygenase | CYP | Cytochrome P450 |
| LA | Linoleic acid | ALA | Alpha-linolenic acid |
| AA | Arachidonic acid | DGLA | Dihomo-gamma-linolenic acid |
| EPA | Eicosapentaenoic acid | DHA | Docosahexaenoic acid |
| PG | Prostaglandin | TX | Thromboxane |
| LT | Leukotriene | NAFLD | Non-alcoholic fatty liver disease |
| NAFL | Non-alcoholic fatty liver | NASH | Non-alcoholic steatohepatosis |
| 5-oxo-ETE | 5-Oxo-eicosatetraenoic acid | HETE | Hydroxyeicosatetraenoic acid |
| HEPE | Hydroxyeicosapentaenoic acid | HDHA | Hydroxydocosahexaenoic acid |
| EET | Epoxyeicosatrienoic acid | DHET | Dihydroxyeicosatrienoic acid |
| LTB4 | Leukotriene B4 | LXA4 | LipoxinA4 |
| HODE | Hydroxyoctadecadienoic acid | PGE2 | Prostaglandin E2 |
| PGE1 | Prostaglandin E1 | PGD1 | Prostaglandin D1 |
| TXB2 | Thromboxane B2 | DiHDPA | Dihydroxydocosapentaenoic acid |
| SSG | Sagittaria sagittifolia | T2D | Type 2 diabetes |
| PGF2α | Prostaglandin F2α | DiHODE | Dihydroxyoctadecadienoic acid |
| DiHOME | Dihydroxyoctadecenoic acid | MS | Mass spectrometry |
| MRM | Multiple reaction monitoring | LC-MS | Liquid chromatography-mass spectrometry |
| IMS-MS | IM-MS | IMS | Ion mobility-mass spectrometry |
| SFC | Supercritical fluid | DESI-MSI | Desorption electrospray ionisation mass spectrometry imaging |
References
- Funk, C.D. Prostaglandins and leukotrienes: Advances in eicosanoid biology. Science 2001, 294, 1871–1875. [Google Scholar] [CrossRef] [PubMed]
- Serhan, C.N.; Chiang, N.; Van Dyke, T.E. Resolving inflammation: Dual anti-inflammatory and pro-resolution lipid mediators. Nat. Rev. Immunol. 2008, 8, 349–361. [Google Scholar] [CrossRef] [PubMed]
- Simmons, D.L.; Botting, R.M.; Hla, T. Cyclooxygenase isozymes: The biology of prostaglandin synthesis and inhibition. Pharm. Rev 2004, 56, 387–437. [Google Scholar] [CrossRef]
- Smith, W.L.; DeWitt, D.L.; Garavito, R.M. Cyclooxygenases: Structural, cellular, and molecular biology. Annu. Rev. Biochem. 2000, 69, 145–182. [Google Scholar] [CrossRef] [PubMed]
- Capdevila, J.H.; Falck, J.R.; Imig, J.D. Roles of the cytochrome P450 arachidonic acid monooxygenases in the control of systemic blood pressure and experimental hypertension. Kidney Int. 2007, 72, 683–689. [Google Scholar] [CrossRef] [PubMed]
- Fleming, I. DiscrEET regulators of homeostasis: Epoxyeicosatrienoic acids, cytochrome P450 epoxygenases and vascular inflammation. Trends Pharm. Sci 2007, 28, 448–452. [Google Scholar] [CrossRef]
- Imig, J.D. Targeting epoxides for organ damage in hypertension. J. Cardiovasc. Pharm. 2010, 56, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Roman, R.J. P-450 metabolites of arachidonic acid in the control of cardiovascular function. Physiol. Rev. 2002, 82, 131–185. [Google Scholar] [CrossRef]
- Spector, A.A.; Norris, A.W. Action of epoxyeicosatrienoic acids on cellular function. Am. J. Physiol. Cell Physiol. 2007, 292, C996–C1012. [Google Scholar] [CrossRef]
- Williams, J.M.; Murphy, S.; Burke, M.; Roman, R.J. 20-hydroxyeicosatetraeonic acid: A new target for the treatment of hypertension. J. Cardiovasc. Pharm. 2010, 56, 336–344. [Google Scholar] [CrossRef]
- Balić, A.; Vlašić, D.; Žužul, K.; Marinović, B.; Bukvić Mokos, Z. Omega-3 Versus Omega-6 Polyunsaturated Fatty Acids in the Prevention and Treatment of Inflammatory Skin Diseases. Int. J. Mol. Sci. 2020, 21, 741. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, G.; Ecker, J. The opposing effects of n-3 and n-6 fatty acids. Prog. Lipid Res. 2008, 47, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Muhlhausler, B.S.; Cook-Johnson, R.; James, M.; Miljkovic, D.; Duthoit, E.; Gibson, R. Opposing effects of omega-3 and omega-6 long chain polyunsaturated Fatty acids on the expression of lipogenic genes in omental and retroperitoneal adipose depots in the rat. J. Nutr. Metab. 2010, 2010, 927836. [Google Scholar] [CrossRef] [PubMed]
- Dennis, E.A.; Norris, P.C. Eicosanoid storm in infection and inflammation. Nat. Rev. Immunol. 2015, 15, 511–523. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, A.; Botta, E.; Holinstat, M. Eicosanoids in inflammation in the blood and the vessel. Front. Pharm. 2022, 13, 997403. [Google Scholar] [CrossRef] [PubMed]
- Sheppe, A.E.F.; Edelmann, M.J. Roles of Eicosanoids in Regulating Inflammation and Neutrophil Migration as an Innate Host Response to Bacterial Infections. Infect. Immun. 2021, 89, e0009521. [Google Scholar] [CrossRef]
- Mendoza, S.R.; Zamith-Miranda, D.; Takacs, T.; Gacser, A.; Nosanchuk, J.D.; Guimaraes, A.J. Complex and Controversial Roles of Eicosanoids in Fungal Pathogenesis. J. Fungi 2021, 7, 254. [Google Scholar] [CrossRef]
- Artru, F.; McPhail, M.J.W.; Triantafyllou, E.; Trovato, F.M. Lipids in Liver Failure Syndromes: A Focus on Eicosanoids, Specialized Pro-Resolving Lipid Mediators and Lysophospholipids. Front. Immunol. 2022, 13, 867261. [Google Scholar] [CrossRef]
- Buczynski, M.W.; Dumlao, D.S.; Dennis, E.A. Thematic Review Series: Proteomics. An integrated omics analysis of eicosanoid biology. J. Lipid Res. 2009, 50, 1015–1038. [Google Scholar] [CrossRef]
- Kurzrok, R.; Lieb, C.C. Biochemical Studies of Human Semen. II. The Action of Semen on the Human Uterus. Proc. Soc. Exp. Biol. Med. 1930, 28, 268–272. [Google Scholar] [CrossRef]
- Fitzpatrick, F.A.; Ennis, M.D.; Baze, M.E.; Wynalda, M.A.; McGee, J.E.; Liggett, W.F. Inhibition of cyclooxygenase activity and platelet aggregation by epoxyeicosatrienoic acids. Influence of stereochemistry. J. Biol. Chem. 1986, 261, 15334–15338. [Google Scholar] [CrossRef] [PubMed]
- Vane, J.R. Inhibition of Prostaglandin Synthesis as a Mechanism of Action for Aspirin-Like Drugs. Nat.-New Biol. 1971, 231, 232. [Google Scholar] [CrossRef] [PubMed]
- Breyer, R.M.; Bagdassarian, C.K.; Myers, S.A.; Breyer, M.D. Prostanoid receptors: Subtypes and signaling. Annu. Rev. Pharm. Toxicol 2001, 41, 661–690. [Google Scholar] [CrossRef] [PubMed]
- Capra, V.; Thompson, M.D.; Sala, A.; Cole, D.E.; Folco, G.; Rovati, G.E. Cysteinyl-leukotrienes and their receptors in asthma and other inflammatory diseases: Critical update and emerging trends. Med. Res. Rev. 2007, 27, 469–527. [Google Scholar] [CrossRef]
- Cheng, H.F.; Harris, R.C. Cyclooxygenases, the kidney, and hypertension. Hypertension 2004, 43, 525–530. [Google Scholar] [CrossRef]
- Hao, C.M.; Breyer, M.D. Physiological regulation of prostaglandins in the kidney. Annu. Rev. Physiol. 2008, 70, 357–377. [Google Scholar] [CrossRef]
- Ribeiro, J.D.; Toro, A.A.; Baracat, E.C. Antileukotrienes in the treatment of asthma and allergic rhinitis. J. Pediatr. 2006, 82, S213–S221. [Google Scholar] [CrossRef]
- Sharma, J.N.; Mohammed, L.A. The role of leukotrienes in the pathophysiology of inflammatory disorders: Is there a case for revisiting leukotrienes as therapeutic targets? Inflammopharmacology 2006, 14, 10–16. [Google Scholar] [CrossRef]
- Bedossa, P. Pathology of non-alcoholic fatty liver disease. Liver Int. 2017, 37, 85–89. [Google Scholar] [CrossRef]
- Teli, M.R.; James, O.F.; Burt, A.D.; Bennett, M.K.; Day, C.P. The natural history of nonalcoholic fatty liver: A follow-up study. Hepatology 1995, 22, 1714–1719. [Google Scholar] [CrossRef]
- Zoller, H.; Tilg, H. Nonalcoholic fatty liver disease and hepatocellular carcinoma. Metabolism 2016, 65, 1151–1160. [Google Scholar] [CrossRef] [PubMed]
- Cotter, T.G.; Rinella, M. Nonalcoholic Fatty Liver Disease 2020: The State of the Disease. Gastroenterology 2020, 158, 1851–1864. [Google Scholar] [CrossRef] [PubMed]
- Golabi, P.; Paik, J.; Hwang, J.P.; Wang, S.; Lee, H.M.; Younossi, Z.M. Prevalence and outcomes of non-alcoholic fatty liver disease (NAFLD) among Asian American adults in the United States. Liver Int. 2019, 39, 748–757. [Google Scholar] [CrossRef] [PubMed]
- Younossi, Z.M.; Koenig, A.B.; Abdelatif, D.; Fazel, Y.; Henry, L.; Wymer, M. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 2016, 64, 73–84. [Google Scholar] [CrossRef]
- Bataller, R.; Brenner, D.A. Liver fibrosis. J. Clin. Investig. 2005, 115, 209–218. [Google Scholar] [CrossRef]
- Svegliati-Baroni, G.; Pierantonelli, I.; Torquato, P.; Marinelli, R.; Ferreri, C.; Chatgilialoglu, C.; Bartolini, D.; Galli, F. Lipidomic biomarkers and mechanisms of lipotoxicity in non-alcoholic fatty liver disease. Free Radic. Biol. Med. 2019, 144, 293–309. [Google Scholar] [CrossRef]
- Banaszczak, M.; Maciejewska, D.; Drozd, A.; Ryterska, K.; Milc, D.J.; Raszeja-Wyszomirska, J.; Wunsch, E.; Gonzalez-Muniesa, P.; Stachowska, E. 5-Lipooxygenase Derivatives as Serum Biomarkers of a Successful Dietary Intervention in Patients with NonAlcoholic Fatty Liver Disease. Medicina 2020, 56, 58. [Google Scholar] [CrossRef]
- Kalveram, L.; Schunck, W.H.; Rothe, M.; Rudolph, B.; Loddenkemper, C.; Holzhutter, H.G.; Henning, S.; Bufler, P.; Schulz, M.; Meierhofer, D.; et al. Regulation of the cytochrome P450 epoxyeicosanoid pathway is associated with distinct histologic features in pediatric non-alcoholic fatty liver disease. Prostaglandins Leukot. Essent. Fat. Acids 2021, 164, 102229. [Google Scholar] [CrossRef]
- Arvind, A.; Osganian, S.A.; Sjoquist, J.A.; Corey, K.E.; Simon, T.G. Epoxygenase-Derived Epoxyeicosatrienoic Acid Mediators Are Associated With Nonalcoholic Fatty Liver Disease, Nonalcoholic Steatohepatitis, and Fibrosis. Gastroenterology 2020, 159, 2232–2234.e2234. [Google Scholar] [CrossRef]
- Marchix, J.; Catheline, D.; Duby, C.; Monthean-Boulier, N.; Boissel, F.; Pedrono, F.; Boudry, G.; Legrand, P. Interactive effects of maternal and weaning high linoleic acid intake on hepatic lipid metabolism, oxylipins profile and hepatic steatosis in offspring. J. Nutr. Biochem. 2020, 75, 108241. [Google Scholar] [CrossRef]
- Chalasani, N.; Younossi, Z.; Lavine, J.E.; Diehl, A.M.; Brunt, E.M.; Cusi, K.; Charlton, M.; Sanyal, A.J. The Diagnosis and Management of Non-alcoholic Fatty Liver Disease: Practice Guideline by the American Gastroenterological Association, American Association for the Study of Liver Diseases, and American College of Gastroenterology. Gastroenterology 2012, 142, 1592–1609. [Google Scholar] [CrossRef] [PubMed]
- Dyson, J.K.; Anstee, Q.M.; McPherson, S. Non-alcoholic fatty liver disease: A practical approach to diagnosis and staging. Frontline Gastroenterol. 2014, 5, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Di Mauro, S.; Scamporrino, A.; Filippello, A.; Di Pino, A.; Scicali, R.; Malaguarnera, R.; Purrello, F.; Piro, S. Clinical and Molecular Biomarkers for Diagnosis and Staging of NAFLD. Int. J. Mol. Sci. 2021, 22, 11905. [Google Scholar] [CrossRef] [PubMed]
- Maciejewska, D.; Drozd, A.; Skonieczna-Zydecka, K.; Skorka-Majewicz, M.; Dec, K.; Jakubczyk, K.; Pilutin, A.; Stachowska, E. Eicosanoids in Nonalcoholic Fatty Liver Disease (NAFLD) Progression. Do Serum Eicosanoids Profile Correspond with Liver Eicosanoids Content during NAFLD Development and Progression? Molecules 2020, 25, 2026. [Google Scholar] [CrossRef] [PubMed]
- Camacho-Munoz, D.; Kiezel-Tsugunova, M.; Kiss, O.; Uddin, M.; Sunden, M.; Ryaboshapkina, M.; Lind, L.; Oscarsson, J.; Nicolaou, A. Omega-3 carboxylic acids and fenofibrate differentially alter plasma lipid mediators in patients with non-alcoholic fatty liver disease. FASEB J. 2021, 35, e21976. [Google Scholar] [CrossRef]
- Deng, X.; Ke, X.; Tang, Y.; Luo, W.; Dong, R.; Ge, D.; Han, L.; Yang, Y.; Liu, H.; Reyila, T.; et al. Sagittaria sagittifolia polysaccharide interferes with arachidonic acid metabolism in non-alcoholic fatty liver disease mice via Nrf2/HO-1 signaling pathway. Biomed. Pharm. 2020, 132, 110806. [Google Scholar] [CrossRef]
- Yoshino, K.; Hosooka, T.; Shinohara, M.; Aoki, C.; Hosokawa, Y.; Imamori, M.; Ogawa, W. Canagliflozin ameliorates hepatic fat deposition in obese diabetic mice: Role of prostaglandin E-2. Biochem. Biophys. Res. Commun. 2021, 557, 62–68. [Google Scholar] [CrossRef]
- Tuomisto, K.; Palmu, J.; Long, T.; Watrous, J.D.; Mercader, K.; Lagerborg, K.A.; Andres, A.; Salmi, M.; Jalkanen, S.; Vasan, R.S.; et al. A plasma metabolite score of three eicosanoids predicts incident type 2 diabetes: A prospective study in three independent cohorts. BMJ Open Diabetes Res. Care 2022, 10, e002519. [Google Scholar] [CrossRef]
- Miao, Z.J.; Tang, X.; Schultzberg, M.; Zhao, Y.W.; Wang, X.Z. Plasma Resolvin D2 to Leukotriene B-4 Ratio Is Reduced in Diabetic Patients with Ischemic Stroke and Related to Prognosis. Biomed. Res. Int. 2021, 2021, 6657646. [Google Scholar] [CrossRef]
- Tans, R.; Bande, R.; van Rooij, A.; Molloy, B.J.; Stienstra, R.; Tack, C.J.; Wevers, R.A.; Wessels, H.; Gloerich, J.; van Gool, A.J. Evaluation of cyclooxygenase oxylipins as potential biomarker for obesity-associated adipose tissue inflammation and type 2 diabetes using targeted multiple reaction monitoring mass spectrometry. Prostaglandins Leukot. Essent. Fat. Acids 2020, 160, 102157. [Google Scholar] [CrossRef]
- Pawelzik, S.C.; Avignon, A.; Idborg, H.; Boegner, C.; Stanke-Labesque, F.; Jakobsson, P.J.; Sultan, A.; Back, M. Urinary prostaglandin D2 and E2 metabolites associate with abdominal obesity, glucose metabolism, and triglycerides in obese subjects. Prostaglandins Other Lipid Mediat. 2019, 145, 106361. [Google Scholar] [CrossRef] [PubMed]
- Truchan, N.A.; Fenske, R.J.; Sandhu, H.K.; Weeks, A.M.; Patibandla, C.; Wancewicz, B.; Pabich, S.; Reuter, A.; Harrington, J.M.; Brill, A.L.; et al. Human Islet Expression Levels of Prostaglandin E2 Synthetic Enzymes, But Not Prostaglandin EP3 Receptor, Are Positively Correlated with Markers of beta-Cell Function and Mass in Nondiabetic Obesity. ACS Pharm. Transl. Sci. 2021, 4, 1338–1348. [Google Scholar] [CrossRef] [PubMed]
- Grapov, D.; Fiehn, O.; Campbell, C.; Chandler, C.J.; Burnett, D.J.; Souza, E.C.; Casazza, G.A.; Keim, N.L.; Hunter, G.R.; Fernandez, J.R.; et al. Impact of a weight loss and fitness intervention on exercise-associated plasma oxylipin patterns in obese, insulin-resistant, sedentary women. Physiol. Rep. 2020, 8, e14547. [Google Scholar] [CrossRef] [PubMed]
- Heemskerk, M.M.; Giera, M.; el Bouazzaoui, F.; Lips, M.A.; Pijl, H.; van Dijk, K.W.; van Harmelen, V. Increased PUFA Content and 5-Lipoxygenase Pathway Expression Are Associated with Subcutaneous Adipose Tissue Inflammation in Obese Women with Type 2 Diabetes. Nutrients 2015, 7, 7676–7690. [Google Scholar] [CrossRef] [PubMed]
- Fisk, H.L.; Childs, C.E.; Miles, E.A.; Ayres, R.; Noakes, P.S.; Paras-Chavez, C.; Kuda, O.; Kopecky, J.; Antoun, E.; Lillycrop, K.A.; et al. Modification of subcutaneous white adipose tissue inflammation by omega-3 fatty acids is limited in human obesity-a double blind, randomised clinical trial. EBioMedicine 2022, 77, 103909. [Google Scholar] [CrossRef]
- Sanchez-Fernandez, A.; Zandee, S.; Mastrogiovanni, M.; Charabati, M.; Rubbo, H.; Prat, A.; Lopez-Vales, R. Administration of Maresin-1 ameliorates the physiopathology of experimental autoimmune encephalomyelitis. J. Neuroinflammation 2022, 19, 27. [Google Scholar] [CrossRef]
- Dieckmann, S.; Maurer, S.; Fromme, T.; Colson, C.; Virtanen, K.A.; Amri, E.Z.; Klingenspor, M. Fatty Acid Metabolite Profiling Reveals Oxylipins as Markers of Brown but Not Brite Adipose Tissue. Front. Endocrinol. 2020, 11, 73. [Google Scholar] [CrossRef]
- Zhang, Z.Y.; Huang, S.S.; Wu, S.F.; Qi, J.J.; Li, W.C.; Liu, S.S.; Cong, Y.; Chen, H.W.; Lu, L.W.; Shi, S.T.; et al. Clearance of apoptotic cells by mesenchymal stem cells contributes to immunosuppression via PGE2. Ebiomedicine 2019, 45, 341–350. [Google Scholar] [CrossRef]
- Wallace, J.L. Eicosanoids in the gastrointestinal tract. Br. J. Pharm. 2019, 176, 1000–1008. [Google Scholar] [CrossRef]
- Tsiantas, K.; Konteles, S.J.; Kritsi, E.; Sinanoglou, V.J.; Tsiaka, T.; Zoumpoulakis, P. Effects of Non-Polar Dietary and Endogenous Lipids on Gut Microbiota Alterations: The Role of Lipidomics. Int. J. Mol. Sci. 2022, 23, 4070. [Google Scholar] [CrossRef]
- Avila-Roman, J.; Arreaza-Gil, V.; Cortes-Espinar, A.J.; Soliz-Rueda, J.R.; Mulero, M.; Muguerza, B.; Arola-Arnal, A.; Arola, L.; Torres-Fuentes, C. Impact of gut microbiota on plasma oxylipins profile under healthy and obesogenic conditions. Clin. Nutr. 2021, 40, 1475–1486. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Oh, Y.T.; Wan, D.; Watanabe, R.M.; Hammock, B.D.; Youn, J.H. Postprandial effect to decrease soluble epoxide hydrolase activity: Roles of insulin and gut microbiota. J. Nutr. Biochem. 2017, 49, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Crittenden, S.; Goepp, M.; Pollock, J.; Robb, C.T.; Smyth, D.J.; Zhou, Y.; Andrews, R.; Tyrrell, V.; Gkikas, K.; Adima, A.; et al. Prostaglandin E2 promotes intestinal inflammation via inhibiting microbiota-dependent regulatory T cells. Sci. Adv. 2021, 7, eabd7954. [Google Scholar] [CrossRef] [PubMed]
- Chistyakov, D.V.; Gavrish, G.E.; Goriainov, S.V.; Chistyakov, V.V.; Astakhova, A.A.; Azbukina, N.V.; Sergeeva, M.G. Oxylipin Profiles as Functional Characteristics of Acute Inflammatory Responses in Astrocytes Pre-Treated with IL-4, IL-10, or LPS. Int. J. Mol. Sci. 2020, 21, 780. [Google Scholar] [CrossRef] [PubMed]
- Chistyakov, D.V.; Astakhova, A.A.; Goriainov, S.V.; Sergeeva, M.G. Comparison of PPAR Ligands as Modulators of Resolution of Inflammation, via Their Influence on Cytokines and Oxylipins Release in Astrocytes. Int. J. Mol. Sci. 2020, 21, 9577. [Google Scholar] [CrossRef]
- Na, Y.R.; Jung, D.; Stakenborg, M.; Jang, H.; Gu, G.J.; Jeong, M.R.; Suh, S.Y.; Kim, H.J.; Kwon, Y.H.; Sung, T.S.; et al. Prostaglandin E2 receptor PTGER4-expressing macrophages promote intestinal epithelial barrier regeneration upon inflammation. Gut 2021, 70, 2249–2260. [Google Scholar] [CrossRef]
- Hinz, C.; Liggi, S.; Mocciaro, G.; Jung, S.; Induruwa, I.; Pereira, M.; Bryant, C.E.; Meckelmann, S.W.; O′Donnell, V.B.; Farndale, R.W.; et al. A Comprehensive UHPLC Ion Mobility Quadrupole Time-of-Flight Method for Profiling and Quantification of Eicosanoids, Other Oxylipins, and Fatty Acids. Anal. Chem. 2019, 91, 8025–8035. [Google Scholar] [CrossRef]
- Mabud, M.D.A.; Dekrey, M.J.; Cooks, R.G. Surface-Induced Dissociation of Molecular-Ions. Int. J. Mass Spectrom. 1985, 67, 285–294. [Google Scholar] [CrossRef]
- Syka, J.E.; Coon, J.J.; Schroeder, M.J.; Shabanowitz, J.; Hunt, D.F. Peptide and protein sequence analysis by electron transfer dissociation mass spectrometry. Proc. Natl. Acad. Sci. USA 2004, 101, 9528–9533. [Google Scholar] [CrossRef]
- Zubarev, R.A.; Horn, D.M.; Fridriksson, E.K.; Kelleher, N.L.; Kruger, N.A.; Lewis, M.A.; Carpenter, B.K.; McLafferty, F.W. Electron capture dissociation for structural characterization of multiply charged protein cations. Anal. Chem. 2000, 72, 563–573. [Google Scholar] [CrossRef]
- Chait, B.T. Mass spectrometry in the postgenomic era. Annu. Rev. Biochem. 2011, 80, 239–246. [Google Scholar] [CrossRef] [PubMed]
- Kind, T.; Fiehn, O. Advances in structure elucidation of small molecules using mass spectrometry. Bioanal. Rev. 2010, 2, 23–60. [Google Scholar] [CrossRef] [PubMed]
- McCullagh JSO, O.N. Mass Spectrometry; Oxford University Press: Oxford, UK, 2019. [Google Scholar]
- Mann, M.; Hendrickson, R.C.; Pandey, A. Analysis of proteins and proteomes by mass spectrometry. Annu. Rev. Biochem. 2001, 70, 437–473. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.Y.; Zhang, Q.B. Comprehensive analysis of oxylipins in human plasma using reversed-phase liquid chromatography-triple quadrupole mass spectrometry with heatmap-assisted selection of transitions. Anal. Bioanal. Chem. 2019, 411, 367–385. [Google Scholar] [CrossRef] [PubMed]
- Theodoridis, G.A.; Gika, H.G.; Want, E.J.; Wilson, I.D. Liquid chromatography-mass spectrometry based global metabolite profiling: A review. Anal. Chim. Acta 2012, 711, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Molnar, I.; Horvath, C. Reverse-phase chromatography of polar biological substances: Separation of catechol compounds by high-performance liquid chromatography. Clin. Chem. 1976, 22, 1497–1502. [Google Scholar] [CrossRef]
- Kanu, A.B.; Dwivedi, P.; Tam, M.; Matz, L.; Hill, H.H., Jr. Ion mobility-mass spectrometry. J. Mass Spectrom. 2008, 43, 1–22. [Google Scholar] [CrossRef]
- Lanucara, F.; Holman, S.W.; Gray, C.J.; Eyers, C.E. The power of ion mobility-mass spectrometry for structural characterization and the study of conformational dynamics. Nat. Chem. 2014, 6, 281–294. [Google Scholar] [CrossRef]
- Hill, H.H., Jr.; Siems, W.F.; St Louis, R.H.; McMinn, D.G. Ion mobility spectrometry. Anal. Chem. 1990, 62, 1201A–1209A. [Google Scholar] [CrossRef]
- Dodds, J.N.; Baker, E.S. Ion Mobility Spectrometry: Fundamental Concepts, Instrumentation, Applications, and the Road Ahead. J. Am. Soc. Mass Spectrom. 2019, 30, 2185–2195. [Google Scholar] [CrossRef]
- Paglia, G.; Smith, A.J.; Astarita, G. Ion mobility mass spectrometry in the omics era: Challenges and opportunities for metabolomics and lipidomics. Mass Spectrom. Rev. 2022, 41, 722–765. [Google Scholar] [CrossRef] [PubMed]
- Harris, R.A.; Leaptrot, K.L.; May, J.C.; McLean, J.A. New Frontiers in Lipidomics Analyses using Structurally Selective Ion Mobility-Mass Spectrometry. Trends Anal. Chem. 2019, 116, 316–323. [Google Scholar] [CrossRef] [PubMed]
- Domalain, V.; Tognetti, V.; Hubert-Roux, M.; Lange, C.M.; Joubert, L.; Baudoux, J.; Rouden, J.; Afonso, C. Role of cationization and multimers formation for diastereomers differentiation by ion mobility-mass spectrometry. J. Am. Soc. Mass Spectrom. 2013, 24, 1437–1445. [Google Scholar] [CrossRef] [PubMed]
- Stauber, J.; MacAleese, L.; Franck, J.; Claude, E.; Snel, M.; Kaletas, B.K.; Wiel, I.M.; Wisztorski, M.; Fournier, I.; Heeren, R.M. On-tissue protein identification and imaging by MALDI-ion mobility mass spectrometry. J. Am. Soc. Mass Spectrom. 2010, 21, 338–347. [Google Scholar] [CrossRef] [PubMed]
- Jurneczko, E.; Kalapothakis, J.; Campuzano, I.D.; Morris, M.; Barran, P.E. Effects of drift gas on collision cross sections of a protein standard in linear drift tube and traveling wave ion mobility mass spectrometry. Anal. Chem. 2012, 84, 8524–8531. [Google Scholar] [CrossRef] [PubMed]
- Bouza, M.; Li, Y.; Wang, A.C.; Wang, Z.L.; Fernandez, F.M. Triboelectric Nanogenerator Ion Mobility-Mass Spectrometry for In-Depth Lipid Annotation. Anal. Chem. 2021, 93, 5468–5475. [Google Scholar] [CrossRef]
- Fenn, L.S.; Kliman, M.; Mahsut, A.; Zhao, S.R.; McLean, J.A. Characterizing ion mobility-mass spectrometry conformation space for the analysis of complex biological samples. Anal. Bioanal. Chem. 2009, 394, 235–244. [Google Scholar] [CrossRef]
- Fenn, L.S.; McLean, J.A. Biomolecular structural separations by ion mobility-mass spectrometry. Anal. Bioanal. Chem. 2008, 391, 905–909. [Google Scholar] [CrossRef]
- Kliman, M.; May, J.C.; McLean, J.A. Lipid analysis and lipidomics by structurally selective ion mobility-mass spectrometry. Biochim. Biophys. Acta 2011, 1811, 935–945. [Google Scholar] [CrossRef]
- Hellhake, S.; Meckelmann, S.W.; Empl, M.T.; Rentmeister, K.; Wissdorf, W.; Steinberg, P.; Schmitz, O.J.; Benter, T.; Schebb, N.H. Non-targeted and targeted analysis of oxylipins in combination with charge-switch derivatization by ion mobility high-resolution mass spectrometry. Anal. Bioanal. Chem. 2020, 412, 5743–5757. [Google Scholar] [CrossRef]
- Chollet, C.; Boutet-Mercey, S.; Laboureur, L.; Rincon, C.; Mejean, M.; Jouhet, J.; Fenaille, F.; Colsch, B.; Touboul, D. Supercritical fluid chromatography coupled to mass spectrometry for lipidomics. J. Mass Spectrom. 2019, 54, 791–801. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Liang, Y.; Yang, J.; Ye, F.; Zhou, T.; Gongke, L. Advances of supercritical fluid chromatography in lipid profiling. J. Pharm. Anal. 2019, 9, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Taylor, L.T. Supercritical fluid chromatography. Anal. Chem. 2010, 82, 4925–4935. [Google Scholar] [CrossRef] [PubMed]
- Saito, M. History of supercritical fluid chromatography: Instrumental development. J. Biosci. Bioeng. 2013, 115, 590–599. [Google Scholar] [CrossRef]
- Kalikova, K.; Slechtova, T.; Vozka, J.; Tesarova, E. Supercritical fluid chromatography as a tool for enantioselective separation; A review. Anal. Chim. Acta 2014, 821, 1–33. [Google Scholar] [CrossRef]
- Jin, W.; Yang, J.; Liu, D.; Zhong, Q.; Zhou, T. Determination of inflammation-related lipids in depressive rats by on-line supercritical fluid extraction-supercritical fluid chromatography-tandem mass spectrometry. J. Pharm. Biomed. Anal. 2021, 203, 114210. [Google Scholar] [CrossRef]
- Kumari, A.U.S.J.; Acharya, S.R.; Bergquist, J. A novel, fast and sensitive supercritical fluid chromatography-tandem mass spectrometry (SFC-MS/MS) method for analysis of arachidonic acid metabolites. Analyst 2018, 143, 3661–3669. [Google Scholar] [CrossRef]
- Qi, K.; Wu, L.; Liu, C.; Pan, Y. Recent Advances of Ambient Mass Spectrometry Imaging and Its Applications in Lipid and Metabolite Analysis. Metabolites 2021, 11, 780. [Google Scholar] [CrossRef]
- Su, H.; Jiang, Z.H.; Chiou, S.F.; Shiea, J.; Wu, D.C.; Tseng, S.P.; Jain, S.H.; Chang, C.Y.; Lu, P.L. Rapid Characterization of Bacterial Lipids with Ambient Ionization Mass Spectrometry for Species Differentiation. Molecules 2022, 27, 2772. [Google Scholar] [CrossRef]
- Bowman, A.P.; Heeren, R.M.A.; Ellis, S.R. Advances in mass spectrometry imaging enabling observation of localised lipid biochemistry within tissues. TrAC Trends Anal. Chem. 2019, 120, 115197. [Google Scholar] [CrossRef]
- Passarelli, M.K.; Winograd, N. Lipid imaging with time-of-flight secondary ion mass spectrometry (ToF-SIMS). Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2011, 1811, 976–990. [Google Scholar] [CrossRef] [PubMed]
- Takats, Z.; Wiseman, J.M.; Gologan, B.; Cooks, R.G. Mass spectrometry sampling under ambient conditions with desorption electrospray ionization. Science 2004, 306, 471–473. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Qi, F.; Pan, Y. Imaging of Polar and Nonpolar Lipids Using Desorption Electrospray Ionization/Post-photoionization Mass Spectrometry. Methods Mol. Biol. 2021, 2306, 285–298. [Google Scholar] [CrossRef] [PubMed]
- Bowman, A.P.; Bogie, J.F.J.; Hendriks, J.J.A.; Haidar, M.; Belov, M.; Heeren, R.M.A.; Ellis, S.R. Evaluation of lipid coverage and high spatial resolution MALDI-imaging capabilities of oversampling combined with laser post-ionisation. Anal. Bioanal. Chem. 2020, 412, 2277–2289. [Google Scholar] [CrossRef] [PubMed]
- Coras, R.; Kavanaugh, A.; Kluzniak, A.; Holt, D.; Weilgosz, A.; Aaron, A.; Quehenberger, O.; Ritchlin, C.; Guma, M. Differences in oxylipin profile in psoriasis versus psoriatic arthritis. Arthritis Res. 2021, 23, 200. [Google Scholar] [CrossRef]
- Bansal, K.; Narayana, Y.; Patil, S.A.; Balaji, K.N.M. bovis BCG induced expression of COX-2 involves nitric oxide-dependent and -independent signaling pathways. J. Leukoc. Biol. 2009, 85, 804–816. [Google Scholar] [CrossRef]
- Eckmann, L.; Stenson, W.F.; Savidge, T.C.; Lowe, D.C.; Barrett, K.E.; Fierer, J.; Smith, J.R.; Kagnoff, M.F. Role of intestinal epithelial cells in the host secretory response to infection by invasive bacteria. Bacterial entry induces epithelial prostaglandin h synthase-2 expression and prostaglandin E2 and F2alpha production. J. Clin. Investig. 1997, 100, 296–309. [Google Scholar] [CrossRef]
- Teng, O.; Ang, C.K.E.; Guan, X.L. Macrophage–Bacteria Interactions—A Lipid-Centric Relationship. Front. Immunol. 2017, 8, 1836. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).