Syndecan-4 as a Marker of Endothelial Dysfunction in Patients with Resistant Hypertension
Abstract
1. Introduction
2. Methods
2.1. Patients, Baroreflex Activation Therapy (BAT) and Study Protocol
2.2. Patient Collective of the Control Group
2.3. Office Blood Pressure (BP) and 24 h Measurements
2.4. Enzyme-Linked Immunosorbent Assay (ELISA) Analysis of Human Syndecan-4 (SDC4)
2.5. Measurement of Pulse Wave Velocity (PWV)
2.6. Statistical Analysis
3. Results
3.1. Patients and Blood Pressure
3.2. SDC4 is Elevated in Patients with Resistant Arterial Hypertension (HTN)
3.3. Patients with Diabetes Mellitus (DM) Type II Show Higher Serum SDC4-Levels
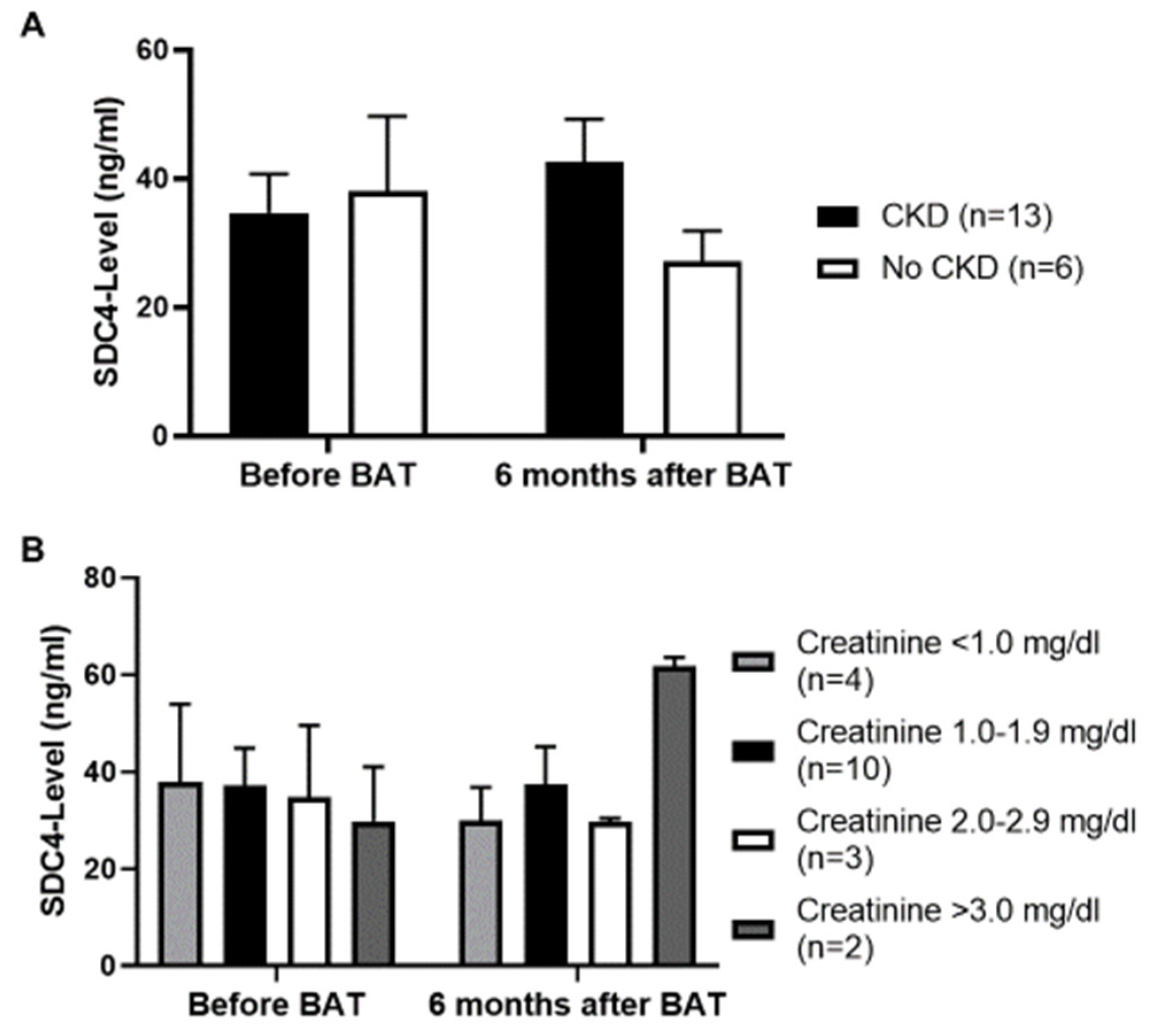
3.4. No Influence of Chronic Kidney Disease (CKD) on Serum SDC4-Levels
3.5. β-Blockers and Thiazide Diuretics Decrease, and Calcium Channel Blockers Increase the Serum Level of SDC4
4. Discussion
5. Limitation
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ACE | angiotensin-converting enzyme |
| ADAM-17 | metalloproteinase domain-containing protein 17 |
| ARB | angiotensin receptor blocker |
| BAT | baroreflex activation therapy |
| BMI | body mass index |
| BP | blood pressure |
| CKD | chronic kidney disease |
| DM | diabetes mellitus |
| eNOS | endothelial nitric oxide synthase |
| GFR | glomerular filtration rate |
| HLP | hyperlipoproteinemia |
| HTN | arterial hypertension |
| MMP | matrix metalloproteinase |
| SD | standard deviation |
| SDC4 | syndecan-4 |
| SEM | standard error of the mean |
References
- Hildebrandt, D.A.; Irwin, E.D.; Lohmeier, T.E. Prolonged baroreflex activation abolishes salt-induced hypertension after reductions in kidney mass. Hypertension 2016, 68, 1400–1406. [Google Scholar] [CrossRef]
- Lipphardt, M.; Koziolek, M.J.; Lehnig, L.Y.; Schäfer, A.K.; Müller, G.A.; Lüders, S.; Wallbach, M. Effect of baroreflex activation therapy on renal sodium excretion in patients with resistant hypertension. Clin. Res. Cardiol. 2019, 108, 1287–1296. [Google Scholar] [CrossRef] [PubMed]
- Wallbach, M.; Koziolek, M.J. Baroreceptors in the carotid and hypertension-systematic review and meta-analysis of the effects of baroreflex activation therapy on blood pressure. Nephrol. Dial. Transplant. 2018, 33, 1485–1493. [Google Scholar] [CrossRef] [PubMed]
- Wallbach, M.; Lehnig, L.Y.; Helms, H.J.; Schroer, C.; Müller, G.A.; Wachter, R.; Koziolek, M.J. Long-term effects of baroreflex activation therapy on glucose metabolism. Acta Diabetol. 2015, 52, 829–835. [Google Scholar] [CrossRef]
- Wallbach, M.; Lehnig, L.Y.; Schroer, C.; Helms, H.J.; Lüders, S.; Patschan, D.; Patschan, S.; Müller, G.A.; Wachter, R.; Koziolek, M.J. Effects of baroreflex activation therapy on arterial stiffness and central hemodynamics in patients with resistant hypertension. J. Hypertens. 2015, 33, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Wallbach, M.; Lehnig, L.-Y.; Schroer, C.; Hasenfuss, G.; Müller, G.A.; Wachter, R.; Koziolek, M.J. Impact of baroreflex activation therapy on renal function—A pilot study. Am. J. Nephrol. 2014, 40, 371–380. [Google Scholar] [CrossRef] [PubMed]
- Esler, M.D. Sympathetic nervous system moves toward center stage in cardiovascular medicine: From Thomas Willis to resistant hypertension. Hypertension 2014, 63, e25–e32. [Google Scholar] [CrossRef]
- Smith, P.A.; Graham, L.N.; Mackintosh, A.F.; Stoker, J.B.; Mary, D.A. Relationship between central sympathetic activity and stages of human hypertension. Am. J. Hypertens. 2004, 17, 217–222. [Google Scholar] [CrossRef]
- Grassı, G.; Seravalle, G.; Brambilla, G.; Pini, C.; Alimento, M.; Facchetti, R.; Spaziani, D.; Cuspidi, C.; Mancia, G. Marked sympathetic activation and baroreflex dysfunction in true resistant hypertension. Int. J. Cardiol. 2014, 177, 1020–1025. [Google Scholar] [CrossRef]
- Kaplon, R.E.; Walker, A.E.; Seals, D.R. Plasma norepinephrine is an independent predictor of vascular endothelial function with aging in healthy women. J. Appl. Physiol. 2011, 111, 1416–1421. [Google Scholar] [CrossRef]
- Sverrisdóttir, Y.B.; Jansson, L.M.; Hägg, U.; Gan, L.M. Muscle sympathetic nerve activity is related to a surrogate marker of endothelial function in healthy individuals. PLoS ONE 2010, 5, e9257. [Google Scholar] [CrossRef] [PubMed]
- Schlaich, M.P.; Sobotka, P.A.; Krum, H.; Whitbourn, R.; Walton, A.; Esler, M.D. Renal denervation as a therapeutic approach for hypertension: Novel implications for an old concept. Hypertension 2009, 54, 1195–1201. [Google Scholar] [CrossRef] [PubMed]
- Hasking, G.J.; Esler, M.D.; Jennings, G.L.; Burton, D.; Johns, J.A.; Korner, P.I. Norepinephrine spillover to plasma in patients with congestive heart failure: Evidence of increased overall and cardiorenal sympathetic nervous activity. Circulation 1986, 73, 615–621. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.J.; Gona, P.; Larson, M.G.; Levy, D.; Benjamin, E.J.; Tofler, G.H.; Jacques, P.F.; Meigs, J.B.; Rifai, N.; Selhub, J.; et al. Multiple biomarkers and the risk of incident hypertension. Hypertension 2007, 49, 432–438. [Google Scholar] [CrossRef] [PubMed]
- Vuong, T.T.; Reine, T.M.; Sudworth, A.; Jenssen, T.G.; Kolset, S.O. Syndecan-4 is a major syndecan in primary human endothelial cells in vitro, modulated by inflammatory stimuli and involved in wound healing. J. Histochem. Cytochem. 2015, 63, 280–292. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.W.; Goldberger, O.A.; Gallo, R.L.; Bernfield, M. Members of the syndecan family of heparan sulfate proteoglycans are expressed in distinct cell-, tissue-, and development-specific patterns. Mol. Biol. Cell 1994, 5, 797–805. [Google Scholar] [CrossRef]
- Echtermeyer, F.; Streit, M.; Wilcox-Adelman, S.; Saoncella, S.; Denhez, F.; Detmar, M.; Goetinck, P.F. Delayed wound repair and impaired angiogenesis in mice lacking syndecan-4. J. Clin. Investig. 2001, 107, R9–R14. [Google Scholar] [CrossRef] [PubMed]
- Matsui, Y.; Ikesue, M.; Danzaki, K.; Morimoto, J.; Sato, M.; Tanaka, S.; Kojima, T.; Tsutsui, H.; Uede, T. Syndecan-4 prevents cardiac rupture and dysfunction after myocardial infarction. Circ. Res. 2011, 108, 1328–1339. [Google Scholar] [CrossRef]
- Ishiguro, K.; Kadomatsu, K.; Kojima, T.; Muramatsu, H.; Iwase, M.; Yoshikai, Y.; Yanada, M.; Yamamoto, K.; Matsushita, T.; Nishimura, M.; et al. Syndecan-4 deficiency leads to high mortality of lipopolysaccharide-injected Mice. J. Biol. Chem. 2001, 276, 47483–47488. [Google Scholar] [CrossRef]
- Baeyens, N.; Mulligan-Kehoe, M.J.; Corti, F.; Simon, D.D.; Ross, T.D.; Rhodes, J.M.; Wang, T.Z.; Mejean, C.O.; Simons, M.; Humphrey, J.; et al. Syndecan 4 is required for endothelial alignment in flow and atheroprotective signaling. Proc. Natl. Acad. Sci. USA 2014, 111, 17308–17313. [Google Scholar] [CrossRef]
- Finsen, A.V.; Woldbaek, P.R.; Li, J.; Wu, J.; Lyberg, T.; Tønnessen, T.; Christensen, G. Increased syndecan expression following myocardial infarction indicates a role in cardiac remodeling. Physiol. Genom. 2004, 16, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Corti, F.; Finetti, F.; Ziche, M.; Simons, M. The syndecan-4/protein kinase cα pathway mediates prostaglandin E2-induced extracellular regulated kinase (ERK) activation in endothelial cells and angiogenesis in vivo. J. Biol. Chem. 2013, 288, 12712–12721. [Google Scholar] [CrossRef] [PubMed]
- Nunes, S.S.; Juliano, L.; Vardiero, F.; Nader, H.B.; Woods, A.; Legrand, C.; Morandi, V.; Outeiro-Bernstein, M.A.F.D. Syndecan-4 contributes to endothelial tubulogenesis through interactions with two motifs inside the pro-angiogenic N-terminal domain of thrombospondin-1. J. Cell. Physiol. 2007, 214, 828–837. [Google Scholar] [CrossRef] [PubMed]
- Ju, R.; Zhuang, Z.W.; Zhang, J.; Lanahan, A.A.; Kyriakides, T.; Sessa, W.; Simons, M. Angiopoietin-2 secretion by endothelial cell exosomes: Regulation by the phosphatidylinositol 3-kinase (PI3K)/Akt/endothelial nitric oxide synthase (eNOS) and syndecan-4/syntenin pathways. J. Biol. Chem. 2013, 289, 510–519. [Google Scholar] [CrossRef] [PubMed]
- Elfenbein, A.; Simons, M. Syndecan-4 signaling at a glance. J. Cell Sci. 2013, 126, 3799–3804. [Google Scholar] [CrossRef]
- Simons, M.; Horowitz, A. Syndecan-4-mediated signalling. Cell. Signal. 2001, 13, 855–862. [Google Scholar] [CrossRef]
- Strand, M.E.; Herum, K.M.; Rana, Z.A.; Škrbić, B.; Askevold, E.T.; Dahl, C.P.; Vistnes, M.; Hasic, A.; Kvaløy, H.; Sjaastad, I.; et al. Innate immune signaling induces expression and shedding of the heparan sulfate proteoglycan syndecan-4 in cardiac fibroblasts and myocytes, affecting inflammation in the pressure-overloaded heart. FEBS J. 2013, 280, 2228–2247. [Google Scholar] [CrossRef]
- Gamboa, A.; Figueroa, R.; Paranjape, S.Y.; Farley, G.; Diedrich, A.; Biaggioni, I. Autonomic blockade reverses endothelial dysfunction in obesity-associated hypertension. Hypertension 2016, 68, 1004–1010. [Google Scholar] [CrossRef]
- Chobanian, A.V.; Bakris, G.L.; Black, H.R.; Cushman, W.C.; Green, L.A.; Izzo, J.L.; Jones, D.W.; Materson, B.J.; Oparil, S.; Wright, J.T.; et al. Seventh report of the joint national committee on prevention, detection, evaluation, and treatment of high blood pressure. Hypertension 2003, 42, 1206–1252. [Google Scholar] [CrossRef]
- Mancia, G.; De Backer, G.; Dominiczak, A.; Cifkova, R.; Fagard, R.; Germano, G.; Grassi, G.; Heagerty, A.M.; Kjeldsen, S.E.; Laurent, S.; et al. 2007 Guidelines for the management of arterial hypertension: The task force for the management of arterial hypertension of the european society of hypertension (ESH) and of the european society of cardiology (ESC). Eur. Heart J. 2013, 34, 2159–2219. [Google Scholar] [CrossRef]
- Williams, B.; Mancia, G.; Spiering, W.; Rosei, E.A.; Azizi, M.; Burnier, M.; Clement, D.L.; Coca, A.; De Simone, G.; Dominiczak, A.F.; et al. 2018 ESC/ESH Guidelines for the management of arterial hypertension. Eur. Heart J. 2018, 39, 3021–3104. [Google Scholar] [CrossRef] [PubMed]
- Tordoir, J.H.M.; Scheffers, I.; Schmidli, J.; Savolainen, H.; Liebeskind, U.; Hansky, B.; Herold, U.; Irwin, E.; Kroon, A.; De Leeuw, P.; et al. An implantable carotid sinus baroreflex activating system: Surgical technique and short-term outcome from a multi-center feasibility trial for the treatment of resistant hypertension. Eur. J. Vasc. Endovasc. Surg. 2007, 33, 414–421. [Google Scholar] [CrossRef] [PubMed]
- Hoppe, U.C.; Brandt, M.-C.; Wachter, R.; Beige, J.; Rump, L.C.; Kroon, A.A.; Cates, A.W.; Lovett, E.G.; Haller, H. Minimally invasive system for baroreflex activation therapy chronically lowers blood pressure with pacemaker-like safety profile: Results from the Barostim neo trial. J. Am. Soc. Hypertens. 2012, 6, 270–276. [Google Scholar] [CrossRef] [PubMed]
- Wallbach, M.; Lehnig, L.-Y.; Schroer, C.; Lüders, S.; Böhning, E.; Müller, G.A.; Wachter, R.; Koziolek, M.J. Effects of baroreflex activation therapy on ambulatory blood pressure in patients with resistant hypertension. Hypertension 2016, 67, 701–709. [Google Scholar] [CrossRef]
- Levin, A.; Stevens, P.E. Summary of KDIGO 2012 CKD Guideline: Behind the scenes, need for guidance, and a framework for moving forward. Kidney Int. 2014, 85, 49–61. [Google Scholar] [CrossRef]
- Nikaido, T.; Tanino, Y.; Wang, X.; Sato, S.; Misa, K.; Fukuhara, N.; Sato, Y.; Fukuhara, A.; Uematsu, M.; Suzuki, Y.; et al. Serum Syndecan-4 as a Possible Biomarker in Patients With Acute Pneumonia. J. Infect. Dis. 2015, 212, 1500–1508. [Google Scholar] [CrossRef]
- Sato, Y.; Tanino, Y.; Wang, X.; Nikaido, T.; Sato, S.; Misa, K.; Togawa, R.; Frevert, C.W.; Munakata, M. Baseline serum syndecan-4 predicts prognosis after the onset of acute exacerbation of idiopathic interstitial pneumonia. PLoS ONE 2017, 12, e0176789. [Google Scholar] [CrossRef]
- Takahashi, R.; Negishi, K.; Watanabe, A.; Arai, M.; Naganuma, F.; Ohyama, Y.; Kurabayashi, M. Serum syndecan-4 is a novel biomarker for patients with chronic heart failure. J. Cardiol. 2011, 57, 325–332. [Google Scholar] [CrossRef]
- Van Bortel, L.M.; Laurent, S.; Boutouyrie, P.; Chowienczyk, P.; Cruickshank, J.; De Backer, T.; Filipovsky, J.; Huybrechts, S.; Mattace-Raso, F.U.; Protogerou, A.; et al. Expert consensus document on the measurement of aortic stiffness in daily practice using carotid-femoral pulse wave velocity. J. Hypertens. 2012, 30, 445–448. [Google Scholar] [CrossRef]
- The Reference Values for Arterial Stiffness’ Collaboration; Reference Values for Arterial Stiffness’ Collaboration Determinants of pulse wave velocity in healthy people and in the presence of cardiovascular risk factors: ‘establishing normal and reference values’. Eur. Hear. J. 2010, 31, 2338–2350. [CrossRef]
- Ben-Shlomo, Y.; Spears, M.; Boustred, C.; May, M.; Anderson, S.G.; Benjamin, E.J.; Boutouyrie, P.; Cameron, J.D.; Chen, C.-H.; Cruickshank, J.K.; et al. Aortic pulse wave velocity improves cardiovascular event prediction: An individual participant meta-analysis of prospective observational data from 17,635 subjects. J. Am. Coll. Cardiol. 2013, 63, 636–646. [Google Scholar] [CrossRef] [PubMed]
- Scarpellini, A.; Huang, L.; Burhan, I.; Schroeder, N.; Funck, M.; Johnson, T.S.; Verderio, E.A. Syndecan-4 Knockout Leads to Reduced Extracellular Transglutaminase-2 and Protects against Tubulointerstitial Fibrosis. J. Am. Soc. Nephrol. 2013, 25, 1013–1027. [Google Scholar] [CrossRef] [PubMed]
- Lipphardt, M.; Dihazi, H.; Song, J.W.; Ratliff, B.B.; Müller, G.A.; Goligorsky, M.S. Endothelial dysfunction is a superinducer of syndecan-4: Fibrogenic role of its ectodomain. Am. J. Physiol. Circ. Physiol. 2018, 314, H484–H496. [Google Scholar] [CrossRef] [PubMed]
- Wee, Y.M.; Go, H.; Choi, M.Y.; Jung, H.R.; Cho, Y.M.; Kim, Y.H.; Han, D.J.; Shin, S. Tissue-resident natural killer cells exacerbate tubulointerstitial fibrosis by activating transglutaminase 2 and syndecan-4 in a model of aristolochic acid-induced nephropathy. BMB Rep. 2019, 52, 554–559. [Google Scholar] [CrossRef]
- Strand, M.E.; Aronsen, J.M.; Braathen, B.; Sjaastad, I.; Kvaløy, H.; Tønnessen, T.; Christensen, G.; Lunde, I.G. Shedding of syndecan-4 promotes immune cell recruitment and mitigates cardiac dysfunction after lipopolysaccharide challenge in mice. J. Mol. Cell. Cardiol. 2015, 88, 133–144. [Google Scholar] [CrossRef]
- Lunde, I.G.; Herum, K.M.; Carlson, C.C.; Christensen, G. Syndecans in heart fibrosis. Cell. Tissue. Res. 2016, 365, 539–552. [Google Scholar] [CrossRef]
- Lipphardt, M.; Song, J.W.; Goligorsky, M.S. Sirtuin 1 and endothelial glycocalyx. Pflüg. Arch. Eur. J. Phy. 2020, 472, 991–1002. [Google Scholar] [CrossRef]
- Lipphardt, M.; Dihazi, H.; Müller, G.A.; Goligorsky, M.S. Fibrogenic Secretome of Sirtuin 1-Deficient Endothelial Cells: Wnt, Notch and Glycocalyx Rheostat. Front. Physiol. 2018, 9, 9. [Google Scholar] [CrossRef]
- Yasmin; Wallace, S.; McEniery, C.M.; Dakham, Z.; Pusalkar, P.; Maki-Petaja, K.; Ashby, M.J.; Cockcroft, J.R.; Wilkinson, I.B. Matrix Metalloproteinase-9 (MMP-9), MMP-2, and Serum Elastase Activity Are Associated With Systolic Hypertension and Arterial Stiffness. Arter. Thromb. Vasc. Biol. 2005, 25, 372–378. [Google Scholar] [CrossRef]
- DeRosa, G.; D’Angelo, A.; Ciccarelli, L.; Piccinni, M.N.; Pricolo, F.; Salvadeo, S.; Montagna, L.; Gravina, A.; Ferrari, I.; Galli, S.; et al. Matrix Metalloproteinase-2, -9, and Tissue Inhibitor of Metalloproteinase-1 in Patients with Hypertension. Endothelium 2006, 13, 227–231. [Google Scholar] [CrossRef]
- Ramnath, R.; Foster, R.R.; Qiu, Y.; Cope, G.; Butler, M.J.; Salmon, A.H.; Mathieson, P.W.; Coward, R.J.; Welsh, G.I.; Satchell, S. Matrix metalloproteinase 9-mediated shedding of syndecan 4 in response to tumor necrosis factor α: A contributor to endothelial cell glycocalyx dysfunction. FASEB J. 2014, 28, 4686–4699. [Google Scholar] [CrossRef] [PubMed]
- Partovian, C.; Ju, R.; Zhuang, Z.W.; Martin, K.A.; Simons, M. Syndecan-4 Regulates Subcellular Localization of mTOR Complex2 and Akt Activation in a PKCα-Dependent Manner in Endothelial Cells. Mol. Cell 2008, 32, 140–149. [Google Scholar] [CrossRef] [PubMed]
- Schulz, E.; Gori, T.; Münzel, T. Oxidative stress and endothelial dysfunction in hypertension. Hypertens. Res. 2011, 34, 665–673. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Herrera, A.H.; Li, Y.; Belani, K.K.; Walcheck, B. Regulation of mature ADAM17 by redox agents for L-selectin shedding. J. Immunol. 2009, 182, 2449–2457. [Google Scholar] [CrossRef] [PubMed]
- Kawahara, R.; Lima, R.N.; Domingues, R.R.; Pauletti, B.A.; Meirelles, G.V.; Assis, M.; Figueira, A.C.M.; Leme, A.F.P. Deciphering the Role of the ADAM17-Dependent Secretome in Cell Signaling. J. Proteome Res. 2014, 13, 2080–2093. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.W.; Song, K.E.; Shin, D.S.; Ahn, S.M.; Ha, E.S.; Kim, D.J.; Nam, M.S.; Lee, K.W. Alterations in peripheral blood levels of TIMP-1, MMP-2, and MMP-9 in patients with type-2 diabetes. Diabetes Res. Clin. Pract. 2005, 69, 175–179. [Google Scholar] [CrossRef] [PubMed]
- Menghini, R.; Fiorentino, L.; Casagrande, V.; Lauro, R.; Federici, M. The role of ADAM17 in metabolic inflammation. Atherosclerosis 2013, 228, 12–17. [Google Scholar] [CrossRef]
- Subramanian, S.V.; Fitzgerald, M.L.; Bernfield, M. Regulated Shedding of Syndecan-1 and -4 Ectodomains by Thrombin and Growth Factor Receptor Activation. J. Biol. Chem. 1997, 272, 14713–14720. [Google Scholar] [CrossRef]
- Kainulainen, V.; Wang, H.; Schick, C.; Bernfield, M. Syndecans, heparan sulfate proteoglycans, maintain the proteolytic balance of acute wound fluids. J. Biol. Chem. 1998, 273, 11563–11569. [Google Scholar] [CrossRef]
- De La Sierra, A.; Larrousse, M. Endothelial dysfunction is associated with increased levels of biomarkers in essential hypertension. J. Hum. Hypertens. 2009, 24, 373–379. [Google Scholar] [CrossRef] [PubMed]
- Montezano, A.C.; Dulak-Lis, M.; Tsiropoulou, S.; Harvey, A.; Briones, A.M.; Touyz, R.M. Oxidative Stress and Human Hypertension: Vascular Mechanisms, Biomarkers, and Novel Therapies. Can. J. Cardiol. 2015, 31, 631–641. [Google Scholar] [CrossRef] [PubMed]
- Paniagua, O.A.; Bryant, M.B.; Panza, J.A. Role of Endothelial Nitric Oxide in Shear Stress–Induced Vasodilation of Human Microvasculature. Circulation 2001, 103, 1752–1758. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.-X.; Yan, Z.-P.; Zhang, Y.-Y.; Wu, J.; Liu, X.-H.; Zeng, Y. Hemodynamic shear stress regulates the transcriptional expression of heparan sulfate proteoglycans in human umbilical vein endothelial cell. Cell. Mol. Biol. 2016, 62, 28–34. [Google Scholar]
- Fang, Y.; Wu, D.; Birukov, K.G. Mechanosensing and Mechanoregulation of Endothelial Cell Functions. Compr. Physiol. 2019, 9, 873–904. [Google Scholar] [CrossRef] [PubMed]
- Lyle, A.N.; Raaz, U. Killing Me Unsoftly. Arter. Thromb. Vasc. Biol. 2017, 37, e1–e11. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.J.; Park, C.G.; Park, J.S.; Suh, S.Y.; Choi, C.U.; Kim, J.W.; Kim, S.H.; Lim, H.E.; Rha, S.W.; Seo, H.S.; et al. Relationship between blood pressure parameters and pulse wave velocity in normotensive and hypertensive subjects: Invasive study. J. Hum. Hypertens. 2006, 21, 141–148. [Google Scholar] [CrossRef]
- Annabi, B.B.; Vaillancourt-Jean, E.; Weil, A.G.; Béliveau, R. Pharmacological targeting of β-adrenergic receptor functions abrogates NF-κB signaling and MMP-9 secretion in medulloblastoma cells. OncoTargets Ther. 2010, 3, 219–226. [Google Scholar] [CrossRef]
- Haas, M.J.; Kurban, W.; Shah, H.; Onstead-Haas, L.; Mooradian, A.D. Beta Blockers Suppress Dextrose-Induced Endoplasmic Reticulum Stress, Oxidative Stress, and Apoptosis in Human Coronary Artery Endothelial Cells. Am. J. Ther. 2016, 23, e1524–e1531. [Google Scholar] [CrossRef]
- Zervoudaki, A.; Economou, E.; Pitsavos, C.; Vasiliadou, K.; Aggeli, C.; Tsioufis, K.; Toutouza, M.; Toutouzas, P.; Stefanadis, C. The effect of Ca2+ channel antagonists on plasma concentrations of matrix metalloproteinase-2 and -9 in essential hypertension. Am. J. Hypertens. 2004, 17, 273–276. [Google Scholar] [CrossRef][Green Version]
- Manon-Jensen, T.; Multhaupt, H.A.B.; Couchman, J.R. Mapping of matrix metalloproteinase cleavage sites on syndecan-1 and syndecan-4 ectodomains. FEBS J. 2013, 280, 2320–2331. [Google Scholar] [CrossRef]
- Tanner, R.M.; Lynch, A.I.; Brophy, V.H.; Eckfeldt, J.H.; Davis, B.R.; Ford, C.E.; Boerwinkle, E.; Arnett, D.K. Pharmacogenetic Associations of MMP9 and MMP12 Variants with Cardiovascular Disease in Patients with Hypertension. PLoS ONE 2011, 6, e23609. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Echtermeyer, F.; Thilo, F.; Theilmeier, G.; Schmidt, A.; Schülein, R.; Jensen, B.L.; Loddenkemper, C.; Jankowski, V.; Marcussen, N.; et al. The proteoglycan syndecan 4 regulates transient receptor potential canonical 6 channels via RhoA/Rho-associated protein kinase signaling. Arter. Thromb. Vasc. Biol. 2011, 32, 378–385. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.Y.; Roshanravan, H.; Dryer, S.E. Syndecan-4 ectodomain evokes mobilization of podocyte TRPC6 channels and their associated pathways: An essential role for integrin signaling. Biochim. Biophys. Acta (BBA) Bioenerg. 2015, 1853, 2610–2620. [Google Scholar] [CrossRef] [PubMed]
- Pyke, C.; Kristensen, P.; Østergaard, P.B.; Oturai, P.; Rømer, J. Proteoglycan Expression in the Normal Rat Kidney. Nephron 1997, 77, 461–470. [Google Scholar] [CrossRef]





| Parameter | Control Group | Resistant Hypertension Group | p |
|---|---|---|---|
| n | 35 | 19 | |
| female n (%) Male n (%) | 18 (51%) 17 (49%) | 10 (53%) 9 (47%) | 0.99 0.99 |
| age (years) | 25.4 ± 3.5 | 61.1 ± 10 | <0.01 |
| BMI (kg/m2) | 25.0 ± 4.9 | 32.4 ± 6.6 | <0.01 |
| smokers n (%) | 4 (11%) | 13 (68%) | <0.01 |
| relevant concomitant diseases | |||
| CKD n (%) | - | 13 (68%) | |
| CKD ≥ STAGE 3 | - | 12 (63%) | |
| DM n (%) | - | 7 (36%) | |
| HLP n (%) | - | 16 (84%) | |
| CAD n (%) | - | 6 (31%) | |
| CHF n (%) | - | 4 (21%) | |
| adipositas stage ≥1 (BMI ≥ 30 kg/m2) | - | 12 (63%) | |
| number of antihypertensive medications | 0.0 ± 0.0 | 6.8 ± 1.4 | >0.99 |
| office BP | |||
| systolic diastolic | - - | 160.2 ± 23.6 81.3 ± 14.9 | |
| 24-H ABP | |||
| systolic diastolic | - - | 143.1 ± 17.2 76.6 ± 9.9 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lipphardt, M.; Dihazi, H.; Maas, J.-H.; Schäfer, A.-K.; Amlaz, S.I.; Ratliff, B.B.; Koziolek, M.J.; Wallbach, M. Syndecan-4 as a Marker of Endothelial Dysfunction in Patients with Resistant Hypertension. J. Clin. Med. 2020, 9, 3051. https://doi.org/10.3390/jcm9093051
Lipphardt M, Dihazi H, Maas J-H, Schäfer A-K, Amlaz SI, Ratliff BB, Koziolek MJ, Wallbach M. Syndecan-4 as a Marker of Endothelial Dysfunction in Patients with Resistant Hypertension. Journal of Clinical Medicine. 2020; 9(9):3051. https://doi.org/10.3390/jcm9093051
Chicago/Turabian StyleLipphardt, Mark, Hassan Dihazi, Jens-Holger Maas, Ann-Kathrin Schäfer, Saskia I. Amlaz, Brian B. Ratliff, Michael J. Koziolek, and Manuel Wallbach. 2020. "Syndecan-4 as a Marker of Endothelial Dysfunction in Patients with Resistant Hypertension" Journal of Clinical Medicine 9, no. 9: 3051. https://doi.org/10.3390/jcm9093051
APA StyleLipphardt, M., Dihazi, H., Maas, J.-H., Schäfer, A.-K., Amlaz, S. I., Ratliff, B. B., Koziolek, M. J., & Wallbach, M. (2020). Syndecan-4 as a Marker of Endothelial Dysfunction in Patients with Resistant Hypertension. Journal of Clinical Medicine, 9(9), 3051. https://doi.org/10.3390/jcm9093051






