Effects of Different Concentrations of Lactobacillus plantarum and Bacillus licheniformis on Silage Quality, In Vitro Fermentation and Microbial Community of Hybrid Pennisetum
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Silage Material and Preparation
2.2. Sample Preparation
2.3. In Vitro Fermentation
2.3.1. Animals and Experimental Diets
2.3.2. Operating Steps
2.4. Analyses of Bacterial Community
2.5. Statistical Analysis
3. Results
3.1. Chemical Compositions of HP Silage
3.2. Fermentation Quality of HP Silage
3.3. Gas Production of HP Silage
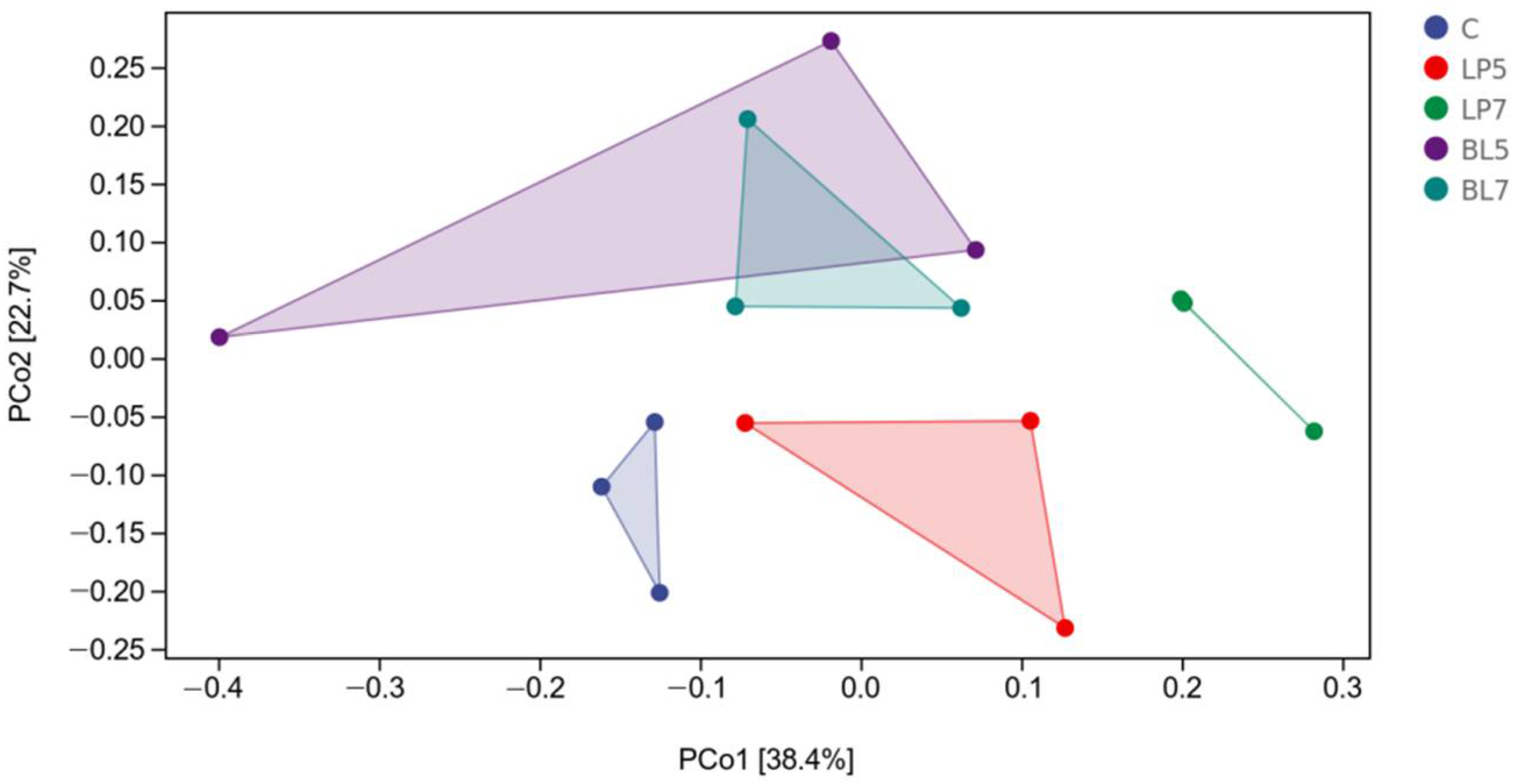
3.4. Bacterial Community of HP Silage
3.4.1. Bacterial Diversity
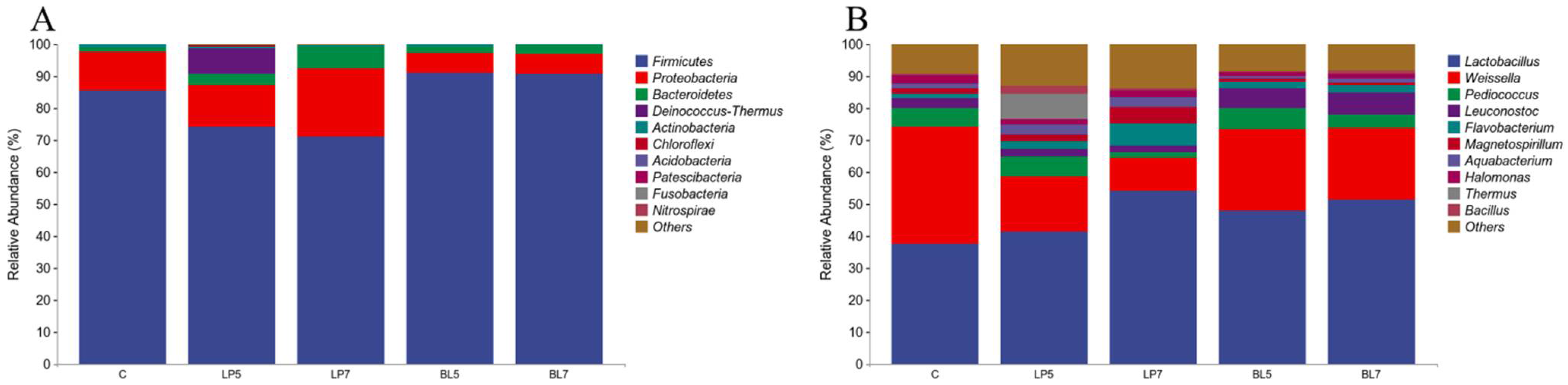
3.4.2. Bacterial Composition
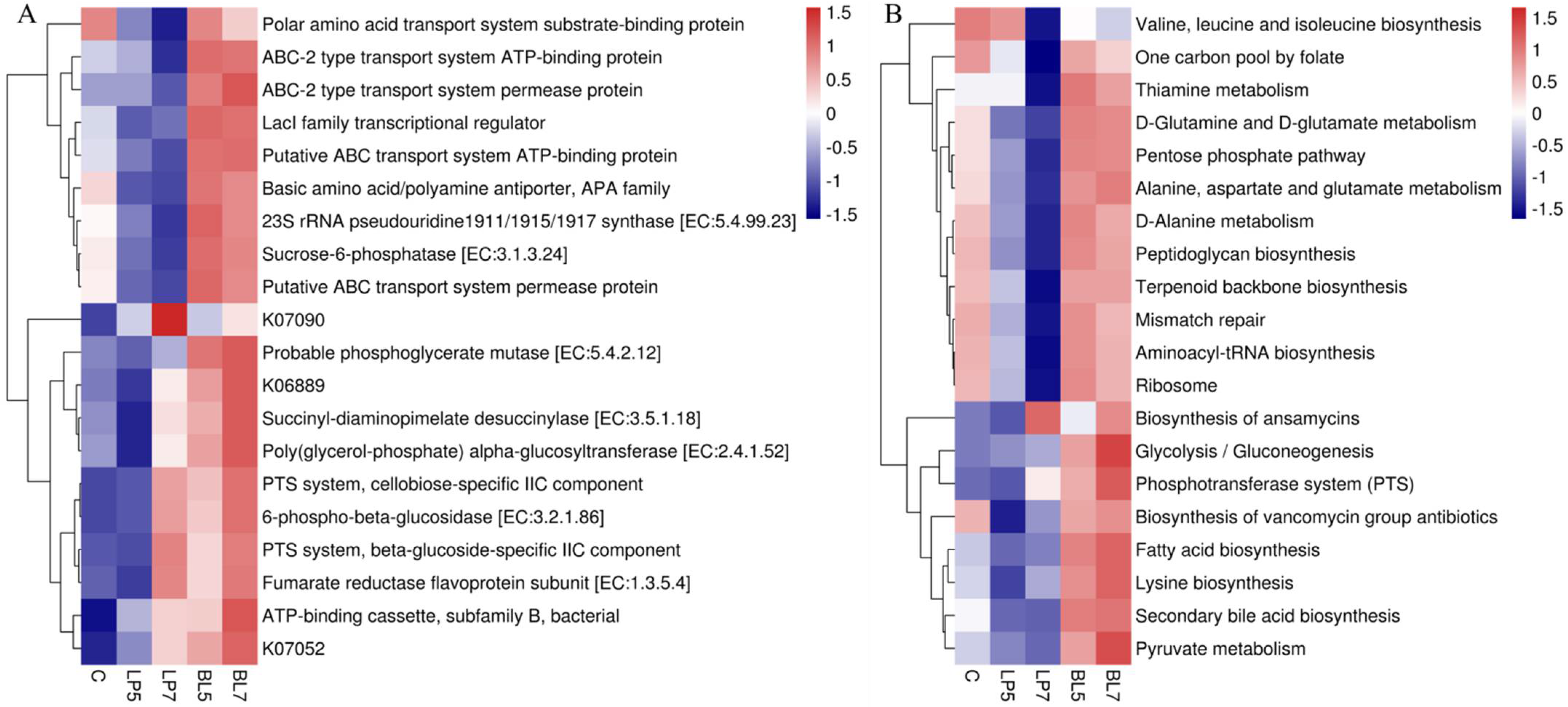
3.4.3. Predicted Functions and Pathways
4. Discussions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Li, Y.; Wang, J.; Mei, J.; Huang, L.; Liu, H. Effects of Mulberry Branch and Leaves Silage on Microbial Community, Rumen Fermentation Characteristics, and Milk Yield in Lactating Dairy Cows. Fermentation 2022, 8, 86. [Google Scholar] [CrossRef]
- Xihui, K.; Yi, Z.; Bing, S.; Yongming, S.; Lianhua, L.; Yu, H.; Xiaoying, K.; Xinjian, L.; Zhenhong, Y. The effect of mechanical pretreatment on the anaerobic digestion of Hybrid Pennisetum. Fuel 2019, 252, 469–474. [Google Scholar] [CrossRef]
- Jingfeng, W.; Donglin, X.; Xincun, H.; Juying, W.; Xifeng, F.; Kena, L.; Junhua, Z. Structural properties and hydrolysabilities of Chinese Pennisetum and Hybrid Pennisetum: Effect of aqueous ammonia pretreatment. Bioresour. Technol. 2016, 199, 211–219. [Google Scholar] [CrossRef]
- Shah, A.A.; Wu, J.; Qian, C.; Liu, Z.; Mobashar, M.; Tao, Z.; Zhang, X.; Zhong, X. Ensiling of whole-plant hybrid pennisetum with natamycin and Lactobacillus plantarum impacts on fermentation characteristics and meta-genomic microbial community at low temperature. J. Sci. Food Agric. 2020, 100, 3378–3385. [Google Scholar] [CrossRef]
- Mu, L.; Xie, Z.; Hu, L.; Chen, G.; Zhang, Z. Lactobacillus plantarum and molasses alter dynamic chemical composition, microbial community, and aerobic stability of mixed (amaranth and rice straw) silage. J. Sci. Food Agric. 2021, 101, 5225–5235. [Google Scholar] [CrossRef]
- Chen, L.; Bao, X.; Guo, G.; Huo, W.; Li, Q.; Xu, Q.; Wang, C.; Liu, Q. Evaluation of gallnut tannin and Lactobacillus plantarum as natural modifiers for alfalfa silage: Ensiling characteristics, in vitro ruminal methane production, fermentation profile and microbiota. J. Appl. Microbiol. 2021, 132, 907–918. [Google Scholar] [CrossRef]
- Assar, A.S.; Chen, Q.; Juanzi, W.; Zhiwei, L.; Salman, K.; Zhujun, T.; Xiaomin, Z.; Irfan, U.K.; Xiaoxian, Z. Effects of natamycin and Lactobacillus plantarum on the chemical composition, microbial community, and aerobic stability of Hybrid pennisetum at different temperatures. RSC Adv. 2020, 10, 8692–8702. [Google Scholar] [CrossRef] [Green Version]
- Muck, R.E.; Nadeau, E.M.G.; McAllister, T.A.; Contreras-Govea, F.E.; Santos, M.C.; Kung, L., Jr. Silage review: Recent advances and future uses of silage additives. J. Dairy Sci. 2018, 101, 3980–4000. [Google Scholar] [CrossRef]
- Blajman, J.E.; Paez, R.B.; Vinderola, C.G.; Lingua, M.S.; Signorini, M.L. A meta-analysis on the effectiveness of homofermentative and heterofermentative lactic acid bacteria for corn silage. J. Appl. Microbiol. 2018, 125, 1655–1669. [Google Scholar] [CrossRef]
- Yang, G.; Yang, D.; Wang, X.; Cao, W. A novel thermostable cellulase-producing Bacillus licheniformis A5 acts synergistically with Bacillus subtilis B2 to improve degradation of Chinese distillers’ grains. Bioresource Technol. 2021, 325, 124729. [Google Scholar] [CrossRef]
- Pattnaik, P.; Kaushik, J.K.; Grover, S.; Batish, V.K. Purification and characterization of a bacteriocin-like compound (Lichenin) produced anaerobically by Bacillus licheniformis isolated from water buffalo. J. Appl. Microbiol. 2001, 91, 636–645. [Google Scholar] [CrossRef]
- Bampidis, V.; Azimonti, G.; Bastos, M.D.L.; Christensen, H.; Dusemund, B.; Kouba, M.; Kos, D.M.; Lópezalonso, M.; López, P.S.; Marcon, F.; et al. Safety and efficacy of Bacillus licheniformis DSM 32457 as a silage additive for all animal species. EFSA J. 2019, 17, 5787. [Google Scholar] [CrossRef]
- Mara, G.; Becze, A.; Varga, O.; Laslo, É.; Oancea, F. Screening of multifunctional bacterial inoculants with lignocellulose degradation ability for agricultural applications. Studia Univ. Vasile Goldis Ser. Stiintele Vietii 2016, 26, 225–233. [Google Scholar]
- Oliveira, C.A.; Sousa, D.O.; Penso, J.F.; Menegucci, P.F.; Silva, L.F.P. Effect of different doses of a Bacillus-based probiotic on the in vitro digestibility of concentrates and forages. J. Anim. Sci. 2016, 94, 654. [Google Scholar] [CrossRef]
- Shao, T.; Shimojo, M.; Wang, T.; Masuda, Y. Effect of Additives on the Fermentation Quality and Residual Mono- and Disaccharides Compositions of Forage Oats (Avena sativa L.) and Italian Ryegrass (Lolium multiflorum Lam.) Silages. Asian Austral. J. Anim. 2005, 18, 1582–1588. [Google Scholar] [CrossRef]
- Josefa, M.; Antonio, M.T.; Fuensanta, H.; María, D.M. A comparative study on the determination of lactic acid in silage juice by colorimetric, high-performance liquid chromatography and enzymatic methods. J. Sci. Food Agric. 1999, 79, 1722–1726. [Google Scholar] [CrossRef]
- Ke, W.C.; Ding, W.R.; Xu, D.M.; Ding, L.M.; Zhang, P.; Li, F.D.; Guo, X.S. Effects of addition of malic or citric acids on fermentation quality and chemical characteristics of alfalfa silage. J. Dairy Sci. 2017, 100, 8958–8966. [Google Scholar] [CrossRef] [Green Version]
- Chances In Official Methods Of Analysis. J. Aoac Int. 1990, 73, 189–191. [CrossRef]
- Van Soest, P.J.; Robertson, J.B.; Lewis, B.A. Methods for Dietary Fiber, Neutral Detergent Fiber, and Nonstarch Polysaccharides in Relation to Animal Nutrition. J. Dairy Sci. 1991, 74, 3583–3597. [Google Scholar] [CrossRef]
- Owens, V.N.; Albrecht, K.A.; Muck, R.E.; Duke, S.H. Protein Degradation and Fermentation Characteristics of Red Clover and Alfalfa Silage Harvested with Varying Levels of Total Nonstructural Carbohydrates. Crop Sci. 1999, 39, 1873–1880. [Google Scholar] [CrossRef]
- Menke, K.H.; Raab, L.; Salewski, A.; Steingass, H.; Fritz, D.; Schneider, W. The estimation of the digestibility and metabolizable energy content of ruminant feedingstuffs from the gas production when they are incubated with rumen liquor in vitro. J. Agric. Sci. 1979, 93, 217–222. [Google Scholar] [CrossRef] [Green Version]
- Roni, R.; Iman, R.; Yantyati, W.; Komang, G.W.; Bambang, P.; Mitsuo, S.; Moriya, O. Fermentation Characteristics and Microbial Diversity of Tropical Grass-legumes Silages. Asian Austral. J. Anim. 2015, 28, 515–518. [Google Scholar] [CrossRef] [Green Version]
- Borreani, G.; Tabacco, E.; Schmidt, R.J.; Holmes, B.J.; Muck, R.E. Silage review: Factors affecting dry matter and quality losses in silages. J. Dairy Sci. 2018, 101, 3952–3979. [Google Scholar] [CrossRef] [Green Version]
- Daniel, O. Protein-surfactant interactions: A tale of many states. BBA-Proteins Proteom. 2011, 1814, 562–591. [Google Scholar] [CrossRef]
- Zhang, G.; Chen, Y.; Li, Q.; Zhou, J.; Li, J.; Du, G. Growth-coupled evolution and high-throughput screening assisted rapid enhancement for amylase-producing Bacillus licheniformis. Bioresour. Technol. 2021, 337, 125467. [Google Scholar] [CrossRef]
- Grant, R.J.; Ferraretto, L.F. Silage review: Silage feeding management: Silage characteristics and dairy cow feeding behavior. J. Dairy Sci. 2018, 101, 4111–4121. [Google Scholar] [CrossRef]
- Zhang, Y.X.; Huang, Z.P.; Vyas, D.; Adesogan, A.T.; Franco, M.; Ke, W.C.; Li, F.H.; Bai, J.; Ding, Z.T.; Guo, X.S. Antioxidant status, chemical composition and fermentation profile of alfalfa silage ensiled at two dry matter contents with a novel Lactobacillus plantarum strain with high-antioxidant activity. Anim. Feed. Sci. Technol. 2020, 272, 114751. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, C.; Zhou, W.; Yang, F.; Chen, X.; Zhang, Q. Effects of Wilting and Lactobacillus plantarum Addition on the Fermentation Quality and Microbial Community of Moringa oleifera Leaf Silage. Front. Microbiol. 2018, 9, 1817. [Google Scholar] [CrossRef] [Green Version]
- Amer, S.; Hassanat, F.; Berthiaume, R.; Seguin, P.; Mustafa, A.F. Effects of water soluble carbohydrate content on ensiling characteristics, chemical composition and in vitro gas production of forage millet and forage sorghum silages. Anim. Feed. Sci. Technol. 2012, 177, 23–29. [Google Scholar] [CrossRef]
- Zi, X.; Li, M.; Chen, Y.; Lv, R.; Zhou, H.; Tang, J. Effects of Citric Acid and Lactobacillus plantarum on Silage Quality and Bacterial Diversity of King Grass Silage. Front. Microbiol. 2021, 12, 631096. [Google Scholar] [CrossRef]
- Kung, L., Jr.; Shaver, R.D.; Grant, R.J.; Schmidt, R.J. Silage review: Interpretation of chemical, microbial, and organoleptic components of silages. J. Dairy Sci. 2018, 101, 4020–4033. [Google Scholar] [CrossRef]
- Liu, Q.H.; Shao, T.; Bai, Y.F. The effect of fibrolytic enzyme, Lactobacillus plantarum and two food antioxidants on the fermentation quality, alpha-tocopherol and beta-carotene of high moisture napier grass silage ensiled at different temperatures. Anim. Feed Sci. Tech. 2016, 221, 1–11. [Google Scholar] [CrossRef]
- Li, J.; Yuan, X.; Desta, S.T.; Dong, Z.; Mugabe, W.; Shao, T. Characterization of Enterococcus faecalis JF85 and Enterococcus faecium Y83 isolated from Tibetan yak (Bos grunniens) for ensiling Pennisetum sinese. Bioresour. Technol. 2018, 257, 76–83. [Google Scholar] [CrossRef]
- Cheng, W.; Liwen, H.; Yaqi, X.; Wei, Z.; Fuyu, Y.; Xiaoyang, C.; Qing, Z. Fermentation quality and microbial community of alfalfa and stylo silage mixed with Moringa oleifera leaves. Bioresour. Technol. 2019, 284, 240–247. [Google Scholar] [CrossRef]
- Dong, Z.; Shao, T.; Li, J.; Yang, L.; Yuan, X. Effect of alfalfa microbiota on fermentation quality and bacterial community succession in fresh or sterile Napier grass silages. J. Dairy Sci. 2020, 103, 4288–4301. [Google Scholar] [CrossRef]
- Tian, H.; Wang, Y.; Liu, Z.; Hu, Z.; Guo, Y.; Deng, M.; Liu, G.; Sun, B. Effects of Malic Acid and Sucrose on the Fermentation Parameters, CNCPS Nitrogen Fractions, and Bacterial Community of Moringa oleifera Leaves Silage. Microorganisms 2021, 9, 2102. [Google Scholar] [CrossRef]
- Musa, B.B.; Duan, Y.; Khawar, H.; Sun, Q.; Ren, Z.; Elsiddig, M.M.A.; Abbasi, I.H.R.; Yang, X. Bacillus subtilis B21 and Bacillus licheniformis B26 improve intestinal health and performance of broiler chickens with Clostridium perfringens-induced necrotic enteritis. J. Anim. Physiol. Anim. Nutr. 2019, 103, 1039–1049. [Google Scholar] [CrossRef]
- Chen, Y.C.; Yu, Y.H. Bacillus licheniformis-fermented products improve growth performance and the fecal microbiota community in broilers. Poult. Sci. 2020, 99, 1432–1443. [Google Scholar] [CrossRef]
- Chen, L.; Bai, S.; You, M.; Xiao, B.; Li, P.; Cai, Y. Effect of a low temperature tolerant lactic acid bacteria inoculant on the fermentation quality and bacterial community of oat round bale silage. Anim. Feed. Sci. Technol. 2020, 269, 114669. [Google Scholar] [CrossRef]
- Romero, J.J.; Zhao, Y.; Balseca-Paredes, M.A.; Tiezzi, F.; Gutierrez-Rodriguez, E.; Castillo, M.S. Laboratory silo type and inoculation effects on nutritional composition, fermentation, and bacterial and fungal communities of oat silage. J. Dairy Sci. 2017, 100, 1812–1828. [Google Scholar] [CrossRef] [Green Version]
- Silva, V.P.; Pereira, O.G.; Leandro, E.S.; Da Silva, T.C.; Ribeiro, K.G.; Mantovani, H.C.; Santos, S.A. Effects of lactic acid bacteria with bacteriocinogenic potential on the fermentation profile and chemical composition of alfalfa silage in tropical conditions. J. Dairy Sci. 2016, 99, 1895–1902. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.A.; Liu, Z.; Qian, C.; Wu, J.; Zhong, X.; Umm-E-Kalsoom. Effect of endophytic Bacillus megaterium colonization on structure strengthening, microbial community, chemical composition and stabilization properties of Hybrid Pennisetum. J. Sci. Food Agric. 2020, 100, 1164–1173. [Google Scholar] [CrossRef] [PubMed]
- Graf, K.; Ulrich, A.; Idler, C.; Klocke, M. Bacterial community dynamics during ensiling of perennial ryegrass at two compaction levels monitored by terminal restriction fragment length polymorphism. J. Appl. Microbiol. 2016, 120, 1479–1497. [Google Scholar] [CrossRef] [Green Version]
- Ogunade, I.M.; Jiang, Y.; Cervantes, A.P.; Kim, D.H.; Oliveira, A.S.; Vyas, D.; Weinberg, Z.G.; Jeong, K.C.; Adesogan, A.T. Bacterial diversity and composition of alfalfa silage as analyzed by Illumina MiSeq sequencing: Effects of Escherichia coli O157:H7 and silage additives. J. Dairy Sci. 2018, 101, 2048–2059. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Wang, M.; Chen, J.; Xu, Z.; Wang, S.; Xia, X.; Liu, D.; Wang, S.; Xie, C.; Wu, J.; et al. Preventive Effects of Bacillus licheniformis on Heat Stroke in Rats by Sustaining Intestinal Barrier Function and Modulating Gut Microbiota. Front. Microbiol. 2021, 12, 630841. [Google Scholar] [CrossRef]
- Ul, H.Z.; Al, T.R.; Alnaimi, H.; Migheli, Q.; Jaoua, S. Investigation and Application of Bacillus licheniformis Volatile Compounds for the Biological Control of Toxigenic Aspergillus and Penicillium spp. ACS Omega 2019, 4, 17186–17193. [Google Scholar] [CrossRef]
- Bernardet, J.F.; Segers, P.; Vancanneyt, M.; Berthe, F.; Kersters, K.; Vandamme, P. Cutting a Gordian Knot: Emended Classification and Description of the Genus Flavobacterium, Emended Description of the Family Flavobacteriaceae, and Proposal of Flavobacterium hydatis nom. nov. (Basonym, Cytophaga aquatilis Strohl and Tait 1978). Int. J. Syst. Bacteriol. 1996, 46, 128–148. [Google Scholar] [CrossRef]
- Ayasan, T.; Ülger, I.; Kaliber, M.; Ergül, S.; Inci, H.; Mart, D.; Türkeri, M. Comparison of in vitro gas production, nutritive value, metabolizable energy and organic matter digestibility of some chickpea varieties. Iran. J. Appl. Anim. Sci. 2018, 8, 517–522. [Google Scholar] [CrossRef]
- Calsamiglia, S.; Cardozo, P.W.; Ferret, A.; Bach, A. Changes in rumen microbial fermentation are due to a combined effect of type of diet and pH. J. Anim. Sci. 2008, 86, 702–711. [Google Scholar] [CrossRef]
- Bannink, A.; France, J.; Lopez, S.; Gerrits, W.J.J.; Kebreab, E.; Tamminga, S.; Dijkstra, J. Modelling the implications of feeding strategy on rumen fermentation and functioning of the rumen wall. Anim. Feed. Sci. Technol. 2007, 143, 3–26. [Google Scholar] [CrossRef]
| Items | |
|---|---|
| Flake corn | 6.02 |
| Corn | 13.27 |
| Soybean meal | 15.42 |
| Sunflower meal | 0.73 |
| Beet meal | 1.68 |
| Whole cottonseed | 6.14 |
| Soybean hull | 4.29 |
| Puffed soybeans | 0.84 |
| Molasses | 1.55 |
| 1 Premix | 0.42 |
| CaHPO4 | 0.43 |
| Stone powder | 0.87 |
| NaHCO3 | 0.75 |
| MgO | 0.22 |
| NaCl | 0.28 |
| Corn silage | 25.55 |
| Alfalfa | 15.26 |
| Oat grass | 6.29 |
| Nutrient levels | |
| CP | 17.2 |
| 2 NEL(Mcal/kg) | 6.71 |
| NDF | 35.66 |
| ADF | 23.65 |
| Ca | 0.87 |
| P | 0.4 |
| Starch | 22.35 |
| NFC | 26.61 |
| EE | 3.89 |
| Items | Treatments | 0 | 105 cfu/g | 107 cfu/g | SEM | p | ||
|---|---|---|---|---|---|---|---|---|
| A | C | A × C | ||||||
| DM | LP | 32.40 | 33.06 | 32.61 | 0.086 | 0.872 | 0.005 | 0.475 |
| BL | 32.40 b | 33.28 a | 32.30 b | |||||
| CP (DM%) | LP | 4.39 | 4.25 B | 4.32 | 0.041 | 0.003 | 0.162 | 0.016 |
| BL | 4.39 b | 4.92 aA | 4.54 ab | |||||
| WSC (DM%) | LP | 2.58 b | 2.87 ab | 3.18 a | 0.083 | 0.093 | 0.236 | 0.445 |
| BL | 2.58 | 2.47 | 2.68 | |||||
| NDF (DM%) | LP | 68.86 | 67.70 | 68.32 | 0.230 | 0.801 | 0.172 | 0.965 |
| BL | 68.86 | 67.77 | 68.61 | |||||
| ADF (DM%) | LP | 43.12 a | 40.37 b | 41.55 b | 0.141 | 0.165 | <0.001 | 0.588 |
| BL | 43.12 a | 41.04 c | 42.13 b | |||||
| HC (DM%) | LP | 25.74 | 27.33 | 26.77 | 0.194 | 0.458 | 0.051 | 0.821 |
| BL | 25.74 | 26.73 | 26.48 | |||||
| Items | Treatments | 0 | 105 cfu/g | 107 cfu/g | SEM | p | ||
|---|---|---|---|---|---|---|---|---|
| A | C | A × C | ||||||
| pH | LP | 3.71 | 3.72 | 3.71 | 0.010 | 0.555 | 0.111 | 0.258 |
| BL | 3.71 | 3.74 | 3.65 | |||||
| AN (DM%) | LP | 0.91 a | 0.42 bB | 0.39 b | 0.021 | 0.011 | <0.001 | 0.017 |
| BL | 0.91 a | 0.75 aA | 0.44 b | |||||
| LA (DM%) | LP | 6.78 b | 6.92 b | 8.50 aA | 0.229 | 0.004 | 0.535 | 0.029 |
| BL | 6.78 | 5.51 | 5.04 B | |||||
| AA (mmol/L) | LP | 3.58 bc | 4.25 a | 3.31 bB | 0.045 | 0.334 | <0.001 | 0.028 |
| BL | 3.58 b | 4.05 a | 3.78 abA | |||||
| Items | Treatments | 0 | 105 cfu/g | 107 cfu/g | SEM | p | ||
|---|---|---|---|---|---|---|---|---|
| A | C | A × C | ||||||
| 3 h | LP | 4.17 b | 4.17 bB | 7.83 a | 0.167 | 0.011 | <0.001 | <0.001 |
| BL | 4.17 b | 8.17 aA | 6.83 a | |||||
| 6 h | LP | 8.67 b | 9.00 b | 12.00 a | 0.236 | 0.818 | 0.005 | 0.009 |
| BL | 8.67 b | 11.33 a | 10.00 ab | |||||
| 12 h | LP | 13.83 b | 14.83 b | 17.50 a | 0.266 | 0.684 | 0.008 | 0.013 |
| BL | 13.83 b | 16.83 a | 14.83 ab | |||||
| 24 h | LP | 20.83 b | 22.17 b | 25.17 aA | 0.342 | 0.635 | 0.013 | 0.028 |
| BL | 20.83 b | 24.50 a | 21.83 abB | |||||
| 48 h | LP | 29.50 b | 31.17 ab | 34.50 aA | 0.487 | 0.277 | 0.090 | 0.064 |
| BL | 29.50 | 32.50 | 29.83 B | |||||
| Items | Treatments | 0 | 105 cfu/g | 107 cfu/g | SEM | p | ||
|---|---|---|---|---|---|---|---|---|
| A | C | A × C | ||||||
| pH | LP | 6.76 | 6.74 | 6.74 | 0.002 | 1.000 | 0.041 | 0.133 |
| BL | 6.76 a | 6.75 ab | 6.74 b | |||||
| TVFA (mmol/L) | LP | 76.13 | 79.02 | 78.61 | 0.735 | 0.357 | 0.573 | 0.798 |
| BL | 76.13 | 76.97 | 76.44 | |||||
| AA (mmol/L) | LP | 46.17 | 48.39 | 47.96 | 0.483 | 0.268 | 0.513 | 0.720 |
| BL | 46.17 | 46.71 | 46.27 | |||||
| PA (mmol/L) | LP | 16.87 | 17.37 | 17.38 | 0.150 | 0.372 | 0.646 | 0.810 |
| BL | 16.87 | 16.96 | 16.96 | |||||
| IBA (mmol/L) | LP | 1.31 | 1.32 | 1.35 | 0.012 | 0.970 | 0.623 | 0.536 |
| BL | 1.31 | 1.35 | 1.32 | |||||
| BA (mmol/L) | LP | 7.71 | 7.87 | 7.90 | 0.067 | 0.942 | 0.546 | 0.980 |
| BL | 7.71 | 7.88 | 7.85 | |||||
| IVA (mmol/L) | LP | 2.80 | 2.81 | 2.79 | 0.022 | 0.887 | 0.876 | 0.993 |
| BL | 2.80 | 2.80 | 2.78 | |||||
| VA (mmol/L) | LP | 1.26 | 1.17 | 1.23 | 0.011 | 0.671 | 0.764 | 0.792 |
| BL | 1.26 | 1.26 | 1.26 | |||||
| Items | Treatments | 0 | 105 cfu/g | 107 cfu/g | SEM | p | ||
|---|---|---|---|---|---|---|---|---|
| A | C | A × C | ||||||
| Observed species | LP | 722.23 | 1386.67 A | 806.33 | 55.49 | 0.033 | 0.355 | 0.077 |
| BL | 722.23 a | 515.93 bB | 655.97 ab | |||||
| Chao1 index | LP | 863.99 | 1703.77 | 1024.28 | 70.95 | 0.028 | 0.417 | 0.079 |
| BL | 863.99 ab | 587.00 b | 734.00 ab | |||||
| Shannon index | LP | 4.34 b | 5.27 a | 4.25 b | 0.08 | 0.358 | 0.048 | 0.052 |
| BL | 4.34 | 4.47 | 4.57 | |||||
| Simpson index | LP | 0.83 ab | 0.86 a | 0.81 b | 0.01 | 0.489 | 0.522 | 0.434 |
| BL | 0.83 | 0.81 | 0.82 | |||||
| Pielou evenness | LP | 0.46 b | 0.51 a | 0.44 b | 0.01 | 0.429 | 0.084 | 0.257 |
| BL | 0.46 | 0.50 | 0.49 | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, Y.; Xiong, H.; Wen, Z.; Tian, H.; Chen, Y.; Wu, L.; Guo, Y.; Sun, B. Effects of Different Concentrations of Lactobacillus plantarum and Bacillus licheniformis on Silage Quality, In Vitro Fermentation and Microbial Community of Hybrid Pennisetum. Animals 2022, 12, 1752. https://doi.org/10.3390/ani12141752
Zhu Y, Xiong H, Wen Z, Tian H, Chen Y, Wu L, Guo Y, Sun B. Effects of Different Concentrations of Lactobacillus plantarum and Bacillus licheniformis on Silage Quality, In Vitro Fermentation and Microbial Community of Hybrid Pennisetum. Animals. 2022; 12(14):1752. https://doi.org/10.3390/ani12141752
Chicago/Turabian StyleZhu, Yanchen, Haoming Xiong, Zhiying Wen, Hanchen Tian, Yiye Chen, Longfei Wu, Yongqing Guo, and Baoli Sun. 2022. "Effects of Different Concentrations of Lactobacillus plantarum and Bacillus licheniformis on Silage Quality, In Vitro Fermentation and Microbial Community of Hybrid Pennisetum" Animals 12, no. 14: 1752. https://doi.org/10.3390/ani12141752
APA StyleZhu, Y., Xiong, H., Wen, Z., Tian, H., Chen, Y., Wu, L., Guo, Y., & Sun, B. (2022). Effects of Different Concentrations of Lactobacillus plantarum and Bacillus licheniformis on Silage Quality, In Vitro Fermentation and Microbial Community of Hybrid Pennisetum. Animals, 12(14), 1752. https://doi.org/10.3390/ani12141752





