Therapeutic Applications of Halloysite
Abstract
1. Introduction
2. Nanoclay Types
2.1. Montmorillonite
2.2. Bentonite
2.3. Laponite
3. Halloysite Structure and Applications
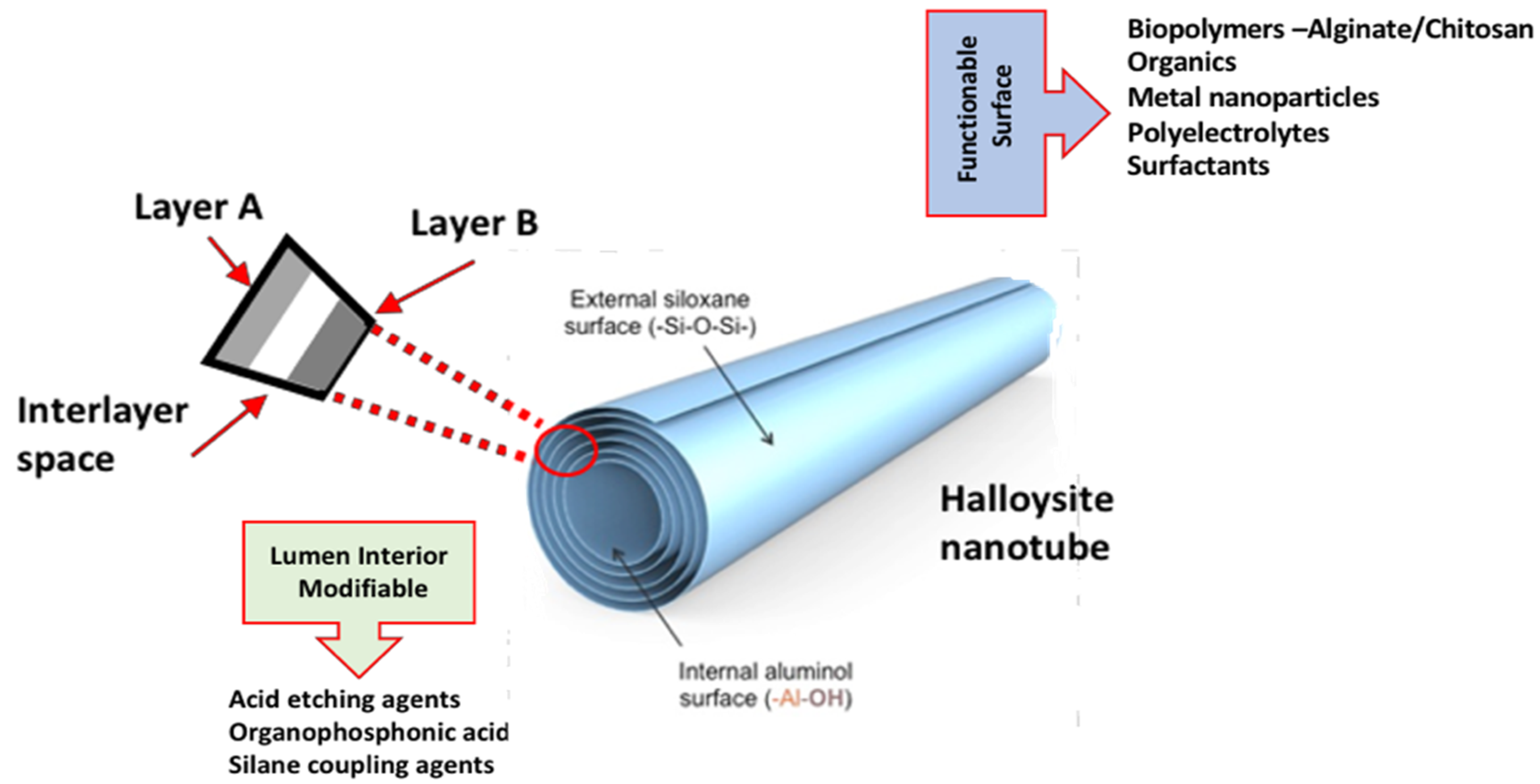
3.1. HNT Structure
3.2. HNTs in Cancer Therapeutics
3.3. HNTs in Drug Delivery
3.4. HNTs in Tissue Engineering
3.5. HNTs in Wound Healing
4. Recent Trends in HNT Research
4.1. HNTs as a Drug Carrier
4.2. HNTs in Coatings and Films
4.3. HNTs and 3D Printing
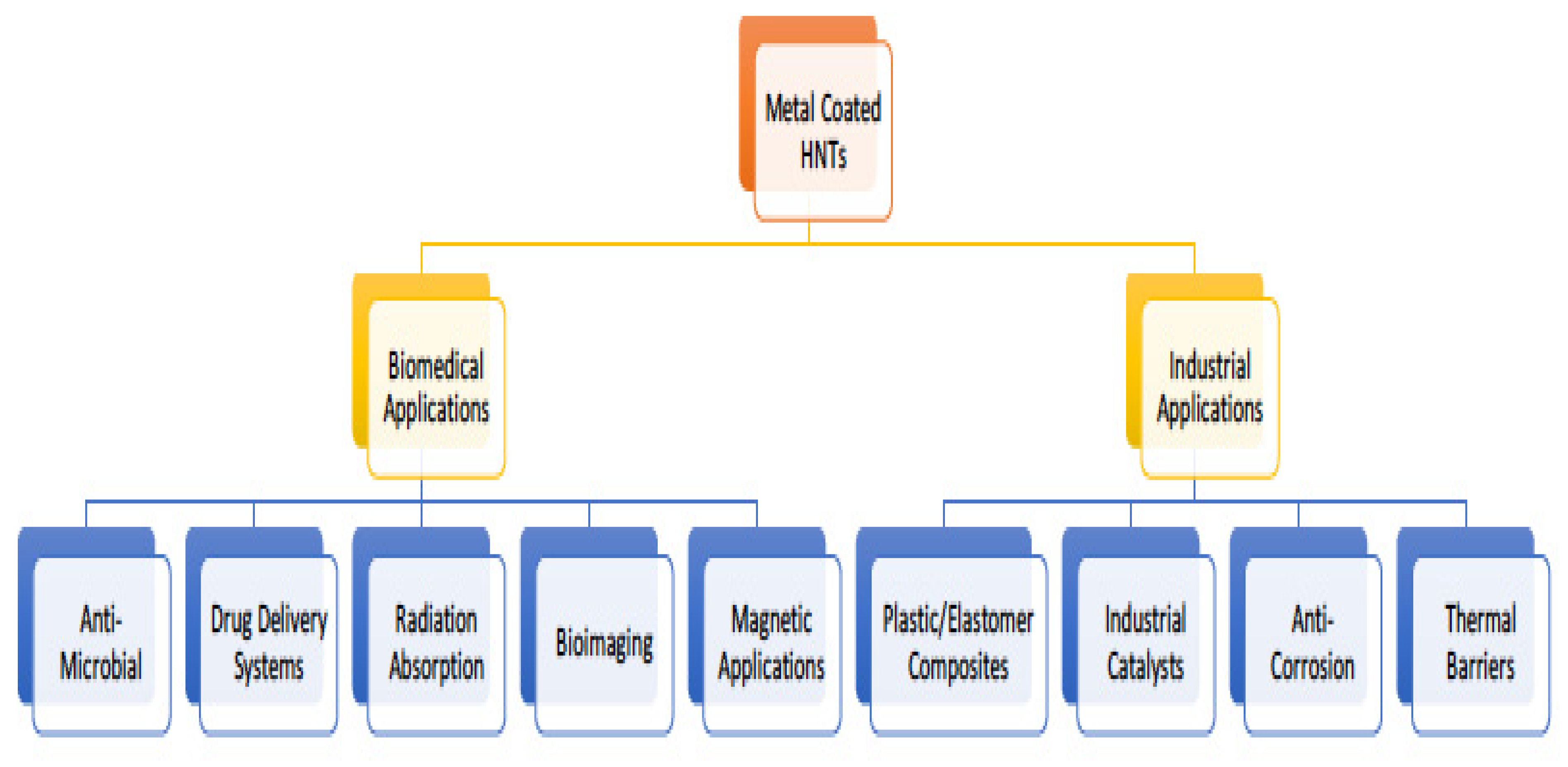
4.4. Metalized HNT Applications
5. Future Directions in HNT Research
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Savic, I.; Stojiljkovic, S.; Savic, I.; Gajic, D. Industrial application of clays and clay minerals. In Clays and Clay Minerals: Geological Origin, Mechanical Properties and Industrial Applications; Wesley, L.R., Ed.; Nova Science Publishers: New York, NY, USA, 2014; pp. 379–402. [Google Scholar]
- Guo, F.; Aryana, S.; Han, Y.; Jiao, Y. A review of the synthesis and applications of polymer–nanoclay composites. Appl. Sci. 2018, 8, 1696. [Google Scholar] [CrossRef]
- Sandri, G.; Bonferoni, M.C.; Rossi, S.; Ferrari, F.; Aguzzi, C.; Viseras, C.; Caramella, C. Clay minerals for tissue regeneration, repair, and engineering. In Wound Healing Biomaterials; Ågren, M.S., Ed.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 385–402. ISBN 9781782424567. [Google Scholar]
- Suresh, R.; Borkar, N.S.; Sawant, V.A.; Shende, V.S.; Dimble, S.K. Nanoclay drug delivery system. Inter. J. Pharma. Sci. Nanotech. 2010, 3, 901–905. [Google Scholar]
- Kotal, M.; Bhowmick, A.K. Polymer nanocomposites from modified clays: Recent advances and challenges. Prog. Polym. Sci. 2015, 51, 127–187. [Google Scholar] [CrossRef]
- Sánchez-Fernández, A.; Peña-Parás, L.; Vidaltamayo, R.; Cué-Sampedro, R.; Mendoza-Martínez, A.; Zomosa-Signoret, V.C.; Rivas-Estilla, A.M.; Riojas, P. Synthesization, characterization, and in vitro evaluation of cytotoxicity of biomaterials based on halloysite nanotubes. Materials 2014, 7, 7770–7780. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Long, M.; Huang, P.; Yang, H.; Chang, S.; Hu, Y.; Tang, A.; Mao, L. Emerging integrated nanoclay-facilitated drug delivery system for papillary thyroid cancer therapy. Sci. Rep. 2016, 6, 33335. [Google Scholar] [CrossRef]
- Jawaid, M.; Qaiss, A.K.; Bouhfid, R. Nanoclay Reinforced Polymer Composites: Nanocomposites and Bionanocomposites; Springer: Singapore, 2016. [Google Scholar]
- Lau, L.; Gu, C.; Hui, D. A critical review on nanotube and nanotube/nanoclay related polymer composite materials. Compos. Part B Eng. 2006, 37, 425–436. [Google Scholar] [CrossRef]
- Uddin, F. Montmorillonite: An Introduction to Properties and Utilization; IntechOpen: London, UK, 2018. [Google Scholar]
- Ali, A. Effect of incorporation of montmorillonite on Xylan/Chitosan conjugate scaffold. Colloids Surf. Part B 2019, 180, 75–82. [Google Scholar] [CrossRef]
- Cui, Z.-K.; Kim, S.; Baljon, J.J.; Wu, B.M.; Aghaloo, T.; Lee, M. Microporous methacrylated glycol chitosan-montmorillonite nanocomposite hydrogel for bone tissue engineering. Nat. Commun. 2019, 10, 3523. [Google Scholar] [CrossRef] [PubMed]
- Haroun, A.A.; Gamal-Eldeen, A.; Harding, D.R.K. Preparation, characterization and in vitro biological study of biomimetic three-dimensional gelatin–montmorillonite/cellulose scaffold for tissue engineering. J. Mater. Sci. Mater. Med. 2009, 20, 2527–2540. [Google Scholar] [CrossRef] [PubMed]
- Mauro, N.; Chiellini, F.; Bartoli, C.; Gazzarri, M.; Laus, M.; Antonioli, D.; Griffiths, P.; Manfredi, A.; Ranucci, E.; Ferruti, P. RGD-mimic polyamidoamine–montmorillonite composites with tunable stiffness as scaffolds for bone tissue-engineering applications. J. Tissue Eng. Regen. Med. 2017, 1, 2164–2175. [Google Scholar] [CrossRef]
- Jamshidi, M. Nanoclay reinforced starch-polycaprolactone scaffolds for bone tissue Engineering. J. Tissues Mater. 2019, 2, 55–63. [Google Scholar]
- Chen, M. Fabrication and characterization of a rapid prototyped tissue engineering scaffold with embedded multicomponent matrix for controlled drug release. Int. J. Nanomed. 2012, 2012, 4285–4297. [Google Scholar] [CrossRef]
- Noori, S. Nanoclay enhanced the mechanical properties of poly(vinylalcohol)/chitosan/montmorillonite nanocomposite hydrogel as wound dressing. Procedia Mater. Sci. 2015, 1, 52–156. [Google Scholar]
- Garcia-Villen, F. Montmorillonite-norfloxacin nanocomposite intended for healing of infected wounds. Int. J. Nanomed. 2019, 14, 5051–5060. [Google Scholar] [CrossRef]
- Emami-Razavi, S.H. Effect of bentonite on skin wound healing: Experimental study in the rat model. Acta Med. Iran. Vol. 2016, 44, 235–240. [Google Scholar]
- Alavi, M.; Totonchi, A.; Okhovat, M.A.; Motazedian, M.; Rezaei, P.; Atefi, M. The effect of a new impregnated gauze containing bentonite and halloysite minerals on blood coagulation and wound healing. Blood Coagul. Fbrinolysis 2014, 25, 856–859. [Google Scholar] [CrossRef]
- Khoshmohabat, H.; Dalfardi, B.; Dehghanian, A.; Rasouli, H.R.; Mortazavi, S.M.J.; Paydar, S. The effect of CoolClot hemostatic agent on skin wound healing in rats. J. Surg. Res. 2015, 200, 732–737. [Google Scholar] [CrossRef] [PubMed]
- Wilson, R. Kaolin and halloysite deposits of China. Clay Miner. 2004, 39, 1–15. [Google Scholar] [CrossRef]
- Liu, M.; Shen, Y.; Daib, L.; Zhou, C. The improvement of hemostatic and wound healing property of chitosan by halloysite nanotubes. RSC Adv. 2014, 4, 23540–23553. [Google Scholar] [CrossRef]
- Fang, F.; Xu, Y.; Wang, Z.; Zhou, W.; Yan, L.; Fan, F.; Liu, H. 3D porous chitin sponge with high absorbency, rapid shape recovery, and excellent antibacterial activities for noncompressible wound. Chem. Eng. J. 2020, 388, 124169. [Google Scholar] [CrossRef]
- Devi, N. Preparation and characterization of chitosan-bentonite nanocomposite films for wound healing application. Int. J. Biol. Macromol. 2017, 104, 1897–1904. [Google Scholar] [CrossRef] [PubMed]
- Adams, L.A. Bentonite clay and waterglass porous monoliths Via the sol-gel process. J. Met. Mat. Min. 2011, 21, 1–6. [Google Scholar]
- Essien, E.R.; Adams, L.A.; Shaibu, R.O.; Oki, A. Sol-gel bioceramic material from bentonite clay. J. Biomed. Sci. Eng. 2013, 6, 258–264. [Google Scholar] [CrossRef][Green Version]
- Shahbuddin, N.S. Alumina Foam (AF) Fabrication optimization and SBF immersion studies for AF, hydroxyapatite (HA) coated AF (HACAF) and HA-bentonite coated AF (HABCAF) Bone Tissue Scaffolds. Procedia Chem. 2016, 19, 884–890. [Google Scholar] [CrossRef]
- Adams, L.A.; Essien, E.R.; Kaufmann, E.E. Mechanical and bioactivity assessment of wollastonite/PVA composite synthesized from bentonite clay. Ceramica 2019, 65, 246–251. [Google Scholar] [CrossRef]
- Bajaj, H.; Kevadiya, B.; Joshi, G.; Patel, H.; Abdi, S. Montmorillonite-alginate composites as a drug delivery system: Intercalation and In vitro release of diclofenac sodium. Indian J. Pharm. Sci. 2010, 72, 732–737. [Google Scholar] [CrossRef]
- Park, J.-H.; Shin, H.-J.; Kim, M.H.; Kim, J.-S.; Kang, N.; Lee, J.-Y.; Kim, K.-T.; Lee, J.I.; Kim, D.-D. Application of montmorillonite in bentonite as a pharmaceutical excipient in drug delivery systems. J. Pharm. Investig. 2016, 46, 363–375. [Google Scholar] [CrossRef]
- Hosseini, F.; Hosseini, F.; Jafari, S.M.; Taheri, A. Bentonite nanoclay-based drug-delivery systems for treating melanoma. Clay Miner. 2018, 53, 53–63. [Google Scholar] [CrossRef]
- Aguzzi, C.; Cerezo, P.; Viseras, C.; Caramella, C. Use of clays as drug delivery systems: Possibilities and limitations. Appl. Clay Sci. 2007, 36, 22–36. [Google Scholar] [CrossRef]
- Thomas, H.; Alves, C.S.; Rodrigues, J. Laponite®: A key nanoplatform for biomedical applications? Nanomed. Nanotechnol. Biol. Med. 2018, 14, 2407–2420. [Google Scholar] [CrossRef]
- Das, S.S.; Neelam; Hussain, K.; Singh, S.; Hussain, A.; Faruk, A.; Tebyetekerwa, M. Laponite-based nanomaterials for biomedical applications: A Review. Curr. Pharm. Des. 2019, 25, 424–443. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Wang, S.; Li, K.; Ju, Y.; Li, J.; Zhang, Y.; Li, J.; Liu, X.; Shi, X.; Zhao, Q. Preparation of laponite bioceramics for potential Bbne tissue engineering applications. PLoS ONE 2014, 9, e99585. [Google Scholar] [CrossRef]
- Levis, S.; Deasy, P. Characterisation of halloysite for use as a microtubular drug delivery system. Int. J. Pharm. 2002, 243, 125–134. [Google Scholar] [CrossRef]
- Jousseni, E.; Petit, S.; Churchman, J.; Theng, B.; Delvaux, D.R.B. Halloysite clay minerals: A review. Clay Min. 2005, 40, 383–426. [Google Scholar] [CrossRef]
- Noro, H. Hexagonal platy halloysite in an altered tuff bed, Komaki City, Aichi Prefecture, Central Japan. Clay Miner. 1986, 21, 401. [Google Scholar] [CrossRef]
- Wei, W.; Abdllayev, E.; Goeders, A.; Hollister, A.; Lvov, L.; Mills, D.K. Clay nanotube/poly(methyl methacrylate) bone cement composite with sustained antibiotic release. Macromol. Mat. Eng. 2012, 297, 645–653. [Google Scholar] [CrossRef]
- De Silva, R.T.; Dissanayake, R.K.; Mantilaka, M.M.M.G.P.G.; Wijesinghe, W.P.S.L.; Kaleel, S.S.; Premachandra, T.N.; Weerasinghe, L.; Amaratunga, G.A.J.; de Silva, K.M.N. Drug-loaded halloysite nanotube-reinforced electrospun alginate-based nanofibrous scaffolds with sustained antimicrobial protection. ACS Appl. Mater. Interfaces 2018, 10, 33913–33922. [Google Scholar] [CrossRef]
- Karnik, S.; Mills, D.K. Clay nanotubes as growth factor delivery vehicle for bone tissue engineering. J. Nanomed. Nanotechnnol. 2013, 4, 102. [Google Scholar]
- Satish, S.; Tharmavaram, M.; Rawtani, D. Halloysite nanotubes as a nature’s boon for biomedical applications. Nanobiomedicine 2019, 6, 1849543519863625. [Google Scholar] [CrossRef]
- Pasbakhsh, P.; De Silva, R.; Vahedi, V.; Churchman, G.J. Halloysite nanotubes: Prospects and challenges of their use as additives and carriers—A focused review. Clay Miner. 2016, 51, 479–487. [Google Scholar] [CrossRef]
- Lvov, Y.; Abdullayev, E. Functional polymer–clay nanotube composites with sustained release of chemical agents. Prog. Polym. Sci. 2013, 38, 1690–1719. [Google Scholar] [CrossRef]
- Mousavi, S.M.; Hashemi, S.A.; Salahi, S.; Hosseini, M.; Ali Mohammad Amani, A.M.; Aziz Babapoor, A. Development of clay nanoparticles toward bio and medical applications. In Chapter 8 in Current Topics in the Utilization of Clay in Industrial and Medical Applications; InTech Open: Zagreb, Croatia, 2018. [Google Scholar]
- Tharmavaram, M.; Gaurav, P.; Deepak, R. Surface modified halloysite nanotubes: A flexible interface for biological, environmental and catalytic applications. Adv. Colloid Interface Sci. 2018, 261, 82–101. [Google Scholar] [CrossRef]
- Patel, S.; Jammalamadaka, U.; Sun, L.; Tappa, K.; Mills, D.K. Sustained release of antibacterial agents from doped halloysite nanotubes. Bioengineering 2015, 3, 1. [Google Scholar] [CrossRef] [PubMed]
- Karnik, S.; Hines, K.; Mills, D.K. Nanoenhanced hydrogel system with sustained release capabilities. J. Biomed. Mater. Res. Part A 2014, 103, 2416–2426. [Google Scholar] [CrossRef]
- Romero-Trigueros, C.; Parra, M.; Bayona, J.M.; Nortes, P.; Alarcón, J.J.; Nicolás, E. Effect of deficit irrigation and reclaimed water on yield and quality of grapefruits at harvest and postharvest. LWT Food Sci. Technol. 2017, 85, 405–411. [Google Scholar] [CrossRef]
- Fakhrullina, G.I.; Akhatova, F.S.; Lvov, Y.M.; Fakhrullin, R.F. Toxicity of halloysite clay nanotubes in vivo: A Caenorhabditis elegans study. Environ. Sci. Nano 2014, 2, 54–59. [Google Scholar] [CrossRef]
- Kryuchkova, M.; Danilushkina, A.; Lvov, Y.; Fakhrullin, R. Evaluation of toxicity of nanoclays and graphene oxide in vivo: A Paramecium caudatum study. Environ. Sci. Nano 2016, 3, 442–452. [Google Scholar] [CrossRef]
- Yang, J.; Wu, Y.; Shen, Y.; Zhou, C.; Li, Y.-F.; He, R.-R.; Liu, M. Enhanced Therapeutic Efficacy of Doxorubicin for Breast Cancer Using Chitosan Oligosaccharide-Modified Halloysite Nanotubes. ACS Appl. Mater. Interfaces 2016, 8, 26578–26590. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Chang, Y.; Yang, J.; You, Y.; He, R.; Chen, T.; Zhou, C. Functionalized halloysite nanotube by chitosan grafting for drug delivery of curcumin to achieve enhanced anticancer efficacy. J. Mater. Chem. B 2016, 4, 2253–2263. [Google Scholar] [CrossRef]
- Wu, Y.-P.; Yang, J.; Gao, H.-Y.; Shen, Y.; Jiang, L.; Zhou, C.; Li, Y.-F.; He, R.-R.; Liu, M. Folate-Conjugated Halloysite Nanotubes, an Efficient Drug Carrier, Deliver Doxorubicin for Targeted Therapy of Breast Cancer. ACS Appl. Nano Mater. 2018, 1, 595–608. [Google Scholar] [CrossRef]
- Lvov, Y.M.; Devilliers, M.M.; Fakhrullin, R.F. The application of halloysite tubule nanoclay in drug delivery. Expert Opin. Drug Deliv. 2016, 13, 977–986. [Google Scholar] [CrossRef] [PubMed]
- Fizir, M.; Dramou, P.; Dahiru, N.S.; Ruya, W.; Huang, T.; He, H. Halloysite nanotubes in analytical sciences and in drug delivery: A review. Microchim. Acta 2018, 185, 389. [Google Scholar] [CrossRef] [PubMed]
- Tohidi, S.; Ghaee, A.; Barzin, J. Preparation and characterization of poly (lactic-co-glycolic acid)/chitosan electrospun membrane containing amoxicillin-loaded halloysite nanoclay. Poly. Adv. Techn. 2016, 27, 1020–1028. [Google Scholar] [CrossRef]
- Li, H.; Zhu, X.; Zhou, H.; Zhong, S. Functionalization of halloysite nanotubes by enlargement and hydrophobicity for sustained release of analgesic. Colloids Surf. Physicochem. Eng. Asp. 2015, 487, 154–161. [Google Scholar] [CrossRef]
- Tan, D.; Yuan, P.; Annabi-Bergaya, F.; Yu, H.; Liu, D.; Liu, H.; He, H. Natural halloysite nanotubes as mesoporous carriers for the loading of ibuprofen. Microporous Mesoporous Mater. 2013, 179, 89–98. [Google Scholar] [CrossRef]
- Ganguly, S.; Das, T.K.; Mondal, S.; Das, N.C. Synthesis of polydopamine-coated halloysite nanotube-based hydrogel for controlled release of a calcium channel blocker. RSC Adv. 2016, 6, 105350–105362. [Google Scholar] [CrossRef]
- Shi, Y.-F.; Tian, Z.; Zhang, Y.; Shen, H.-B.; Jia, N.-Q. Functionalized halloysite nanotube-based carrier for intracellular delivery of antisense oligonucleotides. Nanoscale Res. Lett. 2011, 6, 608. [Google Scholar] [CrossRef]
- Santos, A.C.; Ferreira, C.; Veiga, F.; Ribeiro, A.; Panchal, A.; Lvov, Y.; Agarwal, A. Halloysite clay nanotubes for life sciences applications: From drug encapsulation to bioscaffold. Adv. Colloid Interface Sci. 2018, 257, 58–70. [Google Scholar] [CrossRef]
- Bottino, M.C.; Batarseh, G.; Palasuk, J.; Alkatheeri, M.S.; Windsor, L.J.; Platt, J.A. Nanotube-modified dentin adhesive—Physicochemical and dentin bonding characterizations. Dent. Mater. 2013, 29, 1158–1165. [Google Scholar] [CrossRef]
- Chen, Q.; Zhao, Y.; Wu, W.; Xu, T.; Fong, H. Fabrication and evaluation of Bis-GMA/TEGDMA dental resins/composites containing halloysite nanotubes. Dent. Mater. 2012, 28, 1071–1079. [Google Scholar] [CrossRef]
- Feitosa, S.A.; Münchow, E.A.; Al-Zain, A.O.; Kamocki, K.; Platt, J.A.; Bottino, M.C. Synthesis and characterization of novel halloysite-incorporated adhesive resins. J. Dent. 2015, 43, 1316–1322. [Google Scholar] [CrossRef]
- Barot, T.; Rawtani, D.; Kulkarni, P. Physicochemical and biological assessment of silver nanoparticles immobilized halloysite nanotubes-based resin composite for dental applications. Heliyon 2020, 6, e03601. [Google Scholar] [CrossRef] [PubMed]
- Drury, J.L.; Mooney, D.J. Hydrogels for tissue engineering: Scaffold design variables and applications. Biomaterials 2003, 24, 4337–4351. [Google Scholar] [CrossRef]
- Fakhrullin, R.F.; Lvov, Y.M. Halloysite clay nanotubes for tissue engineering. Nanomedicine 2016, 11, 2243–2246. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Han, S.S. Enhanced mechanical, biomineralization, and cellular response of nanocomposite hydrogels by bioactive glass and halloysite nanotubes for bone tissue regeneration. Mater. Sci. Eng. 2021, 128, 112236. [Google Scholar] [CrossRef]
- Sandri, G.; Aguzzi, C.; Rossi, S.; Bonferoni, M.C.; Bruni, G.; Boselli, C.; Cornaglia, A.I.; Riva, F.; Viseras, C.; Caramella, C.; et al. Halloysite and chitosan oligosaccharide nanocomposite for wound healing. Acta Biomater. 2017, 57, 216–224. [Google Scholar] [CrossRef]
- Wang, L.; You, X.; Dai, C.; Tong, T.; Wu, J. Hemostatic nanotechnologies for external and internal hemorrhage management. Biomater. Sci. 2020, 8, 4396–4412. [Google Scholar] [CrossRef]
- Sandri, G.; Faccendini, A.; Longo, M.; Ruggeri, M.; Rossi, S.; Bonferoni, M.C.; Miele, D.; Prina-Mello, A.; Aguzzi, C.; Viseras, C.; et al. Halloysite- and Montmorillonite-Loaded Scaffolds as Enhancers of Chronic Wound Healing. Pharmaceutics 2020, 12, 179. [Google Scholar] [CrossRef]
- Wali, A.; Gorain, M.; Inamdar, S.; Kundu, G.C.; Badiger, M.V. In vivo wound healing performance of halloysite clay and gentamicin-incorporated cellulose ether-PVA electrospun nanofiber mats. ACS Appl. Bio Mater. 2019, 2, 4324–4334. [Google Scholar] [CrossRef]
- Cheng, C.; Gao, Y.; Song, W.; Zhao, Q.; Zhang, H.; Zhang, H. Halloysite nanotube-based H2O2-responsive drug delivery system with a turn on effect on fluorescence for real-time monitoring. Chem. Eng. J. 2020, 380, 122474. [Google Scholar] [CrossRef]
- Lisuzzo, L.; Cavallaro, G.; Parisi, F.; Milioto, S.; Fakhrullin, R.; Lazzara, G. Core/shell gel beads with embedded halloysite nanotubes for controlled drug release. Coatings 2019, 9, 70. [Google Scholar] [CrossRef]
- Akrami-Hasan-Kohal, M.; Ghorbani, M.; Mahmoodzadeh, F.; Nikzad, B. Development of reinforced aldehyde-modified kappa-carrageenan/gelatin film by incorporation of halloysite nanotubes for biomedical applications. Int. J. Biol. Macromol. 2020, 160, 669–676. [Google Scholar] [CrossRef] [PubMed]
- Xie, M.; Huang, K.; Yang, F.; Wang, R.; Han, L.; Yu, H.; Ye, Z.; Wu, F. Chitosan nanocomposite films based on halloysite nanotubes modification for potential biomedical applications. Int. J. Biol. Macromol. 2019, 151, 1116–1125. [Google Scholar] [CrossRef]
- Devi, N.; Dutta, J. Development and in vitro characterization of chitosan/starch/halloysite nanotubes ternary nanocomposite films. Int. J. Biol. Macromol. 2019, 127, 222–231. [Google Scholar] [CrossRef]
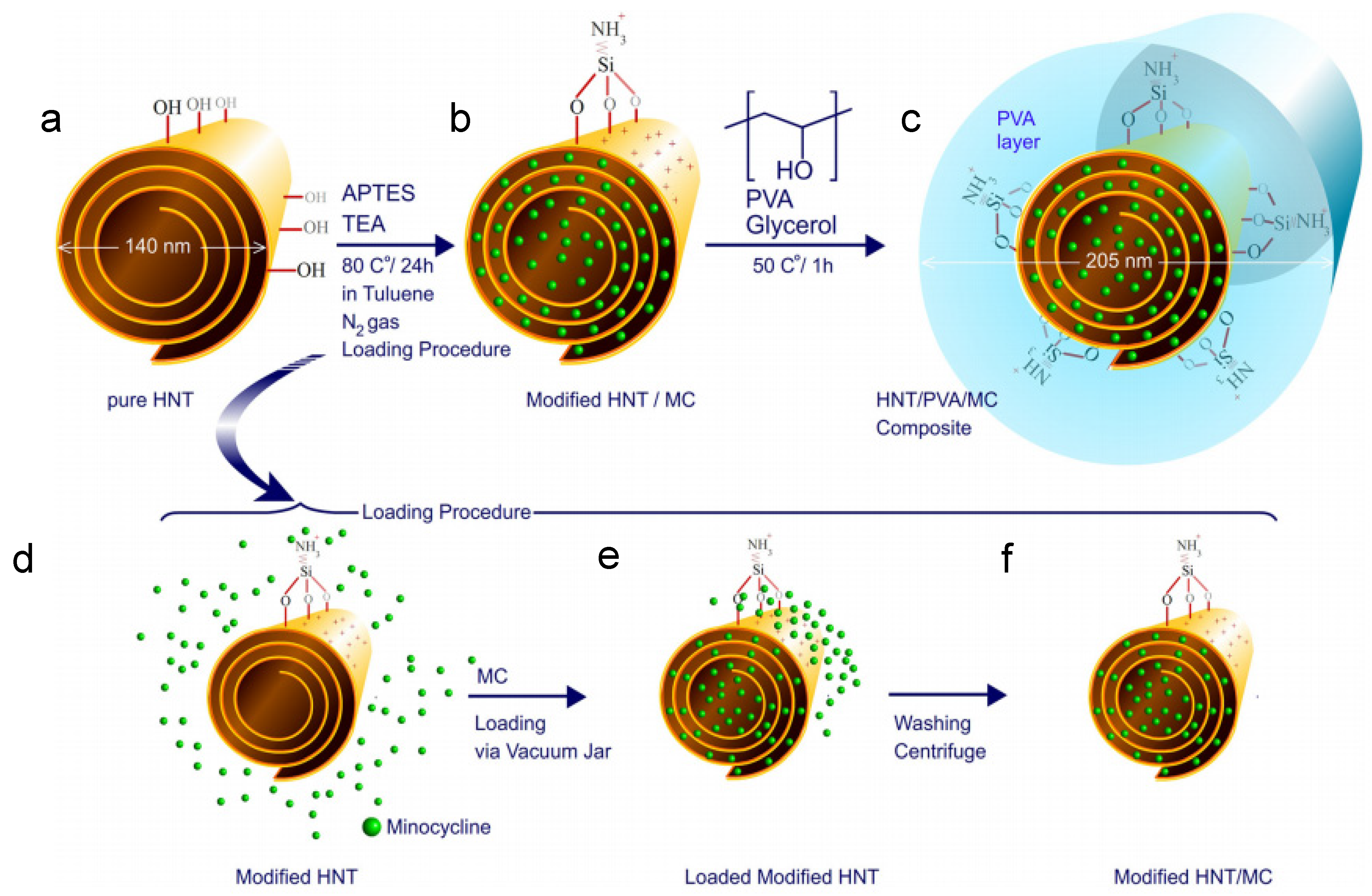
- Mohebalia, A.; Abdoussa, M.; Faramarz, A.; Taromib, A. Fabrication of biocompatible antibacterial nanowafers based on HNT/PVA nanocomposites loaded with minocycline for burn wound dressing. Mater. Sci. Eng. 2020, 110, 110685. [Google Scholar] [CrossRef]
- Weisman, J.A.; Jammalamadaka, U.; Tappa, K.; Mills, D.K. Doped Halloysite Nanotubes for Use in the 3D Printing of Medical Devices. Bioengineering 2017, 4, 96. [Google Scholar] [CrossRef] [PubMed]
- Tappa, K.; Jammalamadaka, U.; Weisman, J.A.; Ballard, D.H.; Wolford, D.D.; Pascual-Garrido, C.; Wolford, L.M.; Woodard, P.K.; Mills, D.K. 3D Printing Custom Bioactive and Absorbable Surgical Screws, Pins, and Bone Plates for Localized Drug Delivery. J. Funct. Biomater. 2019, 10, 17. [Google Scholar] [CrossRef]
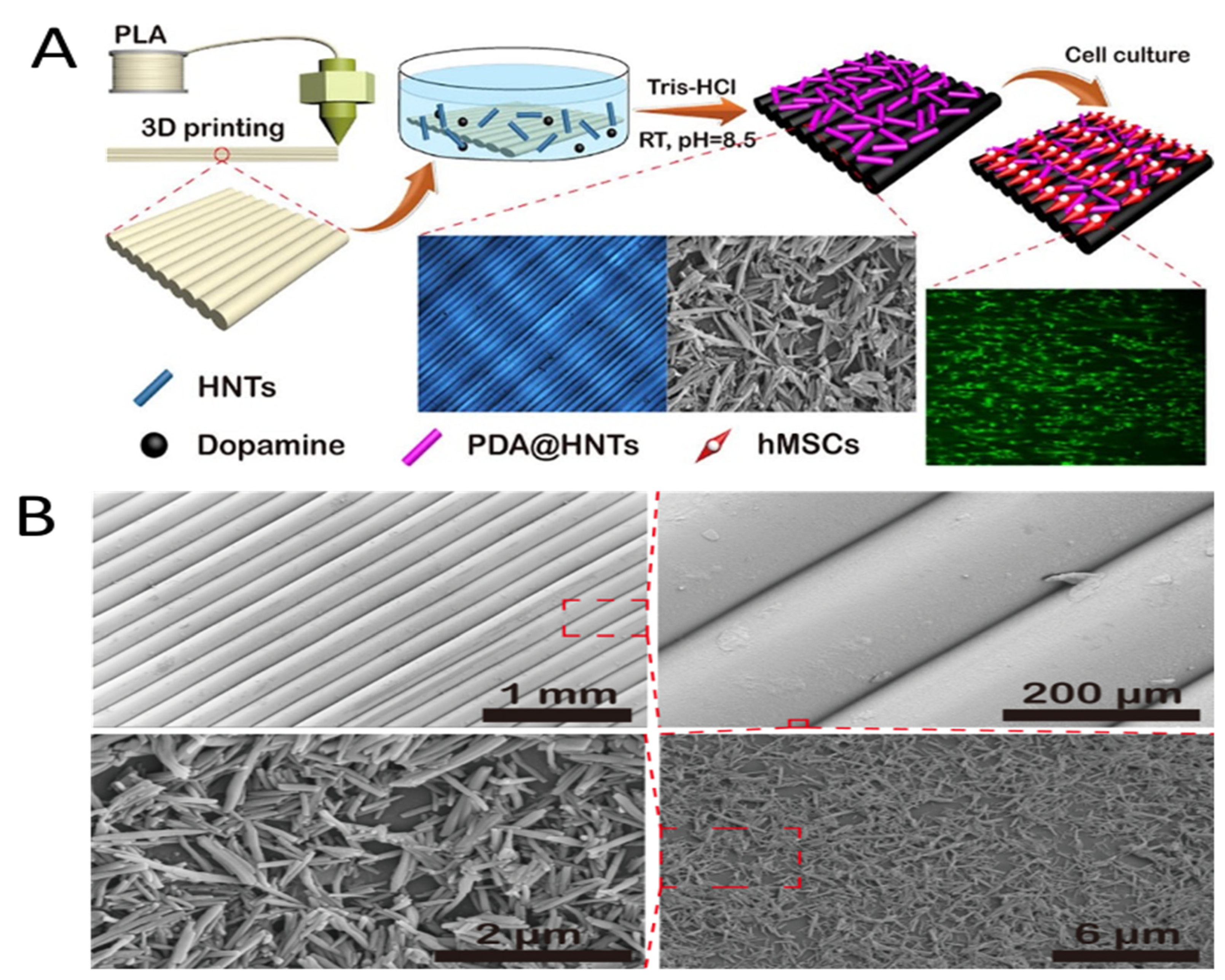
- Wu, F.; Zheng, J.; Li, Z.; Liu, M. Halloysite nanotubes coated 3D printed PLA pattern for guiding human mesenchymal stem cells (hMSCs) orientation. Chem. Eng. J. 2018, 359, 672–683. [Google Scholar] [CrossRef]
- Arvizo, R.R.; Bhattacharyya, S.; Kudgus, R.A.; Giri, K.; Bhattacharya, R.; Mukherjee, P. Intrinsic therapeutic applications of noble metal nanoparticles: Past, present and future. Chem. Soc. Rev. 2012, 41, 2943–2970. [Google Scholar] [CrossRef]
- Azharuddin, M.; Zhu, G.H.; Das, D.; Ozgur, E.; Uzun, L.; Turner, A.P.F.; Patra, H.K. A repertoire of biomedical applications of noble metal nanoparticles. Chem. Commun. 2019, 55, 6964–6996. [Google Scholar] [CrossRef]
- Massaro, M.; Cavallaro, G.; Colletti, C.G.; Lazzara, G.; Milioto, S.; Noto, R.; Riela, S. Chemical modification of halloysite nanotubes for controlled loading and release. J. Mater. Chem. B 2018, 6, 3415–3433. [Google Scholar] [CrossRef] [PubMed]
- Boyer, C.; Mills, D.K. Method for Preparing Halloysite Supported Metal Nanoparticles through Electrolysis. U.S. Patent US9981074B1, 25 September 2015. [Google Scholar]
- Luo, Y.; Humayun, A.; Mills, D.K. Surface Modification of 3D Printed PLA/Halloysite Composite Scaffolds with Antibacterial and Osteogenic Capabilities. Appl. Sci. 2020, 10, 3971. [Google Scholar] [CrossRef]
- Humayun, A.; Luo, Y.; Mills, D.K. 3D printed antimicrobial PLA constructs functionalized with zinc-coated halloysite nanotubes-Ag-chitosan oligosaccharide lactate. Mater. Technol. 2020, 8, 1–8. [Google Scholar]
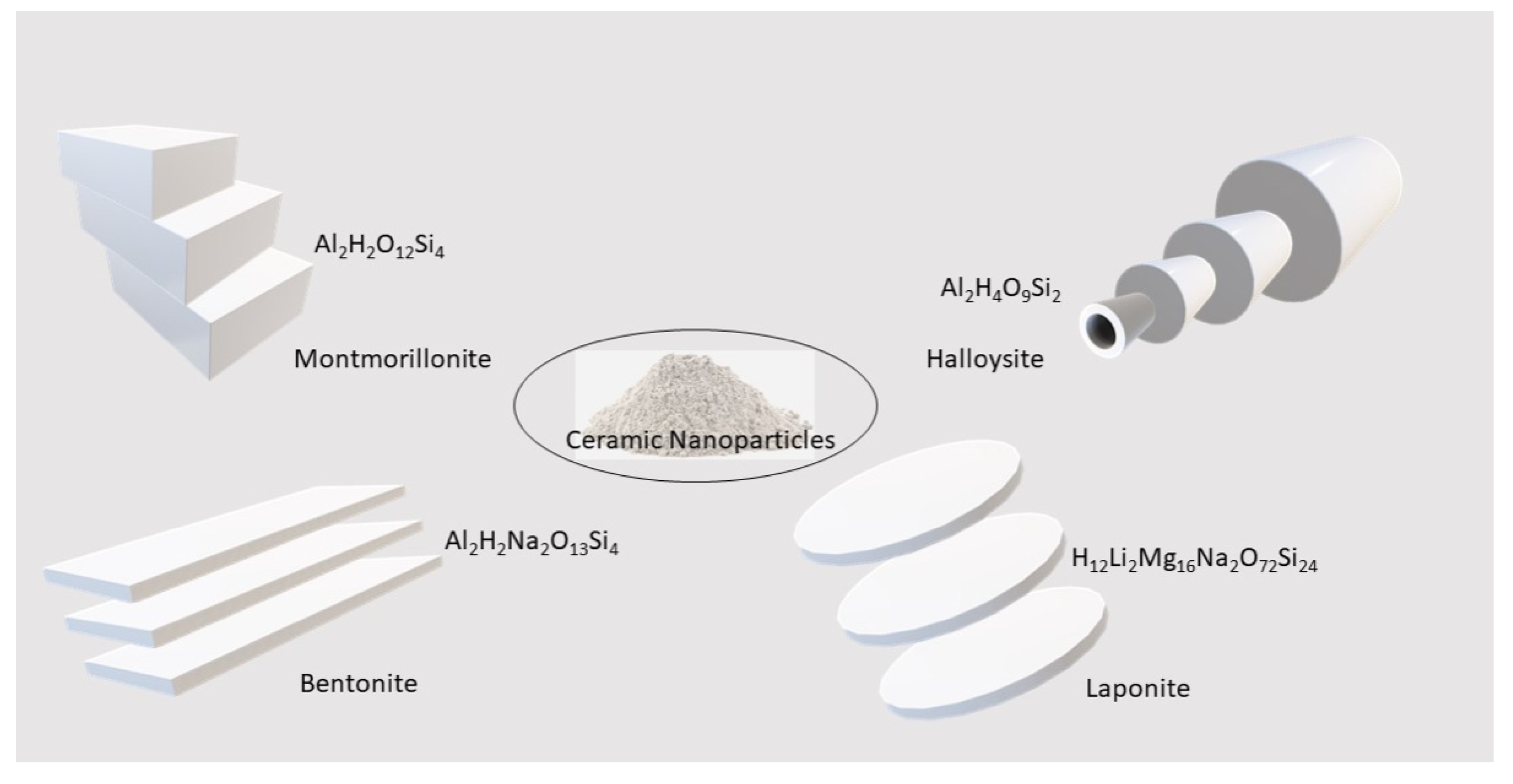


| Clay Type | General Formula | Structure | Therapeutic Applications |
|---|---|---|---|
| Bentonite | Al2H2Na2O13Si4 | Layered | Absorption, detoxification, drug delivery, filler |
| Halloysite | Al2Si2O5(OH)4 | Nanotube | Absorption, bioink additive bioremediation, detoxification, drug delivery, filler, tissue engineering |
| Kaolinite | Al2H4O9Si2 | Layered | Absorption, drug delivery, filler, tissue engineering |
| Laponite | H12Li2Mg16Na2O72Si24 | Discoidal | Bioink additive, bioimaging, drug delivery, tissue engineering |
| Montmorillonite | Al2H2O12Si4 | Multi-layered | Antimicrobial, drug delivery, filler, tissue engineering |
| Antimicrobial | Sustained Release | Targeted Drug Delivery | Tissue Engineering | Wound Healing |
|---|---|---|---|---|
| antibiotics | antimicrobials | brain | bone | anti-infection |
| antifungals | anti-cancer agents | breast cancer | guided nerve regeneration | hemostasis |
| anti-biofouling | dyes | colon cancer | cartilage | healing |
| anti-viral | growth factors | osteosarcoma | skin | revascularization |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mobaraki, M.; Karnik, S.; Li, Y.; Mills, D.K. Therapeutic Applications of Halloysite. Appl. Sci. 2022, 12, 87. https://doi.org/10.3390/app12010087
Mobaraki M, Karnik S, Li Y, Mills DK. Therapeutic Applications of Halloysite. Applied Sciences. 2022; 12(1):87. https://doi.org/10.3390/app12010087
Chicago/Turabian StyleMobaraki, Mohammadmahdi, Sonali Karnik, Yue Li, and David K. Mills. 2022. "Therapeutic Applications of Halloysite" Applied Sciences 12, no. 1: 87. https://doi.org/10.3390/app12010087
APA StyleMobaraki, M., Karnik, S., Li, Y., & Mills, D. K. (2022). Therapeutic Applications of Halloysite. Applied Sciences, 12(1), 87. https://doi.org/10.3390/app12010087







