Metabolic Basis and Clinical Evidence for Skin Lightening Effects of Thiol Compounds
Abstract
:1. Introduction
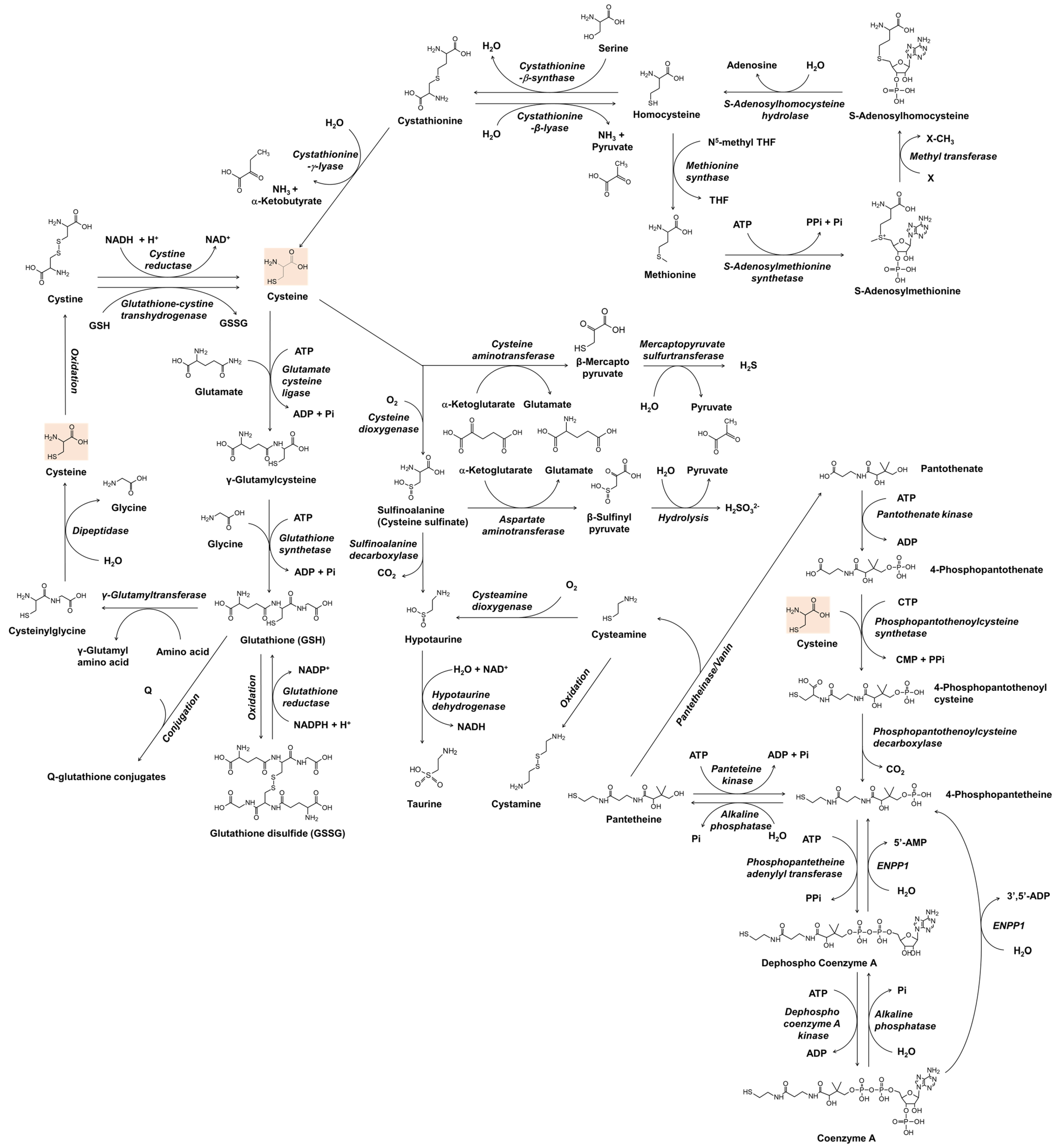
2. The Metabolism of Cysteine and Related Thiol Compounds
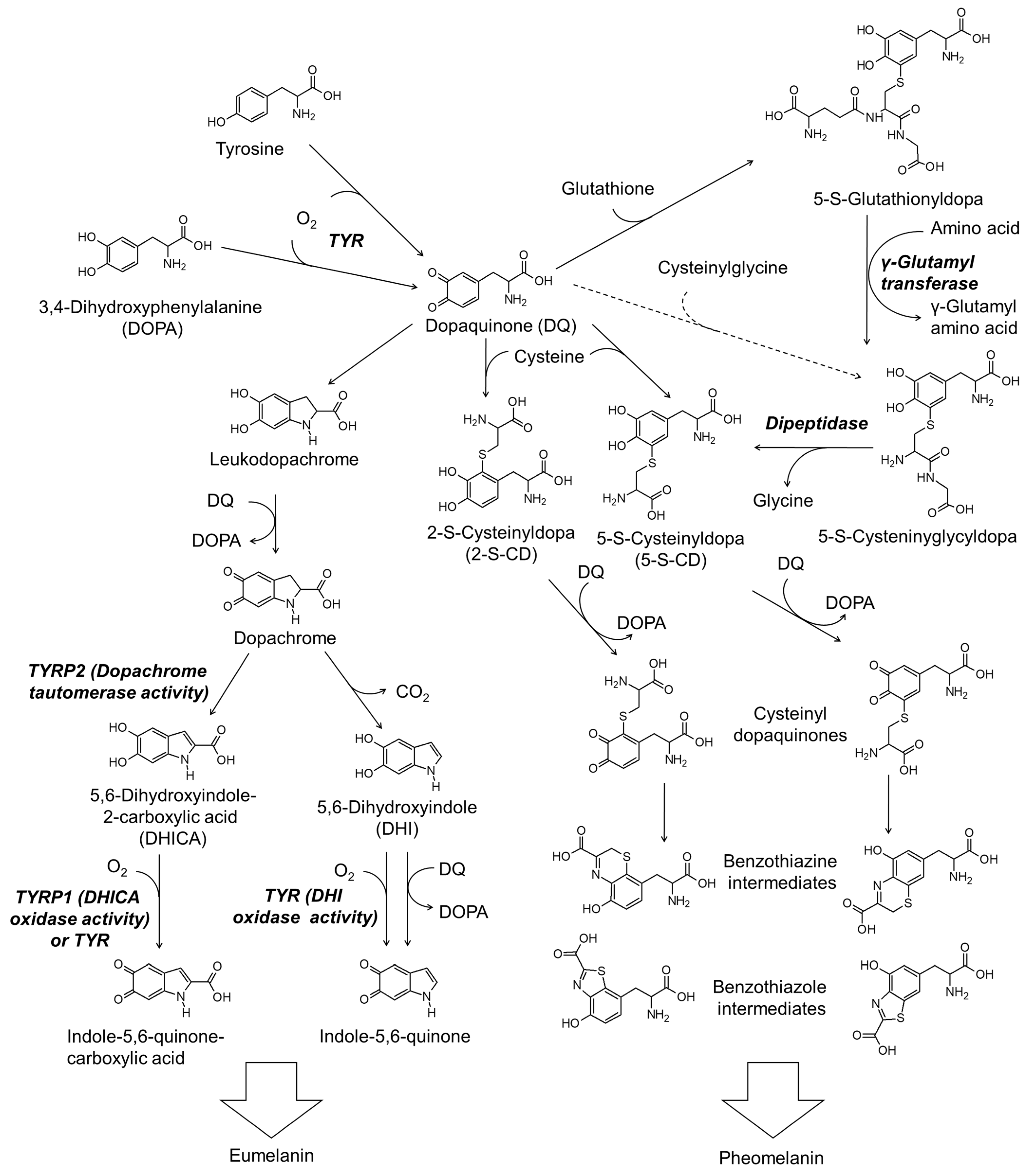
3. The Role of Thiol Compounds in Melanin Synthesis
4. TYR-Inhibitory and Antimelanogenic Effects of Various Thiol Compounds
4.1. TYR-Inhibitory Effects In Vitro
4.2. Anti-Melanogenic Effects in Cells
5. Clinical Trials on the Skin Lightening Efficacy of Thiol Compounds
5.1. Glutathione and Glutathione Disulfide
5.2. Cysteine, N-Acetyl Cysteine and Cystine
5.3. Cysteamine
6. Discussion
7. Conclusions
Funding
Conflicts of Interest
References
- Slominski, A.; Kim, T.K.; Brozyna, A.A.; Janjetovic, Z.; Brooks, D.L.; Schwab, L.P.; Skobowiat, C.; Jozwicki, W.; Seagroves, T.N. The role of melanogenesis in regulation of melanoma behavior: Melanogenesis leads to stimulation of HIF-1alpha expression and HIF-dependent attendant pathways. Arch. Biochem. Biophys. 2014, 563, 79–93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Slominski, R.M.; Zmijewski, M.A.; Slominski, A.T. The role of melanin pigment in melanoma. Exp. Dermatol. 2015, 24, 258–259. [Google Scholar] [CrossRef] [Green Version]
- Costin, G.E.; Hearing, V.J. Human skin pigmentation: Melanocytes modulate skin color in response to stress. FASEB J. 2007, 21, 976–994. [Google Scholar] [CrossRef]
- Schiaffino, M.V. Signaling pathways in melanosome biogenesis and pathology. Int. J. Biochem. Cell Biol. 2010, 42, 1094–1104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamaguchi, Y.; Beer, J.Z.; Hearing, V.J. Melanin mediated apoptosis of epidermal cells damaged by ultraviolet radiation: Factors influencing the incidence of skin cancer. Arch. Dermatol. Res. 2008, 300 (Suppl. 1), S43–S50. [Google Scholar] [CrossRef] [PubMed]
- Boo, Y.C. Emerging Strategies to Protect the Skin from Ultraviolet Rays Using Plant-Derived Materials. Antioxidants 2020, 9, 637. [Google Scholar] [CrossRef]
- Rose, P.T. Pigmentary disorders. Med. Clin. N. Am. 2009, 93, 1225–1239. [Google Scholar] [CrossRef]
- Ganju, P.; Nagpal, S.; Mohammed, M.H.; Nishal Kumar, P.; Pandey, R.; Natarajan, V.T.; Mande, S.S.; Gokhale, R.S. Microbial community profiling shows dysbiosis in the lesional skin of Vitiligo subjects. Sci. Rep. 2016, 6, 18761. [Google Scholar] [CrossRef] [Green Version]
- Spritz, R.A.; Andersen, G.H. Genetics of Vitiligo. Dermatol. Clin. 2017, 35, 245–255. [Google Scholar] [CrossRef] [Green Version]
- Ganceviciene, R.; Liakou, A.I.; Theodoridis, A.; Makrantonaki, E.; Zouboulis, C.C. Skin anti-aging strategies. Dermatoendocrinology 2012, 4, 308–319. [Google Scholar] [CrossRef] [Green Version]
- Ramos-e-Silva, M.; Celem, L.R.; Ramos-e-Silva, S.; Fucci-da-Costa, A.P. Anti-aging cosmetics: Facts and controversies. Clin. Dermatol. 2013, 31, 750–758. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Gao, J. The use of botanical extracts as topical skin-lightening agents for the improvement of skin pigmentation disorders. J. Investig. Dermatol. Symp. Proc. 2008, 13, 20–24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Niu, C.; Aisa, H.A. Upregulation of Melanogenesis and Tyrosinase Activity: Potential Agents for Vitiligo. Molecules 2017, 22, 1303. [Google Scholar] [CrossRef] [Green Version]
- Pillaiyar, T.; Namasivayam, V.; Manickam, M.; Jung, S.H. Inhibitors of Melanogenesis: An Updated Review. J. Med. Chem. 2018, 61, 7395–7418. [Google Scholar] [CrossRef]
- Maymone, M.B.C.; Neamah, H.H.; Secemsky, E.A.; Vashi, N.A. Correlating the Dermatology Life Quality Index and Skin Discoloration Impact Evaluation Questionnaire tools in disorders of hyperpigmentation. J. Dermatol. 2018, 45, 361–362. [Google Scholar] [CrossRef] [PubMed]
- Nautiyal, A.; Wairkar, S. Management of hyperpigmentation: Current treatments and emerging therapies. Pigment Cell Melanom. Res. 2021, 34, 1000–1014. [Google Scholar] [CrossRef] [PubMed]
- Perez-Bernal, A.; Munoz-Perez, M.A.; Camacho, F. Management of facial hyperpigmentation. Am. J. Clin. Dermatol. 2000, 1, 261–268. [Google Scholar] [CrossRef]
- Draelos, Z.D. Skin lightening preparations and the hydroquinone controversy. Dermatol. Ther. 2007, 20, 308–313. [Google Scholar] [CrossRef]
- Tse, T.W. Hydroquinone for skin lightening: Safety profile, duration of use and when should we stop? J. Dermatol. Treat. 2010, 21, 272–275. [Google Scholar] [CrossRef]
- Boo, Y.C. Mechanistic Basis and Clinical Evidence for the Applications of Nicotinamide (Niacinamide) to Control Skin Aging and Pigmentation. Antioxidants 2021, 10, 1315. [Google Scholar] [CrossRef]
- Boo, Y.C. Arbutin as a Skin Depigmenting Agent with Antimelanogenic and Antioxidant Properties. Antioxidants 2021, 10, 1129. [Google Scholar] [CrossRef] [PubMed]
- Kelm, R.C.; Zahr, A.S.; Kononov, T.; Ibrahim, O. Effective lightening of facial melasma during the summer with a dual regimen: A prospective, open-label, evaluator-blinded study. J. Cosmet. Dermatol. 2020, 19, 3251–3257. [Google Scholar] [CrossRef] [PubMed]
- Raposo, G.; Marks, M.S. The dark side of lysosome-related organelles: Specialization of the endocytic pathway for melanosome biogenesis. Traffic 2002, 3, 237–248. [Google Scholar] [CrossRef] [PubMed]
- Boo, Y.C. Up- or Downregulation of Melanin Synthesis Using Amino Acids, Peptides, and Their Analogs. Biomedicines 2020, 8, 322. [Google Scholar] [CrossRef]
- Kim, J.H.; Seok, J.K.; Kim, Y.M.; Boo, Y.C. Identification of small peptides and glycinamide that inhibit melanin synthesis using a positional scanning synthetic peptide combinatorial library. Br. J. Dermatol. 2019, 181, 128–137. [Google Scholar] [CrossRef]
- Boo, Y.C.; Jo, D.J.; Oh, C.M.; Lee, S.Y.; Kim, Y.M. The First Human Clinical Trial on the Skin Depigmentation Efficacy of Glycinamide Hydrochloride. Biomedicines 2020, 8, 257. [Google Scholar] [CrossRef]
- Boo, Y.C. Human Skin Lightening Efficacy of Resveratrol and Its Analogs: From in Vitro Studies to Cosmetic Applications. Antioxidants 2019, 8, 332. [Google Scholar] [CrossRef] [Green Version]
- Boo, Y.C. p-Coumaric Acid as An Active Ingredient in Cosmetics: A Review Focusing on its Antimelanogenic Effects. Antioxidants 2019, 8, 275. [Google Scholar] [CrossRef] [Green Version]
- Cooksey, C.J.; Garratt, P.J.; Land, E.J.; Pavel, S.; Ramsden, C.A.; Riley, P.A.; Smit, N.P. Evidence of the indirect formation of the catecholic intermediate substrate responsible for the autoactivation kinetics of tyrosinase. J. Biol. Chem. 1997, 272, 26226–26235. [Google Scholar] [CrossRef] [Green Version]
- Simon, J.D.; Peles, D.; Wakamatsu, K.; Ito, S. Current challenges in understanding melanogenesis: Bridging chemistry, biological control, morphology, and function. Pigment Cell Melanoma Res. 2009, 22, 563–579. [Google Scholar] [CrossRef]
- Olivares, C.; Solano, F. New insights into the active site structure and catalytic mechanism of tyrosinase and its related proteins. Pigment Cell Melanoma Res. 2009, 22, 750–760. [Google Scholar] [CrossRef] [PubMed]
- Thody, A.J.; Higgins, E.M.; Wakamatsu, K.; Ito, S.; Burchill, S.A.; Marks, J.M. Pheomelanin as Well as Eumelanin Is Present in Human Epidermis. J. Investig. Dermatol. 1991, 97, 340–344. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Halprin, K.M.; Ohkawara, A. Glutathione and Human Pigmentation. Arch. Dermatol. 1966, 94, 355. [Google Scholar] [CrossRef] [PubMed]
- Benedetto, J.P.; Ortonne, J.P.; Voulot, C.; Khatchadourian, C.; Prota, G.; Thivolet, J. Role of Thiol Compounds in Mammalian Melanin Pigmentation.1. Reduced and Oxidized Glutathione. J. Investig. Dermatol. 1981, 77, 402–405. [Google Scholar] [CrossRef] [Green Version]
- Liu, M.J.; Prakash, C.; Nauta, A.; Siezen, R.J.; Francke, C. Computational Analysis of Cysteine and Methionine Metabolism and Its Regulation in Dairy Starter and Related Bacteria. J. Bacteriol. 2012, 194, 3522–3533. [Google Scholar] [CrossRef] [Green Version]
- Romero, L.C.; Aroca, M.A.; Laureano-Marin, A.M.; Moreno, I.; Garcia, I.; Gotor, C. Cysteine and Cysteine-Related Signaling Pathways in Arabidopsis thaliana. Mol. Plant 2014, 7, 264–276. [Google Scholar] [CrossRef] [Green Version]
- Stipanuk, M.H.; Dominy, J.E.; Lee, J.I.; Coloso, R.M. Mammalian cysteine metabolism: New insights into regulation of cysteine metabolism. J. Nutr. 2006, 136, 1652s–1659s. [Google Scholar] [CrossRef]
- Mato, J.M.; Corrales, F.J.; Lu, S.C.; Avila, M.A. S-adenosylmethionine: A control switch that regulates liver function. FASEB J. 2002, 16, 15–26. [Google Scholar] [CrossRef] [Green Version]
- Conter, C.; Fruncillo, S.; Fernandez-Rodriguez, C.; Martinez-Cruz, L.A.; Dominici, P.; Astegno, A. Cystathionine beta-synthase is involved in cysteine biosynthesis and-H2S generation in Toxoplasma gondii. Sci. Rep. 2020, 10, 1–14. [Google Scholar] [CrossRef]
- Guggenheim, S.; Flavin, M. Cystathionine Gamma-Synthase—A Pyridoxal Phosphate Enzyme Catalyzing Rapid Exchanges of Beta and Alpha Hydrogen Atoms in Amino Acids. J. Biol. Chem. 1969, 244, 6217. [Google Scholar] [CrossRef]
- Grillo, M.A.; Colombatto, S. S-adenosylmethionine and its products. Amino Acids 2008, 34, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Paul, B.D.; Sbodio, J.I.; Snyder, S.H. Cysteine Metabolism in Neuronal Redox Homeostasis. Trends Pharmacol. Sci. 2018, 39, 513–524. [Google Scholar] [CrossRef] [PubMed]
- Koppula, P.; Zhuang, L.; Gan, B.Y. Cystine transporter SLC7A11/xCT in cancer: Ferroptosis, nutrient dependency, and cancer therapy. Protein Cell 2021, 12, 599–620. [Google Scholar] [CrossRef]
- Lo, M.; Wang, Y.Z.; Gout, P.W. The x(c)(-) cystine/glutamate antiporter: A potential target for therapy of cancer and other diseases. J. Cell. Physiol. 2008, 215, 593–602. [Google Scholar] [CrossRef] [PubMed]
- Mironov, A.; Seregina, T.; Shatalin, K.; Nagornykh, M.; Shakulov, R.; Nudler, E. CydDC functions as a cytoplasmic cystine reductase to sensitize Escherichia coli to oxidative stress and aminoglycosides. Proc. Natl. Acad. Sci. USA 2020, 117, 23565–23570. [Google Scholar] [CrossRef] [PubMed]
- States, B.; Segal, S. Interrelationship of Glutathione-Cystine Transhydrogenase and Glutathione Reductase in Developing Rat Intestine. Biochem. J. 1973, 132, 623–631. [Google Scholar] [CrossRef] [Green Version]
- Bjorn-Yoshimoto, W.E.; Underhill, S.M. The importance of the excitatory amino acid transporter 3 (EAAT3). Neurochem. Int. 2016, 98, 4–18. [Google Scholar] [CrossRef] [Green Version]
- Adelmann, C.H.; Traunbauer, A.K.; Chen, B.; Condon, K.J.; Chan, S.H.; Kunchok, T.; Lewis, C.A.; Sabatini, D.M. MFSD12 mediates the import of cysteine into melanosomes and lysosomes. Nature 2020, 588, 699–704. [Google Scholar] [CrossRef]
- Poole, L.B. The basics of thiols and cysteines in redox biology and chemistry. Free Radic. Biol. Med. 2015, 80, 148–157. [Google Scholar] [CrossRef] [Green Version]
- Bansal, A.; Simon, M.C. Glutathione metabolism in cancer progression and treatment resistance. J. Cell Biol. 2018, 217, 2291–2298. [Google Scholar] [CrossRef] [Green Version]
- McBean, G.J.; Aslan, M.; Griffiths, H.R.; Torrao, R.C. Thiol redox homeostasis in neurodegenerative disease. Redox Biol. 2015, 5, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.C. Glutathione synthesis. Biochim. Biophys. Acta-Gen. Subj. 2013, 1830, 3143–3153. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Forman, H.J.; Zhang, H.Q.; Rinna, A. Glutathione: Overview of its protective roles, measurement, and biosynthesis. Mol. Asp. Med. 2009, 30, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Couto, N.; Wood, J.; Barber, J. The role of glutathione reductase and related enzymes on cellular redox homoeostasis network. Free Radic. Biol. Med. 2016, 95, 27–42. [Google Scholar] [CrossRef]
- Kennedy, L.; Sandhu, J.K.; Harper, M.E.; Cuperlovic-Culf, M. Role of Glutathione in Cancer: From Mechanisms to Therapies. Biomolecules 2020, 10, 1429. [Google Scholar] [CrossRef]
- Ballatori, N.; Krance, S.M.; Marchan, R.; Hammond, C.L. Plasma membrane glutathione transporters and their roles in cell physiology and pathophysiology. Mol. Asp. Med. 2009, 30, 13–28. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.Q.; Forman, H.J.; Choi, J. gamma-Glutamyl transpeptidase in glutathione biosynthesis. Methods Enzymol. 2005, 401, 468–483. [Google Scholar]
- Gaucher, C.; Boudier, A.; Bonetti, J.; Clarot, I.; Leroy, P.; Parent, M. Glutathione: Antioxidant Properties Dedicated to Nanotechnologies. Antioxidants 2018, 7, 62. [Google Scholar] [CrossRef] [Green Version]
- Czumaj, A.; Szrok-Jurga, S.; Hebanowska, A.; Turyn, J.; Swierczynski, J.; Sledzinski, T.; Stelmanska, E. The Pathophysiological Role of CoA. Int. J. Mol. Sci. 2020, 21, 9057. [Google Scholar] [CrossRef]
- Genschel, U. Coenzyme A biosynthesis: Reconstruction of the pathway in archaea and an evolutionary scenario based on comparative genomics. Mol. Biol. Evol. 2004, 21, 1242–1251. [Google Scholar] [CrossRef] [Green Version]
- Srinivasan, B.; Baratashvili, M.; van der Zwaag, M.; Kanon, B.; Colombelli, C.; Lambrechts, R.A.; Schaap, O.; Nollen, E.A.; Podgorsek, A.; Kosec, G.; et al. Extracellular 4’-phosphopantetheine is a source for intracellular coenzyme A synthesis. Nat. Chem. Biol. 2015, 11, 784–792. [Google Scholar] [CrossRef] [PubMed]
- Naquet, P.; Kerr, E.W.; Vickers, S.D.; Leonardi, R. Regulation of coenzyme A levels by degradation: The ‘Ins and Outs’. Prog. Lipid Res. 2020, 78, 101028. [Google Scholar] [CrossRef] [PubMed]
- Pitari, G.; Malergue, F.; Martin, F.; Philippe, J.M.; Massucci, M.T.; Chabret, C.; Maras, B.; Dupre, S.; Naquet, P.; Galland, F. Pantetheinase activity of membrane-bound Vanin-1: Lack of free cysteamine in tissues of Vanin-1 deficient mice. FEBS Lett. 2000, 483, 149–154. [Google Scholar] [CrossRef] [Green Version]
- Besouw, M.; Masereeuw, R.; van den Heuvel, L.; Levtchenko, E. Cysteamine: An old drug with new potential. Drug Discov. Today 2013, 18, 785–792. [Google Scholar] [CrossRef]
- Paul, B.D.; Snyder, S.H. Therapeutic Applications of Cysteamine and Cystamine in Neurodegenerative and Neuropsychiatric Diseases. Front. Neurol. 2019, 10, 1315. [Google Scholar] [CrossRef] [Green Version]
- Qaradakhi, T.; Gadanec, L.K.; McSweeney, K.R.; Abraham, J.R.; Apostolopoulos, V.; Zulli, A. The Anti-Inflammatory Effect of Taurine on Cardiovascular Disease. Nutrients 2020, 12, 2847. [Google Scholar] [CrossRef]
- Wen, C.Y.; Li, F.N.; Zhang, L.Y.; Duan, Y.H.; Guo, Q.P.; Wang, W.L.; He, S.P.; Li, J.Z.; Yin, Y.L. Taurine is Involved in Energy Metabolism in Muscles, Adipose Tissue, and the Liver. Mol. Nutr. Food Res. 2019, 63, 1800536. [Google Scholar] [CrossRef]
- Miyamoto, R.; Otsuguro, K.; Yamaguchi, S.; Ito, S. Contribution of cysteine aminotransferase and mercaptopyruvate sulfurtransferase to hydrogen sulfide production in peripheral neurons. J. Neurochem. 2014, 130, 29–40. [Google Scholar] [CrossRef] [Green Version]
- Cao, X.; Ding, L.; Xie, Z.Z.; Yang, Y.; Whiteman, M.; Moore, P.K.; Bian, J.S. A Review of Hydrogen Sulfide Synthesis, Metabolism, and Measurement: Is Modulation of Hydrogen Sulfide a Novel Therapeutic for Cancer? Antioxid. Redox Signal. 2019, 31, 1–38. [Google Scholar] [CrossRef]
- Stipanuk, M.H.; Ueki, I.; Dominy, J.E.; Simmons, C.R.; Hirschberger, L.L. Cysteine dioxygenase: A robust system for regulation of cellular cysteine levels. Amino Acids 2009, 37, 55–63. [Google Scholar] [CrossRef] [Green Version]
- Stipanuk, M.H. Metabolism of Sulfur-Containing Amino Acids: How the Body Copes with Excess Methionine, Cysteine, and Sulfide. J. Nutr. 2020, 150, 2494s–2505s. [Google Scholar] [CrossRef] [PubMed]
- Steinhoff, M.; Stander, S.; Seeliger, S.; Ansel, J.C.; Schmelz, M.; Luger, T. Modern aspects of cutaneous neurogenic inflammation. Arch Dermatol. 2003, 139, 1479–1488. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.; Tobin, D.J.; Shibahara, S.; Wortsman, J. Melanin pigmentation in mammalian skin and its hormonal regulation. Physiol. Rev. 2004, 84, 1155–1228. [Google Scholar] [CrossRef] [PubMed]
- Flaherty, K.T.; Hodi, F.S.; Fisher, D.E. From genes to drugs: Targeted strategies for melanoma. Nat. Rev. Cancer 2012, 12, 349–361. [Google Scholar] [CrossRef]
- Serre, C.; Busuttil, V.; Botto, J.M. Intrinsic and extrinsic regulation of human skin melanogenesis and pigmentation. Int. J. Cosmet. Sci. 2018, 40, 328–347. [Google Scholar] [CrossRef] [Green Version]
- Rzepka, Z.; Buszman, E.; Beberok, A.; Wrzesniok, D. From tyrosine to melanin: Signaling pathways and factors regulating melanogenesis. Postepy Hig. Med. Dosw. 2016, 70, 695–708. [Google Scholar] [CrossRef]
- D’Mello, S.A.; Finlay, G.J.; Baguley, B.C.; Askarian-Amiri, M.E. Signaling Pathways in Melanogenesis. Int. J. Mol. Sci. 2016, 17, 1144. [Google Scholar] [CrossRef] [Green Version]
- Yuan, X.H.; Jin, Z.H. Paracrine regulation of melanogenesis. Br. J. Dermatol. 2018, 178, 632–639. [Google Scholar] [CrossRef]
- Ito, S.; Wakamatsu, K. Chemistry of mixed melanogenesis--pivotal roles of dopaquinone. Photochem. Photobiol. 2008, 84, 582–592. [Google Scholar] [CrossRef]
- Kishida, R.; Saputro, A.G.; Kasai, H. Mechanism of dopachrome tautomerization into 5,6-dihydroxyindole-2-carboxylic acid catalyzed by Cu(II) based on quantum chemical calculations. Biochim. Biophys. Acta-Gen. Subj. 2015, 1850, 281–286. [Google Scholar] [CrossRef] [Green Version]
- Sugumaran, M. Molecular Mechanisms for Mammalian Melanogenesis—Comparison with Insect Cuticular Sclerotization. FEBS Lett. 1991, 295, 233–239. [Google Scholar] [CrossRef] [Green Version]
- Jara, J.R.; Aroca, P.; Solano, F.; Martinez, J.H.; Lozano, J.A. The role of sulfhydryl compounds in mammalian melanogenesis: The effect of cysteine and glutathione upon tyrosinase and the intermediates of the pathway. Biochim. Biophys. Acta 1988, 967, 296–303. [Google Scholar] [CrossRef]
- Ito, S. A chemist’s view of melanogenesis. Pigment Cell Res. 2003, 16, 230–236. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hansson, C.; Rorsman, H.; Rosengren, E. 5-S-Cysteinyldopa as a Substrate for Tyrosinase. Acta Dermatol. Venereol. 1980, 60, 399–402. [Google Scholar]
- Wakamatsu, K.; Ohtara, K.; Ito, S. Chemical analysis of late stages of pheomelanogenesis: Conversion of dihydrobenzothiazine to a benzothiazole structure. Pigment Cell Melanoma Res. 2009, 22, 474–486. [Google Scholar] [CrossRef]
- Ito, S.; Palumbo, A.; Prota, G. Tyrosinase-catalyzed conjugation of dopa with glutathione. Experientia 1985, 41, 960–961. [Google Scholar] [CrossRef]
- Carstam, R.; Hansson, C.; Lindbladh, C.; Rorsman, H.; Rosengren, E. Dopaquinone addition products in cultured human melanoma cells. Acta Dermatol. Venereol. 1987, 67, 100–105. [Google Scholar]
- Benathan, M.; Virador, V.; Furumura, M.; Kobayashi, N.; Panizzon, R.G.; Hearing, V.J. Co-regulation of melanin precursors and tyrosinase in human pigment cells: Roles of cysteine and glutathione. Cell. Mol. Biol. 1999, 45, 981–990. [Google Scholar]
- Sanada, H.; Suzue, R.; Nakashima, Y.; Kawada, S. Effect of thiol compounds on melanin formation by tyrosinase. Biochim. Biophys. Acta Gen. Subj. 1972, 261, 258–266. [Google Scholar] [CrossRef]
- Kahn, V. Effect of Proteins, Protein Hydrolyzates and Amino Acids on o-Dihydroxyphenolase Activity of Polyphenol Oxidase of Mushroom, Avocado, and Banana. J. Food Sci. 1985, 50, 111–115. [Google Scholar] [CrossRef]
- Kermasha, S.; Goetghebeur, M.; Monfette, A.; Metche, M.; Rovel, B. Inhibitory Effects of Cysteine and Aromatic-Acids on Tyrosinase Activity. Phytochemistry 1993, 34, 349–353. [Google Scholar] [CrossRef]
- Wang, S.F.; Oh, S.; Si, Y.X.; Wang, Z.J.; Han, H.Y.; Lee, J.; Qian, G.Y. Computational prediction of protein-protein interactions of human tyrosinase. Enzym. Res. 2012, 2012, 192867. [Google Scholar] [CrossRef] [PubMed]
- Jergil, B.; Lindbladh, C.; Rorsman, H.; Rosengren, E. Inactivation of human tyrosinase by cysteine. Protection by dopa and tyrosine. Acta Dermatol. Venereol. 1984, 64, 155–157. [Google Scholar]
- Tseng, T.S.; Tsai, K.C.; Chen, W.C.; Wang, Y.T.; Lee, Y.C.; Lu, C.K.; Don, M.J.; Chang, C.Y.; Lee, C.H.; Lin, H.H.; et al. Discovery of Potent Cysteine-Containing Dipeptide Inhibitors against Tyrosinase: A Comprehensive Investigation of 20 × 20 Dipeptides in Inhibiting Dopachrome Formation. J. Agric. Food Chem. 2015, 63, 6181–6188. [Google Scholar] [CrossRef]
- Hsiao, N.W.; Tseng, T.S.; Lee, Y.C.; Chen, W.C.; Lin, H.H.; Chen, Y.R.; Wang, Y.T.; Hsu, H.J.; Tsai, K.C. Serendipitous Discovery of Short Peptides from Natural Products as Tyrosinase Inhibitors. J. Chem. Inf. Model. 2014, 54, 3099–3111. [Google Scholar] [CrossRef]
- Luisi, G.; Stefanucci, A.; Zengin, G.; Dimmito, M.P.; Mollica, A. Anti-Oxidant and Tyrosinase Inhibitory In Vitro Activity of Amino Acids and Small Peptides: New Hints for the Multifaceted Treatment of Neurologic and Metabolic Disfunctions. Antioxidants 2018, 8, 7. [Google Scholar] [CrossRef] [Green Version]
- Lee, H.K.; Ha, J.W.; Hwang, Y.J.; Boo, Y.C. Identification of L-Cysteinamide as a Potent Inhibitor of Tyrosinase-Mediated Dopachrome Formation and Eumelanin Synthesis. Antioxidants 2021, 10, 1202. [Google Scholar] [CrossRef]
- Kwon, B.S.; Haq, A.K.; Pomerantz, S.H.; Halaban, R. Isolation and sequence of a cDNA clone for human tyrosinase that maps at the mouse c-albino locus. Proc. Natl. Acad. Sci. USA 1987, 84, 7473–7477. [Google Scholar] [CrossRef] [Green Version]
- Wichers, H.J.; Recourt, K.; Hendriks, M.; Ebbelaar, C.E.; Biancone, G.; Hoeberichts, F.A.; Mooibroek, H.; Soler-Rivas, C. Cloning, expression and characterisation of two tyrosinase cDNAs from Agaricus bisporus. Appl. Microbiol. Biotechnol. 2003, 61, 336–341. [Google Scholar] [CrossRef]
- An, S.M.; Koh, J.S.; Boo, Y.C. p-coumaric acid not only inhibits human tyrosinase activity in vitro but also melanogenesis in cells exposed to UVB. Phytother. Res. 2010, 24, 1175–1180. [Google Scholar] [CrossRef]
- Mann, T.; Gerwat, W.; Batzer, J.; Eggers, K.; Scherner, C.; Wenck, H.; Stab, F.; Hearing, V.J.; Rohm, K.H.; Kolbe, L. Inhibition of Human Tyrosinase Requires Molecular Motifs Distinctively Different from Mushroom Tyrosinase. J. Investig. Dermatol. 2018, 138, 1601–1608. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- del Marmol, V.; Ito, S.; Bouchard, B.; Libert, A.; Wakamatsu, K.; Ghanem, G.; Solano, F. Cysteine deprivation promotes eumelanogenesis in human melanoma cells. J. Investig. Dermatol. 1996, 107, 698–702. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smit, N.P.M.; VanderMeulen, H.; Koerten, H.K.; Kolb, R.M.; Mommaas, A.M.; Lentjes, E.G.W.M.; Pavel, S. Melanogenesis in cultured melanocytes can be substantially influenced by L-tyrosine and L-cysteine. J. Investig. Dermatol. 1997, 109, 796–800. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qiu, L.; Zhang, M.; Sturm, R.A.; Gardiner, B.; Tonks, I.; Kay, G.; Parsons, P.G. Inhibition of melanin synthesis by cystamine in human melanoma cells. J. Investig. Dermatol. 2000, 114, 21–27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chung, B.Y.; Choi, S.R.; Moon, I.J.; Park, C.W.; Kim, Y.H.; Chang, S.E. The Glutathione Derivative, GSH Monoethyl Ester, May Effectively Whiten Skin but GSH Does Not. Int. J. Mol. Sci. 2016, 17, 629. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choi, H.R.; Kang, Y.A.; Lee, H.S.; Park, K.C. Disulfanyl peptide decreases melanin synthesis via receptor-mediated ERK activation and the subsequent downregulation of MITF and tyrosinase. Int. J. Cosmet. Sci. 2015, 38, 279–285. [Google Scholar] [CrossRef]
- Villarama, C.D.; Maibach, H.I. Glutathione as a depigmenting agent: An overview. Int. J. Cosmet. Sci. 2005, 27, 147–153. [Google Scholar] [CrossRef]
- Dilokthornsakul, W.; Dhippayom, T.; Dilokthornsakul, P. The clinical effect of glutathione on skin color and other related skin conditions: A systematic review. J. Cosmet. Dermatol. 2019, 18, 728–737. [Google Scholar] [CrossRef]
- Sonthalia, S.; Jha, A.K.; Lallas, A.; Jain, G.; Jakhar, D. Glutathione for skin lightening: A regnant myth or evidence-based verity? Dermatol. Pract. Concept. 2018, 8, 15–21. [Google Scholar] [CrossRef] [Green Version]
- Yamada, M.; Ogawa, Y.; Ikeda, T. Clinical use of synthetic glutathione (Tathione) in various skin diseases. Acta Dermatol.-Kyoto. Engl. Ed. 1967, 62, 219–223. [Google Scholar]
- Han, J.Y.; Park, S.O.; Hahm, J.H.; Kook, H.I. Clinical Effect of Glutathione (Tathione) on Melasma. Korean J. Dermatol. 1977, 15, 57. [Google Scholar]
- Arjinpathana, N.; Asawanonda, P. Glutathione as an oral whitening agent: A randomized, double-blind, placebo-controlled study. J. Dermatol. Treat. 2012, 23, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, F.; Hashizume, E.; Chan, G.P.; Kamimura, A. Skin-whitening and skin-condition-improving effects of topical oxidized glutathione: A double-blind and placebo-controlled clinical trial in healthy women. Clin. Cosmet. Investig. Dermatol. 2014, 7, 267–274. [Google Scholar] [CrossRef] [Green Version]
- Weschawalit, S.; Thongthip, S.; Phutrakool, P.; Asawanonda, P. Glutathione and its antiaging and antimelanogenic effects. Clin. Cosmet. Investig. Dermatol. 2017, 10, 147–153. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zubair, S.; Hafeez, S.; Mujtaba, G. Efficacy of intravenous glutathione vs. placebo for skin tone lightening. J. Pak. Assoc. Dermatol. 2016, 26, 177. [Google Scholar]
- Handog, E.B.; Datuin, M.S.L.; Singzon, I.A. An open-label, single-arm trial of the safety and efficacy of a novel preparation of glutathione as a skin-lightening agent in Filipino women. Int. J. Dermatol. 2016, 55, 153–157. [Google Scholar] [CrossRef]
- Duperray, J.; Sergheraert, R.; Chalothorn, K.; Tachalerdmanee, P.; Perin, F. The effects of the oral supplementation of L-Cystine associated with reduced L-Glutathione-GSH on human skin pigmentation: A randomized, double-blinded, benchmark- and placebo-controlled clinical trial. J. Cosmet. Dermatol. 2022, 21, 802–813. [Google Scholar] [CrossRef]
- Wilkes, M.; Wright, C.Y.; du Plessis, J.L.; Reeder, A. Fitzpatrick Skin Type, Individual Typology Angle, and Melanin Index in an African Population: Steps Toward Universally Applicable Skin Photosensitivity Assessments. JAMA Dermatol. 2015, 151, 902–903. [Google Scholar] [CrossRef] [Green Version]
- Adil, M.; Amin, S.S.; Mohtashim, M. N-acetylcysteine in dermatology. Indian J. Dermatol. Venereol. Leprol. 2018, 84, 652–659. [Google Scholar] [CrossRef]
- Njoo, M.D.; Menke, H.E.; Pavel, S.; Westerhof, W. N-acetylcysteine as a bleaching agent in the treatment of melasma. J. Eur. Acad. Dermatol. Venereol. 1997, 9, 86–87. [Google Scholar] [CrossRef]
- Fujiwara, Y.; Sahashi, Y.; Aritro, M.; Hasegawa, S.; Akimoto, K.; Ninomiya, S.; Sakaguchi, Y.; Seyama, Y. Effect of simultaneous administration of vitamin C, L-cysteine and vitamin E on the melanogenesis. Biofactors 2004, 21, 415–418. [Google Scholar] [CrossRef] [PubMed]
- Ahramiyanpour, N.; Saki, N.; Akbari, Z.; Shamsi-Meymandi, S.; Amiri, R.; Heiran, A. Efficacy of topical cysteamine hydrochloride in treating melasma: A systematic review. J. Cosmet. Dermatol. 2021, 20, 3593–3602. [Google Scholar] [CrossRef] [PubMed]
- Mansouri, P.; Farshi, S.; Hashemi, Z.; Kasraee, B. Evaluation of the efficacy of cysteamine 5% cream in the treatment of epidermal melasma: A randomized double-blind placebo-controlled trial. Br. J. Dermatol. 2015, 173, 209–217. [Google Scholar] [CrossRef]
- Farshi, S.; Mansouri, P.; Kasraee, B. Efficacy of cysteamine cream in the treatment of epidermal melasma, evaluating by Dermacatch as a new measurement method: A randomized double blind placebo controlled study. J. Dermatol. Treat. 2018, 29, 182–189. [Google Scholar] [CrossRef] [PubMed]
- Karrabi, M.; David, J.; Sahebkar, M. Clinical evaluation of efficacy, safety and tolerability of cysteamine 5% cream in comparison with modified Kligman’s formula in subjects with epidermal melasma: A randomized, double-blind clinical trial study. Ski. Res. Technol. 2021, 27, 24–31. [Google Scholar] [CrossRef]
- Nguyen, J.; Remyn, L.; Chung, I.Y.; Honigman, A.; Wutami, I.; Mane, S.; Wong, C.; Rodrigues, M. Evaluation of the efficacy of cysteamine cream compared to hydroquinone in the treatment of melasma: A randomised, double-blinded, trial. Australas. J. Dermatol. 2021, 62, e41–e46. [Google Scholar] [CrossRef]
- Karrabi, M.; Mansournia, M.A.; Sharestanaki, E.; Abdollahnejad, Y.; Sahebkar, M. Clinical evaluation of efficacy and tolerability of cysteamine 5% cream in comparison with tranexamic acid mesotherapy in subjects with melasma: A single-blind, randomized clinical trial study. Arch. Dermatol. Res. 2021, 313, 539–547. [Google Scholar] [CrossRef]
- Singh, S.; Singh, U.; Pandey, S.S. Increased Level of Serum Homocysteine in Vitiligo. J. Clin. Lab. Anal. 2011, 25, 110–112. [Google Scholar] [CrossRef]
- Silverberg, J.; Silverberg, N. Serum homocysteine is associated with extent of vitiligo vulgaris. J. Am. Acad. Dermatol. 2011, 64, Ab142. [Google Scholar]
- Tsai, T.Y.; Kuo, C.Y.; Huang, Y.C. Serum homocysteine, folate, and vitamin B-12 levels in patients with vitiligo and their potential roles as disease activity biomarkers: A systematic review and meta-analysis. J. Am. Acad. Dermatol. 2019, 80, 646. [Google Scholar] [CrossRef]
- Chen, J.; Zhuang, T.; Chen, J.; Tian, Y.; Yi, X.; Ni, Q.; Zhang, W.; Song, P.; Jian, Z.; Liu, L.; et al. Homocysteine induces melanocytes apoptosis via PERK-eIF2alpha-CHOP pathway in vitiligo. Clin. Sci. 2020, 134, 1127–1141. [Google Scholar] [CrossRef] [PubMed]
- Reish, O.; Townsend, D.; Berry, S.A.; Tsai, M.Y.; King, R.A. Tyrosinase Inhibition Due to Interaction of Homocyst(E)Ine with Copper—The Mechanism for Reversible Hypopigmentation in Homocystinuria Due to Cystathionine Beta-Synthase Deficiency. Am. J. Hum. Genet. 1995, 57, 127–132. [Google Scholar] [PubMed]
- Dey-Rao, R.; Sinha, A.A. Interactome analysis of gene expression profile reveals potential novel key transcriptional regulators of skin pathology in vitiligo. Genes Immun. 2016, 17, 30–45. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.J.; Boo, Y.C. Melanogenesis Promotion by 3-Deazaneplanocin A, a Specific Inhibitor of S-Adenosylhomocysteine Hydrolase, in B16/F10 Melanoma Cells. J. Soc. Cosmet. Sci. Korea 2021, 47, 107–121. [Google Scholar]
- Denat, L.; Kadekaro, A.L.; Marrot, L.; Leachman, S.A.; Abdel-Malek, Z.A. Melanocytes as instigators and victims of oxidative stress. J. Investig. Dermatol. 2014, 134, 1512–1518. [Google Scholar] [CrossRef] [Green Version]
- Abbas, K.; Qadir, M.I.; Anwar, S. The Role of Melanin in Skin Cancer. Crit. Rev. Eukaryot. Gene Expr. 2019, 29, 17–24. [Google Scholar] [CrossRef]
- Smit, N.P.M.; Nieuwpoort, F.A.; Marrot, L.; Out, C.; Poorthuis, B.; van Pelt, H.; Meunier, J.R.; Pavel, S. Increased melanogenesis is a risk factor for oxidative DNA damage—Study on cultured melanocytes and atypical nevus cells. Photochem. Photobiol. 2008, 84, 550–555. [Google Scholar] [CrossRef]
- Lu, Y.Y.; Tonissen, K.F.; Di Trapani, G. Modulating skin colour: Role of the thioredoxin and glutathione systems in regulating melanogenesis. Biosci. Rep. 2021, 41, BSR20210427. [Google Scholar] [CrossRef]
- Atallah, C.; Charcosset, C.; Greige-Gerges, H. Challenges for cysteamine stabilization, quantification, and biological effects improvement. J. Pharm. Anal. 2020, 10, 499–516. [Google Scholar] [CrossRef]
- Khan, N.U.; Ali, A.; Khan, H.; Khan, Z.U.; Ahmed, Z. Stability Studies and Characterization of Glutathione-Loaded Nanoemulsion. J. Cosmet. Sci. 2018, 69, 257–267. [Google Scholar]
- Demirkol, O.; Adams, C.; Ercal, N. Biologically important Thiols in various vegetables and fruits. J. Agric. Food Chem. 2004, 52, 8151–8154. [Google Scholar] [CrossRef] [PubMed]
- Hand, C.E.; Honek, J.F. Biological chemistry of naturally occurring thiols of microbial and marine origin. J. Nat. Prod. 2005, 68, 293–308. [Google Scholar] [CrossRef] [PubMed]
| Literature | Study Format | No. of Subjects | Tested Materials | Treatment | Key Findings |
|---|---|---|---|---|---|
| [112] | A double-blind, randomized, placebo-controlled study | 30 | Placebo capsules | The capsules were orally taken twice daily for 4 weeks. | Compared to the baseline values, the melanin indices and the number of ultraviolet (UV) spots at all six skin sites decreased consistently in subjects who received glutathione for 4 weeks. The reductions were statistically significantly greater than those receiving placebo at some skin sites. |
| 30 | Glutathione 250 mg capsules | ||||
| [113] | A randomized, double-blind, matched-pair, placebo-controlled study | 30 | A placebo lotion | Subjects applied test lotion to one side of the face and a placebo lotion to the other side twice daily for 10 weeks. | The skin melanin index was significantly lower with GSSG lotion treatment compared with placebo lotion treatment from the first week after the start of the trial through to the end of the study period (10th week). |
| Glutathione disulfide 2% lotion | |||||
| [114] | A randomized, double-blind, placebo-controlled, parallel, three-arm study | 20 | Glutathione 250 mg capsule | A capsule was orally taken daily for 12 weeks | Melanin index and UV spots were reduced in the glutathione group and the glutathione disulfide group compared to the placebo group. |
| 18 | Glutathione disulfide 250 mg | ||||
| 19 | Placebo capsule | ||||
| [115] | A placebo-controlled study | 16 | Placebo | Patients were given 2 intravenous injections (glutathione 1200 mg, ascorbic acid, hydrolyzed collagen, NaCl, and aqua) per week for 6 weeks | After 12 injections of glutathione, 6 of 16 (37.5%) subjects showed significant improvement in skin tone, whereas 3 (18.7%) subjects improved with placebo. After stopping the treatment, this improvement was gradually lost in most patients in 6 months. |
| 16 | Glutathione 1200 mg | ||||
| [116] | An open-label, single-arm clinical trial | 30 | Glutathione 500 mg lozenges | Subjects put one lozenge in the mouth against the inner cheek (buccal mucosa) until completely dissolved every morning for 8 weeks. | There was a significant decrease in melanin indices from baseline to end that became evident as early as 2 weeks. The skin lightening effect was observed both in sun-exposed and sun-protected skin. |
| [117] | A randomized, double-blind, parallel-group, benchmark- and placebo-controlled trial | 32 | Placebo | Subjects ingested tablets or capsules daily or twice daily for 12 weeks in a blinded format. | Oral supplementation of cystine plus glutathione induced a significant skin lightening after 12 weeks. This combination also induced a significant reduction in the size of facial dark spots after 6 and 12 weeks. The changes were significantly bigger than those obtained not only with placebo but also with cystine alone or glutathione alone. |
| 31 | Glutathione 250 mg | ||||
| 30 | Cystine 500 mg | ||||
| 31 | Cystine 500 mg plus glutathione 250 mg |
| Literature | Study Format | No. of Subjects | Tested Materials | Treatment | Key Findings |
|---|---|---|---|---|---|
| [123] | A randomized, double-blind placebo-controlled study | 25 | A placebo cream | Cysteamine cream or placebo was applied on the lesions once a day at bedtime over 4 months. | The mean differences between pigmented and normal skin (calculated by Mexameter) were reduced after 2 and 4 months of application of cysteamine cream. At the end of the treatment, the melasma area severity index (MASI) scores were significantly lower in the cysteamine group vs. placebo. |
| 25 | Cysteamine 5% cream | ||||
| [124] | A double-blind placebo-controlled study | 20 | A placebo cream | Cysteamine cream or placebo was applied on the lesions once a day at bedtime throughout the 4-month study period. | Cysteamine cream significantly reduced the degree of pigmentation measured by Mexameter and Dermacatch after 2 weeks compared to the pre-use and placebo-using groups. After 4 weeks, MASI and IGA scores were significantly lower in the cysteamine group compared to the placebo group. |
| 20 | Cysteamine 5% cream | ||||
| [125] | A randomized, double-blind clinical study | 25 | Modified Kligman’s formula (MKF, 4% hydroquinone, 0.05% retinoic acid, and 0.1% betamethasone) | Cysteamine cream was applied once (15 min) daily, and MKF was applied once (whole night) daily over 4 months. | The cysteamine treatment decreased the modified MASI score to a greater degree (32.3%, 51.3%) compared to MKF (23.7%, 42.3%) at 2 and 4 months, respectively, and these differences were strongly statistically significant. The differences between the MKF and cysteamine groups were not statistically significant in IGA assessments at 2 and 4 months. |
| 25 | Cysteamine 5% cream | ||||
| [126] | A randomized, double-blind trial | 5 | Cysteamine 5% cream | Cysteamine cream was applied once (15 min exposure) daily, and hydroquinone cream was applied once daily over 16 weeks. | There was a 19.7% and 39.1% reduction in modified MASI for the cysteamine group and a 39.2% and 33% reduction in the hydroquinone group at 8 and 16 weeks, respectively. The difference between groups was not statistically significant. Hydroquinone cream was better tolerated than cysteamine cream. |
| 9 | Hydroquinone 4% cream | ||||
| [127] | A single-blind randomized clinical study | 27 | Cysteamine 5% cream | Cysteamine 5% cream was applied on the melasma lesions 30 min before bed for 4 months. Tranexamic acid mesotherapy (0.05 mL) was performed every 4 weeks until 8 weeks. | The MASI scores and Dermacatch values were significantly decreased in both cysteamine (2, 4 months) and tranexamic acid mesotherapy groups (4, 8 weeks) compared with baseline. The improvement rates between the two groups were similar (cysteamine for 2 months vs. tranexamic acid mesotherapy for 4 weeks; cysteamine for 4 months vs. tranexamic acid mesotherapy for 8 weeks). |
| 27 | Tranexamic acid (4 mg mL−1) mesotherapy |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boo, Y.C. Metabolic Basis and Clinical Evidence for Skin Lightening Effects of Thiol Compounds. Antioxidants 2022, 11, 503. https://doi.org/10.3390/antiox11030503
Boo YC. Metabolic Basis and Clinical Evidence for Skin Lightening Effects of Thiol Compounds. Antioxidants. 2022; 11(3):503. https://doi.org/10.3390/antiox11030503
Chicago/Turabian StyleBoo, Yong Chool. 2022. "Metabolic Basis and Clinical Evidence for Skin Lightening Effects of Thiol Compounds" Antioxidants 11, no. 3: 503. https://doi.org/10.3390/antiox11030503
APA StyleBoo, Y. C. (2022). Metabolic Basis and Clinical Evidence for Skin Lightening Effects of Thiol Compounds. Antioxidants, 11(3), 503. https://doi.org/10.3390/antiox11030503






