Efficacy and Effectiveness of SARS-CoV-2 Vaccines: A Systematic Review and Meta-Analysis
Abstract
:1. Introduction
2. Materials and Methods
- Reported COVID-19 vaccine efficacy (RCTs) or effectiveness (observational studies).
- Had a comparator group receiving either a placebo or another vaccine.
- The intervention group were either partially vaccinated (received only one dose of COVID-19 vaccine) or fully vaccinated.
- No restriction regarding country, race, gender, or age.
- We excluded abstract only letters to the editor, reviews, conference reports, study protocols, author responses, case reports, case series, and surveillance studies with no control group, in addition to any studies that had unreliable data for extraction or duplicates.
- Efficacy and effectiveness of COVID-19 vaccines to prevent COVID-19 mortality.
- Efficacy and effectiveness of the vaccine to prevent severe disease.
- Efficacy and effectiveness of the vaccine to prevent SARS-CoV-2 infection within 7, 14, 21, and 28 days of the 1st dose.
- Efficacy and effectiveness of the vaccine to prevent SARS-CoV-2 infection any time after the 1st dose.
- Efficacy and effectiveness of the vaccine to prevent SARS-CoV-2 infection within 7 and 14 days of the 2nd dose, and 7,14 days after the 2nd dose.
- Efficacy and effectiveness of the vaccine to prevent SARS-CoV-2 infection any time after the 2nd dose.
3. Data Collection and Extraction
4. Data Analysis
- Visual inspection of the forest plot was carried out to analyze the consistency of intervention effects across the included studies. If the same intervention effect is estimated, there should be an overlap between the confidence intervals for each effect estimate on the forest plot. However, if the overlap is weak, or there are outliers, statistical heterogeneity is likely to be present.
- Statistical test for variation: heterogeneity was assessed by inspecting the forest plots to detect overlapping confidence intervals (CIs) and the I2 statistic used to denote levels of heterogeneity as defined in the Cochrane Handbook for Systematic Reviews of Interventions [30].
- 0% to 40%: might not be important.
- 30% to 60%: may represent moderate heterogeneity.
- 50% to 90%: may represent substantial heterogeneity.
- 75% to 100%: considerable heterogeneity.
5. Results
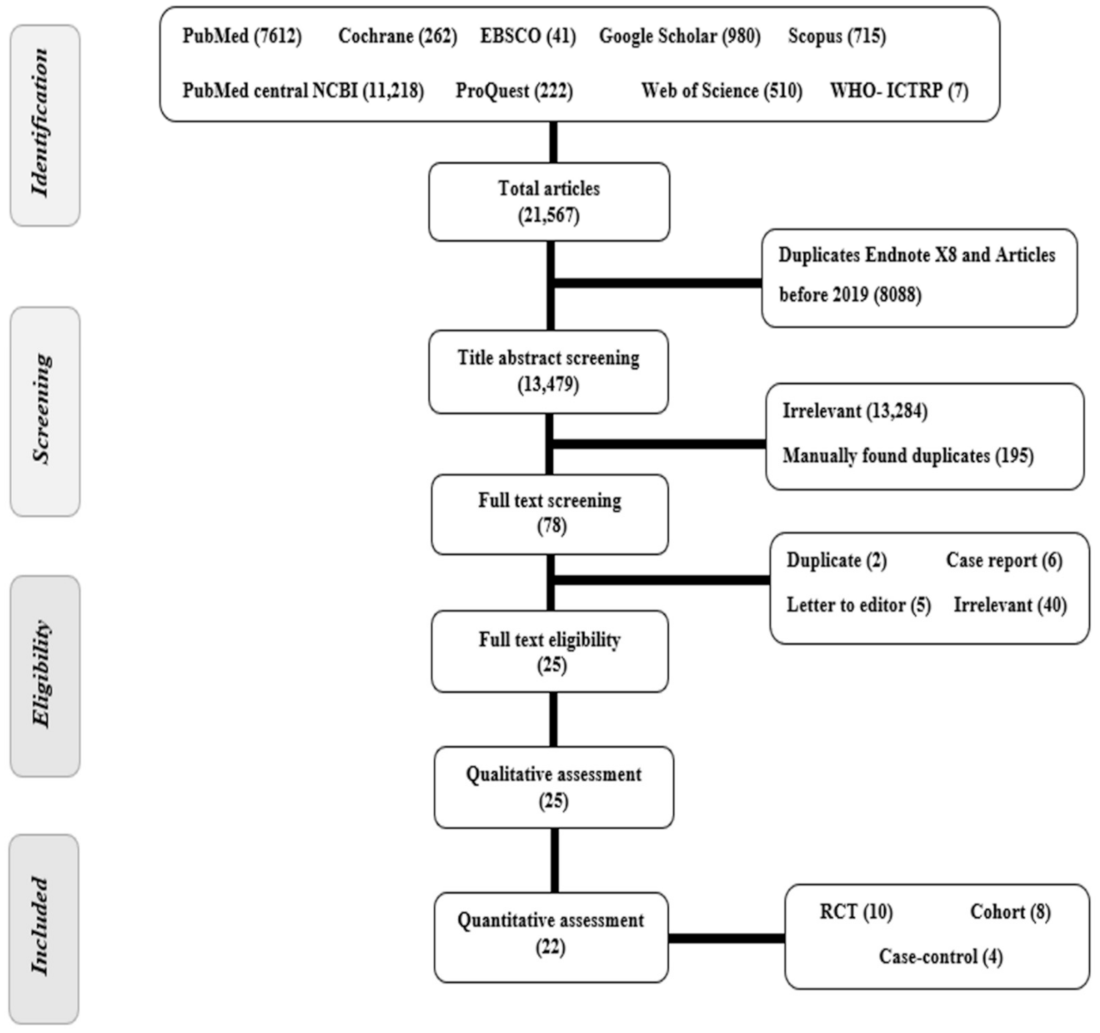
5.1. Study Selection Process
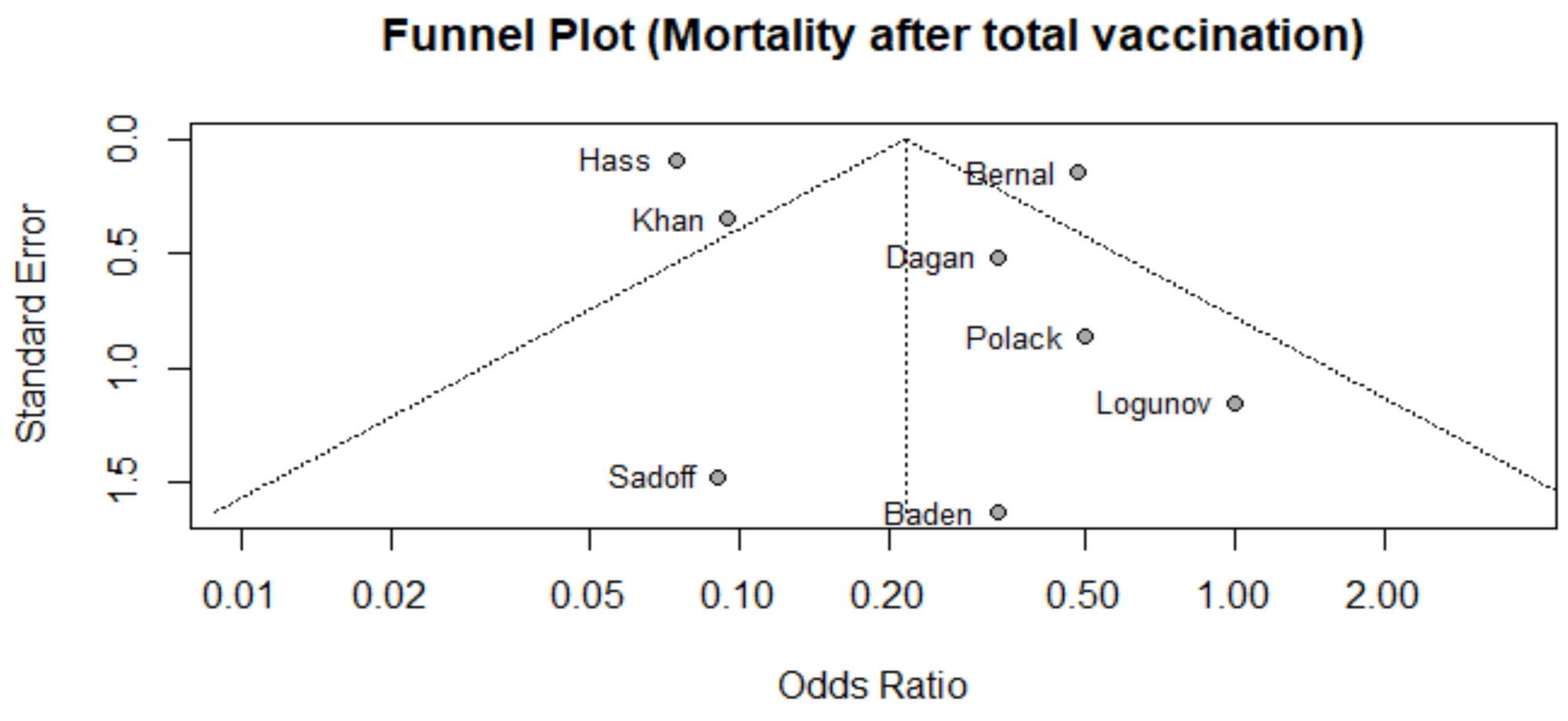
5.2. Publication Bias
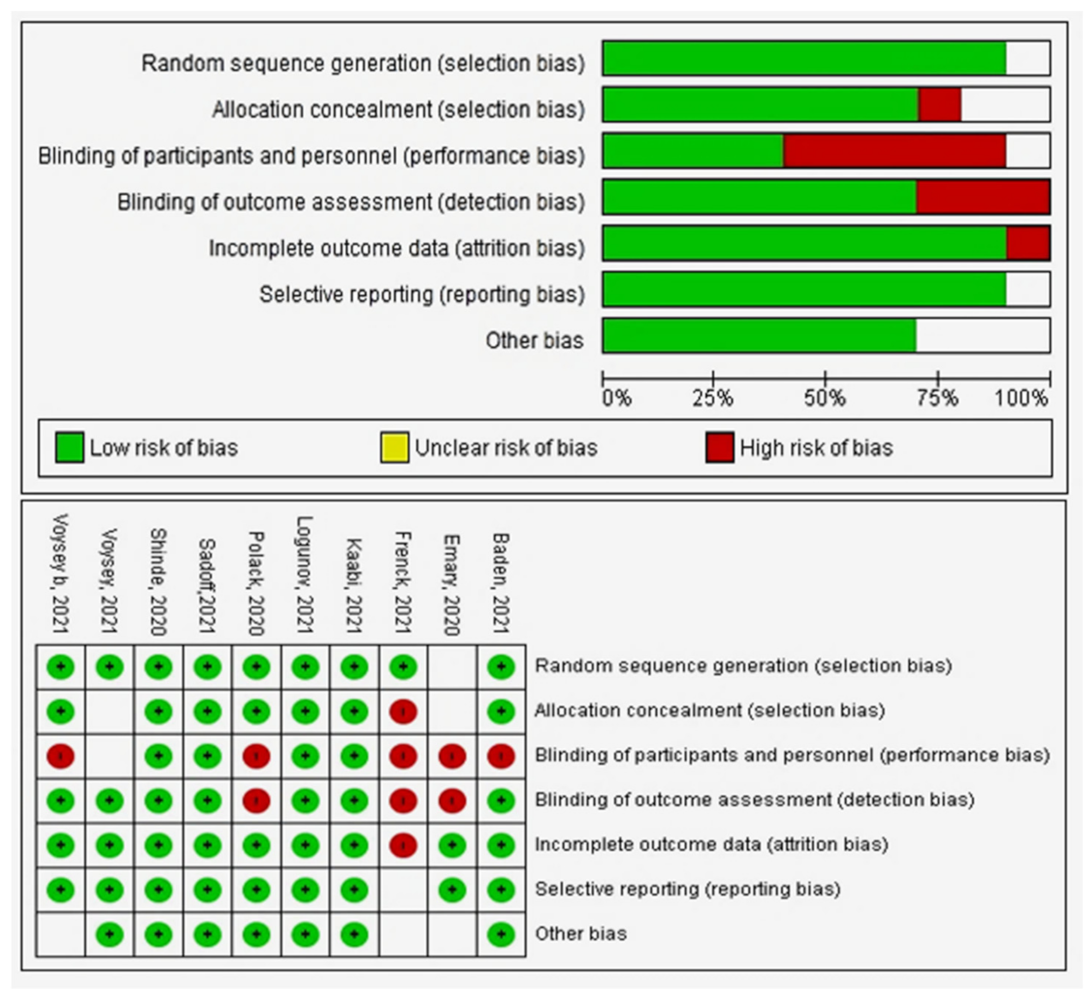
6. Quality Assessment
6.1. Primary Outcome
Mortality after 7 and 14 Days after 2nd Dose
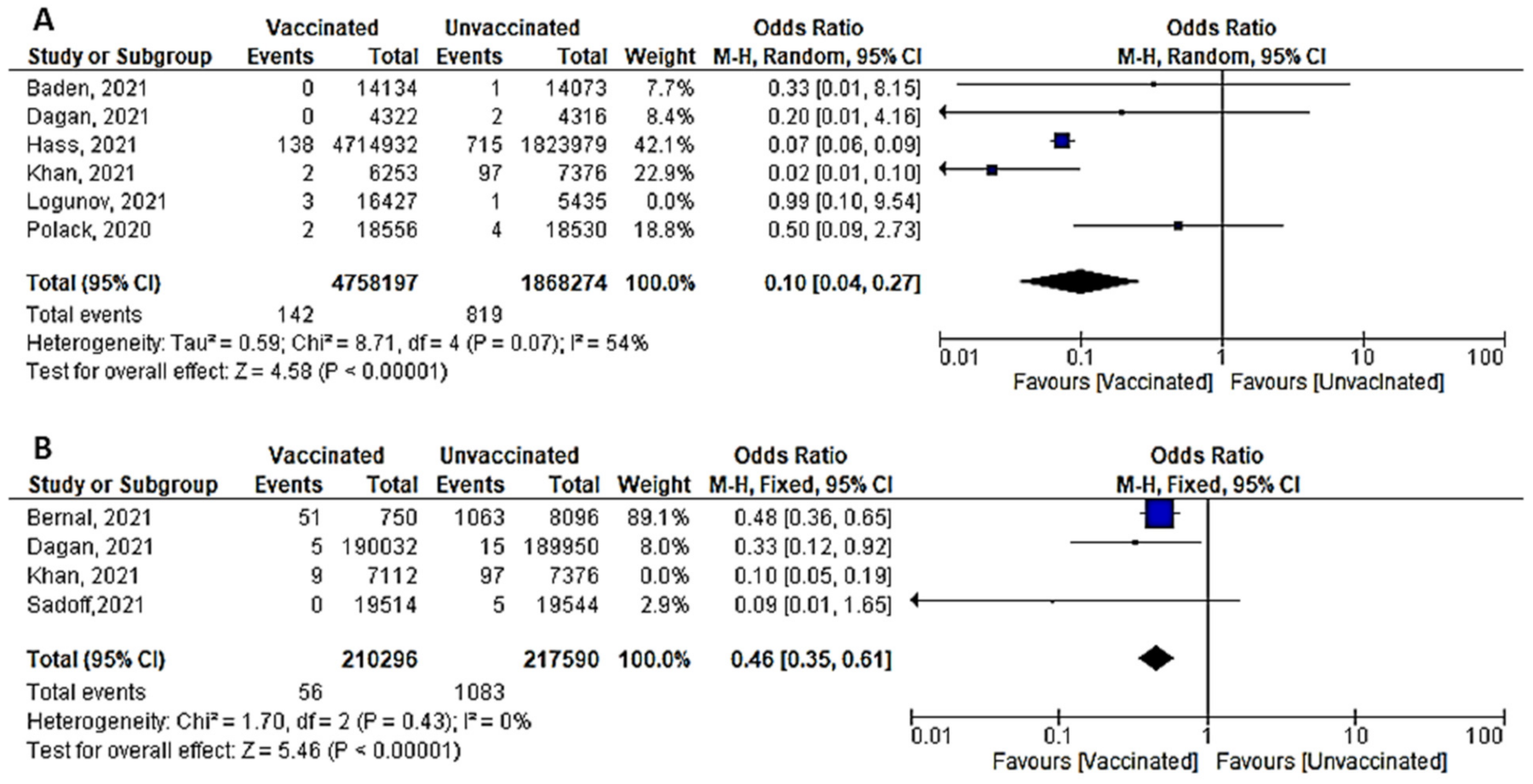
- A total of six studies (4 RCTs and 2 observational studies) assessed mortality due to COVID-19, 7 days after the 2nd dose of vaccination. Mortality among vaccinated subjects decreased significantly (OR = 0.14, [95% CI, 0.05–0.41], I2 = 63%). After conducting sensitivity analysis, the study of Logunov et al. [40] was removed, heterogeneity decreased to 54%; and the odds ratio became 0.10 [95% CI, 0.04–0.27], p < 0.001 (Figure 4A). It is important to note that in the study of Khan et al. [51] deaths due to COVID-19 were not separated from other causes of death.
- Four studies addressed mortality 14 days after 2nd dose, all of which were observational, except for the study by Sadoff et al. [30]. The overall mortality among vaccinated population decreased significantly (OR = 0.34, [95% CI, 0.26–0.44], I2 = 85%). After conducting a leave-one-out sensitivity analysis, the study of Khan et al. [51] was omitted. The observed heterogeneity dropped to 0% with an odds ratio of 0.46 [95% CI, 0.35–0.61], p < 0.001 (Figure 4B).
- Main findings: COVID-19 vaccination significantly decreased deaths 7 and 14 days after 2nd dose.
6.2. Secondary Outcomes
Severe COVID-19 Infection 7 Days after the 1st and 2nd Doses
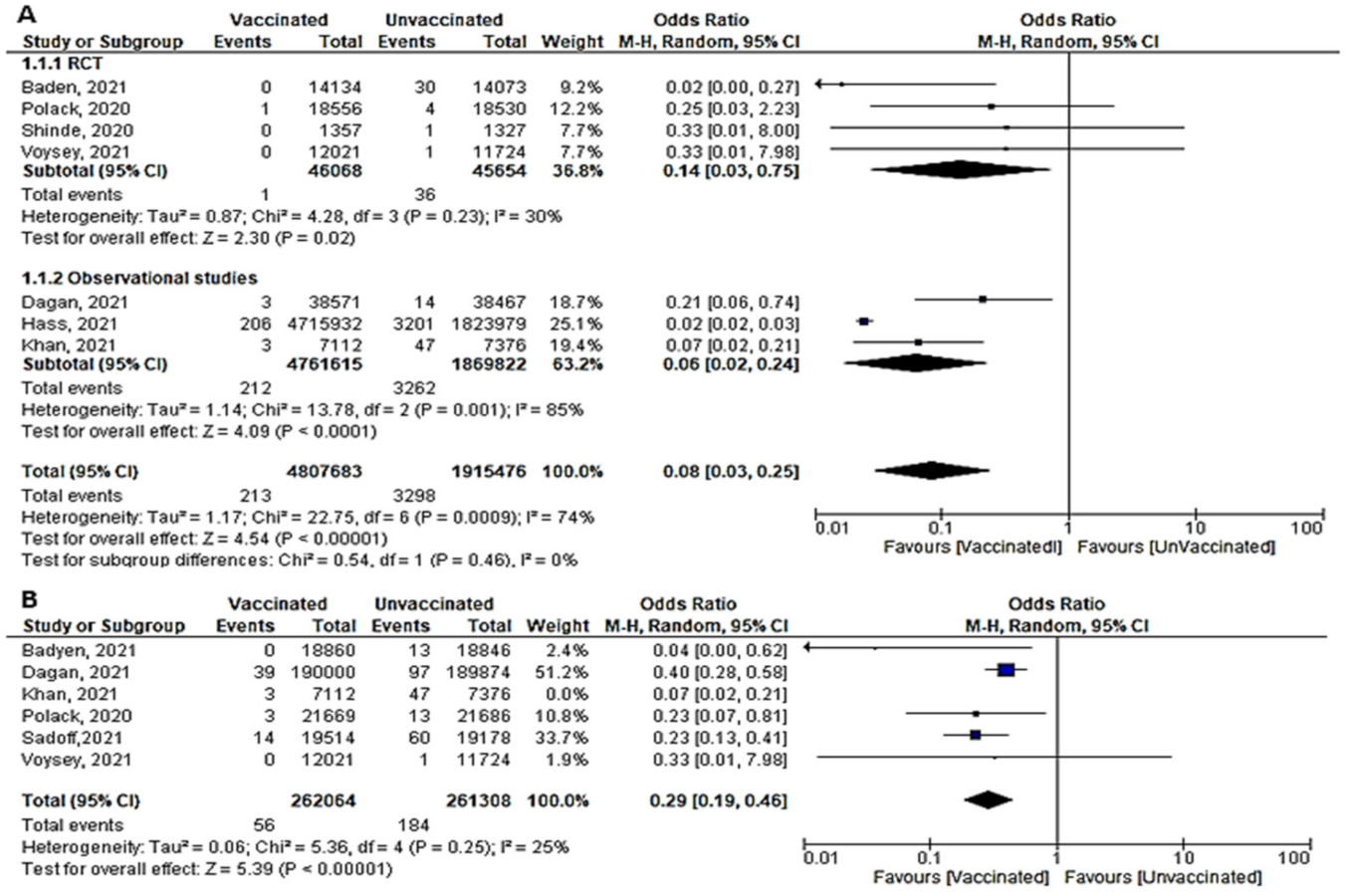
- Seven papers studied the incidence of severe COVID-19 infection 7 days after the 2nd dose. In total, 213 of 480,7683 vaccinated people developed severe COVID-19, compared to 3298 out of 191,5476 unvaccinated subjects. The odds ratio of developing severe COVID-19 among vaccinated subjects was 0.08 [95% CI, 0.03–0.25], I2 = 74%. After subgrouping the included studies into RCTs and observational, the heterogeneity dropped to 0%. The difference between observational and interventional studies was not significant (p = 0.46). The odds ratio of severe COVID-19 among vaccinated population in RCTs was 0.14 (95% CI, 0.03–0.75), I2 = 30%, p < 0.001 while in observational studies was 0.06 [0.02–0.24], I2 = 85%, p < 0.001. (Figure 5A).
- Main findings: COVID-19 vaccination significantly decreased severe COVID-19 after vaccination with either 1 or 2 doses
6.3. Efficacy and Effectiveness of COVID-19 Vaccine in Reducing Infection Incidence after the 1st Dose
6.3.1. Cases Reported within 7 Days of 1st Dose (Total Cases, Symptomatic and Asymptomatic)
- Two studies evaluated the efficacy and effectiveness of the COVID-19 vaccines in reducing SARS-CoV-2 infection (symptomatic and asymptomatic) within 7 days of the 1st dose. Dagan et al., [46] reported that 1965 of 596,618 vaccinated subjects got infection compared to 2362 of 596,618 unvaccinated subjects, OR = 0.83 [95% CI, 0.78–0.88]. 2362 of 596,618 unvaccinated subjects, OR = 0.83 [95% CI, 0.78–0.88]. Hall et al. [50] highlighted that the incidence of SARS-CoV-2 was 140 out of 20,641 unvaccinated subjects; it was lower than cases reported among unvaccinated (977 out of 2683), with OR = 0.01 [95% CI, 0.01–0.01]. Due to the significant heterogeneity, we could not pool the findings of these two outcomes.
- Bernal et al. [52] reported that 346 out of 864 vaccinated subjects versus 8988 out of 24,706 unvaccinated subjects got symptomatic SARS-CoV-2 infection, OR = 1.17 [95% CI, 1.02–1.34]. However, Dagan et al. [46] found that the COVID-19 vaccine had a protective effect, with an OR of 0.78 [95% CI, 0.72–0.84]. Among 596,618 vaccinated individuals, about 1103 subjects developed symptomatic SARS-CoV-2 infection, while among 596,618 unvaccinated individuals, about 1419 subjects developed symptomatic SARS-CoV-2 infection. Due to the significant heterogeneity, we could not pool the findings of these two outcomes.
- No studies reported asymptomatic cases within 7 days of the 1st dose.
6.3.2. Cases Reported within 14 Days of 1st Dose:
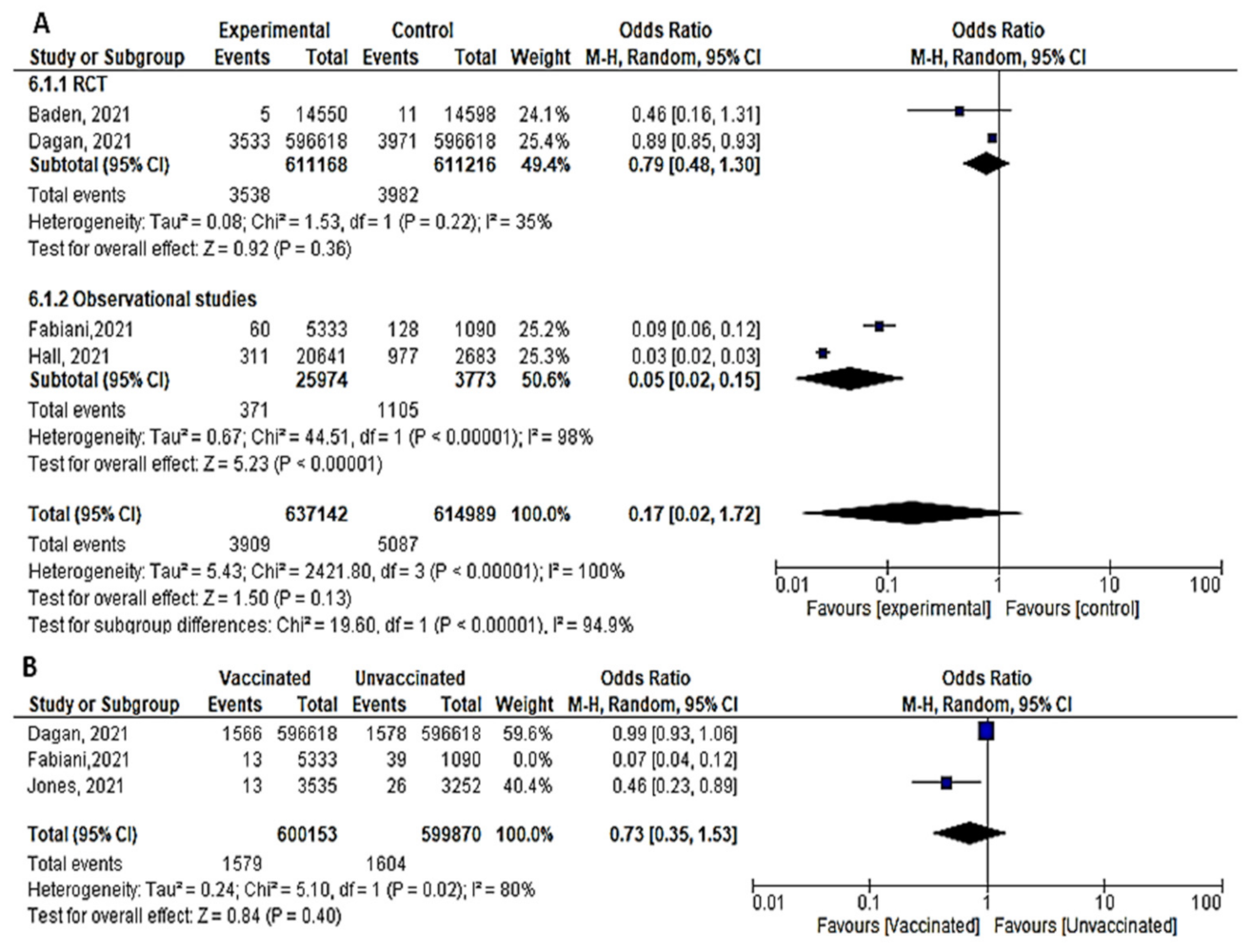
- Four studies assessed the efficacy and effectiveness of COVID-19 vaccines within 14 days of the 1st dose. In total, 3909 of 637,142 vaccinated subjects developed SARS-CoV-2 infection (symptomatic or asymptomatic) within 2 weeks after the 1st dose, compared to 5087 of 614,989 unvaccinated subjects. The odds ratio of getting SARS-CoV-2 infection decreased significantly (OR = 0.17 ([95% CI, 0.02–1.72], I2 = 100%). After we sub-grouped the included studies into RCTs and observational, the heterogeneity decreased to 94%. The difference between observational and interventional studies was significant (p = 0.001). The odds ratio of getting infection with SARS-CoV-2 in the RCTs was 0.79 [95% CI, 0.48–1.3], p = 0.36 while that of observational studies was 0.05 [95% CI, 0.02–0.15], p < 0.001. (Figure 6A).
- Bernal et al. [52] reported 958 symptomatic cases out of 1154 vaccinated subjects compared to 89 out of 8988 unvaccinated subjects, OR = 83.84 [95% CI, 68.07–103.26]. On the other hand, the protective effect of the COVID-19 vaccine was addressed by Dagan et al. [46] 1967 out of 596,618 vaccinated subjects versus 2393 out of 596,618 unvaccinated subjects OR = 0.82, [95% CI, 0.77–0.87]. On the same line, Fabiani et al. [47] reported that 47 out of 5333 vaccinated subjects versus 89 out of 1090 unvaccinated subjects developed symptomatic diseases, OR = 0.1, [95% CI, 0.07–0.14]. The results could not be pooled due to the significant heterogeneity.
- Three studies addressed the efficacy and effectiveness of vaccination in reducing the risk of having asymptomatic COVID-19 within 14 days of the 1st dose (OR = 0.23 [95% CI, 0.06–1.63], I2 = 97%). After conducting a leave-one-out sensitivity analysis, the study by Fabiani et al., [47] was omitted. The probability of getting asymptomatic infection did not reduce significantly. (OR = 0.73 [95% CI, 0.35–1.53], I2= 80%), p < 0.02. (Figure 6B).
- Main finding: incidence of SARS-CoV-2 infection in observational studies decreased significantly after vaccination within 14 days of 1st dose.
6.3.3. Cases Reported within 21 Days of 1st Dose
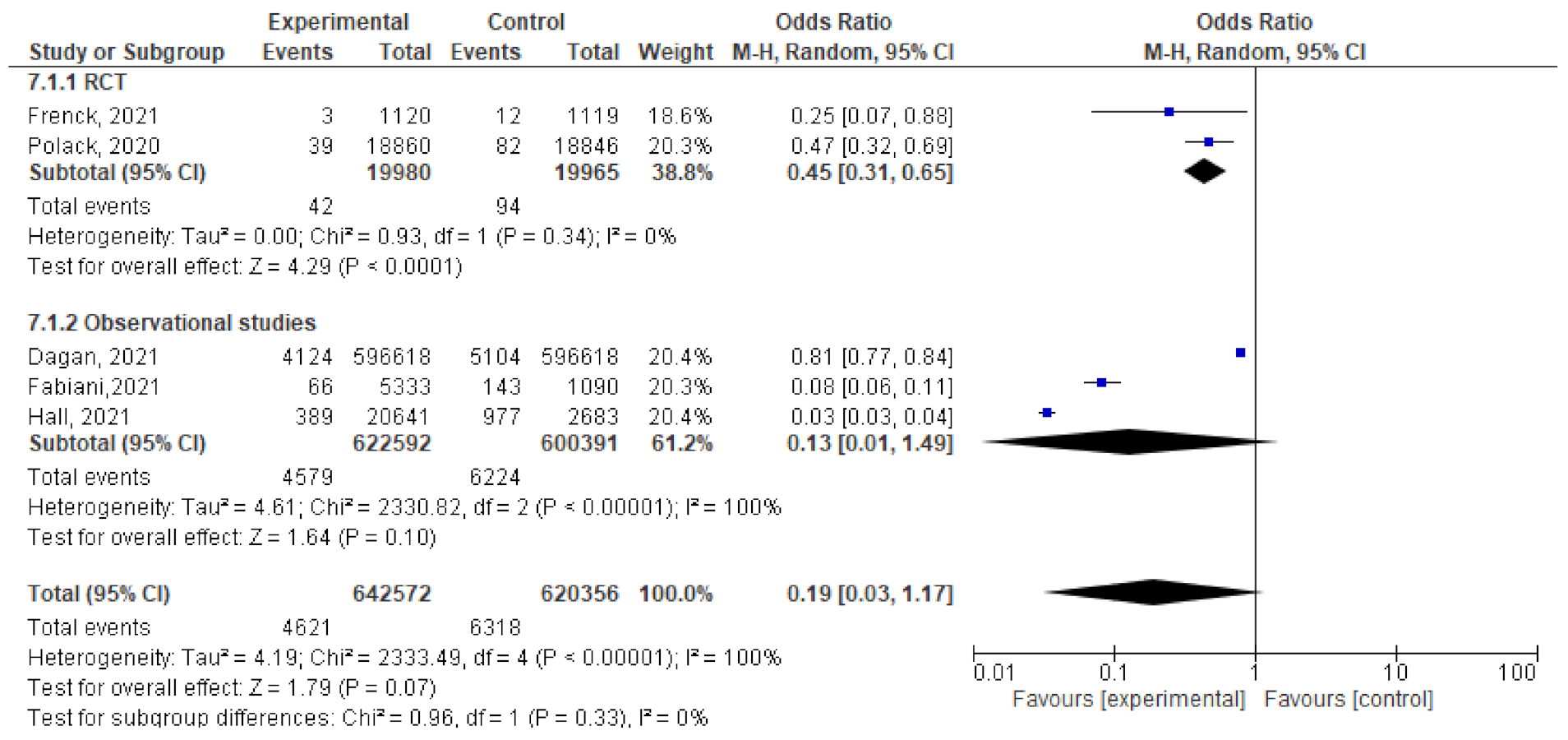
- Five studies assessed the efficacy and effectiveness of COVID-19 vaccines within 21 days of the 1st dose. In total, 4621 of 642,572 vaccinated subjects developed SARS-CoV-2 infection (symptomatic and asymptomatic) within 3 weeks of the 1st dose, compared to 6318 out of 620,356 unvaccinated subjects. The odds ratio of having infection was 0.19 [95% CI, 0.03–1.17], I2 = 100%. After subgrouping the included studies into RCTs and observational, the heterogeneity dropped to 0%. The difference between observational and interventional studies was insignificant (p = 0.33). The odds ratio of infection with COVID-19 in the RCTs was 0.45 [95% CI, 0.31–0.65], p < 0.001, while the OR in observational studies was 0.13 [95% CI, 0.01–1.49], p < 0.001 (Figure 7).
- For Dagan et al. [46], the odds ratio of catching symptomatic SARS-CoV-2 after vaccination was 0.73 [95% CI, 0.69–0.77]. Of the 596,618 vaccinated subjects, 2250 developed symptomatic SARS-CoV-2 infection, compared to 3079 out of 596,618 unvaccinated subjects. Fabiani et al. [47] reported that 51 out of 53,333 vaccinated subjects developed symptomatic disease compared to 97 out of 1090 unvaccinated subjects, OR = 0.1 (95% CI, 0.07–0.14). The results could not be pooled due to the significant heterogeneity.
- Only the study by Fabiani et al. [47] addressed the effectiveness of COVID-19 against asymptomatic infection three weeks after the 1st dose. In total, 15 out of 5333 vaccinated subjects developed asymptomatic SARS-CoV-2 compared to 46 out of 1090 unvaccinated subjects, OR = 0.06 [95% CI, 0.04–0.12].
- Main findings: incidence of SARS-CoV-2 infection within 21 days of the 1st dose of vaccination reduced significantly.
6.3.4. Cases Reported within 28 Days of 1st Dose
- The total number of cases (symptomatic and asymptomatic) reported 28 days after the 1st dose was reported in two studies; Dagan et al. [46] documented 4405 confirmed cases of 596,618 vaccinated individuals compared to 5775 confirmed cases of 596,618 unvaccinated subjects. Hall et al. [50] diagnosed 427 cases of 20,641 vaccinated subjects, and 977 cases of 2683 unvaccinated subject. The odds ratios were 0.76 [95% CI, 0.73–0.79] and 0.04 [95% CI, 0.03–0.4], respectively. The research team could not pool the odds ratios due to the significant heterogeneity.
- No studies reported asymptomatic or symptomatic cases within 28 days of the 1st dose.
6.3.5. Cases Reported Any Time after 1st Dose
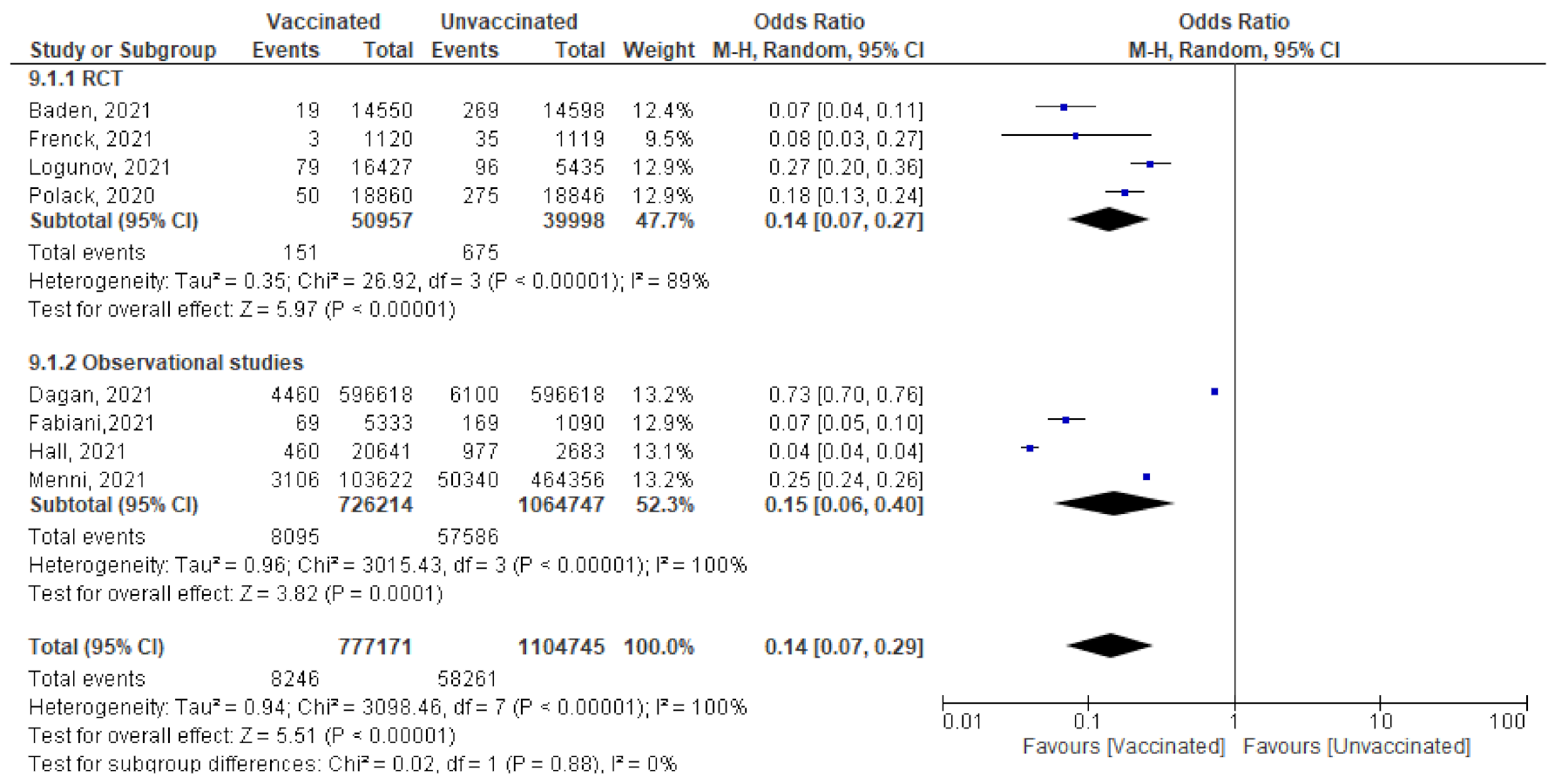
- Among 777,171 vaccinated subjects with a single dose of COVID-19 vaccine, 8246 got SARS-CoV-2 infection (symptomatic or asymptomatic) compared to 58,261 of 1,104,745 unvaccinated subjects (OR = 0.14 [95% CI, 0.07–0.4], I2 = 100%). After subgrouping based on the study design, the heterogeneity dropped to 0%, the odds ratio of RCTs was 0.14 [95% CI, 0.07–0.27], p < 0.001 and of observational studies was 0.15 [95% CI, 0.06–0.4] p < 0.001. The difference between subgroups was not significant, p = 0.88. (Figure 8)
- Main findings: incidence of SARS-CoV-2 infection any time after the 1st dose decreased significantly among vaccinated compared to unvaccinated.
6.4. Efficacy/Effectiveness of the 2nd Dose
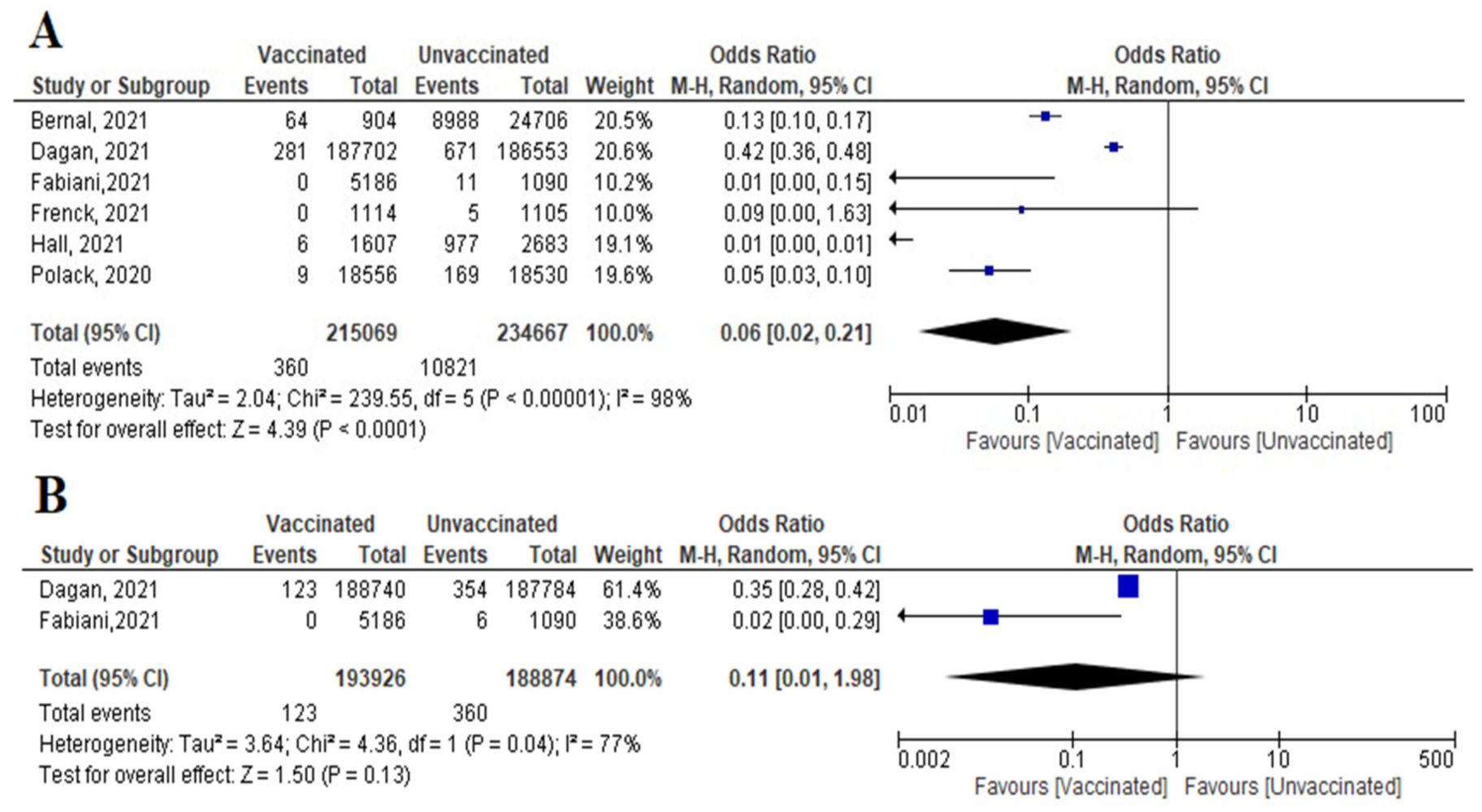
6.4.1. Cases Reported within 7 days of 2nd Dose
- Six studies reported COVID-19 cases within 7 days of vaccination; two were RCTs and four were observational studies. The number of cases reported decreased significantly among the vaccinated versus non-vaccinated population (OR = 0.06 (95% CI, 0.02–0.21), I2 = 98%). We conducted a meta-regression to explain this substantial heterogeneity, using the type of vaccine and country as predictors [mRNA and non-mRNA]. The output of the developed meta-regression model explained 100.0% of the heterogeneity (variability in our data); [mRNA type, β = −3.33, p = 0.028; country UK, β = −3.7, p = 0.025; country USA, β = −2.03, p = 0.037: Israel was the reference country] (Figure 9A). Meaning that vaccination using mRNA decreases the incidence of SARS-CoV-2 infection by 3.33 compared to other vaccines, and vaccination in developed countries like UK and USA decreased the incidence of infection by 3.7 and 2.03 times less than the reference country, respectively.
- Incidence of symptomatic SARS-CoV-2 infection did not decrease significantly among vaccinated compared to unvaccinated (OR = 0.11 [95% CI, 0.01–1.98], I2 = 77%), p = 0.13 (Figure 9B).
- No studies reported asymptomatic cases within 7 days of the 2nd dose.
- Main findings: COVID-19 vaccination decreased incidence of SARS-CoV-2 infection within 7 days of 2nd dose. The main predictor of vaccine effectiveness/efficacy were the country where the vaccine was supplied and the type of vaccine.
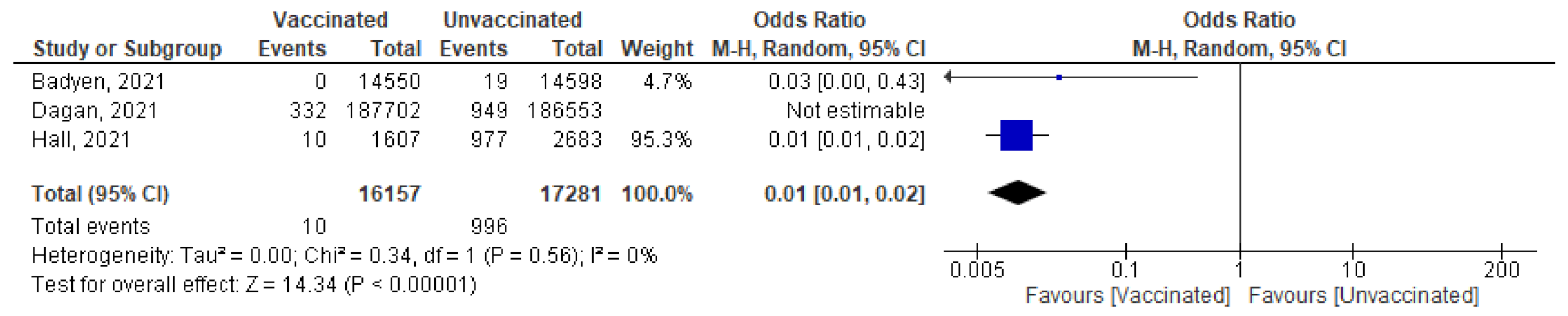
6.4.2. Cases within 14 Days of 2nd Dose
- Three studies highlighted the number of new cases (symptomatic and asymptomatic) reported within 14 days of 2nd dose. Baden et al. [37] reported no cases among 14,550 vaccinated subjects compared to 19 cases out of 14,598 unvaccinated subjects. Dagan et al. [46] reported 332 confirmed cases out of 187,702 vaccinated subjects and 949 cases out of 186,553 unvaccinated subjects. Hall et al. [50] reported 10 cases out of 1607 vaccinated subjects and 977 cases out of 2683 unvaccinated subjects. Pooling of these findings showed significant reduction in incidence of reported cases (OR = 0.05 [95% CI, 0.0–1.34], I2 = 99%). After conducting leave-one-out sensitivity analysis, the study by Dagan et al. [46] was omitted, the vaccine was effective in reducing the number of cases reported within 14 days of the 2nd dose (OR = 0.01 [95% CI, 0.01–0.02], I2 = 0%), p < 0.001 (Figure 10).
- No studies reported asymptomatic or symptomatic cases within 14 days of 2nd dose.
- Main findings: incidence of SARS-CoV-2 infection within 14 days of the 2nd dose decreased significantly after vaccination.
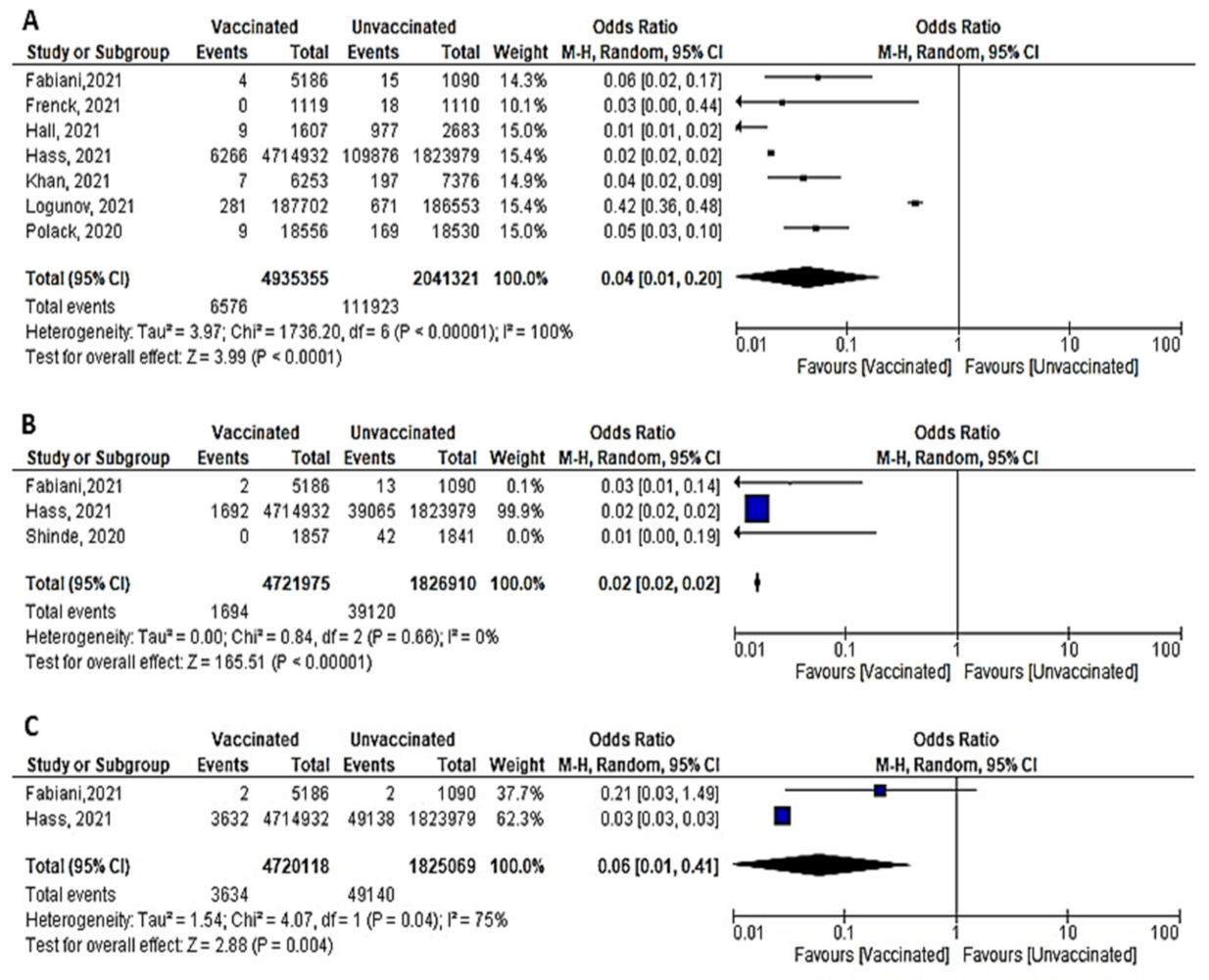
6.4.3. Cases Reported 7 Days after 2nd Dose
- Among 4,935,355 vaccinated subjects, the number of confirmed SARS-CoV-2 infection (symptomatic and asymptomatic) 7 days after 2nd dose was 6576, while 111,923 confirmed cases were reported among 2,041,321 unvaccinated subjects. The OR was 0.04 [95% CI, 0.01–0.2], I2=100, p < 0.001, indicating that COVID-19 vaccines had a protective effect. We were able to explain 100% of this heterogeneity by conducting meta-regression analysis. Vaccine type and country were significant predictors of response; [non-mRNA vaccine (β = 2.99, p = 0.0001): mRNA is the reference type, country UK (β = –0.75, p = 0.038), country USA (β = 0.8, p = 0.02); reference country was Israel] (Figure 11A). In this case, non-mRNA (the highest OR) vaccine and vaccination in the USA increased the risk of SARS-CoV-2 infection by 2.99 and 0.8 times, respectively. On the other hand, vaccination in the UK decreased risk of SARS-CoV-2 infection by 0.75 comparing to reference country (Israel).
- COVID-19 vaccines were effective in preventing symptomatic COVID-19 infection 7 days after the 2nd dose (OR = 0.02 [95% CI, 0.02–0.02], I2 = 0%), p < 0.001 (Figure 11B).
- Two studies (recruiting 4,720,118 vaccinated subjects and 1,825,069 unvaccinated subjects) assessed asymptomatic SARS-CoV-2 infection after 7 days of vaccination. Fabini et al. [47] diagnosed two cases among vaccinated and non-vaccinated subjects with a statistically non-significant odds ratio (OR = 0.21, [95% CI, 0.03–1.49]). Meanwhile, Hass et al. [49] reported 3632 asymptomatic cases among vaccinated subjects and 49,138 among unvaccinated subjects (OR = 0.03, [95% CI, 0.03–0.03]) (Figure 11C). Vaccination was protective against asymptomatic infection 7 days after the 2nd dose (OR = 0.06 [95% CI, 0.01–0.41), I2 = 75%), p = 0.04.
- Main findings: incidence of SARS-CoV-2 (symptomatic and asymptomatic) reported 7 days after the 2nd dose decreased significantly.
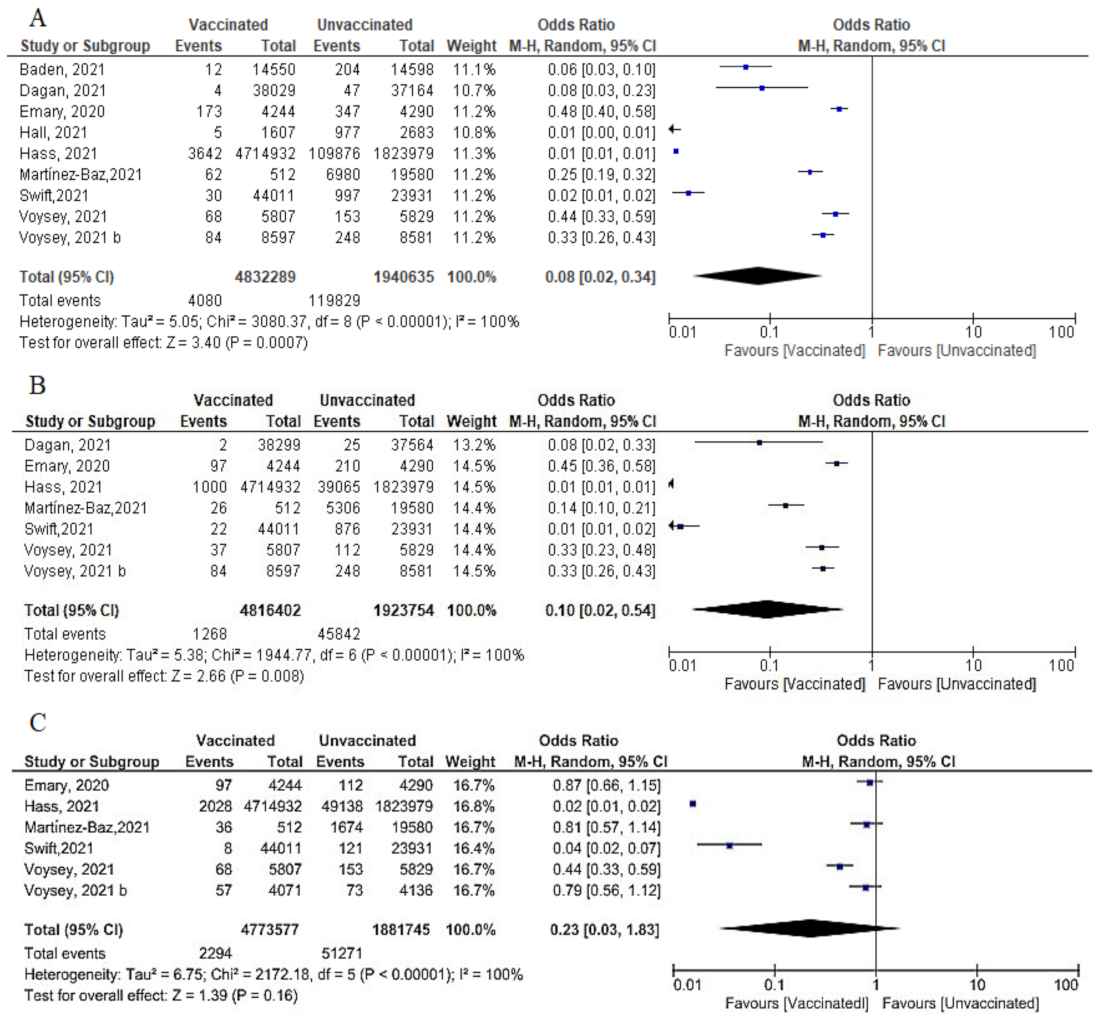
6.4.4. Cases Reported 14 Days after 2nd Dose
- We pooled nine studies that addressed the total number of symptomatic or asymptomatic cases confirmed 14 days after the 2nd dose of vaccination. The total number of cases was 4080 out of 4,832,289 vaccinated subjects and 119,829 out of 1,940,635 unvaccinated subjects. The vaccine had a protective effect against getting SARS-CoV-2 infection 14 days after the 2nd dose (OR = 0.08, [95% CI, 0.02, 0.34], I2 = 100%), p < 0.001. Thus, we carried out a meta-regression analysis to understand the main predictors of this heterogeneity. Again, the type of vaccine and the country were responsible for 88.21% of the heterogeneity; [non-mRNA vaccine β = 3.519, p = 0.004; country Spain β = 2.6256, p = 0.028: Israel was the reference country) (Figure 1A).
- Seven studies reported the incidence of symptomatic infection 14 days after the 2nd dose. Vaccination had a protective effect against symptomatic infection. The odds among vaccinated subjects was (0.10 [95% CI, 0.02–0.54], I2 = 100%), p < 0.001. To further explain this substantial heterogeneity, we performed meta-regression analysis that explained 100% of the heterogeneity using type of vaccine and country as predictors; [non-mRNA type, β = 3.16, p = 0.0044; country Spain β = 2.7, p = 0.0048; Israel is the reference country] (Figure 12B).
- Regarding asymptomatic cases after 14 days of the 2nd dose, 2294 of 4,773,577 vaccinated subjects compared to 51,271 of 1,881,745 unvaccinated subjects developed asymptomatic infection. (OR = 0.23 [95% CI, 0.03–1.83], I2 = 100%.), p < 0.001. Meta-regression was performed and explain 96.29% of heterogeneity, using type of vaccine and the country as predictors, [non-mRNA type, β = 2.9, p = 0.033; country Spain β = 3.96, p = 0.016: reference Israel]. (Figure 12C).
- Main findings: incidence of SARS-CoV-2 cases (total cases and asymptomatic cases) two weeks after the 2nd dose was significantly reduced.
6.4.5. Cases Reported 7–14 Days after 2nd Dose
6.4.6. All Cases Reported after the 2nd Dose
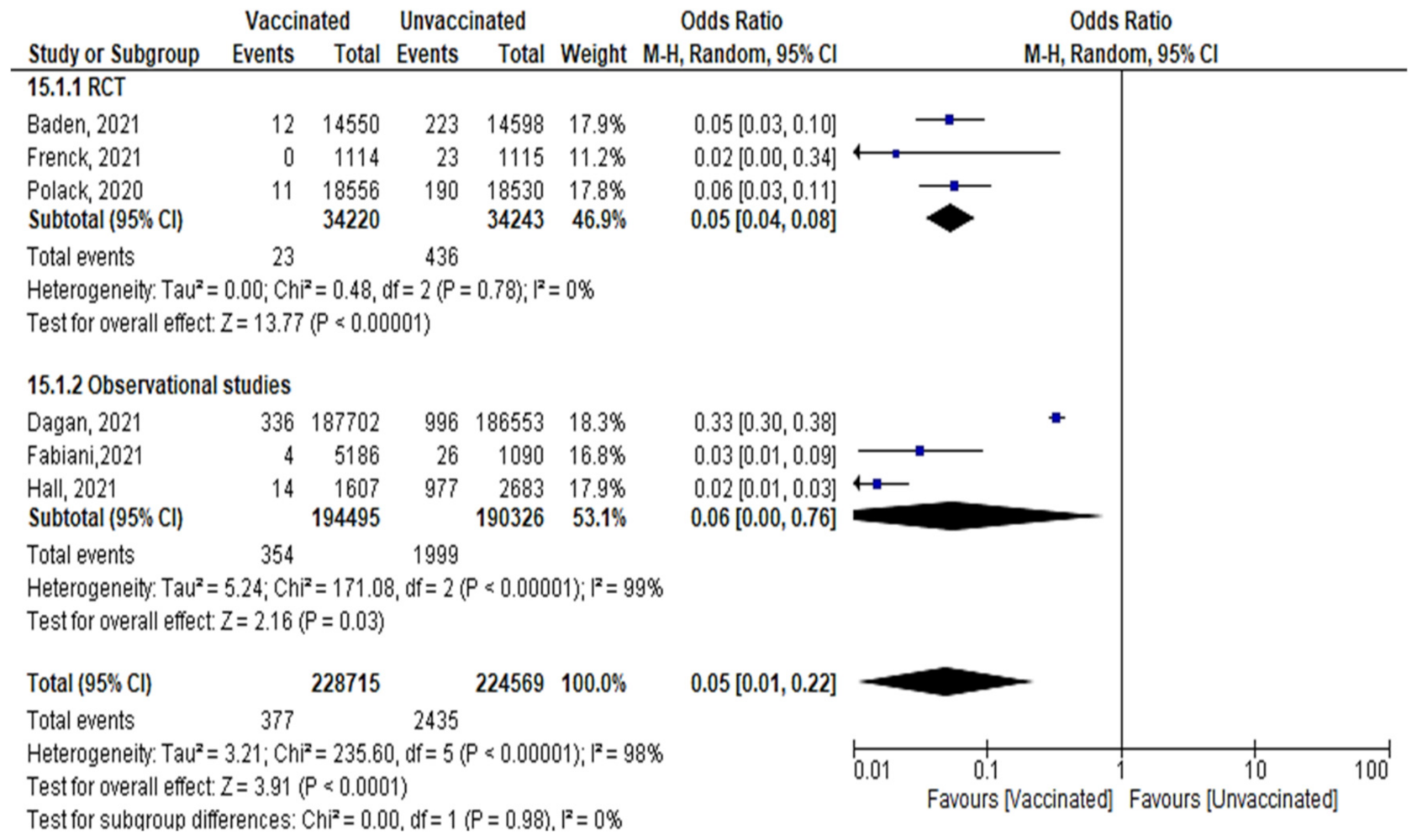
- A total of 377 confirmed cases out of 228,715 vaccinated subjects, and 2435 cases out of 224,569 unvaccinated subjects were reported after the 2nd dose, (OR = 0.179 [95% CI, 0.15–0.19], I2 = 98%). The test for subgroup differences suggested that there was no statistically significant subgroup effect (p = 0.98), meaning that the type of study did not significantly modify the efficacy and effectiveness of vaccination. Vaccination decreased the number of cases regardless of the study design, although the protective effect was greater in RCT than in observational studies. There was no heterogeneity between the results of the RCT studies, while the heterogeneity of observational studies was I2 = 99% (Figure 13).
- Main findings: vaccination against SARS-CoV-2 decreased incidence of infection after the 2nd dose regardless of the duration.
7. Discussion
7.1. Mortality and Severe COVID-19
7.2. Infection after Vaccination
7.3. Strengths and Limitations
8. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- De Wit, E.; van Doremalen, N.; Falzarano, D.; Munster, V.J. SARS and MERS: Recent insights into emerging coronaviruses. Nat. Rev. Microbiol. 2016, 14, 523–534. [Google Scholar] [CrossRef]
- Memish, Z.A.; Perlman, S.; Van Kerkhove, M.D.; Zumla, A. Middle East respiratory syndrome. Lancet 2020, 395, 1063–1077. [Google Scholar] [CrossRef]
- World Health Organization. Epidemic and Pandemic-Prone Diseases: MERS Situation Update. Available online: http://www.emro.who.int/pandemic-epidemic-diseases/mers-cov/mers-situation-update-december-2019.html (accessed on 3 August 2021).
- Mubarak, A.; Alturaiki, W.; Hemida, M.G. Middle East Respiratory Syndrome Coronavirus (MERS-CoV): Infection, Immunological Response, and Vaccine Development. J. Immunol. Res. 2019, 2019, 6491738. [Google Scholar] [CrossRef] [PubMed]
- Roper, R.L.; Rehm, K.E. SARS vaccines: Where are we? Expert Rev. Vaccines 2009, 8, 887–898. [Google Scholar] [CrossRef] [PubMed]
- Shinde, V.; Bhikha, S.; Hoosain, Z.; Archary, M.; Bhorat, Q.; Fairlie, L.; Lalloo, U.; Masilela, M.S.L.; Moodley, D.; Hanley, S.; et al. Efficacy of NVX-CoV2373 COVID-19 Vaccine against the B.1.351 Variant. N. Engl. J. Med. 2021, 384, 1899–1909. [Google Scholar] [CrossRef]
- Adney, D.; Wang, L.; van Doremalen, N.; Shi, W.; Zhang, Y.; Kong, W.-P.; Miller, M.; Bushmaker, T.; Scott, D.; de Wit, E.; et al. Efficacy of an Adjuvanted Middle East Respiratory Syndrome Coronavirus Spike Protein Vaccine in Dromedary Camels and Alpacas. Viruses 2019, 11, 212. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hilgenfeld, R.; Peiris, M. From SARS to MERS: 10 years of research on highly pathogenic human coronaviruses. Antivir. Res. 2013, 100, 286–295. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Stratton, C.W.; Tang, Y.W. Outbreak of pneumonia of unknown etiology in Wuhan, China: The mystery and the miracle. J. Med. Virol. 2020, 92, 401. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef] [Green Version]
- Vaninov, N. In the eye of the COVID-19 cytokine storm. Nat. Rev. Immunol. 2020, 20, 277. [Google Scholar] [CrossRef]
- Worldometer. COVID-19 Coronavirus Pandemic. Available online: https://www.worldometers.info/coronavirus/ (accessed on 1 January 2022).
- World Health Organization. Estimating Mortality from COVID-19. 2020. Available online: https://www.who.int/publications/i/item/WHO-2019-nCoV-Sci-Brief-Mortality-2020.1 (accessed on 2 January 2022).
- Pilishvili, T.; Fleming-Dutra, K.E.; Farrar, J.L.; Gierke, R.; Mohr, N.M.; Talan, D.A.; Krishnadasan, A.; Harland, K.K.; Smithline, H.A.; Hou, P.C.; et al. Interim Estimates of Vaccine Effectiveness of Pfizer-BioNTech and Moderna COVID-19 Vaccines Among Health Care Personnel—33 U.S. Sites, January–March 2021. MMWR. Morb. Mortal. Wkly. Rep. 2021, 70, 753–758. [Google Scholar] [CrossRef]
- World Health Organization. COVID-19 Vaccine Tracker and Landscape. Available online: https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccines (accessed on 2 January 2022).
- Chen, W.-H.; Strych, U.; Hotez, P.J.; Bottazzi, M.E. The SARS-CoV-2 Vaccine Pipeline: An Overview. Curr. Trop. Med. Rep. 2020, 7, 61–64. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- World Health Organization. Draft Landscape of COVID-19 Candidate Vaccines. Available online: https://www.who.int/docs/default-source/a-future-for-children/novel-coronavirus_landscape_covid-19.pdf?sfvrsn=4d8bd201_1 (accessed on 2 January 2022).
- Tanne, J.H. COVID-19: FDA approves Pfizer-BioNTech vaccine in record time. BMJ 2021, 374, n2096. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration. FDA Authorizes Pfizer-BioNTech COVID-19 Vaccine for Emergency Use in Children 5 through 11 Years of Age. Available online: https://www.fda.gov/news-events/press-announcements/fda-authorizes-pfizer-biontech-covid-19-vaccine-emergency-use-children-5-through-11-years-age (accessed on 6 January 2022).
- Africa CDC. COVID-19 Vaccination. Available online: https://africacdc.org/covid-19-vaccination/ (accessed on 2 January 2022).
- Covidvax. Live COVID-19 Vaccination Tracker Asia. Available online: https://covidvax.live/continent/asia (accessed on 2 January 2022).
- The New New York Times. Covid Vaccinations Tracker. Available online: https://www.nytimes.com/interactive/2021/world/covid-vaccinations-tracker.html (accessed on 2 January 2022).
- European Centre for Disease Prevention and Control. COVID-19 Vaccine Tracker. Available online: https://qap.ecdc.europa.eu/public/extensions/COVID-19/vaccine-tracker.html#uptake-tab (accessed on 2 January 2022).
- Funk, C.D.; Laferrière, C.; Ardakani, A. A Snapshot of the Global Race for Vaccines Targeting SARS-CoV-2 and the COVID-19 Pandemic. Front. Pharmacol. 2020, 11, 937. [Google Scholar] [CrossRef]
- Abdou, M.S.; Kheirallah, K.A.; Aly, M.O.; Ramadan, A.; Elhadi, Y.A.M.; Elbarazi, I.; Deghidy, E.A.; El Saeh, H.M.; Salem, K.M.; Ghazy, R.M. The coronavirus disease 2019 (COVID-19) vaccination psychological antecedent assessment using the Arabic 5c validated tool: An online survey in 13 Arab countries. PLoS ONE 2021, 16, e0260321. [Google Scholar] [CrossRef]
- National Health Service. Coronavirus (COVID-19) Vaccines Side Effects and Safety. Available online: https://www.nhs.uk/conditions/coronavirus-covid-19/coronavirus-vaccination/safety-and-side-effects/ (accessed on 25 December 2021).
- World Health Organization. Vaccine Efficacy, Effectiveness and Protection. Available online: https://www.who.int/news-room/feature-stories/detail/vaccine-efficacy-effectiveness-and-protection (accessed on 12 December 2021).
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Ann. Intern. Med. 2009, 151, 264–269. [Google Scholar] [CrossRef] [Green Version]
- Higgins, J.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions; Wiley: Hoboken, NJ, USA, 2009; Volume 5. [Google Scholar]
- Sadoff, J.; Gray, G.; Vandebosch, A.; Cárdenas, V.; Shukarev, G.; Grinsztejn, B.; Goepfert, P.A.; Truyers, C.; Fennema, H.; Spiessens, B.; et al. Safety and Efficacy of Single-Dose Ad26.COV2.S Vaccine against COVID-19. N. Engl. J. Med. 2021, 384, 2187–2201. [Google Scholar] [CrossRef]
- World Health Organization. WHO COVID-19 Case Definition. Available online: https://www.who.int/publications/i/item/WHO-2019-nCoV-Surveillance_Case_Definition-2020.2 (accessed on 28 December 2021).
- World Health Organization. COVID-19 Clinical Management: Living Guidance. Available online: https://apps.who.int/iris/handle/10665/338882 (accessed on 12 December 2021).
- Vandenbroucke, J.P.; Brickley, E.B.; Vandenbroucke-Grauls, C.M.J.E.; Pearce, N. A Test-Negative Design with Additional Population Controls Can Be Used to Rapidly Study Causes of the SARS-CoV-2. Epidemiology 2020, 31, 836–843. [Google Scholar] [CrossRef]
- Cochrane. Revised Cochrane Risk-of-Bias Tool for Randomized Trials (RoB 2). Available online: https://methods.cochrane.org/bias/resources/rob-2-revised-cochrane-risk-bias-tool-randomized-trials (accessed on 21 December 2021).
- National Institution of Health. Study Quality Assessment Tools. Available online: https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools (accessed on 18 May 2021).
- Al Kaabi, N.; Zhang, Y.; Xia, S.; Yang, Y.; Al Qahtani, M.M.; Abdulrazzaq, N.; Al Nusair, M.; Hassany, M.; Jawad, J.S.; Abdalla, J.; et al. Effect of 2 Inactivated SARS-CoV-2 Vaccines on Symptomatic COVID-19 Infection in Adults: A Randomized Clinical Trial. JAMA 2021, 326, 35–45. [Google Scholar] [CrossRef]
- Baden, L.R.; El Sahly, H.M.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; Diemert, D.; Spector, S.A.; Rouphael, N.; Creech, C.B.; et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N. Engl. J. Med. 2021, 384, 403–416. [Google Scholar] [CrossRef]
- Emary, K.R.W.; Golubchik, T.; Aley, P.K.; Ariani, C.V.; Angus, B.; Bibi, S.; Blane, B.; Bonsall, D.; Cicconi, P.; Charlton, S.; et al. Efficacy of ChAdOx1 nCoV-19 (AZD1222) vaccine against SARS-CoV-2 variant of concern 202012/01 (B.1.1.7): An explora-tory analysis of a randomised controlled trial. Lancet 2021, 397, 1351–1362. [Google Scholar] [CrossRef]
- Frenck, R.W.; Klein, N.P.; Kitchin, N.; Gurtman, A.; Absalon, J.; Lockhart, S.; Perez, J.L.; Walter, E.B.; Senders, S.; Bailey, R.; et al. Safety, Immunogenicity, and Efficacy of the BNT162b2 COVID-19 Vaccine in Adolescents. N. Engl. J. Med. 2021, 385, 239–250. [Google Scholar] [CrossRef] [PubMed]
- Logunov, D.Y.; Dolzhikova, I.V.; Shcheblyakov, D.V.; Tukhvatulin, A.I.; Zubkova, O.V.; Dzharullaeva, A.S.; Kovyrshina, A.V.; Lubenets, N.L.; Grousova, D.M.; Erokhova, A.S.; et al. Safety and efficacy of an rAd26 and rAd5 vector-based heterol-ogous prime-boost COVID-19 vaccine: An interim analysis of a randomised controlled phase 3 trial in Russia. Lancet 2021, 397, 671–681. [Google Scholar] [CrossRef]
- Madhi, S.A.; Baillie, V.; Cutland, C.L.; Voysey, M.; Koen, A.L.; Fairlie, L.; Padayachee, S.D.; Dheda, K.; Barnabas, S.L.; Bhorat, Q.E.; et al. Efficacy of the ChAdOx1 nCoV-19 COVID-19 Vaccine against the B.1.351 Variant. N. Engl. J. Med. 2021, 384, 1885–1898. [Google Scholar] [CrossRef]
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Pérez Marc, G.; Moreira, E.D.; Zerbini, C.; et al. Safety and Efficacy of the BNT162b2 mRNA COVID-19 Vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef]
- Voysey, M.; Clemens, S.A.C.; Madhi, S.A.; Weckx, L.Y.; Folegatti, P.M.; Aley, P.K.; Angus, B.; Baillie, V.L.; Barnabas, S.L.; Bhorat, Q.E.; et al. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: An interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet 2021, 397, 99–111. [Google Scholar] [CrossRef]
- Voysey, M.; Costa Clemens, S.A.; Madhi, S.A.; Weckx, L.Y.; Folegatti, P.M.; Aley, P.K.; Angus, B.; Baillie, V.L.; Barnabas, S.L.; Bhorat, Q.E.; et al. Single-dose administration and the influence of the timing of the booster dose on immunogenicity and ef-ficacy of ChAdOx1 nCoV-19 (AZD1222) vaccine: A pooled analysis of four randomised trials. Lancet 2021, 397, 881–891. [Google Scholar] [CrossRef]
- Britton, A.; Jacobs Slifka, K.M.; Edens, C.; Nanduri, S.A.; Bart, S.M.; Shang, N.; Harizaj, A.; Armstrong, J.; Xu, K.; Ehrlich, H.Y.; et al. Effectiveness of the Pfizer-BioNTech COVID-19 Vaccine Among Residents of Two Skilled Nursing Facilities Experiencing COVID-19 Outbreaks—Connecticut, December 2020–February 2021. MMWR. Morb. Mortal. Wkly. Rep. 2021, 70, 396–401. [Google Scholar] [CrossRef]
- Dagan, N.; Barda, N.; Kepten, E.; Miron, O.; Perchik, S.; Katz, M.A.; Hernán, M.A.; Lipsitch, M.; Reis, B.; Balicer, R.D. BNT162b2 mRNA COVID-19 Vaccine in a Nationwide Mass Vaccination Setting. N. Engl. J. Med. 2021, 384, 1412–1423. [Google Scholar] [CrossRef]
- Fabiani, M.; Ramigni, M.; Gobbetto, V.; Mateo-Urdiales, A.; Pezzotti, P.; Piovesan, C. Effectiveness of the Comirnaty (BNT162b2, BioNTech/Pfizer) vaccine in preventing SARS-CoV-2 infection among healthcare workers, Treviso province, Veneto region, Italy, 27 December 2020 to 24 March 2021. Eurosurveillance 2021, 26, 2100420. [Google Scholar] [CrossRef]
- Gras-Valentí, P.; Chico-Sánchez, P.; Algado-Sellés, N.; Jiménez-Sepúlveda, N.J.; Gómez-Sotero, I.L.; Fuster-Pérez, M.; Carta-gena-Llopis, L.; Sánchez-Valero, M.; Cerezo-Milán, P.; Martínez-Tornero, I.; et al. Effectiveness of the first dose of BNT162b2 vaccine to preventing COVID-19 in healthcare personnel. Rev. Esp. Salud Publica 2021, 95, 319–325. [Google Scholar] [CrossRef]
- Haas, E.J.; Angulo, F.J.; McLaughlin, J.M.; Anis, E.; Singer, S.R.; Khan, F.; Brooks, N.; Smaja, M.; Mircus, G.; Pan, K.; et al. Impact and effectiveness of mRNA BNT162b2 vaccine against SARS-CoV-2 infections and COVID-19 cases, hospitalisations, and deaths following a nationwide vaccination campaign in Israel: An observational study using national surveillance data. Lancet 2021, 397, 1819–1829. [Google Scholar] [CrossRef]
- Hall, V.J.; Foulkes, S.; Saei, A.; Andrews, N.; Oguti, B.; Charlett, A.; Wellington, E.; Stowe, J.; Gillson, N.; Atti, A.; et al. COVID-19 vaccine coverage in health-care workers in England and effectiveness of BNT162b2 mRNA vaccine against infec-tion (SIREN): A prospective, multicentre, cohort study. Lancet 2021, 397, 1725–1735. [Google Scholar] [CrossRef]
- Khan, N.; Mahmud, N. Effectiveness of SARS-CoV-2 Vaccination in a Veterans Affairs Cohort of Patients with Inflammatory Bowel Disease with Diverse Exposure to Immunosuppressive Medications. Gastroenterology 2021, 161, 827–836. [Google Scholar] [CrossRef]
- Lopez Bernal, J.; Andrews, N.; Gower, C.; Robertson, C.; Stowe, J.; Tessier, E.; Simmons, R.; Cottrell, S.; Roberts, R.; O’Doherty, M.; et al. Effectiveness of the Pfizer-BioNTech and Oxford-AstraZeneca vaccines on COVID-19 related symptoms, hospital ad-missions, and mortality in older adults in England: Test negative case-control study. BMJ. 2021, 373, n1088. [Google Scholar] [CrossRef]
- Martínez-Baz, I.; Miqueleiz, A.; Casado, I.; Navascués, A.; Trobajo-Sanmartín, C.; Burgui, C.; Guevara, M.; Ezpeleta, C.; Castilla, J. Effectiveness of COVID-19 vaccines in preventing SARS-CoV-2 infection and hospitalisation, Navarre, Spain, January to April 2021. Eurosurveillance 2021, 26, 2100438. [Google Scholar] [CrossRef]
- Menni, C.; Klaser, K.; May, A.; Polidori, L.; Capdevila, J.; Louca, P.; Sudre, C.H.; Nguyen, L.H.; Drew, D.A.; Merino, J.; et al. Vaccine side-effects and SARS-CoV-2 infection after vaccination in users of the COVID Symptom Study app in the UK: A prospective observational study. Lancet Infect. Dis. 2021, 21, 939–949. [Google Scholar] [CrossRef]
- Swift, M.D.; Breeher, L.E.; Tande, A.J.; Tommaso, C.P.; Hainy, C.M.; Chu, H.; Murad, M.H.; Berbari, E.F.; Virk, A. Effec-tiveness of Messenger RNA Coronavirus Disease 2019 (COVID-19) Vaccines Against Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Infection in a Cohort of Healthcare Personnel. Clin. Infect. Dis. 2021, 73, e1376–e1379. [Google Scholar] [CrossRef]
- Tenforde, M.W.; Olson, S.M.; Self, W.H.; Talbot, H.K.; Lindsell, C.J.; Steingrub, J.S.; Shapiro, N.I.; Ginde, A.A.; Douin, D.J.; Prekker, M.E.; et al. Effectiveness of Pfizer-BioNTech and Moderna Vaccines Against COVID-19 Among Hospitalized Adults Aged ≥65 Years—United States, January–March 2021. MMWR. Morb. Mortal. Wkly. Rep. 2021, 70, 674–679. [Google Scholar] [CrossRef]
- Vasileiou, E.; Simpson, C.R.; Shi, T.; Kerr, S.; Agrawal, U.; Akbari, A.; Bedston, S.; Beggs, J.; Bradley, D.; Chuter, A.; et al. Interim findings from first-dose mass COVID-19 vaccination roll-out and COVID-19 hospital admissions in Scotland: A na-tional prospective cohort study. Lancet 2021, 397, 1646–1657. [Google Scholar] [CrossRef]
- McGill University. COVID19 Vaccine Tracker. Available online: https://covid19.trackvaccines.org/country/egypt/ (accessed on 21 December 2021).
- Ritchie, H.; Mathieu, E.; Rodés-Guirao, L.; Appel, C.; Giattino, C.; Ortiz-Ospina, E.; Hasell, J.; Macdonald, B.; Beltekian, D.; Roser, M. Coronavirus Pandemic (COVID-19). Available online: https://ourworldindata.org/covid-vaccinations (accessed on 7 January 2022).
- World Health Organization. Vaccine Inequity Undermining Global Economic Recovery. Available online: https://www.who.int/news/item/22-07-2021-vaccine-inequity-undermining-global-economic-recovery (accessed on 2 January 2022).
- Griffin, J.B.; Haddix, M.; Danza, P.; Fisher, R.; Koo, T.H.; Traub, E.; Gounder, P.; Jarashow, C.; Balter, S. SARS-CoV-2 infections and hospitalizations among persons aged≥ 16 years, by vaccination status—Los Angeles County, California, May 1–July 25, 2021. Morb. Mortal. Wkly. Rep. 2021, 70, 1170. [Google Scholar] [CrossRef]
- Havers, F.P.; Pham, H.; Taylor, C.A.; Whitaker, M.; Patel, K.; Anglin, O.; Kambhampati, A.K.; Milucky, J.; Zell, E.; Chai, S.J. COVID-19-associated hospitalizations among vaccinated and unvaccinated adults ≥18 years–COVID-NET, 13 states, January 1–July 24, 2021. medRxiv 2021. [Google Scholar] [CrossRef]
- Scobie, H.M.; Johnson, A.G.; Suthar, A.B.; Severson, R.; Alden, N.B.; Balter, S.; Bertolino, D.; Blythe, D.; Brady, S.; Cadwell, B. Monitoring incidence of COVID-19 cases, hospitalizations, and deaths, by vaccination status—13 US jurisdictions, April 4–July 17, 2021. Morb. Mortal. Wkly. Rep. 2021, 70, 1284. [Google Scholar] [CrossRef]
- Fiolet, T.; Kherabi, Y.; MacDonald, C.-J.; Ghosn, J.; Peiffer-Smadja, N. Comparing COVID-19 vaccines for their characteristics, efficacy and effectiveness against SARS-CoV-2 and variants of concern: A narrative review. Clin. Microbiol. Infect. 2021, 28, 202–221. [Google Scholar] [CrossRef]
- Hasan, S.A.W. Interim Statement on Booster Doses for COVID-19 Vaccination. Available online: https://www.who.int/news/item/22-12-2021-interim-statement-on-booster-doses-for-covid-19-vaccination---update-22-december-2021 (accessed on 2 January 2022).
- Rotshild, V.; Hirsh-Raccah, B.; Miskin, I.; Muszkat, M.; Matok, I. Comparing the clinical efficacy of COVID-19 vaccines: A systematic review and network meta-analysis. Sci. Rep. 2021, 11, 22777. [Google Scholar] [CrossRef]
- Andrews, N.; Tessier, E.; Stowe, J.; Gower, C.; Kirsebom, F.; Simmons, R.; Gallagher, E.; Thelwall, S.; Groves, N.; Dabrera, G. Duration of Protection against Mild and Severe Disease by COVID-19 Vaccines. N. Engl. J. Med. 2022, 386, 340–350. [Google Scholar] [CrossRef]
- Alagoz, O.; Sethi, A.K.; Patterson, B.W.; Churpek, M.; Alhanaee, G.; Scaria, E.; Safdar, N. The impact of vaccination to control COVID-19 burden in the United States: A simulation modeling approach. PLoS ONE 2021, 16, e0254456. [Google Scholar] [CrossRef]
- Speiser, D.E.; Bachmann, M.F. COVID-19: Mechanisms of Vaccination and Immunity. Vaccines 2020, 8, 404. [Google Scholar] [CrossRef]
- Pormohammad, A.; Zarei, M.; Ghorbani, S.; Mohammadi, M.; Razizadeh, M.H.; Turner, D.L.; Turner, R.J. Efficacy and Safety of COVID-19 Vaccines: A Systematic Review and Meta-Analysis of Randomized Clinical Trials. Vaccines 2021, 9, 467. [Google Scholar] [CrossRef]
- Francis, A.I.; Ghany, S.; Gilkes, T.; Umakanthan, S. Review of COVID-19 vaccine subtypes, efficacy and geographical dis-tributions. Postgrad. Med. J. 2021. [Google Scholar] [CrossRef]
- Olliaro, P.; Torreele, E.; Vaillant, M. COVID-19 vaccine efficacy and effectiveness-the elephant (not) in the room. Lancet Microbe 2021, 2, e279–e280. [Google Scholar] [CrossRef]
- Africa Centers for Diseases Control and Prevention. Responding to COVID-19 in Africa: Finding the Balance (Part IV) and Calls to action. Available online: https://africacdc.org/download/responding-to-covid-19-in-africa-finding-the-balance-part-iv-and-calls-to-action/ (accessed on 4 January 2022).
- Centers for Disease Control and Prevention. COVID-19 Vaccine Effectiveness Research. Available online: https://www.cdc.gov/vaccines/covid-19/effectiveness-research/protocols.html (accessed on 7 January 2022).
- Thompson, M.G.; Burgess, J.L.; Naleway, A.L.; Tyner, H.L.; Yoon, S.K.; Meece, J.; Olsho, L.E.; Caban-Martinez, A.J.; Fowlkes, A.; Lutrick, K. Interim estimates of vaccine effectiveness of BNT162b2 and mRNA-1273 COVID-19 vaccines in preventing SARS-CoV-2 infection among health care personnel, first responders, and other essential and frontline workers—eight US locations, December 2020–March 2021. Morb. Mortal. Wkly. Rep. 2021, 70, 495. [Google Scholar] [CrossRef] [PubMed]
- Pilishvili, T.; Gierke, R.; Fleming-Dutra, K.E.; Farrar, J.L.; Mohr, N.M.; Talan, D.A.; Krishnadasan, A.; Harland, K.K.; Smithline, H.A.; Hou, P.C. Effectiveness of mRNA COVID-19 vaccine among US health care personnel. N. Engl. J. Med. 2021, 385, e90. [Google Scholar] [CrossRef]
- Krause, P.R.; Fleming, T.R.; Peto, R.; Longini, I.M.; Figueroa, J.P.; Sterne, J.A.; Cravioto, A.; Rees, H.; Higgins, J.P.; Boutron, I. Considerations in boosting COVID-19 vaccine immune responses. Lancet 2021, 398, 1377–1380. [Google Scholar] [CrossRef]
- Feikin, D.; Higdon, M.M.; Abu-Raddad, L.J.; Andrews, N.; Araos, R.; Goldberg, Y.; Groome, M.; Huppert, A.; O’Brien, K.; Smith, P.G. Duration of Effectiveness of Vaccines against SARS-CoV-2 Infection and COVID-19 Disease: Results of a Systematic Review and Meta-Regression. 2021. Available online: https://papers.ssrn.com/sol3/papers.cfm?abstract_id=3961378 (accessed on 2 January 2022).
- Ghazy, R.M.; Taha, S.H.N.; Elhadi, Y.A.M. Letter from Egypt. Respirology 2022, 27, 242–244. [Google Scholar] [CrossRef]
| Author, Year | Country | Study Design | Study Population (Criteria) | Inclusion/Exclusion Criteria | Primary and Secondary Outcomes | Type of Vaccine/No of Doses | Time Points of Analysis | Adjusted Vaccine Efficacy/Effectiveness | |
|---|---|---|---|---|---|---|---|---|---|
| 1st Dose | 2nd Dose | ||||||||
| Baden, 2021 [37] | USA | RCT | Sample size = (30,420) Intervention = (14,550) Control = (14,598) Mean age = (51.4 years) Sex = (47.3% F) | Inclusion: -Age ≥ 18 years. -No known history of SARS-CoV-2. -Absence of SARS-CoV-2–binding antibodies. | Prevention of COVID-19 illness ≥ 14 days after the 2nd dose. Prevention of severe COVID-19 or efficacy of the vaccine at preventing COVID-19 after a single dose or at preventing COVID-19 according to a secondary (CDC, less restrictive case | Moderna/2 doses | 14 days after the 1st dose | 94.10% for symptomatic infection | |
| Polack, 2021 [42] | USA | RCT | Sample size = (37,706) | Inclusion: -Age ≥ 16 years, healthy or had stable chronic medical conditions -No medical history of COVID-19 -Did not receive immunosuppressive therapy -Had immunocompromising condition. | Prevention of COVID-19 illness ≥ 7 days after the 2nd dose. Efficacy in participants ± evidence of prior infection | Pfizer–BioNTech/2 doses | ≥7 days after the 2nd dose | 94.6% | |
| Voysey, 2021a [43] | UK, Brazil, and South Africa | RCT | Sample size = (17,178) Intervention = (8597) Control = (8581) | Inclusion: - Individuals ≥ 18 years | Virologically confirmed symptomatic COVID-19 disease more than 14 days after the 2nd dose. COVID-19 infection at least 22 days after the 1st dose | ChAdOx1 nCoV-19/AstraZeneca/2 doses | 3 months after the 1st dose and 14 days after the 2nd dose | 76.0% | 66.7% |
| Shinde, 2020 [6] | Saudi Arabia | RCT | Sample size = (4387) Intervention = (2199) Control = (2188)Mean age = (32.0 years) Sex = (43% F) | Inclusion: -Age > 18–84 years, without human immunodeficiency virus infection or a subgroup aged 18–64 years with medically stable HIV. Exclusion: -Pregnancy -Receipt of immunosuppressive therapy autoimmune or immunodeficiency disease -A history of confirmed or suspected COVID-19, and SARS-CoV-2 infection as confirmed on a nucleic acid amplification test (NAAT). | Safety and vaccine efficacy against laboratory-confirmed symptomatic COVID-19 at 7 days or more after the 2nd dose among participants without previous SARS-CoV-2 infection | Novavax/2 doses | ≥7 days after the 2nd dose | 60.1% | |
| Emary, 2021 [38] | UK | RCT | Sample size = (8534) Intervention = (4244) Control = (4290) Sex = (59% F) | Inclusion: -Age ≥ 18 years who were enrolled in phase 2/3 vaccine efficacy studies in the United Kingdom (UK), and who were randomly assigned (1:1) to receive ChAdOx1 nCoV-19 or a meningococcal conjugate control (MenACWY) vaccine, -people who received 2 doses of the intervention. Exclusion: -Single-dose recipients were excluded. -Cases were excluded if they occurred <15 days after the 2nd dose of vaccine or occurred in participants who were not seronegative on a SARS-CoV-2 N protein assay at baseline. | Symptomatic COVID-19 disease, defined as a positive NAAT result on an upper airway swab in a participant with at least one symptom, including cough, fever of 37.8 °C or higher, shortness of breath, anosmia, or ageusia. The efficacy analysis included symptomatic COVID-19 in seronegative participants with a NAAT positive swab > 14 days after the 2nd dose of vaccine | ChAdOx1 nCoV-19/AstraZeneca/2 doses | ≥7 days after the 2nd dose | 70.4% for B.1.1.7 and 81.5% for non-B.1.1.7 lineages | |
| Logunov, 2021 [40] | Russia | RCT | Sample size = (19,866 Intervention = (14,964) Control = (4902) [46] Mean age (45·3 years) Sex (38.9% F) | Inclusion: -Age ≥ 18 years -Negative HIV, hepatitis B and C, and syphilis test results. Exclusion: -known history of SARS-CoV-2 -Positive drug and alcohol tests at screening visit -History of vaccine-induced reactions. -Pregnancy or breastfeeding. -The active form of a disease caused by HIV, syphilis, or hepatitis B or C. | Confirmed COVID-19 by PCR from day 21 after receiving the 1st dose | Gam-COVID-Vac/2 doses | ≥7 days after the 1st dose. | 91.6% | |
| Kaabi, 2021 [36] | UAE and Bahrain | RCT | Sample size = (40,382) (13,459 Received SARS-CoV-2WIV04 and 13,465 received HB02) Control = (13, 458) Mean age = (36.1 years) Sex = (15.6% F) | Inclusion: -Age ≥ 18 years -Non-pregnant -With self-ability to understand the study procedures and sign the informed consent form. Exclusion: -Confirmed acute cases of SARS-CoV-2 infection -a medical history of SARS, MERS virus infection -with severe chronic illness and other circumstances judged by investigators | Symptomatic laboratory-confirmed COVID-19 case that occurred ≥ 14 days after 2nd dose | SARS-CoV-2 WIV04/2 doses and HB02/2 doses | ≥7 days after the 2nd dose | 72.8%for SARS-CoV-2 WIV04 and 78.1% for HB02 | |
| Frenck, 2021 [39] | Multinational | RCT | Sample size = (2260) Intervention = (1131) Control = (1129) Sex = (49.9% F) | Inclusion: -Age 12–15 years -Healthy or had stable pre-existing disease (hepatitis B, hepatitis C, or HIV. Exclusion: -History of SARS-CoV-2 infection -Had immunocompromising or immunodeficiency disorder, or treatment with immunosuppressive therapy. | Safety objectives included the assessment of local or systemic reactogenicity events, immunogenicity assessments (SARS-CoV-2 serum neutralization assay, and receptor-binding domain (RBD). The efficacy of BNT162b2 against confirmed COVID-19 with an onset 7 or more days after dose 2 | BNT162b2/2 doses | ≥7 days after the 2nd dose | 100% | |
| Sadoff, 2021 [30] | Multinational | RCT | Sample size = (39,321) Intervention = (19,630) Control = (19,691) | Inclusion: -Body Mass Index (BMI) <30 kg/m2, -Healthy or had stable pre-existing disease (including hepatitis B, hepatitis C, or HIV infection). Exclusion: -History of anaphylaxis or other serious adverse drug reaction (ADR) to vaccines or their excipients. -has an abnormal function of the immune system resulting from clinical conditions -Persons with a previous clinical or virologic COVID-19 diagnosis or SARS-CoV-2 infection | Vaccine efficacy against moderate to severe–critical COVID- 2019) with an onset ≥14 days and ≥28 days after administration | Ad26.COV2. S/1 dose | After the 1st dose | 66.9% | |
| Dagan, 2021 [46] | Israel | Observational study | Sample size = (71,152) | Inclusion: -Age ≥ 16 Y -No known history of SARS-CoV-2 -A member of the health care organization during the previous year. -No Clalit membership (health service organization) -Unmapped place of residence -Being health care workers, and residence in a long-term care facility. | Documented SARS-CoV-2 infection, symptomatic COVID-19, hospital admission for COVID-19, and death from COVID-19 | BNT162b2/2 doses | days 14 through 20 (after 1st dose. 7 days after the 2nd dose | 57% for symptomatic infection, 74% for hospitalization | 94% for symptomatic infection, 87% for hospitalization |
| Martínez-Baz, 2021 [53] | Spain | Prospective cohort | Sample size = (20,961) (801 received Comirnaty, 524 (Vaxzevria and 56 Moderna vaccine) | Inclusion: -Age ≥ 18 years covered by the Navarre Health Service. -Had been close contacts of laboratory-confirmed -COVID-19 cases from January to April 2021. Exclusion: -Close contacts with a positive test for SARS-CoV-2 before January 2021 -Nursing home residents and those who did not complete the testing protocol | Preventing severe SARS-CoV-2infections, symptomatic confirmed SARS-CoV-2 infections, and COVID-19 hospitalizations | Comirnaty/2 doses, Vaxzervria/2 doses and Moderna/2 doses | After the 1st and 2nd doses | 35% for infection, 42% for symptomatic infection, and 72% for hospitalization | 66% for infections and 82% for symptomatic infection, and 95% for hospitalization |
| Swift, 2021 [55] | USA | Retrospective cohort | Sample size = (71,152) Cohort = (47,221)Controls = (23,931) | Inclusion: -Health care workers (HCWs). Exclusion: -Individuals with a positive molecular assay prior to 1 Januaray 2021 or inactive employment status. | Vaccine effectiveness in the subpopulation of health care personnel (HCP) reached through employer vaccination programs | BNT162b2/2 doses and Moderna/2 doses | After the 1st and 2nd doses | 96% | |
| Tenforde, 2021 [56] | USA | Case control | Sample size = (417) Cases = (187) Control = (230) | Inclusion: -Age ≥ 65 years -SARS-CoV-2 negative Exclusion: -Participants with unverified vaccination status -Vaccination with Janssen COVID-19 vaccine. | Effectiveness of Pfizer and Moderna vaccines among adults aged >65 years | BNT162b2/2 doses and Moderna/2 doses | After the 1st and 2nd doses | 64% | 94% |
| Khan, 2021 [51] | USA | Retrospective cohort | Sample size (14,697) Cohort (7321) Controls (7376) | Inclusion: -Age ≥ 18 years confirmed negative SARS-CoV-2 infection. -who were taking an inflammatory bowel disease (IBD) medication, and who had ≥ 6 months of Veterans Health Administration outpatient visit data prior to the index date. Exclusion: -Janssen COVID-19 vaccine. | Time to SARS-CoV-2 infection, determined by PCR testing. All-cause mortality and severe SARS-CoV-2 infection | BNT162b2/2 doses and Moderna/2 doses | >7 days after the 2nd dose | 80.4% | |
| Bernal, 2021 [52] | UK | Case control | Sample size = (156,930) Cases = (138,869) Controls = (18,061) | Inclusion: -Age ≥ 70 years and older who reported symptoms of COVID-19 between 8 December 2020and 19 February 2021 and were successfully linked to vaccination data in the National Immunization Management System. Exclusion: -Those with a previous positive PCR or antibody test result at any time | Confirmed symptomatic SARS-CoV-2 infections. Admissions to hospital for COVID-19, and deaths with COVID-19 | BNT162b2/2 doses and ChAdOx1-S)/2 doses | ≥42 days after the 1st and 14 after the 2nd dose | 60–70% for BNT162b2 and 60–75% for ChAdOx1-S after the 1st dose | 89% For BNT162b2 |
| Fabiani, 2021 [47] | Italy | Retrospective cohort | Sample size = (6423) Cases = (5333) Controls = (1090) | Inclusion: -HCWs. -HCWs infected with COVID-19 before the vaccination campaign, -HCWs working outside hospitals and district outpatient centers, support staff, and administrative staff. | Effectiveness of the vaccine | BNT162b2/2 doses | 14–21 days After 1st dose and ≥7 days after 2nd dose | 84% | 95% |
| Gras-Valentí, 2021 [48] | Spain | Case control | Sample size = (268) Cases = (70) Controls = (198) | Inclusion: -HCP with suspected COVID-19 and HCP close contacts of COVID-19 cases were included and PCR tested for COVID-19; those with positive PCR were considered cases, and those with negative PCR were considered controls. | Effectiveness of a dose of (BNT162b2) after 12 days of administration in health personnel of a department of Health | BNT162b2/1 dose | 12 days After the 1st dose | 52.6%, the adjusted vaccine efficacy was 74.6% | |
| Hall, 2021 [50] | UK | Prospective cohort | Sample size = (1,106,905) Cohort = (396,318) Control = (710,587) Median age = (46·1 years) Sex (84% F) | Inclusion: -HCWs, support staff, and administrative staff (aged ≥18 years) working at hospital sites participating in SIREN who could provide written informed consent and anticipated remaining engaged in follow-up for 12 months. Exclusion: -Had no PCR tests after 7 December 2020, or had insufficient PCR and antibody to complete cohort assignment | Vaccinated participants for the vaccine coverage analysis and SARS-CoV-2 infection confirmed by a PCR test for the vaccine effectiveness analysis | BNT162b2/2 doses | After the 1st and 2nd doses | 70% | 85% |
| Haas 2021 [49] | Israel | Observational study | Sample size = (6,538,911) Vaccinated = (4,714,932) Controls = (1,823,979) | Inclusion: -Residents of Israel who aged ≥16 years. Exclusion: -Had received only one dose or had received two doses of BNT162b2 and fewer than 7 days had passed since the 2nd dose. | COVID-19 infection Hospitalization, severe infection, symptomatic infection, and death | BNT162b/2 doses | >7 days after the 2nd dose | 91.5% for asymptomatic infection, 97.0% for symptomatic COVID-19 and 96.7% against COVID-19-related death | |
| Pilishvili, 2021 [14] | USA | Cases-control | Sample size = (1843) Cases = (623) Controls = (1220) | Inclusion: -HCWs with positive SARS-CoV-2 test symptoms were enrolled as case-patients, and HCWs with negative SARS-CoV-2 PCR test results, regardless of symptoms, were eligible for enrolment as controls. Exclusion: -Positive SARS-CoV-2 PCR or antigen-based test result >60 days earlier | BNT162b2/2 doses and Moderna/2 doses | 14 days after the 1st dose and ≥7 days after the 2nd dose | 82% | 94% | |
| Vasileiou, 2021 [57] | UK | Prospective cohort | Sample size = (4,409,588) Cohort = (1,331,993) Controls = (3,077,595) | Inclusion: -Received a single dose of the vaccine between 8 December 2020 and 22 February 2021, with maximum follow-up time censored on 22 February 2021. Exclusion: -Previously tested positive with real-time reverse transcription-PCR (RT- PCR) for SARS-CoV-2 infection. | Any hospital admission with COVID-19 as the main cause, or hospital admission within 28 days of a positive RT-PCR test for SARS-CoV-2 infection from 8 December 2020 to 22 February 2021 | BNT162b2/2 doses and ChAdOx1-S)/2 doses | 28–34 days post the 1st dose vaccination. | 91% for BNT162b2 and 88% for the ChAdOx1 vaccine | |
| Britton, 2021 [45] | USA | Retrospective cohort | Sample size (463) (304 had received 2 doses of vaccine, 72 had received 1 dose and 87 had not received any doses). | Inclusion: -Residents of skilled nursing facilities were included if they were admitted at either facility during one or more rounds of facility-wide SARS-CoV-2 testing during the week before or any time after their facility’s first vaccination clinic. | BNT162b2/2 doses | After the 1st dose | 63% | ||
| Madhi, 2021 [41] | South Africa | RCT | Sample size: 2026 Intervention (1013) Control (1013) Median age: 30 years Sex: 56.5% M | HIV-negative adults aged 18 to >65 years. HIV-positive adults at screening, previous or current laboratory- confirmed COVID-19, a history of anaphylaxis in relation to vaccination, and morbid obesity (BMI ≥ 40). | Safety and efficacy of the vaccine against laboratory-confirmed symptomatic COVID-19 more than 14 days after the 2nd dose | ChAdOx1/2 doses | 14 days after the 2nd dose | 21.9% | |
| Menni, Klaser, 2021 [54] | UK | Prospective observational study | Sample Size (67,293 users received BNT162b2 and 36,329 received ChAdOx1 nCoV-19 Compared to 464,356 unvaccinated users) | Not reported. | The proportion of app users reporting adverse effects within 8 days after vaccination. Infection rates in individuals after receiving the 1st dose of either the vaccines | BNT162b2/2 doses and ChAdOx1-S)/2 doses | 8 days after the 1st dose | 64% for BNT162b2 and 52% for ChAdOx1 | |
| Voysey, 2021b [44] | UK, Brazil, and South Africa | RCT | Sample size COV002 (UK; LD/SD; n = 2741) COV002 (UK; SD/SD; n = 4807) COV003 (Brazil; all SD/SD; n = 4088) | Inclusion: -The first symptom or first NAAT-positive result was on or before the data cutoff date (4 November 2020). Exclusion: -Seropositive participants at baseline or those who had no baseline result were excluded. -NAAT-positive swabs within 14 days after the 2nd dose. | Virologically confirmed, symptomatic/asymptomatic COVID-19 infection, in COV002 in UK tested weekly by self-administered nose and throat swab from 1 week after 1st dose of vaccination using kits provided Adverse events | COV002: LD (2.2 × 10¹⁰ viral particles), SD (5 × 10¹⁰ viral particles) COV003/2 doses of the vaccine at a dose of 3.5–6.5 × 10¹⁰ viral particles with administration up to 12 weeks apart (target 4 weeks) | >14 days after their 2nd dose | 70.4% | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghazy, R.M.; Ashmawy, R.; Hamdy, N.A.; Elhadi, Y.A.M.; Reyad, O.A.; Elmalawany, D.; Almaghraby, A.; Shaaban, R.; Taha, S.H.N. Efficacy and Effectiveness of SARS-CoV-2 Vaccines: A Systematic Review and Meta-Analysis. Vaccines 2022, 10, 350. https://doi.org/10.3390/vaccines10030350
Ghazy RM, Ashmawy R, Hamdy NA, Elhadi YAM, Reyad OA, Elmalawany D, Almaghraby A, Shaaban R, Taha SHN. Efficacy and Effectiveness of SARS-CoV-2 Vaccines: A Systematic Review and Meta-Analysis. Vaccines. 2022; 10(3):350. https://doi.org/10.3390/vaccines10030350
Chicago/Turabian StyleGhazy, Ramy Mohamed, Rasha Ashmawy, Noha Alaa Hamdy, Yasir Ahmed Mohammed Elhadi, Omar Ahmed Reyad, Dina Elmalawany, Abdallah Almaghraby, Ramy Shaaban, and Sarah Hamed N. Taha. 2022. "Efficacy and Effectiveness of SARS-CoV-2 Vaccines: A Systematic Review and Meta-Analysis" Vaccines 10, no. 3: 350. https://doi.org/10.3390/vaccines10030350
APA StyleGhazy, R. M., Ashmawy, R., Hamdy, N. A., Elhadi, Y. A. M., Reyad, O. A., Elmalawany, D., Almaghraby, A., Shaaban, R., & Taha, S. H. N. (2022). Efficacy and Effectiveness of SARS-CoV-2 Vaccines: A Systematic Review and Meta-Analysis. Vaccines, 10(3), 350. https://doi.org/10.3390/vaccines10030350










