Serological Response to Three, Four and Five Doses of SARS-CoV-2 Vaccine in Kidney Transplant Recipients
Abstract
:1. Introduction
2. Methods
Study Population
3. Outcome
3.1. Multivariable Analysis of Predictors of Serological Response
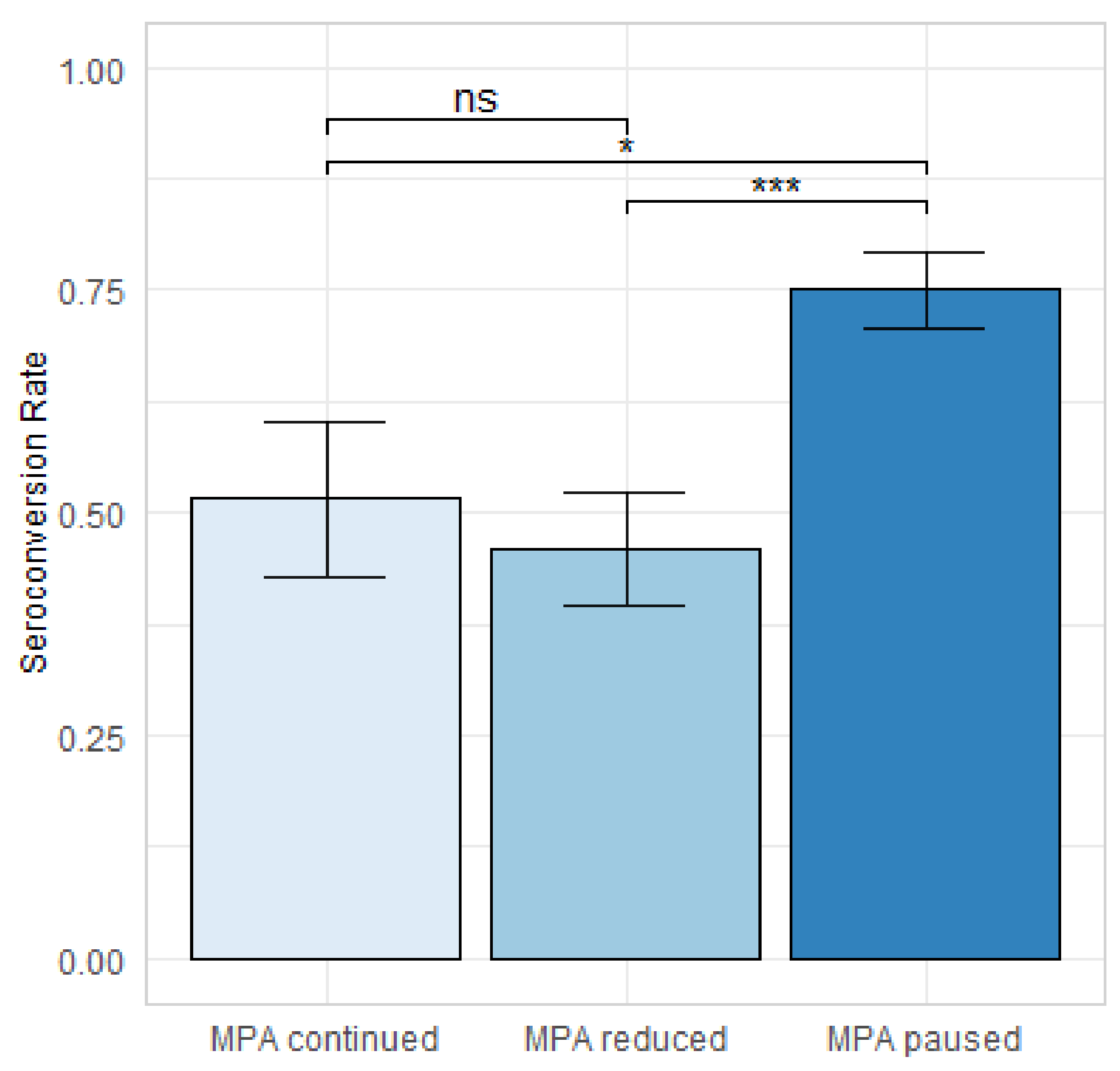
3.2. Comparison of Mycophenolic Acid (MPA) Dose Adjustment Regimens
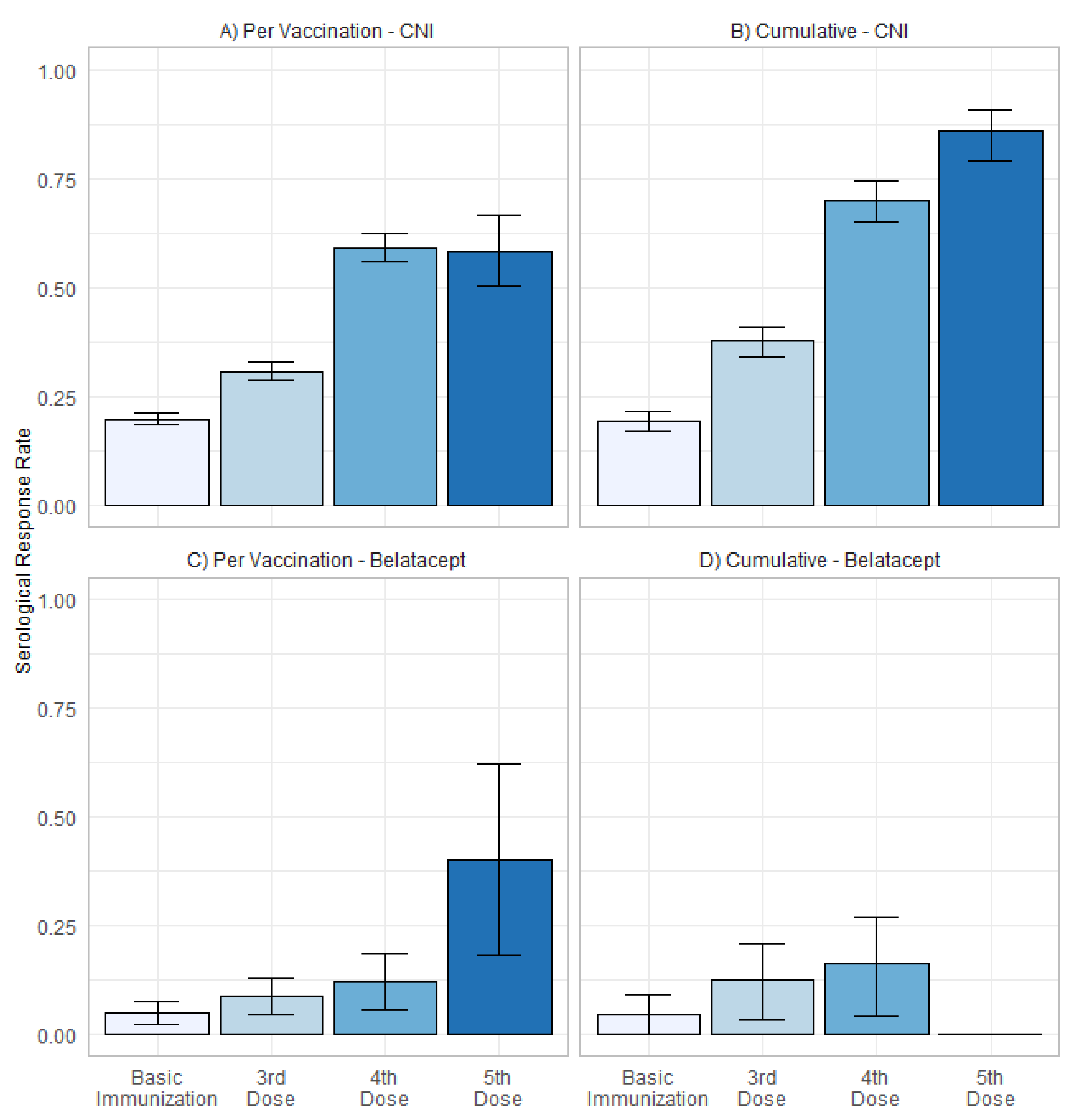
3.3. Serological Response in Patients with CNI and Belatacept Maintenance Immunosuppression
4. Results
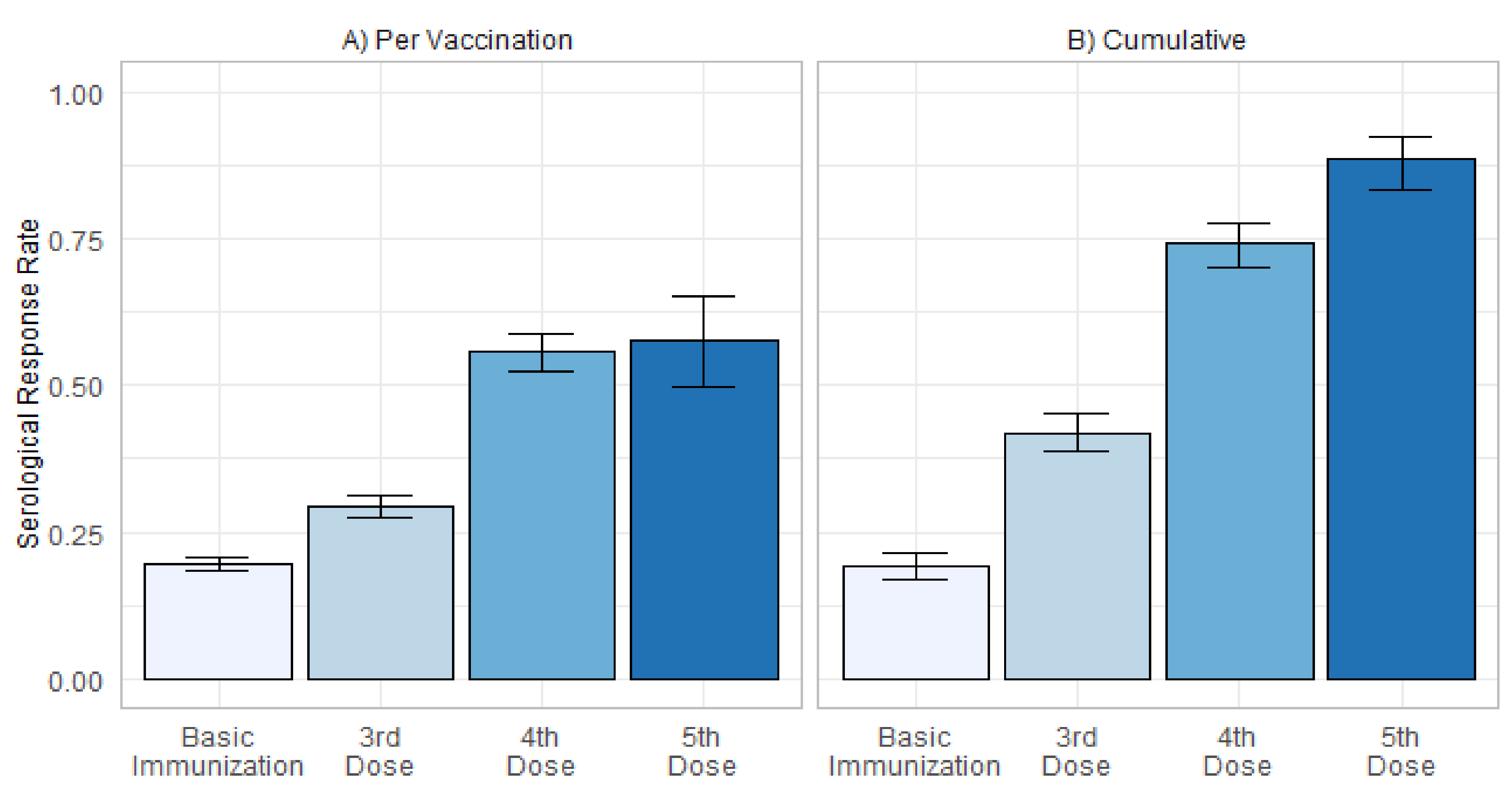
4.1. Serological Response to Immunization against SARS-CoV-2
4.2. Predictors of Serological Response to Immunization against SARS-CoV-2
4.3. Change in MPA Dose as Predictor of Serological Response after Four Doses of SARS-CoV-2 Vaccine
4.4. Belatacept-Based Immunosuppression as Predictor of Serological Response
5. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CNI | calcineurin inhibitor |
| COVID-19 | coronavirus disease 2019 |
| eGFR | estimated glomerular filtration rate |
| ELISA | enzyme-linked immunosorbent assays |
| KTR | kidney transplant recipients |
| MPA | mycophenolic acid |
| N | nucleocapsid protein |
| RBD | receptor binding domain |
| S | spike protein |
| SARS-CoV-2 | severe acute respiratory syndrome coronavirus 2 |
| SOT | solid organ transplantation |
| TCMR | T cell mediated rejection |
References
- Andrews, N.; Tessier, E.; Stowe, J.; Gower, C.; Kirsebom, F.; Simmons, R.; Gallagher, E.; Thelwall, S.; Groves, N.; Dabrera, G.; et al. Duration of Protection against Mild and Severe Disease by Covid-19 Vaccines. N. Engl. J. Med. 2022, 386, 340–350. [Google Scholar] [CrossRef]
- Phadke, V.K.; Scanlon, N.; Jordan, S.C.; Rouphael, N.G. Immune Responses to SARS-CoV-2 in Solid Organ Transplant Recipients. Curr. Transplant. Rep. 2021, 8, 127–139. [Google Scholar] [CrossRef] [PubMed]
- Reischig, T.; Kacer, M.; Vlas, T.; Drenko, P.; Kielberger, L.; Machova, J.; Topolcan, O.; Kucera, R.; Kormunda, S. Insufficient response to mRNA SARS-CoV-2 vaccine and high incidence of severe COVID-19 in kidney transplant recipients during pandemic. Am. J. Transplant. 2021, 22, 801–812. [Google Scholar] [CrossRef] [PubMed]
- Osmanodja, B.; Mayrdorfer, M.; Halleck, F.; Choi, M.; Budde, K. Undoubtedly, kidney transplant recipients have a higher mortality due to COVID-19 disease compared to the general population. Transpl. Int. 2021, 34, 769–771. [Google Scholar] [CrossRef] [PubMed]
- CDC. CDC COVID Data Tracker. Available online: https://covid.cdc.gov/covid-data-tracker/#rates-by-vaccine-status (accessed on 28 January 2022).
- Boyarsky, B.J.; Werbel, W.A.; Avery, R.K.; Tobian, A.A.R.; Massie, A.B.; Segev, D.L.; Garonzik-Wang, J.M. Antibody Response to 2-Dose SARS-CoV-2 mRNA Vaccine Series in Solid Organ Transplant Recipients. JAMA 2021, 325, 2204–2206. [Google Scholar] [CrossRef] [PubMed]
- Sattler, A.; Schrezenmeier, E.; Weber, U.A.; Potekhin, A.; Bachmann, F.; Straub-Hohenbleicher, H.; Budde, K.; Storz, E.; Pross, V.; Bergmann, Y.; et al. Impaired humoral and cellular immunity after SARS-CoV-2 BNT162b2 (tozinameran) prime-boost vaccination in kidney transplant recipients. J. Clin. Investig. 2021, 131, 150175. [Google Scholar] [CrossRef] [PubMed]
- Rincon-Arevalo, H.; Choi, M.; Stefanski, A.L.; Halleck, F.; Weber, U.; Szelinski, F.; Jahrsdorfer, B.; Schrezenmeier, H.; Ludwig, C.; Sattler, A.; et al. Impaired humoral immunity to SARS-CoV-2 BNT162b2 vaccine in kidney transplant recipients and dialysis patients. Sci. Immunol. 2021, 6, eabj1031. [Google Scholar] [CrossRef] [PubMed]
- Feng, S.; Phillips, D.J.; White, T.; Sayal, H.; Aley, P.K.; Bibi, S.; Dold, C.; Fuskova, M.; Gilbert, S.C.; Hirsch, I.; et al. Correlates of protection against symptomatic and asymptomatic SARS-CoV-2 infection. Nat. Med. 2021, 27, 2032–2040. [Google Scholar] [CrossRef] [PubMed]
- Khoury, D.S.; Cromer, D.; Reynaldi, A.; Schlub, T.E.; Wheatley, A.K.; Juno, J.A.; Subbarao, K.; Kent, S.J.; Triccas, J.A.; Davenport, M.P. Neutralizing antibody levels are highly predictive of immune protection from symptomatic SARS-CoV-2 infection. Nat. Med. 2021, 27, 1205–1211. [Google Scholar] [CrossRef]
- Kamar, N.; Abravanel, F.; Marion, O.; Couat, C.; Izopet, J.; Del Bello, A. Three Doses of an mRNA Covid-19 Vaccine in Solid-Organ Transplant Recipients. N. Engl. J. Med. 2021, 385, 661–662. [Google Scholar] [CrossRef]
- Schrezenmeier, E.; Rincon-Arevalo, H.; Stefanski, A.L.; Potekhin, A.; Straub-Hohenbleicher, H.; Choi, M.; Bachmann, F.; Pross, V.; Hammett, C.; Schrezenmeier, H.; et al. B and T Cell Responses after a Third Dose of SARS-CoV-2 Vaccine in Kidney Transplant Recipients. J. Am. Soc. Nephrol. JASN 2021, 32, 3027–3033. [Google Scholar] [CrossRef]
- Schrezenmeier, E.; Rincon-Arevalo, H.; Jens, A.; Stefanski, A.L.; Hammett, C.; Osmanodja, B.; Koch, N.; Zukunft, B.; Beck, J.; Oellerich, M.; et al. Temporary antimetabolite treatment hold boosts SARS-CoV-2 vaccination-specific humoral and cellular immunity in kidney transplant recipients. JCI Insight 2022, in press. [Google Scholar] [CrossRef] [PubMed]
- Qin, C.X.; Moore, L.W.; Anjan, S.; Rahamimov, R.; Sifri, C.D.; Ali, N.M.; Morales, M.K.; Tsapepas, D.S.; Basic-Jukic, N.; Miller, R.A.; et al. Risk of Breakthrough SARS-CoV-2 Infections in Adult Transplant Recipients. Transplantation 2021, 105, e265–e266. [Google Scholar] [CrossRef] [PubMed]
- Schrezenmeier, E.; Bergfeld, L.; Hillus, D.; Lippert, J.D.; Weber, U.; Tober-Lau, P.; Landgraf, I.; Schwarz, T.; Kappert, K.; Stefanski, A.L.; et al. Immunogenicity of COVID-19 Tozinameran Vaccination in Patients on Chronic Dialysis. Front. Immunol. 2021, 12, 690698. [Google Scholar] [CrossRef] [PubMed]
- Jahrsdorfer, B.; Kroschel, J.; Ludwig, C.; Corman, V.M.; Schwarz, T.; Korper, S.; Rojewski, M.; Lotfi, R.; Weinstock, C.; Drosten, C.; et al. Independent Side-by-Side Validation and Comparison of 4 Serological Platforms for SARS-CoV-2 Antibody Testing. J. Infect. Dis. 2021, 223, 796–801. [Google Scholar] [CrossRef]
- Caillard, S.; Thaunat, O. COVID-19 vaccination in kidney transplant recipients. Nat. Rev. Nephrol. 2021, 17, 785–787. [Google Scholar] [CrossRef]
- Kaplan, E.L.; Meier, P. Nonparametric-Estimation from Incomplete Observations. J. Am. Stat. Assoc. 1958, 53, 457–481. [Google Scholar] [CrossRef]
- Liefeldt, L.; Glander, P.; Klotsche, J.; Straub-Hohenbleicher, H.; Budde, K.; Eberspacher, B.; Friedersdorff, F.; Halleck, F.; Hambach, P.; Hofmann, J.; et al. Predictors of Serological Response to SARS-CoV-2 Vaccination in Kidney Transplant Patients: Baseline Characteristics, Immunosuppression, and the Role of IMPDH Monitoring. J. Clin. Med. 2022, 11, 1697. [Google Scholar] [CrossRef]
- Caillard, S.; Thaunat, O.; Benotmane, I.; Masset, C.; Blancho, G. Antibody Response to a Fourth Messenger RNA COVID-19 Vaccine Dose in Kidney Transplant Recipients: A Case Series. Ann. Intern. Med. 2022, 175, 455–456. [Google Scholar] [CrossRef]
- Alejo, J.L.; Mitchell, J.; Chiang, T.P.; Abedon, A.T.; Boyarsky, B.J.; Avery, R.K.; Tobian, A.A.R.; Levan, M.L.; Massie, A.B.; Garonzik-Wang, J.M.; et al. Antibody Response to a Fourth Dose of a SARS-CoV-2 Vaccine in Solid Organ Transplant Recipients: A Case Series. Transplantation 2021, 105, e280–e281. [Google Scholar] [CrossRef]
- Chavarot, N.; Morel, A.; Leruez-Ville, M.; Vilain, E.; Divard, G.; Burger, C.; Serris, A.; Sberro-Soussan, R.; Martinez, F.; Amrouche, L.; et al. Weak antibody response to three doses of mRNA vaccine in kidney transplant recipients treated with belatacept. Am. J. Transplant. 2021, 21, 4043–4051. [Google Scholar] [CrossRef] [PubMed]
- Noble, J.; Langello, A.; Bouchut, W.; Lupo, J.; Lombardo, D.; Rostaing, L. Immune Response Post-SARS-CoV-2 mRNA Vaccination in Kidney Transplant Recipients Receiving Belatacept. Transplantation 2021, 105, e259–e260. [Google Scholar] [CrossRef] [PubMed]
- Debska-Slizien, A.; Slizien, Z.; Muchlado, M.; Kubanek, A.; Piotrowska, M.; Dabrowska, M.; Tarasewicz, A.; Chamienia, A.; Biedunkiewicz, B.; Renke, M.; et al. Predictors of Humoral Response to mRNA COVID19 Vaccines in Kidney Transplant Recipients: A Longitudinal Study-The COViNEPH Project. Vaccines 2021, 9, 1165. [Google Scholar] [CrossRef] [PubMed]
- Stumpf, J.; Siepmann, T.; Lindner, T.; Karger, C.; Schwobel, J.; Anders, L.; Faulhaber-Walter, R.; Schewe, J.; Martin, H.; Schirutschke, H.; et al. Humoral and cellular immunity to SARS-CoV-2 vaccination in renal transplant versus dialysis patients: A prospective, multicenter observational study using mRNA-1273 or BNT162b2 mRNA vaccine. Lancet Reg. Health Eur. 2021, 9, 100178. [Google Scholar] [CrossRef] [PubMed]
- Tober-Lau, P.; Schwarz, T.; Vanshylla, K.; Hillus, D.; Gruell, H.; Group, E.C.S.; Suttorp, N.; Landgraf, I.; Kappert, K.; Seybold, J.; et al. Long-term immunogenicity of BNT162b2 vaccination in older people and younger health-care workers. Lancet Respir. Med. 2021, 9, e104–e105. [Google Scholar] [CrossRef]
- Jahrsdorfer, B.; Fabricius, D.; Scholz, J.; Ludwig, C.; Grempels, A.; Lotfi, R.; Korper, S.; Adler, G.; Schrezenmeier, H. BNT162b2 Vaccination Elicits Strong Serological Immune Responses Against SARS-CoV-2 Including Variants of Concern in Elderly Convalescents. Front. Immunol. 2021, 12, 743422. [Google Scholar] [CrossRef]
- Wang, J.; Tong, Y.; Li, D.; Li, J.; Li, Y. The Impact of Age Difference on the Efficacy and Safety of COVID-19 Vaccines: A Systematic Review and Meta-Analysis. Front. Immunol. 2021, 12, 758294. [Google Scholar] [CrossRef] [PubMed]
- Semenzato, L.; Botton, J.; Drouin, J.; Cuenot, F.; Dray-Spira, R.; Weill, A.; Zureik, M. Chronic diseases, health conditions and risk of COVID-19-related hospitalization and in-hospital mortality during the first wave of the epidemic in France: A cohort study of 66 million people. Lancet Reg. Health Eur. 2021, 8, 100158. [Google Scholar] [CrossRef]
- Espi, M.; Charmetant, X.; Barba, T.; Mathieu, C.; Pelletier, C.; Koppe, L.; Chalencon, E.; Kalbacher, E.; Mathias, V.; Ovize, A.; et al. A prospective observational study for justification, safety, and efficacy of a third dose of mRNA vaccine in patients receiving maintenance hemodialysis. Kidney Int. 2022, 101, 390–402. [Google Scholar] [CrossRef]
- Wendel, S.; Fontao-Wendel, R.; Fachini, R.; Candelaria, G.; Scuracchio, P.; Achkar, R.; Brito, M.; Reis, L.F.; Camargo, A.; Amano, M.; et al. A longitudinal study of convalescent plasma (CCP) donors and correlation of ABO group, initial neutralizing antibodies (nAb), and body mass index (BMI) with nAb and anti-nucleocapsid (NP) SARS-CoV-2 antibody kinetics: Proposals for better quality of CCP collections. Transfusion 2021, 61, 1447–1460. [Google Scholar] [CrossRef]
- Lee, S.W.; Moon, J.Y.; Lee, S.K.; Lee, H.; Moon, S.; Chung, S.J.; Yeo, Y.; Park, T.S.; Park, D.W.; Kim, T.H.; et al. Anti-SARS-CoV-2 Spike Protein RBD Antibody Levels After Receiving a Second Dose of ChAdOx1 nCov-19 (AZD1222) Vaccine in Healthcare Workers: Lack of Association with Age, Sex, Obesity, and Adverse Reactions. Front. Immunol. 2021, 12, 779212. [Google Scholar] [CrossRef] [PubMed]
- Pellini, R.; Venuti, A.; Pimpinelli, F.; Abril, E.; Blandino, G.; Campo, F.; Conti, L.; De Virgilio, A.; De Marco, F.; Di Domenico, E.G.; et al. Initial observations on age, gender, BMI and hypertension in antibody responses to SARS-CoV-2 BNT162b2 vaccine. EClinicalMedicine 2021, 36, 100928. [Google Scholar] [CrossRef] [PubMed]
- Dürr, M.; Lachmann, N.; Zukunft, B.; Schmidt, D.; Budde, K.; Brakemeier, S. Late Conversion to Belatacept After Kidney Transplantation: Outcome and Prognostic Factors. Transpl. Proc. 2017, 49, 1747–1756. [Google Scholar] [CrossRef] [PubMed]
- Brakemeier, S.; Kannenkeril, D.; Dürr, M.; Braun, T.; Bachmann, F.; Schmidt, D.; Wiesener, M.; Budde, K. Experience with belatacept rescue therapy in kidney transplant recipients. Transpl. Int. 2016, 29, 1184–1195. [Google Scholar] [CrossRef] [Green Version]
- Darres, A.; Ulloa, C.; Brakemeier, S.; Garrouste, C.; Bestard, O.; del Bello, A.; Soussan, R.S.; Dürr, M.; Budde, K.; Legendre, C.; et al. Conversion to Belatacept in Maintenance Kidney Transplant Patients: A Retrospective Multicenter European Study. Transplantation 2018, 102, 1545–1552. [Google Scholar] [CrossRef]

| Inclusion Criteria per Patient |
|---|
|
|
|
| Exclusion Criteria per Vaccination |
|
|
|
|
|
|
|
| Vaccination Number | 3 | 4 | 5 |
|---|---|---|---|
| Total Patients | 603 | 250 | 40 |
| Demographics and Comorbidities | |||
| Female/male patients | 38%/62% | 33%/67% | 45%/55% |
| Median age in years (IQR) | 59 (48–68) | 61 (51–70) | 63 (56–72) |
| BMI in kg/m2 | 25.4 ± 4.7 | 25.0 ± 4.4 | 24.76 ± 4.77 |
| Diabetes | 21.7% | 21.8% | 18.0% |
| Transplantation | |||
| Median transplant age in years (IQR) | 8.2 (3.1–13.5) | 7.7 (3.0–12.7) | 7.2 (2.2–11.5) |
| Tacrolimus-based IS | 73.6% | 73.6% | 77.5% |
| Ciclosporin-based IS | 16.8% | 16.4% | 12.5% |
| Belatacept-based IS | 7.6% | 10% | 12.5% |
| Patients with MPA | 93.7% | 50.4% | 42.5% |
| Patients with mTORi | 1.0% | - | - |
| Patients with Azathioprine | 0.8% | - | - |
| Patients with more than 2 immunosuppressive drugs | 60.7% | 39.6% | 32.5% |
| Laboratory Values | |||
| Baseline eGFR in mL/min/1.73 m2 | 51.2 ± 20.0 | 47.5 ± 19.6 | 43.35 ± 18.68 |
| Urine albumin creatinine ratio in g/g | 0.14 ± 0.45 | 0.19 ± 0.62 | 0.44 ± 1.45 |
| Hemoglobin in g/dL | 12.68 ± 1.65 | 12.56 ± 1.52 | 12.73 ± 1.69 |
| Leukocyte count in /nL | 7.33 ± 2.38 | 7.41 ± 2.53 | 7.86 ± 2.44 |
| Vaccination | |||
| Baseline SARS-CoV-2 IgG low positive | 3.5% | 11.6% | 45% |
| mRNA Vaccination | 72.8% | 86.8% | 100% |
| Median time since previous vaccination in days (IQR) | 71 (53–102) | 64 (55–84) | 62 (46–70) |
| Variable | Odds Ratio (95% CI) | Adj. p-Value |
|---|---|---|
| Low positive anti-SARS-CoV-2-S-protein IgG before vaccination | 28.5 (7.18–201) | <0.001 |
| Age | 0.98 (0.96–0.99) | 0.016 |
| BMI | 1.06 (1.01–1.11) | 0.012 |
| Transplant age | 1.06 (1.03–1.09) | <0.001 |
| Belatacept | 0.15 (0.03–0.45) | 0.008 |
| MPA dose in MMF equivalent in g | 0.29 (0.20–0.43) | <0.001 |
| eGFR in mL/min/1.73 m2 | 1.02 (1.01–1.04) | <0.001 |
| Hemoglobin | 1.29 (1.13–1.49) | 0.001 |
| Variable | Odds Ratio (95% CI) | Adj. p-Value |
|---|---|---|
| Low positive anti-SARS-CoV-2-S-protein IgG before vaccination | 18.7 (4.68–134) | 0.001 |
| Age | 0.96 (0.94–0.99) | 0.004 |
| Transplant age | 1.09 (1.04–1.16) | 0.002 |
| Belatacept | 0.03 (0.004–0.13) | <0.001 |
| MPA dose in MMF equivalent in g | 0.34 (0.18–0.59) | 0.001 |
| MPA Pause | MPA Reduction | MPA Continuation | |
|---|---|---|---|
| Number of Patients | 104 | 63 | 33 |
| Median age in years (IQR) | 60 (51–70) | 63 (47–70) | 64 (55–71) |
| Female | 33.7% | 25.4% | 33.3% |
| BMI in kg/m2 | 25.5 ± 4.3 | 24.2 ± 4.3 | 24.6 ± 4.2 |
| Median transplant Age in years (IQR) | 7.6 (3.0–7.5) | 6.8 (2.1–11.4) | 4.9 (2.3–9.6) |
| eGFR in mL/min/1.73 m2 | 47.6 ± 19.0 | 55.1 ± 17.8 | 44.7 ± 8.1 |
| Change in eGFR in mL/min/1.73 m2 | −1.09 ± 6.73 | −2.47 ± 9.30 | 0.53 ± 5.96 |
| ACR in g/g | 0.18 ± 0.77 | 0.11 ± 0.21 | 0.13 ± 0.23 |
| Change in ACR in g/g | 0.02 ± 0.21 | 0.03 ± 0.30 | −0.05 ± 0.16 |
| mRNA Vaccination | 96.2% | 76.2% | 75.8% |
| IgG low positive before vaccination | 12.5% | 7.9% | 24.3% |
| MPA dose in g MMF equivalent | 0 | 0.87 ± 0.25 | 1.18 ± 0.43 |
| Patients with steroid treatment | 100% | 76.2% | 57.6% |
| Steroid dose in mg methylprednisolone equivalent | 4.1 ± 0.8 | 2.8 ± 1.8 | 1.8 ± 1.9 |
| Median time since last vaccination in days (IQR) | 65 (56–83) | 65 (58–94) | 65 (48–87) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Osmanodja, B.; Ronicke, S.; Budde, K.; Jens, A.; Hammett, C.; Koch, N.; Seelow, E.; Waiser, J.; Zukunft, B.; Bachmann, F.; et al. Serological Response to Three, Four and Five Doses of SARS-CoV-2 Vaccine in Kidney Transplant Recipients. J. Clin. Med. 2022, 11, 2565. https://doi.org/10.3390/jcm11092565
Osmanodja B, Ronicke S, Budde K, Jens A, Hammett C, Koch N, Seelow E, Waiser J, Zukunft B, Bachmann F, et al. Serological Response to Three, Four and Five Doses of SARS-CoV-2 Vaccine in Kidney Transplant Recipients. Journal of Clinical Medicine. 2022; 11(9):2565. https://doi.org/10.3390/jcm11092565
Chicago/Turabian StyleOsmanodja, Bilgin, Simon Ronicke, Klemens Budde, Annika Jens, Charlotte Hammett, Nadine Koch, Evelyn Seelow, Johannes Waiser, Bianca Zukunft, Friederike Bachmann, and et al. 2022. "Serological Response to Three, Four and Five Doses of SARS-CoV-2 Vaccine in Kidney Transplant Recipients" Journal of Clinical Medicine 11, no. 9: 2565. https://doi.org/10.3390/jcm11092565
APA StyleOsmanodja, B., Ronicke, S., Budde, K., Jens, A., Hammett, C., Koch, N., Seelow, E., Waiser, J., Zukunft, B., Bachmann, F., Choi, M., Weber, U., Eberspächer, B., Hofmann, J., Grunow, F., Mikhailov, M., Liefeldt, L., Eckardt, K.-U., Halleck, F., & Schrezenmeier, E. (2022). Serological Response to Three, Four and Five Doses of SARS-CoV-2 Vaccine in Kidney Transplant Recipients. Journal of Clinical Medicine, 11(9), 2565. https://doi.org/10.3390/jcm11092565






