Abstract
During the last decades, the discovery of nanolime and its introduction in the field of Cultural Heritage has entailed a significant advance for the consolidation of historic wall paintings. Nevertheless, its use is not completely generalized yet within the conservation practitioner’s community due to its undesired white veiling deposit on the surface after treatment which usually covers the pictorial layer. Given the scarcity of existing literature which specify how to mitigate this undesired side-effect, the aim of this work is to carry out the first assessment of possible cleaning and treatment methods to eliminate those deposits and, at the same time, to analyse their effects on the consolidation properties. To do that, we have developed laboratory fresco wall paintings specimens. After applying an artificial ageing cycle to all of them, we consolidated them with CaloSil IP25, one of the most currently used commercial product in the practice. The elimination of the whitish deposits has been investigated with different methods: (1) wiping off the superficial excess of product immediately after each application during the treatment; (2) mechanical cleaning (scalpel) after consolidation; chemical cleaning with rigid gels of water (3), ethylenediaminetetraacetic acid (EDTA) (4) and citric acid (5) after consolidation. The effectiveness of each cleaning method has been assessed by non-destructive methods before and after the consolidation, and repeated after cleaning, by measuring differences in colour, water absorption by capillarity (i.e., sponge test), superficial adhesion (i.e., Scotch Tape Test) and by quantifying the thickness of the deposits eliminated by means of photogrammetry. Results show that both the mechanical cleaning and the removal of the excess of product after each application during the treatment yielded the most promising results for conservation practice.
1. Introduction
Consolidation is one of the most important tasks when it comes to intervening in a historic wall painting since it restores the cohesion of both the supporting material and the pictorial film, favouring its conservation. However, this consolidation presents several challenges [1,2,3]: (1) the treatment must have certain penetration; (2) it must fix the pictorial layer, but it must not cause a loss of permeability in the surface and (3) must not modify the colour or the aesthetics characteristics of the substrate. In this light, nanolime treatments have recently emerged as a very promising option for wall-paintings conservation due to: (1) their high compatibility with the constitutive material of the artwork, not generating an impermeable layer on the surface, in comparison with other consolidating products used in the conservation field such as acrylic or tetra-ethoxysilane (TEOS)-based consolidants [4]; (2) their potentially high penetration depth due to the nanometric size of the particles compared to traditional limewater method [5]; and (3) the high reactivity of their particles which accelerates carbonation [5,6,7,8]. Since their discovery in the early 2000s by the University of Florence-CSGI (Florence, Italy), the effectiveness of nanolimes has been tested specially in wall paintings, obtaining successful results in terms of superficial cohesion recovery, reduction of water absorption by capillarity and increase of the mechanical properties of the pictorial layer and support [7,9,10,11,12,13]. However, despite the significant increase in superficial cohesion, the consolidation usually generates whitish deposits on the surface which frequently covers the pictorial layer, modifying its aspect, colour and interpretation of the historical artwork [5,14,15]. Such a “veiling” or white “glazing” is highly detrimental and there is a strong need for research aimed at preventing its appearance.
Whereas this side-effect is recurrently mentioned in most of scientific articles concerning nanolime consolidation treatments [6,7,8,9,10,11,12,13,14,15], and it is mentioned in the information technical data sheet provided by both nanolime distributors (e.g., Calosil® and Nanorestore®) [16,17], there is no literature or scientific evidence which clearly specify neither the most convenient application method to avoid the generation of those glazings without altering the consolidating properties, nor the possible subsequent treatments to remove them. Following the technical recommendations provided by both nanolime commercially available brands (Calosil® and Nanorestore®), it is also not clear which guidelines should be followed: whereas Nanorestore suggests washing the pictorial layer after each application to remove the excess of product remaining [16], Calosil suggests the possibility of diluting the consolidating product to obtain lower concentrations, as a way to prevent the appearance of superficial glazings [17].
Therefore, this study aims to assess, the different cleaning methods and treatments for the elimination of these deposits The elimination of the whitish deposits has been investigated with different methods: (1) removal of the superficial excess of product immediately after each application during the treatment; (2) mechanical cleaning after consolidation; (3) chemical cleaning with rigid gels with water after consolidation; (4) ethylenediaminetetraacetic acid (EDTA) and (5) citric acid.
2. Materials and Methods
2.1. Wall Painting Mortar Specimens Preparation
The tests were carried out on laboratory-developed wall painting specimens, which were produced with a volumetric binder: sand ratio, layer thickness and pigments following the traditional roman recipes and materials (Pliny y Vitruvius) [18,19]. The use of these laboratory-developed wall painting specimens instead of decontextualised fragments of a real artworks was intentionally decided since they are more homogeneous, thus facilitating the assessment of the cleaning treatments. Besides, such mock-ups allow the use of certain assessing techniques which might cause a deterioration of the pictorial layer, which is prohibitive in the case real artworks.
A total of ten wall paintings specimens were created, with 7 cm diameter and 1 cm height, simulating the most superficial layers of a traditional Roman wall painting (Figure 1). Wall painting specimens have two layers of mortar, presenting a lower thickness and finer granulometry in the most superficial layer, as indicated in the classic recipe of Pliny and Vitruvius [18,19]:

Figure 1.
Laboratory developed wall painting specimens prior the consolidation.
- The first “stratum”, corresponding to the intonaco, has an approximate thickness of 0.7cm. It is composed of a 1:3 proportion of lime (lime putty, CTS commercial brand), aggregate (silica-based aggregate with diameter of 0.4–0.8 mm and marble powder (magnesium calcium carbonate) in 2:1 proportion); this proportion of aggregate and binder has been used because it is the one that has been identified in different examples of roman wall painting [20].
- The second “stratum”, corresponding to the intonachino, presented an approximate thickness of 0.3 cm. It was composed of a 1:2 proportion of lime and aggregate, being the latter a mixture of silica sand (diameter of 0.4–0.8 mm) and marble powder (magnesium calcium carbonate) in 1:3 proportion; this proportion of aggregate and binder has been used because it is the one identified in different roman wall paintings [20].
Mortars were batched by volume following the traditional protocol consisting of mixing the dry sand with lime putty in a bowl, and stirring with a spatula until the obtention of a homogeneous mortar paste. The pictorial layer was executed following the traditional fresco technique, mixing the pigment with water and applying it by brush on the last “stratum” of the mortar, still wet. In this case, the pigment used was yellow ochre (Yellow Ochre from Kremer Pigment, Aichstetten, Germany), composed by iron oxide-hydroxide, Fe2O3 + Fe(OH)3/FeOOH. This pigment was chosen as it is one of the most common tones in the Roman palette. Samples were cast in 70 mm diameter × 10 mm height moulds. After casting, fresh mortars were transferred to a laboratory room with controlled temperature and humidity maintained at 20 °C (±10 °C) and 65% relative humidity (RH) (±10% RH) for a curing period of 28 days.
2.2. Weathering Process of Wall Painting Specimens
After curing, wall painting mortar specimens were artificially weathered in order to generate decay on the samples prior to the consolidation. The ageing process consisted of several cycles of thermal weathering, following the protocol developed by Franzoni et al. [21] and tested by Sena da Fonseca [22]. This artificial weathering method has been reported to induce controllable microstructural, physical and mechanical alterations, tested especially in lime-based substrates [22]. However, since temperatures higher than 200 °C can induce changes on the iron oxide-hydroxide (Fe2O3 + Fe(OH)3/FeOOH) altering its aesthetic appearance (from ochre to red), Franzoni’s test was adapted to lower temperatures. The induced damage was monitored between each cycle by means of Ultra-sounds Pulse Velocity (P-wave) to ascertain a noticeable degree of degradation. The ageing protocol consisted of the following steps:
- Specimens were dried until they reached constant mass at 70 °C (24 h) and left to cool at room temperature in a desiccator. We recorded the initial weight.
- Specimens were heated in a pre-heated oven at 100 °C for 1 h. After that, they were left for 2 h cooling at room temperature (in a desiccator with silica gel).
- Specimens were heated in a pre-heated oven at 200 °C for 1 h, followed by 2 h cooling in room condition (in a desiccator with silica gel).
- Specimens were completely immersed in deionized water until full saturation for 2 h and samples were heated in a pre-heated oven at 200 °C for 1 h, followed by 2 h cooling at room temperature (in a desiccator with silica gel).
This sequence was repeated three times and was stopped when no more decay was noticed by measuring differences in the P-wave velocity, (m/s) which decreased from 828 ± 21.5 m/s to 656.21 ± 16.1.5 m/s.
2.3. Nanolime Treatment Application
Calosil IP25® (by IBZ Salzchemie GmbH & Co.KG, Halsbrücke, Germany, containing 25 g/L of Ca(OH)2 nanoparticles in isopropanol) was used for this experiment. This product was intentionally selected since it is considered one of the most popular products used in conservation practice. Calosil IP25 was diluted to 12.5 g/L to increase penetrability [23,24], as commonly carried out in practice activities. Before the treatment, the Calosil’s bottle was vigorously shaked, and nanolime was gently applied by brush on the top face (painted layer) of the mortar, allowing the nanolime to be fully absorbed by the specimen between two consecutive brushstrokes. Specimens’ edges were sealed with parafilm to avoid the penetration of the product through the sides. The bottle of nanolime was also shaken between each brushstroke. The penetration of consolidant in the specimens was monitored by measuring the increase in weight over time. The treatment was stopped when samples obtained asymptotic values of weight increase during the treatment. At this time, the application was interrupted and repeated after 48 h, once each specimen was completely dried. All samples were weighed before and after each application to obtain the amount of nanolime absorbed by each specimen at the moment of the saturation of each application. The difference in weight between the “saturated” and “dry” values was considered the amount of nanolime dispersion absorbed in each application. Final total amount of absorbed nanolime corresponded to the sum of the three saturation–application cycles. The amount of absorbed dispersion product was expressed as the weight of dispersion product per sample volume (g/cm3) which corresponded to 2 (±0.1) g/cm3 for each of the samples. This was carried out to ascertain that all specimens absorbed the same amount of product during the treatment facilitating the comparison between treatments. Additionally, since the nanolime concentration was known (i.e., 12.5 g/L), the total amount of Ca(OH)2 particles absorbed by the specimen as a result of the treatment (g) could also be estimated, which corresponded to 0.255 g of Ca(OH)2 in each specimen. This application protocol has also been used in previous studies concerning nanolime treatments [25,26,27]. There are a vast number of articles in the literature testing the effectiveness of nanolime. However, most papers just indicate the number of applications (most commonly 1, 2, 4, 6 and 8) without providing an empirical number of absorbed consolidating material. This could be misleading since it does not provide the total amount of particles each testing sample absorbed in each application, thus making it difficult to establish a reliable comparison between different experiments. This protocol was carried out to ensure that all samples obtained the same amount of particles, so that the influence of the cleaning method could be analysed on similar conditions. The authors consider that this is slightly more accurate than just indicating the number of applications without providing an empiric number. After the application, the samples were stored for a period of 28 days at RH ~75% (±10% RH) in an outdoor sheltered storeroom to accelerate carbonation and foster the formation of pure calcite [28].
In the case of M1, M2, AG1, AG2, ED1, ED2, AC1 and AC2 (Table 1), the treatment application procedure was as described before, without removing the excess of product accumulated on the surface after each application. However, in the case of NC1 and NC2, the treatment consisted on the same procedure but the surface of the samples was immediately wiped off with an alcohol-damped cloth after reaching saturation, to remove the excess of nanolime product accumulated over the surface of each specimen. The objective of NC1 and NC2 samples was to assess the influence of the wiping off on the consolidation effectiveness since this praxis is commonly carried out in the field, but its effects have never been tested before.

Table 1.
Testing specimens and cleaning methods applied to each of them.
2.4. Cleaning Methods after Consolidation
The cleaning treatments were carried out on the eight specimens in which the excess of consolidant was not removed after each application (i.e., M1, M2, AG1, AG2, ED1, ED2, AC1 and AC2).
The effectiveness after consolidation of both mechanical and chemical cleaning methods has been investigated. In the case of chemical cleaning, we considered the literature related to the elimination of this type of glazing generated by carbonate crusts [13,16,29,30,31,32]. In this combination treatment, chemical cleaning did not aim to completely remove the deposit, as this could have been too aggressive for the pictorial layer, but aimed to reduce it in companion with the mechanical cleaning without affecting the mechanical properties of the polychromic layer.
In order to facilitate the superficial cleaning action of the chemical treatments and to control their penetration, they were applied with a supporting agent (Gellano Kelkogel, a rigid gel composed of a polysaccharide complex [33]). This agent reduces the diffusion of the solvent into the crust on which it had been applied and provides a more localized acting effectiveness on the material which is being eliminated. It also slows down the diffusion of the solvent in the surface, reducing the possibilities of penetration in the substrate and interaction with other elements of the same [32].
To achieve more representative results and facilitate their assessment, each of the cleaning methods was applied on two different testing specimens (Table 1):
- Mechanical cleaning (M1 and M2): a homogeneous mechanical cleaning was carried out by means of a glass fibre pencil and a scalpel. In both cases, we used circular movements with moderate pressure.
- Aqueous rigid gel (AG1 and AG2): the aqueous gel used was Gellano Kelkogel at 3% and it was left to act for 60 min. Water is considered a universal solvent; in this case, it has been chosen because it is the solvent indicated by nanolime commercially available brands for remove the excess of product remaining after the treatment.
- EDTA rigid gel (ED1 and ED2): EDTA salt was applied with pH 10 by means of Gellano Kelkogel rigid gel at 3% and was left to act for 60 min. EDTA is a chelating agent; at pH 10 complexes calcium ions that form the whitish crust helping it removal [29].
- Citric acid rigid gel (AC1 and AC2): citric acid was applied with Gellano Kelkogel rigid gel at 3% and was left to act for 30 min. Due to its chelating power in addition to its acid character that reacts with basis substances such as lime, citric acid wakens the whitish layer formed by nanolimes, favouring their elimination [29].
A mechanical cleaning was also carried out after the chemical treatments, this time with only a scalpel.
2.5. Assessment of Cleaning Effectiveness
Cleaning effectiveness was analysed by using several non-destructive techniques. To assess the effect of the cleaning procedure on the consolidation effectiveness of Calosil, experimental analyses were carried out before and after the consolidation treatment and repeated after the cleaning procedure for the samples in which the excess of consolidant has not been removed after each application (M1, M2, AG1, AG2, ED1, ED2, AC1 and AC2). However, in the case of samples in which the excess of product has been removed after each application (NC1 and NC2), the measurements were taken only before and after the consolidation, since the excess of product from the surface has been previously wiped off during the consolidation treatment and is considered a cleaning method. During the experimental analysis, in order to assure that the measurements were always taken at the same point and to avoid small variations caused by a minor heterogeneity of the pictorial layer, a template was made of transparent acetate where it was indicated the exact point of each measurement (Figure 2). This template was individually created for each specimen and used for the colour measurements, the stereoscopic microscopy and the Scotch Tape Test (STT). Measurements recorded changes before and after the consolidation treatment and after the cleaning method in the exact same location.
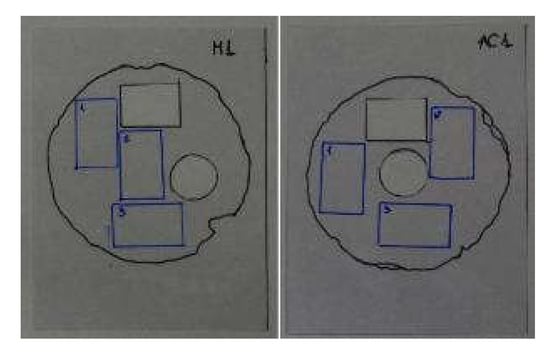
Figure 2.
Examples of two measuring templates: the black rectangle corresponds to the stereoscopic microscopy analysis; the circle corresponds to the measurements taken with the spectrophotometer and the blue rectangles correspond to the Scotch Tape Test.
2.5.1. Photographic Documentation
One of the most immediate methods for the assessment of a treatment consists of a visual examination of the initial and final appearance of the surface. For this reason, photographic record of the surface was conducted before and after the application of the consolidation treatment, as well as after the cleaning. This allowed a general a visu examination of the surface, providing a global evaluation of the treatments as a basis for the subsequent evaluations carried out on more specific aspects.
The photographic record was conducted using a photographic camera Nikon D5300 (Tokyo, Japan) with collapsible screen and AF-S Nikkor 18–105 mm lens with VR. The photographs were taken with colour chart and scale and placing the photographic camera on a tripod.
2.5.2. Assessment of the Surface by Means of the Stereoscopic Microscope
In order to carry out a more detailed analysis of the surface, we have chosen a representative area of the pictorial layer and this has been analysed by means of stereoscopic microscopy (NIKON SZM1000, Tokyo, Japan), where a photograph of the same area has been taken before and after consolidation and after cleaning.
2.5.3. Superficial Adhesion Strength (Scotch Tape Test)
The influence of the consolidation and cleaning methods on surface cohesion was evaluated by “Scotch Tape Test” (STT) according to ASTM 2009 [34], and following the guidelines established by Drdácký et al. to avoid systematic errors of the ASTM standard [35]. The test has been carried out on each of the specimen before and after consolidation treatment, on same locations visually indicated on Figure 2. Scotch Tape Tests consisted of three measurements for each sample (area:1.5 cm × 2.5 cm), to estimate consistency of results. STT was used to study if the increase of surface cohesion induced by the consolidation action of nanolime treatments was also noticed after cleaning methods.
2.5.4. Water Absorption Test by Capillarity (Sponge Test)
The influence of the consolidation and cleaning methods was also evaluated by observing changes in the water absorption by capillary kinetics. For this, we followed the contact sponge method, developed by the Istituto per la Conservazione e la Valorizzazione dei Beni Culturali in Florence and converted into UNI STANDARD 11432.2011 [36]. These measurements were carried out before and after the consolidation treatment, and repeated again after the cleaning methods. As established by the standard, the data consisted of three measurements per specimen obtained on three consecutive days, registering the initial and final weight of the sponge impregnated with 5 mL of distilled water and positioning it in contact with the pictorial layer during the established time of 5 min.
2.5.5. Analysis of Colour Variations of the Pictorial Layer
One of the most important concerns when assessing the effectiveness of a consolidation treatment on wall painting is the colour change that it might induce on the pictorial surface. For this reason, we took colour measurements on the surface in order to record any chromatic change induced by the consolidation as well as by each cleaning method.
To study the differences in colour, we used the CIEL*a*b* colour space established by the Commission Internationale de l’Eclairage (CIE) in 1976, we analysed the colour differences registered after the application of the consolidant and after each cleaning method. We calculated this colour difference following the CIEL*a*b* formula ΔE*ab,10. Even though the colour difference CIEDE 2000 (IOS/CIE 116-6:2014) is currently the recommended one by the ISO and the CIE for certain reference conditions, in this case, as not all the required conditions were fulfilled, it was considered appropriate to use the CEILAB colour difference formula which is, on the other hand, the most widely used in the cultural heritage research field [37,38].
Measurements were taken using a Konica-Minolta CM700d spectrophotometer (Tokyo, Japan) equipped with a pulsed Xe lamp (diffuse reflectance geometry, D65 illuminant at 10°) and using the CIELAB colour space. For each specimen, 10 measurements were taken in the same location shown in the acetate template shown in Figure 2.
2.5.6. Quantification of the Thickness of the Eliminated Whitish Deposits
Finally, in order to further assess the effectiveness of each of the cleaning methods, we quantified the thickness of the deposits which was eliminated in the eight specimens in which the excess of consolidant has not been removed after each application (M1, M2, AG1, AG2, ED1, ED2, AC1 and AC2).
To do that, we digitalised the pictorial layer of each specimen before and after the cleaning, by means of photogrammetry. After that, both models were aligned so that the existing distance between them has been calculated, giving as a result a colour map in which the red areas represent the areas in which a larger thickness of the deposit has been removed and the green areas represented those in which the deposit has been remained unchanged, that is, those in which the cleaning has not been significantly effective [39]. The programme can analyse up to ±0.5 mm difference between both models.
Photogrammetric models were carried out with an average of 120 photographs per test specimen, with a 0.66 mm ground sampling distance (GSD), using a light box and a photographic camera Nikon D5300 (Tokyo, Japan) with collapsible screen and an AF-S Nikkor 18–105 mm lens with VR. The size of the meshes varied between 160,000 and 242,000 triangles. The photogrammetric models were generated with Agisoft Photoscan (V.1.7.1.11797), while the comparison of both meshes was carried out using the open-source programme Cloud Compare.
3. Results
3.1. Visual Analysis
The visual analysis of the photographic documentation and the stereoscopic microscopy demonstrated a noticeable difference after the consolidation treatment between those specimens which were treated with the removal of the excess of product after each application (NC1) and those in which that excess of product was not removed. While some whitish deposits appeared in localized areas in the first case, in the second case those deposits presented a larger extension and created a continuous white stratum which completely covered the pictorial layer (Figure 3). Furthermore, the study of the general photographs suggests an acceptable effectiveness of the cleaning in those specimens which were subjected to mechanical cleaning (M1 and M2), as well as EDTA rigid gel (ED1 and ED2) and citric acid rigid gel (AC1 and AC2) chemical cleaning. However, the detailed analysis of the surface carried out with the stereoscopic microscope revealed some damages in form of disintegration caused in the pictorial layer during cleaning in the specimens treated with citric acid which could compromise its effectiveness (Figure 4).

Figure 3.
Photographic record of the specimens before the treatment, after the consolidation and after the cleaning. Due to the similarities between samples, only one sample per testing condition is included.

Figure 4.
Detailed analysis of the pictorial layer by means of stereoscopic microscopy: (a) initial state of AC1; (b) state of AC1 after the cleaning; (c) initial state of AC2; (d) state of AC2 after the cleaning.
3.2. Superficial Adhesion Strength (Scotch Tape Test)
The results of the Scotch Tape Test (Table 2) showed, in all cases, an increase of the superficial cohesion of the pictorial layer after the consolidation treatment. This effect is even noticeable in the specimens in which the excess of product was removed after each application (NC1 and NC2) where the superficial cohesion increased by approximately 70% and 57%, respectively. This increase is very similar to that obtained after the consolidation of samples in which the excess of product on the surface was not removed during application. This result clearly suggests that removing the excess of nanolime product accumulated on the surface during treatment does not decrease the superficial mechanical properties of specimens.

Table 2.
Results of the Scotch Tape Test (STT) after treatment and cleaning processes.
Regarding the superficial cohesion of the pictorial layer after cleaning, results showed that the four cleaning methods slightly reduced the superficial cohesion induced by the consolidation. As it can be seen in Table 2, the mg/cm2 variation in the measurement taken after the cleaning was, in most cases, higher than the obtained in the measurement taken after the consolidation, therefore suggesting that in all cases the applied cleaning treatments decreased the superficial cohesion of the pictorial layer induced by the consolidant. In this sense, both the mechanical cleaning (M) and the citric acid cleaning (AC) were the treatments which presented slightly lower variation in the mechanical properties after cleaning. This result could suggest that the cleaning procedure might have not removed some of the superficial cohesion obtained by the consolidation with nanolime.
3.3. Water Absorption Test by Capillarity (Sponge Test)
Results of the sponge test (Table 3) showed, in all cases, a reduction of the absorption after the consolidation treatment. This effect is even more noticeable in the samples in which the excess of product was removed after each application (NC1 and NC2) where water absorption (Wa) was reduced by 20.77% and 21.1%, respectively. This reduction was very similar to Wa reductions after consolidation in those samples in which the excess of product in the pictorial layer was not eliminated during application (i.e., between 15.21% to 28.66%).

Table 3.
Results of the Capillary Absorption by the Sponge Test after treatment and cleaning processes.
After cleaning, Wa absorption values were maintained relatively stable, showing small fluctuations with respect to the measurements taken after the consolidation. Samples cleaned with Mechanical (M) seemed to present a lower variation in Wa after cleaning, which could suggest a lesser modification in the amount of micropores of the surface after the cleaning. This was in line with the STT results, which also suggested that those samples also presented low levels of variation in the surface cohesion values before and after cleaning. On the other hand, samples cleaned with citric acid presented a higher variation in the Wa kinetics after cleaning presenting increasing values, which suggests that the cleaning procedure might have partially removed some of the superficial nanolime in the pores of the specimen.
3.4. Analysis of Colour Variations
Colour measurements (Table 4) indicated a clearly perceptible colour change after the application of the consolidation treatment, as could also be seen in the visual analysis (Figure 4). It is considered that colour difference values (ΔE) of around 3.0 are noticeable by people with a normal colour vision [40] and values ΔE > 5 are not appropriate for the consolidation of historical structures [41]. Colorimetric results have shown significant colour differences on all treated samples where the excess of nanolime has not been removed from the surface after treatment, obtaining ΔE values between 24.17 to 54.19. However, in the case of the two specimens in which the excess of product was removed during the treatment, this colour difference was considerably lower, being ΔE 3.03 and 4.95, slightly above the noticeable threshold of human eye [40]. As expected, all samples treated without removing the excess of product from the surface, presented a significant increase in the L* after treatment, which corresponded to the whitening induced by the nanolime, as shown in Figure 4, being the main reason for the high ΔE values in those samples. In particular, no significant white glazing was detected in specimens NC1 and NC2, as reflected by the absence of change of L* parameter (average values 60.22 ± 0.17 and 60.20 ± 0.23 for NC1, and 61.14 ± 0.18 and 63.09 ± 0.23 for NC2, before and after treatment, respectively). The absence of white glazing or any other undesired chromatic side-effect suggests that the tested protocol used for the application of Ca(OH)2 nanoparticle dispersions on NC meets the requirements for practical conservation interventions, since it increases the surface cohesion and slightly reduces water absorption by capillarity without inducing significant aesthetic alterations.

Table 4.
Chromatic alterations for treated and cleaned samples.
Colour measurements taken after cleaning (Table 4), showed that colour values were closer to the initial values of specimens before treatment, even though it still existed a noticeable colour difference, which ranged from ΔE~14.44 (AG2) in the smaller case and ΔE~43.33 (AC2) in the higher case. Samples cleaned by mechanical cleaning (i.e., M1 and M2) presented a higher reduction in ΔE values (a reduction of 53.3% and 38.3%, respectively) which suggests this method presents higher effectiveness, as it was also suggested by the visual observation, STT and the Sponge test.
3.5. Quantification of the Thickness of the Eliminated Whitish Deposits
The analysis performed by means of photogrammetry for measuring the thickness of the removed whitish deposits showed that there was a small difference between the initial and final models of the specimens treated with rigid gels (Figure 5), while a higher difference was recorded in those models corresponding to specimens treated with mechanical cleaning, quantifying the thickness of the removed deposits in up to 0.1 mm (Figure 6). This is also in line with the above results that suggest this cleaning method presents higher effectiveness in terms of higher reduction of excess of product in the surface without altering the consolidating properties induced by the nanolime.
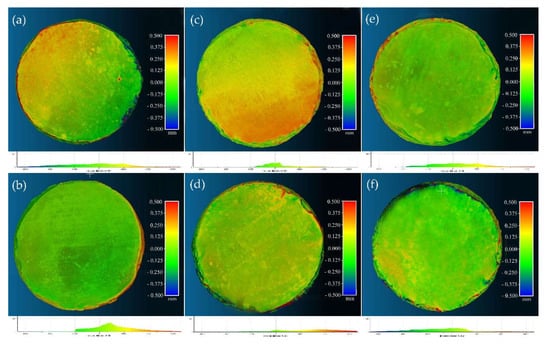
Figure 5.
Rough quantification of the thickness of the whitish deposits removed in those specimens cleaned with rigid gels; the green areas are those in which the result of the treatment was hardly noticeable, and the yellow and red areas corresponded to those in which the treatment has been more noticeable: (a) AG1; (b) AG2; (c) ED1; (d) ED2; (e) AC1; (f) AC2.
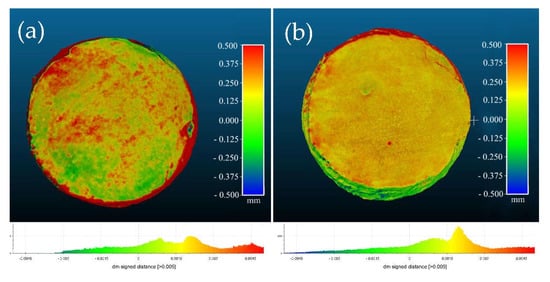
Figure 6.
Rough quantification of the thickness of the whitish deposits eliminated in the specimens which has been carried out mechanical cleaning; the green areas were those in which the treatment has hardly been noticeable, and the yellow and red areas were those in which the treatment had been more appreciable: (a) M1; (b) M2.
4. Discussion
First of all, the specimens in which the consolidation treatment has been applied removing the excess of product after each application (NC1 and NC2) showed, in general terms, an acceptable consolidation effectiveness since they presented a significant increase in superficial cohesion (57–70%), a reduction in Wa (20–21% reduction) while did not significantly induced the so-called white glazing side-effect commonly caused by the nanolime treatments, as reported in the literature [5,6,7,8,14,15,25,26,27,28]. While the rest of consolidation treatments showed a regular white “stratum” covering up the pictorial layer, NC1 and NC2 only showed some small whitish deposits, therefore the difference of aspect and colour they showed after the application of the treatment is significantly lower (ΔE* 3.99 ± 0.96). On the other hand, the cleaning of the excess of product during the application did not involve a reduction of the consolidating properties, as the results of the absorption test and the Scotch Tape Test in these cases were similar to those of the other specimens. Likewise, as no subsequent cleaning was needed, the pictorial layer did not suffer the possible damages derived from the cleaning.
Secondly, thanks to the comparison of the photogrammetric models, as well as the colour measurements carried out, we demonstrate that specimens that have been mechanically cleaned (M1 and M2) also showed acceptable results in terms of removing a large quantity of whitish deposits, that were covering the pictorial layer, without showing a significant colour difference with respect to the original appearance after treatment (ΔE* = 14.75 ± 0.17). Additionally, the stereoscopic microscope analysis suggests that the cleaning treatment does not mechanically affect the pictorial layer. Besides the visual observations, absorption tests and the Scotch Tape Test confirm that this mechanical cleaning does not significantly affect the induced consolidation effectiveness.
The specimens in which a chemical cleaning was carried out with rigid gels (AG1, AG2, ED1, ED2, AC1 and AC2) showed similar results among them. In those samples, cleaning treatments contributed to the softening of the white deposits, thus facilitating their posterior removal by mechanic action. However, those cleaning methods also weakened the physical-mechanical properties of the pictorial layer, as it was noticeable in absorption, STT and in the photogrammetric models, since while those from M1 and M2 showed some yellow and red areas, in which the whitish deposit which covered the pictorial layer has been removal, in AG1, AG2, ED1, ED2, AC1 and AC2 most of the surface showed in green, meaning that the cleaning has been significantly superficial.
The specimens that were used for the chemical cleaning, those in which aqueous rigid gels were applied (AG1 and AG2) showed less colour variation with respect to the original appearance (ΔE* 16.71 ± 2.26). Despite the cleaning treatment not causing a significant variation in water absorption of the pictorial layer, it showed a significant reduction of superficial adhesion strength obtained in the Scotch Tape Test.
The specimens in which EDTA rigid gel was applied (ED1 and ED2) showed a higher colour variation with respect to the original appearance (ΔE* 24.43 ± 5.31). This cleaning treatment is the one which caused a higher loss of superficial adhesion strength induced after consolidation; however, after the cleaning treatment, these specimens presented lower water absorption kinetics, even lower than that showed before the cleaning treatment.
Finally, the specimens treated with citric acid gel (AC1 and AC2) were those which presented a higher colour difference with respect to the original appearance (ΔE* 38.4 ± 4.92), being also those which experienced, according to the data collected by the stereoscopic microscope, some mechanical damage in form of disintegration in the pictorial layer. Despite AC1 and AC2 showed the lower change in the superficial adhesion strength by Scotch Tape Test before and after cleaning, taking into consideration the induced damage in the form of disintegration, this cleaning method could be considered inappropriate since it could potentially be aggressive to the pictorial layer. A full study of this damage will be carried out in future research.
5. Conclusions
This work aimed to respond one of the most important practical issues within nanolime consolidation treatments in wall painting: the creation of a whitish deposits which covers the surface.
The first conclusion regards to the application method of the consolidation treatment, which is of great importance. In this sense, the results showed that the removal of the excess of consolidating product after each application avoids, to a large extent, the subsequent appearance of whitish deposits which cover the pictorial layer, without significantly reducing the consolidating effect of the consolidation treatment in terms of an increase of superficial cohesion and a reduction of water absorption by capillarity. On the contrary, the application of the consolidation treatment without removing the excess of product at the time, generated a large whitish layer which covered the surface. Therefore, despite the fact that this layer could be removed afterwards with cleaning treatments, the stress caused to the pictorial layer and the possible deterioration this might cause, it is preferable to dismiss this application method.
Secondly, the results also showed that the subsequent removal of the whitish deposits, when possible, is preferable with an exclusively mechanical cleaning. If the deposits show an excessive hardness, it is possible to try to soften them with chemical cleaning; in this case, the cleaning with rigid gels showed good results as it reduced the penetration of solvent in the supporting material and increased its superficial action, allowing to increase the time of contact of the treatment. In this sense, aqueous and EDTA gels were those with intermediate results, and the use of citric acid was considered inappropriate since it could potentially cause chemical and mechanical damage, being too aggressive to the pictorial layer.
Finally, despite this is not presented in this work, it is possible to combine both options: the application methodology of the consolidant and the cleaning treatment. This approach could potentially obtain good results since it could eliminate the excess of product after each application during the consolidation and, after that, eliminating the possible whitish deposits which it generates by mechanical cleaning. A full study of this approach will be carried out in future research. However, it is necessary to emphasize that if it is possible, it is preferable and advisable to remove the excess of product after each application; according to the results obtained in this work, it does not interfere with the consolidating action of the treatment and it avoids the generation of whitish veils and the consequent stress that occurs to the wall painting when attempting their subsequent removal.
Author Contributions
Conceptualization, T.L.-M. and J.O.; methodology, T.L.-M. and J.O.; software, T.L.-M. and J.O.; validation, T.L.-M. and J.O.; formal analysis, T.L.-M. and J.O.; investigation, T.L.-M. and J.O.; resources, T.L.-M. and J.O.; data curation, T.L.-M. and J.O.; writing—original draft preparation, T.L.-M. and J.O.; writing—review and editing, T.L.-M. and J.O. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Ministerio de Ciencia e Innovación, Gobierno de España, with the project PID 2019-105706GB-I00/SRA (State Research Agency/10.13039/501100011033). J. Otero’s research in nanolime is currently being funded by the European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie (MSCA-IF) grant agreement No 893762 (NANOMORT).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data is contained within the article.
Acknowledgments
The authors would like to thank Ana García Bueno, for her counsel and disposition. Authors thank Carlos Rodriguez-Navarro for his useful advice, comments and revision.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Doehne, E.; Price, C.A. An overview of current research. In Stone Conservation, 2nd ed.; Research in Conservation, the Getty Conservation Institute, Getty Publications: Los Angeles, CA, USA, 2011; pp. 35–37. [Google Scholar]
- Mora, P.; Mora, L.; Philippot, P. La Conservazione Delle Pitture Murali, 2nd ed.; Editrice Compositori: Bologna, Italy, 2003. [Google Scholar]
- Young, M.E.; Murray, M.; Cordiner, P. Stone Consolidants and Chemical Treatments in Scotland; Edinburg: Historic Scotland, UK, 1999. [Google Scholar]
- Hansen, E.; Doehne, E.; Fidler, J.; Larson, J.; Martin, B.; Matteini, M.; Rodriguez-Navarro, C.; Pardo, E.S.; Price, C.; de Tagle, A.; et al. A review of selected inorganic consolidants and protective treatments for porous calcareous materials. Rev. Conserv. 2013, 4, 3630. [Google Scholar] [CrossRef]
- Rodriguez-Navarro, C.; Ruiz-Agudo, E. Nanolimes: From synthesis to application. Pure Appl. Chem. 2017, 90, 523–550. [Google Scholar] [CrossRef]
- Otero, J.; Charola, A.E.; Grissom, C.A.; Starinieri, V. An overview of nanolime as a consolidation method for calcareous substrates. GE Conserv. 2017, 1, 71–78. [Google Scholar] [CrossRef] [Green Version]
- Baglioni, P.; Chelazzi, D.; Giorgi, R.; Carretti, E.; Toccafondi, N.; Jaidar, Y. Commercial Ca (OH)2 nanoparticles for the consolidation of immovable works of art. Appl. Phys. A 2014, 114, 723–732. [Google Scholar] [CrossRef]
- Baglioni, P.; Chelazzi, D.; Giorgi, R. Nanotechnologies in the Conservation of Cultural Heritage: A Compendium of Materials and Techniques; Springer: New York, NY, USA, 2015. [Google Scholar]
- Ambrosi, M.; Dei, L.; Giorgi, R.; Neto, C.; Baglioni, P. Colloidal particles of Ca(OH)2: Properties and application to restoration of frescoes. J. Am. Chem. Soc. 2001, 17, 4251–4255. [Google Scholar] [CrossRef]
- Giorgi, R.; Baglioni, M.; Berti, D.; Baglioni, P. New methodologies for conservation of cultural heritage: Micellar solutions microemulsions and hydroxide nanoparticles. Acc. Chem. Res. 2010, 43, 695–704. [Google Scholar] [CrossRef]
- Baglioni, P.; Vargas, R.C.; Chelazzi, D.; Gonzàlez, M.C.; Desprat, A.; Giorgi, R. The Maya site of Calakmul: In situ preservation of wall paintings and limestone using nanotechnology. Stud. Conserv. 2016, 51, 162–169. [Google Scholar] [CrossRef]
- Natali, I.; Saladino, M.L.; Andriulo, F.; Chillura Martino, D.; Caponetti, E.; Carretti, E.; Dei, L. Consolidation and protection by nanolime: Recent advances for the conservation of the graffiti, Carceri dello Steri Palermo of the 18th century lunettes, SS. Giuda e Simone Cloister, Corniola (Empoli). J. Cult. Herit. 2014, 15, 151–158. [Google Scholar] [CrossRef]
- Esquivel Álvarez, A.; Filloy Nadal, L. Evaluación del proceso de carbonatación de nanocales aplicadas a pinturas murales prehispánicas de origen maya. Intervención 2007, 14, 31–41. [Google Scholar]
- Otero, J.; Starinieri, V.; Charola, A.E. Nanolime for the consolidation of lime mortars: A comparison of three available products. Constr. Build. Mater. 2018, 181, 394–407. [Google Scholar] [CrossRef] [Green Version]
- Otero, J.; Charola, A.E.; Starinieri, V. Sticky rice–nanolime as a consolidation treatment for lime mortars. J. Mater. Sci. 2019, 54, 10217–10234. [Google Scholar] [CrossRef] [Green Version]
- CTS. Nanorestore. Ficha Técnica. Available online: https://shop-espana.ctseurope.com/documentacioncts/fichastecnicasweb2018/2.1consolidantes2016/nanorestoreesp.pdf (accessed on 17 May 2021).
- Calosil IP5. Ficha Técnica. Available online: https://www.calosil.de/downloads-en (accessed on 17 May 2021).
- Plinio Segundo, C. Historia natural de Cayo Plinio Segundo. XXXV. De la pintura, colores y pintores. Traductor, F. Hernández y J. de Huerta; VISOR: Madrid, Spain, 1998. [Google Scholar]
- Vitruvio Polión, M. Los Diez Libros de Arquitectura. VII; Cicón Ediciones: Cáceres, Spain, 1999. [Google Scholar]
- López-Martínez, T.; Blanc-García, M.R.; García-Bueno, A. Metodología para el estudio de morteros arqueológicos de revestimiento. Ge-Conserv. 2021, 19, 31–44. [Google Scholar] [CrossRef]
- Franzoni, E.; Sassoni, E.; Sherer, G.W.; Naidu, S. Artificial weathering of stone by heating. J. Cult. Herit. 2013, 145, 85–93. [Google Scholar] [CrossRef]
- Sena da Fonseca, B.; Ferreira Pinto, A.P.; Rodrigues, A.; Picarra, S.; Fonseca, D.; Montemor, M.F. On the estimation of marbles weathering by thermal action using drilling resistance. J. Build. Eng. 2021, 42, 102494. [Google Scholar] [CrossRef]
- Arizzi, A.; Gomez-Villalba, L.S.; Lopez-Arce, P.; Cultrone, G.; Fort, R. Lime mortar consolidation with nanostructured calcium hydroxide dispersions: The efficacy of different consolidating products for heritage conservation. Eur. J. Mineral. 2015, 27, 311–323. [Google Scholar] [CrossRef] [Green Version]
- Slizkova, Z.; Frankeová, D. Consolidation of a porous limestone with nanolime. In Proceedings of the 12th International Congress on the Deterioration and Conservation of Stone, New York, NY, USA, 21–25 October 2012. [Google Scholar]
- Otero, J.; Pozo-Antonio, J.S.; Montojo, C. Influence of application method and number of applications of nanolime on the effectiveness of the Doulting limestone treatments. Mater. Struct. 2021, 54, 1–19. [Google Scholar] [CrossRef]
- Otero, J.; Starinieri, V.; Charola, A.E.; Taglieri, G. Influence of different types of solvent on the effectiveness of nanolime treatments on highly porous mortar substrates. Constr. Build. Mater. 2020, 230, 117112. [Google Scholar] [CrossRef]
- Otero, J.; Starinieri, V.; Charola, A.E. Influence of substrate pore structure and nanolime particle size on the effectiveness of nanolime treatments. J. Constr. Build. Mater. 2019, 209, 701–708. [Google Scholar] [CrossRef] [Green Version]
- López-Arce, P.; Gomez-Villalba, L.S.; Pinho, L.; Fernández-Valle, M.E.; de Buergo, M.Á.; Fort, R. Influence of porosity and relative humidity on consolidation of dolostone with calcium hydroxide nanoparticles: Effectiveness assessment with non-destructive techniques. Mater. Charact. 2010, 61, 168–184. [Google Scholar] [CrossRef] [Green Version]
- Barros García, J.M.; Llano Torre, S. Utilización de ácido cítrico y EDTA en la limpieza de estructuras pictóricas. Estud. de Conserv. e Restauro 2005, 3, 32–45. [Google Scholar]
- Del Ordi Castilla, B.; Regidor Ros, J.L.; Pasíes Oviedo, T. Revisión de tratamientos de limpieza de pintura mural arqueológica. Eliminación de concreciones carbonáticas. ARCHÉ 2010, 4, 73–80. [Google Scholar]
- Nicola, M.; Scalesse, R.; Coluccia, S. Elaborazione ed applicazione di un sistema di pulitura per l’assottigliamento di strati di carbonatazione su superficie murale dipinta sensibile, realizzato mediante soluzioni acquosa addensate a ph debolmente acido. In Proceedings of the IV Congresso Nazionale IGIIC-Lo Stato dell'Arte-Siena, Siena, Italy, 28–30 September 2006. [Google Scholar]
- Cremonesi, P. L’ambiente Acquoso per il Trattamento di Opere Polirome; Il Prato Publishing House: Villatora, Italy, 2012. [Google Scholar]
- Gellano Kelcogel. Ficha Técnica. Available online: https://shop-espana.ctseurope.com/documentacioncts/fichastecnicasweb2018/3.1disolventes2016/gellanokelocogelesp.pdf (accessed on 17 May 2021).
- ASTM. Standard Test Methods for Measuring Adhesion by Tape Test; ASTM D3359-02; ASTM International: West Conshohocken, PA, USA, 2002. [Google Scholar]
- Drdácký, M.; Lesák, J.; Rescic, S.; Slížková, Z.; Tiano, P.; Valach, J. Standardization of peeling tests for assessing the cohesion and consolidation characteristics of historic stone surfaces. Mater. Struct. 2012, 45, 505–520. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- UNI. Cultural Heritage—Natural And Artificial Stone—Determination of the Water Absortption By Contact Sponge; UNI 11432:2011; Ente Nazionale Italiano di Unificazione: Roma, Italy, 2011. [Google Scholar]
- Collado-Montero, F.J.; Calero-Castillo, A.I.; Melgosa, M.; Medina-Flórez, V.J. Colorimetric evaluation of pictorial coatings in conservacion of plasterworks from the Islamic tradititon. Stud. Conserv. 2019, 2, 90–100. [Google Scholar] [CrossRef]
- Prestileo, F.; Bruno, G.; Alberghina, M.F.; Schiavone, S.; Pellegrino, L. I mosaici della villa romana del Casale di Piazza Armerina: Il contributo della indagini colorimetriche per la stesura del protocollo di intervento. In Proceedings of the Terza Conferenza Nazionale del Gruppo del Colore, Torino, Italy, 24–26, October, 2007. [Google Scholar]
- López-Martínez, T.; García-Bueno, A.; Medina-Flórez, V.J. New methodology for the assessment of cleaning treatments. Applications of photogrammetry for restoration. J. Cult. Herit. 2018, 30, 123–177. [Google Scholar] [CrossRef]
- Huang, M.; Liu, H.; Cui, G.; Luo, M.; Melgosa, M. Evaluation of threshold color differences using printed samples. J. Opt. Soc. Am. A 2012, 29, 883–891. [Google Scholar] [CrossRef] [PubMed]
- Grossi, C.M.; Blimblecombe, P.; Esbert, R.M.; Alonso, F.J. Color changes in architectural limestones from pollution and cleaning. Color Res. Appl. 2007, 32, 320–331. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).