Neurological Disorders following COVID-19 Vaccination
Abstract
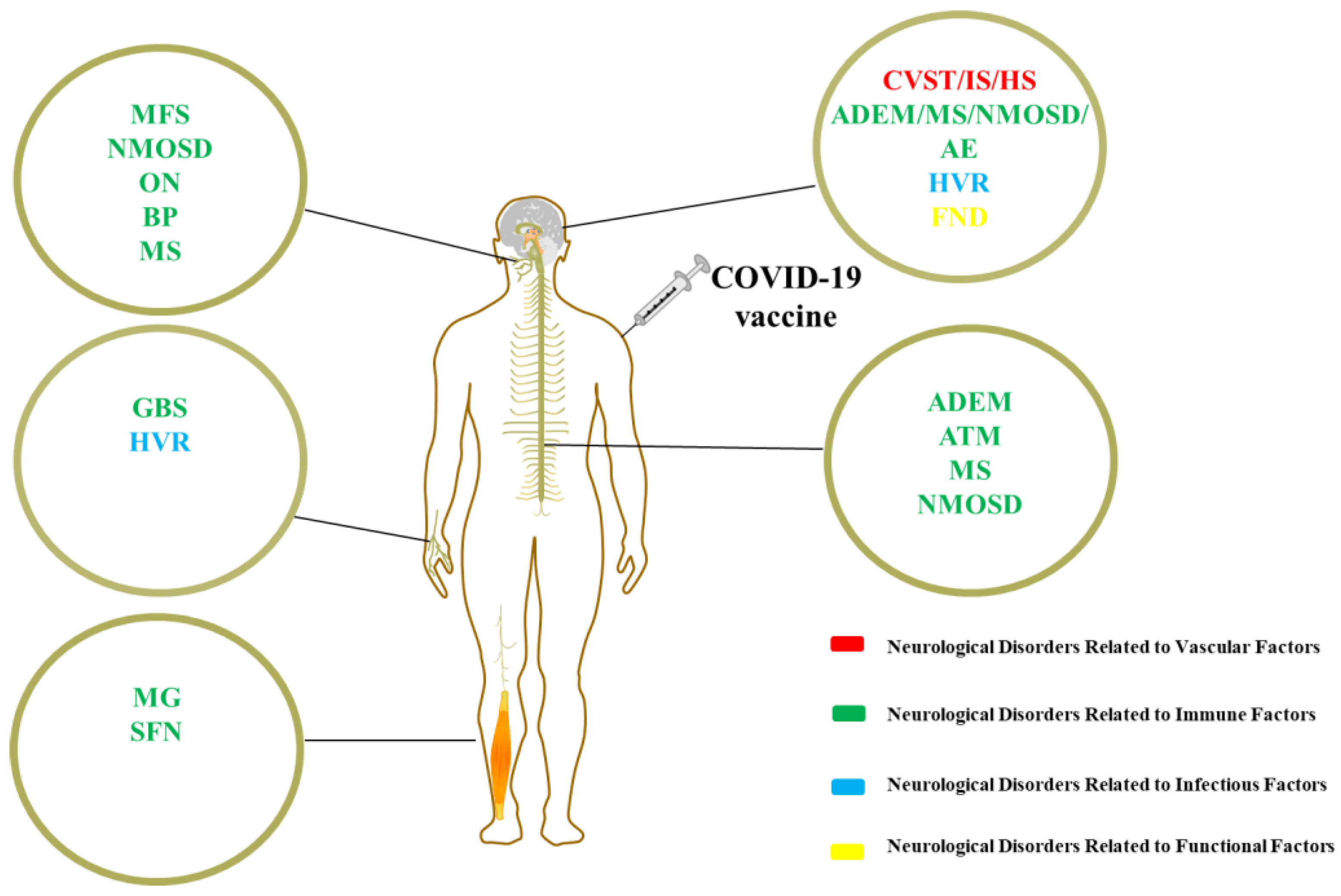
1. Introduction
2. Discussion
2.1. Neurological Disorders Related to Vascular Factors
2.1.1. Cranial Venous Sinus Thrombosis
2.1.2. Ischemic Stroke
2.1.3. Hemorrhagic Stroke
2.2. Neurological Disorders Related to Immune Factors
2.2.1. Bell’s Palsy
2.2.2. Guillain–Barre Syndrome and Miller–Fisher Syndrome
2.2.3. Myasthenia Gravis
2.2.4. Demyelinating Diseases
Optic Neuritis
Acute Transverse Myelitis
Acute Disseminated Encephalomyelitis
Multiple Sclerosis
Neuromyelitis Optica Spectrum Disorder
2.2.5. Autoimmune Encephalitis
2.2.6. Small Fiber Neuropathy
2.3. Neurological Disorders Related to Infectious Factors
2.4. Neurological Disorders Related to Functional Factors
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO Coronavirus (COVID-19) Dashboard. Available online: https://covid19.who.int/ (accessed on 17 May 2023).
- Mendez-Lizarraga, C.A.; Chacon-Cruz, E.; Carrillo-Meza, R.; Hernández-Milán, N.S.; Inustroza-Sánchez, L.C.; Ovalle-Marroquín, D.F.; Machado-Contreras, J.R.; Ceballos Zuñiga, O.; Bejarano-Ramírez, V.; Aguilar-Aguayo, C.; et al. Report of Adverse Effects Following Population-Wide COVID-19 Vaccination: A Comparative Study between Six Different Vaccines in Baja-California, Mexico. Vaccines 2022, 10, 1196. [Google Scholar] [CrossRef] [PubMed]
- Mohseni Afshar, Z.; Sharma, A.; Babazadeh, A.; Alizadeh-Khatir, A.; Sio, T.T.; Taghizadeh Moghadam, M.A.; Tavakolli Pirzaman, A.; Mojadad, A.; Hosseinzadeh, R.; Barary, M.; et al. A review of the potential neurological adverse events of COVID-19 vaccines. Acta Neurol. Belg. 2023, 123, 9–44. [Google Scholar] [CrossRef] [PubMed]
- Allahyari, F.; Molaee, H.; Hosseini Nejad, J. COVID-19 vaccines and neurological complications: A systematic review. Z. Naturforschung. C J. Biosci. 2023, 78, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Azorin, D.; Baykan, B.; Beghi, E.; Doheim, M.F.; Fernandez-de-Las-Penas, C.; Gezegen, H.; Guekht, A.; Hoo, F.K.; Santacatterina, M.; Sejvar, J.; et al. Timing of headache after COVID-19 vaccines and its association with cerebrovascular events: An analysis of 41,700 VAERS reports. Cephalalgia Int. J. Headache 2022, 42, 1207–1217. [Google Scholar] [CrossRef]
- Iba, T.; Levy, J.H.; Warkentin, T.E. Recognizing Vaccine-Induced Immune Thrombotic Thrombocytopenia. Crit. Care Med. 2022, 50, e80–e86. [Google Scholar] [CrossRef] [PubMed]
- Palaiodimou, L.; Stefanou, M.I.; de Sousa, D.A.; Coutinho, J.M.; Papadopoulou, M.; Papaevangelou, V.; Vassilakopoulos, T.I.; Tsiodras, S.; Filippou, D.K.; Tsivgoulis, G. Cerebral venous sinus thrombosis in the setting of COVID-19 vaccination: A systematic review and meta-analysis. J. Neurol. 2022, 269, 3413–3419. [Google Scholar] [CrossRef]
- Krzywicka, K.; van de Munckhof, A.; Sánchez van Kammen, M.; Heldner, M.R.; Jood, K.; Lindgren, E.; Tatlisumak, T.; Putaala, J.; Kremer Hovinga, J.A.; Middeldorp, S.; et al. Age-Stratified Risk of Cerebral Venous Sinus Thrombosis After SARS-CoV-2 Vaccination. Neurology 2022, 98, e759–e768. [Google Scholar] [CrossRef]
- Fervers, P.; Kottlors, J.; Persigehl, T.; Lennartz, S.; Maus, V.; Fischer, S.; Styczen, H.; Deuschl, C.; Schlamann, M.; Mpotsaris, A.; et al. Meaningful use of imaging resources to rule out cerebral venous sinus thrombosis after ChAdOx1 COVID-19 vaccination: Evaluation of the AHA diagnostic algorithm with a clinical cohort and a systematic data review. J. Clin. Neurosci. Off. J. Neurosurg. Soc. Australas. 2022, 102, 5–12. [Google Scholar] [CrossRef]
- Sánchez van Kammen, M.; Aguiar de Sousa, D.; Poli, S.; Cordonnier, C.; Heldner, M.R.; van de Munckhof, A.; Krzywicka, K.; van Haaps, T.; Ciccone, A.; Middeldorp, S.; et al. Characteristics and Outcomes of Patients With Cerebral Venous Sinus Thrombosis in SARS-CoV-2 Vaccine-Induced Immune Thrombotic Thrombocytopenia. JAMA Neurol. 2021, 78, 1314–1323. [Google Scholar] [CrossRef]
- Wittstock, M.; Walter, U.; Volmer, E.; Storch, A.; Weber, M.A.; Großmann, A. Cerebral venous sinus thrombosis after adenovirus-vectored COVID-19 vaccination: Review of the neurological-neuroradiological procedure. Neuroradiology 2022, 64, 865–874. [Google Scholar] [CrossRef]
- Islam, A.; Bashir, M.S.; Joyce, K.; Rashid, H.; Laher, I.; Elshazly, S. An Update on COVID-19 Vaccine Induced Thrombotic Thrombocytopenia Syndrome and Some Management Recommendations. Molecules 2021, 26, 5004. [Google Scholar] [CrossRef] [PubMed]
- Sharifian-Dorche, M.; Bahmanyar, M.; Sharifian-Dorche, A.; Mohammadi, P.; Nomovi, M.; Mowla, A. Vaccine-induced immune thrombotic thrombocytopenia and cerebral venous sinus thrombosis post COVID-19 vaccination; a systematic review. J. Neurol. Sci. 2021, 428, 117607. [Google Scholar] [CrossRef] [PubMed]
- Koh, J.S.; Hoe, R.H.M.; Yong, M.H.; Chiew, H.J.; Goh, Y.; Yong, K.P.; Tu, T.M.; Chan, D.W.S.; Tan, B.Y.; Yeo, L.L.L.; et al. Hospital-based observational study of neurological disorders in patients recently vaccinated with COVID-19 mRNA vaccines. J. Neurol. Sci. 2021, 430, 120030. [Google Scholar] [CrossRef] [PubMed]
- Stefanou, M.I.; Palaiodimou, L.; Aguiar de Sousa, D.; Theodorou, A.; Bakola, E.; Katsaros, D.E.; Halvatsiotis, P.; Tzavellas, E.; Naska, A.; Coutinho, J.M.; et al. Acute Arterial Ischemic Stroke Following COVID-19 Vaccination: A Systematic Review and Meta-analysis. Neurology 2022, 99, e1465–e1474. [Google Scholar] [CrossRef] [PubMed]
- López-Mena, D.; García-Grimshaw, M.; Saldivar-Dávila, S.; Hernandez-Vanegas, L.E.; Saniger-Alba, M.D.M.; Gutiérrez-Romero, A.; Carrillo-Mezo, R.; Valdez-Ruvalcaba, H.E.; Cano-Nigenda, V.; Flores-Silva, F.D.; et al. Stroke Among SARS-CoV-2 Vaccine Recipients in Mexico: A Nationwide Descriptive Study. Neurology 2022, 98, e1933–e1941. [Google Scholar] [CrossRef] [PubMed]
- Klein, N.P.; Lewis, N.; Goddard, K.; Fireman, B.; Zerbo, O.; Hanson, K.E.; Donahue, J.G.; Kharbanda, E.O.; Naleway, A.; Nelson, J.C.; et al. Surveillance for Adverse Events After COVID-19 mRNA Vaccination. JAMA 2021, 326, 1390–1399. [Google Scholar] [CrossRef]
- Botton, J.; Jabagi, M.J.; Bertrand, M.; Baricault, B.; Drouin, J.; Le Vu, S.; Weill, A.; Farrington, P.; Zureik, M.; Dray-Spira, R. Risk for Myocardial Infarction, Stroke, and Pulmonary Embolism Following COVID-19 Vaccines in Adults Younger Than 75 Years in France. Ann. Intern. Med. 2022, 175, 1250–1257. [Google Scholar] [CrossRef]
- Hippisley-Cox, J.; Patone, M.; Mei, X.W.; Saatci, D.; Dixon, S.; Khunti, K.; Zaccardi, F.; Watkinson, P.; Shankar-Hari, M.; Doidge, J.; et al. Risk of thrombocytopenia and thromboembolism after COVID-19 vaccination and SARS-CoV-2 positive testing: Self-controlled case series study. BMJ 2021, 374, n1931. [Google Scholar] [CrossRef]
- Gorenflo, M.P.; Davis, P.B.; Kaelber, D.C.; Xu, R. Ischemic stroke after COVID-19 bivalent vaccine administration in patients aged 65 years and older: Analysis of nation-wide patient electronic health records in the United States. medRxiv 2023. [Google Scholar] [CrossRef]
- Ye, X.; Huang, C.; Yan, V.K.C.; Kang, W.; Fan, M.; Tsang, G.K.C.; Ho, C.M.Y.; Lip, G.Y.H.; Yiu, K.H.; Tse, H.F.; et al. Sex-based differences in risk of ischemic stroke or systemic embolism after BNT162b2 or CoronaVac COVID-19 vaccination in patients with atrial fibrillation: A self-controlled case series and nested case-control study. Eur. Heart J. Cardiovasc. Pharmacother. 2023; in press. [Google Scholar] [CrossRef]
- Kim, H.J.; Lee, S.J.; Sa, S.; Bae, J.H.; Song, G.; Lee, C.W.; Kim, J.H.; Shim, S.R.; Hong, M.; Han, H.W. Safety of COVID-19 Vaccines among Patients with Type 2 Diabetes Mellitus: Real-World Data Analysis. Diabetes Metab. J. 2023, 47, 356–365. [Google Scholar] [CrossRef]
- Stamenković, M.; Radmilo, L.; Jovićević, M.; Rabi-Žikić, T.; Žarkov, M.; Ružička-Kaloci, S.; Simić, S.; Stamenković, A.; Dangić, J.; Knezović, G.; et al. Clinical Characteristics of Patients with Acute Ischemic Stroke Previously Vaccinated Against COVID-19. J. Stroke Cerebrovasc. Dis. Off. J. Natl. Stroke Assoc. 2022, 31, 106483. [Google Scholar] [CrossRef] [PubMed]
- Kolahchi, Z.; Khanmirzaei, M.; Mowla, A. Acute ischemic stroke and vaccine-induced immune thrombotic thrombocytopenia post COVID-19 vaccination; a systematic review. J. Neurol. Sci. 2022, 439, 120327. [Google Scholar] [CrossRef] [PubMed]
- Cascio Rizzo, A.; Giussani, G.; Agostoni, E.C. Ischemic Stroke and Vaccine-Induced Immune Thrombotic Thrombocytopenia following COVID-19 Vaccine: A Case Report with Systematic Review of the Literature. Cerebrovasc. Dis. 2022, 51, 722–734. [Google Scholar] [CrossRef] [PubMed]
- Powers, W.J.; Rabinstein, A.A.; Ackerson, T.; Adeoye, O.M.; Bambakidis, N.C.; Becker, K.; Biller, J.; Brown, M.; Demaerschalk, B.M.; Hoh, B.; et al. 2018 Guidelines for the Early Management of Patients With Acute Ischemic Stroke: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke 2018, 49, e46–e110. [Google Scholar] [CrossRef]
- Mowla, A.; Razavi, S.M.; Lail, N.S.; Mohammadi, P.; Shirani, P.; Kavak, K.S.; Sawyer, R.N.; Kamal, H. Hyperdense middle cerebral artery sign and response to combination of mechanical Thrombectomy plus intravenous thrombolysis in acute stroke patients. J. Neurol. Sci. 2021, 429, 117618. [Google Scholar] [CrossRef] [PubMed]
- Kenda, J.; Lovrič, D.; Škerget, M.; Milivojević, N. Treatment of ChAdOx1 nCoV-19 Vaccine-Induced Immune Thrombotic Thrombocytopenia Related Acute Ischemic Stroke. J. Stroke Cerebrovasc. Dis. Off. J. Natl. Stroke Assoc. 2021, 30, 106072. [Google Scholar] [CrossRef]
- Patone, M.; Handunnetthi, L.; Saatci, D.; Pan, J.; Katikireddi, S.V.; Razvi, S.; Hunt, D.; Mei, X.W.; Dixon, S.; Zaccardi, F.; et al. Neurological complications after first dose of COVID-19 vaccines and SARS-CoV-2 infection. Nat. Med. 2021, 27, 2144–2153. [Google Scholar] [CrossRef]
- Moll, K.; Lufkin, B.; Fingar, K.R.; Ke Zhou, C.; Tworkoski, E.; Shi, C.; Hobbi, S.; Hu, M.; Sheng, M.; McCarty, J.; et al. Background rates of adverse events of special interest for COVID-19 vaccine safety monitoring in the United States, 2019–2020. Vaccine 2023, 41, 333–353. [Google Scholar] [CrossRef]
- Chui, C.S.L.; Fan, M.; Wan, E.Y.F.; Leung, M.T.Y.; Cheung, E.; Yan, V.K.C.; Gao, L.; Ghebremichael-Weldeselassie, Y.; Man, K.K.C.; Lau, K.K.; et al. Thromboembolic events and hemorrhagic stroke after mRNA (BNT162b2) and inactivated (CoronaVac) COVID-19 vaccination: A self-controlled case series study. EClinicalMedicine 2022, 50, 101504. [Google Scholar] [CrossRef]
- Lazcano, U.; Cuadrado-Godia, E.; Grau, M.; Subirana, I.; Martínez-Carbonell, E.; Boher-Massaguer, M.; Rodríguez-Campello, A.; Giralt-Steinhauer, E.; Fernández-Pérez, I.; Jiménez-Conde, J.; et al. Increased COVID-19 Mortality in People With Previous Cerebrovascular Disease: A Population-Based Cohort Study. Stroke 2022, 53, 1276–1284. [Google Scholar] [CrossRef]
- Nachamkin, I.; Shadomy, S.V.; Moran, A.P.; Cox, N.; Fitzgerald, C.; Ung, H.; Corcoran, A.T.; Iskander, J.K.; Schonberger, L.B.; Chen, R.T. Anti-ganglioside antibody induction by swine (A/NJ/1976/H1N1) and other influenza vaccines: Insights into vaccine-associated Guillain-Barré syndrome. J. Infect. Dis. 2008, 198, 226–233. [Google Scholar] [CrossRef] [PubMed]
- Keddie, S.; Pakpoor, J.; Mousele, C.; Pipis, M.; Machado, P.M.; Foster, M.; Record, C.J.; Keh, R.Y.S.; Fehmi, J.; Paterson, R.W.; et al. Epidemiological and cohort study finds no association between COVID-19 and Guillain-Barré syndrome. Brain A J. Neurol. 2021, 144, 682–693. [Google Scholar] [CrossRef] [PubMed]
- Messina, N.L.; Zimmermann, P.; Curtis, N. The impact of vaccines on heterologous adaptive immunity. Clin. Microbiol. Infect. 2019, 25, 1484–1493. [Google Scholar] [CrossRef] [PubMed]
- van Aalst, S.; Ludwig, I.S.; van der Zee, R.; van Eden, W.; Broere, F. Bystander activation of irrelevant CD4+ T cells following antigen-specific vaccination occurs in the presence and absence of adjuvant. PLoS ONE 2017, 12, e0177365. [Google Scholar] [CrossRef]
- Wraith, D.C.; Goldman, M.; Lambert, P.H. Vaccination and autoimmune disease: What is the evidence? Lancet 2003, 362, 1659–1666. [Google Scholar] [CrossRef]
- Teijaro, J.R.; Farber, D.L. COVID-19 vaccines: Modes of immune activation and future challenges. Nat. Rev. Immunol. 2021, 21, 195–197. [Google Scholar] [CrossRef]
- Soeiro, T.; Salvo, F.; Pariente, A.; Grandvuillemin, A.; Jonville-Béra, A.P.; Micallef, J. Type I interferons as the potential mechanism linking mRNA COVID-19 vaccines to Bell’s palsy. Therapie 2021, 76, 365–367. [Google Scholar] [CrossRef]
- Alonso Castillo, R.; Martínez Castrillo, J.C. Neurological manifestations associated with COVID-19 vaccine. Neurologia, 2022; in press. [Google Scholar] [CrossRef]
- Wan, E.Y.F.; Chui, C.S.L.; Lai, F.T.T.; Chan, E.W.Y.; Li, X.; Yan, V.K.C.; Gao, L.; Yu, Q.; Lam, I.C.H.; Chun, R.K.C.; et al. Bell’s palsy following vaccination with mRNA (BNT162b2) and inactivated (CoronaVac) SARS-CoV-2 vaccines: A case series and nested case-control study. Lancet Infect. Dis. 2022, 22, 64–72. [Google Scholar] [CrossRef]
- Ozonoff, A.; Nanishi, E.; Levy, O. Bell’s palsy and SARS-CoV-2 vaccines. Lancet Infect. Dis. 2021, 21, 450–452. [Google Scholar] [CrossRef]
- Bertin, B.; Grenet, G.; Pizzoglio-Billaudaz, V.; Lepelley, M.; Atzenhoffer, M.; Vial, T. Vaccines and Bell’s palsy: A narrative review. Therapie 2022, 78, 279–292. [Google Scholar] [CrossRef] [PubMed]
- Khurshid, M.; Ansari, I.; Ahmad, H.; Ghaffar, H.; Khurshid, A.; Shahid, A.; Essar, M.Y.; Ullah, I.; Cheema, H.A. Development of facial palsy following COVID-19 vaccination: A systematic review. Ann. Med. Surg. 2022, 82, 104758. [Google Scholar] [CrossRef] [PubMed]
- Shibli, R.; Barnett, O.; Abu-Full, Z.; Gronich, N.; Najjar-Debbiny, R.; Doweck, I.; Rennert, G.; Saliba, W. Association between vaccination with the BNT162b2 mRNA COVID-19 vaccine and Bell’s palsy: A population-based study. Lancet Reg. Health. Eur. 2021, 11, 100236. [Google Scholar] [CrossRef] [PubMed]
- Shahsavarinia, K.; Mahmoodpoor, A.; Sadeghi-Ghyassi, F.; Nedayi, A.; Razzaghi, A.; Zehi Saadat, M.; Salehi-Pourmehr, H. Bell’s Palsy and COVID-19 Vaccination: A Systematic Review. Med. J. Islam. Repub. Iran 2022, 36, 85. [Google Scholar] [CrossRef]
- Shemer, A.; Pras, E.; Hecht, I. Peripheral Facial Nerve Palsy Following BNT162b2 (COVID-19) Vaccination. Isr. Med. Assoc. J. IMAJ 2021, 23, 143–144. [Google Scholar]
- Renoud, L.; Khouri, C.; Revol, B.; Lepelley, M.; Perez, J.; Roustit, M.; Cracowski, J.L. Association of Facial Paralysis With mRNA COVID-19 Vaccines: A Disproportionality Analysis Using the World Health Organization Pharmacovigilance Database. JAMA Intern. Med. 2021, 181, 1243–1245. [Google Scholar] [CrossRef]
- Albakri, K.; Khaity, A.; Atwan, H.; Saleh, O.; Al-Hajali, M.; Cadri, S.; Diab, R.A.; Albazee, E.; Negida, A. Bell’s Palsy and COVID-19 Vaccines: A Systematic Review and Meta-Analysis. Vaccines 2023, 11, 236. [Google Scholar] [CrossRef]
- Sato, K.; Mano, T.; Niimi, Y.; Toda, T.; Iwata, A.; Iwatsubo, T. Facial nerve palsy following the administration of COVID-19 mRNA vaccines: Analysis of a self-reporting database. Int. J. Infect. Dis. 2021, 111, 310–312. [Google Scholar] [CrossRef]
- Shemer, A.; Pras, E.; Einan-Lifshitz, A.; Dubinsky-Pertzov, B.; Hecht, I. Association of COVID-19 Vaccination and Facial Nerve Palsy: A Case-Control Study. JAMA Otolaryngol.-Head Neck Surg. 2021, 147, 739–743. [Google Scholar] [CrossRef]
- Alkhalifah, J.M.; Al Seraihi, A.; Al-Tawfiq, J.A.; Alshehri, B.F.; Alhaluli, A.H.; Alsulais, N.M.; Alessa, M.M.; Seddiq, W.; Aljeri, T.; Qahtani, M.H.; et al. Pattern of self-reported adverse events related to COVID-19 vaccines in Saudi Arabia: A nationwide study. Front. Public Health 2023, 11, 1043696. [Google Scholar] [CrossRef]
- Lahoz Fernandez, P.E.; Miranda Pereira, J.; Fonseca Risso, I.; Baleeiro Rodrigues Silva, P.; Freitas Barboza, I.C.; Vieira Silveira, C.G.; Diogo Silva, G.; Marzorati Kuntz Puglia, P.; Genaro Mutarelli, E. Guillain-Barre syndrome following COVID-19 vaccines: A scoping review. Acta Neurol. Scand. 2022, 145, 393–398. [Google Scholar] [CrossRef] [PubMed]
- Abolmaali, M.; Rezania, F.; Behnagh, A.K.; Hamidabad, N.M.; Gorji, A.; Mirzaasgari, Z. Guillain-Barré syndrome in association with COVID-19 vaccination: A systematic review. Immunol. Res. 2022, 70, 752–764. [Google Scholar] [CrossRef] [PubMed]
- Finsterer, J.; Scorza, F.A.; Scorza, C.A. Post SARS-CoV-2 vaccination Guillain-Barre syndrome in 19 patients. Clinics 2021, 76, e3286. [Google Scholar] [CrossRef] [PubMed]
- García-Grimshaw, M.; Michel-Chávez, A.; Vera-Zertuche, J.M.; Galnares-Olalde, J.A.; Hernández-Vanegas, L.E.; Figueroa-Cucurachi, M.; Paredes-Ceballos, O.; Reyes-Terán, G.; Carbajal-Sandoval, G.; Ceballos-Liceaga, S.E.; et al. Guillain-Barré syndrome is infrequent among recipients of the BNT162b2 mRNA COVID-19 vaccine. Clin. Immunol. 2021, 230, 108818. [Google Scholar] [CrossRef]
- Tondo, G.; Virgilio, E.; Naldi, A.; Bianchi, A.; Comi, C. Safety of COVID-19 Vaccines: Spotlight on Neurological Complications. Life 2022, 12, 1338. [Google Scholar] [CrossRef]
- Germano, F.; Bellucci, M.; Grisanti, S.; Beronio, A.; Grazzini, M.; Coco, E.; Tassinari, T.; Della Cava, F.; De Michelis, C.; Baldi, O.; et al. COVID-19 vaccine-related Guillain-Barré syndrome in the Liguria region of Italy: A multicenter case series. J. Neurol. Sci. 2022, 440, 120330. [Google Scholar] [CrossRef]
- Ha, J.; Park, S.; Kang, H.; Kyung, T.; Kim, N.; Kim, D.K.; Kim, H.; Bae, K.; Song, M.C.; Lee, K.J.; et al. Real-world data on the incidence and risk of Guillain-Barré syndrome following SARS-CoV-2 vaccination: A prospective surveillance study. Sci. Rep. 2023, 13, 3773. [Google Scholar] [CrossRef]
- Yu, M.; Nie, S.; Qiao, Y.; Ma, Y. Guillain-Barre syndrome following COVID-19 vaccines: A review of literature. Front. Immunol. 2023, 14, 1078197. [Google Scholar] [CrossRef]
- Li, X.; Ostropolets, A.; Makadia, R.; Shoaibi, A.; Rao, G.; Sena, A.G.; Martinez-Hernandez, E.; Delmestri, A.; Verhamme, K.; Rijnbeek, P.R.; et al. Characterising the background incidence rates of adverse events of special interest for COVID-19 vaccines in eight countries: Multinational network cohort study. BMJ 2021, 373, n1435. [Google Scholar] [CrossRef]
- Hanson, K.E.; Goddard, K.; Lewis, N.; Fireman, B.; Myers, T.R.; Bakshi, N.; Weintraub, E.; Donahue, J.G.; Nelson, J.C.; Xu, S.; et al. Incidence of Guillain-Barré Syndrome After COVID-19 Vaccination in the Vaccine Safety Datalink. JAMA Netw. Open 2022, 5, e228879. [Google Scholar] [CrossRef]
- Atzenhoffer, M.; Auffret, M.; Pegat, A.; Masmoudi, K.; Khouri, C.; Bertin, B.; Vial, T. Guillain-Barré Syndrome Associated with COVID-19 Vaccines: A Perspective From Spontaneous Report Data. Clin. Drug Investig. 2022, 42, 581–592. [Google Scholar] [CrossRef] [PubMed]
- Shao, S.C.; Wang, C.H.; Chang, K.C.; Hung, M.J.; Chen, H.Y.; Liao, S.C. Guillain-Barré Syndrome Associated with COVID-19 Vaccination. Emerg. Infect. Dis. 2021, 27, 3175–3178. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.H.; Kim, J.E.; Yoon, B.A.; Kim, J.K.; Bae, J.S. Bilateral Facial Weakness with Distal Paresthesia Following COVID-19 Vaccination: A Scoping Review for an Atypical Variant of Guillain-Barré Syndrome. Brain Sci. 2022, 12, 1046. [Google Scholar] [CrossRef] [PubMed]
- Chua, S.K.K.; Soh, Q.Y.; Saffari, S.E.; Tan, E.K. Prognosis of Guillain-Barré Syndrome Linked to COVID-19 Vaccination. Brain Sci. 2022, 12, 711. [Google Scholar] [CrossRef]
- Gupta, A.; Ranga, A.; Prakash, N.B.; Khanna, M. Rehabilitation outcomes in patients with post-COVID-19 vaccine-associated Guillain-Barre syndrome. J. Neurosci. Rural Pract. 2022, 13, 684–690. [Google Scholar] [CrossRef]
- Pritchard, J.; Mukherjee, R.; Hughes, R.A. Risk of relapse of Guillain-Barré syndrome or chronic inflammatory demyelinating polyradiculoneuropathy following immunisation. J. Neurol. Neurosurg. Psychiatry 2002, 73, 348–349. [Google Scholar] [CrossRef]
- Baars, A.E.; Kuitwaard, K.; de Koning, L.C.; Luijten, L.W.G.; Kok, W.M.; Eftimov, F.; Wieske, L.; Goedee, H.S.; van der Pol, W.L.; Blomkwist-Markens, P.H.; et al. SARS-CoV-2 Vaccination Safety in Guillain-Barré Syndrome, Chronic Inflammatory Demyelinating Polyneuropathy, and Multifocal Motor Neuropathy. Neurology 2023, 100, e182–e191. [Google Scholar] [CrossRef]
- Kim, J.E.; Yoon, B.A.; Kim, Y.H.; Kim, J.K.; Bae, J.S. Miller Fisher syndrome following COVID-19 vaccines: A scoping review. Acta Neurol. Scand. 2022, 146, 604–609. [Google Scholar] [CrossRef]
- Nishiguchi, Y.; Matsuyama, H.; Maeda, K.; Shindo, A.; Tomimoto, H. Miller Fisher syndrome following BNT162b2 mRNA coronavirus 2019 vaccination. BMC Neurol. 2021, 21, 452. [Google Scholar] [CrossRef]
- Siddiqi, A.R.; Khan, T.; Tahir, M.J.; Asghar, M.S.; Islam, M.S.; Yousaf, Z. Miller Fisher syndrome after COVID-19 vaccination: Case report and review of literature. Medicine 2022, 101, e29333. [Google Scholar] [CrossRef]
- Doron, A.; Piura, Y.; Vigiser, I.; Kolb, H.; Regev, K.; Nesher, N.; Karni, A. BNT162b2 mRNA COVID-19 vaccine three-dose safety and risk of COVID-19 in patients with myasthenia gravis during the alpha, delta, and omicron waves. J. Neurol. 2022, 269, 6193–6201. [Google Scholar] [CrossRef]
- Mirmosayyeb, O.; Moases Ghaffary, E.; Mazdak, M.; Bagheri, Z.; Bagherieh, S.; Shaygannejad, V. Is Myasthenia Gravis a Real Complication of the COVID-19 Vaccine? A Case Report-Based Systematic Review. Can. J. Infect. Dis. Med. Microbiol. J. Can. Mal. Infect. Microbiol. Med. 2022, 2022, 5009450. [Google Scholar] [CrossRef] [PubMed]
- Ruan, Z.; Tang, Y.; Li, C.; Sun, C.; Zhu, Y.; Li, Z.; Chang, T. COVID-19 Vaccination in Patients with Myasthenia Gravis: A Single-Center Case Series. Vaccines 2021, 9, 1112. [Google Scholar] [CrossRef] [PubMed]
- Maher, D.I.; Hogarty, D.; Ben Artsi, E. Acute onset ocular myasthenia gravis after vaccination with the Oxford-AstraZeneca COVID-19 vaccine. Orbit 2022, 1–5. [Google Scholar] [CrossRef]
- Ishizuchi, K.; Takizawa, T.; Sekiguchi, K.; Motegi, H.; Oyama, M.; Nakahara, J.; Suzuki, S. Flare of myasthenia gravis induced by COVID-19 vaccines. J. Neurol. Sci. 2022, 436, 120225. [Google Scholar] [CrossRef]
- Lotan, I.; Hellmann, M.A.; Friedman, Y.; Stiebel-Kalish, H.; Steiner, I.; Wilf-Yarkoni, A. Early safety and tolerability profile of the BNT162b2 COVID-19 vaccine in myasthenia gravis. Neuromuscul. Disord. NMD 2022, 32, 230–235. [Google Scholar] [CrossRef]
- Lee, M.A.; Lee, C.; Park, J.H.; Lee, J.H. Early-Onset Myasthenia Gravis Following COVID-19 Vaccination. J. Korean Med. Sci. 2022, 37, e50. [Google Scholar] [CrossRef] [PubMed]
- Koh, J.S.; Hoe, R.H.M.; Chen, G.J.; Goh, Y.; Tan, B.Y.Q.; Yong, M.H.; Hui, A.C.; Tu, T.M.; Yong, K.P.; Angon, J.; et al. Low incidence of neurological recurrent side-effects following COVID-19 reimmunization. QJM Mon. J. Assoc. Physicians 2023, 116, 221–226. [Google Scholar] [CrossRef]
- Tagliaferri, A.R.; Narvaneni, S.; Azzam, M.H.; Grist, W. A Case of COVID-19 Vaccine Causing a Myasthenia Gravis Crisis. Cureus 2021, 13, e15581. [Google Scholar] [CrossRef]
- Sonigra, K.J.; Sarna, K.; Vaghela, V.P.; Guthua, S. An Interesting Case of Fatal Myasthenic Crisis Probably Induced by the COVID-19 Vaccine. Cureus 2022, 14, e23251. [Google Scholar] [CrossRef]
- Camelo-Filho, A.E.; Silva, A.M.S.; Estephan, E.P.; Zambon, A.A.; Mendonça, R.H.; Souza, P.V.S.; Pinto, W.; Oliveira, A.S.B.; Dangoni-Filho, I.; Pouza, A.F.P.; et al. Myasthenia Gravis and COVID-19: Clinical Characteristics and Outcomes. Front. Neurol. 2020, 11, 1053. [Google Scholar] [CrossRef] [PubMed]
- Elnahry, A.G.; Asal, Z.B.; Shaikh, N.; Dennett, K.; Abd Elmohsen, M.N.; Elnahry, G.A.; Shehab, A.; Vytopil, M.; Ghaffari, L.; Athappilly, G.K.; et al. Optic neuropathy after COVID-19 vaccination: A report of two cases. Int. J. Neurosci. 2021, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Huang, S.; Yu, Z.; Zhang, S.; Hou, G.; Xu, S. Unilateral optic neuritis after vaccination against the coronavirus disease: Two case reports. Doc. Ophthalmol. Adv. Ophthalmol. 2022, 145, 65–70. [Google Scholar] [CrossRef] [PubMed]
- García-Estrada, C.; Gómez-Figueroa, E.; Alban, L.; Arias-Cárdenas, A. Optic neuritis after COVID-19 vaccine application. Clin. Exp. Neuroimmunol. 2022, 13, 72–74. [Google Scholar] [CrossRef] [PubMed]
- Motegi, S.; Kanda, T.; Takeuchi, M. A Case of Atypical Unilateral Optic Neuritis Following BNT162b2 mRNA COVID-19 Vaccination. Vaccines 2022, 10, 1574. [Google Scholar] [CrossRef]
- Lee, G.W.; Park, H.S.; Lee, D. Silent progressive bilateral papillitis after COVID-19 vaccination: A case report. Medicine 2022, 101, e31112. [Google Scholar] [CrossRef]
- Shah, P.P.; Gelnick, S.; Zhu, D.; Wong, A.; Verma, R. Optic neuritis secondary to the Pfizer-BioNTech-162b2 COVID-19 vaccine managed with plasmapheresis: A case report and review. Oman J. Ophthalmol. 2022, 15, 397–402. [Google Scholar]
- Kim, J.E.; Park, J.; Song, T.J. A disproportionality analysis for the association of central nervous system demyelinating diseases with COVID-19 vaccination using the World Health Organization pharmacovigilance database. Mult. Scler. 2022, 28, 2112–2123. [Google Scholar] [CrossRef]
- Gao, J.J.; Tseng, H.P.; Lin, C.L.; Shiu, J.S.; Lee, M.H.; Liu, C.H. Acute Transverse Myelitis Following COVID-19 Vaccination. Vaccines 2021, 9, 1008. [Google Scholar] [CrossRef]
- Ostovan, V.R.; Sahraian, M.A.; Karazhian, N.; Rostamihosseinkhani, M.; Salimi, M.; Marbooti, H. Clinical characteristics, radiological features and prognostic factors of transverse myelitis following COVID-19 vaccination: A systematic review. Mult. Scler. Relat. Disord. 2022, 66, 104032. [Google Scholar] [CrossRef]
- Ismail, I.I.; Salama, S. A systematic review of cases of CNS demyelination following COVID-19 vaccination. J. Neuroimmunol. 2022, 362, 577765. [Google Scholar] [CrossRef] [PubMed]
- Erdem, N.; Demirci, S.; Özel, T.; Mamadova, K.; Karaali, K.; Çelik, H.T.; Uslu, F.I.; Özkaynak, S.S. Acute transverse myelitis after inactivated COVID-19 vaccine. Ideggyogy. Szle. 2021, 74, 273–276. [Google Scholar] [CrossRef] [PubMed]
- Román, G.C.; Gracia, F.; Torres, A.; Palacios, A.; Gracia, K.; Harris, D. Acute Transverse Myelitis (ATM):Clinical Review of 43 Patients With COVID-19-Associated ATM and 3 Post-Vaccination ATM Serious Adverse Events With the ChAdOx1 nCoV-19 Vaccine (AZD1222). Front. Immunol. 2021, 12, 653786. [Google Scholar] [CrossRef] [PubMed]
- Garg, R.K.; Paliwal, V.K. Spectrum of neurological complications following COVID-19 vaccination. Neurol. Sci. Off. J. Ital. Neurol. Soc. Ital. Soc. Clin. Neurophysiol. 2022, 43, 3–40. [Google Scholar] [CrossRef]
- Khan, E.; Shrestha, A.K.; Colantonio, M.A.; Liberio, R.N.; Sriwastava, S. Acute transverse myelitis following SARS-CoV-2 vaccination: A case report and review of literature. J. Neurol. 2022, 269, 1121–1132. [Google Scholar] [CrossRef]
- Maramattom, B.V.; Lotlikar, R.S.; Sukumaran, S. Central nervous system adverse events after ChAdOx1 vaccination. Neurol. Sci. Off. J. Ital. Neurol. Soc. Ital. Soc. Clin. Neurophysiol. 2022, 43, 3503–3507. [Google Scholar] [CrossRef]
- Gustavsen, S.; Nordling, M.M.; Weglewski, A. Acute disseminated encephalomyelitis following the COVID-19 vaccine Ad26.COV2.S, a case report. Bull. Natl. Res. Cent. 2023, 47, 5. [Google Scholar] [CrossRef]
- Rinaldi, V.; Bellucci, G.; Buscarinu, M.C.; Reniè, R.; Marrone, A.; Nasello, M.; Zancan, V.; Nistri, R.; Palumbo, R.; Salerno, A.; et al. CNS inflammatory demyelinating events after COVID-19 vaccines: A case series and systematic review. Front. Neurol. 2022, 13, 1018785. [Google Scholar] [CrossRef]
- Permezel, F.; Borojevic, B.; Lau, S.; de Boer, H.H. Acute disseminated encephalomyelitis (ADEM) following recent Oxford/AstraZeneca COVID-19 vaccination. Forensic Sci. Med. Pathol. 2022, 18, 74–79. [Google Scholar] [CrossRef]
- Netravathi, M.; Dhamija, K.; Gupta, M.; Tamborska, A.; Nalini, A.; Holla, V.V.; Nitish, L.K.; Menon, D.; Pal, P.K.; Seena, V.; et al. COVID-19 vaccine associated demyelination & its association with MOG antibody. Mult. Scler. Relat. Disord. 2022, 60, 103739. [Google Scholar] [CrossRef]
- Poli, K.; Poli, S.; Ziemann, U. Multiple Autoimmune Syndromes Including Acute Disseminated Encephalomyelitis, Myasthenia Gravis, and Thyroiditis Following Messenger Ribonucleic Acid-Based COVID-19 Vaccination: A Case Report. Front. Neurol. 2022, 13, 913515. [Google Scholar] [CrossRef]
- Bastide, L.; Perrotta, G.; Lolli, V.; Mathey, C.; Vierasu, O.I.; Goldman, S.; Vandergheynst, F. Atypical acute disseminated encephalomyelitis with systemic inflammation after a first dose of AztraZaneca COVID-19 vaccine. A case report. Front. Neurol. 2022, 13, 995875. [Google Scholar] [CrossRef] [PubMed]
- Hada, M.; Mosholder, A.D.; Leishear, K.; Perez-Vilar, S. Systematic review of risk of SARS-CoV-2 infection and severity of COVID-19 with therapies approved to treat multiple sclerosis. Neurol. Sci. Off. J. Ital. Neurol. Soc. Ital. Soc. Clin. Neurophysiol. 2022, 43, 1557–1567. [Google Scholar] [CrossRef] [PubMed]
- Nojszewska, M.; Kalinowska, A.; Adamczyk-Sowa, M.; Kułakowska, A.; Bartosik-Psujek, H. COVID-19 mRNA vaccines (Pfizer-BioNTech and Moderna) in patients with multiple sclerosis: A statement by a working group convened by the Section of Multiple Sclerosis and Neuroimmunology of the Polish Neurological Society. Neurol. I Neurochir. Pol. 2021, 55, 8–11. [Google Scholar] [CrossRef]
- Yamout, B.I.; Zakaria, M.; Inshasi, J.; Al-Jumah, M.; Zeineddine, M.; Dahdaleh, M.; Bohlega, S.; Gouider, R.; Alroughani, R. MENACTRIMS practice guideline for COVID-19 vaccination in patients with multiple sclerosis. Mult. Scler. Relat. Disord. 2021, 56, 103225. [Google Scholar] [CrossRef]
- Fujimori, J.; Miyazawa, K.; Nakashima, I. Initial clinical manifestation of multiple sclerosis after immunization with the Pfizer-BioNTech COVID-19 vaccine. J. Neuroimmunol. 2021, 361, 577755. [Google Scholar] [CrossRef]
- Nabizadeh, F.; Ramezannezhad, E.; Kazemzadeh, K.; Khalili, E.; Ghaffary, E.M.; Mirmosayyeb, O. Multiple sclerosis relapse after COVID-19 vaccination: A case report-based systematic review. J. Clin. Neurosci. Off. J. Neurosurg. Soc. Australas. 2022, 104, 118–125. [Google Scholar] [CrossRef]
- Koch-Henriksen, N.; Thygesen, L.C.; Stenager, E.; Laursen, B.; Magyari, M. Incidence of MS has increased markedly over six decades in Denmark particularly with late onset and in women. Neurology 2018, 90, e1954–e1963. [Google Scholar] [CrossRef]
- Murtonen, A.; Sumelahti, M.L. Multiple sclerosis prevalence in 2000 and 2010 in Western Finland. Acta Neurol. Scand. 2020, 141, 311–318. [Google Scholar] [CrossRef]
- Magyari, M.; Sorensen, P.S. The changing course of multiple sclerosis: Rising incidence, change in geographic distribution, disease course, and prognosis. Curr. Opin. Neurol. 2019, 32, 320–326. [Google Scholar] [CrossRef] [PubMed]
- Khayat-Khoei, M.; Bhattacharyya, S.; Katz, J.; Harrison, D.; Tauhid, S.; Bruso, P.; Houtchens, M.K.; Edwards, K.R.; Bakshi, R. COVID-19 mRNA vaccination leading to CNS inflammation: A case series. J. Neurol. 2022, 269, 1093–1106. [Google Scholar] [CrossRef]
- Toljan, K.; Amin, M.; Kunchok, A.; Ontaneda, D. New diagnosis of multiple sclerosis in the setting of mRNA COVID-19 vaccine exposure. J. Neuroimmunol. 2022, 362, 577785. [Google Scholar] [CrossRef] [PubMed]
- Achiron, A.; Dolev, M.; Menascu, S.; Zohar, D.N.; Dreyer-Alster, S.; Miron, S.; Shirbint, E.; Magalashvili, D.; Flechter, S.; Givon, U.; et al. COVID-19 vaccination in patients with multiple sclerosis: What we have learnt by February 2021. Mult. Scler. 2021, 27, 864–870. [Google Scholar] [CrossRef]
- Kavosh, A.; Ashtari, F.; Naghavi, S.; Adibi, I.; Shaygannejad, V.; Karimi, Z.; Arabi, S.; Rahimi, M.; Mazaheri, S. Safety of Sinopharm vaccine for people with Multiple Sclerosis: Study of adverse reactions and disease activity. Mult. Scler. Relat. Disord. 2022, 61, 103708. [Google Scholar] [CrossRef] [PubMed]
- Paybast, S.; Emami, A.; Baghalha, F.; Naser Moghadasi, A. Watch out for neuromyelitis optica spectrum disorder onset or clinical relapse after COVID-19 vaccination: What neurologists need to know? Mult. Scler. Relat. Disord. 2022, 65, 103960. [Google Scholar] [CrossRef]
- Shirah, B.; Mulla, I.; Aladdin, Y. Optic Neuritis Following the BNT162b2 mRNA COVID-19 Vaccine in a Patient with Systemic Lupus Erythematosus Uncovering the Diagnosis of Neuromyelitis Optica Spectrum Disorders. Ocul. Immunol. Inflamm. 2022, 1–3. [Google Scholar] [CrossRef]
- Motahharynia, A.; Naghavi, S.; Shaygannejad, V.; Adibi, I. Fulminant neuromyelitis optica spectrum disorder (NMOSD) following COVID-19 vaccination: A need for reconsideration? Mult. Scler. Relat. Disord. 2022, 66, 104035. [Google Scholar] [CrossRef]
- Mealy, M.A.; Cook, L.J.; Pache, F.; Velez, D.L.; Borisow, N.; Becker, D.; Arango, J.A.J.; Paul, F.; Levy, M. Vaccines and the association with relapses in patients with neuromyelitis optica spectrum disorder. Mult. Scler. Relat. Disord. 2018, 23, 78–82. [Google Scholar] [CrossRef]
- Samim, M.M.; Dhar, D.; Goyal, S.; Dey, T.; Parvin, N.; Shah, R.D.; Singh, V.; Chowdhury, S.; Lal, B.M.; Varghese, N.; et al. AI-CoV Study: Autoimmune Encephalitis Associated with COVID-19 and Its Vaccines—A Systematic Review. J. Clin. Neurol. 2022, 18, 692–710. [Google Scholar] [CrossRef]
- Zlotnik, Y.; Gadoth, A.; Abu-Salameh, I.; Horev, A.; Novoa, R.; Ifergane, G. Case Report: Anti-LGI1 Encephalitis Following COVID-19 Vaccination. Front. Immunol. 2021, 12, 813487. [Google Scholar] [CrossRef] [PubMed]
- Abu-Abaa, M.; Dawood, G.; Arshad, H.; Jumaah, O.; Landau, D. A Possible Case of Autoimmune Encephalitis After mRNA COVID-19 Booster Vaccine: A Case Report. Cureus 2022, 14, e31118. [Google Scholar] [CrossRef]
- Huang, Y.F.; Ho, T.C.; Chang, C.C.; Shen, D.H.; Chan, H.P.; Chuang, K.P.; Tyan, Y.C.; Yang, M.H. A Rare Adverse Effect of the COVID-19 Vaccine on Autoimmune Encephalitis. Vaccines 2022, 10, 1114. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Guo, K.; Lu, L.; Luo, R.; Liu, J.; Zhou, D.; Hong, Z. Safety of inactivated COVID-19 vaccines in autoimmune encephalitis: A real-world cross-sectional survey. Mult. Scler. Relat. Disord. 2022, 70, 104495. [Google Scholar] [CrossRef]
- Mastropaolo, M.; Hasbani, M.J. Small Fiber Neuropathy Triggered by COVID-19 Vaccination: Association with FGFR3 Autoantibodies and Improvement during Intravenous Immunoglobulin Treatment. Case Rep. Neurol. 2023, 15, 6–10. [Google Scholar] [CrossRef]
- Schelke, M.W.; Barcavage, S.; Lampshire, E.; Brannagan, T.H., 3rd. Post-COVID-19 vaccine small-fiber neuropathy and tinnitus treated with plasma exchange. Muscle Nerve 2022, 66, E21–E23. [Google Scholar] [CrossRef]
- Trevino, J.A.; Novak, P. TS-HDS and FGFR3 antibodies in small fiber neuropathy and Dysautonomia. Muscle Nerve 2021, 64, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Watad, A.; De Marco, G.; Mahajna, H.; Druyan, A.; Eltity, M.; Hijazi, N.; Haddad, A.; Elias, M.; Zisman, D.; Naffaa, M.E.; et al. Immune-Mediated Disease Flares or New-Onset Disease in 27 Subjects Following mRNA/DNA SARS-CoV-2 Vaccination. Vaccines 2021, 9, 435. [Google Scholar] [CrossRef]
- Waheed, W.; Carey, M.E.; Tandan, S.R.; Tandan, R. Post COVID-19 vaccine small fiber neuropathy. Muscle Nerve 2021, 64, E1–E2. [Google Scholar] [CrossRef]
- Fathy, R.A.; McMahon, D.E.; Lee, C.; Chamberlin, G.C.; Rosenbach, M.; Lipoff, J.B.; Tyagi, A.; Desai, S.R.; French, L.E.; Lim, H.W.; et al. Varicella-zoster and herpes simplex virus reactivation post-COVID-19 vaccination: A review of 40 cases in an International Dermatology Registry. J. Eur. Acad. Dermatol. Venereol. JEADV 2022, 36, e6–e9. [Google Scholar] [CrossRef]
- Walsh, E.E.; Frenck, R.W., Jr.; Falsey, A.R.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Neuzil, K.; Mulligan, M.J.; Bailey, R.; et al. Safety and Immunogenicity of Two RNA-Based COVID-19 Vaccine Candidates. N. Engl. J. Med. 2020, 383, 2439–2450. [Google Scholar] [CrossRef] [PubMed]
- Psichogiou, M.; Samarkos, M.; Mikos, N.; Hatzakis, A. Reactivation of Varicella Zoster Virus after Vaccination for SARS-CoV-2. Vaccines 2021, 9, 572. [Google Scholar] [CrossRef] [PubMed]
- Hertel, M.; Heiland, M.; Nahles, S.; von Laffert, M.; Mura, C.; Bourne, P.E.; Preissner, R.; Preissner, S. Real-world evidence from over one million COVID-19 vaccinations is consistent with reactivation of the varicella-zoster virus. J. Eur. Acad. Dermatol. Venereol. JEADV 2022, 36, 1342–1348. [Google Scholar] [CrossRef]
- Rodríguez-Jiménez, P.; Chicharro, P.; Cabrera, L.M.; Seguí, M.; Morales-Caballero, Á.; Llamas-Velasco, M.; Sánchez-Pérez, J. Varicella-zoster virus reactivation after SARS-CoV-2 BNT162b2 mRNA vaccination: Report of 5 cases. JAAD Case Rep. 2021, 12, 58–59. [Google Scholar] [CrossRef] [PubMed]
- Katsikas Triantafyllidis, K.; Giannos, P.; Mian, I.T.; Kyrtsonis, G.; Kechagias, K.S. Varicella Zoster Virus Reactivation Following COVID-19 Vaccination: A Systematic Review of Case Reports. Vaccines 2021, 9, 1013. [Google Scholar] [CrossRef]
- Wan, E.Y.F.; Chui, C.S.L.; Wang, Y.; Ng, V.W.S.; Yan, V.K.C.; Lai, F.T.T.; Li, X.; Wong, C.K.H.; Chan, E.W.Y.; Wong, C.S.M.; et al. Herpes zoster related hospitalization after inactivated (CoronaVac) and mRNA (BNT162b2) SARS-CoV-2 vaccination: A self-controlled case series and nested case-control study. Lancet Reg. Health West. Pac. 2022, 21, 100393. [Google Scholar] [CrossRef]
- Shahrudin, M.S.; Mohamed-Yassin, M.S.; Nik Mohd Nasir, N.M. Herpes Zoster Following COVID-19 Vaccine Booster. Am. J. Case Rep. 2023, 24, e938667. [Google Scholar] [CrossRef]
- Préta, L.H.; Contejean, A.; Salvo, F.; Treluyer, J.M.; Charlier, C.; Chouchana, L. Association study between herpes zoster reporting and mRNA COVID-19 vaccines (BNT162b2 and mRNA-1273). Br. J. Clin. Pharmacol. 2022, 88, 3529–3534. [Google Scholar] [CrossRef]
- Chen, J.; Li, F.; Tian, J.; Xie, X.; Tang, Q.; Chen, Y.; Ge, Y. Varicella zoster virus reactivation following COVID-19 vaccination in patients with autoimmune inflammatory rheumatic diseases: A cross-sectional Chinese study of 318 cases. J. Med. Virol. 2023, 95, e28307. [Google Scholar] [CrossRef] [PubMed]
- May Lee, M.; Macchi, S.; Mora, E.; Feliciani, C. Potential risk factors for Varicella-zoster virus reactivation after COVID-19 vaccination. J. Cosmet. Dermatol. 2022, 21, 1347–1349. [Google Scholar] [CrossRef]
- Butler, M.; Coebergh, J.; Safavi, F.; Carson, A.; Hallett, M.; Michael, B.; Pollak, T.A.; Solomon, T.; Stone, J.; Nicholson, T.R. Functional Neurological Disorder After SARS-CoV-2 Vaccines: Two Case Reports and Discussion of Potential Public Health Implications. J. Neuropsychiatry Clin. Neurosci. 2021, 33, 345–348. [Google Scholar] [CrossRef] [PubMed]
- Ercoli, T.; Lutzoni, L.; Orofino, G.; Muroni, A.; Defazio, G. Functional neurological disorder after COVID-19 vaccination. Neurol. Sci. Off. J. Ital. Neurol. Soc. Ital. Soc. Clin. Neurophysiol. 2021, 42, 3989–3990. [Google Scholar] [CrossRef]
- Espay, A.J.; Aybek, S.; Carson, A.; Edwards, M.J.; Goldstein, L.H.; Hallett, M.; LaFaver, K.; LaFrance, W.C., Jr.; Lang, A.E.; Nicholson, T.; et al. Current Concepts in Diagnosis and Treatment of Functional Neurological Disorders. JAMA Neurol. 2018, 75, 1132–1141. [Google Scholar] [CrossRef]
- Fasano, A.; Daniele, A. Functional disorders after COVID-19 vaccine fuel vaccination hesitancy. J. Neurol. Neurosurg. Psychiatry 2022, 93, 339–340. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, O.; Sakakibara, R.; Sawai, S.; Ogata, T. Functional neurological disorders after COVID-19 vaccination: Case series and literature review. Psychiatry Clin. Neurosci. 2022, 76, 529–531. [Google Scholar] [CrossRef] [PubMed]
- de Souza, A.; Jacques, R.; Mohan, S. Vaccine-Induced Functional Neurological Disorders in the COVID-19 Era. Can. J. Neurol. Sci. J. Can. Sci. Neurol. 2022, 50, 346–350. [Google Scholar] [CrossRef]
- Alonso-Canovas, A.; Kurtis, M.M.; Gomez-Mayordomo, V.; Macías-García, D.; Gutiérrez Viedma, Á.; Mondragón Rezola, E.; Pagonabarraga, J.; Aranzabal Orgaz, L.; Masjuan, J.; Martinez-Castrillo, J.C.; et al. Functional neurological disorders after COVID-19 and SARS-CoV-2 vaccines: A national multicentre observational study. J. Neurol. Neurosurg. Psychiatry, 2023; online first. [Google Scholar] [CrossRef]
- Linden, S.C.; Carson, A.J.; Wessely, S. Functional neurological disorder after vaccination: A balanced approach informed by history. J. R. Coll. Physicians Edinb. 2021, 51, 330–331. [Google Scholar] [CrossRef] [PubMed]
| VITT-CVST | Non-VITT-CVST | |
|---|---|---|
| Age | Younger | Older |
| Vaccine type | Adenoviral vector-based vaccines | Non-vector-based vaccines |
| Onset time | Shorter interval | Longer interval |
| Thrombocytopenia | Yes | Usually no |
| PF4 antibodies assay | Usually positive | Negative |
| D-dimer | Higher than 2000 μg/L | Lower than 2000 μg/L |
| Other risk factors for thromboses | Usually absent | Usually present |
| Intracerebral hemorrhage | More common | Less common |
| Thromboses in other sites | More common | Less common |
| Mortality | Higher | Lower |
| Treatment | Non-heparin anticoagulants and IVIG | Heparin anticoagulants |
| Guillain–Barre Syndrome | Miller–Fisher Syndrome | |
|---|---|---|
| Vaccine type | Most adenoviral vector-based vaccines | Various kinds of vaccines |
| Anti-ganglioside antibodies | Usually absent | Usually present |
| Respiratory muscle involvement | More frequent | Less frequent |
| Treatment | IVIG, plasma exchange | Usually IVIG only |
| Prognosis | Various | Good |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, Y.; Huang, L. Neurological Disorders following COVID-19 Vaccination. Vaccines 2023, 11, 1114. https://doi.org/10.3390/vaccines11061114
Yang Y, Huang L. Neurological Disorders following COVID-19 Vaccination. Vaccines. 2023; 11(6):1114. https://doi.org/10.3390/vaccines11061114
Chicago/Turabian StyleYang, Ying, and Lisu Huang. 2023. "Neurological Disorders following COVID-19 Vaccination" Vaccines 11, no. 6: 1114. https://doi.org/10.3390/vaccines11061114
APA StyleYang, Y., & Huang, L. (2023). Neurological Disorders following COVID-19 Vaccination. Vaccines, 11(6), 1114. https://doi.org/10.3390/vaccines11061114






