Explaining Global Turkey Biometric Diversity Through Principal Component Analysis
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Collection
2.2. Ethics Statement
2.3. Data Analysis
3. Results
3.1. Descriptive Statistics
3.2. Correlation Matrix Among Explanatory Variables
3.3. Principal Components Analysis and Model Generated
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ferrari, C.; Marelli, S.P.; Bagnato, A.; Cerolini, S.; Strillacci, M.G. Sequencing and characterization of complete mitogenome DNA of worldwide turkey (Meleagris gallopavo) populations. Anim. Biotechnol. 2024, 35, 2397682. [Google Scholar] [CrossRef]
- Lawal, R.A.; Martin, S.H.; Vanmechelen, K.; Vereijken, A.; Silva, P.; Al-Atiyat, R.M.; Aljumaah, R.S.; Mwacharo, J.M.; Wu, D.-D.; Zhang, Y.-P. The wild species genome ancestry of domestic chickens. BMC Biol. 2020, 18, 13. [Google Scholar] [CrossRef]
- Aslam, M.L.; Bastiaansen, J.W.; Elferink, M.G.; Megens, H.-J.; Crooijmans, R.P.; Blomberg, L.A.; Fleischer, R.C.; Van Tassell, C.P.; Sonstegard, T.S.; Schroeder, S.G. Whole genome SNP discovery and analysis of genetic diversity in Turkey (Meleagris gallopavo). BMC Genom. 2012, 13, 391. [Google Scholar] [CrossRef]
- Thornton, E.K.; Emery, K.F. The uncertain origins of Mesoamerican turkey domestication. J. Archaeol. Method Theory 2017, 24, 328–351. [Google Scholar] [CrossRef]
- Crawford, R. Introduction to Europe and diffusion of domesticated turkeys from the America. Arch. De Zootec. 1992, 41, 2. [Google Scholar]
- De Grossi Mazzorin, I.; Epifani, I. Introduzione e diffusione in Italia di animali esotici dal Nuovo Mondo: Il caso del tacchino (Meleagris gallopavo L.). L’Idomeneo 2016, 2015, 55–74. [Google Scholar]
- Portillo-Salgado, R.; Herrera Haro, J.; Bautista-Ortega, J.; Chay-Canul, A.; Cigarroa Vázquez, F. Guajolote–A poultry genetic resource native to Mexico. World’s Poult. Sci. J. 2022, 78, 467–482. [Google Scholar] [CrossRef]
- Cucchi, T.; Arbuckle, B. Animal domestication: From distant past to current development and issues. Oxf. Univ. Press US 2021, 11, 6–9. [Google Scholar] [CrossRef]
- Sponenberg, D.P.; Martin, A.; Couch, C.; Beranger, J. Conservation strategies for local breed biodiversity. Diversity 2019, 11, 177. [Google Scholar] [CrossRef]
- González Ariza, A.; Arando Arbulu, A.; León Jurado, J.M.; Navas González, F.J.; Delgado Bermejo, J.V.; Camacho Vallejo, M.E. Discriminant canonical tool for differential biometric characterization of multivariety endangered hen breeds. Animals 2021, 11, 2211. [Google Scholar] [CrossRef]
- FAO. Domestic Animal Diversity Information System (DAD-IS). Available online: https://www.fao.org/dad-is/en/ (accessed on 20 March 2025).
- FAO. Phenotypic characterization of animal genetic resources. In FAO Animal Production and Health; FAO: Rome, Italy, 2012; Volume 11. [Google Scholar]
- Marelli, S.P.; Zaniboni, L.; Strillacci, M.G.; Madeddu, M.; Cerolini, S. Morphological characterization of two light Italian turkey breeds. Animals 2022, 12, 571. [Google Scholar] [CrossRef] [PubMed]
- Nestor, K.; Anderson, J.; Patterson, R. Effects of selection for increased body weight, egg production, and shank width on developmental stability in turkeys. Poult. Sci. 2000, 79, 937–945. [Google Scholar] [CrossRef] [PubMed]
- Portillo-Salgado, R.; Cigarroa-Vázquez, F.; Herrera-Haro, J.; Vázquez-Martínez, I. Prediction of body weight of native Mexican guajolotes trough morphometric measurements. ITEA-Inf. Tec. Econ. Agrar. 2020, 116, 150–160. [Google Scholar]
- Portillo-Salgado, R.; Herrera-Haro, J.; Bautista-Ortega, J.; Chay-Canul, A.; Ramírez-Bribiesca, J.; Ortega-Cerrilla, M. Predictive Equations of Carcass Characteristics and Primal Cut Weights of Native Mexican Guajolotes Using Body Measurements. Braz. J. Poult. Sci. 2022, 24, eRBCA-2022-1633. [Google Scholar] [CrossRef]
- Delgado, J.V.; Landi, V.; Barba, C.J.; Fernández, J.; Gómez, M.M.; Camacho, M.E.; Martínez, M.A.; Navas, F.J.; León, J.M. Murciano-Granadina Goat: A Spanish local breed ready for the challenges of the twenty-first century. In Sustainable Goat Production in Adverse Environments: Volume II: Local Goat Breeds; Springer: Berlin/Heidelberg, Germany, 2018; pp. 205–219. [Google Scholar]
- Adeoye, A.; Udoh, J.; Oladepo, A. Sexual differentiation and phenotypic correlations among biometric traits of Nigerian local turkey (Meleagris gallopavo). Niger. J. Anim. Prod. 2019, 46, 28–33. [Google Scholar] [CrossRef]
- Chana, I.; Kabir, M.; Orunmuyi, O.; Musa, A. Effect of Breed and Sex on Body Weight and Linear Body Measurements of Turkeys (Meleagris gallopavo). Am. J. Exp. Agric. 2019, 33, 1–5. [Google Scholar] [CrossRef]
- Dudusola, I.; Bashiru, H.; Awojimi, I. Morphometric traits of turkey (Meleagris gallopavo) as affected by genotype and sex. Niger. J. Anim. Prod. 2020, 47, 1–6. [Google Scholar] [CrossRef]
- Jáuregui Jiménez, R.; Galdámez Cabrera, N.W. Morfoestructura del Pavo Criollo (Meleagris gallopavo) del Traspatio de Guatemala; Universidad de San Carlos de Guatemala: Guatemala City, Guatemala, 2021. [Google Scholar]
- Laryea, D. Phenotypic Characterization of Indigenous Turkeys (Meleagris gallopavo) in the Ashanti, Ahafo, Bono and Bono East Region of Ghana; University of Education: Winneba, Ghana, 2023. [Google Scholar]
- Portillo-Salgado, R.; Herrera-Haro, J.G.; Bautista-Ortega, J.; Sánchez-Villarreal, A.; Cigarroa-Vázquez, F.A.; Chay-Canul, A.J.; Yakubu, A. Study of racial profile of the native Guajolote (Meleagris gallopavo gallopavo) in two regions of Mexico: Morphometric characterization. Trop. Anim. Health Prod. 2022, 54, 93. [Google Scholar] [CrossRef]
- Stange, M.; Núñez-León, D.; Sánchez-Villagra, M.R.; Jensen, P.; Wilson, L.A. Morphological variation under domestication: How variable are chickens? R. Soc. Open Sci. 2018, 5, 180993. [Google Scholar] [CrossRef]
- McLean, A.K.; Gonzalez, F.J.N. Can scientists influence donkey welfare? Historical perspective and a contemporary view. J. Equine Vet. Sci. 2018, 65, 25–32. [Google Scholar] [CrossRef]
- Salgado Pardo, J.I.; Navas González, F.J.; González Ariza, A.; León Jurado, J.M.; Galán Luque, I.; Delgado Bermejo, J.V.; Camacho Vallejo, M.E. Study of meat and carcass quality-related traits in Turkey populations through discriminant canonical analysis. Foods 2023, 12, 3828. [Google Scholar] [CrossRef]
- Schlosser, R.W.; Wendt, O.; Bhavnani, S.; Nail-Chiwetalu, B. Use of information-seeking strategies for developing systematic reviews and engaging in evidence-based practice: The application of traditional and comprehensive Pearl Growing. A review. Int. J. Lang. Commun. Disord. 2006, 41, 567–582. [Google Scholar] [CrossRef]
- Arando, A.; González-Ariza, A.; Lupi, T.; Nogales, S.; León, J.; Navas-González, F.; Delgado, J.; Camacho, M. Comparison of non-linear models to describe the growth in the Andalusian turkey breed. Ital. J. Anim. Sci. 2021, 20, 1156–1167. [Google Scholar] [CrossRef]
- Süzer, B.; Serbest, A.; Arican, I.; Yonkova, P.; Yilmaz, B. A morphometric study on the skull of the turkeys (Meleagris gallopavo). Uludağ Üniversitesi Vet. Fakültesi Derg. 2018, 37, 93–100. [Google Scholar] [CrossRef][Green Version]
- Adeoye, A.; Rotimi, E.; Oluyode, M. Biometric differentiation of local and exotic turkeys (Meleagris gallopavo) in southwest Nigeria. Appl. Trop. Agric. 2017, 22, 63–66. [Google Scholar]
- Adeoye, A.; Oladepo, A. Sexual dimorphism and phenotypic correlations among growth traits of exotic turkey (Meleagris gallopavo). Niger. J. Anim. Prod. 2018, 45, 1–5. [Google Scholar] [CrossRef]
- Havenstein, G.; Nestor, K.; Toelle, V.; Bacon, W. Estimates of Genetic Parameters in Turkeys: 1. Body Weight and Skeletal Characteristics. Poult. Sci. 1988, 67, 1378–1387. [Google Scholar] [CrossRef]
- Johnson, A.; Asmundson, V. Genetic and Environment Factors Affecting Size of Body and Body Parts of Turkeys: 1. The Heritability and Interrelationships of Body Weight and Live Body Measurements. Poult. Sci. 1957, 36, 296–301. [Google Scholar] [CrossRef]
- Kabalin, A.E.; Menčik, S.; Ostović, M.; Štoković, I.; Grgas, A.; Horvath, Š.; Balenović, T.; Sušić, V.; Karadjole, I.; Pavičić, Ž. Morphological characteristics of Dalmatian Turkey: Preliminary results. Maked. J. Anim. Sci. 2012, 2, 277–280. [Google Scholar]
- Janječić, Z.; Mužic, S. Phenotypic traits in Zagorje turkey. Poljoprivreda 2007, 13, 205–208. [Google Scholar]
- Kabir, M.; Habred, H.; Chana, I. Body mass index (BMI) and production performance of two strains of unselected breeder turkey (Meleagris gallopavo) in Zaria, Nigeria. In Proceedings of The 40th Annual Conference of the Nigerian Society for Animal Production (NSAP), NAPRI/ABU, Zaria, Nigeria, 15–19 March 2015; pp. 1–4. [Google Scholar]
- Kumar Roy, K. Study on Phenotipic and Reproductive Characterizationn of Turkey (Meleagris gallopavo) in Chittagong, Bangladesh; Chattogram Veterinary and Animal Sciences University: Chattogram, Bangladesh, 2017. [Google Scholar]
- Aubin, T.S.; Felix, M.; Francis, D.D.; Omer, K.T.R.; Aristide, F.T.; Yacouba, M. First Study of the Turkey (Meleagris Gallopavo) in Cameroon: Assessing Turkey Biodiversity in the Highlands of West-Cameroon. Int. J. Food Sci. Biotechnol. 2017, 2, 16–23. [Google Scholar]
- Available online: http://ir.jkuat.ac.ke/bitstream/handle/123456789/4514/Macharia%2C%20Joyce%20Janet%20Wangui%20MSc%20Biotech.%2C%202018.pdf?sequence=1&isAllowed=y (accessed on 25 July 2025).
- Ríos Utrera, Á.; Román Ponce, S.I.; Vélez Izquierdo, A.; Cabrera Torres, E.; Cantú Covarrubias, A.; Cruz Colín, L.D.l.; Durán Aguilar, M.; Maldonado Jaquez, J.A.; Martínez Silva, F.E.; Martínez Velázquez, G. Analysis of morphological variables in Mexican backyard turkeys (Meleagris gallopavo gallopavo). Rev. Mex. Cienc. Pecu. 2016, 7, 377–389. [Google Scholar] [CrossRef]
- Cigarroa-Vázquez, F.; Herrera-Haro, J.G.; Ruiz-Sesma, B.; Cuca-García, J.M.; Rojas-Martínez, R.I.; Lemus-Flores, C. Caracterización fenotípica del guajolote autóctono (Meleagris gallopavo) y sistema de producción en la región centro norte de Chiapas, México. Agrociencia 2013, 47, 579–591. [Google Scholar]
- Adeoye, A.; Oladepo, A.; Udoh, J. Biometric prediction of live body weight using morphometric traits in Nigerian local turkey (Meleagris gallopavo). Afr. J. Agric. Technol. Environ. Vol 2018, 7, 47–53. [Google Scholar]
- Adeyemi, M.; Oseni, S. Canonical discriminant analysis applied to biometric data of nigerian indigenous turkeys. Arch. Zootec 2018, 67, 7–12. [Google Scholar] [CrossRef]
- Ogah, D. Assessing size and conformation of the body of Nigerian indigenous turkey. Slovak J. Anim. Sci. 2011, 44, 21–27. [Google Scholar]
- Ajayi, O.O.; Yakubu, A.; Jayeola, O.O.; Imumorin, I.G.; Takeet, M.I.; Ozoje, M.O.; Ikeobi, C.O.; Peters, S.O. Multivariate analysis of sexual size dimorphism in local turkeys (Meleagris gallopavo) in Nigeria. Trop. Anim. Health Prod. 2012, 44, 1089–1095. [Google Scholar] [CrossRef]
- Djebbi, A.; M’hamdi, N.; Haddad, I.; Chriki, A. Phenotypic characterization of the indigenous turkey (Meleagris gallopavo) in the North West Regions of Tunisia. Sci. Agric. 2014, 2, 51–56. [Google Scholar] [CrossRef]
- Bhuiyan, M.; Ferdaus, A.J.; Bhuiyan, A.; Hassin, B.; Ali, M. Body conformation, morphometry indices and inheritance pattern of indigenous Dwarf chickens of Bangladesh. J. Poult. Res. 2019, 16, 55–61. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Routledge: London, UK, 1988. [Google Scholar]
- Jolliffe, I.T.; Cadima, J. Principal component analysis: A review and recent developments. Philos. Trans. R Soc. A Math. Phys. Eng. Sci. 2016, 374, 20150202. [Google Scholar] [CrossRef]
- Abdi, H.; Williams, L.J. Principal component analysis. Wiley Interdiscip. Rev. Comput. Stat. 2010, 2, 433–459. [Google Scholar] [CrossRef]
- Groth, D.; Hartmann, S.; Klie, S.; Selbig, J. Principal components analysis. Methods Mol Biol. 2013, 930, 527–547. [Google Scholar] [PubMed]
- Cigarroa-Vázquez, F.; Herrera-Haro, J.G.; Ruiz-Sesma, B.; Cuca-García, J.M.; Rojas-Martínez, R.I.; Lemus-Flores, C. Phenotypic characterization of the indigenous turkey (Meleagris gallopavo) and production system in the north-central region of Chiapas, México. Agrociencia 2013, 47, 579–591. [Google Scholar]
- Timmwood, K.; Hyde, D.; Plopper, C. Lung growth of the turkey, Meleagris gallopavo: II. Comparison of two genetic lines. Am. J. Anat. 1987, 178, 158–169. [Google Scholar] [CrossRef] [PubMed]
- Huchzermeyer, F.W. Broiler ascites: A review of the ascites work done at the poultry section of the Onderstepoort Veterinary Institute 1981–1990. World’s Poult. Sci. J. 2012, 68, 41–50. [Google Scholar] [CrossRef]
- Rajkumar, U.; Muthukumar, M.; Haunshi, S.; Niranjan, M.; Raju, M.; Rama Rao, S.; Chatterjee, R. Comparative evaluation of carcass traits and meat quality in native Aseel chickens and commercial broilers. Br. Poult. Sci. 2016, 57, 339–347. [Google Scholar] [CrossRef]
- Salgado Pardo, J.I.; González Ariza, A.; Navas González, F.J.; León Jurado, J.M.; Díaz Ruiz, E.; Delgado Bermejo, J.V.; Camacho Vallejo, M.E. Discriminant canonical analysis as a tool for genotype traceability testing based on turkey meat and carcass traits. Front. Vet. Sci. 2024, 11, 1326519. [Google Scholar] [CrossRef]
- Velleman, S.; Coy, C.; Anderson, J.; Patterson, R.; Nestor, K. Effect of selection for growth rate on embryonic breast muscle development in turkeys. Poult. Sci. 2002, 81, 1113–1121. [Google Scholar] [CrossRef]
- Livingstone, D.J.; Salt, D.W. Variable selection—Spoilt for choice? Rev. Comput. Chem. 2005, 21, 287–348. [Google Scholar] [CrossRef]
- Yakubu, A.; Jegede, P.; Wheto, M.; Shoyombo, A.J.; Adebambo, A.O.; Popoola, M.A.; Osaiyuwu, O.H.; Olafadehan, O.A.; Alabi, O.O.; Ukim, C.I. Multivariate characterisation of morpho-biometric traits of indigenous helmeted Guinea fowl (Numida meleagris) in Nigeria. PLoS ONE 2022, 17, e0261048. [Google Scholar] [CrossRef]
- Wideman, N.; O’bryan, C.; Crandall, P. Factors affecting poultry meat colour and consumer preferences-A review. World’s Poult. Sci. J. 2016, 72, 353–366. [Google Scholar] [CrossRef]
- Nestor, K.; Bacon, W.; Saif, Y.M.; Renner, P. The influence of genetic increases in shank width on body weight, walking ability, and reproduction of turkeys. Poult. Sci. 1985, 64, 2248–2255. [Google Scholar] [CrossRef] [PubMed]
- Taha, N.; Farran, M. Comparative study of thigh muscles and bones conformation and some carcass traits of local vs. imported turkey strain. Int. J. Poult. Sci 2009, 8, 368–372. [Google Scholar] [CrossRef]
- Mediouni, M.R.; Said, S.; Ameur, A.A.; Gaouar, S.B.S. Phenotypic and morphometric diversity of Indigenous Turkey (Meleagris Gallopavo) from Wilaya of Tlemcen, Northwest of Algeria. Genet. Biodivers. J. 2020, 4, 1–10. [Google Scholar] [CrossRef]
- Kyselý, R.; Meduna, P. The early history of the turkey (Meleagris gallopavo) in the Czech Republic. Archaeol. Anthropol. Sci. 2019, 11, 6431–6449. [Google Scholar] [CrossRef]
- Canales, A.M.; Camacho, M.E.; Beltrán, A.H.; Delgado, J.V.; Landi, V.; Martínez, A.M. Genetic diversity in 10 populations of domestic Turkeys by using microsatellites markers. Poult. Sci. 2023, 102, 102311. [Google Scholar] [CrossRef]
- Smith, E.J.; Xu, J.; Adikari, J. Additional Molecular Evidence that The Royal Palm is Probably a Turkey Breed; VirginiaTech: Blacksburg, VA, USA, 2019. [Google Scholar]
- Russell, K.L. DNA Sequence and Haplotype Variation Analysis of Inflammatory Response Genes NLRX1, IL6, and IL8 in the Turkey (Meleagris gallopavo); Virginia Polytechnic Institute and State University: Blacksburg, VA, USA, 2018. [Google Scholar]
- Goddeeris, B.M.; Goddeeris, B.R. History of the crested turkey, a rare variant of the domesticated turkey (Meleagris gallopavo gallopavo). Arch. Nat. Hist. 2023, 50, 370–384. [Google Scholar] [CrossRef]
- Corso, J.; Hepp, D.; Ledur, M.C.; Peixoto, J.O.; Fagundes, N.J.; Freitas, T.R. Genetic variation of the bronze locus (MC1R) in turkeys from Southern Brazil. Genet. Mol. Biol. 2017, 40, 104–108. [Google Scholar] [CrossRef]

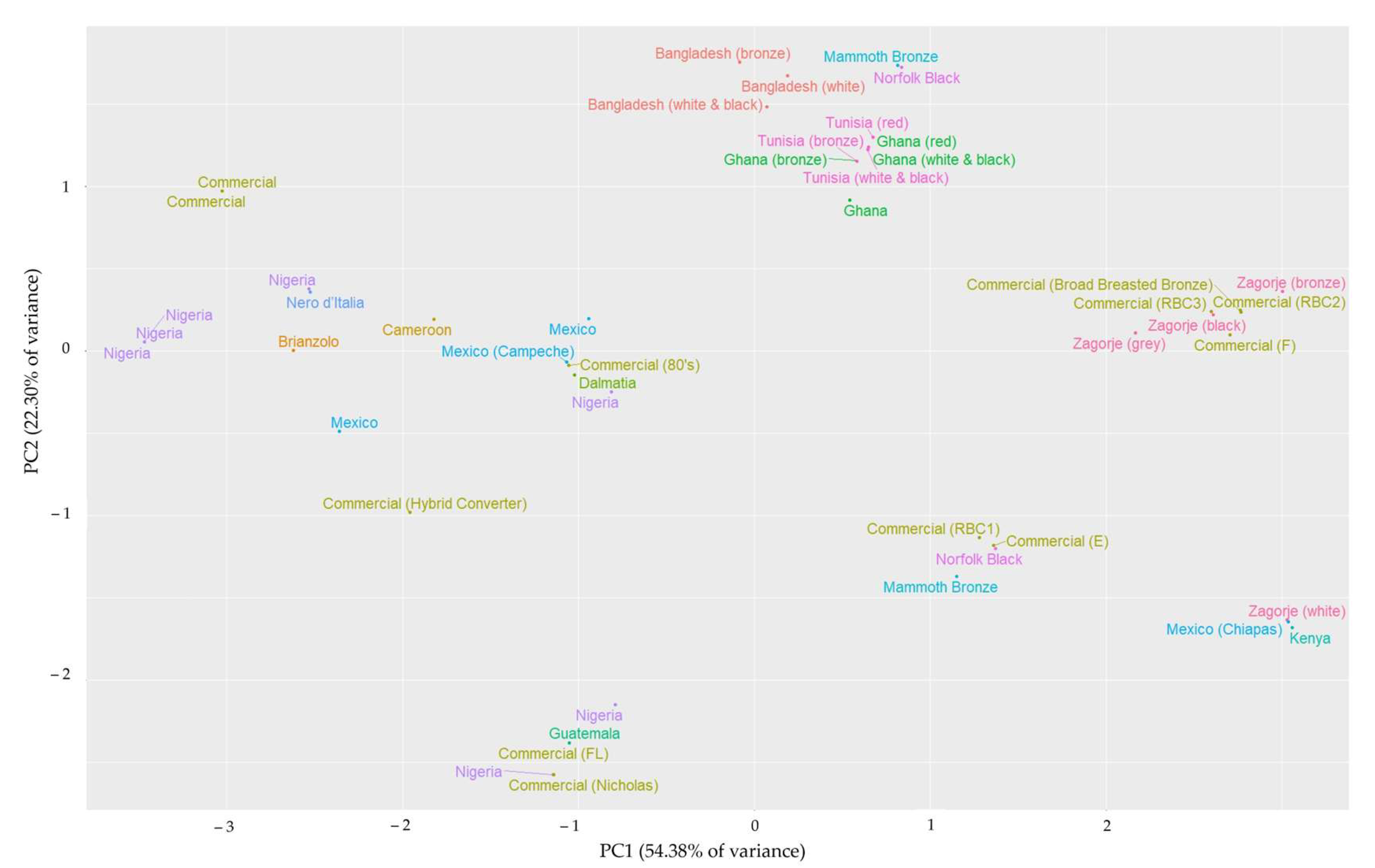

| Abbreviation | Genotype | Distribution | Measured Animals in Studies | References |
|---|---|---|---|---|
| COM | Commercial | Worldwide | 1183 toms and 1149 hens | [14,20,29,30,31,32,33] |
| BRI | Brianzolo | Italy | 17 toms and 29 hens | [13] |
| NDI | Nero d’Italia | Italy | 17 toms and 29 hens | [13] |
| DAL | Dalmatian turkey | Croatia | 10 toms and 20 hens | [34] |
| ZAG | Zagorje turkey | Croatia | 20 toms and 80 hens | [35] |
| MAM | Mammoth Bronze | Worldwide | 41 toms and 41 hens | [19,36] |
| NOR | Norfolk Black | Worldwide | 41 toms and 41 hens | [19,36] |
| BAN | Local Bangladeshi | Bangladesh | 25 toms and 25 hens | [37] |
| CAM | Local Cameroonian | Cameroon | 95 toms and 141 hens | [38] |
| GHA | Local Ghanaian | Ghana | 195 toms and 105 hens | [22] |
| GUA | Local Guatemalan | Guatemala | 181 toms and 210 hens | [21] |
| KEN | Local Kenyan | Kenya | 9 toms and 9 hens | [39] |
| MEX | Local Mexican | Mexico | 526 toms and 332 hens | [16,23,40,41] |
| NIG | Local Nigerian | Nigerian | 382 toms and 475 hens | [18,20,30,42,43,44,45] |
| TUN | Local Tunisian | Tunisia | 93 toms and 108 hens | [46] |
| Abbreviation | Variable | Description |
|---|---|---|
| SkL | Skull length | Distance between the occiput to the tip of the beak |
| SkW | Skull width | Lateral distance between both zygomatic arches |
| BeL | Beak length | Distance from the base to the tip of the beak |
| BeW | Beak width | The maximum lateral distance, at the base of the beak |
| SnL | Snood length | Distance from the base to the tip of the snood |
| W | Weight | Bird’s live weight |
| WS | Wingspan | Distance between the tip of both extended wings |
| BoL | Body length | Distance between the beak’s tip and end of the non-feathered tail |
| NL | Neck length | Maximum distance between the occiput to the neck’s base |
| BrC | Breast circumference | Maximum thoracic perimeter through the keel and wings |
| KL | Keel length | Distance between both keel’s tips |
| ThL | Thigh length | Distance from the joint of the femur to the base of the shank |
| TaL | Tarsus length | Distance from the tibiotarsal joint to the beginning of the toe |
| TaD | Tarsus depth | Cranio-caudal distance at the middle of the metatarsal bone |
| TaW | Tarsus width | Lateral distance at the middle of the metatarsal bone |
| MDL | Middle toe length | Distance from the 3rd phalanx to the tip of the toe |
| SKULL | Skull ratio | Skull length/skull width |
| TARS | Tarsus ratio | Tarsus depth/tarsus width |
| STOCK | Stockiness | (Breast circumference/body length) × 100 |
| MAS | Massiveness | (Weight/body length) × 100 |
| LLEG | Long-leggedness | (Tarsus length/body length)100 |
| BMI | Body mass index | Weight/(body length)2 |
| SHAPE | Shape index | Tarsus length/3√(weight) |
| Commercial | Cameroonian | Kenyan | Dalmatian | Nero D’Italia | Zagorje | Tunisian | Ghanaian | Brianzolo | Bangladeshi | Norfolk Black | Mammoth Bronze | Mexican | Nigerian | Guatemalan | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| W (Kg) | 10.96 ± 3.92 | 7.93 sr | 7.20 sr | 7.18 sr | 6.76 sr | 6.53 ± 0.51 | 6.25 ± 0.12 | 6.04 ± 0.47 | 5.47 sr | 5.66 ± 0.15 | 5.62 ± 2.08 | 5.44 ± 2.46 | 5.36 ± 0.57 | 4.29 ± 1.70 | 3.69 sr |
| WS (cm) | 80.27 | - | - | - | 60.38 sr | - | - | - | 58.45 sr | - | - | - | 69.80 sr | 72.78 ± 0.15 | - |
| BoL (cm) | 65.48 ± 18.69 | 62.83 sr | - | - | 63.31 sr | 29.92 ± 0.91 | 34.58 ± 0.14 | 34.16 ± 0.92 | 62.91 sr | 33.34 ± 1.16 | 33.85 ± 0.67 | 33.61 ± 1.48 | 49.74 ± 14.13 | 50.38 ± 13.90 | 55.01 sr |
| SkL (cm) | 9.60 sr | - | - | 11.35 sr | - | 11.98 ± 0.35 | - | 7.20 sr | - | - | - | - | - | 9.39 sr | 11.25 sr |
| SkW (cm) | 3.62 sr | - | - | 4.35 sr | - | 4.23 ± 0.17 | - | - | - | - | - | - | - | - | 4.26 sr |
| BeL (cm) | 6.33 sr | - | - | - | - | 3.64 ± 0.05 | 2.92 ± 0.01 | - | - | 5.48 ± 0.19 | - | - | 5.06 ± 0.15 | 5.19 ± 0.15 | - |
| BeW (cm) | 2.90 sr | - | - | - | - | - | - | 2.86 ± 0.14 | - | - | - | - | - | - | - |
| SnL (mm) | - | - | - | - | - | - | - | - | - | - | - | - | 8.30 sr | - | 7.38 sr |
| NL (cm) | 25.00 sr | - | - | - | - | - | - | - | - | - | - | - | 27.55 ± 1.73 | 23.72 ± 2.54 | - |
| BrC (cm) | 60.20 sr | 62.02 sr | - | - | 55.00 sr | - | - | - | 53.62 sr | - | 53.78 ± 8.79 | 52.14 ± 12.05 | 53.23 ± 7.93 | 47.35 ± 8.86 | - |
| KL (cm) | 19.70 ± 2.12 | - | - | 16.65 sr | 17.66 sr | 16.27 ± 0.75 | 14.63 ± 0.43 | 14.38 | 15.50 sr | - | - | - | 16.29 sr | 16.52 ± 2.76 | - |
| ThL (cm) | 23.47 ± 2.89 | - | - | 21.10 sr | - | 22.16 ± 0.99 | 22.15 ± 0.42 | 21.80 ± 0.86 | - | 16.69 ± 0.56 | 20.31 sr | 20.29 sr | 15.07 sr | 18.96 ± 4.50 | - |
| TaL (cm) | 17.74 ± 4.32 | 14.57 sr | 16.9 sr4 | 15.77 sr | 12.08 sr | 15.70 ± 0.57 | 13.45 ± 0.03 | 13.40 ± 0.13 | 11.56 sr | 11.06 ± 0.39 | 14.8 ± 2.54 | 14.53 ± 2.17 | 14.14 ± 0.84 | 11.34 ± 2.25 | - |
| TaD (mm) | 22.75 ± 3.17 | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| TaW (cm) | 15.97 ± 4.19 | 25.40 sr | - | - | 10.32 sr | - | - | - | 10.98 sr | - | - | - | 15.35 ± 4.59 | - | - |
| MDL (cm) | 9.95 sr | - | - | - | - | - | - | - | - | - | - | - | - | 8.02 sr | - |
| BdL (cm) | - | 10.35 sr | - | - | - | - | - | - | - | - | - | - | 6.83 sr | - | - |
| Commercial | Cameroonian | Kenyan | Dalmatian | Nero D’italia | Zagorje | Tunisian | Ghanaian | Brianzolo | Bangladeshi | Norfolk Black | Mamooth Bronze | Mexican | Nigerian | Guatemalan | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| W (Kg) | 8.07 ± 2.75 | 4.89 sr | 3.39 sr | 4.26 sr | 3.02 sr | 3.85 ± 0.22 | 3.57 ± 0.11 | 3.69 ± 0.29 | 2.67 sr | 4.01 ± 0.26 | 3.45 ± 0.92 | 3.36 ± 0.48 | 3.26 ± 0.05 | 3.346 ± 1.41 | 2.52 sr |
| WS (cm) | 73.65 sr | - | - | - | 49.71 sr | - | - | - | 49.85 sr | - | - | - | 58.40 sr | 63.45 ± 0.92 | - |
| BoL (cm) | 59.15 ± 16.15 | 53.65 sr | - | - | 53.59 sr | 24.20 ± 0.78 | 28.66 ± 0.36 | 28.16 ± 0.74 | 51.40 sr | 30.62 ± 0.69 | 29.84 ± 0.22 | 30.00 ± 0.28 | 45.51 ± 14.26 | 44.01 ± 12.47 | 46.73 sr |
| SkL (cm) | 11.60 sr | - | - | 9.62 sr | - | 10.01 ± 0.19 | - | 4.76 sr | - | - | - | - | - | 6.71 sr | 9.99 sr |
| SkW (cm) | 4.30 sr | - | - | 3.68 sr | - | 3.51 ± 0.04 | - | - | - | - | - | - | - | - | 3.74 sr |
| BeL (cm) | 5.55 sr | - | - | - | - | 3.30 ± 0.08 | 2.56 ± 0.11 | - | - | 4.36 ± 0.23 | - | - | 4.73 ± 0.34 | 4.79 ± 0.51 | - |
| BeW (cm) | 2.34 sr | - | - | - | - | - | - | 2.50 ± 0.17 | - | - | - | - | - | - | - |
| SnL (mm) | - | - | - | - | - | - | - | - | - | - | - | - | 3.20 sr | - | 1.95 sr |
| NL (cm) | 22.92 sr | - | - | - | - | - | - | - | - | - | - | - | 23.67 ± 1.47 | 18.93 ± 1.91 | - |
| BrC (cm) | 57.24 sr | 48.70 sr | - | - | 41.93 sr | - | - | - | 41.02 sr | - | 42.86 ± 1.61 | 42.24 ± 0.79 | 45.69 ± 11.56 | 41.51 ± 8.43 | - |
| KL (cm) | 17.89 ± 3.38 | - | - | 11.95 sr | 12.50 sr | 12.46 ± 0.27 | 11.49 ± 0.22 | 10.50 sr | 12.00 sr | - | - | - | - | 13.03 ± 1.66 | - |
| ThL (cm) | 21.07 ± 2.19 | - | - | 17.78 sr | - | 18.27 ± 0.49 | 17.79 ± 0.54 | 17.87 ± 0.51 | - | 13.41 ± 0.36 | 16.96 sr | 17.50 sr | 13.01 sr | 15.89 ± 3.66 | - |
| TaL (cm) | 14.54 ± 3.34 | 11.66 sr | 13.50 sr | 12.13 sr | 9.63 sr | 12.60 ± 0.35 | 10.64 ± 0.35 | 10.55 ± 0.37 | 9.59 sr | 9.76 ± 0.22 | 12.16 ± 3.06 | 11.84 ± 2.61 | 11.71 ± 0.79 | 9.23 ± 1.99 | - |
| TaD (mm) | 20.25 ± 3.29 | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| TaW (cm) | 14.54 ± 4.09 | 25.20 sr | - | - | 6.70 sr | - | - | - | 7.58 sr | - | - | - | 9.00 sr | - | - |
| MDL (cm) | 8.17 sr | - | - | - | - | - | - | - | - | - | - | - | - | 7.20 sr | - |
| SKULL | TARS | STOCK | MASS | LLEG | BMI | SHAPE | ||
|---|---|---|---|---|---|---|---|---|
| COM | M | 2.65 sr | 1.43 ± 0.21 | 78.93 sr | 11.59 sr | 21.01 ± 13.76 | 0.101 ± 0.087 | 8.32 ± 1.95 |
| F | 2.69 sr | 1.38 ± 0.16 | 83.59 sr | 11.91 sr | 21.06 ± 15.46 | 0.116 ± 0.100 | 7.44 ± 1.84 | |
| CAM | M | - | - | 98.71 sr | 12.62 sr | 23.19 sr | 0.201 sr | 7.31 sr |
| F | - | - | 90.77 sr | 9.11 sr | 21.73 sr | 0.170 sr | 6.87 sr | |
| KEN | M | - | - | - | - | - | - | 8.77 sr |
| F | - | - | - | - | - | - | 8.99 sr | |
| DAL | M | 2.61 sr | - | - | - | - | - | 8.17 sr |
| F | 2.61 sr | - | - | - | - | - | 7.48 sr | |
| NDI | M | - | - | 86.87 sr | 10.68 sr | 19.08 sr | 0.168 sr | 6.39 sr |
| F | - | - | 78.24 sr | 5.64 sr | 17.97 sr | 0.110 sr | 6.66 sr | |
| ZAG | M | 2.83 ± 0.03 | - | - | 21.84 ± 1.57 | 52.49 ± 2.38 | 0.730 ± 0.056 | 8.40 ± 0.27 |
| F | 2.85 ± 0.05 | - | - | 15.90 ± 0.61 | 52.07 ± 0.66 | 0.657 ± 0.026 | 8.04 ± 0.12 | |
| TUN | M | - | - | - | 18.09 ± 0.36 | 38.91 ± 0.11 | 0.523 ± 0.011 | 7.30 ± 0.06 |
| F | - | - | - | 12.46 ± 0.54 | 37.15 ± 1.57 | 0.435 ± 0.024 | 6.96 ± 0.19 | |
| GHA | M | - | - | - | 17.67 ± 0.98 | 39.23 ± 0.72 | 0.517 ± 0.017 | 7.36 ± 0.14 |
| F | - | - | - | 13.14 ± 1.38 | 37.47 ± 1.02 | 0.468 ± 0.062 | 6.83 ± 0.34 | |
| BRI | M | - | - | 85.23 sr | 8.69 sr | 18.38 sr | 0.138 sr | 6.56 sr |
| F | - | - | 79.81 sr | 5.19 sr | 18.66 sr | 0.100 sr | 6.91 sr | |
| BAN | M | - | - | - | 17.00 ± 0.33 | 33.19 ± 1.63 | 0.510 ± 0.026 | 6.20 ± 0.25 |
| F | - | - | - | 13.09 ± 0.61 | 31.91 ± 1.41 | 0.427 ± 0.014 | 6.15 ± 0.27 | |
| NOR | M | - | - | 158.64 ± 22.81 | 16.56 ± 5.83 | 43.79 ± 8.38 | 0.487 ± 0.162 | 8.54 ± 2.52 |
| F | - | - | 143.61 ± 4.31 | 11.56 ± 3.01 | 40.80 ± 10.57 | 0.387 ± 0.098 | 8.20 ± 2.77 | |
| MAM | M | - | - | 154.49 ± 29.02 | 16.03 ± 6.61 | 43.43 ± 8.38 | 0.473 ± 0.176 | 8.56 ± 2.58 |
| F | - | - | 140.81 ± 3.97 | 11.20 ± 1.71 | 39.44 ± 8.32 | 0.374 ± 0.060 | 7.96 ± 2.12 | |
| MEX | M | - | - | 103.25 ± 18.16 | 11.15 ± 2.46 | 28.89 ± 6.39 | 0.242 ± 0.098 | 8.08 ± 0.41 |
| F | - | - | 89.49 ± 23.04 | 7.50 ± 2.47 | 25.93 ± 6.91 | 0.182 ± 0.111 | 7.89 ± 0.53 | |
| NIG | M | - | - | 93.60 ± 40.93 | 7.18 ± 1.66 | 24.74 ± 11.51 | 0.128 ± 0.097 | 6.81 ± 1.41 |
| F | - | - | 90.39 ± 35.64 | 6.25 ± 1.39 | 22.92 ± 11.02 | 0.125 ± 0.093 | 6.01 ± 1.31 | |
| GUA | M | 2.64 sr | - | - | 6.71 sr | - | 0.122 sr | - |
| F | 2.67 sr | - | - | 5.39 sr | - | 0.12 sr | - |
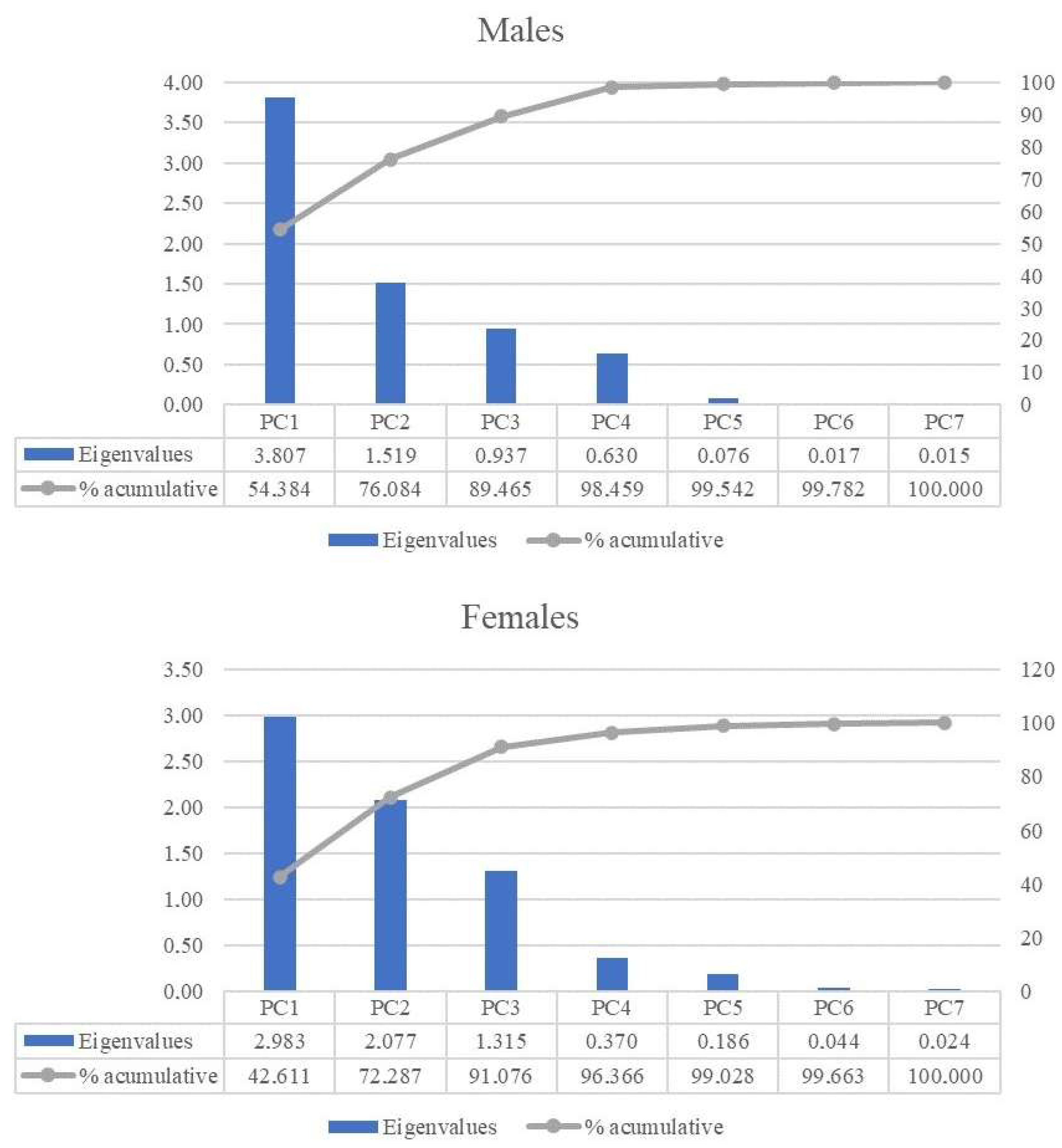
| PC1 | PC2 | PC3 | PC4 | PC5 | ||
| MALES | SKULL | 0.498 | 0.168 | 0.250 | 0.058 | 0.026 |
| TARS | 0.003 | 0.489 | 0.290 | 0.216 | 0.002 | |
| STOCK | 0.471 | 0.064 | 0.388 | 0.055 | 0.021 | |
| MAS | 0.721 | 0.211 | 0.001 | 0.048 | 0.013 | |
| LLEG | 0.943 | 0.032 | 0.000 | 0.014 | 0.000 | |
| BMI | 0.825 | 0.127 | 0.003 | 0.032 | 0.003 | |
| SHAPE | 0.345 | 0.429 | 0.005 | 0.207 | 0.012 | |
| PC1 | PC2 | PC3 | PC4 | PC5 | ||
| FEMALES | SKULL | 0.458 | 0.002 | 0.464 | 0.003 | 0.067 |
| TARS | 0.153 | 0.588 | 0.025 | 0.233 | 0.002 | |
| STOCK | 0.062 | 0.329 | 0.548 | 0.004 | 0.053 | |
| MAS | 0.781 | 0.027 | 0.127 | 0.036 | 0.012 | |
| LLEG | 0.614 | 0.347 | 0.004 | 0.010 | 0.006 | |
| BMI | 0.892 | 0.002 | 0.077 | 0.008 | 0.006 | |
| SHAPE | 0.023 | 0.783 | 0.071 | 0.077 | 0.040 |
| MALES | FEMALES | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| F1 (90.37%) | F2 (97.57%) | F3 (99.06%) | F4 (99.80%) | F5 (100%) | F1 (60.56%) | F2 (79.34%) | F3 (97.04%) | F4 (99.52%) | F3 (100%) | ||
| HEAD | SkL | 0.747 | 0.245 | 0.006 | 0.002 | 0.000 | 0.782 | 0.052 | 0.109 | 0.054 | 0.003 |
| SkW | 0.931 | 0.051 | 0.001 | 0.013 | 0.003 | 0.356 | 0.255 | 0.385 | 0.000 | 0.004 | |
| BeL | 0.942 | 0.001 | 0.057 | 0.000 | 0.000 | 0.789 | 0.141 | 0.005 | 0.063 | 0.002 | |
| BeW | 0.923 | 0.063 | 0.008 | 0.000 | 0.005 | 0.179 | 0.487 | 0.329 | 0.005 | 0.000 | |
| SnL | 0.975 | 0.000 | 0.002 | 0.021 | 0.002 | 0.923 | 0.004 | 0.057 | 0.001 | 0.015 | |
| F1 (41.80%) | F2 (67.41%) | F3 (85.60%) | F4 (94.45%) | F5 (98.36%) | F1 (38.05%) | F2 (64.86%) | F3 (81.57%) | F4 (92.52%) | F5 (96.84%) | ||
| TORSO | W | 0.109 | 0.075 | 0.750 | 0.053 | 0.002 | 0.657 | 0.079 | 0.083 | 0.079 | 0.056 |
| WS | 0.633 | 0.081 | 0.002 | 0.252 | 0.019 | 0.685 | 0.132 | 0.079 | 0.000 | 0.009 | |
| BoL | 0.633 | 0.009 | 0.099 | 0.192 | 0.067 | 0.040 | 0.400 | 0.392 | 0.131 | 0.037 | |
| NL | 0.337 | 0.520 | 0.042 | 0.001 | 0.090 | 0.554 | 0.038 | 0.280 | 0.024 | 0.091 | |
| BrC | 0.465 | 0.327 | 0.143 | 0.022 | 0.000 | 0.088 | 0.701 | 0.090 | 0.028 | 0.068 | |
| KL | 0.332 | 0.525 | 0.055 | 0.010 | 0.057 | 0.260 | 0.259 | 0.078 | 0.395 | 0.000 | |
| F1 (37.87%) | F2 (64.68%) | F3 (85.13%) | F4 (93.99%) | F5 (100%) | F1 (57.95%) | F2 (85.65%) | F3 (95.63%) | F4 (99.54%) | F5 (100%) | ||
| LEGS | ThL | 0.172 | 0.604 | 0.038 | 0.184 | 0.002 | 0.486 | 0.170 | 0.340 | 0.004 | 0.000 |
| TaL | 0.734 | 0.050 | 0.059 | 0.006 | 0.152 | 0.710 | 0.093 | 0.121 | 0.074 | 0.001 | |
| TaD | 0.592 | 0.179 | 0.073 | 0.035 | 0.121 | 0.568 | 0.396 | 0.000 | 0.027 | 0.009 | |
| TaW | 0.382 | 0.036 | 0.435 | 0.133 | 0.014 | 0.554 | 0.427 | 0.001 | 0.008 | 0.011 | |
| MDL | 0.013 | 0.471 | 0.418 | 0.086 | 0.012 | 0.580 | 0.299 | 0.037 | 0.082 | 0.002 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salgado Pardo, J.I.; González Ariza, A.; Carranco Medina, L.; León Jurado, J.M.; Delgado Bermejo, J.V.; Marelli, S.P.; Cerolini, S.; Zaniboni, L.; Camacho Vallejo, M.E. Explaining Global Turkey Biometric Diversity Through Principal Component Analysis. Animals 2025, 15, 2537. https://doi.org/10.3390/ani15172537
Salgado Pardo JI, González Ariza A, Carranco Medina L, León Jurado JM, Delgado Bermejo JV, Marelli SP, Cerolini S, Zaniboni L, Camacho Vallejo ME. Explaining Global Turkey Biometric Diversity Through Principal Component Analysis. Animals. 2025; 15(17):2537. https://doi.org/10.3390/ani15172537
Chicago/Turabian StyleSalgado Pardo, José Ignacio, Antonio González Ariza, Laura Carranco Medina, José Manuel León Jurado, Juan Vicente Delgado Bermejo, Stefano Paolo Marelli, Silvia Cerolini, Luisa Zaniboni, and María Esperanza Camacho Vallejo. 2025. "Explaining Global Turkey Biometric Diversity Through Principal Component Analysis" Animals 15, no. 17: 2537. https://doi.org/10.3390/ani15172537
APA StyleSalgado Pardo, J. I., González Ariza, A., Carranco Medina, L., León Jurado, J. M., Delgado Bermejo, J. V., Marelli, S. P., Cerolini, S., Zaniboni, L., & Camacho Vallejo, M. E. (2025). Explaining Global Turkey Biometric Diversity Through Principal Component Analysis. Animals, 15(17), 2537. https://doi.org/10.3390/ani15172537








