Evaluation of Antioxidant Activity, Toxicity, and Phenolic Profile of Aqueous Extracts of Chamomile (Matricaria chamomilla L.) and Sage (Salvia officinalis L.) Prepared at Different Temperatures
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Plant Material
2.3. Sample Preparation
2.4. Determination of Total Phenolic Content
2.5. Determination of Antioxidant Capacity
2.6. Microtox Assay
2.7. Statistical Analysis
2.8. LC-MS Analysis of Phenolic Compounds
3. Results and Discussion
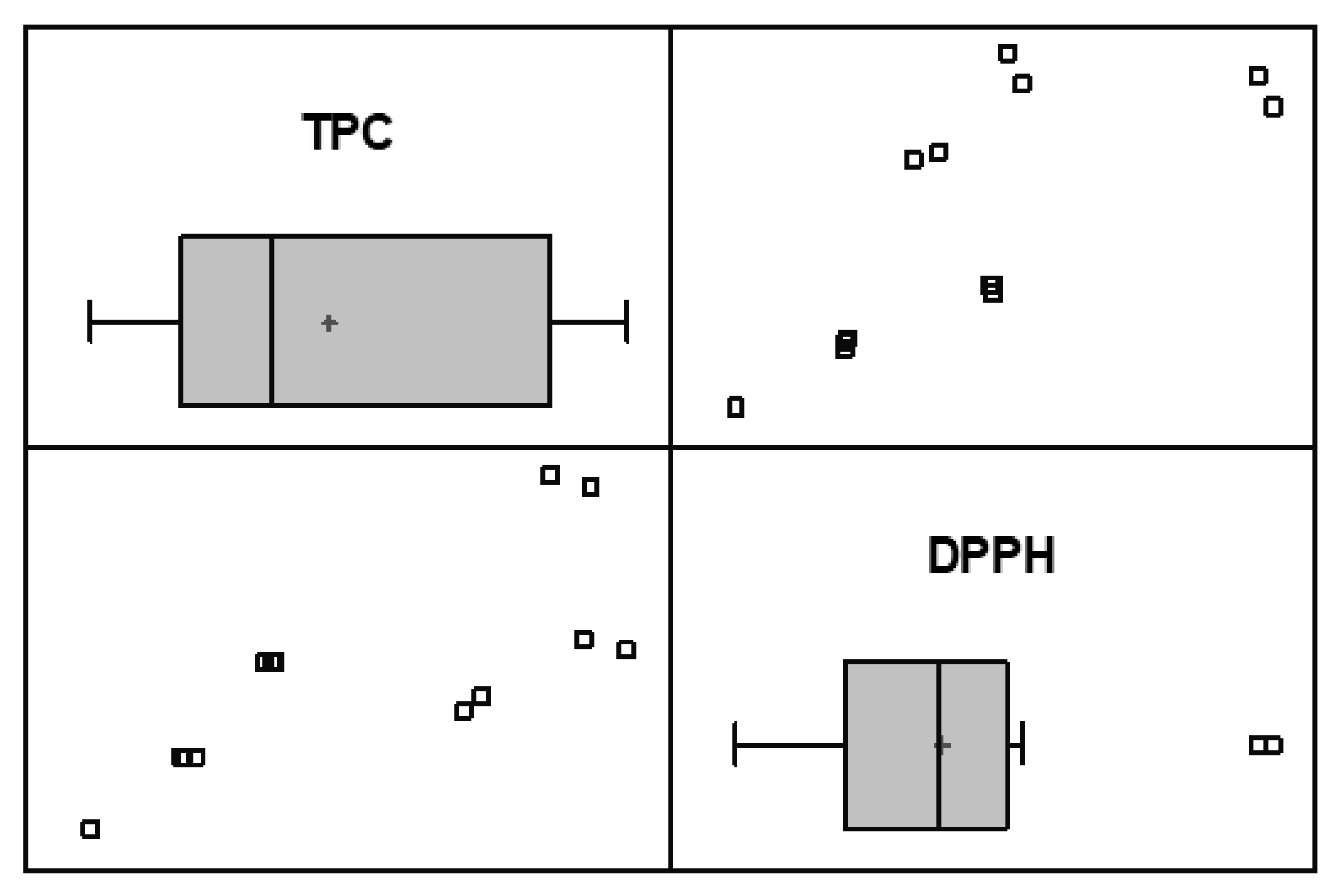
3.1. Total Phenolic Content
3.2. Antioxidant Capacity
3.3. Evaluation of Acute Toxicity towards Vibrio fischeri
3.4. Statistical Data and Correlation between Examined Assays
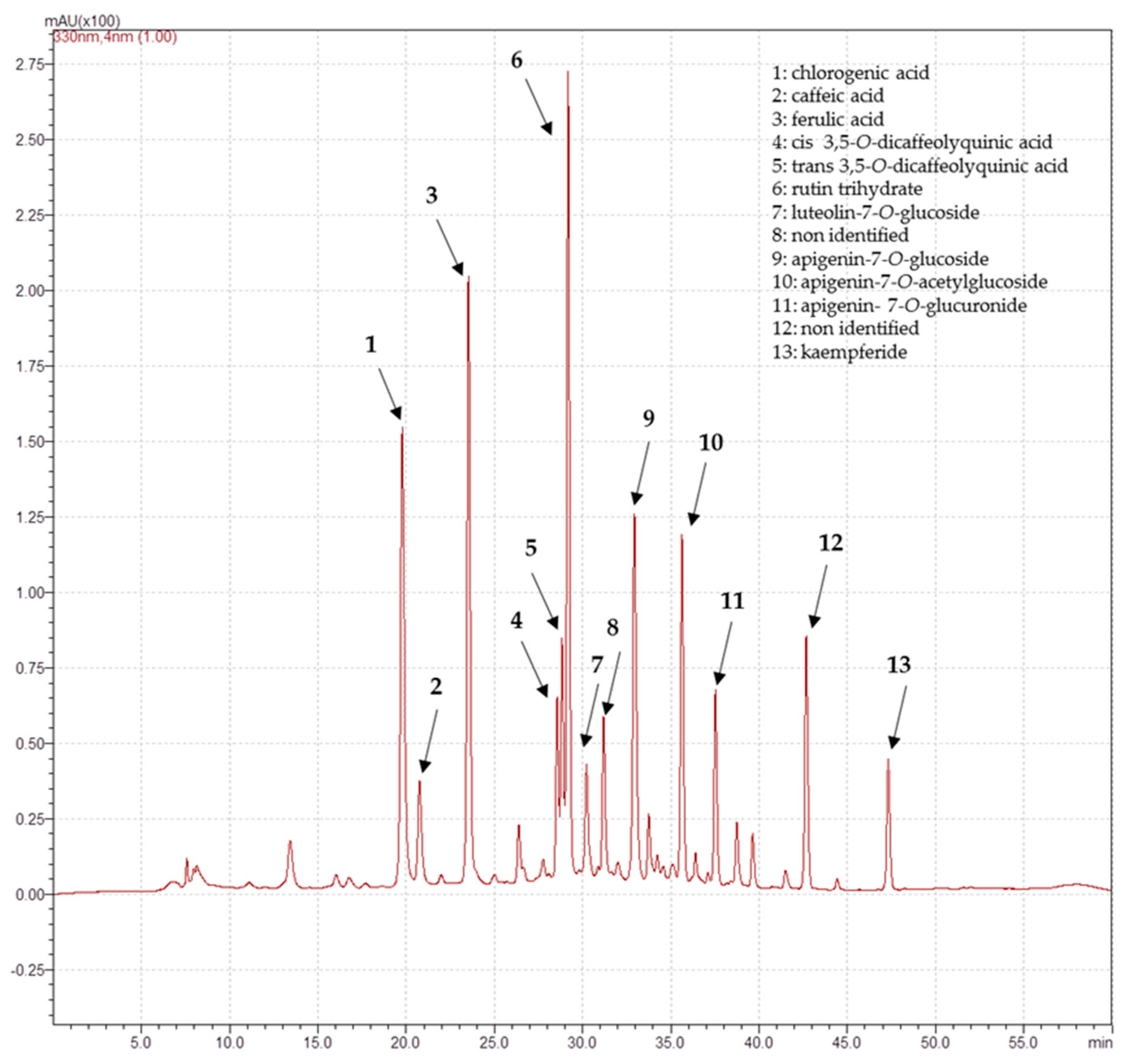
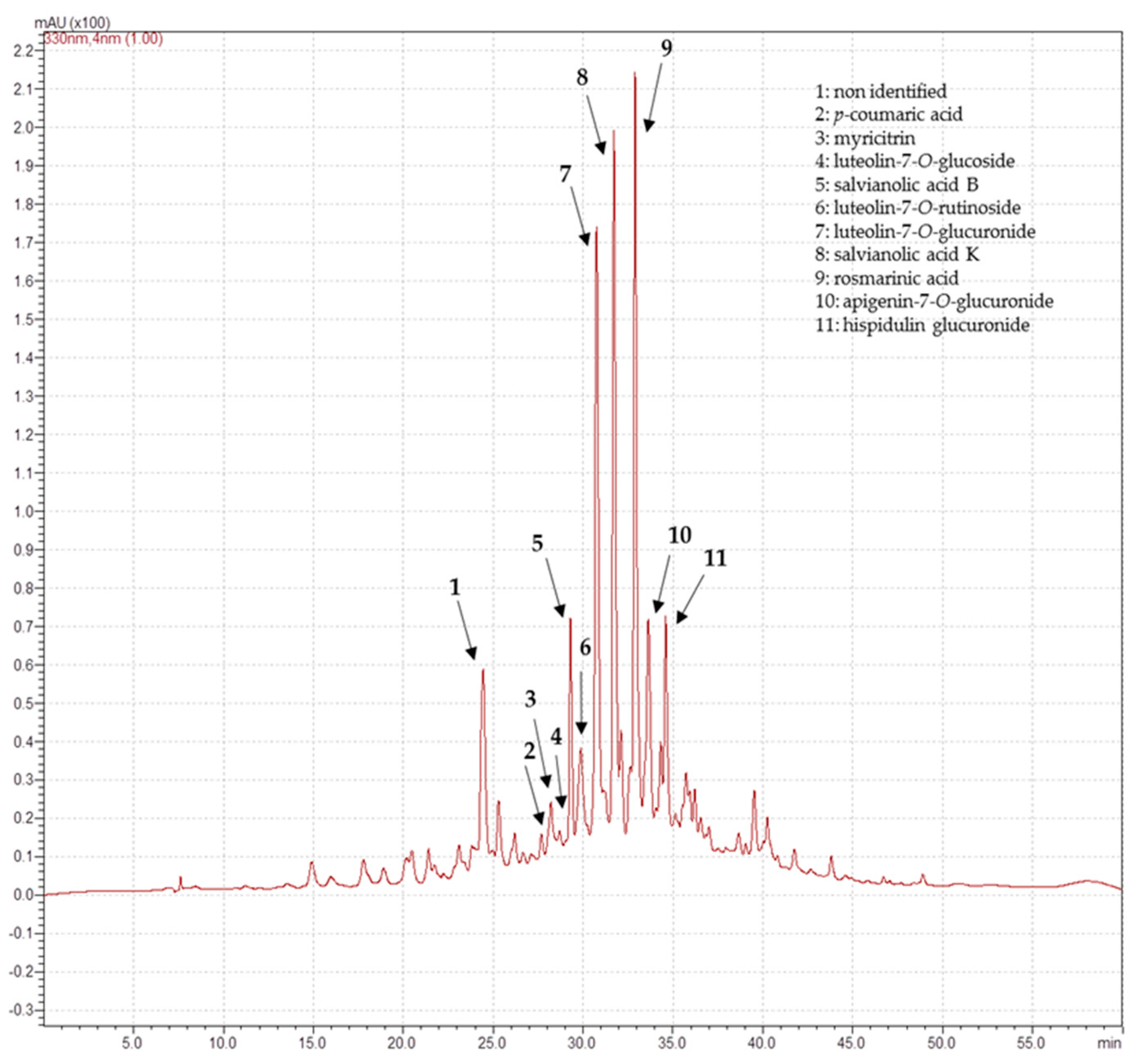
3.5. Determination of Phenolic Compounds
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Horžić, D.; Komes, D.; Belščak, A.; Ganić, K.K.; Iveković, D.; Karlović, D. The composition of polyphenols and methylxanthines in teas and herbal infusions. Food Chem. 2009, 115, 441–448. [Google Scholar] [CrossRef]
- Da Silva Pinto, M. Tea: A new perspective on health benefits. Food Res. Int. 2013, 53, 558–567. [Google Scholar] [CrossRef]
- Ekor, M. The growing use of herbal medicines: Issues relating to adverse reactions and challenges in monitoring safety. Front. Neurol. 2014, 4, 177. [Google Scholar] [CrossRef] [PubMed]
- Abbas, M.; Adil, M.; Ehtisham-ul-Haque, S.; Munir, B.; Yameen, M.; Ghaffar, A.; Shar, G.A.; Asif Tahir, M.; Iqbal, M. Vibrio fischeri bioluminescence inhibition assay for ecotoxicity assessment: A review. Sci. Total Environ. 2018, 626, 1295–1309. [Google Scholar] [CrossRef] [PubMed]
- Skotti, E.; Anastasaki, E.; Kanellou, G.; Polissiou, M.; Tarantilis, P.A. Total phenolic content, antioxidant activity and toxicity of aqueous extracts from selected Greek medicinal and aromatic plants. Ind. Crops Prod. 2014, 53, 46–54. [Google Scholar] [CrossRef]
- Conforti, F.; Ioele, G.; Statti, G.A.; Marrelli, M.; Ragno, G.; Menichini, F. Antiproliferative activity against human tumor cell lines and toxicity test on Mediterranean dietary plants. Food Chem. Toxicol. 2008, 46, 3325–3332. [Google Scholar] [CrossRef]
- McKay, D.L.; Blumberg, J.B. A Review of the Bioactivity and Potential Health Benefits of Chamomile Tea (Matricaria recutita L.). Phyther. Res. 2006, 20, 519–530. [Google Scholar] [CrossRef]
- Guimarães, R.; Barros, L.; Dueñas, M.; Calhelha, R.C.; Carvalho, A.M.; Santos-Buelga, C.; Queiroz, M.J.R.P.; Ferreira, I.C.F.R. Infusion and decoction of wild German chamomile: Bioactivity and characterization of organic acids and phenolic compounds. Food Chem. 2013, 136, 947–954. [Google Scholar] [CrossRef]
- Srivastava, J.K.; Gupta, S. Health Promoting Benefits of Chamomile in the Elderly Population. In Complementary and Alternative Therapies and the Aging Population; Academic Press: Cambridge, MA, USA, 2009; pp. 135–158. ISBN 9780123742285. [Google Scholar]
- Guzelmeric, E.; Ristivojević, P.; Vovk, I.; Milojković-Opsenica, D.; Yesilada, E. Quality assessment of marketed chamomile tea products by a validated HPTLC method combined with multivariate analysis. J. Pharm. Biomed. Anal. 2017, 132, 35–45. [Google Scholar] [CrossRef]
- Martins, N.; Barros, L.; Santos-Buelga, C.; Henriques, M.; Silva, S.; Ferreira, I.C.F.R. Evaluation of bioactive properties and phenolic compounds in different extracts prepared from Salvia officinalis L. Food Chem. 2014, 170, 378–385. [Google Scholar] [CrossRef]
- Dent, M.; Dragović-Uzelac, V.; Penić, M.; Brñić, M.; Bosiljkov, T.; Levaj, B. The effect of extraction solvents, temperature and time on the composition and mass fraction of polyphenols in dalmatian wild sage (Salvia officinalis L.) extracts. Food Technol. Biotechnol. 2013, 51, 84–91. [Google Scholar]
- Kaliora, A.C.; Kogiannou, D.A.A.; Kefalas, P.; Papassideri, I.S.; Kalogeropoulos, N. Phenolic profiles and antioxidant and anticarcinogenic activities of Greek herbal infusions; Balancing delight and chemoprevention? Food Chem. 2014, 142, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Farzaneh, V.; Carvalho, I.S. A review of the health benefit potentials of herbal plant infusions and their mechanism of actions. Ind. Crops Prod. 2015, 65, 247–258. [Google Scholar] [CrossRef]
- Giacometti, J.; Bursać Kovačević, D.; Putnik, P.; Gabrić, D.; Bilušić, T.; Krešić, G.; Stulić, V.; Barba, F.J.; Chemat, F.; Barbosa-Cánovas, G.; et al. Extraction of bioactive compounds and essential oils from mediterranean herbs by conventional and green innovative techniques: A review. Food Res. Int. 2018, 113, 245–262. [Google Scholar] [CrossRef]
- Rivas Romero, M.P.; Estévez Brito, R.; Rodríguez Mellado, J.M.; González-Rodríguez, J.; Ruiz Montoya, M.; Rodríguez-Amaro, R. Exploring the relation between composition of extracts of healthy foods and their antioxidant capacities determined by electrochemical and spectrophotometrical methods. LWT 2018, 95, 157–166. [Google Scholar] [CrossRef]
- Harbourne, N.; Marete, E.; Jacquier, J.C.; O’Riordan, D. Stability of phytochemicals as sources of anti-inflammatory nutraceuticals in beverages-A review. Food Res. Int. 2013, 50, 480–486. [Google Scholar] [CrossRef]
- Cvetanović, A.; Švarc-Gajić, J.; Mašković, P.; Savić, S.; Nikolić, L. Antioxidant and biological activity of chamomile extracts obtained by different techniques: Perspective of using superheated water for isolation of biologically active compounds. Ind. Crops Prod. 2015, 65, 582–591. [Google Scholar] [CrossRef]
- Cvetanović, A.; Švarc-Gajić, J.; Zeković, Z.; Jerković, J.; Zengin, G.; Gašić, U.; Tešić, Ž.; Mašković, P.; Soares, C.; Fatima Barroso, M.; et al. The influence of the extraction temperature on polyphenolic profiles and bioactivity of chamomile (Matricaria chamomilla L.) subcritical water extracts. Food Chem. 2019, 271, 328–337. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods Enzymol. 1998, 299, 152–178. [Google Scholar]
- Huang, D.; Boxin, O.U.; Prior, R.L. The chemistry behind antioxidant capacity assays. J. Agric. Food Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef]
- Surveswaran, S.; Cai, Y.Z.; Corke, H.; Sun, M. Systematic evaluation of natural phenolic antioxidants from 133 Indian medicinal plants. Food Chem. 2007, 102, 938–953. [Google Scholar] [CrossRef]
- Foti, M.C.; Daquino, C.; Geraci, C. Electron-Transfer Reaction of Cinnamic Acids and Their Methyl Esters with the DPPH. Radical in Alcoholic Solutions. J. Org. Chem. 2004, 69, 2309–2314. [Google Scholar] [CrossRef] [PubMed]
- Kováts, N.; Gölöncsér, F.; Ács, A.; Refaey, M. Quantification of the antibacterial properties of Artemisia absinthium, A. vulgaris, Chrysanthemum leucanthemum and Achillea millefolium using the Vibrio fischeri bacterial bioassay. Acta Bot. Hung. 2010, 25, 137–144. [Google Scholar] [CrossRef]
- Sotiropoulou, N.D.; Kokkini, M.K.; Megremi, S.P.; Daferera, D.J.; Skotti, E.P.; Kimbaris, A.C. Determination of ɑ -and β-thujone in Wormwood and Sage Infusions of Greek flora and Estimation of their Average Toxicity. Curr. Res. Nutr Food Sci. 2016, 4, 152–160. [Google Scholar] [CrossRef]
- Mierzejewska, E.; Baran, A.; Urbaniak, M. Biodegradation Potential and Ecotoxicity Assessment in Soil Extracts Amended with Phenoxy Acid Herbicide (2,4-D) and a Structurally-Similar Plant Secondary Metabolite (Ferulic Acid). Bull. Environ. Contam. Toxicol. 2019, 104, 200–205. [Google Scholar] [CrossRef]
- Aoshima, H.; Hirata, S.; Ayabe, S. Antioxidative and anti-hydrogen peroxide activities of various herbal teas. Food Chem. 2007, 103, 617–622. [Google Scholar] [CrossRef]
- Jiménez-Zamora, A.; Delgado-Andrade, C.; Rufián-Henares, J.A. Antioxidant capacity, total phenols and color profile during the storage of selected plants used for infusion. Food Chem. 2016, 199, 339–346. [Google Scholar] [CrossRef]
- Lim, Y.Y.; Murtijaya, J. Antioxidant properties of Phyllanthus amarus extracts as affected by different drying methods. LWT-Food Sci. Technol. 2007, 40, 1664–1669. [Google Scholar] [CrossRef]
- Herrera, T.; Aguilera, Y.; Rebollo-Hernanz, M.; Bravo, E.; Benítez, V.; Martínez-Sáez, N.; Arribas, S.M.; del Castillo, M.D.; Martín-Cabrejas, M.A. Teas and herbal infusions as sources of melatonin and other bioactive non-nutrient components. LWT-Food Sci. Technol. 2018, 89, 65–73. [Google Scholar] [CrossRef]
- Ho, S.C.; Wu, S.P.; Lin, S.M.; Tang, Y.L. Comparison of anti-glycation capacities of several herbal infusions with that of green tea. Food Chem. 2010, 122, 768–774. [Google Scholar] [CrossRef]
- Kogiannou, D.A.A.; Kalogeropoulos, N.; Kefalas, P.; Polissiou, M.G.; Kaliora, A.C. Herbal infusions; their phenolic profile, antioxidant and anti-inflammatory effects in HT29 and PC3 cells. Food Chem. Toxicol. 2013, 61, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Stagos, D.; Portesis, N.; Spanou, C.; Mossialos, D.; Aligiannis, N.; Chaita, E.; Panagoulis, C.; Reri, E.; Skaltsounis, L.; Tsatsakis, A.M.; et al. Correlation of total polyphenolic content with antioxidant and antibacterial activity of 24 extracts from Greek domestic Lamiaceae species. Food Chem. Toxicol. 2012, 50, 4115–4124. [Google Scholar] [CrossRef] [PubMed]
- Gerolis, L.G.L.; Lameiras, F.S.; Krambrock, K.; Neves, M.J. Effect of gamma radiation on antioxidant capacity of green tea, yerba mate, and chamomile tea as evaluated by different methods. Radiat. Phys. Chem. 2017, 130, 177–185. [Google Scholar] [CrossRef]
- Caleja, C.; Barros, L.; Antonio, A.L.; Ciric, A.; Barreira, J.C.M.; Sokovic, M.; Oliveira, M.B.P.P.; Santos-Buelga, C.; Ferreira, I.C.F.R. Development of a functional dairy food: Exploring bioactive and preservation effects of chamomile (Matricaria recutita L.). J. Funct. Foods 2015, 16, 114–124. [Google Scholar] [CrossRef]
- Harbourne, N.; Jacquier, J.C.; O’Riordan, D. Optimisation of the extraction and processing conditions of chamomile (Matricaria chamomilla L.) for incorporation into a beverage. Food Chem. 2009, 115, 15–19. [Google Scholar] [CrossRef]
- Guzelmeric, E.; Vovk, I.; Yesilada, E. Development and validation of an HPTLC method for apigenin 7-O-glucoside in chamomile flowers and its application for fingerprint discrimination of chamomile-like materials. J. Pharm. Biomed. Anal. 2015, 107, 108–118. [Google Scholar] [CrossRef]
- Raal, A.; Orav, A.; Püssa, T.; Valner, C.; Malmiste, B.; Arak, E. Content of essential oil, terpenoids and polyphenols in commercial chamomile (Chamomilla recutita L. Rauschert) teas from different countries. Food Chem. 2012, 131, 632–638. [Google Scholar] [CrossRef]
- Cvetanović, A.; Švarc-Gajić, J.; Zeković, Z.; Gašić, U.; Tešić, Ž.; Zengin, G.; Mašković, P.; Mahomoodally, M.F.; Đurović, S. Subcritical water extraction as a cutting edge technology for the extraction of bioactive compounds from chamomile: Influence of pressure on chemical composition and bioactivity of extracts. Food Chem. 2018, 266, 389–396. [Google Scholar] [CrossRef]
- Cvetkovikj, I.; Stefkov, G.; Acevska, J.; Stanoeva, J.P.; Karapandzova, M.; Stefova, M.; Dimitrovska, A.; Kulevanova, S. Polyphenolic characterization and chromatographic methods for fast assessment of culinary Salvia species from South East Europe. J. Chromatogr. A 2013, 1282, 38–45. [Google Scholar] [CrossRef]

| Temperature of Extraction | Herb | |||
|---|---|---|---|---|
| Chamomile | Sage | |||
| Total phenolic content expressed as mg of gallic acid equivalents (GAE)/mL of extract | ||||
| Median | Range | Median | Range | |
| 25 °C | Nd1 | Nd1 | 0.222 | 0.019 |
| 80 °C | 0.165 | 0.007 | 0.207 | 0.018 |
| 100 °C | 0.041 | 0.006 | 0.079 | 0.005 |
| Antioxidant activity expressed as ug Trolox equivalents (TE)/ mL of extract | ||||
| Median | Range | Median | Range | |
| 25 °C | Nd1 | Nd1 | 6.39 | 0.331 |
| 80 °C | 4.62 | 0.616 | 11.9 | 1.80 |
| 100 °C | 2.53 | 0.022 | 5.84 | 0.022 |
| Rt (min) | λmax (nm) | Molecular Ion [M H]− (m/z) | Proposed Compound | Chamomile % | Sage % |
|---|---|---|---|---|---|
| 19.78 | 327 | 353 | Chlorogenic acid | 12.6 ± 0.2 | |
| 20.75 | 222, 298, 324 | 179 | Caffeic acid | 2.9 ± 0.5 | |
| 23.53 | 242, 323 | 193 | Ferulic acid | 13.3 ± 0.4 | |
| 24.44 | 325 | Ni2 | 5.7 ± 0.2 | ||
| 27.68 | 235, 309 | 163 | p-Coumaric acid | 0.42 ± 0.05 | |
| 28.21 | 231, 256, 352 | 463 | Myricitrin | 1.6 ± 0.4 | |
| 28.55 | 220, 326 | 515 | cis 3,5-O-Dicaffeolyquinic acid1 | 3.1 ± 0.2 | |
| 28.69 | 255, 349 | 447 | Luteolin-7-O-glucoside | 2.5 ± 0.1 | 0.37 ± 0.02 |
| 28.83 | 220, 328 | 515 | trans 3,5-O-Dicaffeolyquinic acid1 | 4.4 ± 0.05 | |
| 29.17 | 255, 356 | 609 | Rutin trihydrate | 15.1 ± 0.1 | |
| 29.30 | 253, 286, 308 | 717 | Salvianolic acid B | 4.1 ± 0.4 | |
| 29.87 | 268, 337 | 593 | Luteolin-7-O-rutinoside1 | 3.2 ± 0.4 | |
| 30.75 | 348 | 461 | Luteolin- 7-O-glucuronide1 | 15 ± 1 | |
| 31.19 | 229, 328 | Ni2 | 3.1 ± 0.2 | ||
| 31.72 | 236, 286, 326 | 555 | Salvianolic acid K1 | 15.4 ± 0.4 | |
| 32.89 | 243, 289, 330 | 359 | Rosmarinic acid | 15 ± 1 | |
| 32.94 | 242, 267, 338 | 431 | Apigenin-7-O-glucoside | 9.1 ± 0.1 | |
| 33.62 | 445 | Apigenin- 7-O-glucuronide1 | 3.8 ± 0.6 | 5 ± 0.3 | |
| 33.74 | 235, 322 | Ni2 | 1.3 ± 0.1 | ||
| 34.59 | 229, 273, 332 | 475 | Hispidulin glucuronide1 | 4.5 ± 0.4 | |
| 35.62 | 238, 267, 332 | 473 | Apigenin-7-O-acetylglucoside1 | 6.9 ± 0.2 | |
| 42.66 | 242, 254, 312 | Ni2 | 5.2 ± 0.3 | ||
| 47.32 | 266, 324, 365 | 299 | Kaempferide | 2.8 ± 0.3 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sotiropoulou, N.S.; Megremi, S.F.; Tarantilis, P. Evaluation of Antioxidant Activity, Toxicity, and Phenolic Profile of Aqueous Extracts of Chamomile (Matricaria chamomilla L.) and Sage (Salvia officinalis L.) Prepared at Different Temperatures. Appl. Sci. 2020, 10, 2270. https://doi.org/10.3390/app10072270
Sotiropoulou NS, Megremi SF, Tarantilis P. Evaluation of Antioxidant Activity, Toxicity, and Phenolic Profile of Aqueous Extracts of Chamomile (Matricaria chamomilla L.) and Sage (Salvia officinalis L.) Prepared at Different Temperatures. Applied Sciences. 2020; 10(7):2270. https://doi.org/10.3390/app10072270
Chicago/Turabian StyleSotiropoulou, Nefeli S., Stiliani F. Megremi, and Petros Tarantilis. 2020. "Evaluation of Antioxidant Activity, Toxicity, and Phenolic Profile of Aqueous Extracts of Chamomile (Matricaria chamomilla L.) and Sage (Salvia officinalis L.) Prepared at Different Temperatures" Applied Sciences 10, no. 7: 2270. https://doi.org/10.3390/app10072270
APA StyleSotiropoulou, N. S., Megremi, S. F., & Tarantilis, P. (2020). Evaluation of Antioxidant Activity, Toxicity, and Phenolic Profile of Aqueous Extracts of Chamomile (Matricaria chamomilla L.) and Sage (Salvia officinalis L.) Prepared at Different Temperatures. Applied Sciences, 10(7), 2270. https://doi.org/10.3390/app10072270






