Abstract
In rural Africa, income generating activities of many households heavily depend on agricultural activities. In this paper, we present the results of a multi-year intervention whereby dairy farmers and small-scale entrepreneurs were taught to convert their milk into a probiotic yoghurt using an innovative bacterial starter culture and basic equipment. This intervention creates additional sources of income and employment for people involved in the delivery of milk as well as production, distribution, and sales of yoghurt. Besides the economic benefits, the consumption of the probiotic yoghurt can contribute to reduction of the incidence and severity of diarrhea, respiratory tract infections, atopic diseases, alleviate the symptoms of stomach ulcers, and decrease the uptake of aflatoxins in the body. With minimal external financial support, 116 communities or small entrepreneurs have been able to start, expand, and maintain a business by production and sales of probiotic yoghurt. Applied business models and success rate in terms of revenues and profitability varied per region and depended on location, culture, ownership structure, wealth status, and gender.
1. Introduction
In Uganda, the farmland area per capita is decreasing due to population growth [1,2]. Therefore, occupational diversification such as non-farm rural incomes are necessary for successful income growth, and critical to the achievement of sustainable livelihoods. Several studies indicate that additional non-farm income is the best driver for agricultural growth, as the monetary income reduces vulnerability and allows households to buy material to improve agricultural yields [1,3]. In sub-Saharan Africa, the degree of economy-wide diversification is the lowest in the world, with women being even less involved in off-farm activities than men [4]. Today in Uganda only 24% of the household income is obtained from non-agricultural enterprises [5]. Many regions and their populations face systemic constraints for steady economic development such as geographical isolation, rough climate conditions, low quality of physical infrastructure, including roads and electricity, poor human capital development, lack of investment opportunities, low population density, underdeveloped markets, irregular seasonal income, and general low purchasing power of the population [4,6]. A typical example of an underdeveloped market is reflected by the milk market in Uganda where communities either have a lack of market, no access to the market for the milk they produce, lack of negotiation skills and power (Central, North, and East Uganda), or low profit margins (South-West Uganda) [7,8]. Despite all these challenges, Uganda is marked as one of the countries with the highest economic growth rates in the world [9]. This translates in a trend of accelerated commercialization and urbanization and increased purchasing power over the past years, which has improved the market system. At the same time, increased market regulation might limit local entrepreneurs with narrow financial means to effectively implement value adding activities in agriculture and food production. Overall, with a GDP per capita of 615 USD (as compared to e.g., 55,972 USD in North-America), Uganda is still a developing country by all classification systems [10]. Moreover, the growth is said to be non-inclusive and of benefit for a minor part of the population [11]. In spite of the high growth rate of the national economy, the growth rate per capita is still relatively low, due to the strong population growth [9].
Apart from the economical hurdles, many African countries also face challenges in public health, food safety, and food security. In Uganda, 12% of the population lacks reliable access to healthy food [12]. Malnutrition undermines work productivity, increases the risk and severity of disease, and can lead to death. The average life expectancy in Uganda is 60 years [13], 29% of children under age of five are stunted [14], 13% of child death (1–59 months) is caused by diarrhea, and 23% by respiratory tract infections [15]. In addition, approximately 1.5 million Ugandans (7% of the population) suffer from AIDS [13].
The incidence of bacterial related infections and environmental risk factors such as the presence of aflatoxins in food put a burden on the society. Treatment is often not available or not accessible due to high costs. Another example is the incidence of diarrhea, which is high under Ugandan children and can have fatal consequences. A solution could be provided by consumption of probiotic food. Numerous studies have shown the inhibitory or scavenging effects of certain probiotic bacteria (for example, Lactobacillus rhamnosus GG [16]) on health risk factors and relieve of associated pathogenic symptoms, including diarrhea. However, probiotic products are hardly available in Africa, highly priced, and the related health effects unknown by the population.
Many ethnic groups across Uganda have a culture of home-production and consumption of fermented milk, which is commonly named ‘bongo’ or ‘makamo’. These products are either produced by spontaneous fermentation through environmental yeasts and bacteria, or by back-slopping with previously made fermented milk [17]. However, products made through these processes usually have a short shelf life, inconsistent quality, and even food safety concerns. Hence, commercialization of these traditionally fermented foods faces many challenges.
In this paper, we describe an intervention with an education program and an innovative bacterial starter culture with a presumed sustainable and scalable effect on improving public health and income generation by (i) stimulation of the local milk-market, (ii) increasing employment and income by value addition, (iii) facilitating access to an affordable healthy product, and (iv) reduction of spoilage and spillage of milk. The intervention occurred in several regions in Uganda between 2012 and 2018. Over 170 communities and small entrepreneurs were trained in processing locally produced milk into probiotic yoghurt in line with market standards and without high requirements for (technical) resources. By the end of the period, 116 of the trained groups were able to start, expand and maintain a business in locally made probiotic yoghurts. In order to view the intervention in its right context, it is essential to discriminate between the dairy situation in South-West Uganda and parts of central Uganda from the rest of the country (Box 1).
Although mass scale has not yet been reached, the developed concept and way of working applied elements of the theory of ‘fortune at the bottom of the pyramid’, as developed by Prahalad and Hart [18], and has further potential to reaching scale. Figure 1 portraits the original four BoP principles, together with a description of the approach to stimulate demand and supply as was done in this intervention.
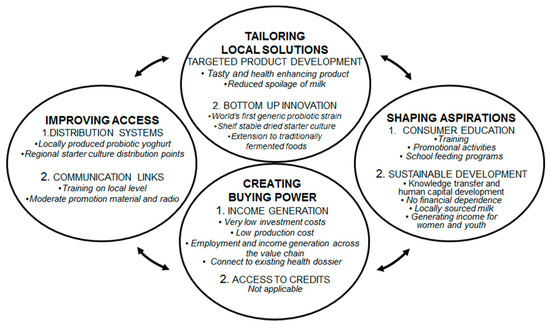
Figure 1.
Stimulating supply and demand for small scale probiotic yoghurt business development at the bottom of the pyramid. Adapted from Pralahad and Hart [18]. Capital letters: original text. Italics: Translation to small scale probiotic yoghurt business development.
Box 1. Description of dairy farming in geographical regions of Uganda as context for the intervention.
South-Western Uganda, as well as areas around the capital Kampala, can be considered the milk-shed of the country, where dairy farming is performed at a commercial scale and where large dairy processing plants in the country are located. These processing plants are responsible for the fact that Uganda is the second-largest, and soon possibly the largest, dairy exporter of Africa [19]. The dairy sector in this region is generally well organized, with 681 milk cooling centers spread over the region. The milk in these coolers is subsequently collected by the milk processors or traders of raw milk. Considering this scale of operations, small-scale ‘back yard’ milk processing (cottage industry) does not have a significant impact on the dairy sector at large in this area. Developments in the dairy sector do however influence the cottage industry. On one hand, the wide availability of milk makes it a relatively accessibly business, especially for dairy farmers themselves, but also extending to other residents of the region. In addition, the improved wealth status and associated purchasing power in the region, allows the products to be sold above the national average selling price. On the other hand, due to the demand and competition in the sector, the milk prices have raised in the recent past, and the improved infrastructure of coolers has reduced the incentive for farmers to search for alternatives for selling fresh milk.
In the remaining areas of the country, milk is produced as a form of subsistence farming, and the dairy sector is dominated by farmers who own 1–4 cows and sell their milk to their neighbors or in the nearby trading centers. In these regions, cottage industry has a more profound impact on the sector at large. The sudden increase of demand for milk as induced by yoghurt business could stimulate the local farmers to produce more milk (more cows, better breeds, better feeding) and organize themselves in groups to join forces for milk collection and transport to the yoghurt producer. The dairy sector in turn also influences the cottage industry, as in the dry season the milk production goes down and no longer meets the demand for milk by the small-scale processor.
2. Results
2.1. Start-Up and Development
In 2012 the locally produced probiotic yoghurt project started at a small yoghurt production site called Life Dairy, in Kateete, Mukono district, Uganda. The owner of this business was an early adopter, willing to take a risk, well informed, and respected in the community. In 2013, five more small sized companies joined the probiotic yoghurt production project. Initial weekly production volumes varied between 50 and 200 L. The developed bacterial starter cultures were stored at and distributed from the Ugandan Industrial Research Institute in Kampala.
After 2014, the project started to grow rapidly when contacts were made with dairy cooperatives in rural areas through Heifer International under the East African Dairy Development project (EADD). At the same time, the project team changed from working with short term volunteers to a small team of two professionals who selected potential probiotic yoghurt producers, coordinated trainings and measured progress. In addition, the team scouted for more small-scale operating dairy companies, women groups and local entrepreneurs motivated to start a probiotic yoghurt production. As a result, the total number of probiotic yoghurt producers increased, as well as the weekly production volumes per production unit. By mid-2016, 99 processing units were trained across several regions in Uganda and 59 of them were well into business. Weekly production volumes varied between 20 and 1000 L. Five regional starter culture distribution centers facilitated distribution and sales of the starter cultures.
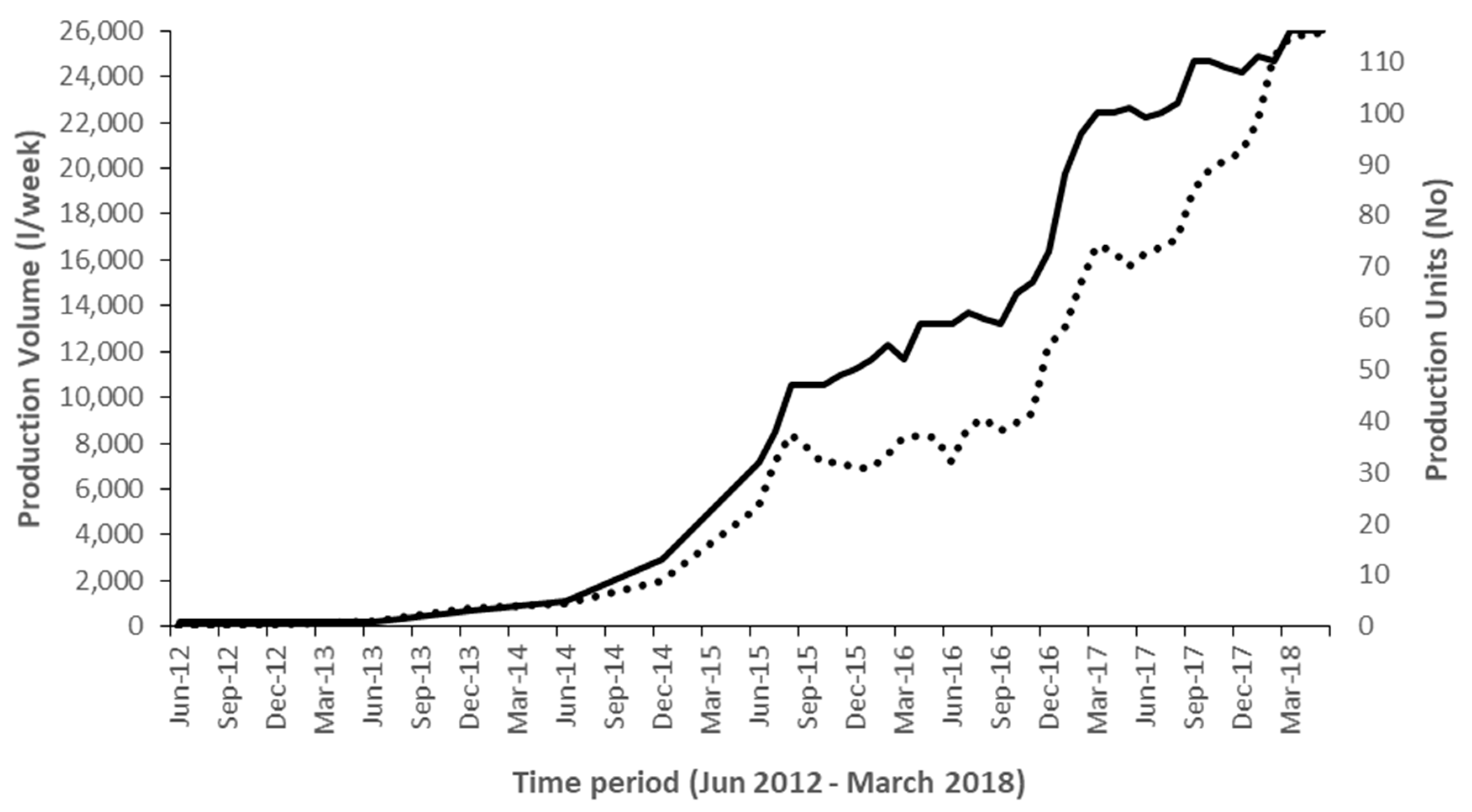
This successful pilot phase then enabled Yoba for Life Foundation in collaboration Heifer International and the University of Western Ontario (UWO) to acquire funding from the International Development Research Centre (IDRC) to implement a two-year project (May 2016–June 2018) entitled the ‘Fermented Food for Life’ (FFFL) in Uganda, Kenya and Tanzania. In Uganda, this funding allowed employment for four fulltime staff spending most of their time in the field, to implement a wide range of activities form increasing awareness on probiotic yoghurt, to building capacity and competence of production units by improvement of product quality as well as business administration. The trainers’ team organized workshops, provided coaching and empowered production units to participate in 36 shows and exhibitions throughout the country. At such events, the new probiotic yoghurt producers could promote and sell their products and create awareness on probiotics. By June 2018, 116 production units were in business, making cumulative volumes of close to 26,000 L per week (20–2000 L per unit) (Figure 2 and Figure 3). Furthermore, the trainers had started a collaboration with the Dairy Development Authority (DDA) in Uganda, leading to a joined training program, enabling 75 production units to acquire a certificate of analysis. This certificate indicated that the product complied to Ugandan National Bureau of Standards (UNBS) microbial specifications for yoghurt. By the time of writing, several other production units are still in the process of acquiring the same certification. Simultaneous to the increase of the number of production units all over Uganda, the number of probiotic starter culture distribution units, who purchased their stock on wholesale from Kampala, increased to 12, (Figure 3).
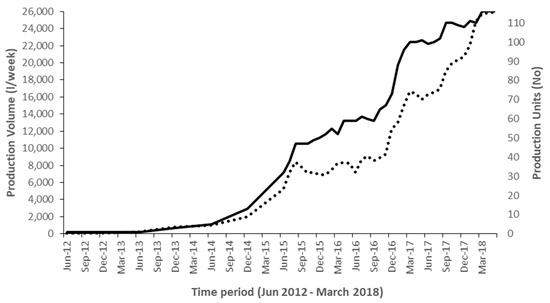
Figure 2.
The number of active probiotic yoghurt production units and accumulated weekly production volumes in liters of probiotic yoghurt production over the period 2012–2018. Solid line: number of production units (right vertical axis); Dotted line: accumulated weekly production volumes (left vertical axis).
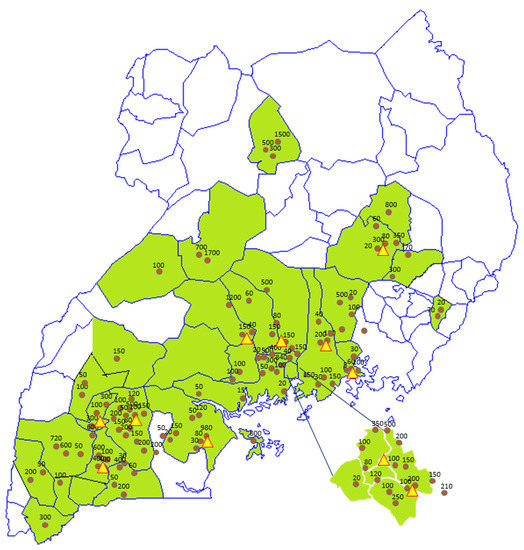
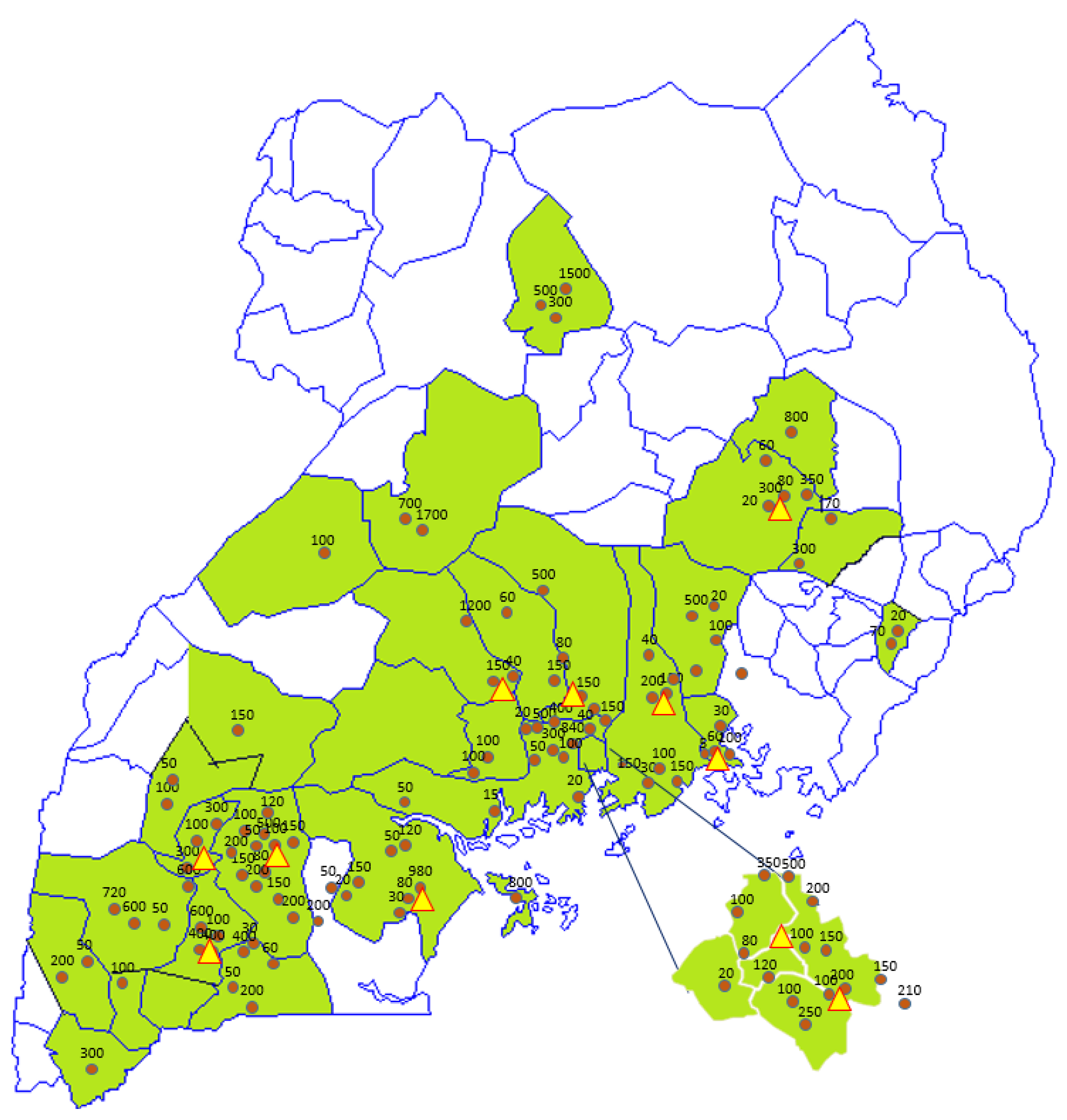
Figure 3.
Geographical footprint of the intervention in Uganda. Date: May 2018. Spheres: Probiotic yoghurt production units including average weekly production volumes; Triangles: Starter culture distribution units.
In terms of gender, age, and number of people involved, 53% of the production units were owned and run by women, and 22% by youth (defined as people below 35 years of age). The age of people involved varied from 16 to 60 years old. In terms of employment, 704 people were involved in milk collection, production, packaging, distribution, and sales, of which 57% were women and 43% were youth. The number of people involved per production unit ranged from 2 to 22. The production units were spread over 39 out of Uganda’s 112 districts, and these districts were located in 11 out of Uganda’s 15 sub-regions (as defined by the Uganda Bureau of Statistics [20]). The vast majority of the groups were located in Central, South-East (Busoga), East (Teso), and Southwest (Ankole) Uganda (Table 1).

Table 1.
Probiotic yoghurt business and production data for the six sub-regions in which most of the production takes place.
2.2. Production Volume as Performance Indicator
Evaluation of the effectiveness of the intervention showed that 68% of all 170 trained groups turned into effective production units. The average production volume of these units is 209 ± 248 L per week. A total of 30 of these production units were in the business of regular yoghurt production, before they received additional support from the project and started to use the probiotic starter culture. These units produce an average volume of 413 L per week, as opposed to an average of 157 L per week among the 86 production units, which had no previous experience with dairy processing.
Other factors influencing performance related to gender (female- or male-owned), age (youth- or adult-owned), and governance structure (dairy cooperative, group or individually owned) of the production units. A dairy cooperative is an organization with typically over 100 farmer members, who elect a board that decides on the daily operations of the cooperative and employs staff to run the operations. Groups designated to yoghurt production are usually much smaller (5–20 members), typically do not employ people, but divide the work as well as the profits among themselves. Youth organized in groups reached the highest production volumes (Table 2). All these youth-groups were male-owned.

Table 2.
Average production volumes of production units categorized by gender, age, or governance structure.
Table 3 lists the production units that showed significant growth, i.e., a constantly rising production volume curve between 2016 and 2018, and production units that have either stable or fluctuating production volumes. Hence, business performance in terms of growth potential from 111 of the 116 active production units was analyzed. Growing production units were characterized by a steady increase in production volumes, while stable production units produced approximately the same volumes every week during the period of analysis. Fluctuating units were unstable in weekly production volumes and business development.

Table 3.
A total of 111 production units categorized by size and performance in terms of production volume development between 2016 and 2018. The number of production units in each category is given in absolute numbers as well as a percentage of the total number of active production units (five remaining production units had been in production for a short time and their performance trends could not be assessed).
Production units with growing production volumes were characterized by enthusiastic, motivated, ambitious and well-organized owners, who were continuously looking for new markets. Also, profits made from the yoghurt sales were reinvested in better and bigger equipment and premises with the aim to expand and upgrade the production capacity. Lastly, owners of growing production units were open to exploration and experimentation (i.e., they tried out probiotic yoghurt with different flavors, different packaging material, or ventured into new markets or market segments) and built good relationships with their clients as well as other stakeholders and partners.
Table 4 lists characteristics and reasons of production units that showed static or fluctuating production volumes. From all 179 production units trained, 24 production units were only in production for a number of months, but were not able to sustain the business.

Table 4.
Reasons for hampering business development at probiotic yoghurt production units.
Those who never started yoghurt business after receiving the training revealed financial constraints to buy packaging material and other start-up equipment, absence of electricity, or absence of potential yoghurt selling points equipped with fridges.
2.3. Sales and Income Generation
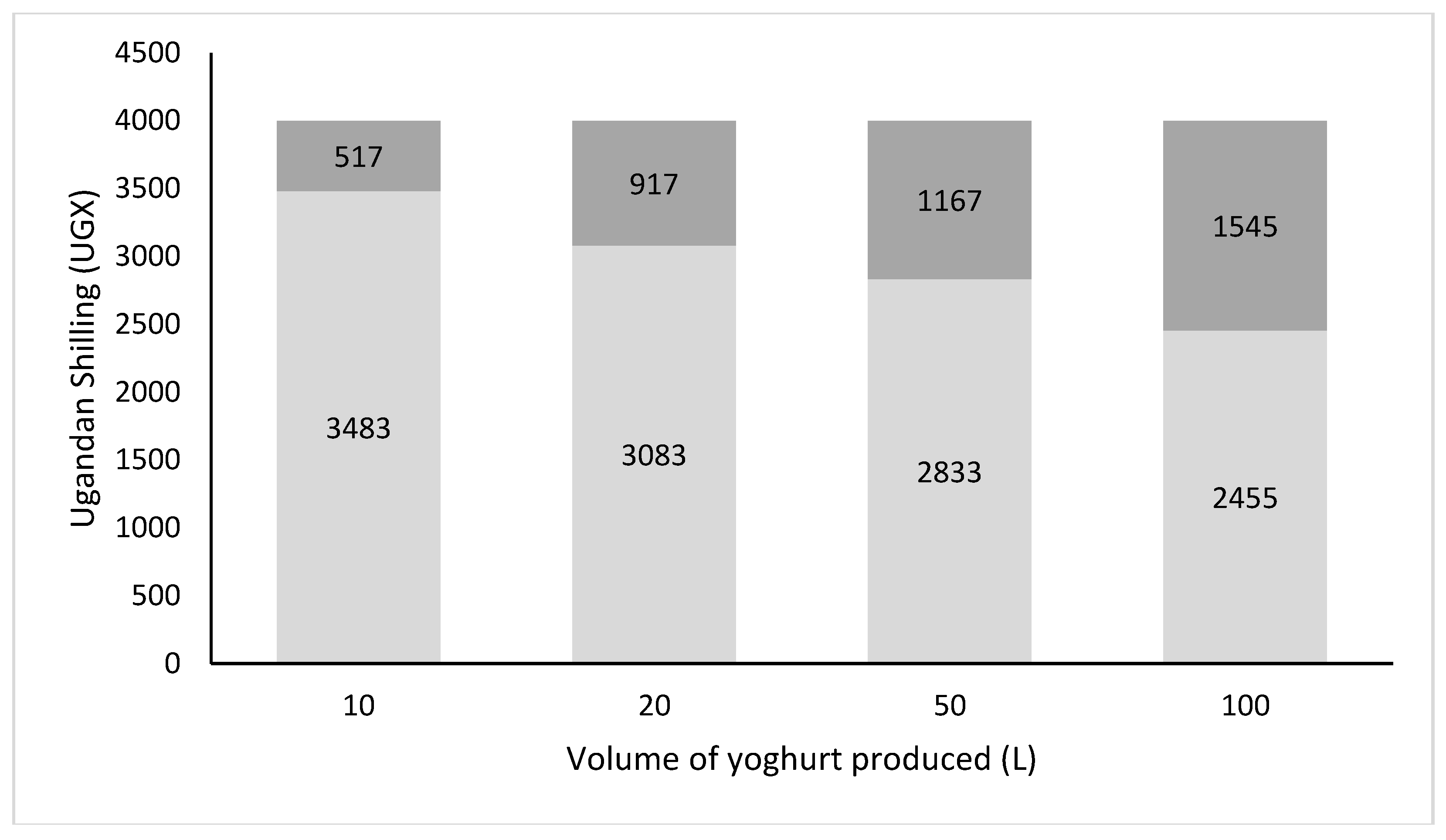
Despite a relatively low sales price, making the product affordable for the rural (poor) population, attractive profit margins were obtained, since transaction cost related to storage, transport, and mid-men were low and the yoghurt could be sold locally [21]. Figure 4 shows the revenues and profits generated by the production and sales of the probiotic Yoba yoghurt in Uganda. Profits could exceed 1600 UGX per liter yoghurt produced. The data also show that yoghurt production benefits from the economy of scale, thereby favoring those businesses that have more working capital, storage capacity and can afford the risk to produce larger batches of yoghurt. A detailed average cost breakdown is shown in Table S2. From consumption point of view, we noticed that the probiotic yoghurt could be a healthy alternative for soft drinks when the price of a portion remained slightly below the price of 1500 UGX that is usually charged for a soft drink.
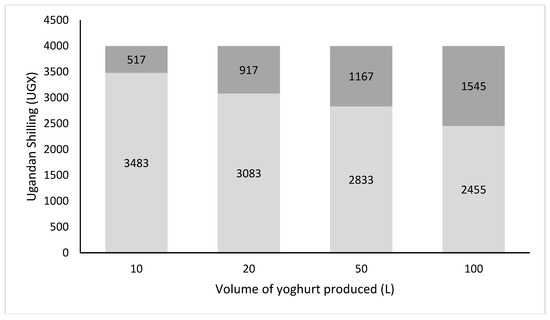
Figure 4.
Average cost and profits per liter of probiotic yoghurt in Uganda as function of production volumes. Light grey bars: Cost per liter of yoghurt (UGX); Dark grey bars: Profit (UGX); Total bars: Sales price per liter probiotic yoghurt. Note: Cost breakdown as shown in Table S2. Sales price is set at 4000 UGX per liter which resembles an average wholesale price when the yoghurt is sold through external shops.
A market survey conducted among 634 respondents, showed that there is almost complete ignorance among the consumers about the meaning of the word ‘probiotic’. However, this same survey showed that Ugandan consumers are (very) health conscious in their general purchase behavior, and express interest and appreciation for the concept of probiotic yoghurt when it is explained to them. On the downside, consumers are skeptical about health claims on packaging material. This is especially true in more rural areas where there is little regulation, and anybody can put anything on the market, including labels with far-fetched health claims.
2.4. Reduction of Milk Losses
Fresh milk in rural Uganda often has a very short shelf life, as a result of high initial bacterial load in the fresh milk originating from poor milking and milk handling practices in combination with a relatively high ambient temperature. In addition, refrigeration facilities are limited due to absence of adequate cooling equipment or power cuts. Prior to our intervention, milk collected by the farmers who lack cooling equipment had to be sold relatively fast before getting spoiled. Contrarily, when milk is fermented, the resulting probiotic yoghurt produced in this study has a shelf life of at least 4 weeks under refrigeration and does not spoil when exposed for several hours to ambient temperature. The intervention also includes milk quality tests, which was quite unknown in the informal milk sector comprising 80% of the total market [8]. Such quality tests stimulated farmers to work more hygienically and deliver clean, unadulterated, and antibiotic-free milk. As a consequence, milk spoilage could be further reduced.
2.5. Potential Health Benefits
Apart from the economic advantages, the second final outcome of the intervention is expected to be the enhancement of health of consumers by the probiotic Lactobacillus rhamnosus yoba 2012 strain present in the locally produced yoghurt [16]. This strain is the generic variant of world’s mostly studied probiotic bacterium L. rhamnosus GG. Probiotics are generally defined as “live microorganisms that, when administered in adequate amounts, confer a health benefit on the host” [22]. Although this intervention did not specifically measure generated health benefits, Table 5 provides an overview of the outcome of several studies previously conducted with L. rhamnosus GG. Since all produced probiotic yoghurts generated during the six years intervention were sold at prices below regular yoghurts, and especially below the widely available and very popular soda drinks, we conclude that the study succeeded to provide access to a healthy and affordable probiotic yoghurt that has the potential to alleviate commonly faced health challenges in sub-Saharan Africa. Based on a survey that counted and classified consumers at a representative number of retail outlets of the probiotic yoghurt, we estimate that due to this intervention at approximately 111 production units, 215,000 people per month (107,000 regular and 108,000 irregular) and 1.4 million people per year (107,000 regular and 1,302,000 irregular) consume probiotic yoghurt and hence can profit from expected health benefits.

Table 5.
A selection of commonly faced public health challenges in sub-Saharan Africa the related health benefits of L. rhamnosus GG (generic name: L. rhamnosus yoba 2012).
Although the efficacy of L. rhamnosus GG has been tested and confirmed in numerous independent studies (Table 5), health effects of the probiotic yoghurt containing this strain has not been tested to date on specific populations in Uganda. Therefore, the first pilot study in rural Uganda was conducted on children in 2017 at Aparisa-Asamuk primary school in Uganda. In order to get insight in the health status of the children, parents were asked if any of their children were ill or had suffered from any illness in the two weeks prior to the start of the study (baseline), during the study (midline) and at the end of the study (endline). Evaluation of the responses indicated a decrease in the rate of infection/illness as observed with increasing number of children with no illness throughout the study period, from 10% to 32% to 43% in the baseline, midline and endline, respectively. Most remarkable was the reduction in the occurrence of skin rashes as observed during skin inspection by a qualified nurse. Where at the start of the study 101 children were diagnosed with skin rashes, 5.5 weeks later this number had rapidly and significantly decreased to 15 (Table 6).

Table 6.
Interpretation and classification of skin inspections at three different time points among 245 children daily consuming probiotic yoghurt for 5.5 weeks.
The number of children who missed school during the study period also reduced continuously during the study from 40% in the baseline to 36% in the midline and to 23% in the endline.
3. Discussion
Although knowledge intensive technologies have been recognized as important drivers for development, they usually do not match the pressing needs of the region, do not reach their full potential, and often are not inclusive when generating products for the poor without inducing substantial degrees of empowerment [71]. The present intervention aimed to take up the challenge of using new technology in development work, ensure full participation of the targeted beneficiaries, make it sustainable and achieve impact on scale. The intervention started in 2012 by training the rural population on how to diversify their income from farming by processing locally produced milk into probiotic yoghurt in agreement with market standards and without high demands for (technical) resources.
3.1. Evaluation of Economic and Political Related Factors
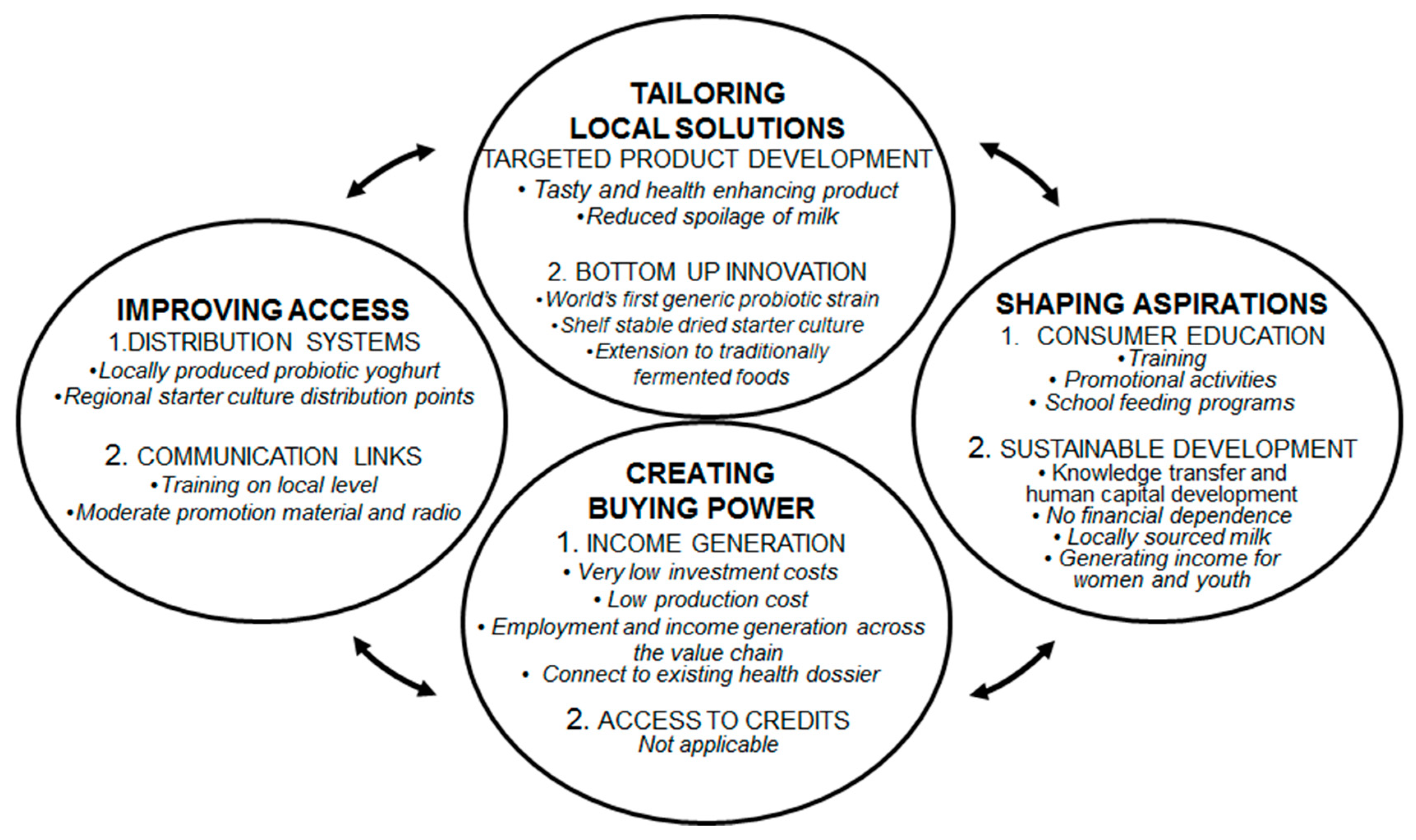
The increasing popularity and attention for probiotics in the western world by both consumers and the scientific world, and the associated growth in probiotic products, cannot be replicated directly in Africa. Hence, the intervention that enables local communities to produce and sell their own probiotic yoghurts was tailored to the local circumstances in a continuous process of searching for appropriate solutions [16,71,72,73,74,75]. The production of locally produced probiotic yoghurt by means of an affordable shelf stable bacterial starter culture can be seen as a bottom-up innovation that complies with local needs and capabilities, facilitates employment, increases purchasing power and improves access for many people to an affordable, tasty and healthy product that can be part of the daily diet. In addition, the described intervention empowers local producers and let them adjust product formulation, serving sizes, packaging, and distribution channels according to local wishes and practices.
In Uganda, typical characteristics of businesses with the highest growth rates historically relate to young businesses that start small, are male owned (as males are less risk-adverse compared to their female counterparts), and are operating in the manufacturing sector [76]. Although more than half of our production units were run by women, we noticed that the female-owned production units have lower average production volumes of 193 L/week, compared to male-owned production units, who reported an average of 257 L/week (see Table 2). This indicates that male owned businesses are larger and potentially more successful.
The activities described in this paper followed a bottom-up approach, without active support from authorities. From a more top-down perspective we can say that African governments focus on urban, medium-large scale formal industries and pay relatively little attention to rural, informal, and usually small-scale industries, as we targeted by the probiotic yoghurt concept. For example, stringent regulations and high costs associated with certification (from the Uganda National Bureau of Standards) seem to aim to discourage small-scale producers from even trying to come up, and thereby favoring the bigger, well-established businesses. However, evidence from China indicates that labor-intensive township and village enterprises (TVE’s) are an engine for growth, and can offer distributional benefits through lowering prices for the poor [2]. Rural poverty reduction strategy in Africa is more likely to be effective when it harnesses the potential of the nonfarm sector, as illustrated by the local production of probiotic yoghurt. Furthermore, increased consumption of probiotics among the population might be an efficient strategy to cost-effectively improve public health. It could be worth for policy makers to consider the economic impact of allocating health care budget to promoting the consumption of probiotics for those who might need it most [16,71,77,78,79]. Furthermore, in order to increase income and reduce non-income related poverty-including school attendance, health, nutrition, safety nets, and household food security-, it remains essential to build well-developed public services, especially education and infrastructure that facilitate access to markets.
The lack of effective support from authorities are not the only challenges related to the political environment around the intervention. For instance, cronyism and corruption favors politicians and the politically connected at the expense of those who are not, most especially at district and local level. The privileged can avoid paying taxes and have greater access to financial resources and information. One way in which production units can harness themselves against such practices is to cooperate in a network and support each other by sharing knowledge about best practices, practices to avoid or to be aware of, input suppliers and other opportunities. In other words, apart from the technical aspects, also socio-economic, political, and cultural factors are essential to successfully implement bottom-up innovations as described in this paper.
3.2. Stimulating Demand
Although from the supply side we do not expect major hurdles for further expansion of the locally made probiotic fermented food, stimulating demand in Uganda might be more challenging, especially in rural areas where awareness on health benefits of probiotic yoghurts is almost non-existent. Frequently used media to raise consumer awareness such as radio, local television, and satellite television have limited applicability, for instance because location and fees of the avenues as well as the viewing-time and male focused content restricts women from benefitting from this development [6].
During our intervention, the majority of the demand for the yoghurt produced was not driven by its special probiotic attribute, but mainly by the quality, taste and affordability of the product. A market survey showed that this was due to ignorance about the product, not due to a lack of interest in health promoting products. We therefore aim for the future to put more emphasis on trainings and interactions with the producers on business skills and product knowledge, as means to further empower local producers to educate customers on probiotic fermented food and stimulate the demand side [6,80]. Furthermore, we envision that active engagement of policy makers can also result in further awareness and stimulation of consumption, as can be concluded from a government sponsored probiotic yoghurt school feeding project in Argentina [71]. This initiative started gaining significant attention of all potential stakeholders involved after a locally conducted double blind trial, involving young children consuming probiotic yoghurt, clearly showed improvements in a large number of health factors including the reduction of diarrhea. Notwithstanding the solid health dossier of the generic probiotic strain used in our intervention, we envision that the results of local health impact studies in Africa, as are currently running in Uganda with probiotic yoghurt, will help to gain further attention and acceptance for probiotic products by the targeted consumers and other stakeholders. Another strong element of the Argentinian project is the inclusion of lectures on probiotics in the school curriculum in combination with field visits to production units and laboratories. This increases awareness among children, and their parents, for a healthy diet and nutrition in general and benefits of consumption of probiotic fermented food in particular. Hereto NGOs The Dutch Development Organization (SNV) and Yoba for Life Foundation recently started the implementation of the pre-primary probiotic program. In this program, parents are encouraged to pay approximately 3 USD per school term of 3 months, for their child to take 125 mL of probiotic yoghurt twice per week, as produced by one of the local probiotic yoghurt producers. Preliminary results of this program are very promising, as in a period of 6 months already 5,000 children enjoy yoghurt twice per week, as paid for by their parents.
3.3. Regional Differences
In terms of number of production units and cumulative yoghurt production volume, the uptake of the intervention is the highest in the South-West, an area covering about 7% of Uganda. This region is among the wealthiest parts of Uganda (after Kampala) [20], responsible for 34% of the produced milk in the country [7], and contains 67% of the milk collection centers [8]. The local milk-culture already includes the consumption of naturally fermented milk. Many people have their own relatively large dairy farms with on average 25 cows, and are therefore interested in value addition to their produce. In terms of gender balance, many yoghurt producing entities in these regions are deployed by women groups, which can be seen from the fact that 66%–75% of the people involved are women, with an average group size of 6.5–9.8 people (Table 1). These are commonly women above a certain age (seen from the low number of youth involved) who are not fully occupied with the care for young children anymore and can free up time for other activities [81]. Historically, the women in this area are in charge of the production of traditionally fermented milk called ‘bongo’ or ‘makamo’. This region also has a strong social cohesion and a group-culture, as is reflected by the relatively large average number of people actively involved in production units [81].
In South-East Uganda, where poverty rates are four times higher compared to South-West Uganda [20], preparing yoghurt is not seen as a social activity for groups, rather as a livelihood strategy [82]. Businesses are mainly deployed as a form of income diversification by older male individuals or male-owned dairy cooperatives (75% male-owned, but 0% youth-owned) who employ young men to do the work (Table 1). The population in this region is less likely to unite in groups compared to the national average [81] due to financial constraints related to entrance barriers (subscription fees), low direct returns when initial profits are retained by the groups, and the luxury of ‘leisure’ for spending time in groups. With an average of 2.6 people involved per production unit and the lowest average weekly volumes of 115 L per week, this region is dominated by scattered individual small businesses (Table 1). Because of these lower levels of social capital and trust, governance issues of production units that work in collectives or cooperative setting have proven to be a challenge in this area. In order to reach a large group of consumers in East Uganda where the purchasing power of the population is small, yoghurt selling prices are lower compared to other areas.
The Teso sub-region is a rather remote, rural, and less developed area. Due to the large distance from Kampala, which is the economic center of Uganda, and the poor infrastructure in large parts of the region, not many industrial yoghurt companies have penetrated this area. Yet this region has a culture of consuming locally fermented milk, and yoghurt is hence a popular product, as long as it is sold at affordable prices. Local yoghurt producers hence find themselves almost a monopoly in a wide area, and venture into production of large volumes with low profit margins. This is reflected the highest average production volumes compared to other sub-regions. Besides, youth ownership and participation are the highest in this region (Table 1).
Contrary to South-West and East Uganda, where a larger proportion of the producers lives in deep rural areas and the probiotic yoghurt is supplied at wholesale price to for example shops, restaurants, and/or schools, the yoghurt producers in Central Uganda are mostly located in trading centers and urban areas. These regions comprise of a mixture of ethnic groups and their sub-cultures, hence no clear trends in production characteristics could be derived.
3.4. Health Benefits
Although in the school study in Aparisa-Asamuk primary school no placebo was used and the occurrence of seasonal influences cannot be excluded, during the 5.5 weeks of daily consumption of probiotic yoghurt with the L. rhamnosus yoba 2012 bacterium we observed a decrease in the occurrence of illness and use of medication, as indicated by the parents, a reduction of the incidence of skin rashes, as indicated by a nurse, and a decrease of absenteeism from school, as indicated by the teachers. Similar to what has been reported in an Argentinean intervention study with probiotic yoghurt [83], the obtained data suggests that the daily provision of 100 mL of probiotic yoghurt could bring health benefits and increase school attendance. In order to better investigate the health promoting benefits of probiotic yoghurt, we recommend to perform a placebo-controlled study with school children in Uganda over a longer time period. Ideally, in such a such a two-arm study, the health status of children consuming the probiotic yoghurt containing L. rhamnosus yoba 2012 is monitored and compared to a group of children consuming an equal amount of non-fermented milk.
3.5. Outlook
At the conducted scale, the concept presented here has shown to function as a rural poverty reduction strategy. Since the probiotic starter culture can also be used for the fermentation of cereals such as millet and corn [73,84], the scope of application can further extend. This is particularly relevant for the purpose of detoxification (reduction of aflatoxins) [54]. We envision that this concept and the direct way of working together with the local communities can also be implemented in other African countries. For future expansion, moderate financial and institutional sustainability needs to be granted. Ideally, African governments should be brought on board as the local production of affordable healthy foods directly affects health economic related parameters and general economic development opportunities [71]. Furthermore, a favorable political climate, including access to education, infrastructure, and finance, will positively impact business outcomes [21]. Table S3 summarizes the mechanisms that are in our view needed to increase the number of sustainable yoghurt production units, or related businesses. The greater income diversification through non-farm activities is expected to cause more rapid growth in earnings and consumption. This will induce a positive feedback loop, wherein those participating in the rural non-farm economy will gain faster income growth, which can be invested again into expanded farm or nonfarm activities. Subsequently, rural development is expected to be improved and poverty to be reduced.
4. Methods
4.1. Intervention Set-Up
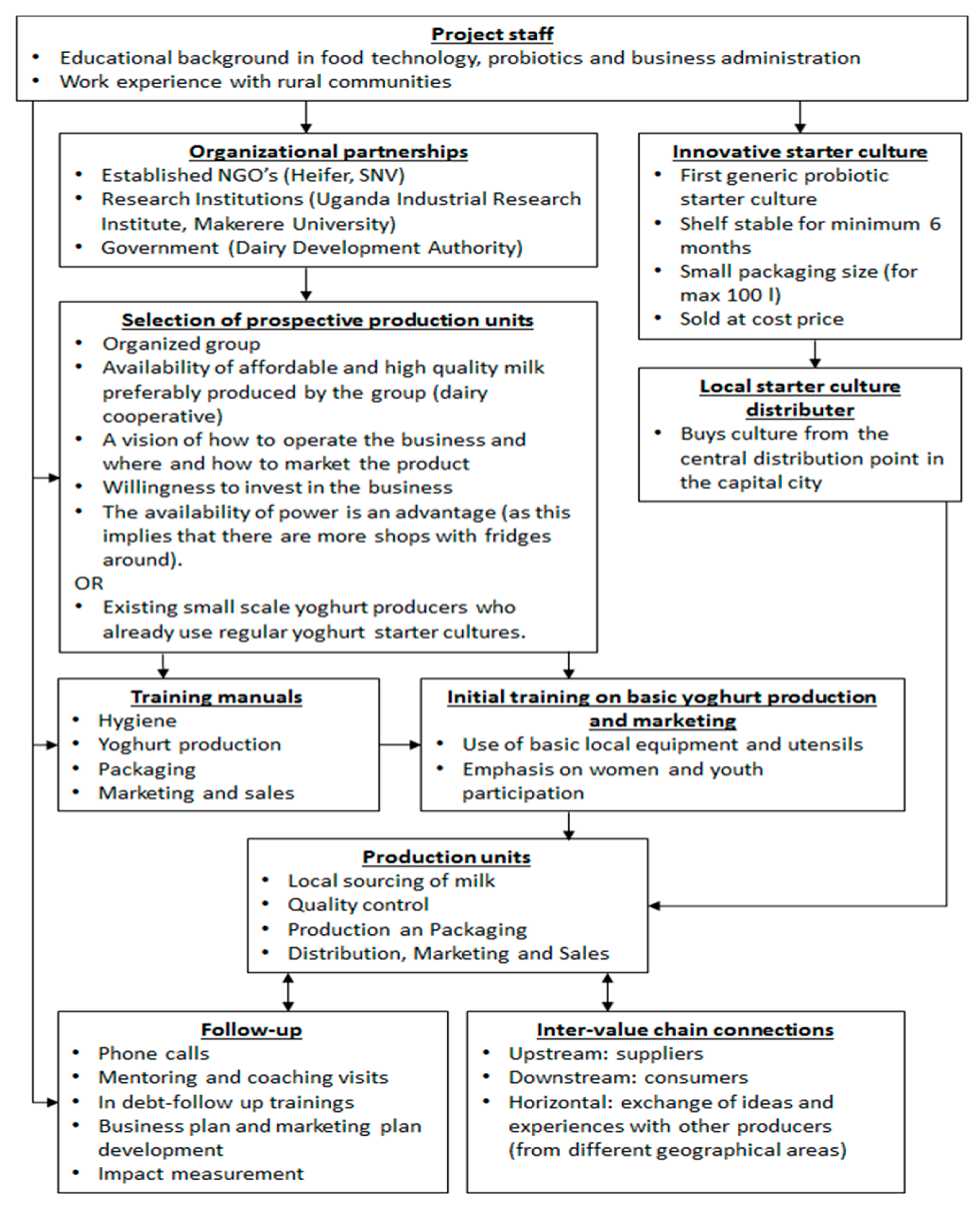
A schematic overview of the intervention set-up is given in Figure 5. One of the key components is connecting to already existing dairy communities and/or small-scale dairy processors. Next, farmers and dairy processors are taught how to upgrade fresh milk production to probiotic yoghurt business. Focus is on hygiene and quality awareness, technical capacity building, packaging, as well as marketing, business administration and creation of linkages within the value-chain. In order maximize the sustainability of the concept, the processing units invest in equipment and raw materials, without any financial support, and have full ownership over their yoghurt production and sales. Only the training and coaching are provided for free.
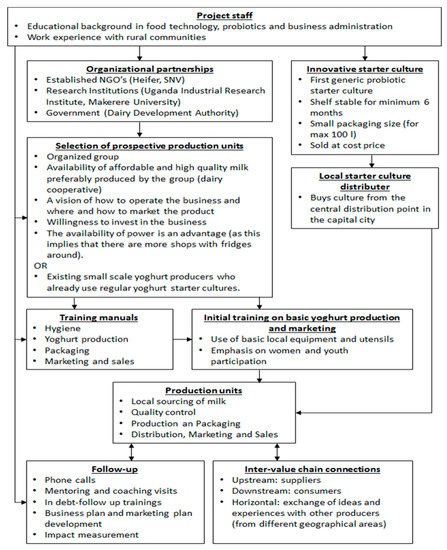
Figure 5.
Systematic outline of the set-up of the intervention of locally produced probiotic yoghurt. The bacterial starter culture and the specific production protocol are described by Kort et al. [73] and Westerik et al. [74], respectively.
4.2. Production of Probiotic Yoghurt
The characteristics of the probiotic starter culture and its specific use have recently been described [73,74]. In short, the dried starter culture consists of L. rhamnosus yoba 2012 and an adjuvant strain Streptococcus thermophilus C106. Upon rehydration in milk and at a temperature of approximately 37 °C, the bacteria revitalize and start growing and converting the raw milk into yoghurt. The S. thermophilus strain enables the lactose negative L. rhamnosus strain to propagate in milk. The L. rhamnosus yoba 2012 strain is the generic variant of world’s best documented probiotic bacterium L. rhamnosus GG [72]. Sachets containing one gram of starter culture were produced by the Yoba for Life Foundation (Amsterdam, The Netherlands) in collaboration with CSK Food Ingredients (Leeuwarden, The Netherlands), Lactosan (Kapfenberg, Austria). The sachets are sold from regional distribution centers to yoghurt producers for a price of 2000 UGX (approx. 0.56 US dollar) per sachet. 1500 UGX covers the sachet production and distribution cost, and 500 UGX is a compensation for the sachet distribution center. One gram of starter culture allows for the production of up to 100 L of probiotic yoghurt.
Prior to production, the quality of the locally sourced milk is tested by the producers. After production, the yoghurt is packed in polythene bags, bottles, or small jerry cans, with volumes ranging between 125 mL and 5 L. Producers decide on the addition of colorants, sugar, and flavors like strawberry, vanilla, or mango to adjust color and taste according to local consumer demands and national food standards. Occasionally, producers use potassium sorbate from local suppliers as an accepted food preservative. Yoghurt is sold either at the production site, distributed to trading centers or nearby selling points like kiosks and supermarkets.
In order to maximize ownership for the probiotic yoghurt production by each production unit, yoghurts are sold under the brand name of the local producer. A brand added benefit mark, in form of a “Yoba inside” stamp, is used to differentiate the probiotic yoghurt from non-probiotic variants, see examples in Figure S1. Besides the empowerment of local communities in setting-up their own business in probiotic yoghurt, a second component of the intervention is the contribution to the improvement of health by creating access for the (rural) Ugandan population to locally produced yoghurt that is naturally enriched with the probiotic strain L. rhamnosus GG.
4.3. Participating Dairy Companies and NGOs
In the period 2012–2018, 170 dairy companies, including cooperatives, existing dairy processing factories, women groups and new starting entrepreneurs received training for production of the so-called Yoba probiotic yoghurt. Bacterial starter culture and training materials were developed by the Yoba for Life Foundation (Amsterdam, NL). Access to the dairy cooperative networks was provided through local branches of Heifer International under the East African Dairy Development project and later under the Fermented Food for Life project, and Dutch Development Organization SNV under The Inclusive Dairy Enterprise project. The main partners collaborating in this intervention and their specific roles are listed in Table S1.
4.4. Data Collection and Evaluation
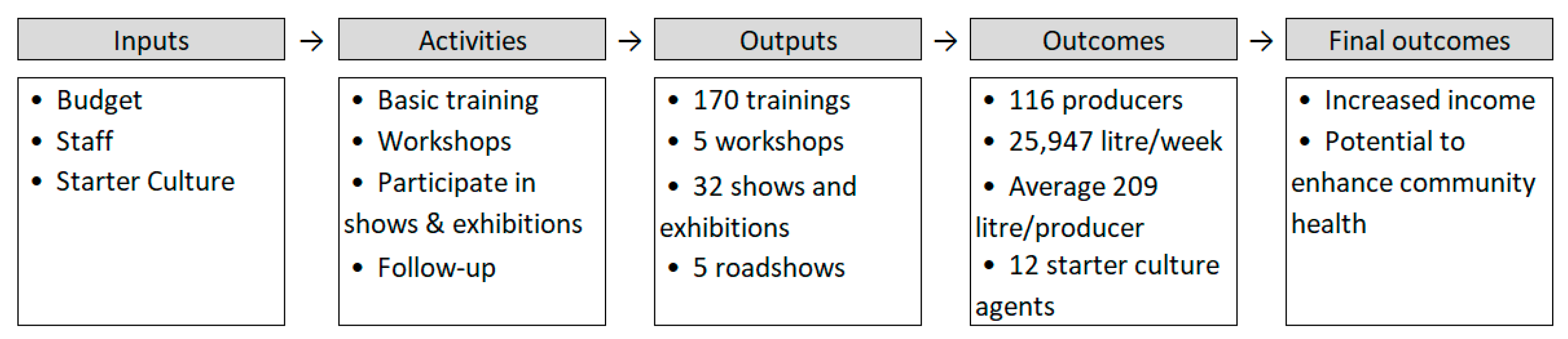
The intervention was studied by means of a non-experimental longitudinal descriptive study. The study represents a first inquiry and assessment of the possible short and long-term outcomes of the intervention, over a period of six years. This study therefore forms a starting point for further impact evaluation, which may confirm and quantify inferences or causal relationships proposed by the current study. A result chain as shown in Figure 6 was guiding the investigations in this study [85]. During the present evaluation two main hypotheses have been assessed: (1) Upon training and by providing access to bacterial starter cultures, local groups and entrepreneurs will be able to set up a successful yoghurt business. (2) A probiotic yoghurt production business will lead to additional income under the local circumstances. In addition, based on available data from previously conducted studies, we have evaluated the potential health impact on consumers upon frequent probiotic yoghurt consumption.
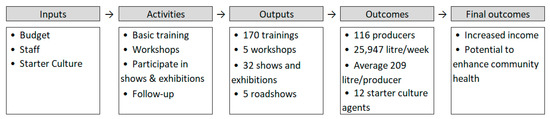
Figure 6.
Result chain of the intervention. The first three stages (inputs, activities, and outputs) are controlled by the intervention. The outcomes are achieved when the beneficiaries choose to use the opportunities created by the intervention. The final outcomes represent the goal of the intervention.
Data collection methods included periodic interviews, written inquiries, and focus group discussions with the producers and other stakeholders, like NGOs operating in the dairy sector, local leaders, and players in the fresh milk sector, as well as the government represented by the Dairy Development Authority. Questions related to (i.) production volumes, (ii.) number, age, and gender of people involved in yoghurt production and sales, and (iii.) type of products produced (size, packaging material, and price). A market survey was conducted among 634 respondents in urban, semi-urban and rural areas about their perception of the concept of probiotic yoghurt, and their general consumption preferences for similar products. In addition, anecdotal data was obtained through continuous face-to-face interactions with the production units and on-site observations. Probiotic yoghurt producers were categorized as dairy cooperatives, collectives, or individual dairy processors with or without prior experience in yoghurt production.
Health benefits were investigated by means of literature review looking at clinical trials and health impact assessments of the consumption of Lactobacillus rhamnosus GG. Besides, a nutritional trial testing the health impact of consumption of Yoba yoghurt was conducted in 2017 at Aparisa-Asamuk primary school in Uganda. A total of 245 pupils aged between 3 and 10 received 100 mL of probiotic yoghurt every day from Monday till Friday for 5.5 weeks. Yoghurt was consumed in addition to the regular family meals at the children’s homes or at school as packed lunch. The Yoba yoghurt had an estimated caloric load of 80 kCal and a cell count of approximately 2.5 × 107 CFU/mL, implying that portions of 100 mL contain 2.5 × 109 CFU. By means of a questionnaire parents of the children were asked about the health status of the children and the use of medication before, during, and at the end of the intervention. With the same frequency, a nurse checked the condition of the skin and the teachers were asked to monitor the absence level of the pupils.
Probiotic yoghurt containing L. rhamnosus yoba 2012 and S. thermophilus C106 was prepared by Amuria Farmers’ Cooperative Society Limited, Amuria town, Amuria, Uganda, on a daily basis in agreement with the Yoba for Life production protocols [74]. The yoghurt was packed in 100 mL polythene packaging bags as supplied by Heifer International. The cooperative was responsible for daily transport of the packed yoghurt to the study site. Yoghurt had to be delivered before 12 PM from Monday–Friday. Data was analyzed using SPSS software, ENA for Smart and Anthroplus software.
5. Conclusions
We conclude that the combination of a training program and access to an affordable and stable probiotic starter culture to increase health and wealth of local communities by the use of existing infrastructures and preferences can be taken as a positive reference for deployment of self-sustainable activities that contribute to value addition at multiple sites along the food chain in sub Saharan Africa.
Supplementary Materials
The following are available online at http://www.mdpi.com/2078-1547/10/1/2/s1, Figure S1: Figure S1. Picture of the “Yoba Inside” brand added benefit mark and two examples of packaging design used by local producers, Table S1: Improving Health and Wealth by an intervention with an affordable starter for locally produced probiotic yoghurt in Uganda—Main partners, Table S2: Average cost and profits per liter of probiotic yoghurt in Uganda as function of production volumes. Note: Sales price is set at 4.000 UGX per liter, Table S3: Mechanisms behind the implementation of sustainable probiotic yoghurt production units and related effects. Inspired by the McKinsey Psychology of Change Management (Lawson E, Price C. The psychology of change management. McKinsey Q. 2003;30–41).
Author Contributions
Conceptualization, R.K., W.S., W.M. and G.R.; Methodology, N.W., A.P.W., W.M. and E.A.; Investigation, E.A., N.W.; Writing—Original Draft Preparation, N.W.; Writing—Review & Editing, A.P.W., G.R., R.K. and W.S.
Funding
Funding was received from the International Development Research Centre (IDRC), Canada (project number 108122) and the Yoba for Life Foundation.
Acknowledgments
We thank all partners described in Table S1 for their support. We especially would like to acknowledge Heifer Uganda team, led by Susan Atyang, for the early collaboration within the East Africa Dairy Development Project. We thank Robert Sebbandeke and Arinda Nelson for their tireless efforts in the Fermented Food for Life project, which led to the outcomes as presented in this paper. We gratefully acknowledge Rinus van Klinken, project manager of the Inclusive Dairy Enterprise project (SNV) for his support, guidance and advise for the yoghurt programs, as well as for his critical reading of this article. We acknowledge Amuria Farmers’ Cooperative Society Ltd. for the daily preparation of yoghurt and Joseph Okia for the daily supply of yoghurt to Aparisa Primary School during the school feeding study. We thank Mary Amuge and the Staff of Aparisa primary school for all support during data collection.
Conflicts of Interest
Remco Kort and Wilbert Sybesma are co-founders of the Yoba for Life foundation (2009), a non-profit organization, accredited by the Dutch Tax Authorities as a Public Benevolent Institution (PBI), which aims to promote local production and consumption of fermented products in Africa. Nieke Westerik is the Country Coordinator of the Yoba for Life Foundation in Uganda. Alex Paul Wacoo is a part-time employee and associated researcher at the Yoba for Life Foundation in Uganda. African fermented products made with the Yoba starter culture, are not marketed by the foundation as such, but the Yoba for Life foundation stimulates local production and ownership, allowing income-generating activities for African small-scale entrepreneurs in the food sector. The Yoba for Life foundation distributes and sells ready-to-use sachets with dried bacterial starter cultures at cost price, through a network of partners and volunteers to facilitate the local production of dairy and cereal-based products by controlled bacterial fermentation. The other authors declare no conflict of interest.
References
- Bahiigwa, G.; Mdoe, N.; Ellis, F. Livelihoods research findings and agriculture-led growth. IDS Bull. 2005, 36, 115–120. [Google Scholar] [CrossRef]
- Lanjouw, J.O.; Lanjouw, P. The rural non-farm sector: Issues and evidence from developing countries. Agric. Econ. 2001, 26, 1–23. [Google Scholar] [CrossRef]
- Barrett, C.B.; Reardon, T.; Webb, P. Nonfarm income diversification and household livelihood strategies in rural Africa: Concepts, dynamics, and policy implications. Food Policy 2001, 26, 315–331. [Google Scholar] [CrossRef]
- Ajani, E.N.; Igbowke, E.M. Prospects of occupational diversification among rural women in sub-Saharan Africa: A review. J. Agric. Ext. Rural Dev. 2014, 6, 69–74. [Google Scholar]
- Uganda Bureau of Statistics (UBOS). Uganda National Household Survey 2012/13; UBOS: Kampala, Uganda, 2014.
- Livingstone, J.; Ruhindi, E. Economic Empowerment for pastoralist Women: A Comparative Look at Program Experience in Uganda, Somaliland and Sudan; Land Deals Politics Initiative: Sussex, UK, 2011. [Google Scholar]
- Balikowa, D. A Review of Uganda’s Dairy Industry, Dairy Development in Uganda; Dairy Development Authority: Kampala, Uganda, 2011.
- East Africa Dairy Development Project Uganda Dairy Market Study Report; Tig Marketing Consultant: Kampala, Uganda, 2015.
- McKenney, C. Growth Projections | The Atlas of Economic Complexity. Available online: http://atlas.cid.harvard.edu/rankings/growth-predictions/ (accessed on 2 September 2017).
- World Bank GDP per Capita (Current US$) | Data. Available online: https://data-worldbank-org.vu-nl.idm.oclc.org/indicator/NY.GDP.PCAP.CD (accessed on 9 September 2017).
- Lagarde, C.; Kampala, I.M.D. Becoming the Champion: Uganda’s Development Challenge. Available online: https://www-imf-org.vu-nl.idm.oclc.org/en/News/Articles/2017/01/27/ sp01272016-Becoming-the-Champion-Ugandas-Development-Challenge (accessed on 9 September 2017).
- UN FAO Uganda at a Glance | FAO in Uganda. Available online: http://www.fao.org/uganda/fao-in-uganda/uganda-at-a-glance/en/ (accessed on 7 August 2018).
- World Bank Uganda | Data. Available online: https://data.worldbank.org/country/uganda (accessed on 7 August 2018).
- Uganda Bureau of Statistics (UBOS); ICF. Key Indicators Report; UBOS; ICF: Kampala, Uganda; Rockville, MD, USA, 2017.
- World Health Organization GHO | By Category | Rate of Deaths by Country—Uganda. Available online: http://apps.who.int/gho/data/view.main.ghe2002015-UGA?lang=en (accessed on 17 April 2018).
- Westerik, N.; Kort, R.; Sybesma, W.; Reid, G. Lactobacillus rhamnosus probiotic food as a tool for empowerment across the value chain in Africa. Front. Microbiol. 2018, 9, 1501. [Google Scholar] [CrossRef]
- Franz, C.M.A.P.; Huch, M.; Mathara, J.M.; Abriouel, H.; Benomar, N.; Reid, G.; Galvez, A.; Holzapfel, W.H. African fermented foods and probiotics. Int. J. Food Microbiol. 2014, 190, 84–96. [Google Scholar] [CrossRef]
- Prahalad, C.K.; Hart, S.L. Strategies for the Bottom of the Pyramid: Creating Sustainable Development. Available online: http://pdf.wri.org/2001summit_hartarticle.pdf (accessed on 7 December 2018).
- Van Klinken, R. Uganda Poised to Become the Largest Dairy Exporter in Africa. Available online: http://www.busiweek.com/uganda-largest-dairy-exporter/ (accessed on 16 August 2018).
- Uganda Bureau of Statistics (UBOS). Uganda National Household Survey 2016/17; UBOS: Kampala, Uganda, 2018.
- Deininger, K.; Okidi, J. Rural households: Incomes, productivity, and nonfarm enterprises. In Uganda’s Recovery: The Role of Farms, Firms, and Government; The World Bank: Washington, DC, USA, 2001; pp. 123–175. [Google Scholar]
- FAO/WHO Working Group. Guidelines for the Evaluation of Probiotics in Food; FAO; WHO: London, UK, 2002. [Google Scholar]
- Okeke, I.N.; Aboderin, O.A.; Byarugaba, D.K.; Ojo, K.K.; Opintan, J.A. Growing problem of multidrug-resistant enteric pathogens in Africa. Emerg. Infect. Dis. 2007, 13, 1640–1646. [Google Scholar] [CrossRef]
- Fujita, K.; Kaku, M.; Yanagase, Y.; Ezaki, T.; Furuse, K.; Ozawa, A.; Saidi, S.M.; Sang, W.K.; Waiyaki, P.G. Physicochemical characteristics and flora of diarrhoeal and recovery faeces in children with acute gastroenteritis in Kenya. Ann. Trop. Paediatr. 1990, 10, 339–345. [Google Scholar] [CrossRef]
- Mshana, S.E.; Joloba, M.; Kakooza, A.; Kaddu-Mulindwa, D. Campylobacter spp among Children with acute diarrhea attending Mulago hospital in Kampala—Uganda. Afr. Health Sci. 2009, 9, 201–205. [Google Scholar]
- Burkholder, K.M.; Bhunia, A.K. Salmonella enterica serovar Typhimurium adhesion and cytotoxicity during epithelial cell stress is reduced by Lactobacillus rhamnosus GG. Gut Pathog. 2009, 1, 14. [Google Scholar] [CrossRef]
- Castillo, N.A.; de Moreno de LeBlanc, A.; Galdeano, C.M.; Perdigón, G. Probiotics: An alternative strategy for combating salmonellosis: Immune mechanisms involved. Food Res. Int. 2012, 45, 831–841. [Google Scholar] [CrossRef]
- Marianelli, C.; Cifani, N.; Pasquali, P. Evaluation of antimicrobial activity of probiotic bacteria against Salmonella enterica subsp. enterica serovar typhimurium 1344 in a common medium under different environmental conditions. Res. Microbiol. 2010, 161, 673–680. [Google Scholar] [CrossRef]
- Nomoto, K. Prevention of infections by probiotics. J. Biosci. Bioeng. 2005, 100, 583–592. [Google Scholar] [CrossRef]
- Xu, H.; Jeong, H.S.; Lee, H.Y.; Ahn, J. Assessment of cell surface properties and adhesion potential of selected probiotic strains. Lett. Appl. Microbiol. 2009, 49, 434–442. [Google Scholar] [CrossRef]
- Keersmaecker, S.C.J.D.; Verhoeven, T.L.A.; Desair, J.; Marchal, K.; Vanderleyden, J.; Nagy, I. Strong antimicrobial activity of Lactobacillus rhamnosus GG against Salmonella typhimurium is due to accumulation of lactic acid. FEMS Microbiol. Lett. 2006, 259, 89–96. [Google Scholar] [CrossRef]
- Hütt, P.; Shchepetova, J.; Lõivukene, K.; Kullisaar, T.; Mikelsaar, M. Antagonistic activity of probiotic lactobacilli and bifidobacteria against entero- and uropathogens. J. Appl. Microbiol. 2006, 100, 1324–1332. [Google Scholar] [CrossRef]
- Rinkinen, M.; Jalava, K.; Westermarck, E.; Salminen, S.; Ouwehand, A.C. Interaction between probiotic lactic acid bacteria and canine enteric pathogens: A risk factor for intestinal Enterococcus faecium colonization? Vet. Microbiol. 2003, 92, 111–119. [Google Scholar] [CrossRef]
- Kosek, M.; Bern, C.; Guerrant, R.L. The global burden of diarrhoeal disease, as estimated from studies published between 1992 and 2000. Bull. World Health Organ. 2003, 81, 197–204. [Google Scholar]
- Parashar, U.D.; Burton, A.; Lanata, C.; Boschi-Pinto, C.; Shibuya, K.; Steele, D.; Birmingham, M.; Glass, R.I. Global mortality associated with rotavirus disease among children in 2004. J. Infect. Dis. 2009, 200, S9–S15. [Google Scholar] [CrossRef]
- Szajewska, H.; Skórka, A.; Ruszczyński, M.; Gieruszczak-Białek, D. Meta-analysis: Lactobacillus GG for treating acute diarrhoea in children. Aliment. Pharmacol. Ther. 2007, 25, 871–881. [Google Scholar] [CrossRef]
- Guandalini, S. Probiotics for prevention and treatment of diarrhea. J. Clin. Gastroenterol. 2011, 45, S149–S153. [Google Scholar] [CrossRef]
- Guarino, A.; Guandalini, S.; Vecchio, A.L. Probiotics for prevention and treatment of diarrhea. J. Clin. Gastroenterol. 2015, 49, S37–S45. [Google Scholar] [CrossRef]
- Allen, S.J.; Martinez, E.G.; Gregorio, G.V.; Dans, L.F. Probiotics for treating acute infectious diarrhoea. Cochrane Database Syst. Rev. 2010, 11, CD003048. [Google Scholar] [CrossRef]
- De Roos, N.M.; Katan, M.B. Effects of probiotic bacteria on diarrhea, lipid metabolism, and carcinogenesis: A review of papers published between 1988 and 1998. Am. J. Clin. Nutr. 2000, 71, 405–411. [Google Scholar] [CrossRef]
- Larcombe, S.; Hutton, M.L.; Lyras, D. Involvement of bacteria other than Clostridium difficile in antibiotic-associated diarrhoea. Trends Microbiol. 2016, 24, 463–476. [Google Scholar] [CrossRef]
- Okeke, I.N.; Lamikanra, A.; Edelman, R. Socioeconomic and behavioral factors leading to acquired bacterial resistance to antibiotics in developing countries. Emerg. Infect. Dis. 1999, 5, 18–27. [Google Scholar] [CrossRef]
- Hawrelak, J.A.; Whitten, D.L.; Myers, S.P. Is Lactobacillus rhamnosus GG effective in preventing the onset of antibiotic-associated diarrhoea: A systematic review. Digestion 2005, 72, 51–56. [Google Scholar] [CrossRef]
- Kate, V.; Ananthakrishnan, N.; Tovey, F.I. Is Helicobacter pylori infection the primary cause of duodenal ulceration or a secondary factor? A Review of the Evidence. Gastroenterol. Res. Pract. 2013, 2013, e425840. [Google Scholar] [CrossRef]
- Tovey, F.I. Staple diets and duodenal ulcer prevalence. Int. Health 2009, 1, 124–132. [Google Scholar] [CrossRef]
- Newton, R.; Ziegler, J.L.; Casabonne, D.; Carpenter, L.; Gold, B.D.; Owens, M.; Beral, V.; Mbidde, E.; Parkin, D.M.; Wabinga, H.; et al. Helicobacter pylori and cancer among adults in Uganda. Infect. Agents Cancer 2006, 1, 5. [Google Scholar] [CrossRef]
- Westerik, N.; Reid, G.; Sybesma, W.; Kort, R. The probiotic Lactobacillus rhamnosus for alleviation of Helicobacter pylori-associated gastric pathology in East Africa. Front. Microbiol. 2018, 9, 1873. [Google Scholar] [CrossRef]
- Myllyluoma, E.; Kajander, K.; Mikkola, H.; Kyrönpalo, S.; Rasmussen, M.; Kankuri, E.; Sipponen, P.; Vapaatalo, H.; Korpela, R. Probiotic intervention decreases serum gastrin-17 in Helicobacter pylori infection. Dig. Liver Dis. 2007, 39, 516–523. [Google Scholar] [CrossRef]
- Hamilton-Miller, J.M.T. The role of probiotics in the treatment and prevention of Helicobacter pylori infection. Int. J. Antimicrob. Agents 2003, 22, 360–366. [Google Scholar] [CrossRef]
- Gotteland, M.; Cruchet, S.; Verbeke, S. Effect of Lactobacillus ingestion on the gastrointestinal mucosal barrier alterations induced by indometacin in humans. Aliment. Pharmacol. Ther. 2001, 15, 11–17. [Google Scholar] [CrossRef]
- Lam, E.K.Y.; Tai, E.K.K.; Koo, M.W.L.; Wong, H.P.S.; Wu, W.K.K.; Yu, L.; So, W.H.L.; Woo, P.C.Y.; Cho, C.H. Enhancement of gastric mucosal integrity by Lactobacillus rhamnosus GG. Life Sci. 2007, 80, 2128–2136. [Google Scholar] [CrossRef]
- Lam, E.K.Y.; Yu, L.; Wong, H.P.S.; Wu, W.K.K.; Shin, V.Y.; Tai, E.K.K.; So, W.H.L.; Woo, P.C.Y.; Cho, C.H. Probiotic Lactobacillus rhamnosus GG enhances gastric ulcer healing in rats. Eur. J. Pharmacol. 2007, 565, 171–179. [Google Scholar] [CrossRef]
- Kaaya, N.A.; Warren, H.L. A review of past and present research on aflatoxin in Uganda. Afr. J. Food Agric. Nutr. Dev. 2005, 5, 1–18. [Google Scholar]
- Paul Wacoo, A.; Wendiro, D.; Nanyonga, S.; Hawumba, J.; Sybesma, W.; Kort, R. Feasibility of a novel on-site detection method for aflatoxin in maize flour from markets and selected households in Kampala, Uganda. Toxins 2018, 10, 327. [Google Scholar] [CrossRef]
- Haskard, C.; Binnion, C.; Ahokas, J. Factors affecting the sequestration of aflatoxin by Lactobacillus rhamnosus strain GG. Chem. Biol. Interact. 2000, 128, 39–49. [Google Scholar] [CrossRef]
- Gratz, S.W.; Mykkanen, H.; El-Nezami, H.S. Probiotics and gut health: A special focus on liver diseases. World J. Gastroenterol. 2010, 16, 403–410. [Google Scholar] [CrossRef]
- Ahlberg, S.H.; Joutsjoki, V.; Korhonen, H.J. Potential of lactic acid bacteria in aflatoxin risk mitigation. Int. J. Food Microbiol. 2015, 207, 87–102. [Google Scholar] [CrossRef]
- Hernandez-Mendoza, A.; Guzman-de-Peña, D.; Garcia, H.S. Key role of teichoic acids on aflatoxin B1 binding by probiotic bacteria. J. Appl. Microbiol. 2009, 107, 395–403. [Google Scholar] [CrossRef]
- Mbonye, A.K. Risk factors for diarrhoea and upper respiratory tract infections among children in a rural area of Uganda. J. Health Popul. Nutr. 2004, 22, 52–58. [Google Scholar]
- O’Brien, K.L.; Wolfson, L.J.; Watt, J.P.; Henkle, E.; Deloria-Knoll, M.; McCall, N.; Lee, E.; Mulholland, K.; Levine, O.S.; Cherian, T. Burden of disease caused by Streptococcus pneumoniae in children younger than 5 years: Global estimates. Lancet 2009, 374, 893–902. [Google Scholar] [CrossRef]
- Kibuule, D.; Kagoya, H.R. Household management of acute respiratory infections in children under five years in Kampala Uganda. Afr. J. Pharm. Pharmacol. 2015, 9, 730–737. [Google Scholar] [CrossRef]
- Uganda Bureau of Statistics (UBOS). Uganda Demographic and Health Survey 2011; UBOS and ICF International: Kampala, Uganda, 2012.
- Kekkonen, R.A.; Vasankari, T.J.; Vuorimaa, T.; Haahtela, T.; Julkunen, I.; Korpela, R. The effect of probiotics on respiratory infections and gastrointestinal symptoms during training in marathon runners. Int. J. Sport Nutr. Exerc. Metab. 2007, 17, 352–363. [Google Scholar] [CrossRef]
- Hojsak, I.; Abdović, S.; Szajewska, H.; Milošević, M.; Krznarić, Ž.; Kolaček, S. Lactobacillus GG in the prevention of nosocomial gastrointestinal and respiratory tract infections. Pediatrics 2010, 125, e1171–e1177. [Google Scholar] [CrossRef]
- Hojsak, I.; Snovak, N.; Abdović, S.; Szajewska, H.; Mišak, Z.; Kolaček, S. Lactobacillus GG in the prevention of gastrointestinal and respiratory tract infections in children who attend day care centers: A randomized, double-blind, placebo-controlled trial. Clin. Nutr. 2010, 29, 312–316. [Google Scholar] [CrossRef]
- Kumpu, M.; Kekkonen, R.A.; Kautiainen, H.; Järvenpää, S.; Kristo, A.; Huovinen, P.; Pitkäranta, A.; Korpela, R.; Hatakka, K. Milk containing probiotic Lactobacillus rhamnosus GG and respiratory illness in children: A randomized, double-blind, placebo-controlled trial. Eur. J. Clin. Nutr. 2012, 66, 1020–1023. [Google Scholar] [CrossRef]
- Hatakka, K.; Savilahti, E.; Pönkä, A.; Meurman, J.H.; Poussa, T.; Näse, L.; Saxelin, M.; Korpela, R. Effect of long term consumption of probiotic milk on infections in children attending day care centres: Double blind, randomised trial. BMJ 2001, 322, 1327. [Google Scholar] [CrossRef]
- Popova, M.; Molimard, P.; Courau, S.; Crociani, J.; Dufour, C.; Vacon, F.; Carton, T. Beneficial effects of probiotics in upper respiratory tract infections and their mechanical actions to antagonize pathogens. J. Appl. Microbiol. 2012, 113, 1305–1318. [Google Scholar] [CrossRef]
- Vendt, N.; Grünberg, H.; Tuure, T.; Malminiemi, O.; Wuolijoki, E.; Tillmann, V.; Sepp, E.; Korpela, R. Growth during the first 6 months of life in infants using formula enriched with Lactobacillus rhamnosus GG: Double-blind, randomized trial. J. Hum. Nutr. Diet. 2006, 19, 51–58. [Google Scholar] [CrossRef]
- American Academy of Pediatrics. Probiotics Help Extremely Premature Infants Gain Weight. Available online: https://www.sciencedaily.com/releases/2010/05/100501013401.htm (accessed on 22 January 2017).
- Bortz, G.; Thomas, H. Biotechnologies for inclusive development: Scaling up, knowledge intensity and empowerment (the case of the probiotic yoghurt ‘Yogurito’ in Argentina). Innov. Dev. 2017, 7, 37–61. [Google Scholar] [CrossRef]
- Kort, R.; Sybesma, W. Probiotics for every body. Trends Biotechnol. 2012, 30, 613–615. [Google Scholar] [CrossRef]
- Kort, R.; Westerik, N.; Serrano, L.M.; Douillard, F.P.; Gottstein, W.; Mukisa, I.M.; Tuijn, C.J.; Basten, L.; Hafkamp, B.; Meijer, W.C.; et al. A novel consortium of Lactobacillus rhamnosus and Streptococcus thermophilus for increased access to functional fermented foods. Microb. Cell Factories 2015, 14, 195. [Google Scholar] [CrossRef]
- Westerik, N.; Wacoo, A.P.; Sybesma, W.; Kort, R. Novel production protocol for small-scale manufacture of probiotic fermented foods. JoVE J. Vis. Exp. 2016, e54365. [Google Scholar] [CrossRef]
- Reid, G.; Kort, R.; Alvarez, S.; Bourdet-Sicard, R.; Benoit, V.; Cunningham, M.; Saulnier, D.M.; van Hylckama Vlieg, J.E.T.; Verstraelen, H.; Sybesma, W. Expanding the reach of probiotics through social enterprises. Benef. Microbes 2018, 9, 707–715. [Google Scholar] [CrossRef]
- Liedholm, C.; McPherson, M.; Chuta, E. Small enterprise employment growth in rural Africa. Am. J. Agric. Econ. 1994, 76, 1177–1182. [Google Scholar] [CrossRef]
- Lenoir-Wijnkoop, I.; Nuijten, M.J.C.; Gutiérrez-Ibarluzea, I.; Hutton, J.; Poley, M.J.; Segal, L.; Bresson, J.L.; van Ganse, E.; Jones, P.; Moreno, L.; et al. Workshop Report: Concepts and methods in the economics of nutrition—Gateways to better economic evaluation of nutrition interventions. Br. J. Nutr. 2012, 108, 1714–1720. [Google Scholar] [CrossRef]
- Chilton, S.N.; Burton, J.P.; Reid, G. Inclusion of fermented foods in food guides around the world. Nutrients 2015, 7, 390–404. [Google Scholar] [CrossRef]
- Marco, M.L.; Heeney, D.; Binda, S.; Cifelli, C.J.; Cotter, P.D.; Foligne, B.; Gänzle, M.; Kort, R.; Pasin, G.; Pihlanto, A.; et al. Health benefits of fermented foods: Microbiota and beyond. Curr. Opin. Biotechnol. 2017, 44, 94–102. [Google Scholar] [CrossRef]
- Bandiera, O.; Burgess, R.; Gulesci, S.; Rasul, I.; Sulaiman, M. Can You Successfully Teach People How to Run Small Businesses? Available online: http://www.iig.ox.ac.uk/output/briefingpapers/pdfs/iig-briefingpaper-14-teaching-how-to-run-small-businesses.pdf (accessed on 2 January 2019).
- Hassan, R.; Birungi, P. Social capital and poverty in Uganda. Dev. S. Afr. 2011, 28, 19–37. [Google Scholar] [CrossRef]
- Butler, L.M.; Mazur, R.E. Principles and processes for enhancing sustainable rural livelihoods: Collaborative learning in Uganda. Int. J. Sustain. Dev. World Ecol. 2007, 14, 604–617. [Google Scholar] [CrossRef]
- Villena, J.; Salva, S.; Núñez, M.; Corzo, J.; Tolaba, R.; Faedda, J.; Font, G.; Alvarez, S. Probiotics for everyone! The novel immunobiotic Lactobacillus rhamnosus CRL1505 and the beginning of Social Probiotic Programs in Argentina. Int. J. Biotechnol. Wellness Ind. 2012, 1, 189–198. [Google Scholar]
- Di Stefano, E.; White, J.; Seney, S.; Hekmat, S.; McDowell, T.; Sumarah, M.; Reid, G. A novel millet-based probiotic fermented food for the developing world. Nutrients 2017, 9, 529. [Google Scholar] [CrossRef]
- Premand, P.; Rawlings, L.B.; Vermeersch, C. Impact Evaluation in Practice; The World Bank: Washington, DC, USA, 2010; pp. 1–266. [Google Scholar]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).