Taxifolin Modulates Transcriptomic Response to Heat Stress in Rainbow Trout, Oncorhynchus mykiss
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Diet
2.2. Fish Rearing
2.3. Experimental Set-Up and Sampling
2.4. Gyrodactylus sp. Infections
2.5. RNA Isolation, Library Preparation, and Sequencing
2.6. Data Analysis
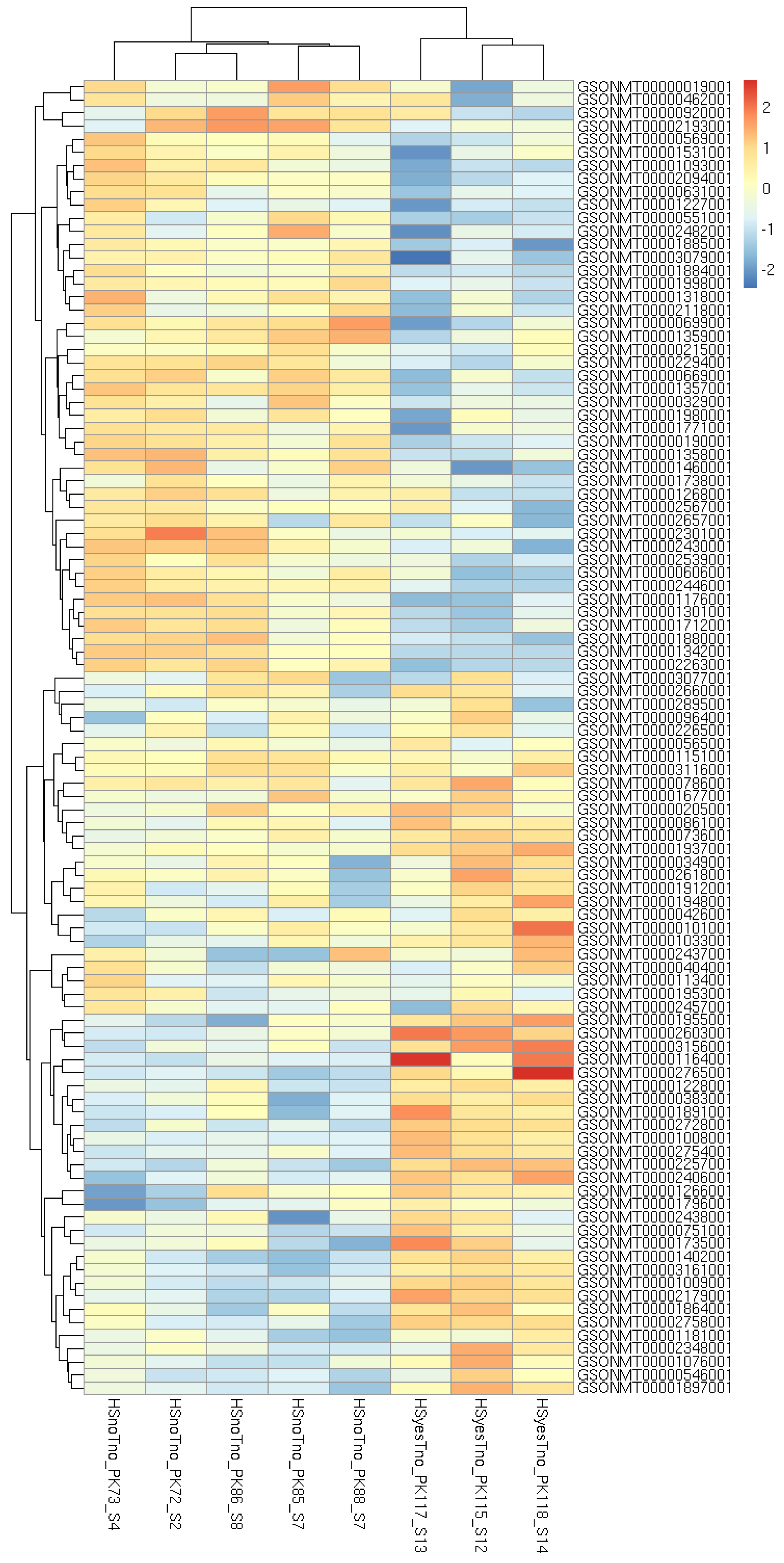
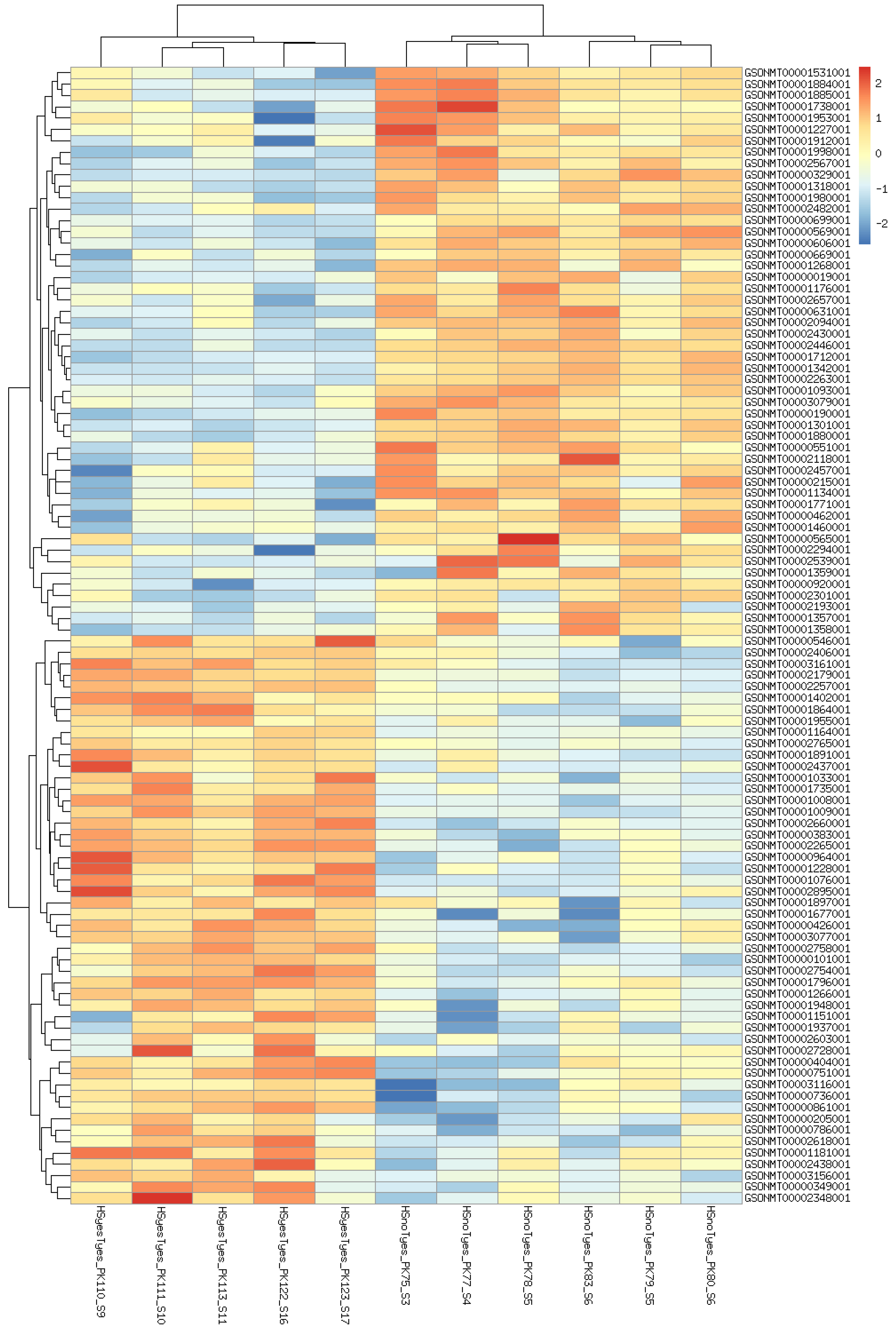
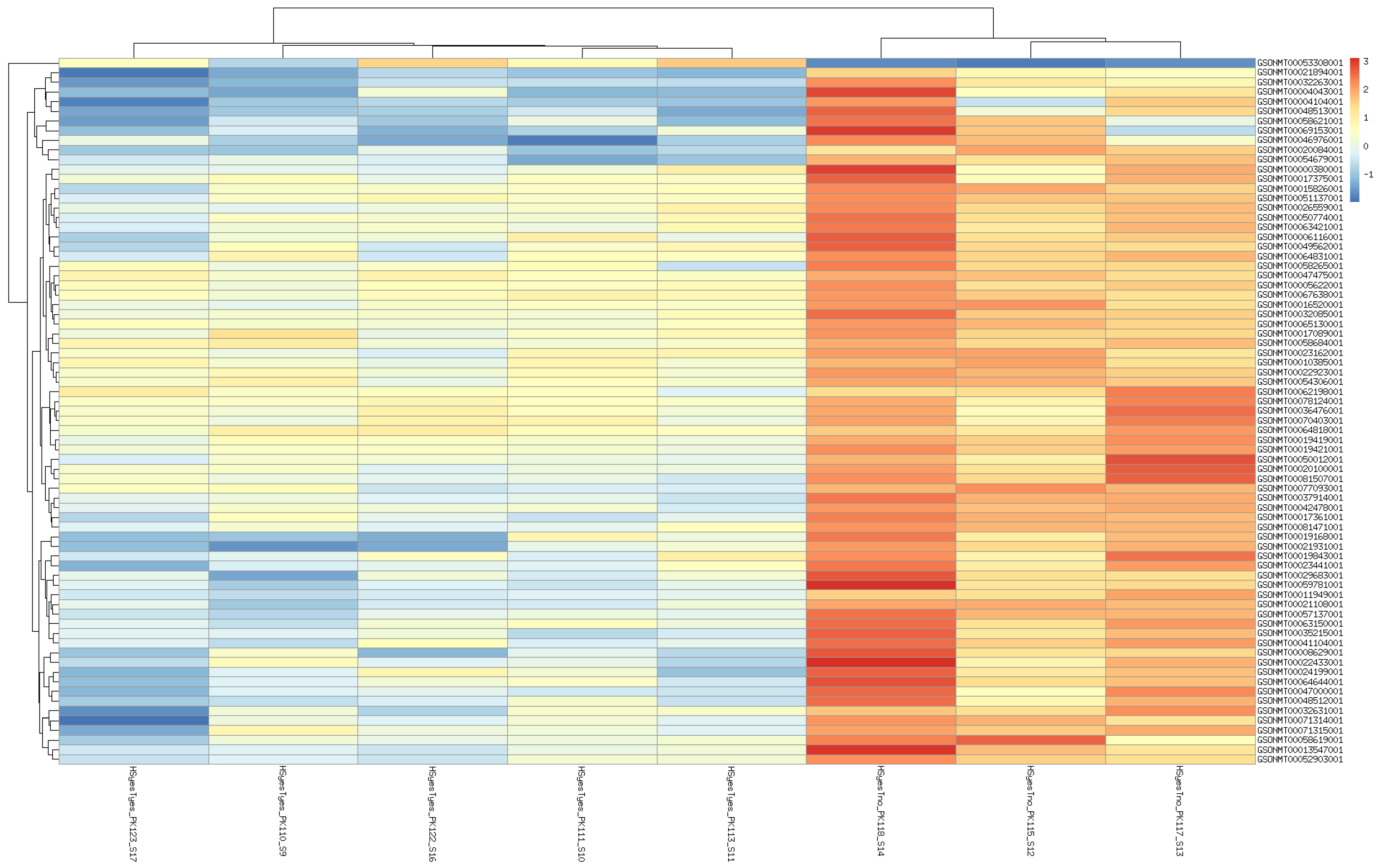
3. Results
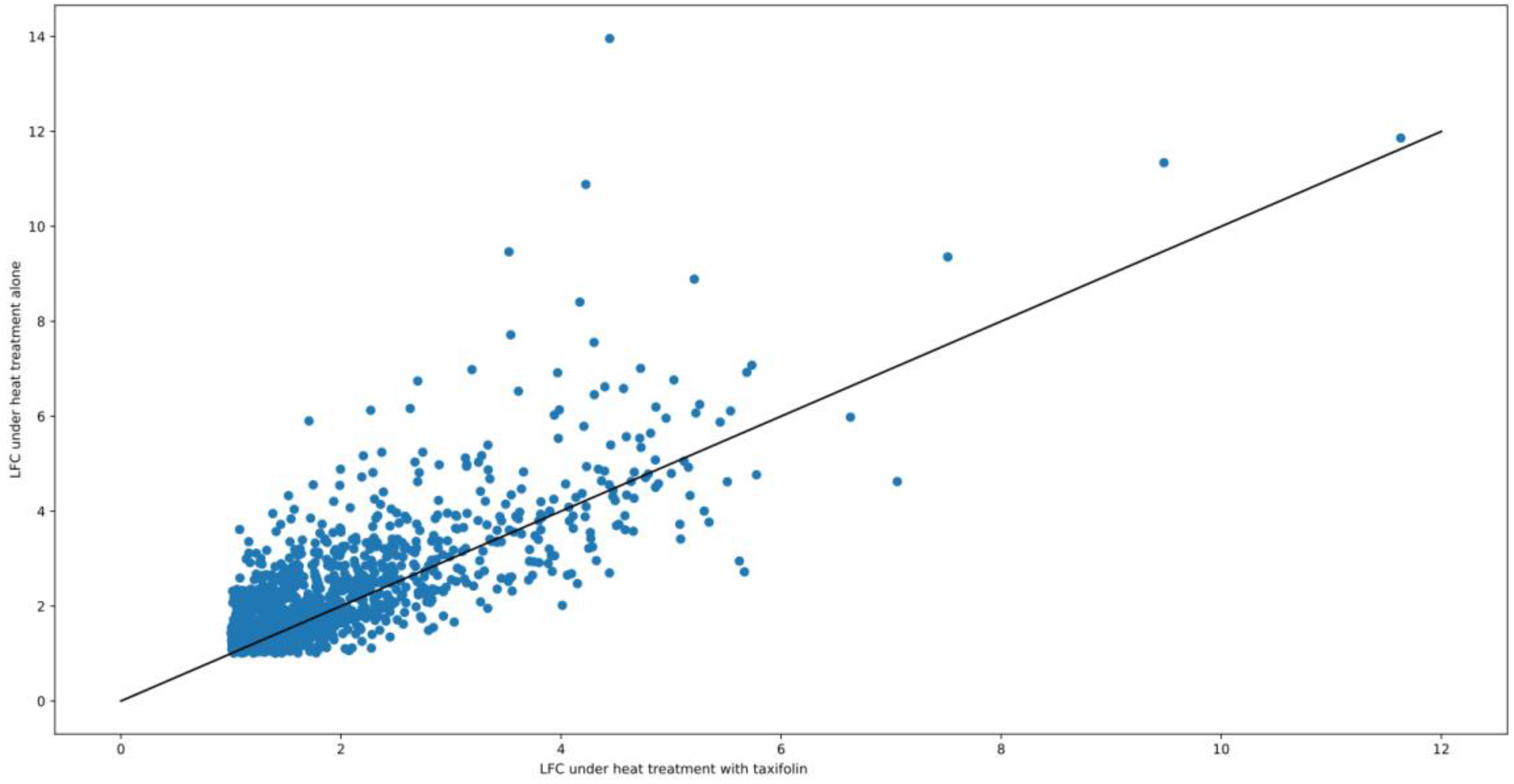
3.1. DEGs and Possible Artifacts
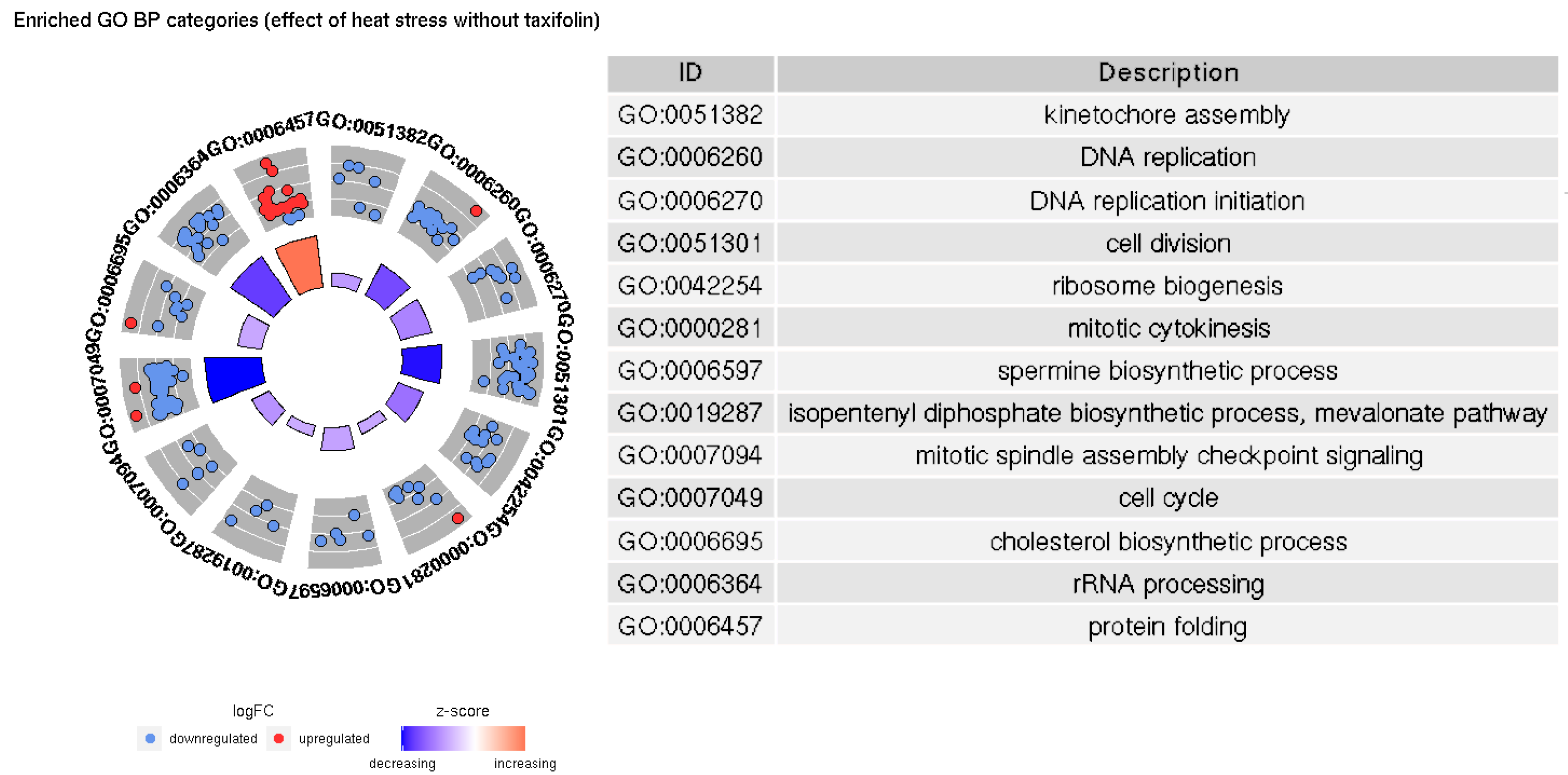
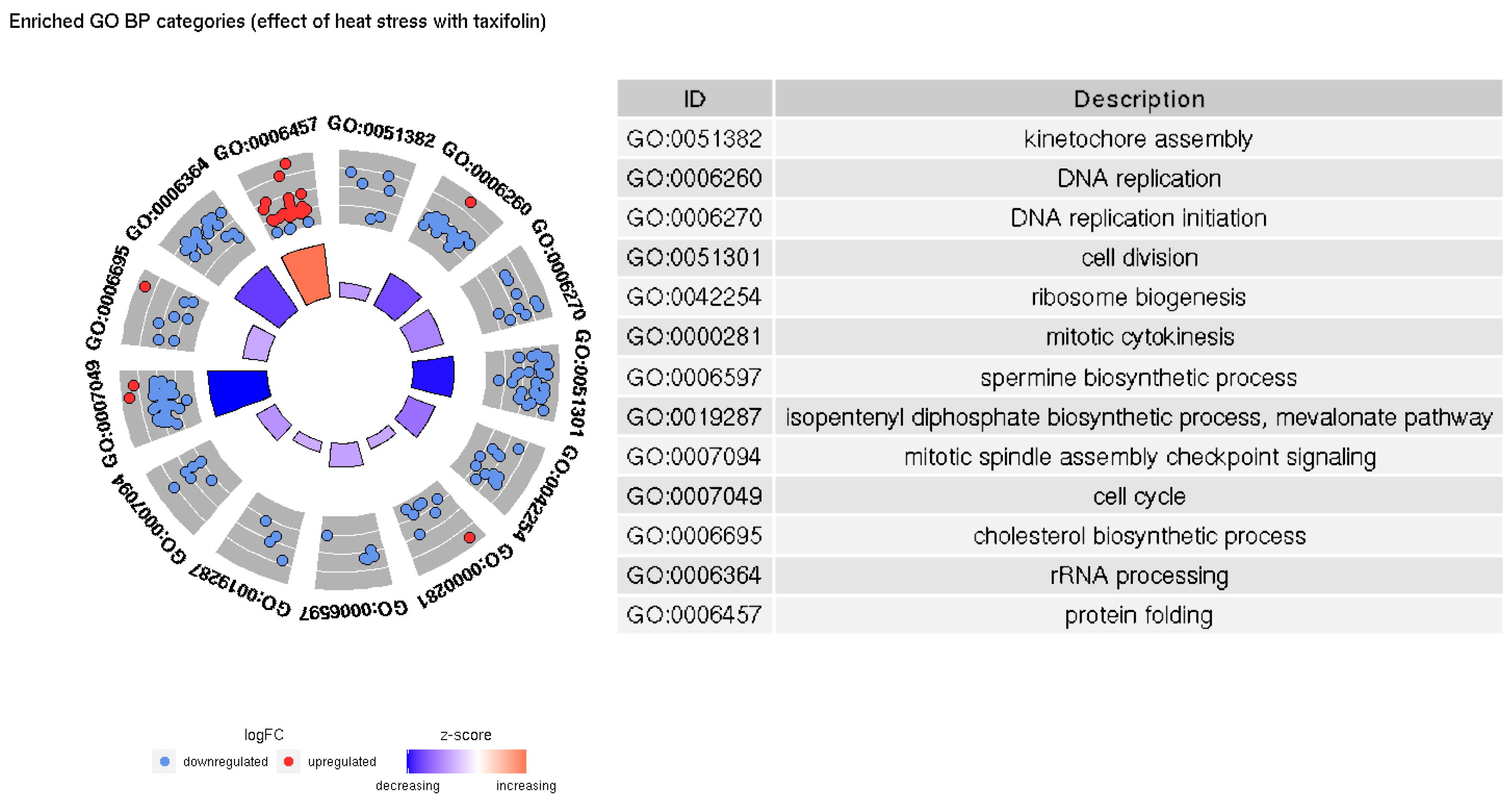
3.2. GO Enrichment
4. Discussion
4.1. Enriched GO Categories with and without Taxifolin
4.2. Other Processes Affected by Taxifolin Treatment and Heat Stress
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Citarasu, T. Herbal biomedicines: A new opportunity for aquaculture industry. Aquac. Int. 2010, 18, 403–414. [Google Scholar] [CrossRef]
- Chakraborty, S.B.; Hancz, C. Application of phytochemicals as immunostimulant, antipathogenic and antistress agents in finfish culture. Rev. Aquac. 2011, 3, 103–119. [Google Scholar] [CrossRef]
- Reverter, M.; Bontemps, N.; Lecchini, D.; Banaigs, B.; Sasal, P. Use of plant extracts in fish aquaculture as an alternative to chemotherapy: Current status and future perspectives. Aquaculture 2014, 433, 50–61. [Google Scholar] [CrossRef]
- Ahmadifar, E.; Yousefi, M.; Karimi, M.; Raieni, R.F.; Dadar, M.; Yilmaz, S.; Dawood, M.A.O.; Abdel-Latif, H.M.R. Benefits of dietary polyphenols and polyphenol-rich additives to aquatic animal health: An Overview. Rev. Fish. Sci. Aquac. 2020, 29, 478–511. [Google Scholar] [CrossRef]
- Alexander, C.P.; Kirubakaran, C.J.; Michael, R.D. Water soluble fraction of Tinospora cordifolia leaves enhanced the non-specific immune mechanisms and disease resistance in Oreochromis mossambicus. Fish. Shellfish Immunol. 2010, 29, 765–772. [Google Scholar] [CrossRef] [PubMed]
- Awad, E.; Austin, D.; Lyndon, A.R. Effect of black cumin seed oil (Nigella sativa) and nettle extract (Quercetin) on enhancement of immunity in rainbow trout, Oncorhynchus mykiss (Walbaum). Aquaculture 2013, 388, 193–197. [Google Scholar] [CrossRef]
- Das, A.; Baidya, R.; Chakraborty, T.; Samanta, A.K.; Roy, S. Pharmacological basis and new insights of taxifolin: A comprehensive review. Biomed. Pharmacother. 2021, 142, 112004. [Google Scholar] [CrossRef]
- Awad, E.; Awaad, A.S.; Esteban, M.A. Effects of dihydroquercetin obtained from deodar (Cedrus deodara) on immune status of gilthead seabream (Sparus aurata L.). Fish. Shellfish Immunol. 2015, 43, 43–50. [Google Scholar] [CrossRef]
- Kantserova, N.; Lysenko, L.; Churova, M.; Tushina, E.; Sukhovskaya, I.; Nemova, N. Dietary supplement with dihydroquercetin and arabinogalactan affects growth performance, intracellular protease activities and muscle-specific gene expression in bacterially infected Oncorhynchus mykiss. Int. Aquatic Res. 2020, 12, 63–73. [Google Scholar] [CrossRef]
- Weidmann, A.E. Dihydroquercetin: More than just an impurity? Eur. J. Pharmacol. 2012, 684, 9–26. [Google Scholar] [CrossRef]
- Sunil, C.; Xu, B. An insight into the health-promoting effects of taxifolin (dihydroquercetin). Phytochemistry 2019, 166, 112066. [Google Scholar] [CrossRef] [PubMed]
- Anthony, K.P.; Saleh, M.A. Free radical scavenging and antioxidant activities of silymarin components. Antioxidants 2013, 2, 398–407. [Google Scholar] [CrossRef] [PubMed]
- Topal, F.; Nar, M.; Gocer, H.; Kalin, P.; Kocyigit, U.M.; Gülçin, İ.; Alwasel, S.H. Antioxidant activity of taxifolin: An activity–structure relationship. J. Enzyme Inhib. Med. Chem. 2016, 31, 674–683. [Google Scholar] [CrossRef]
- Manigandan, K.; Jayaraj, R.L.; Jagatheesh, K.; Elangovan, N. Taxifolin mitigates oxidative DNA damage in vitro and protects zebrafish (Danio rerio) embryos against cadmium toxicity. Environ. Toxicol. Pharmacol. 2015, 39, 1252–1261. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.H.; Wang, W.Y.; Chang, C.C.; Liou, K.T.; Sung, Y.J.; Liao, J.F.; Chen, C.F.; Chang, S.; Hou, Y.C.; Chou, Y.C.; et al. Taxifolin ameliorates cerebral ischemia-reperfusion injury in rats through its anti-oxidative effect and modulation of NF-kappa B activation. J. Biomed. Sci. 2006, 13, 127–141. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Shi, G.; Cai, J.; Yang, J.; Zheng, Y.; Yu, D.; Liu, Q.; Gong, Y.; Zhang, Z. Taxifolin alleviates apoptotic injury induced by DEHP exposure through cytochrome P450 homeostasis in chicken cardiomyocytes. Ecotoxicol. Environ. Saf. 2019, 183, 109582. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.B.; Cha, K.H.; Selenge, D.; Solongo, A.; Nho, C.W. The chemopreventive effect of taxifolin is exerted through ARE-dependent gene regulation. Biol. Pharm. Bull. 2007, 30, 1074–1079. [Google Scholar] [CrossRef]
- Liang, L.; Gao, C.; Luo, M.; Wang, W.; Zhao, C.; Zu, Y.; Efferth, T.; Fu, Y. Dihydroquercetin (DHQ) induced HO-1 and NQO1 expression against oxidative stress through the Nrf2-dependent antioxidant pathway. J. Agric. Food Chem. 2013, 61, 2755–2761. [Google Scholar] [CrossRef]
- Xie, X.; Feng, J.; Kang, Z.; Zhang, S.; Zhang, L.; Zhang, Y.; Li, X.; Tang, Y. Taxifolin protects RPE cells against oxidative stress-induced apoptosis. Mol. Vis. 2017, 23, 520–528. Available online: http://www.molvis.org/molvis/v23/520 (accessed on 15 December 2021).
- Ahiskali, I.; Pinar, C.L.; Kiki, M.; Mammadov, R.; OzbekBilgin, A.; Hacimuftuoglu, A.; Cankaya, M.; KeskinCimen, F.; Altuner, D. Effect of taxifolin on development of retinopathy in alloxan-induced diabetic rats. Cutan. Ocul. Toxicol. 2019, 38, 227–232. [Google Scholar] [CrossRef]
- Wan, F.; Han, H.; Zhong, R.; Wang, M.; Tang, S.; Zhang, S.; Hou, F.; Yi, B.; Zhang, H. Dihydroquercetin supplement alleviates colonic inflammation potentially through improved gut microbiota community in mice. Food Funct. 2021, 12, 11420–11434. [Google Scholar] [CrossRef] [PubMed]
- Cai, C.; Liu, C.; Zhao, L.; Liu, H.; Li, W.; Guan, H.; Zhao, L.; Xiao, J. Effects of taxifolin on osteoclastogenesis in vitro and in vivo. Front. Pharmacol. 2018, 9, 1286. [Google Scholar] [CrossRef] [PubMed]
- Theriault, A.; Wang, Q.; Van Iderstine, S.C.; Chen, B.; Franke, A.A.; Adeli, K. Modulation of hepatic lipoprotein synthesis and secretion by taxifolin, a plant flavonoid. J. Lipid Res. 2000, 41, 1969–1979. [Google Scholar] [CrossRef]
- Casaschi, A.; Rubio, B.K.; Maiyoh, G.K.; Theriault, A.G. Inhibitory activity of diacylglycerol acyltransferase (DGAT) and microsomal triglyceride transfer protein (MTP) by the flavonoid, taxifolin, in HepG2 cells: Potential role in the regulation of apolipoprotein B secretion. Atherosclerosis 2004, 176, 247–253. [Google Scholar] [CrossRef]
- Butt, S.S.; Khan, K.; Badshah, Y.; Rafiq, M.; Shabbir, M. Evaluation of pro-apoptotic potential of taxifolin against liver cancer. PeerJ 2021, 9, e11276. [Google Scholar] [CrossRef]
- Razak, S.; Afsar, T.; Ullah, A.; Almajwal, A.; Alkholief, M.; Alshamsan, A.; Jahan, S. Taxifolin, a natural flavonoid interacts with cell cycle regulators causes cell cycle arrest and causes tumor regression by activating Wnt/β-catenin signaling pathway. BMC Cancer 2018, 18, 1043. [Google Scholar] [CrossRef]
- Wang, R.; Zhu, X.; Wang, Q.; Li, X.; Wang, E.; Zhao, Q.; Wang, Q.; Cao, H. The anti-tumor effect of taxifolin on lung cancer via suppressing stemness and epithelial-mesenchymal transition in vitro and oncogenesis in nude mice. Ann. Transl. Med. 2020, 8, 590. [Google Scholar] [CrossRef]
- Cui, Y.; Zhang, X.; Yin, K.; Qi, X.; Zhang, Y.; Zhang, J.; Li, S.; Lin, H. Dibutyl phthalate-induced oxidative stress, inflammation and apoptosis in grass carp hepatocytes and the therapeutic use of taxifolin. Sci. Total Environ. 2021, 764, 142880. [Google Scholar] [CrossRef]
- Jung, E.H.; Brix, K.V.; Richards, J.G.; Val, A.L.; Brauner, C.J. Reduced hypoxia tolerance and survival at elevated temperatures may limit the ability of Amazonian fish to survive in a warming world. Sci. Total. Environ. 2020, 748, 141349. [Google Scholar] [CrossRef]
- Li, P.; Liu, Q.; Li, J.; Wang, F.; Wen, S.; Li, N. Transcriptomic responses to heat stress in gill and liver of endangered Brachymystax lenok tsinlingensis. Comp. Biochem. Physiol. Part D Genom. Proteom. 2021, 38, 100791. [Google Scholar] [CrossRef]
- Kantserova, N.; Churova, M.; Lysenko, L.; Tushina, E.; Rodin, M.; Krupnova, M.; Sukhovskaya, I. Effect of hyperthermia on proteases and growth regulators in the skeletal muscle of cultivated rainbow trout O. mykiss. Russ. J. Bioorg. Chem. 2020, 46, 1181–1186. [Google Scholar] [CrossRef]
- Morgan, I.J.; D’Cruz, L.M.; Dockray, J.J.; Linton, T.K.; Wood, C.M. The effects of elevated summer temperature and sublethal pollutants (ammonia, low pH) on protein turnover in the gill and liver of rainbow trout (Oncorhynchus mykiss) on a limited food ration. Comp. Biochem. Physiol. Part D 1999, 123, 43–53. [Google Scholar] [CrossRef]
- Rebl, A.; Verleih, M.; Köbis, J.M.; Kühn, C.; Wimmers, K.; Köllner, B.; Goldammer, T. Transcriptome profiling of gill tissue in regionally bred and globally farmed rainbow trout strains reveals different strategies for coping with thermal stress. Mar. Biotechnol. 2015, 15, 445–460. [Google Scholar] [CrossRef] [PubMed]
- Narum, S.R.; Campbell, N.R. Transcriptomic response to heat stress among ecologically divergent populations of redband trout. BMC Genom. 2015, 16, 103. [Google Scholar] [CrossRef] [PubMed]
- Verleih, M.; Borchel, A.; Krasnov, A.; Rebl, A.; Korytář, T.; Kühn, C.; Goldammer, T. Impact of thermal stress on kidney-specific gene expression in farmed regional and imported rainbow trout. Mar. Biotechnol. 2015, 17, 576–592. [Google Scholar] [CrossRef]
- Huang, J.; Li, Y.; Liu, Z.; Kang, Y.; Wang, J. Transcriptomic responses to heat stress in rainbow trout Oncorhynchus mykiss head kidney. Fish Shellfish Immunol. 2018, 82, 32–40. [Google Scholar] [CrossRef]
- Ma, F.; Liu, Z.; Huang, J.; Li, Y.; Kang, Y.; Liu, X.; Wang, J. High-throughput sequencing reveals microRNAs in response to heat stress in the head kidney of rainbow trout (Oncorhynchus mykiss). Funct. Integr. Genom. 2019, 19, 775–786. [Google Scholar] [CrossRef]
- Zhou, C.Q.; Ka, W.; Yuan, W.K.; Wang, J.L. The effect of acute heat stress on the innate immune function of rainbow trout based on the transcriptome. J. Therm. Biol. 2021, 96, 102834. [Google Scholar] [CrossRef]
- Li, Y.; Huang, J.; Liu, Z.; Zhou, Y.; Xia, B.; Wang, Y.; Kang, Y.; Wang, J. Transcriptome analysis provides insights into hepatic responses to moderate heat stress in the rainbow trout (Oncorhynchus mykiss). Gene 2017, 619, 1–9. [Google Scholar] [CrossRef]
- Quan, J.; Kang, Y.; Luo, Z.; Zhao, G.; Ma, F.; Li, L.; Liu, Z. Identification and characterization of long noncoding RNAs provide insight into the regulation of gene expression in response to heat stress in rainbow trout (Oncorhynchus mykiss). Comp. Biochem. Physiol. Part D Genom. Proteom. 2020, 36, 100707. [Google Scholar] [CrossRef]
- Sun, J.; Liu, Z.; Quan, J.; Li, L.; Zhao, G.; Lu, J. RNA-seq analysis reveals alternative splicing under heat stress in rainbow trout (Oncorhynchus mykiss). Mar. Biotechnol. 2022, 24, 5–17. [Google Scholar] [CrossRef] [PubMed]
- Lewis, J.M.; Hori, T.S.; Rise, M.L.; Walsh, P.J.; Currie, S. Transcriptome responses to heat stress in the nucleated red blood cells of the rainbow trout (Oncorhynchus mykiss). Physiol. Genom. 2010, 42, 361–373. [Google Scholar] [CrossRef] [PubMed]
- Sangrador-Vegas, A.; Smith, T.J.; Cairns, M.T. Cloning and characterization of a homologue of the alpha inhibitor of NF-kappaB in rainbow trout (Oncorhynchus mykiss). Vet. Immunol. Immunopathol. 2005, 103, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Andrews, S. FastQC: A Quality Control Tool for High Throughput Sequence Data. 2010. Available online: http://www.bioinformatics.babraham.ac.uk/projects/fastqc/ (accessed on 10 November 2021).
- Berthelot, C.; Brunet, F.; Chalopin, D.; Juanchich, A.; Bernard, M.; Noël, B.; Bento, P.; Da Silva, C.; Labadie, K.; Alberti, A.; et al. The rainbow trout genome provides novel insights into evolution after whole-genome duplication in vertebrates. Nat. Commun. 2014, 5, 3657. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Paggi, J.M.; Park, C.; Bennett, C.; Salzberg, S.L. Graph-based genome alignment and genotyping with HISAT2 and HISAT-genotype. Nat. Biotechnol. 2019, 37, 907–915. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Handsaker, B.; Wysoker, A.; Fennell, T.; Ruan, J.; Homer, N.; Marth, G.; Abecasis, G.; Durbin, R.; 1000 Genome Project Data Processing Subgroup. The Sequence Alignment/Map format and SAMtools. Bioinformatics 2009, 25, 2078–2079. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.D.; McCarthy, D.J.; Smyth, G.K. EdgeR: A Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 2010, 26, 139–140. [Google Scholar] [CrossRef]
- Chen, Y.; Lun, A.T.; Smyth, G.K. From reads to genes to pathways: Differential expression analysis of RNA-Seq experiments using Rsubread and the edgeR quasi-likelihood pipeline. F1000Research 2016, 5, 1438. [Google Scholar] [CrossRef]
- Alexa, A.; Rahnenfuhrer, J. TopGO: Enrichment Analysis for Gene Ontology, R package version 2.46.0. Bioconductor. 2021. Available online: https://bioconductor.org/packages/release/bioc/html/topGO.html (accessed on 20 March 2022).
- Walter, W.; Sánchez-Cabo, F.; Ricote, M. GOplot: An R package for visually combining expression data with functional analysis. Bioinformatics 2015, 31, 2912–2914. [Google Scholar] [CrossRef]
- Grabherr, M.G.; Haas, B.J.; Yassour, M.; Levin, J.Z.; Thompson, D.A.; Amit, I.; Adiconis, X.; Fan, L.; Raychowdhury, R.; Zeng, Q.; et al. Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat. Biotechnol. 2011, 29, 644–652. [Google Scholar] [CrossRef]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glöckner, F.O. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2013, 41, D590–D596. [Google Scholar] [CrossRef] [PubMed]
- Schloss, P.D. Reintroducing Mothur: 10 Years Later. Appl. Environ. Microbiol. 2020, 86, e02343-19. [Google Scholar] [CrossRef] [PubMed]
- Pegg, A.E. The function of spermine. IUBMB Life 2014, 66, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Ha, H.C.; Sirisoma, N.S.; Kuppusamy, P.; Zweier, J.L.; Woster, P.M.; Casero, R.A., Jr. The natural polyamine spermine functions directly as a free radical scavenger. Proc. Natl. Acad. Sci. USA 1998, 95, 11140–11145. [Google Scholar] [CrossRef] [PubMed]
- Cerqueira, N.M.F.S.A.; Oliveira, E.F.; Gesto, D.S.; Santos-Martins, D.; Moreira, C.; Moorthy, H.N.; Ramos, M.J.; Fernandes, P.A. Cholesterol biosynthesis: A mechanistic overview. Biochemistry 2016, 55, 5483–5506. [Google Scholar] [CrossRef] [PubMed]
- Kostyuk, V.A.; Kraemer, T.; Sies, H.; Schewe, T. Myeloperoxidase/nitrite-mediated lipid peroxidation of low-density lipoprotein as modulated by flavonoids. FEBS Lett. 2003, 537, 146–150. [Google Scholar] [CrossRef]
- Casaschi, A.; Wang, Q.; Dang, K.; Richards, A.; Theriault, A. Intestinal apolipoprotein B secretion is inhibited by the flavonoid quercetin: Potential role of microsomal triglyceride transfer protein and diacylglycerol acyltransferase. Lipids 2002, 37, 647–652. [Google Scholar] [CrossRef]
- Cao, X.; Yue, L.; Song, J.; Wu, Q.; Li, N.; Luo, L.; Lan, L.; Yin, Z. Inducible HSP70 antagonizes IL-1β cytocidal effects through inhibiting NF-kB activation via destabilizing TAK1 in HeLa cells. PLoS ONE 2012, 7, e50059. [Google Scholar] [CrossRef]
- Lyu, Q.; Wawrzyniuk, M.; Rutten, V.P.M.G.; van Eden, W.; Sijts, A.J.A.M.; Broere, F. Hsp70 and NF-kB mediated control of innate inflammatory responses in a canine macrophage cell line. Int. J. Mol. Sci. 2020, 21, 6464. [Google Scholar] [CrossRef]
- Ma, F.; Luo, L. Genome-wide identification of Hsp70/110 genes in rainbow trout and their regulated expression in response to heat stress. PeerJ 2020, 8, e10022. [Google Scholar] [CrossRef]
- Kurashova, N.A.; Madaeva, I.M.; Kolesnikova, L.I. Expression of HSP70 heat-shock proteins under oxidative stress. Adv. Gerontol. 2020, 10, 20–25. [Google Scholar] [CrossRef]
- Sahin, K.; Orhan, C.; Yazlak, H.; Tuzcu, M.; Sahin, N. Lycopene improves activation of antioxidant system and Nrf2/HO-1 pathway of muscle in rainbow trout (Oncorhynchus mykiss) with different stocking densities. Aquaculture 2014, 430, 133–138. [Google Scholar] [CrossRef]
- Sun, J.; Liu, Z.; Quan, J.; Li, L.; Zhao, G.; Lu, J. Protective effects of different concentrations of selenium nanoparticles on rainbow trout (Oncorhynchus mykiss) primary hepatocytes under heat stress. Ecotoxicol. Environ. Saf. 2021, 230, 113121. [Google Scholar] [CrossRef] [PubMed]
- Ohtsuji, M.; Katsuoka, F.; Kobayashi, A.; Aburatani, H.; Hayes, J.D.; Yamamoto, M. Nrf1 and Nrf2 play distinct roles in activation of antioxidant response element-dependent genes. J. Biol. Chem. 2008, 283, 33554–33562. [Google Scholar] [CrossRef]
- Biswas, M.; Chan, J.Y. Role of Nrf1 in antioxidant response element-mediated gene expression and beyond. Toxicol. Appl. Pharmacol. 2010, 244, 16–20. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Zhou, B.; Chen, H.; Lu, K.; Wang, Y. Oxidative stress activates the Nrf2-mediated antioxidant response and P38 MAPK pathway: A possible apoptotic mechanism induced by BDE-47 in rainbow trout (Oncorhynchus mykiss) gonadal RTG-2 cells. Environ. Pollut. 2021, 287, 117341. [Google Scholar] [CrossRef] [PubMed]
- Ramsden, R.; Gallagher, E.P. Dual NRF2 paralogs in Coho salmon and their antioxidant response element targets. Redox Biol. 2016, 9, 114–123. [Google Scholar] [CrossRef]
- Léger, J.A.D.; Athanasio, C.G.; Zhera, A.; Chauhan, M.F.; Simmons, D.B.D. Hypoxic responses in Oncorhynchus mykiss involve angiogenesis, lipid, and lactate metabolism, which may be triggered by the cortisol stress response and epigenetic methylation. Comp. Biochem. Physiol. Part D Genom. Proteom. 2021, 39, 100860. [Google Scholar] [CrossRef]

| No of Tank | Group | No of Fish | Weight, g | Length, cm | Number of Gyrodactylus sp. Individuals, n | Transcriptome Sample Name |
|---|---|---|---|---|---|---|
| 1 | HSnoTno | 72 | 181.4 | 25.2 | 1164 | PK72_S2 |
| 1 | HSnoTno | 73 | 203.6 | 25.8 | 304 | PK73_S4 |
| 2 | HSnoTyes | 75 | 259.0 | 35.2 | 36 | PK75_S3 |
| 2 | HSnoTyes | 77 | 139.3 | 23.8 | 631 | PK77_S4 |
| 2 | HSnoTyes | 78 | 183.0 | 24.7 | 36 | PK78_S5 |
| 4 | HSyesTyes | 110 | 154.9 | 24.2 | 21 | PK110_S9 |
| 4 | HSyesTyes | 111 | 205.4 | 26.0 | 48 | PK111_S10 |
| 4 | HSyesTyes | 113 | 107.4 | 20.1 | 13 | PK113_S11 |
| 5 | HSyesTno | 115 | 208.2 | 25.8 | 20 | PK115_S12 |
| 5 | HSyesTno | 117 | 171.6 | 25.4 | 243 | PK117_S13 |
| 5 | HSyesTno | 118 | 150.9 | 24.2 | 40 | PK118_S14 |
| 6 | HSnoTyes | 79 | 144.4 | 23.2 | 16 | PK79_S5 |
| 6 | HSnoTyes | 80 | 205.3 | 25.4 | 48 | PK80_S6 |
| 6 | HSnoTyes | 83 | 136.9 | 23.6 | 64 | PK83_S6 |
| 7 | HSyesTyes | 120 | 169.2 | 25.0 | 22 | PK120_S15 |
| 7 | HSnoTyes | 122 | 182.6 | 25.7 | 33 | PK122_S16 |
| 7 | HSnoTyes | 123 | 187.2 | 25.0 | 4 | PK123_S17 |
| 8 | HSnoTno | 85 | 182.3 | 26.0 | 72 | PK85_S7 |
| 8 | HSnoTno | 86 | 176.4 | 25.0 | 132 | PK86_S8 |
| 8 | HSnoTno | 88 | 106.9 | 22.0 | 262 | PK88_S7 |
| Contrast | Number of DEGs (Specimen 120 Included) | Number of DEGs (Specimen 120 Excluded) | Overlap |
|---|---|---|---|
| Effect of taxifolin without heat stress | 0 | 0 | N/A |
| Effect of heat stress with taxifolin | 2746 | 2580 | 2182 |
| Effect of heat stress without taxifolin | 2253 | 2498 | 2007 |
| Effect of taxifolin under heat stress | 68 | 74 | 59 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sukhovskaya, I.V.; Kantserova, N.P.; Lysenko, L.A.; Morozov, A.A. Taxifolin Modulates Transcriptomic Response to Heat Stress in Rainbow Trout, Oncorhynchus mykiss. Animals 2022, 12, 1321. https://doi.org/10.3390/ani12101321
Sukhovskaya IV, Kantserova NP, Lysenko LA, Morozov AA. Taxifolin Modulates Transcriptomic Response to Heat Stress in Rainbow Trout, Oncorhynchus mykiss. Animals. 2022; 12(10):1321. https://doi.org/10.3390/ani12101321
Chicago/Turabian StyleSukhovskaya, Irina V., Nadezhda P. Kantserova, Liudmila A. Lysenko, and Alexey A. Morozov. 2022. "Taxifolin Modulates Transcriptomic Response to Heat Stress in Rainbow Trout, Oncorhynchus mykiss" Animals 12, no. 10: 1321. https://doi.org/10.3390/ani12101321
APA StyleSukhovskaya, I. V., Kantserova, N. P., Lysenko, L. A., & Morozov, A. A. (2022). Taxifolin Modulates Transcriptomic Response to Heat Stress in Rainbow Trout, Oncorhynchus mykiss. Animals, 12(10), 1321. https://doi.org/10.3390/ani12101321






