The Clinical Role of SRSF1 Expression in Cancer: A Review of the Current Literature
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Source
2.2. Inclusion Criteria
2.3. Screening
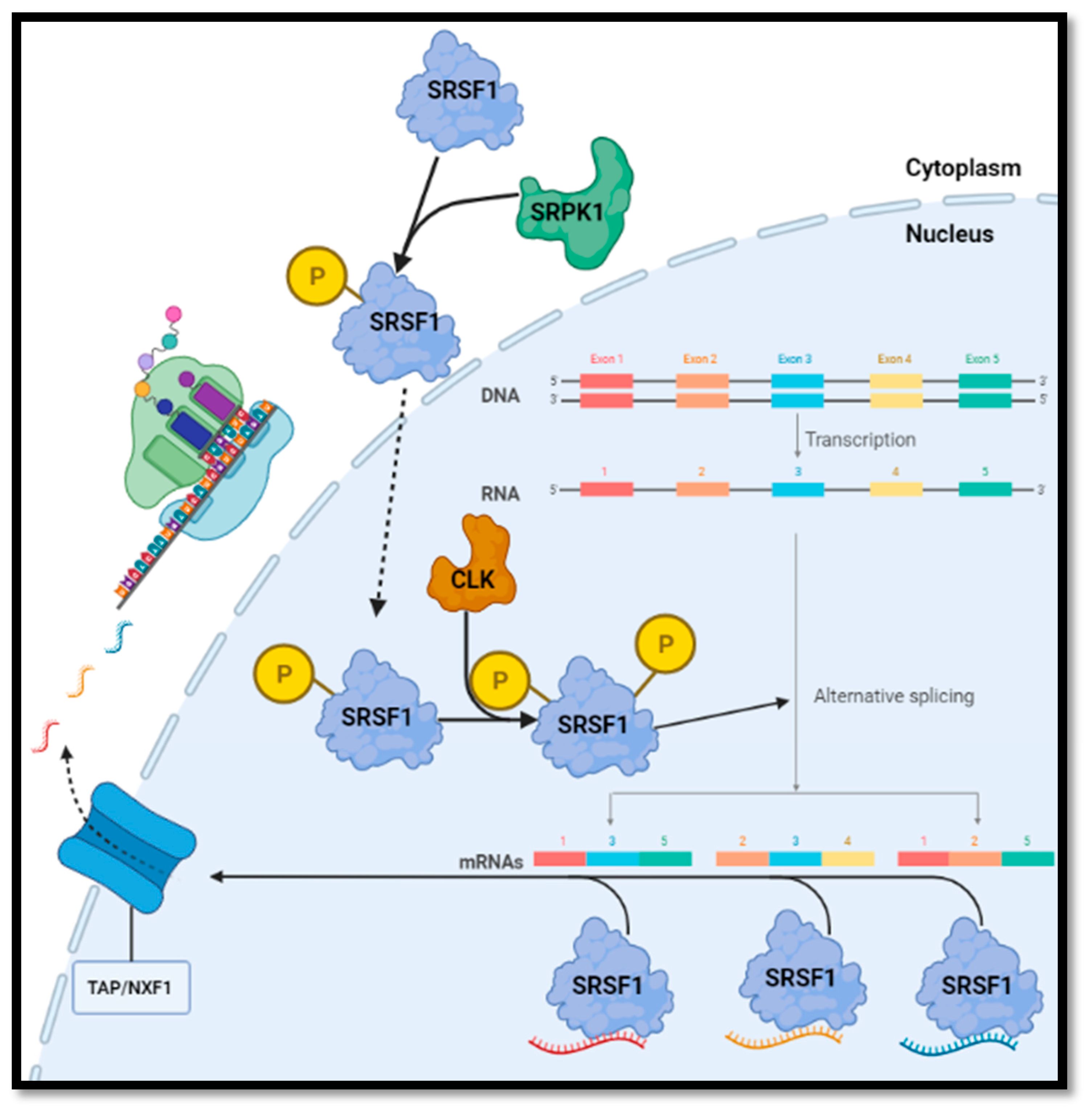
3. Results
3.1. Preliminary Results
3.2. Review
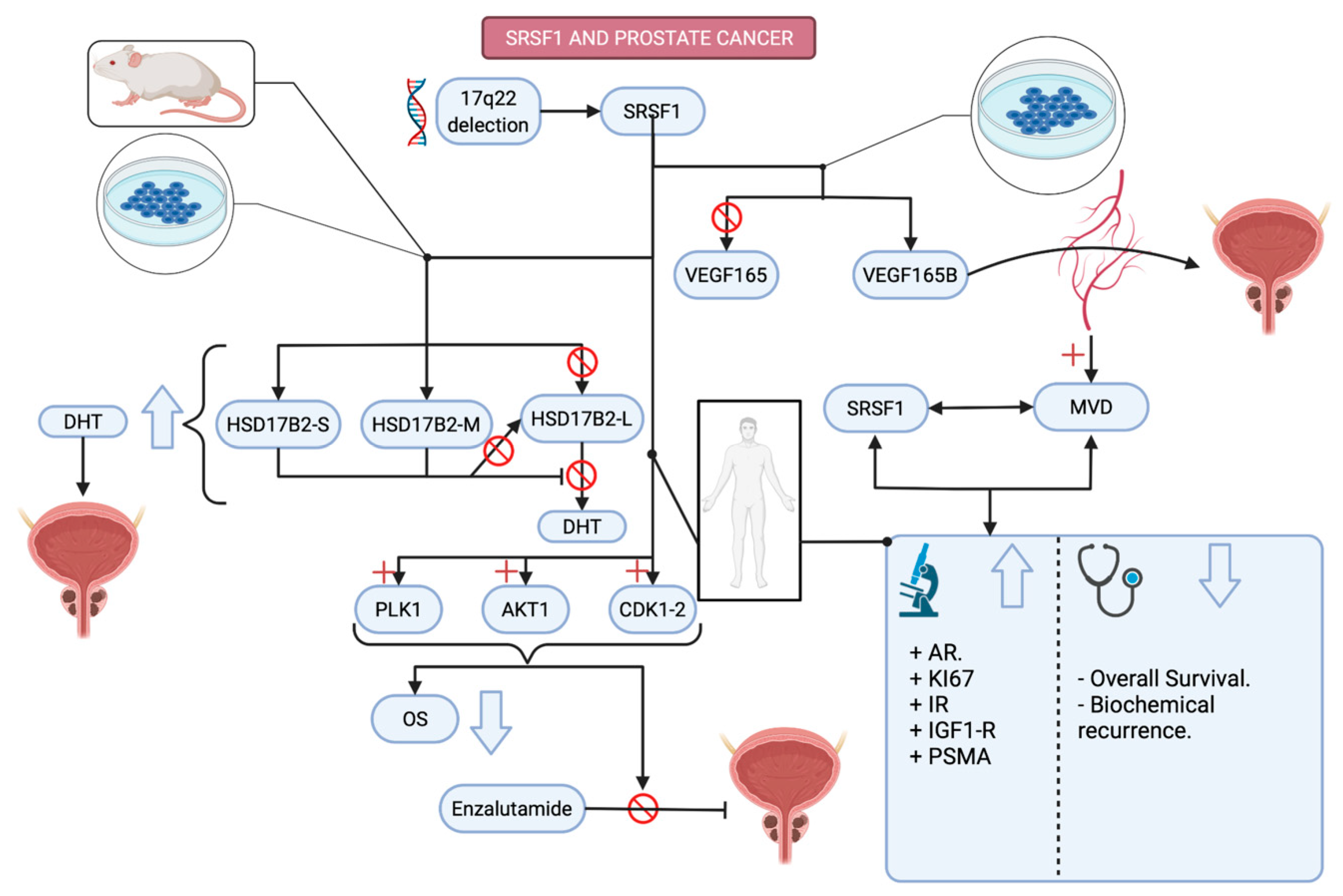
3.2.1. Prostate
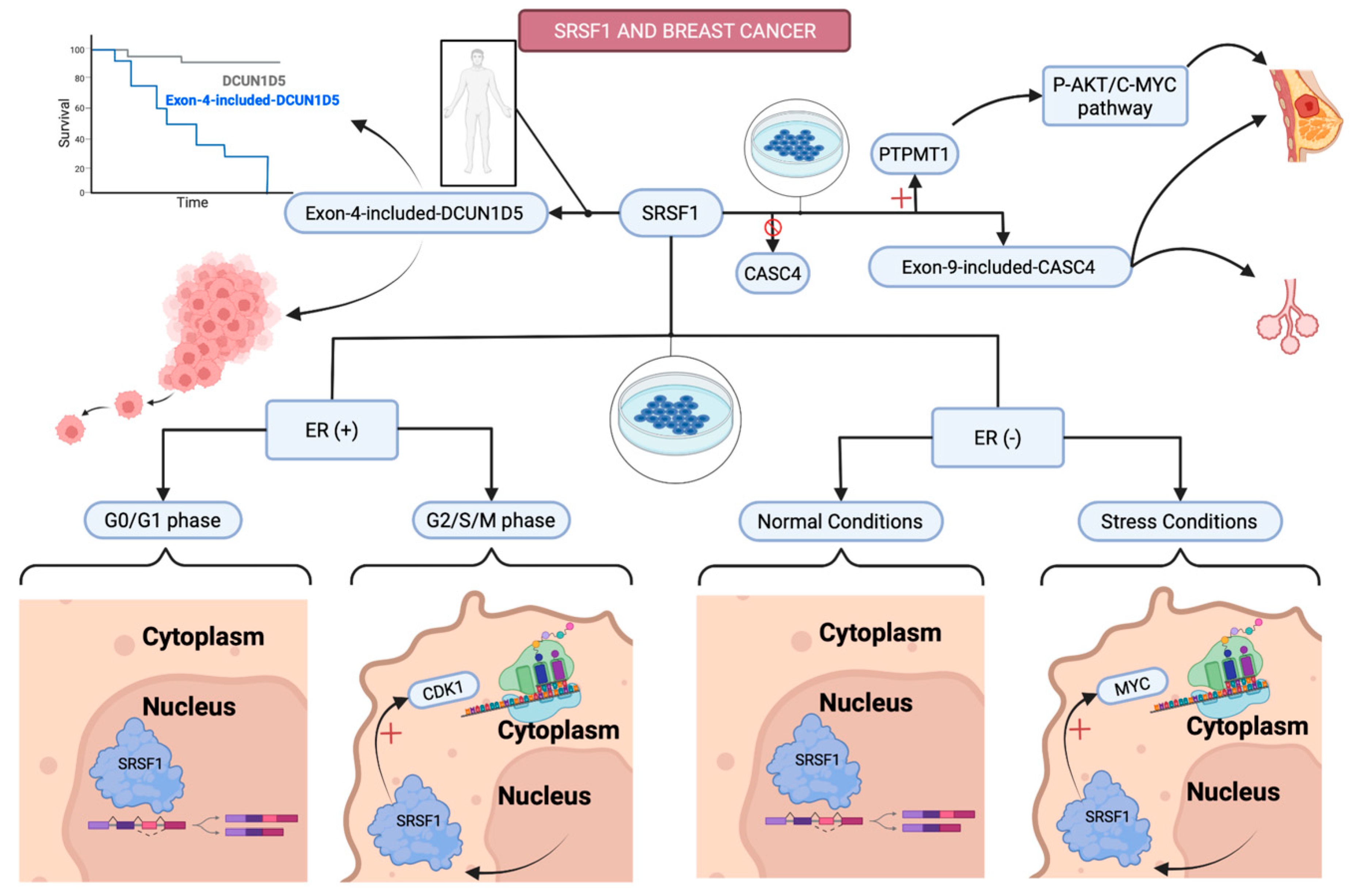
3.2.2. Breast Cancer
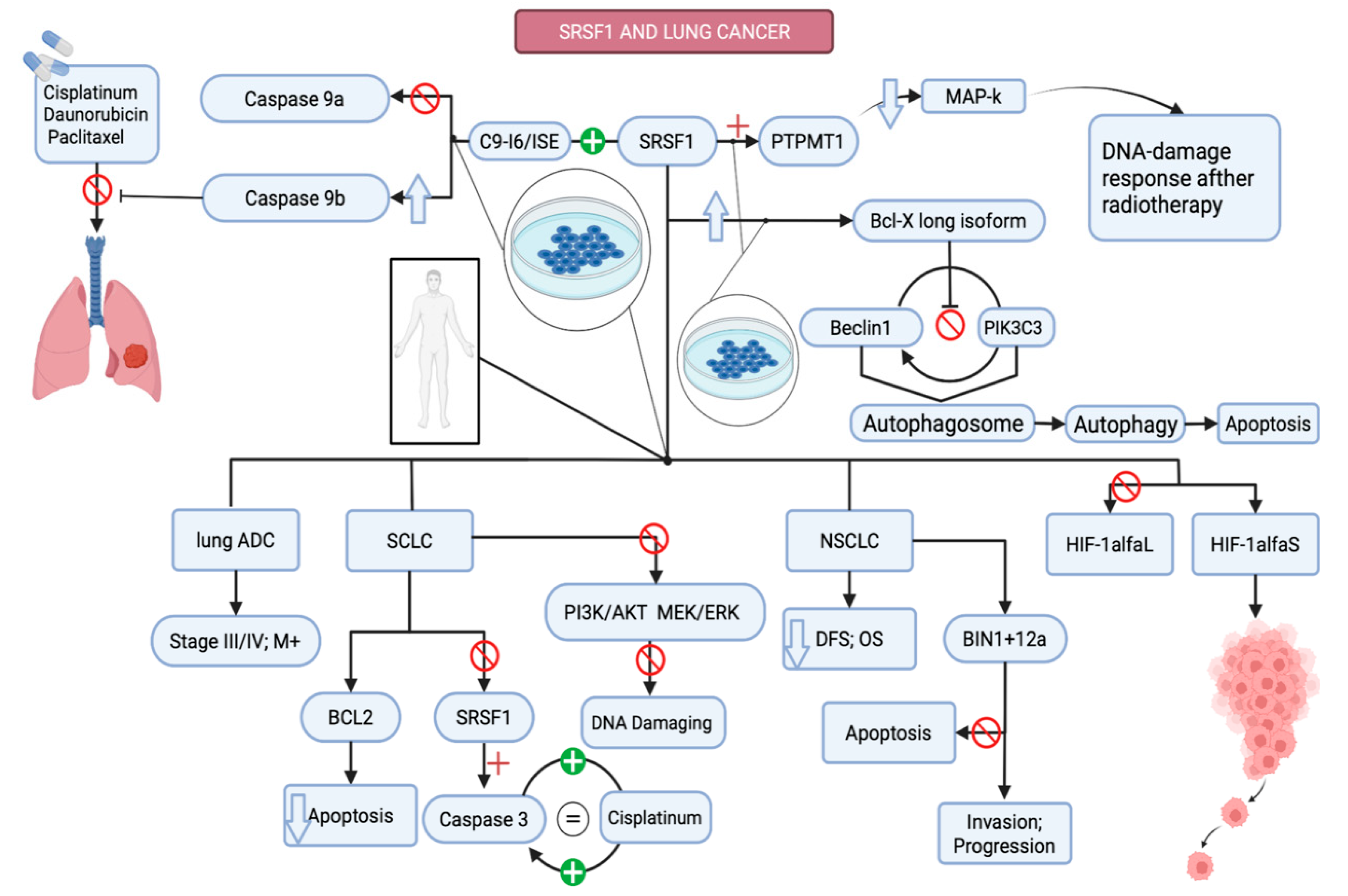
3.2.3. Lung Cancer
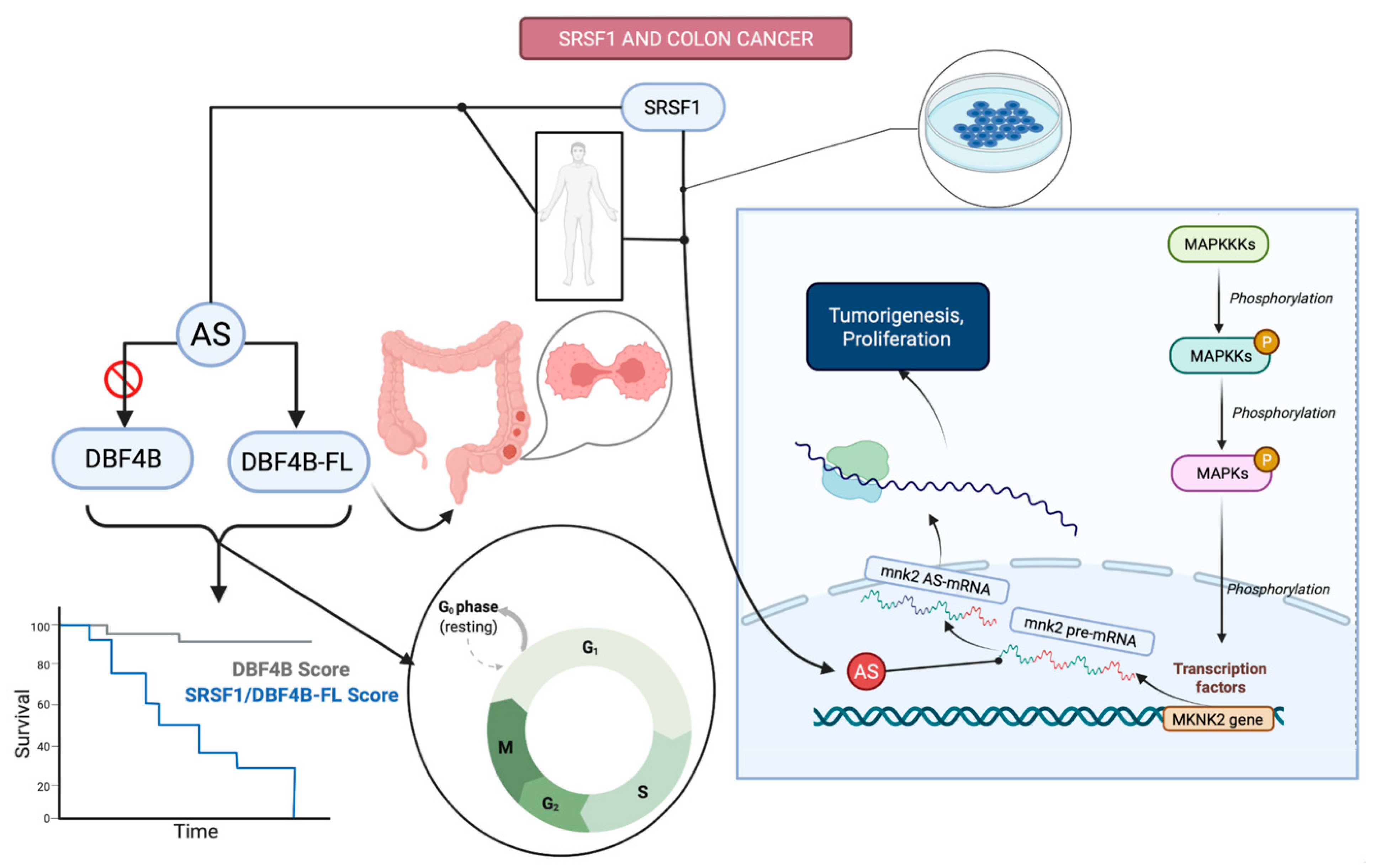
3.2.4. Colorectal Cancer
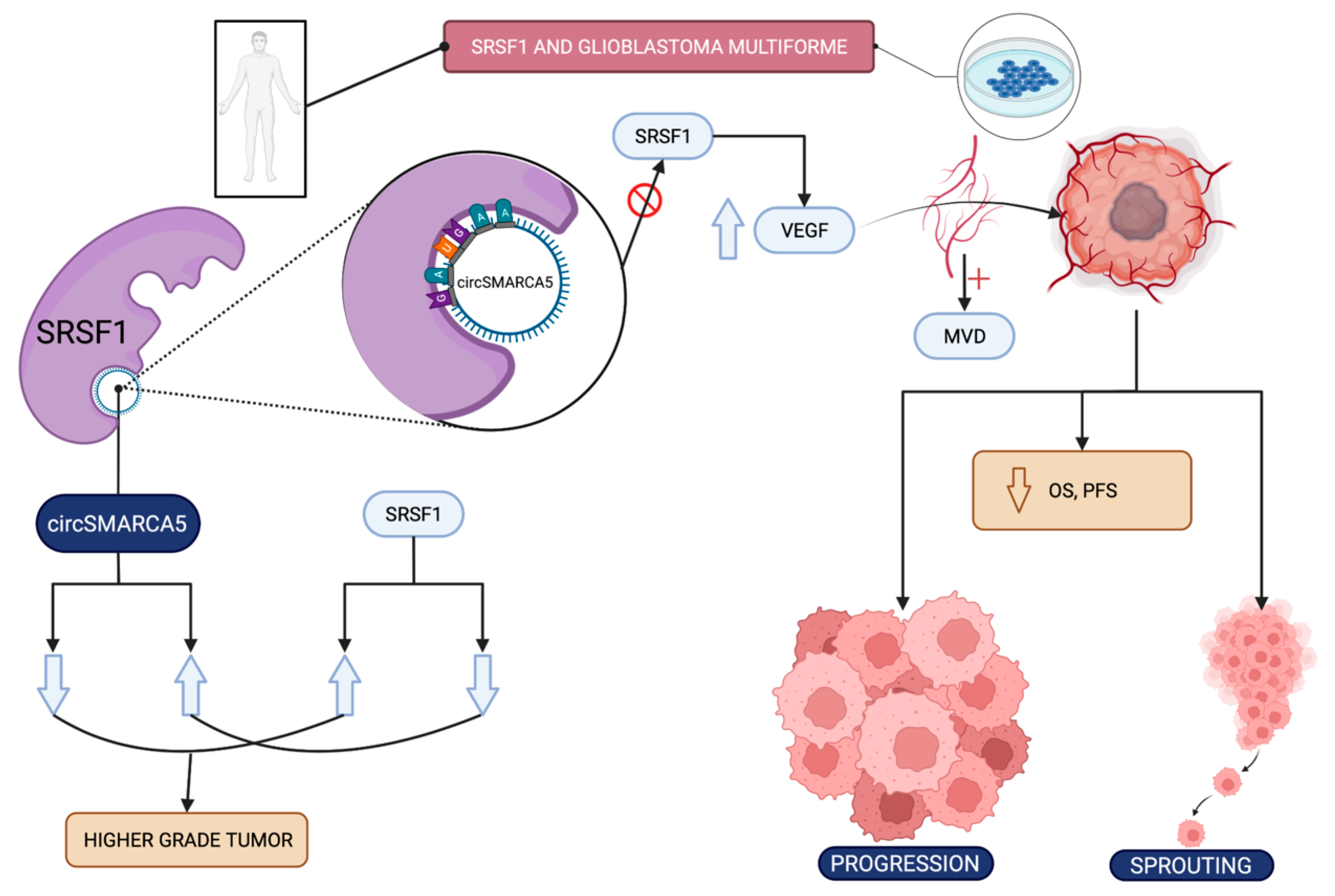
3.2.5. Glioblastoma Multiforme
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Long, J.C.; Caceres, J.F. The SR protein family of splicing factors: Master regulators of gene expression. Biochem. J. 2009, 417, 15–27. [Google Scholar] [CrossRef] [PubMed]
- Howard, J.M.; Sanford, J.R. The RNAissance family: SR proteins as multifaceted regulators of gene expression. Wiley Interdiscip. Rev. RNA 2015, 6, 93–110. [Google Scholar] [CrossRef] [PubMed]
- Anczuków, O.; Akerman, M.; Cléry, A.; Wu, J.; Shen, C.; Shirole, N.H.; Raimer, A.; Sun, S.; Jensen, M.A.; Hua, Y.; et al. SRSF1-Regulated Alternative Splicing in Breast Cancer. Mol. Cell 2015, 60, 105–117. [Google Scholar] [CrossRef] [PubMed]
- Ngo, J.C.K.; Chakrabarti, S.; Ding, J.-H.; Velazquez-Dones, A.; Nolen, B.; Aubol, B.E.; Adams, J.A.; Fu, X.-D.; Ghosh, G. Interplay between SRPK and Clk/Sty Kinases in Phosphorylation of the Splicing Factor ASF/SF2 Is Regulated by a Docking Motif in ASF/SF2. Mol. Cell 2005, 20, 77–89. [Google Scholar] [CrossRef]
- Huang, Y.; Yario, T.A.; Steitz, J.A. A molecular link between SR protein dephosphorylation and mRNA export. Proc. Natl. Acad. Sci. USA 2004, 101, 9666–9670. [Google Scholar] [CrossRef]
- Srebrow, A.; Kornblihtt, A.R. The connection between splicing and cancer. J. Cell Sci. 2006, 119, 2635–2641. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Krainer, A.R. Emerging Functions of SRSF1, Splicing Factor and Oncoprotein, in RNA Metabolism and Cancer. Mol. Cancer Res. 2014, 12, 1195–1204. [Google Scholar] [CrossRef]
- Karni, R.; de Stanchina, E.; Lowe, S.W.; Sinha, R.; Mu, D.; Krainer, A.R. The gene encoding the splicing factor SF2/ASF is a proto-oncogene. Nat. Struct. Mol. Biol. 2007, 14, 185–193. [Google Scholar] [CrossRef]
- Zhou, X.; Wang, R.; Li, X.; Yu, L.; Hua, D.; Sun, C.; Shi, C.; Luo, W.; Rao, C.; Jiang, Z.; et al. Splicing factor SRSF1 promotes gliomagenesis via oncogenic splice-switching of MYO1B. J. Clin. Investig. 2019, 129, 676–693. [Google Scholar] [CrossRef]
- Anczuków, O.; Rosenberg, A.Z.; Akerman, M.; Das, S.; Zhan, L.; Karni, R.; Muthuswamy, S.K.; Krainer, A.R. The splicing factor SRSF1 regulates apoptosis and proliferation to promote mammary epithelial cell transformation. Nat. Struct. Mol. Biol. 2012, 19, 220–228. [Google Scholar] [CrossRef]
- Das, S.; Anczuków, O.; Akerman, M.; Krainer, A.R. Oncogenic Splicing Factor SRSF1 Is a Critical Transcriptional Target of MYC. Cell Rep. 2012, 1, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Arzalluz-Luque, Á.; Cabrera, J.L.; Skottman, H.; Benguria, A.; Bolinches-Amorós, A.; Cuenca, N.; Lupo, V.; Dopazo, A.; Tarazona, S.; Delás, B.; et al. Mutant PRPF8 Causes Widespread Splicing Changes in Spliceosome Components in Retinitis Pigmentosa Patient iPSC-Derived RPE Cells. Front. Neurosci. 2021, 15, 8. [Google Scholar] [CrossRef]
- Cook, D.A.; Reed, D.A. Appraising the Quality of Medical Education Research Methods. Acad. Med. 2015, 90, 1067–1076. [Google Scholar] [CrossRef] [PubMed]
- Wells, G.; Shea, B.; O’Connell, D.; Robertson, J.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Ott. Ott. Hosp. Res. Inst. 2011, 1–12. Available online: www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 18 December 2021).
- Ferrer, F.A.; Miller, L.J.; Andrawis, R.I.; Kurtzman, S.H.; Albertsen, P.C.; Laudone, V.P.; Kreutzer, D.L. Angiogenesis and prostate cancer: In vivo and in vitro expression of angiogenesis factors by prostate cancer cells. Urology 1998, 51, 161–167. [Google Scholar] [CrossRef]
- Ferrer, F.A.; Miller, L.J.; Andrawis, R.I.; Kurtzman, S.H.; Albertsen, P.C.; Laudone, V.P.; Kreutzer, D.L. Vascular endothelial growth factor (VEGF) expression in human prostate cancer: In situ and in vitro expression of VEGF by human prostate cancer cells. J. Urol. 1997, 157, 2329–2333. [Google Scholar] [CrossRef]
- Mavrou, A.; Brakspear, K.; Hamdollah-Zadeh, M.; Damodaran, G.; Babaei-Jadidi, R.; Oxley, J.; Gillatt, D.A.; Ladomery, M.R.; Harper, S.J.; Bates, D.O.; et al. Serine–arginine protein kinase 1 (SRPK1) inhibition as a potential novel targeted therapeutic strategy in prostate cancer. Oncogene 2015, 34, 4311–4319. [Google Scholar] [CrossRef]
- Oltean, S.; Gammons, M.; Hulse, R.; Hamdollah-Zadeh, M.; Mavrou, A.; Donaldson, L.; Salmon, A.H.; Harper, S.J.; Ladomery, M.R.; Bates, D.O. SRPK1 inhibition in vivo: Modulation of VEGF splicing and potential treatment for multiple diseases. Biochem. Soc. Trans. 2012, 40, 831–835. [Google Scholar] [CrossRef]
- Amin, E.M.; Oltean, S.; Hua, J.; Gammons, M.V.R.; Hamdollah-Zadeh, M.; Welsh, G.I.; Cheung, M.-K.; Ni, L.; Kase, S.; Rennel, E.S.; et al. WT1 mutants reveal SRPK1 to be a downstream angiogenesis target by altering VEGF splicing. Cancer Cell 2011, 20, 768–780. [Google Scholar] [CrossRef]
- Gao, X.; Dai, C.; Huang, S.; Tang, J.; Chen, G.; Li, J.; Zhu, Z.; Zhu, X.; Zhou, S.; Gao, Y.; et al. Functional Silencing of HSD17B2 in Prostate Cancer Promotes Disease Progression. Clin. Cancer Res. 2019, 25, 1291–1301. [Google Scholar] [CrossRef]
- Guan, X.; Sun, D.; Lu, E.; Urrutia, J.A.; Reiter, R.E.; Rettig, M.; Evans, C.P.; Lara, P.; Gleave, M.; Beer, T.M.; et al. Copy Number Loss of 17q22 Is Associated with Enzalutamide Resistance and Poor Prognosis in Metastatic Castration-Resistant Prostate Cancer. Clin. Cancer Res. 2020, 26, 4616–4624. [Google Scholar] [CrossRef] [PubMed]
- Broggi, G.; Lo Giudice, A.; Di Mauro, M.; Asmundo, M.G.; Pricoco, E.; Piombino, E.; Caltabiano, R.; Morgia, G.; Russo, G.I. SRSF-1 and microvessel density immunohistochemical analysis by semi-automated tissue microarray in prostate cancer patients with diabetes (DIAMOND study). Prostate 2021, 81, 882–892. [Google Scholar] [CrossRef] [PubMed]
- Stickeler, E.; Kittrell, F.; Medina, D.; Berget, S.M. Stage-specific changes in SR splicing factors and alternative splicing in mammary tumorigenesis. Oncogene 1999, 18, 3574–3582. [Google Scholar] [CrossRef] [PubMed]
- Stickeler, E.; Runnebaum, I.B.; Möbus, V.J.; Kieback, D.G.; Kreienberg, R. Expression of CD44 standard and variant isoforms v5, v6 and v7 in human ovarian cancer cell lines. Anticancer Res. 1997, 17, 1871–1876. [Google Scholar] [PubMed]
- Heider, K.-H.; Mulder, J.-W.R.; Ostermann, E.; Susani, S.; Patzelt, E.; Pals, S.T.; Adolf, G.R. Splice variants of the cell surface glycoprotein CD44 associated with metastatic tumour cells are expressed in normal tissues of humans and cynomolgus monkeys. Eur. J. Cancer 1995, 31, 2385–2391. [Google Scholar] [CrossRef]
- Sinn, H.-P.; Heider, K.-H.; Skroch-Angel, P.; von Minckwitz, G.; Kaufmann, M.; Herrlich, P.; Ponta, H. Human mammary carcinomas express homologues of rat metastasis-associated variants of CD44. Breast Cancer Res. Treat. 1995, 36, 307–313. [Google Scholar] [CrossRef]
- Vaklavas, C.; Blume, S.W.; Grizzle, W.E. Hallmarks and Determinants of Oncogenic Translation Revealed by Ribosome Profiling in Models of Breast Cancer. Transl. Oncol. 2020, 13, 452–470. [Google Scholar] [CrossRef]
- Du, J.X.; Luo, Y.H.; Zhang, S.J.; Wang, B.; Chen, C.; Zhu, G.Q.; Zhu, P.; Cai, C.Z.; Wan, J.L.; Cai, J.L.; et al. Splicing factor SRSF1 promotes breast cancer progression via oncogenic splice switching of PTPMT1. J. Exp. Clin. Cancer Res. 2021, 40, 171. [Google Scholar] [CrossRef]
- Oh, J.; Pradella, D.; Shao, C.; Li, H.; Choi, N.; Ha, J.; Ruggiero, S.; Fu, X.D.; Zheng, X.; Ghigna, C.; et al. Widespread alternative splicing changes in metastatic breast cancer cells. Cells 2021, 10, 858. [Google Scholar] [CrossRef]
- Ezponda, T.; Pajares, M.J.; Agorreta, J.; Echeveste, J.I.; López-Picazo, J.M.; Torre, W.; Pio, R.; Montuenga, L.M. The oncoprotein SF2/ASF promotes non-small cell lung cancer survival by enhancing survivin expression. Clin. Cancer Res. 2010, 16, 4113–4125. [Google Scholar] [CrossRef]
- Shultz, J.C.; Goehe, R.W.; Murudkar, C.S.; Wijesinghe, D.S.; Mayton, E.K.; Massiello, A.; Hawkins, A.J.; Mukerjee, P.; Pinkerman, R.L.; Park, M.A.; et al. SRSF1 Regulates the Alternative Splicing of Caspase 9 Via A Novel Intronic Splicing Enhancer Affecting the Chemotherapeutic Sensitivity of Non–Small Cell Lung Cancer Cells. Mol. Cancer Res. 2011, 9, 889–900. [Google Scholar] [CrossRef] [PubMed]
- Gout, S.; Brambilla, E.; Boudria, A.; Drissi, R.; Lantuejoul, S.; Gazzeri, S.; Eymin, B. Abnormal Expression of the Pre-mRNA Splicing Regulators SRSF1, SRSF2, SRPK1 and SRPK2 in Non Small Cell Lung Carcinoma. PLoS ONE 2012, 7, e46539. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Huang, J.; Higgs, B.W.; Hu, Z.; Xiao, Z.; Yao, X.; Conley, S.; Zhong, H.; Liu, Z.; Brohawn, P.; et al. Genomic Landscape Survey Identifies SRSF1 as a Key Oncodriver in Small Cell Lung Cancer. PLoS Genet. 2016, 12, e1005895. [Google Scholar] [CrossRef]
- Sheng, J.; Zhao, Q.; Zhao, J.; Zhang, W.; Sun, Y.; Qin, P.; Lv, Y.; Bai, L.; Yang, Q.; Chen, L.; et al. SRSF1 modulates PTPMT1 alternative splicing to regulate lung cancer cell radioresistance. EBioMedicine 2018, 38, 113–126. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Terroba, E.; Ezponda, T.; Bértolo, C.; Sainz, C.; Remírez, A.; Agorreta, J.; Garmendia, I.; Behrens, C.; Pio, R.; Wistuba, I.I.; et al. The oncogenic RNA-binding protein SRSF1 regulates LIG1 in non-small cell lung cancer. Lab. Investig. 2018, 98, 1562–1574. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.L.; Lin, J.C. SRSF1 and RBM4 differentially modulate the oncogenic effect of HIF-1α in lung cancer cells through alternative splicing mechanism. Biochim. Biophys. Acta-Mol. Cell Res. 2019, 1866, 118550. [Google Scholar] [CrossRef]
- Wang, J.; Jia, Y.; Zhao, S.; Zhang, X.; Wang, X.; Han, X.; Wang, Y.; Ma, M.; Shi, J.; Liu, L. BIN1 reverses PD-L1-mediated immune escape by inactivating the c-MYC and EGFR/MAPK signaling pathways in non-small cell lung cancer. Oncogene 2017, 36, 6235–6243. [Google Scholar] [CrossRef]
- Wang, J.; Liu, T.; Wang, M.; Lv, W.; Wang, Y.; Jia, Y.; Zhang, R.; Liu, L. SRSF1-dependent alternative splicing attenuates BIN1 expression in non–small cell lung cancer. J. Cell. Biochem. 2020, 121, 946–953. [Google Scholar] [CrossRef]
- Yorimitsu, T.; Klionsky, D.J. Autophagy: Molecular machinery for self-eating. Cell Death Differ. 2005, 12, 1542–1552. [Google Scholar] [CrossRef]
- Boise, L.H.; González-García, M.; Postema, C.E.; Ding, L.; Lindsten, T.; Turka, L.A.; Mao, X.; Nuñez, G.; Thompson, C.B. bcl-x, a bcl-2-related gene that functions as a dominant regulator of apoptotic cell death. Cell 1993, 74, 597–608. [Google Scholar] [CrossRef]
- Peng, Y.; Miao, H.; Wu, S.; Yang, W.; Zhang, Y.; Xie, G.; Xie, X.; Li, J.; Shi, C.; Ye, L.; et al. ABHD5 interacts with BECN1 to regulate autophagy and tumorigenesis of colon cancer independent of PNPLA2. Autophagy 2016, 12, 2167–2182. [Google Scholar] [CrossRef] [PubMed]
- Morselli, E.; Galluzzi, L.; Kepp, O.; Vicencio, J.-M.; Criollo, A.; Maiuri, M.C.; Kroemer, G. Anti- and pro-tumor functions of autophagy. Biochim. Biophys. Acta-Mol. Cell Res. 2009, 1793, 1524–1532. [Google Scholar] [CrossRef] [PubMed]
- Maria Fimia, G.; Stoykova, A.; Romagnoli, A.; Giunta, L.; Di Bartolomeo, S.; Nardacci, R.; Corazzari, M.; Fuoco, C.; Ucar, A.; Schwartz, P.; et al. Ambra1 regulates autophagy and development of the nervous system. Nature 2007, 447, 1121–1125. [Google Scholar] [CrossRef] [PubMed]
- Lv, Y.; Zhang, W.; Zhao, J.; Sun, B.; Qi, Y.; Ji, H.; Chen, C.; Zhang, J.; Sheng, J.; Wang, T.; et al. SRSF1 inhibits autophagy through regulating Bcl-x splicing and interacting with PIK3C3 in lung cancer. Signal. Transduct. Target. Ther. 2021, 6, 108. [Google Scholar] [CrossRef] [PubMed]
- Sheng, J.; Zhao, J.; Xu, Q.; Wang, L.; Zhang, W.; Zhang, Y. Bioinformatics analysis of SRSF1-controlled gene networks in colorectal cancer. Oncol. Lett. 2017, 14, 5393–5399. [Google Scholar] [CrossRef][Green Version]
- Chen, L.; Luo, C.; Shen, L.; Liu, Y.; Wang, Q.; Zhang, C.; Guo, R.; Zhang, Y.; Xie, Z.; Wei, N.; et al. SRSF1 Prevents DNA Damage and Promotes Tumorigenesis through Regulation of DBF4B Pre-mRNA Splicing. Cell Rep. 2017, 21, 3406–3413. [Google Scholar] [CrossRef]
- Montagnoli, A.; Bosotti, R.; Villa, F.; Rialland, M.; Brotherton, D.; Mercurio, C.; Berthelsen, J.; Santocanale, C. Drf1, a novel regulatory subunit for human Cdc7 kinase. EMBO J. 2002, 21, 3171–3181. [Google Scholar] [CrossRef]
- Liu, H.; Gong, Z.; Li, K.; Zhang, Q.; Xu, Z.; Xu, Y. SRPK1/2 and PP1α exert opposite functions by modulating SRSF1-guided MKNK2 alternative splicing in colon adenocarcinoma. J. Exp. Clin. Cancer Res. 2021, 40, 75. [Google Scholar] [CrossRef]
- Stella, M.; Falzone, L.; Caponnetto, A.; Gattuso, G.; Barbagallo, C.; Battaglia, R.; Mirabella, F.; Broggi, G.; Altieri, R.; Certo, F.; et al. Serum Extracellular Vesicle-Derived circHIPK3 and circSMARCA5 Are Two Novel Diagnostic Biomarkers for Glioblastoma Multiforme. Pharmaceuticals 2021, 14, 618. [Google Scholar] [CrossRef]
- Barbagallo, D.; Caponnetto, A.; Brex, D.; Mirabella, F.; Barbagallo, C.; Lauretta, G.; Morrone, A.; Certo, F.; Broggi, G.; Caltabiano, R.; et al. CircSMARCA5 regulates VEGFA mRNA splicing and angiogenesis in glioblastoma multiforme through the binding of SRSF1. Cancers 2019, 11, 194. [Google Scholar] [CrossRef]
- Broggi, G.; Salvatorelli, L.; Barbagallo, D.; Certo, F.; Altieri, R.; Tirrò, E.; Massimino, M.; Vigneri, P.; Guadagno, E.; Maugeri, G.; et al. Diagnostic Utility of the Immunohistochemical Expression of Serine and Arginine Rich Splicing Factor 1 (SRSF1) in the Differential Diagnosis of Adult Gliomas. Cancers 2021, 13, 2086. [Google Scholar] [CrossRef] [PubMed]
- Barbagallo, D.; Caponnetto, A.; Barbagallo, C.; Battaglia, R.; Mirabella, F.; Brex, D.; Stella, M.; Broggi, G.; Altieri, R.; Certo, F.; et al. The gaugaa motif is responsible for the binding between circsmarca5 and srsf1 and related downstream effects on glioblastoma multiforme cell migration and angiogenic potential. Int. J. Mol. Sci. 2021, 22, 1678. [Google Scholar] [CrossRef] [PubMed]
| Num | Authors | Year | Topic | Bioinformatic | In Vitro | In Vivo Animal | Clinical | MERSQI | NOS-E |
|---|---|---|---|---|---|---|---|---|---|
| [1] | Bates, Do. et al. | 2016 | Prostate cancer | X | NA | NA | |||
| [2] | Gao, X. et al. | 2019 | Prostate cancer | X | X | NA | NA | ||
| [3] | Guan, X. et al. | 2020 | Prostate cancer | X | 16 | 10 | |||
| [4] | Broggi, G. et al. | 2021 | Prostate cancer | X | 16 | 10 | |||
| SUM | 4 | ||||||||
| [5] | Anczuków, O. et al. | 2015 | Breast cancer | X | NA | NA | |||
| [6] | Vaklavas, C. et al. | 2019 | Breast cancer | X | NA | NA | |||
| [7] | Du, J.X. et al. | 2021 | Breast cancer | X | 16 | 10 | |||
| [8] | Oh, J. et al. | 2021 | Breast cancer | X | 16 | 10 | |||
| SUM | 4 | ||||||||
| [9] | Shultz, J.C. et al. | 2011 | Lung cancer | X | NA | NA | |||
| [10] | Gout, S. et al. | 2012 | Lung cancer | X | 16 | 10 | |||
| [11] | Jiang, L. et al. | 2016 | Lung cancer | X | 16 | 10 | |||
| [12] | Sheng, J. et al. | 2018 | Lung cancer | X | X | NA | NA | ||
| [13] | Martínez-Terroba, E. et al. | 2018 | Lung cancer | X | 16 | 10 | |||
| [14] | Chang, H.L. et al. | 2019 | Lung cancer | X | NA | NA | |||
| [15] | Wang, J. et al. | 2020 | Lung cancer | X | NA | NA | |||
| [16] | Lv, Y. et al. | 2021 | Lung cancer | X | NA | NA | |||
| SUM | 8 | ||||||||
| [17] | Sheng, J. et al. | 2017 | Colorectal cancer | X | NA | NA | |||
| [18] | Chen, L. et al. | 2017 | Colorectal cancer | X | X | X | 16 | 10 | |
| [19] | Liu, H. et al. | 2021 | Colorectal cancer | X | 16 | 10 | |||
| SUM | 3 | ||||||||
| [20] | Barbagallo, D. et al. | 2018 | Glioblastoma multiforme | X | 16 | 10 | |||
| [21] | Barbagallo, D. et al. | 2019 | Glioblastoma multiforme | X | 16 | 10 | |||
| [22] | Barbagallo, D. et al. | 2021 | Glioblastoma multiforme | X | NA | NA | |||
| SUM | 3 | ||||||||
| SUM | SUM | SUM | SUM | ||||||
| 1 | 11 | 3 | 11 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lo Giudice, A.; Asmundo, M.G.; Broggi, G.; Cimino, S.; Morgia, G.; Di Trapani, E.; Luzzago, S.; Musi, G.; Ferro, M.; de Cobelli, O.; et al. The Clinical Role of SRSF1 Expression in Cancer: A Review of the Current Literature. Appl. Sci. 2022, 12, 2268. https://doi.org/10.3390/app12052268
Lo Giudice A, Asmundo MG, Broggi G, Cimino S, Morgia G, Di Trapani E, Luzzago S, Musi G, Ferro M, de Cobelli O, et al. The Clinical Role of SRSF1 Expression in Cancer: A Review of the Current Literature. Applied Sciences. 2022; 12(5):2268. https://doi.org/10.3390/app12052268
Chicago/Turabian StyleLo Giudice, Arturo, Maria Giovanna Asmundo, Giuseppe Broggi, Sebastiano Cimino, Giuseppe Morgia, Ettore Di Trapani, Stefano Luzzago, Gennaro Musi, Matteo Ferro, Ottavio de Cobelli, and et al. 2022. "The Clinical Role of SRSF1 Expression in Cancer: A Review of the Current Literature" Applied Sciences 12, no. 5: 2268. https://doi.org/10.3390/app12052268
APA StyleLo Giudice, A., Asmundo, M. G., Broggi, G., Cimino, S., Morgia, G., Di Trapani, E., Luzzago, S., Musi, G., Ferro, M., de Cobelli, O., & Russo, G. I. (2022). The Clinical Role of SRSF1 Expression in Cancer: A Review of the Current Literature. Applied Sciences, 12(5), 2268. https://doi.org/10.3390/app12052268









