Clinical Outcomes of Single Mosaic Embryo Transfer: High-Level or Low-Level Mosaic Embryo, Does It Matter?
Abstract
1. Introduction
2. Material and Methods
3. Study Population
4. In Vitro Fertilization, Embryo Culture, TE Biopsy, and Frozen Embryo Transfer
5. PGT-A Using hr-NGS
6. Study Outcomes
7. Statistical Analysis
8. Results
8.1. Impact of the Proportion of Chromosomal Mosaicism on Clinical Outcomes
8.2. Factors Affecting Live Birth in Mosaic Embryo Transfer
8.3. Concordance between TE Biopsy and ICM in Designated as Mosaic Embryos
9. Discussion
10. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Preimplantation Genetic Diagnosis International Society (PGDIS). Guidelines for Mosaic Embryos; PGDIS: Yzeure, France, 2016. [Google Scholar]
- Delhanty, J.D.; Harper, J.C.; Ao, A.; Handyside, A.H.; Winston, R.M. Multicolour FISH detects frequent chromosomal mosaicism and chaotic division in normal preimplantation embryos from fertile patients. Hum. Genet. 1997, 99, 755–760. [Google Scholar] [CrossRef] [PubMed]
- Taylor, T.H.; Gitlin, S.A.; Patrick, J.L.; Crain, J.L.; Wilson, J.M.; Griffin, D.K. The origin, mechanisms, incidence and clinical consequences of chromosomal mosaicism in humans. Hum. Reprod. Update 2014, 20, 571–581. [Google Scholar] [CrossRef] [PubMed]
- Greco, E.; Minasi, M.G.; Fiorentino, F. Healthy Babies after Intrauterine Transfer of Mosaic Aneuploid Blastocysts. N. Engl. J. Med. 2015, 373, 2089–2090. [Google Scholar] [CrossRef] [PubMed]
- Fragouli, E.; Alfarawati, S.; Spath, K.; Babariya, D.; Tarozzi, N.; Borini, A.; Wells, D. Analysis of implantation and ongoing pregnancy rates following the transfer of mosaic diploid-aneuploid blastocysts. Hum. Genet. 2017, 136, 805–819. [Google Scholar] [CrossRef] [PubMed]
- Munne, S.; Blazek, J.; Large, M.; Martinez-Ortiz, P.A.; Nisson, H.; Liu, E.; Tarozzi, N.; Borini, A.; Becker, A.; Zhang, J.; et al. Detailed investigation into the cytogenetic constitution and pregnancy outcome of replacing mosaic blastocysts detected with the use of high-resolution next-generation sequencing. Fertil. Steril. 2017, 108, 62–71. [Google Scholar] [CrossRef] [PubMed]
- Victor, A.R.; Tyndall, J.C.; Brake, A.J.; Lepkowsky, L.T.; Murphy, A.E.; Griffin, D.K.; McCoy, R.C.; Barnes, F.L.; Zouves, C.G.; Viotti, M. One hundred mosaic embryos transferred prospectively in a single clinic: Exploring when and why they result in healthy pregnancies. Fertil. Steril. 2019, 111, 280–293. [Google Scholar] [CrossRef] [PubMed]
- Spinella, F.; Fiorentino, F.; Biricik, A.; Bono, S.; Ruberti, A.; Cotroneo, E.; Baldi, M.; Cursio, E.; Minasi, M.G.; Greco, E. Extent of chromosomal mosaicism influences the clinical outcome of in vitro fertilization treatments. Fertil. Steril. 2018, 109, 77–83. [Google Scholar] [CrossRef]
- Zhang, L.; Wei, D.; Zhu, Y.; Gao, Y.; Yan, J.; Chen, Z.J. Rates of live birth after mosaic embryo transfer compared with euploid embryo transfer. J. Assist. Reprod. Genet. 2019, 36, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Zore, T.; Kroener, L.L.; Wang, C.; Liu, L.; Buyalos, R.; Hubert, G.; Shamonki, M. Transfer of embryos with segmental mosaicism is associated with a significant reduction in live-birth rate. Fertil. Steril. 2019, 111, 69–76. [Google Scholar] [CrossRef]
- Mantikou, E.; Wong, K.M.; Repping, S.; Mastenbroek, S. Molecular origin of mitotic aneuploidies in preimplantation embryos. Biochim. Biophys. Acta 2012, 1822, 1921–1930. [Google Scholar] [CrossRef]
- Lee, C.I.; Chen, C.H.; Huang, C.C.; Cheng, E.H.; Chen, H.H.; Ho, S.T.; Lin, P.Y.; Lee, M.S.; Lee, T.H. Embryo morphokinetics is potentially associated with clinical outcomes of single-embryo transfers in preimplantation genetic testing for aneuploidy cycles. Reprod. Biomed. Online 2019. [Google Scholar] [CrossRef] [PubMed]
- Kushnir, V.A.; Darmon, S.K.; Barad, D.H.; Gleicher, N. Degree of mosaicism in trophectoderm does not predict pregnancy potential: A corrected analysis of pregnancy outcomes following transfer of mosaic embryos. Reprod. Biol. Endocrinol. 2018, 16, 6. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.H.; Huang, C.C.; Cheng, E.H.; Lee, T.H.; Chien, L.F.; Lee, M.S. Optimal timing of blastocyst vitrification after trophectoderm biopsy for preimplantation genetic screening. PLoS ONE 2017, 12, e0185747. [Google Scholar] [CrossRef] [PubMed]
- Inoue, N.; Lopez, R.; Delgado, A.; Nunez, D.; Portella, J.; Noriega-Hoces, L.; Guzman, L. Mosaic embryo transfer after oocyte in vitro maturation in combination with non-invasive prenatal testing (NIPT)-first report of a euploid live birth. J. Assist. Reprod. Genet. 2017, 34, 1199–1205. [Google Scholar] [CrossRef] [PubMed]
- Vera-Rodriguez, M.; Michel, C.E.; Mercader, A.; Bladon, A.J.; Rodrigo, L.; Kokocinski, F.; Mateu, E.; Al-Asmar, N.; Blesa, D.; Simon, C.; et al. Distribution patterns of segmental aneuploidies in human blastocysts identified by next-generation sequencing. Fertil. Steril. 2016, 105, 1047–1055. [Google Scholar] [CrossRef]
- Munne, S.; Wells, D. Detection of mosaicism at blastocyst stage with the use of high-resolution next-generation sequencing. Fertil. Steril. 2017, 107, 1085–1091. [Google Scholar] [CrossRef]
- Victor, A.R.; Griffin, D.K.; Brake, A.J.; Tyndall, J.C.; Murphy, A.E.; Lepkowsky, L.T.; Lal, A.; Zouves, C.G.; Barnes, F.L.; McCoy, R.C.; et al. Assessment of aneuploidy concordance between clinical trophectoderm biopsy and blastocyst. Hum. Reprod. 2019, 34, 181–192. [Google Scholar] [CrossRef]
- Zegers-Hochschild, F.; Adamson, G.D.; de Mouzon, J.; Ishihara, O.; Mansour, R.; Nygren, K.; Sullivan, E.; van der Poel, S.; International Committee for Monitoring Assisted Reproductive Technologies; World Health Organization. The International Committee for Monitoring Assisted Reproductive Technology (ICMART) and the World Health Organization (WHO) Revised Glossary on ART Terminology, 2009. Hum. Reprod. 2009, 24, 2683–2687. [Google Scholar] [CrossRef]
- Kolte, A.M.; Bernardi, L.A.; Christiansen, O.B.; Quenby, S.; Farquharson, R.G.; Goddijn, M.; Stephenson, M.D.; ESHRE Early Pregnancy Special Interest Group. Terminology for pregnancy loss prior to viability: A consensus statement from the ESHRE early pregnancy special interest group. Hum. Reprod. 2015, 30, 495–498. [Google Scholar] [CrossRef]
- Cram, D.S.; Leigh, D.; Handyside, A.; Rechitsky, L.; Xu, K.; Harton, G.; Grifo, J.; Rubio, C.; Fragouli, E.; Kahraman, S.; et al. PGDIS Position Statement on the Transfer of Mosaic Embryos 2019. Reprod. Biomed. Online 2019, 39 (Suppl. 1), e1–e4. [Google Scholar] [CrossRef]
- Fragouli, E.; Lenzi, M.; Ross, R.; Katz-Jaffe, M.; Schoolcraft, W.B.; Wells, D. Comprehensive molecular cytogenetic analysis of the human blastocyst stage. Hum. Reprod. 2008, 23, 2596–2608. [Google Scholar] [CrossRef] [PubMed]
- Barbash-Hazan, S.; Frumkin, T.; Malcov, M.; Yaron, Y.; Cohen, T.; Azem, F.; Amit, A.; Ben-Yosef, D. Preimplantation aneuploid embryos undergo self-correction in correlation with their developmental potential. Fertil. Steril. 2009, 92, 890–896. [Google Scholar] [CrossRef]
- Fragouli, E.; Munne, S.; Wells, D. The cytogenetic constitution of human blastocysts: Insights from comprehensive chromosome screening strategies. Hum. Reprod. Update 2019, 25, 15–33. [Google Scholar] [CrossRef] [PubMed]
- Zhu, P.; Guo, H.; Ren, Y.; Hou, Y.; Dong, J.; Li, R.; Lian, Y.; Fan, X.; Hu, B.; Gao, Y.; et al. Single-cell DNA methylome sequencing of human preimplantation embryos. Nat. Genet. 2018, 50, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Bolton, H.; Graham, S.J.L.; Van der Aa, N.; Kumar, P.; Theunis, K.; Fernandez Gallardo, E.; Voet, T.; Zernicka-Goetz, M. Mouse model of chromosome mosaicism reveals lineage-specific depletion of aneuploid cells and normal developmental potential. Nat. Commun. 2016, 7, 11165. [Google Scholar] [CrossRef]
- Gleicher, N.; Metzger, J.; Croft, G.; Kushnir, V.A.; Albertini, D.F.; Barad, D.H. A single trophectoderm biopsy at blastocyst stage is mathematically unable to determine embryo ploidy accurately enough for clinical use. Reprod. Biol. Endocrinol. 2017, 15, 33. [Google Scholar] [CrossRef]
- Ramos, L.; del Rey, J.; Daina, G.; Martinez-Passarell, O.; Rius, M.; Tunon, D.; Campillo, M.; Benet, J.; Navarro, J. Does the S phase have an impact on the accuracy of comparative genomic hybridization profiles in single fibroblasts and human blastomeres? Fertil. Steril. 2014, 101, 488–495. [Google Scholar] [CrossRef]
- Harton, G.L.; Cinnioglu, C.; Fiorentino, F. Current experience concerning mosaic embryos diagnosed during preimplantation genetic screening. Fertil. Steril. 2017, 107, 1113–1119. [Google Scholar] [CrossRef]
- Palmerola, K.L.; Vitez, S.F.; Amrane, S.; Fischer, C.P.; Forman, E.J. Minimizing mosaicism: Assessing the impact of fertilization method on rate of mosaicism after next-generation sequencing (NGS) preimplantation genetic testing for aneuploidy (PGT-A). J. Assist. Reprod. Genet. 2019, 36, 153–157. [Google Scholar] [CrossRef]
- Kasius, A.; Smit, J.G.; Torrance, H.L.; Eijkemans, M.J.; Mol, B.W.; Opmeer, B.C.; Broekmans, F.J. Endometrial thickness and pregnancy rates after IVF: A systematic review and meta-analysis. Hum. Reprod. Update 2014, 20, 530–541. [Google Scholar] [CrossRef]
- Sermondade, N.; Huberlant, S.; Bourhis-Lefebvre, V.; Arbo, E.; Gallot, V.; Colombani, M.; Freour, T. Female obesity is negatively associated with live birth rate following IVF: A systematic review and meta-analysis. Hum. Reprod. Update 2019, 25, 439–451. [Google Scholar] [CrossRef] [PubMed]
- Boynukalin, F.K.; Gultomruk, M.; Cavkaytar, S.; Turgut, E.; Findikli, N.; Serdarogullari, M.; Coban, O.; Yarkiner, Z.; Rubio, C.; Bahceci, M. Parameters impacting the live birth rate per transfer after frozen single euploid blastocyst transfer. PLoS ONE 2020, 15, e0227619. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Zhang, B.; Jiang, X.; Li, Z.; Zhao, S.; Cui, L.; Chen, Z.J. Metabolic disturbances in non-obese women with polycystic ovary syndrome: A systematic review and meta-analysis. Fertil. Steril. 2019, 111, 168–177. [Google Scholar] [CrossRef] [PubMed]
- Kahraman, S.; Cetinkaya, M.; Yuksel, B.; Yesil, M.; Pirkevi Cetinkaya, C. The birth of a baby with mosaicism resulting from a known mosaic embryo transfer: A case report. Hum. Reprod. 2020, 35, 727–733. [Google Scholar] [CrossRef]
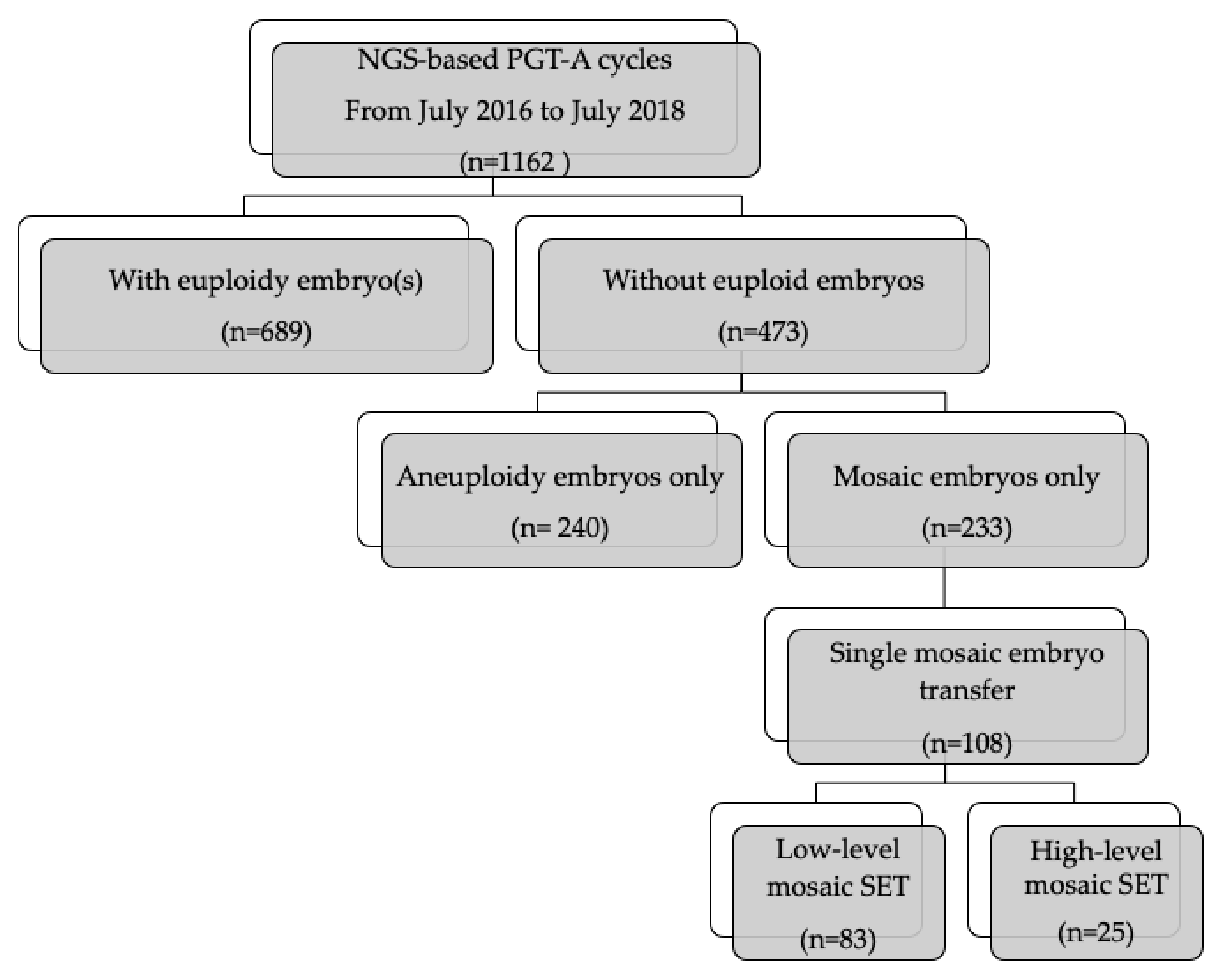
| Low-Level Mosaic SET | High-Level Mosaic SET | p Value | |
|---|---|---|---|
| No. of cycles | 83 | 25 | |
| Age of female partners (y) | 36.9 ± 5.6 | 36.2 ± 4.8 | 0.38 |
| No. of oocytes retrieved | 16.4 ± 11 | 15.6 ± 10.8 | 0.88 |
| No. of good-quality blastocysts | 4.9 ± 4.7 | 2.62 ± 4.4 | 0.2 |
| Blastocyst formation rate (%) | 50.4 ± 27 | 31.5 ± 22 | 0.1 |
| Implantation rate, (%) (n) | 51.8% (43/83) | 52% (13/25) | 0.98 |
| Clinical pregnancy rate, % (n) | 47% (39/83) | 52% (13/25) | 0.66 |
| Ongoing pregnancy rate, % (n) | 47% (39/83) | 36% (9/25) | 0.33 |
| Miscarriage rate, % (n) | 5.1% (2/39) | 30.7% (4/13) | 0.012 a |
| Live birth rate, % (n) | 44.6% (37/83) | 36% (9/25) | 0.45 |
| Multiple gestation, % (n) | 10.8% (4/37) | 0% (0/9) | 0.57 |
| Gestational age at delivery (weeks) | 38.3 ± 1.6 | 38.6 ± 1.2 | 0.53 |
| Birth weight (gm) | 3015 ± 507 | 3076 ± 94 | 0.73 |
| Univariable Analysis | Multivariate Analysis | |||
|---|---|---|---|---|
| Variable | OR (95% CI) | p Value | OR (95% CI) | p Value |
| Age, years | 0.91 (0.84–0.98) | 0.01 a | 0.89 (0.82–0.97) | 0.01 a |
| EM thickness (mm) | 1.51 (1.11–2.05) | 0.008 a | 1.54 (1.12–2.11) | 0.007 a |
| BMI (kg/m2) | 0.86 (0.74–0.99) | 0.04 a | 0.85 (0.73–0.99) | 0.035 a |
| Estradiol (pg/mL) on ET day | 1.00 (1.00–1.02) | 0.03 a | 1.0 (0.99–1.03) | 0.06 |
| EM preparation Nature cycle hormone replacement | 0.30 (0.13–0.66) | 0.003 a | 0.41 (0.16–1.03) | 0.06 |
| Duration of infertility (years) | 0.87 (0.75–1.01) | 0.07 | 0.90 (0.78–1.06) | 0.22 |
| No. of oocytes retrieved | 0.95 (0.88–1.02) | 0.19 | ||
| No. of 2PN | 0.94 (0.83–1.07) | |||
| No. of good–quality blastocysts | 1.19 (0.91–1.56) | 0.20 | ||
| Blastocyst rate (%) | 0.22 (0.01–5.37) | 0.35 | ||
| Day 5/6 blastocyst | 2.2 (0.87–5.61) | 0.1 | ||
| Progesterone (pg/mL) on ET day | 1.0 (0.99–1.02)) | 0.49 | ||
| Mosaic level Low (<50%) High (≥50%) | 0.67 (0.26–1.68) | 0.39 | ||
| Mosaic type Segmental Whole chromosome Complex | 1.75 (0.81–3.78) | 0.15 | ||
| Mosaic No. | 0.94 (0.77–1.15) | 0.57 | ||
| Interaction | ||||
| Mosaic level x type | 0.54 (0.30–0.99) | 0.045 a | 1.74 (0.16–18.43) | 0.65 |
| Mosaic level x No. | 0.90 (0.75–1.09) | 0.29 | ||
| Mosaic type x No. | 1.02 (0.92–1.33) | 0.13 | ||
| Mosaic level x type x No. | 0.89 (0.78–1.03) | 0.11 | ||
| Origin TE Biopsy | ICM Results | Proportion | Concordance | |
|---|---|---|---|---|
| High-level mosaicism (N = 27) | Aneuploid | 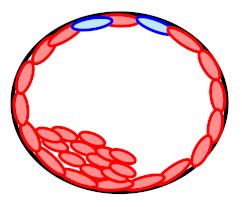 | 22.2% (6/27) | Discordance |
| Mosaicism | 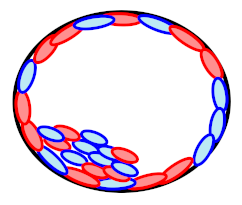 | 37.0% (10/27) | Concordance | |
| Euploid | 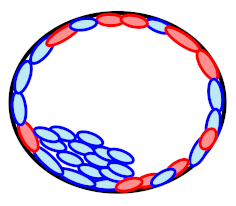 | 40.7% (11/27) | Discordance | |
| Low-level mosaicism (N = 14) | Aneuploid | 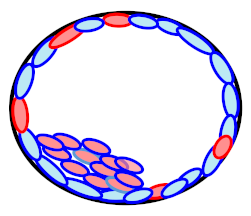 | 0% (0/14) | Discordance |
| Mosaicism | 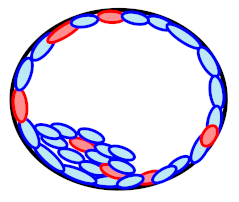 | 50% (7/14) | Concordance | |
| Euploid | 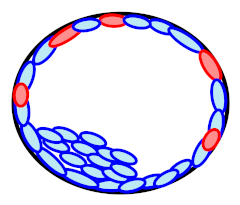 | 50% (7/14) | Discordance | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, P.-Y.; Lee, C.-I.; Cheng, E.-H.; Huang, C.-C.; Lee, T.-H.; Shih, H.-H.; Pai, Y.-P.; Chen, Y.-C.; Lee, M.-S. Clinical Outcomes of Single Mosaic Embryo Transfer: High-Level or Low-Level Mosaic Embryo, Does It Matter? J. Clin. Med. 2020, 9, 1695. https://doi.org/10.3390/jcm9061695
Lin P-Y, Lee C-I, Cheng E-H, Huang C-C, Lee T-H, Shih H-H, Pai Y-P, Chen Y-C, Lee M-S. Clinical Outcomes of Single Mosaic Embryo Transfer: High-Level or Low-Level Mosaic Embryo, Does It Matter? Journal of Clinical Medicine. 2020; 9(6):1695. https://doi.org/10.3390/jcm9061695
Chicago/Turabian StyleLin, Pin-Yao, Chun-I Lee, En-Hui Cheng, Chun-Chia Huang, Tsung-Hsien Lee, Hui-Hsin Shih, Yi-Ping Pai, Yi-Chun Chen, and Maw-Sheng Lee. 2020. "Clinical Outcomes of Single Mosaic Embryo Transfer: High-Level or Low-Level Mosaic Embryo, Does It Matter?" Journal of Clinical Medicine 9, no. 6: 1695. https://doi.org/10.3390/jcm9061695
APA StyleLin, P.-Y., Lee, C.-I., Cheng, E.-H., Huang, C.-C., Lee, T.-H., Shih, H.-H., Pai, Y.-P., Chen, Y.-C., & Lee, M.-S. (2020). Clinical Outcomes of Single Mosaic Embryo Transfer: High-Level or Low-Level Mosaic Embryo, Does It Matter? Journal of Clinical Medicine, 9(6), 1695. https://doi.org/10.3390/jcm9061695






