Acceptance of COVID-19 Vaccination during the COVID-19 Pandemic in China
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design, Population and Sampling
2.2. Measures
2.3. Statistical Analysis
3. Results
3.1. Study Sample Characteristics
3.2. Acceptance, Preferences and Impact Factors of the Future COVID-19 Vaccine
3.3. Influencing Factors of Vaccination Acceptance
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
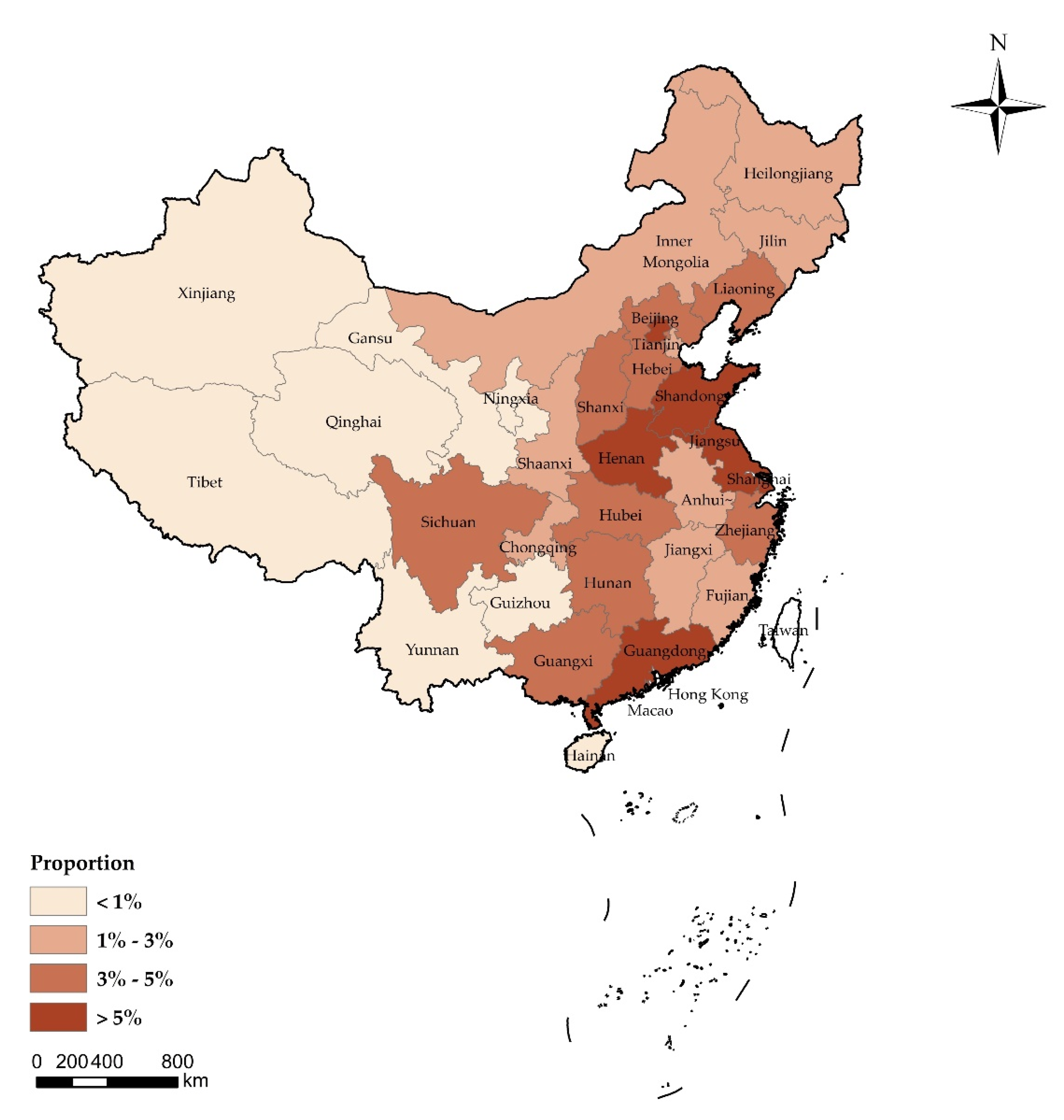
Appendix A
| Provincial Administrative Regions | Respondents (N = 2058) |
|---|---|
| N (%) | |
| Beijing | 138 (6.7) |
| Tianjin | 37 (1.8) |
| Hebei | 96 (4.7) |
| Shanxi | 68 (3.3) |
| Inner Mongolia | 24 (1.2) |
| Liaoning | 69 (3.4) |
| Jilin | 24 (1.2) |
| Heilongjiang | 38 (1.8) |
| Shanghai | 146 (7.1) |
| Jiangsu | 130 (6.3) |
| Zhejiang | 88 (4.3) |
| Anhui | 62 (3.0) |
| Fujian | 62 (3.0) |
| Jiangxi | 38 (1.8) |
| Shandong | 129 (6.3) |
| Henan | 134 (6.5) |
| Hubei | 99 (4.8) |
| Hunan | 68 (3.3) |
| Guangdong | 295 (14.3) |
| Guangxi | 64 (3.1) |
| Hainan | 5 (0.2) |
| Chongqing | 30 (1.5) |
| Sichuan | 86 (4.2) |
| Guizhou | 19 (0.9) |
| Yunnan | 18 (0.9) |
| Tibet | 1 (0.0) |
| Shaanxi | 51 (2.5) |
| Gansu | 17 (0.8) |
| Qinghai | 2 (0.1) |
| Ningxia | 7 (0.3) |
| Xinjiang | 13 (0.6) |
References
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- World Health Organization. WHO Coronavirus Disease (COVID-19) Dashboard. Available online: https://covid19.who.int/?gclid=EAIaIQobChMI2_CM6eDZ6gIVghh9Ch3nDQm1EAAYASAAEgLqwPD_BwE (accessed on 17 July 2020).
- World Health Organization. WHO Director-General’s Opening Remarks at the Media Briefing on COVID-19—11 March. Available online: https://www.who.int/dg/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020 (accessed on 13 April 2020).
- Lurie, N.; Saville, M.; Hatchett, R.; Halton, J. Developing Covid-19 Vaccines at Pandemic Speed. N. Engl. J. Med. 2020, 382, 1969–1973. [Google Scholar] [CrossRef]
- Yang, Y.; Peng, F.; Wang, R.; Guan, K.; Jiang, T.; Xu, G.; Sun, J.; Chang, C. The deadly coronaviruses: The 2003 SARS pandemic and the 2020 novel coronavirus epidemic in China. J. Autoimmun. 2020, 109, 102434. [Google Scholar] [CrossRef]
- World Health Organization. DRAFT Landscape of COVID-19 Candidate Vaccines—14 July. Available online: https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccines (accessed on 14 July 2020).
- Nguyen, T.; Henningsen, K.H.; Brehaut, J.C.; Hoe, E.; Wilson, K. Acceptance of a pandemic influenza vaccine: A systematic review of surveys of the general public. Infect. Drug Resist. 2011, 4, 197–207. [Google Scholar] [CrossRef]
- Yaqub, O.; Castle-Clarke, S.; Sevdalis, N.; Chataway, J. Attitudes to vaccination: A critical review. Soc. Sci. Med. 2014, 112, 1–11. [Google Scholar] [CrossRef]
- Dubé, E.; MacDonald, N.E. Vaccine Acceptance: Barriers, Perceived Risks, Benefits, and Irrational Beliefs. In The Vaccine Book, 2nd ed.; Bloom, B.R., Lambert, P., Eds.; Academic Press: Cambridge, MA, USA, 2016; Chapter 26; pp. 507–528. [Google Scholar]
- Eastwood, K.; Durrheim, D.N.; Jones, A.; Butler, M. Acceptance of pandemic (H1N1) 2009 influenza vaccination by the Australian public. Med. J. Aust. 2010, 192, 33–36. [Google Scholar] [CrossRef]
- Maurer, J.; Harris, K.M.; Parker, A.M.; Lurie, N. Does receipt of seasonal influenza vaccine predict intention to receive novel H1N1 vaccine: Evidence from a nationally representative survey of U.S. adults. Vaccine 2009, 27, 5732–5734. [Google Scholar] [CrossRef][Green Version]
- Schwarzinger, M.; Flicoteaux, R.; Cortarenoda, S.; Obadia, Y.; Moatti, J.-P. Low Acceptability of A/H1N1 Pandemic Vaccination in French Adult Population: Did Public Health Policy Fuel Public Dissonance? PLoS ONE 2010, 5, e10199. [Google Scholar] [CrossRef]
- Maurer, J.; Uscher-Pines, L.; Harris, K.M. Perceived seriousness of seasonal and A(H1N1) influenzas, attitudes toward vaccination, and vaccine uptake among U.S. adults: Does the source of information matter? Prev. Med. 2010, 51, 185–187. [Google Scholar] [CrossRef]
- Sypsa, V.; Livanios, T.; Psichogiou, M.; Malliori, M.; Tsiodras, S.; Nikolakopoulos, I.; Hatzakis, A. Public perceptions in relation to intention to receive pandemic influenza vaccination in a random population sample: Evidence from a cross-sectional telephone survey. Eurosurveillance 2009, 14, 19437. [Google Scholar] [CrossRef]
- Raude, J.; Caille-Brillet, A.-L.; Setbon, M. The 2009 pandemic H1N1 influenza vaccination in France: Who accepted to receive the vaccine and why? PLoS Curr. 2010, 2, rrn1188. [Google Scholar] [CrossRef]
- Rubin, G.J.; Potts, H.; Michie, S. The impact of communications about swine flu (influenza A H1N1v) on public responses to the outbreak: Results from 36 national telephone surveys in the UK. Health Technol. Assess. 2010, 14. [Google Scholar] [CrossRef]
- Seale, H.; Heywood, A.E.; McLaws, M.; Ward, K.F.; Lowbridge, C.P.; Van, D.; MacIntyre, C.R. Why do I need it? I am not at risk! Public perceptions towards the pandemic (H1N1) 2009 vaccine. BMC Infect. Dis. 2010, 10, 99. [Google Scholar] [CrossRef]
- Lau, J.T.F.; Yeung, N.C.; Choi, K.-C.; Cheng, M.Y.; Tsui, H.; Griffiths, S. Factors in association with acceptability of A/H1N1 vaccination during the influenza A/H1N1 pandemic phase in the Hong Kong general population. Vaccine 2010, 28, 4632–4637. [Google Scholar] [CrossRef]
- Zijtregtop, E.A.M.; Wilschut, J.; Koelma, N.; Van Delden, J.J.M.; Stolk, R.P.; Van Steenbergen, J.; Broer, J.; Wolters, B.; Postma, M.; Hak, E. Which factors are important in adults’ uptake of a (pre)pandemic influenza vaccine? Vaccine 2009, 28, 207–227. [Google Scholar] [CrossRef]
- Maddux, J.E.; Rogers, R.W. Protection motivation and self-efficacy: A revised theory of fear appeals and attitude change. J. Exp. Soc. Psychol. 1983, 19, 469–479. [Google Scholar] [CrossRef]
- Rosenstock, I.M. The Health Belief Model: Explaining Health Behavior through Expectancies; Glanz, K., Lewis, F.M., Rimer, B.K., Eds.; The Jossey-Bass health series; Jossey-Bass/Wiley: Hoboken, NJ, USA, 1990; pp. 39–62. [Google Scholar]
- Determann, D.; Korfage, I.J.; Lambooij, M.S.; Bliemer, M.; Richardus, J.H.; Steyerberg, E.W.; De Bekker-Grob, E.W. Acceptance of Vaccinations in Pandemic Outbreaks: A Discrete Choice Experiment. PLoS ONE 2014, 9, e102505. [Google Scholar] [CrossRef]
- Wang, Q.; Yue, N.; Zheng, M.; Wang, D.; Duan, C.; Yu, X.; Zhang, X.; Bao, C.; Jin, H. Influenza vaccination coverage of population and the factors influencing influenza vaccination in mainland China: A meta-analysis. Vaccine 2018, 36, 7262–7269. [Google Scholar] [CrossRef]
- Bone, A.; Guthmann, J.-P.; Nicolau, J.; Levy-Brühl, D. Population and risk group uptake of H1N1 influenza vaccine in mainland France 2009–2010: Results of a national vaccination campaign. Vaccine 2010, 28, 8157–8161. [Google Scholar] [CrossRef]
- Henrich, N.; Holmes, B. The public’s acceptance of novel vaccines during a pandemic: A focus group study and its application to influenza H1N1. Emerg. Health Threat. J. 2009, 2, 7088. [Google Scholar] [CrossRef]
- World Health Organization. Report of the SAGE Working Group on Vaccine Hesitancy. Available online: https://www.who.int/immunization/sage/meetings/2014/october/1_Report_WORKING_GROUP_vaccine_hesitancy_final.pdf (accessed on 14 July 2020).
- Larson, H.J.; Jarrett, C.; Eckersberger, E.; Smith, D.M.D.; Paterson, P. Understanding vaccine hesitancy around vaccines and vaccination from a global perspective: A systematic review of published literature. Vaccine 2014, 32, 2150–2159. [Google Scholar] [CrossRef]
- Larson, H.J.; Cooper, L.Z.; Eskola, J.; Katz, S.L.; Ratzan, S. Addressing the vaccine confidence gap. Lancet 2011, 378, 526–535. [Google Scholar] [CrossRef]
- GAVI. How does COVID-19 Compare to Past Pandemics? Available online: https://www.gavi.org/vaccineswork/how-does-covid-19-compare-past-pandemics (accessed on 14 July 2020).
- World Health Organization. Q&A: Influenza and COVID-19—Similarities and Differences. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/question-and-answers-hub/q-a-detail/q-a-similarities-and-differences-covid-19-and-influenza?gclid=EAIaIQobChMIoJ657eTt6gIVzMDACh3fjQwNEAAYASAAEgLuL_D_BwE (accessed on 17 July 2020).
- Gates, B. Responding to Covid-19—A Once-in-a-Century Pandemic? N. Engl. J. Med. 2020, 382, 1677–1679. [Google Scholar] [CrossRef]
- Leung, K.; Wu, J.T.; Liu, D.; Leung, G.M. First-wave COVID-19 transmissibility and severity in China outside Hubei after control measures, and second-wave scenario planning: A modelling impact assessment. Lancet 2020, 395, 1382–1393. [Google Scholar] [CrossRef]
- Kraemer, M.U.G.; Yang, C.-H.; Gutierrez, B.; Wu, C.-H.; Klein, B.; Pigott, D.M.; Du Plessis, L.; Faria, N.R.; Li, R.; Hanage, W.P.; et al. The effect of human mobility and control measures on the COVID-19 epidemic in China. Science 2020, 368, 493–497. [Google Scholar] [CrossRef]
- Ughasoro, M.D.; Esangbedo, D.O.; Tagbo, B.N.; Mejeha, I.C. Acceptability and Willingness-to-Pay for a Hypothetical Ebola Virus Vaccine in Nigeria. PLoS Negl. Trop. Dis. 2015, 9, e0003838. [Google Scholar] [CrossRef]
- Irwin, K.L.; Jalloh, M.F.; Corker, J.; Mahmoud, B.A.; Robinson, S.J.; Li, W.; James, N.E.; Sellu, M.; Jalloh, M.B.; Diallo, A.A.; et al. Attitudes about vaccines to prevent Ebola virus disease in Guinea at the end of a large Ebola epidemic: Results of a national household survey. Vaccine 2017, 35, 6915–6923. [Google Scholar] [CrossRef]
- Tian, H.; Liu, Y.; Li, Y.; Wu, C.-H.; Chen, B.; Kraemer, M.U.G.; Li, B.; Cai, J.; Xu, B.; Yang, Q.; et al. An investigation of transmission control measures during the first 50 days of the COVID-19 epidemic in China. Science 2020, 368, 638–642. [Google Scholar] [CrossRef]
- Rosenstock, I.M. The Health Belief Model and Preventive Health Behavior. Health Educ. Monogr. 1974, 2, 354–386. [Google Scholar] [CrossRef]
- Cheney, M.K.; John, R. Underutilization of Influenza Vaccine. SAGE Open 2013, 3, 2000242823. [Google Scholar] [CrossRef]
- Kwok, K.O.; Lai, F.; Wei, W.I.; Wong, S.Y.S.; Tang, J.W.T. Herd immunity—Estimating the level required to halt the COVID-19 epidemics in affected countries. J. Infect. 2020, 80, e32–e33. [Google Scholar] [CrossRef]
- Xinhua Net. Update: China to Make COVID-19 Vaccine Global Public Good: Official. Available online: http://www.xinhuanet.com/english/2020-06/07/c_139121625.htm (accessed on 17 July 2020).
| Items | Respondents (n = 2058) N (%) |
|---|---|
| Age group | |
| 18–25 | 475 (23.1) |
| 26–30 | 400 (19.4) |
| 31–40 | 523 (25.4) |
| 41–50 | 510 (24.8) |
| 51 and above | 150 (7.3) |
| Gender | |
| Female | 1115 (54.2) |
| Male | 943 (45.8) |
| Highest level of education | |
| Middle school and below | 123 (6.0) |
| High school | 663 (32.2) |
| Associate or bachelor | 1140 (55.4) |
| Master and above | 132 (6.4) |
| Marriage status | |
| Married | 1385 (67.3) |
| Others (single, divorced or widowed) | 673 (32.7) |
| Location | |
| Central | 531 (25.8) |
| East | 1195 (58.1) |
| West | 332 (16.1) |
| Region | |
| Rural | 420 (20.4) |
| Urban | 1638 (79.6) |
| Employment status | |
| Employed | 1651 (80.2) |
| Unemployed | 407 (19.8) |
| Health status | |
| Good and above (good, very good) | 1527 (74.2) |
| Fair or below (fair, poor, very poor) | 531 (25.8) |
| Total family income in 2019 | |
| ≤CNY 50,000 (USD 7246) | 277 (13.4) |
| CNY 50,000–100,000 (USD 7246–14,492) | 548 (26.6) |
| CNY 100,000–150,000 (USD 14,492–21,739) | 506 (24.6) |
| CNY 150,000–200,000 (USD 21,739–28,986) | 352 (17.1) |
| CNY 200,000–300,000 (USD 28,986–43,478) | 239 (11.7) |
| CNY 300,000 (USD 43,478) | 136 (6.6) |
| There are confirmed or suspected cases in the county | |
| Yes | 1538 (74.7) |
| No or not clear | 520 (25.3) |
| Perceived risk of infection | |
| High or very high | 251 (12.2) |
| Fair | 575 (27.9) |
| Low or very low | 1232 (59.9) |
| Pandemic impact on daily life | |
| Large or very large | 1368 (66.5) |
| Fair | 497 (24.1) |
| Small or very small | 193 (9.4) |
| Pandemic impact on work | |
| Large or very large | 1326 (64.4) |
| Fair | 402 (19.5) |
| Small or very small | 191 (9.3) |
| Missing | 139 (6.8) |
| Pandemic impact on income | |
| Large or very large | 905 (44.0) |
| Fair | 467 (22.7) |
| Small or very small | 325 (15.8) |
| Missing | 361 (17.5) |
| Received vaccination against influenza in the past season | |
| Yes | 301 (14.6) |
| No | 1757 (85.4) |
| Refused vaccination of a certain type of vaccine in the past | |
| Yes | 459 (22.3) |
| No | 1599 (77.7) |
| Items | N (%) |
|---|---|
| Overall respondents (n = 2058) | |
| COVID-19 vaccination is an effective way to prevent and control COVID-19 | |
| Yes | 1842 (89.5) |
| No | 216 (10.5) |
| Accept vaccination if the COVID-19 vaccine is successfully developed and approved for listing in the future | |
| Yes | 1879 (91.3) |
| No | 179 (8.7) |
| Doctor’s recommendation is an important factor in vaccination decision-making | |
| Yes | 1659 (80.6) |
| No | 399 (19.4) |
| Vaccine convenience (vaccination method, frequency, distance to vaccination sites, etc.) is an important factor in vaccination decision-making | |
| Yes | 1558 (75.7) |
| No | 500 (23.3) |
| Vaccine price is an important factor in vaccination decision-making | |
| Yes | 1233 (59.9) |
| No | 825 (40.1) |
| Vaccine accept group (N = 1879) | |
| Want to receive vaccination as soon as possible when the vaccine is available | |
| Yes, as soon as possible | 980 (52.2) |
| No, delay vaccination until I confirmed the vaccine safety | 899 (47.8) |
| Prefer which kind of immunization schedules of the COVID-19 vaccination | |
| Routine immunization | 928 (49.4) |
| Emergency vaccination | 169 (9.0) |
| Both are acceptable | 782 (41.6) |
| Prefer which type of COVID-19 vaccines | |
| Domestic vaccine | 611 (32.5) |
| Imported vaccine | 62 (3.3) |
| Both are acceptable | 1206 (64.2) |
| Characteristics | OR | SE | p-Value | 95% CI |
|---|---|---|---|---|
| Age group | ||||
| 18–25 | Ref | |||
| 26–30 | 1.15 | 0.23 | 0.48 | 0.78–1.70 |
| 31–40 | 1.00 | 0.21 | 0.99 | 0.66–1.52 |
| 41–50 | 1.05 | 0.22 | 0.81 | 0.69–1.60 |
| >51 | 1.65 | 0.48 | 0.09 | 0.93–2.92 |
| Gender | ||||
| Female | Ref | |||
| Male | 1.25 | 0.13 | 0.03 | 1.03–1.52 |
| Highest level of education | ||||
| Middle school and below | Ref | |||
| High school | 1.11 | 0.26 | 0.67 | 0.70–1.75 |
| Associate or Bachelor | 1.27 | 0.31 | 0.33 | 0.79–2.04 |
| Master and above | 1.09 | 0.34 | 0.79 | 0.59–2.00 |
| Marriage status | ||||
| Others (single, divorced or widowed) | Ref | |||
| Married | 1.70 | 0.26 | <0.001 | 1.26–2.29 |
| Location | ||||
| Central | Ref | |||
| East | 0.88 | 0.11 | 0.29 | 0.69–1.11 |
| West | 1.03 | 0.16 | 0.84 | 0.76–1.40 |
| Region | ||||
| Rural | Ref | |||
| Urban | 0.84 | 0.11 | 0.18 | 0.65–1.09 |
| Employment status | ||||
| Unemployed | Ref | |||
| Employed | 1.03 | 0.51 | 0.96 | 0.39–2.7 |
| Health status | ||||
| Fair or below (fair, poor, very poor) | Ref | |||
| Good and above (good, very good) | 1.16 | 0.14 | 0.21 | 0.92–1.46 |
| Total family income in 2019 | ||||
| ≤CNY 50,000 (USD 7246) | Ref | |||
| CNY 50,000–100,000 (USD 7246–14,492) | 0.84 | 0.15 | 0.32 | 0.60–1.18 |
| CNY 100,000–150,000 (USD 14,492–21,739) | 0.73 | 0.13 | 0.08 | 0.51–1.04 |
| CNY 150,000–200,000 (USD 21,739–28,986) | 0.82 | 0.16 | 0.32 | 0.56–1.21 |
| CNY 200,000–300,000 (USD 28,986–43,478) | 0.82 | 0.18 | 0.38 | 0.53–1.27 |
| ≥CNY 300,000 (USD 43,478) | 1.05 | 0.27 | 0.85 | 0.64–1.73 |
| There are confirmed or suspected cases in the county | ||||
| No or not clear | Ref | |||
| Yes | 0.72 | 0.09 | 0.01 | 0.57–0.91 |
| Perceived risk of infection | ||||
| Fair | Ref | |||
| High or very high | 1.46 | 0.25 | 0.03 | 1.04–2.05 |
| Small or very small | 1.02 | 0.12 | 0.89 | 0.81–1.27 |
| Pandemic impact on daily life | ||||
| Fair | Ref | |||
| Large or very large | 1.02 | 0.13 | 0.90 | 0.80–1.30 |
| Small or very small | 0.69 | 0.14 | 0.07 | 0.46–1.04 |
| Pandemic impact on work | ||||
| Fair | Ref | |||
| Large or very large | 1.03 | 0.15 | 0.82 | 0.78–1.36 |
| Small or very small | 0.88 | 0.19 | 0.55 | 0.57–1.34 |
| Pandemic impact on income | ||||
| Fair | Ref | |||
| Large or very large | 0.96 | 0.13 | 0.74 | 0.74–1.24 |
| Small or very small | 0.81 | 0.14 | 0.20 | 0.58–1.12 |
| Received vaccination against influenza in the past season | ||||
| No | Ref | |||
| Yes | 1.90 | 0.27 | <0.001 | 1.43–2.51 |
| Refused vaccination of a certain type of vaccine in the past | ||||
| No | Ref | |||
| Yes | 0.81 | 0.10 | 0.09 | 0.64–1.03 |
| COVID-19 vaccination is an effective way to prevent and control COVID-19 | ||||
| No | Ref | |||
| Yes | 1.56 | 0.29 | 0.02 | 1.08–2.25 |
| Doctor’s recommendation is an important factor in vaccination decision-making | ||||
| No | Ref | |||
| Yes | 2.32 | 0.33 | <0.001 | 1.76–3.07 |
| Vaccine convenience (vaccination method, frequency, distance to vaccination sites, etc.) is an important factor in vaccination decision-making | ||||
| No | Ref | |||
| Yes | 0.69 | 0.09 | <0.001 | 0.54–0.89 |
| Vaccine price is an important factor in vaccination decision-making | ||||
| No | Ref | |||
| Yes | 0.75 | 0.08 | 0.01 | 0.61–0.93 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, J.; Jing, R.; Lai, X.; Zhang, H.; Lyu, Y.; Knoll, M.D.; Fang, H. Acceptance of COVID-19 Vaccination during the COVID-19 Pandemic in China. Vaccines 2020, 8, 482. https://doi.org/10.3390/vaccines8030482
Wang J, Jing R, Lai X, Zhang H, Lyu Y, Knoll MD, Fang H. Acceptance of COVID-19 Vaccination during the COVID-19 Pandemic in China. Vaccines. 2020; 8(3):482. https://doi.org/10.3390/vaccines8030482
Chicago/Turabian StyleWang, Jiahao, Rize Jing, Xiaozhen Lai, Haijun Zhang, Yun Lyu, Maria Deloria Knoll, and Hai Fang. 2020. "Acceptance of COVID-19 Vaccination during the COVID-19 Pandemic in China" Vaccines 8, no. 3: 482. https://doi.org/10.3390/vaccines8030482
APA StyleWang, J., Jing, R., Lai, X., Zhang, H., Lyu, Y., Knoll, M. D., & Fang, H. (2020). Acceptance of COVID-19 Vaccination during the COVID-19 Pandemic in China. Vaccines, 8(3), 482. https://doi.org/10.3390/vaccines8030482





