Date Palm Pollen Extract Avert Doxorubicin-Induced Cardiomyopathy Fibrosis and Associated Oxidative/Nitrosative Stress, Inflammatory Cascade, and Apoptosis-Targeting Bax/Bcl-2 and Caspase-3 Signaling Pathways
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals, Kits, and Reagents
2.2. Date Palm Pollen Grains Collection and Ethanolic Extract Preparation
2.3. Acute Oral Toxicity of DPPE “Median Lethal Dose, LD50”
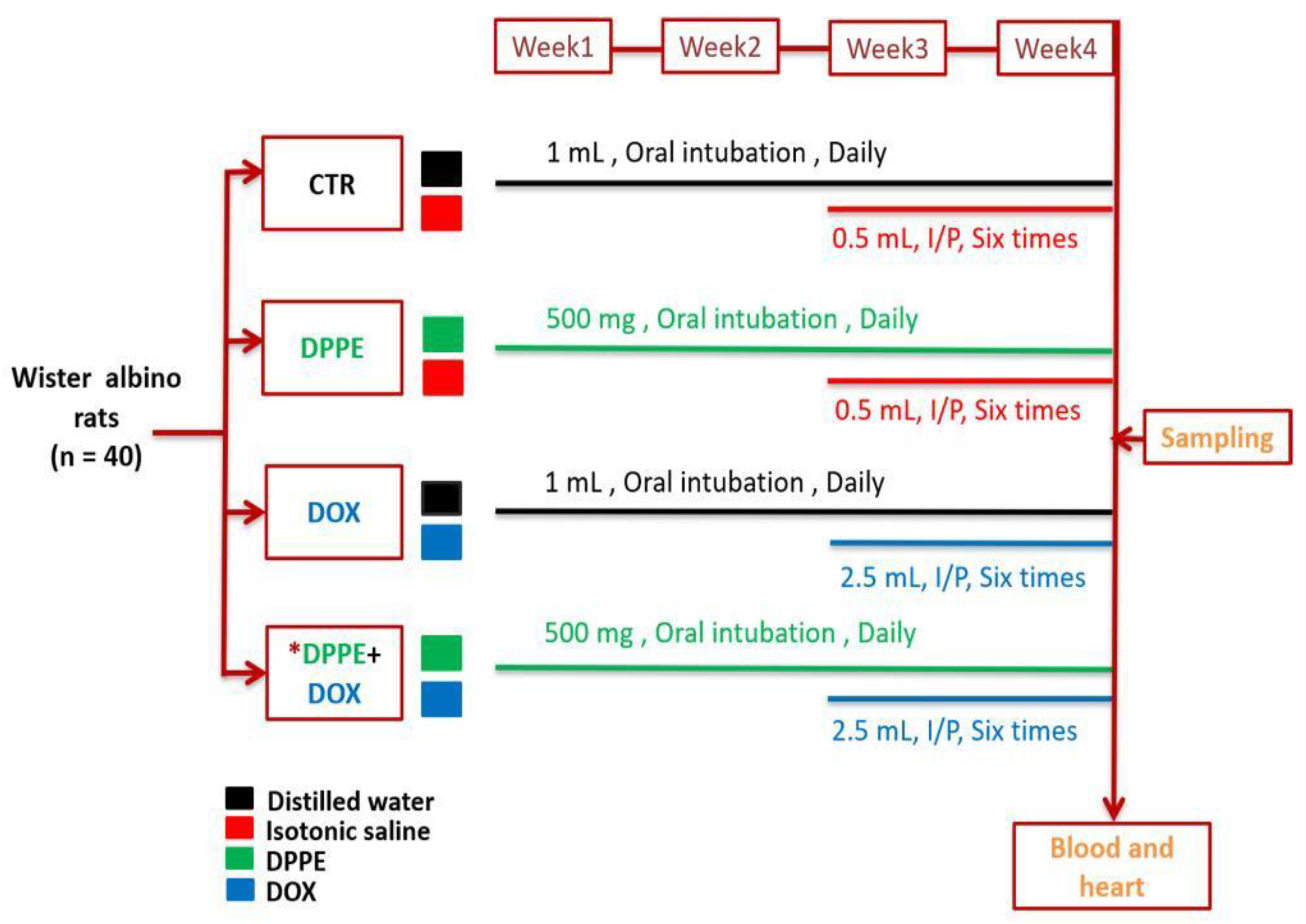
2.4. Animals Experimentation and Sampling
2.5. Assessment of the Body, Heart, and Relative Heart Weights
2.6. Assessment of Cardiac Injury Biomarkers and Cytosolic Calcium (Ca+2)
2.7. Estimation of Cardiac Nitro-Oxidative Stress and Lipid Peroxidation
2.8. Assessment of Inflammatory Markers
2.9. Estimation of Hydroxyproline Content
2.10. Histopathological Assessment and Semi-Quantitative Scoring Approach
2.11. Immunohistochemical Assessment
2.12. Histomorphometric Assessment
2.13. Data Analysis
3. Results
3.1. Median Lethal Dose, Mortality, and Survival Rates
3.2. Body, Heart, and Relative Heart Weights and Myocyte Cross-Sectional Area
3.3. Cardiac Injury Biomarkers and Cardiac Cytosolic Calcium ()
3.4. Cardiac Nitro-Oxidative Stress and Lipid Peroxidation
3.5. Inflammatory Markers
3.6. Cardiac Hydroxyproline Content
3.7. Histopathological Results and Lesions Scoring
3.8. Masson’s Trichrome Staining and Histomorphometric Findings
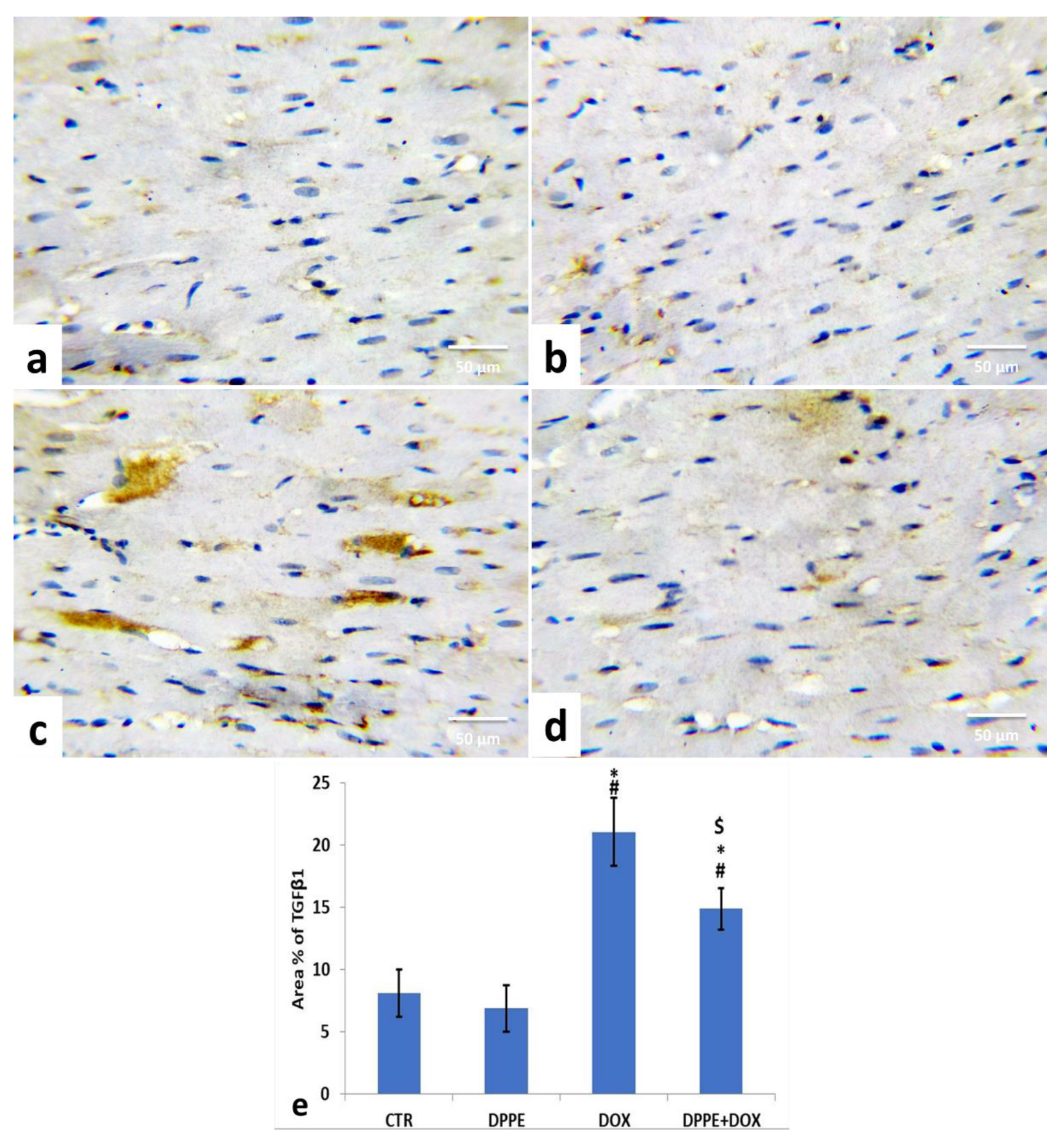
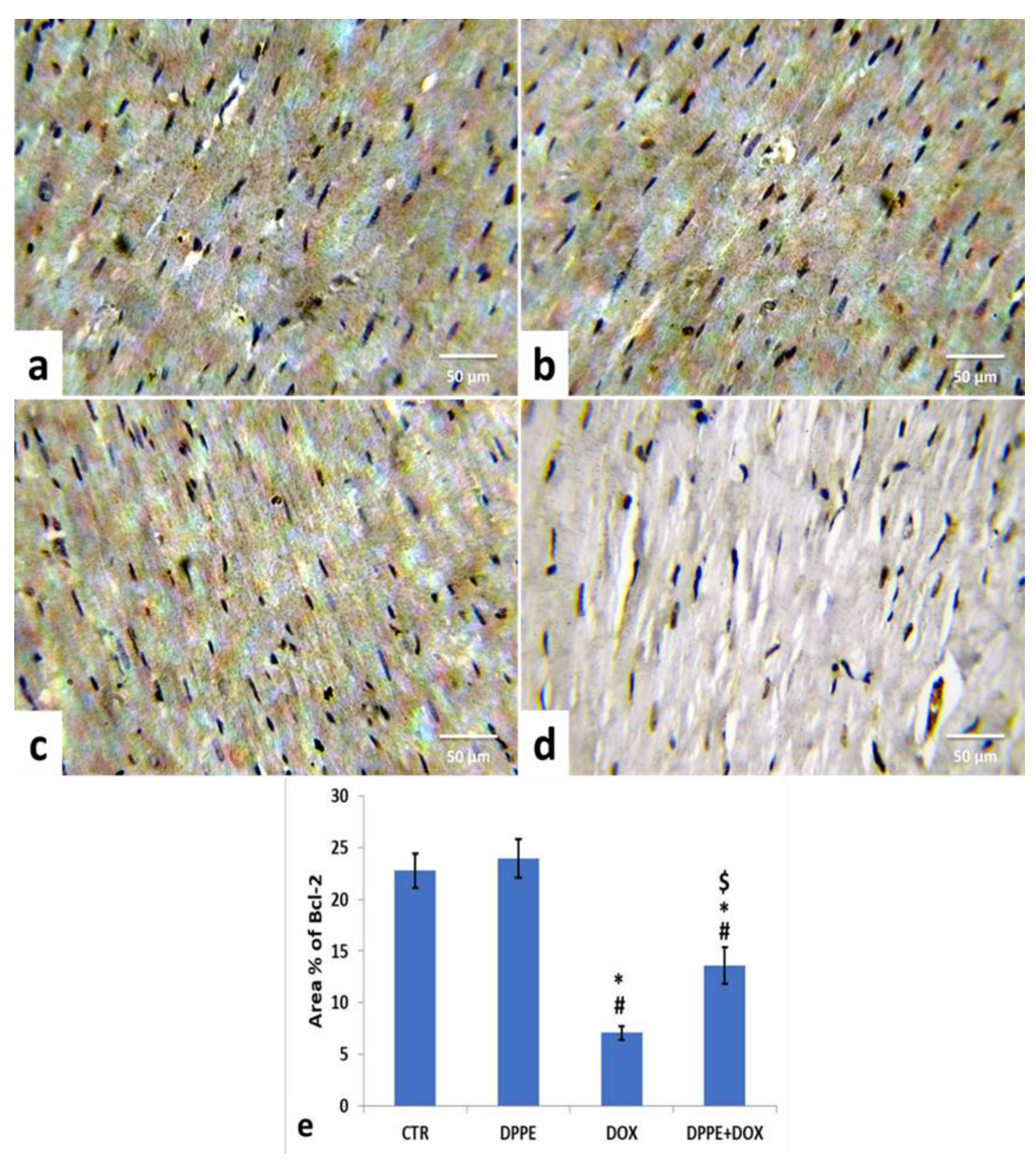
3.9. Immunohistochemical Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ALT | alanine transaminase |
| AST | Aspartate aminotransferase |
| Bax | Alanine aminotransferase |
| Bcl2 | B-cell lymphoma 2 |
| BW | body weight |
| CAT | Catalase |
| CHF | congestive heart failure |
| CK | creatine kinase |
| CK-MB | creatine kinase-cardiac type isoenzyme |
| CVF | collagen volume fraction |
| cTnI | Serum cardiac troponin I |
| cTnT | Troponin T |
| DOX | Doxorubicin |
| DPPE | date palm pollen ethanolic extract |
| GPx | glutathione peroxidase |
| GSH | reduced glutathione |
| IL-1β | interleukin-1 beta |
| IL-6 | interleukin-6 |
| LDH | lactate dehydrogenase |
| MDA | Malondialdehyde |
| NO | nitric oxide |
| NT-pro BNP | N-terminal pro-brain natriuretic peptide |
| PVCA | collagen area |
| RCF | radius centrifugation force |
| RNS | reactive nitrogen species |
| ROS | Redox Oxygen species |
| SOD | superoxide dismutase |
| TNF-α | tumor necrosis factor-alpha |
| UDP | up and down procedure |
References
- Hu, H.; Zhang, W.; Huang, D.; Yang, Q.; Li, J.; Gao, Y. Cardiotoxicity of anthracycline (ANT) treatment in children with malignant tumors. Pediatr. Hematol. Oncol. 2018, 35, 111–120. [Google Scholar] [CrossRef]
- Yu, J.; Wang, C.; Kong, Q.; Wu, X.; Lu, J.J.; Chen, X. Recent progress in doxorubicin-induced cardiotoxicity and protective potential of natural products. Phytomedicine 2018, 40, 125–139. [Google Scholar] [CrossRef] [PubMed]
- Nagiub, M.; Nixon, J.V.; Kontos, M.C. Ability of nonstrain diastolic parameters to predict doxorubicin-induced cardiomyopathy: A Systematic review with meta-analysis. Cardiol. Rev. 2018, 26, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Finn, N.A.; Findley, H.W.; Kemp, M.L. A switching mechanism in doxorubicin bioactivation can be exploited to control doxorubicin toxicity. PLoS Comput. Biol. 2011, 7, e1002151. [Google Scholar] [CrossRef] [PubMed]
- Octavia, Y.; Tocchetti, C.G.; Gabrielson, K.L.; Janssens, S.; Crijns, H.J.; Moens, A.L. Doxorubicin-induced cardiomyopathy: From molecular mechanisms to therapeutic strategies. J. Mol. Cell Cardiol. 2012, 52, 1213–1225. [Google Scholar] [CrossRef] [PubMed]
- Shafei, A.; El-Bakly, W.; Sobhy, A.; Wagdy, O.; Reda, A.; Aboelenin, O.; Marzouk, A.; El Habak, K.; Mostafa, R.; Ali, M.A.; et al. A review on the efficacy and toxicity of different doxorubicin nanoparticles for targeted therapy in metastatic breast cancer. Biomed. Pharmacother. 2017, 95, 1209–1218. [Google Scholar] [CrossRef]
- Tewey, K.M.; Rowe, T.C.; Yang, L.; Halligan, B.D.; Liu, L.F. Adriamycin-induced DNA damage mediated by mammalian DNA topoisomerase II. Science 1984, 226, 466–468. [Google Scholar] [CrossRef]
- Tokarska-Schlattner, M.; Zaugg, M.; Zuppinger, C.; Wallimann, T.; Schlattner, U. New insights into doxorubicin-induced cardiotoxicity: The critical role of cellular energetics. J. Mol. Cell Cardiol. 2006, 41, 389–405. [Google Scholar] [CrossRef]
- Pawlowska, J.; Tarasiuk, J.; Wolf, C.R.; Paine, M.J.; Borowski, E. Differential ability of cytostatics from anthraquinone group to generate free radicals in three enzymatic systems: NADH dehydrogenase, NADPH cytochrome P450 reductase, and xanthine oxidase. Oncol. Res. 2003, 13, 245–252. [Google Scholar] [CrossRef]
- Fogli, S.; Nieri, P.; Breschi, M.C. The role of nitric oxide in anthracycline toxicity and prospects for pharmacologic prevention of cardiac damage. FASEB J. 2004, 18, 664–675. [Google Scholar] [CrossRef]
- Li, D.L.; Wang, Z.V.; Ding, G.; Tan, W.; Luo, X.; Criollo, A.; Xie, M.; Jiang, N.; May, H.; Kyrychenko, V.; et al. Doxorubicin blocks cardiomyocyte autophagic flux by inhibiting lysosome acidification. Circulation 2016, 133, 1668–1687. [Google Scholar] [CrossRef]
- Menna, P.; Salvatorelli, E. Primary prevention strategies for anthracycline cardiotoxicity: A brief overview. Chemotherapy 2017, 62, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, K.; Zhang, J.; Honbo, N.; Karliner, J.S. Doxorubicin cardiomyopathy. Cardiology 2010, 115, 155–162. [Google Scholar] [CrossRef]
- Pecoraro, M.; Del Pizzo, M.; Marzocco, S.; Sorrentino, R.; Ciccarelli, M.; Iaccarino, G.; Pinto, A.; Popolo, A. Inflammatory mediators in a short-time mouse model of doxorubicin-induced cardiotoxicity. Toxicol. Appl. Pharmacol. 2016, 293, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, D.M.; Radwan, R.R.; Fattah, S.M.A. Antioxidant and antiapoptotic effects of sea cucumber and valsartan against doxorubicin-induced cardiotoxicity in rats: The role of low dose gamma irradiation. J. Photochem. Photobiol. B 2017, 170, 70–78. [Google Scholar] [CrossRef]
- McGowan, J.V.; Chung, R.; Maulik, A.; Piotrowska, I.; Walker, J.M.; Yellon, D.M. Anthracycline chemotherapy and cardiotoxicity. Cardiovasc. Drugs Ther. 2017, 31, 63–75. [Google Scholar] [CrossRef] [PubMed]
- Pugazhendhi, A.; Edison, T.; Velmurugan, B.K.; Jacob, J.A.; Karuppusamy, I. Toxicity of doxorubicin (Dox) to different experimental organ systems. Life Sci. 2018, 200, 26–30. [Google Scholar] [CrossRef] [PubMed]
- Goormaghtigh, E.; Huart, P.; Praet, M.; Brasseur, R.; Ruysschaert, J.M. Structure of the adriamycin-cardiolipin complex. Role in mitochondrial toxicity. Biophys. Chem. 1990, 35, 247–257. [Google Scholar] [CrossRef]
- Priya, L.B.; Baskaran, R.; Huang, C.Y.; Padma, V.V. Neferine ameliorates cardiomyoblast apoptosis induced by doxorubicin: Possible role in modulating NADPH oxidase/ROS-mediated NFkappaB redox signaling cascade. Sci. Rep. 2017, 7, 12283. [Google Scholar] [CrossRef]
- Ma, T.; Kandhare, A.D.; Mukherjee-Kandhare, A.A.; Bodhankar, S.L. Fisetin, a plant flavonoid ameliorates doxorubicin-induced cardiotoxicity in experimental rats: The decisive role of caspase-3, COX-II, cTn-I, iNOs and TNF-alpha. Mol. Biol. Rep. 2019, 46, 105–118. [Google Scholar] [CrossRef]
- Zhang, X.; Hu, C.; Kong, C.Y.; Song, P.; Wu, H.M.; Xu, S.C.; Yuan, Y.P.; Deng, W.; Ma, Z.G.; Tang, Q.Z. FNDC5 alleviates oxidative stress and cardiomyocyte apoptosis in doxorubicin-induced cardiotoxicity via activating AKT. Cell Death Differ. 2020, 27, 540–555. [Google Scholar] [CrossRef] [PubMed]
- Wallace, K.B. Adriamycin-induced interference with cardiac mitochondrial calcium homeostasis. Cardiovasc. Toxicol. 2007, 7, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Wenningmann, N.; Knapp, M.; Ande, A.; Vaidya, T.R.; Ait-Oudhia, S. Insights into doxorubicin-induced cardiotoxicity: Molecular mechanisms, preventive strategies, and early monitoring. Mol. Pharmacol. 2019, 96, 219–232. [Google Scholar] [CrossRef]
- Minotti, G.; Recalcati, S.; Menna, P.; Salvatorelli, E.; Corna, G.; Cairo, G. Doxorubicin cardiotoxicity and the control of iron metabolism: Quinone-dependent and independent mechanisms. Methods Enzymol. 2004, 378, 340–361. [Google Scholar] [CrossRef]
- Wang, X.; Wang, Q.; Li, W.; Zhang, Q.; Jiang, Y.; Guo, D.; Sun, X.; Lu, W.; Li, C.; Wang, Y. TFEB-NF-kappaB inflammatory signaling axis: A novel therapeutic pathway of dihydrotanshinone I in doxorubicin-induced cardiotoxicity. J. Exp. Clin. Cancer Res. 2020, 39, 93. [Google Scholar] [CrossRef]
- Shaker, R.A.; Abboud, S.H.; Assad, H.C.; Hadi, N. Enoxaparin attenuates doxorubicin induced cardiotoxicity in rats via interfering with oxidative stress, inflammation and apoptosis. BMC Pharmacol. Toxicol. 2018, 19, 3. [Google Scholar] [CrossRef]
- Zhang, N.; Wei, W.Y.; Li, L.L.; Hu, C.; Tang, Q.Z. Therapeutic potential of polyphenols in cardiac fibrosis. Front. Pharmacol. 2018, 9, 122. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Chen, G.; Xie, Y.; Jiang, D.; Han, J.; Chen, F.; Song, Y. Qiliqiangxin improves cardiac function and attenuates cardiac remodelling in doxorubicin-induced heart failure rats. Pharm. Biol. 2020, 58, 417–426. [Google Scholar] [CrossRef]
- Borriello, M.; Iannuzzi, C.; Sirangelo, I. Pinocembrin protects from age-induced cytotoxicity and inhibits non-enzymatic glycation in human insulin. Cells 2019, 8, 385. [Google Scholar] [CrossRef]
- Salzer, W.L.; Devidas, M.; Carroll, W.L.; Winick, N.; Pullen, J.; Hunger, S.P.; Camitta, B.A. Long-term results of the pediatric oncology group studies for childhood acute lymphoblastic leukemia 1984–2001: A report from the children’s oncology group. Leukemia 2010, 24, 355–370. [Google Scholar] [CrossRef]
- Pandey, K.B.; Rizvi, S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxid. Med. Cell Longev. 2009, 2, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V.K.; Sharma, B. Role of phytochemicals in neurotrophins mediated regulation of Alzheimer’s disease. Int. J. Complement. Altern. Med. 2017, 7, 00231. [Google Scholar] [CrossRef]
- Singh, N.; Sharma, B. Biotoxins mediated DNA damage and role of phytochemicals in DNA protection. Biochem. Mol. Biol. J. 2018, 4, 5. [Google Scholar] [CrossRef]
- Ayza, M.A.; Balasubramanian, R.; Berhe, A.H. Cardioprotective effect of Croton macrostachyus stem bark extract and solvent fractions on cyclophosphamide-induced cardiotoxicity in rats. Evid. Based Complement. Altern. Med. 2020, 2020, 8467406. [Google Scholar] [CrossRef] [PubMed]
- Sandamali, J.A.N.; Hewawasam, R.P.; Jayatilaka, K.; Mudduwa, L.K.B. Cardioprotective potential of Murraya koenigii (L.) Spreng. leaf extract against doxorubicin-induced cardiotoxicity in rats. Evid. Based Complement. Altern. Med. 2020, 2020, 6023737. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chao, X.; Ahmad, F.U.D.; Shi, H.; Mehboob, H.; Hassan, W. Phoenix dactylifera protects against doxorubicin-induced cardiotoxicity and nephrotoxicity. Cardiol. Res. Pract. 2019, 2019, 7395239. [Google Scholar] [CrossRef] [PubMed]
- Bahadir, A.; Kurucu, N.; Kadioglu, M.; Yenilme, E. The role of nitric oxide in doxorubicin-induced cardiotoxicity: Experimental study. Turk. J. Haematol. 2014, 31, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Hassan, H.M.M. Chemical composition and nutritional value of palm pollen grains. Glob. J. Biotechnol. Biochem. 2011, 6, 1–7. [Google Scholar]
- Soliman, F.A.; Soliman, A. The gonad stimulating potency of date palm pollen grains. Experientia 1958, 14, 92–93. [Google Scholar] [CrossRef] [PubMed]
- El-Kashlan, A.M.; Nooh, M.M.; Hassan, W.A.; Rizk, S.M. Therapeutic potential of date palm pollen for testicular dysfunction induced by thyroid disorders in male rats. PLoS ONE 2015, 10, e0139493. [Google Scholar] [CrossRef]
- Kroyer, G.; Hegedus, N. Evaluation of bioactive properties of pollen extracts as functional dietary food supplement. Innov. Food Sci. Emerg. Technol. 2001, 2, 171–174. [Google Scholar] [CrossRef]
- Daoud, A.; Ben Mefteh, F.; Mnafgui, K.; Turki, M.; Jmal, S.; Ben Amar, R.; Ayadi, F.; ElFeki, A.; Abid, L.; Rateb, M.E.; et al. Cardiopreventive effect of ethanolic extract of Date Palm Pollen against isoproterenol induced myocardial infarction in rats through the inhibition of the angiotensin-converting enzyme. Exp. Toxicol. Pathol. 2017, 69, 656–665. [Google Scholar] [CrossRef]
- Bentrad, N.; Gaceb-Terrak, R.; Benmalek, Y.; Rahmania, F. Studies on Chemical Composition and Antimicrobial Activities of Bioactive Molecules from Date Palm (Phoenixdactylifera L.) Pollens and Seeds. Afr. J. Tradit. Complement. Altern. Med. 2017, 14, 242–256. [Google Scholar] [CrossRef]
- Elberry, A.A.; Mufti, S.T.; Al-Maghrabi, J.A.; Abdel-Sattar, E.A.; Ashour, O.M.; Ghareib, S.A.; Mosli, H.A. Anti-inflammatory and antiproliferative activities of date palm pollen (Phoenix dactylifera) on experimentally-induced atypical prostatic hyperplasia in rats. J. Inflamm 2011, 8, 40. [Google Scholar] [CrossRef] [PubMed]
- Abedi, A.; Parviz, M.; Karimian, S.M.; Rodsari, H.R.S. Aphrodisiac activity of aqueous extract of Phoenix dactylifera pollen in male rats. Adv. Sex. Med. 2013, 3, 28–34. [Google Scholar] [CrossRef]
- Metwaly, M.S.; Dkhil, M.A.; Al-Quraishy, S. Anti-coccidial and anti-apoptotic activities of palm pollen grains on Eimeria papillata-induced infection in mice. Biologia 2014, 69, 254–259. [Google Scholar] [CrossRef]
- El-Kholy, W.M.; Soliman, T.N.; Darwish, A.M.G. Evaluation of date palm pollen (Phoenix dactylifera L.) encapsulation, impact on the nutritional and functional properties of fortified yoghurt. PLoS ONE 2019, 14, e0222789. [Google Scholar] [CrossRef] [PubMed]
- Abbas, F.; Ateya, A. Estradiol, esteriol, estrone and novel flavonoids from date palm pollen. Aust. J. Appl. Sci. Res. 2011, 7, 606–614. [Google Scholar]
- El-Neweshy, M.S.; El-Maddawy, Z.K.; El-Sayed, Y.S. Therapeutic effects of date palm (Phoenix dactylifera L.) pollen extract on cadmium-induced testicular toxicity. Andrologia 2013, 45, 369–378. [Google Scholar] [CrossRef]
- OECD. Acute oral toxicity-up-and-down procedure. In OECD Guidelines for the Testing of Chemicals; OECD Publishing: Paris, France, 2001; pp. 1–26. [Google Scholar] [CrossRef]
- Arafa, M.H.; Mohammad, N.S.; Atteia, H.H.; Abd-Elaziz, H.R. Protective effect of resveratrol against doxorubicin-induced cardiac toxicity and fibrosis in male experimental rats. J. Physiol. Biochem. 2014, 70, 701–711. [Google Scholar] [CrossRef]
- Tarpey, M.M.; Wink, D.A.; Grisham, M.B. Methods for detection of reactive metabolites of oxygen and nitrogen: In vitro and in vivo considerations. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2004, 286, R431–R444. [Google Scholar] [CrossRef] [PubMed]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Beutler, E.; Duron, O.; Kelly, B.M. Improved method for the determination of blood glutathione. J. Lab. Clin. Med. 1963, 61, 882–888. [Google Scholar]
- Paglia, D.E.; Valentine, W.N. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J. Lab. Clin. Med. 1967, 70, 158–169. [Google Scholar] [PubMed]
- Aebi, H. Catalase in vitro. Methods Enzymol. 1984, 105, 121–126. [Google Scholar] [CrossRef]
- Nishikimi, M.; Appaji, N.; Yagi, K. The occurrence of superoxide anion in the reaction of reduced phenazine methosulfate and molecular oxygen. Biochem. Biophys. Res. Commun. 1972, 46, 849–854. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- El-Agamy, D.S.; El-Harbi, K.M.; Khoshhal, S.; Ahmed, N.; Elkablawy, M.A.; Shaaban, A.A.; Abo-Haded, H.M. Pristimerin protects against doxorubicin-induced cardiotoxicity and fibrosis through modulation of Nrf2 and MAPK/NF-kB signaling pathways. Cancer Manag. Res. 2019, 11, 47–61. [Google Scholar] [CrossRef]
- Bancroft, J.D.; Layton, C. The hematoxylin and eosin, connective and mesenchymal tissues with their stains. In Bancroft s Theory and Practice of Histological Techniques, 7th ed.; Kim, S., Suvarna, C.L., Eds.; Churchill Livingstone: Philadelphia, PA, USA, 2013; pp. 173–186. [Google Scholar]
- Hsu, S.M.; Raine, L.; Fanger, H. The use of antiavidin antibody and avidin-biotin-peroxidase complex in immunoperoxidase technics. Am. J. Clin. Pathol. 1981, 75, 816–821. [Google Scholar] [CrossRef]
- Fan, Z.D.; Zhang, L.; Shi, Z.; Gan, X.B.; Gao, X.Y.; Zhu, G.Q. Artificial microRNA interference targeting AT(1a) receptors in paraventricular nucleus attenuates hypertension in rats. Gene Ther. 2012, 19, 810–817. [Google Scholar] [CrossRef]
- Ye, F.; He, J.; Wu, X.; Xie, J.; Chen, H.; Tang, X.; Lai, Z.; Huang, R.; Huang, J. The regulatory mechanisms of Yulangsan MHBFC reversing cardiac remodeling in rats based on eNOS-NO signaling pathway. Biomed. Pharmacother. 2019, 117, 109141. [Google Scholar] [CrossRef]
- Elblehi, S.S.; El Euony, O.I.; El-Sayed, Y.S. Apoptosis and astrogliosis perturbations and expression of regulatory inflammatory factors and neurotransmitters in acrylamide-induced neurotoxicity under omega3 fatty acids protection in rats. Neurotoxicology 2020, 76, 44–57. [Google Scholar] [CrossRef]
- Mitry, M.A.; Edwards, J.G. Doxorubicin induced heart failure: Phenotype and molecular mechanisms. Int. J. Cardiol. Heart Vasc. 2016, 10, 17–24. [Google Scholar] [CrossRef]
- Nebigil, C.G.; Desaubry, L. Updates in anthracycline-mediated cardiotoxicity. Front. Pharmacol. 2018, 9, 1262. [Google Scholar] [CrossRef]
- Angsutararux, P.; Luanpitpong, S.; Issaragrisil, S. Chemotherapy-induced cardiotoxicity: Overview of the roles of oxidative stress. Oxid Med. Cell Longev. 2015, 2015, 795602. [Google Scholar] [CrossRef]
- Govender, J.; Loos, B.; Marais, E.; Engelbrecht, A.M. Melatonin improves cardiac and mitochondrial function during doxorubicin-induced cardiotoxicity: A possible role for peroxisome proliferator-activated receptor gamma coactivator 1-alpha and sirtuin activity? Toxicol. Appl. Pharmacol. 2018, 358, 86–101. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Sun, G.B.; Sun, B.; Wu, Y.; He, L.; Wang, X.; Chen, R.C.; Cao, L.; Ren, X.Y.; Sun, X.B. Kaempferol protects against doxorubicin-induced cardiotoxicity in vivo and in vitro. Toxicology 2012, 292, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Geisberg, C.A.; Sawyer, D.B. Mechanisms of anthracycline cardiotoxicity and strategies to decrease cardiac damage. Curr. Hypertens Rep. 2010, 12, 404–410. [Google Scholar] [CrossRef]
- Jardan, Y.A.B.; Ansari, M.A.; Raish, M.; Alkharfy, K.M.; Ahad, A.; Al-Jenoobi, F.I.; Haq, N.; Khan, M.R.; Ahmad, A. Sinapic acid ameliorates oxidative stress, inflammation, and apoptosis in acute doxorubicin-induced cardiotoxicity via the NF-kappaB-mediated pathway. Biomed. Res. Int. 2020, 2020, 3921796. [Google Scholar] [CrossRef]
- Dos Santos, J.M.; Alfredo, T.M.; Antunes, K.A.; da Cunha, J.; Costa, E.M.A.; Lima, E.S.; Silva, D.B.; Carollo, C.A.; Schmitz, W.O.; Boleti, A.P.A.; et al. Guazuma ulmifolia Lam. decreases oxidative stress in blood cells and prevents doxorubicin-induced cardiotoxicity. Oxid. Med. Cell Longev. 2018, 2018, 2935051. [Google Scholar] [CrossRef] [PubMed]
- Cvetkovic, R.S.; Scott, L.J. Dexrazoxane: A review of its use for cardioprotection during anthracycline chemotherapy. Drugs 2005, 65, 1005–1024. [Google Scholar] [CrossRef]
- Zhang, Q.L.; Yang, J.J.; Zhang, H.S. Carvedilol (CAR) combined with carnosic acid (CAA) attenuates doxorubicin-induced cardiotoxicity by suppressing excessive oxidative stress, inflammation, apoptosis and autophagy. Biomed. Pharmacother. 2019, 109, 71–83. [Google Scholar] [CrossRef]
- Wu, R.; Yao, P.A.; Wang, H.L.; Gao, Y.; Yu, H.L.; Wang, L.; Cui, X.H.; Xu, X.; Gao, J.P. Effect of fermented Cordyceps sinensis on doxorubicin-induced cardiotoxicity in rats. Mol. Med. Rep. 2018, 18, 3229–3241. [Google Scholar] [CrossRef] [PubMed]
- Al-Taee, H.; Azimullah, S.; Meeran, M.F.N.; Almheiri, M.K.A.; Al Jasmi, R.A.; Tariq, S.; Ab Khan, M.; Adeghate, E.; Ojha, S. beta-caryophyllene, a dietary phytocannabinoid attenuates oxidative stress, inflammation, apoptosis and prevents structural alterations of the myocardium against doxorubicin-induced acute cardiotoxicity in rats: An in vitro and in vivo study. Eur. J. Pharmacol. 2019, 858, 172467. [Google Scholar] [CrossRef]
- Wu, R.; Mei, X.; Wang, J.; Sun, W.; Xue, T.; Lin, C.; Xu, D. Zn(ii)-curcumin supplementation alleviates gut dysbiosis and zinc dyshomeostasis during doxorubicin-induced cardiotoxicity in rats. Food Funct. 2019, 10, 5587–5604. [Google Scholar] [CrossRef] [PubMed]
- Herman, E.H.; Zhang, J.; Chadwick, D.P.; Ferrans, V.J. Comparison of the protective effects of amifostine and dexrazoxane against the toxicity of doxorubicin in spontaneously hypertensive rats. Cancer Chemother. Pharmacol. 2000, 45, 329–334. [Google Scholar] [CrossRef]
- Jenkins, G.R.; Lee, T.; Moland, C.L.; Vijay, V.; Herman, E.H.; Lewis, S.M.; Davis, K.J.; Muskhelishvili, L.; Kerr, S.; Fuscoe, J.C.; et al. Sex-related differential susceptibility to doxorubicin-induced cardiotoxicity in B6C3F1 mice. Toxicol. Appl. Pharmacol. 2016, 310, 159–174. [Google Scholar] [CrossRef]
- Sahu, B.D.; Kumar, J.M.; Kuncha, M.; Borkar, R.M.; Srinivas, R.; Sistla, R. Baicalein alleviates doxorubicin-induced cardiotoxicity via suppression of myocardial oxidative stress and apoptosis in mice. Life Sci. 2016, 144, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, L.A.; El-Maraghy, S.A. Nicorandil ameliorates mitochondrial dysfunction in doxorubicin-induced heart failure in rats: Possible mechanism of cardioprotection. Biochem. Pharmacol. 2013, 86, 1301–1310. [Google Scholar] [CrossRef]
- Kuzman, J.A.; O’Connell, T.D.; Gerdes, A.M. Rapamycin prevents thyroid hormone-induced cardiac hypertrophy. Endocrinology 2007, 148, 3477–3484. [Google Scholar] [CrossRef]
- Zhang, Y.; Ma, C.; Liu, C.; Wei, F. Luteolin attenuates doxorubicin-induced cardiotoxicity by modulating the PHLPP1/AKT/Bcl-2 signalling pathway. Peer J. 2020, 8, e8845. [Google Scholar] [CrossRef] [PubMed]
- Karagiannis, T.C.; Lin, A.J.; Ververis, K.; Chang, L.; Tang, M.M.; Okabe, J.; El-Osta, A. Trichostatin A accentuates doxorubicin-induced hypertrophy in cardiac myocytes. Aging 2010, 2, 659–668. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Zhang, J.; Ni, T.; Lin, N.; Meng, L.; Gao, F.; Luo, H.; Liu, X.; Chi, J.; Guo, H. Yellow wine polyphenolic compounds prevents doxorubicin-induced cardiotoxicity through activation of the Nrf2 signalling pathway. J. Cell Mol. Med. 2019, 23, 6034–6047. [Google Scholar] [CrossRef] [PubMed]
- Zare, M.F.R.; Rakhshan, K.; Aboutaleb, N.; Nikbakht, F.; Naderi, N.; Bakhshesh, M.; Azizi, Y. Apigenin attenuates doxorubicin induced cardiotoxicity via reducing oxidative stress and apoptosis in male rats. Life Sci. 2019, 232, 116623. [Google Scholar] [CrossRef]
- Folsom, A.R.; Nambi, V.; Bell, E.J.; Oluleye, O.W.; Gottesman, R.F.; Lutsey, P.L.; Huxley, R.R.; Ballantyne, C.M. Troponin T, N-terminal pro-B-type natriuretic peptide, and incidence of stroke: The atherosclerosis risk in communities study. Stroke 2013, 44, 961–967. [Google Scholar] [CrossRef] [PubMed]
- Desai, A.S.; Lewis, E.F.; Li, R.; Solomon, S.D.; Assmann, S.F.; Boineau, R.; Clausell, N.; Diaz, R.; Fleg, J.L.; Gordeev, I.; et al. Rationale and design of the treatment of preserved cardiac function heart failure with an aldosterone antagonist trial: A randomized, controlled study of spironolactone in patients with symptomatic heart failure and preserved ejection fraction. Am. Heart J. 2011, 162, 966–972.e910. [Google Scholar] [CrossRef] [PubMed]
- Argun, M.; Uzum, K.; Sonmez, M.F.; Ozyurt, A.; Derya, K.; Cilenk, K.T.; Unalmis, S.; Pamukcu, O.; Baykan, A.; Narin, F.; et al. Cardioprotective effect of metformin against doxorubicin cardiotoxicity in rats. Anatol. J. Cardiol. 2016, 16, 234–241. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Li, W.R.; Jin, Y.G.; Jie, Q.Q.; Wang, C.Y.; Wu, L. Tetrandrine attenuated doxorubicin-induced acute cardiac injury in mice. Biomed. Res. Int. 2020, 2020, 2616024. [Google Scholar] [CrossRef]
- Quagliariello, V.; Vecchione, R.; Coppola, C.; Di Cicco, C.; De Capua, A.; Piscopo, G.; Paciello, R.; Narciso, V.; Formisano, C.; Taglialatela-Scafati, O.; et al. Cardioprotective effects of nanoemulsions loaded with anti-inflammatory nutraceuticals against doxorubicin-induced cardiotoxicity. Nutrients 2018, 10, 1304. [Google Scholar] [CrossRef]
- Haybar, H.; Goudarzi, M.; Mehrzadi, S.; Aminzadeh, A.; Khodayar, M.J.; Kalantar, M.; Fatemi, I. Effect of gemfibrozil on cardiotoxicity induced by doxorubicin in male experimental rats. Biomed. Pharmacother. 2019, 109, 530–535. [Google Scholar] [CrossRef]
- Mohamed, N.A.; Ahmed, O.M.; Hozayen, W.G.; Ahmed, M.A. Ameliorative effects of bee pollen and date palm pollen on the glycemic state and male sexual dysfunctions in streptozotocin-Induced diabetic wistar rats. Biomed. Pharmacother. 2018, 97, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.F.; Khan, G.; Safhi, M.M.; Alshahrani, S.; Siddiqui, R.; Moni, S.S.; Anwer, T. Thymoquinone ameliorates doxorubicin-induced cardiotoxicity in Swiss Albino mice by modulating oxidative damage and cellular inflammation. Cardiol. Res. Pract. 2018, 2018, 1483041. [Google Scholar] [CrossRef]
- Baniahmad, B.; Safaeian, L.; Vaseghi, G.; Rabbani, M.; Mohammadi, B. Cardioprotective effect of vanillic acid against doxorubicin-induced cardiotoxicity in rat. Res. Pharm. Sci. 2020, 15, 87–96. [Google Scholar] [CrossRef]
- Ohta, S.; Fujimaki, T.; Uy, M.M.; Yanai, M.; Yukiyoshi, A.; Hirata, T. Antioxidant hydroxycinnamic acid derivatives isolated from Brazilian bee pollen. Nat. Prod. Res. 2007, 21, 726–732. [Google Scholar] [CrossRef]
- Hassan, W.; El-kashlan, A.M.; Mohamed, N. Egyptian date palm pollen ameliorates testicular dysfunction induced by cadmium chloride in adult male rats. Am. Sci. J. 2012, 8, 659–669. [Google Scholar]
- Caldwell, C.R. Alkylperoxyl radical scavenging activity of red leaf lettuce (Lactuca sativa L.) phenolics. J. Agric. Food Chem. 2003, 51, 4589–4595. [Google Scholar] [CrossRef]
- Bai, Z.; Wang, Z. Genistein protects against doxorubicin-induced cardiotoxicity through Nrf-2/HO-1 signaling in mice model. Environ. Toxicol. 2019, 34, 645–651. [Google Scholar] [CrossRef]
- Alanazi, A.M.; Fadda, L.; Alhusaini, A.; Ahmad, R.; Hasan, I.H.; Mahmoud, A.M. Liposomal resveratrol and/or carvedilol attenuate doxorubicin-induced cardiotoxicity by modulating inflammation, oxidative stress and S100a1 in rats. Antioxidants 2020, 9, 159. [Google Scholar] [CrossRef]
- Hafez, H.M.; Hassanein, H. Montelukast ameliorates doxorubicin-induced cardiotoxicity via modulation of p-glycoprotein and inhibition of ROS-mediated TNF-alpha/NF-kappaB pathways. Drug Chem. Toxicol. 2020, 5, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Zhang, X.; Zhang, N.; Wei, W.Y.; Li, L.L.; Ma, Z.G.; Tang, Q.Z. Osteocrin attenuates inflammation, oxidative stress, apoptosis, and cardiac dysfunction in doxorubicin-induced cardiotoxicity. Clin. Trans. Med. 2020, 2, 124. [Google Scholar] [CrossRef]
- Asensio-Lopez, M.C.; Soler, F.; Pascual-Figal, D.; Fernandez-Belda, F.; Lax, A. Doxorubicin-induced oxidative stress: The protective effect of nicorandil on HL-1 cardiomyocytes. PLoS ONE 2017, 12, e0172803. [Google Scholar] [CrossRef]
- Xiong, C.; Wu, Y.Z.; Zhang, Y.; Wu, Z.X.; Chen, X.Y.; Jiang, P.; Guo, H.C.; Xie, K.R.; Wang, K.X.; Su, S.W. Protective effect of berberine on acute cardiomyopathy associated with doxorubicin treatment. Oncol. Lett. 2018, 15, 5721–5729. [Google Scholar] [CrossRef] [PubMed]
- Ansari, M.A.; Raish, M.; Ahmad, A.; Alkharfy, K.M.; Ahmad, S.F.; Attia, S.M.; Alsaad, A.M.S.; Bakheet, S.A. Sinapic acid ameliorate cadmium-induced nephrotoxicity: In vivo possible involvement of oxidative stress, apoptosis, and inflammation via NF-kappaB downregulation. Environ. Toxicol. Pharmacol. 2017, 51, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Saad, S.Y.; Najjar, T.A.; Al-Rikabi, A.C. The preventive role of deferoxamine against acute doxorubicin-induced cardiac, renal and hepatic toxicity in rats. Pharmacol. Res. 2001, 43, 211–218. [Google Scholar] [CrossRef]
- Carvalho, F.S.; Burgeiro, A.; Garcia, R.; Moreno, A.J.; Carvalho, R.A.; Oliveira, P.J. Doxorubicin-induced cardiotoxicity: From bioenergetic failure and cell death to cardiomyopathy. Med. Res. Rev. 2014, 34, 106–135. [Google Scholar] [CrossRef] [PubMed]
- Mattila, M.; Soderstrom, M.; Ailanen, L.; Savontaus, E.; Savontaus, M. The Effects of neuropeptide Y overexpression on the mouse model of doxorubicin-induced cardiotoxicity. Cardiovasc. Toxicol. 2020, 20, 328–338. [Google Scholar] [CrossRef] [PubMed]
- Menon, S.; Lawrence, L.; Sivaram, V.P.; Padikkala, J. Oroxylum indicum root bark extract prevents doxorubicin-induced cardiac damage by restoring redox balance. J. Ayurveda Integr. Med. 2019, 10, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Lagranha, C.J.; Deschamps, A.; Aponte, A.; Steenbergen, C.; Murphy, E. Sex differences in the phosphorylation of mitochondrial proteins result in reduced production of reactive oxygen species and cardioprotection in females. Circ. Res. 2010, 106, 1681–1691. [Google Scholar] [CrossRef]
- Li, C.; Liu, Y.; Xie, Z.; Lu, Q.; Luo, S. Stigmasterol protects against Ang II-induced proliferation of the A7r5 aortic smooth muscle cell-line. Food Funct. 2015, 6, 2266–2272. [Google Scholar] [CrossRef] [PubMed]
- Lei, L.; Wang, X.; Huang, W.; Liu, Y.; Zheng, F.; Ma, K.Y.; Li, Y.M.; Wang, L.; Man, S.W.; Zhang, C.; et al. Cholesterol side chain analogs but not its ether analogs possess cholesterol-lowering activity. Food Funct. 2015, 6, 630–634. [Google Scholar] [CrossRef]
- Zou, Z.; Xu, X.; Huang, Y.; Xiao, X.; Ma, L.; Sun, T.; Dong, P.; Wang, X.; Lin, X. High serum level of lutein may be protective against early atherosclerosis: The Beijing atherosclerosis study. Atherosclerosis 2011, 219, 789–793. [Google Scholar] [CrossRef]
- Zou, Z.Y.; Xu, X.R.; Lin, X.M.; Zhang, H.B.; Xiao, X.; Ouyang, L.; Huang, Y.M.; Wang, X.; Liu, Y.Q. Effects of lutein and lycopene on carotid intima-media thickness in Chinese subjects with subclinical atherosclerosis: A randomised, double-blind, placebo-controlled trial. Br. J. Nutr. 2014, 111, 474–480. [Google Scholar] [CrossRef] [PubMed]
- Wong, W.Y.; Ward, L.C.; Fong, C.W.; Yap, W.N.; Brown, L. Anti-inflammatory gamma- and delta-tocotrienols improve cardiovascular, liver and metabolic function in diet-induced obese rats. Eur. J. Nutr. 2017, 56, 133–150. [Google Scholar] [CrossRef]
- Ibarra, M.; Perez-Vizcaino, F.; Cogolludo, A.; Duarte, J.; Zaragoza-Arnaez, F.; Lopez-Lopez, J.G.; Tamargo, J. Cardiovascular effects of isorhamnetin and quercetin in isolated rat and porcine vascular smooth muscle and isolated rat atria. Planta Med. 2002, 68, 307–310. [Google Scholar] [CrossRef]
- Timm, K.N.; Tyler, D.J. The role of AMPK activation for cardioprotection in doxorubicin-induced cardiotoxicity. Cardiovasc. Drugs Ther. 2020, 34, 255–269. [Google Scholar] [CrossRef] [PubMed]
- Hamza, A.A.; Ahmed, M.M.; Elwey, H.M.; Amin, A. Melissa officinalis protects against doxorubicin-induced cardiotoxicity in rats and potentiates its anticancer activity on MCF-7 cells. PLoS ONE 2016, 11, e0167049. [Google Scholar] [CrossRef]
- Wang, B.; Hao, J.; Jones, S.C.; Yee, M.S.; Roth, J.C.; Dixon, I.M. Decreased Smad 7 expression contributes to cardiac fibrosis in the infarcted rat heart. Am. J. Physiol. Heart Circ. Physiol. 2002, 282, H1685–H1696. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.-Y.; Li, K.; Li, Y.; Tian, X.-T.; Ba, H.-X.; Wang, A.; Li, X.-H. Dendrobium candidum aqueous extract attenuates isoproterenol-induced cardiac hypertrophy through the ERK signalling pathway. Pharm. Biol. 2020, 58, 176–183. [Google Scholar] [CrossRef]
- El-Said, N.T.; Mohamed, E.A.; Taha, R.A. Irbesartan suppresses cardiac toxicity induced by doxorubicin via regulating the p38-MAPK/NF-kappaB and TGF-beta1 pathways. Naunyn Schmiedebergs Arch. Pharm. 2019, 392, 647–658. [Google Scholar] [CrossRef]
- Sun, Z.; Lu, W.; Lin, N.; Lin, H.; Zhang, J.; Ni, T.; Meng, L.; Zhang, C.; Guo, H. Dihydromyricetin alleviates doxorubicin-induced cardiotoxicity by inhibiting NLRP3 inflammasome through activation of SIRT1. Biochem. Pharmacol. 2020, 175, 113888. [Google Scholar] [CrossRef]
- Xu, S.; Wang, Y.; Yu, M.; Wang, D.; Liang, Y.; Chen, Y.; Liao, C.; Xie, Z.; Zhao, B.; Han, J.; et al. LongShengZhi capsule inhibits doxorubicin-induced heart failure by anti-oxidative stress. Biomed. Pharmacother. 2020, 123, 109803. [Google Scholar] [CrossRef]
- Deavall, D.G.; Martin, E.A.; Horner, J.M.; Roberts, R. Drug-induced oxidative stress and toxicity. J. Toxicol. 2012, 2012, 645460. [Google Scholar] [CrossRef] [PubMed]
- Radhiga, T.; Rajamanickam, C.; Sundaresan, A.; Ezhumalai, M.; Pugalendi, K.V. Effect of ursolic acid treatment on apoptosis and DNA damage in isoproterenol-induced myocardial infarction. Biochimie 2012, 94, 1135–1142. [Google Scholar] [CrossRef]
- Zamzami, N.; Marchetti, P.; Castedo, M.; Zanin, C.; Vayssiere, J.L.; Petit, P.X.; Kroemer, G. Reduction in mitochondrial potential constitutes an early irreversible step of programmed lymphocyte death in vivo. J. Exp. Med. 1995, 181, 1661–1672. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, B.; Zhou, C.; Bi, Y. Matrine induces apoptosis in angiotensin II-stimulated hyperplasia of cardiac fibroblasts: Effects on Bcl-2/Bax expression and caspase-3 activation. Basic Clin. Pharm. Toxicol. 2007, 101, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Zaki, S.M.; Algaleel, W.A.; Imam, R.A.; Abdelmoaty, M.M. Mesenchymal stem cells pretreated with platelet-rich plasma modulate doxorubicin-induced cardiotoxicity. Hum. Exp. Toxicol. 2019, 38, 857–874. [Google Scholar] [CrossRef] [PubMed]




| Groups | Initial Body Weight (g) | Body Weight (g) | Heart Weight (g) | RHW (%) | Cardiomyocyte Cross-Sectional Area (μm2) |
|---|---|---|---|---|---|
| CTR | 194 ± 4.5 | 218.43 ± 4.86 | 0.85 ± 0.013 | 0.52 ± 0.07 | 138.03 ± 3.90 |
| DPPE | 195 ± 5.2 | 221.85 ± 6.51 | 0.87 ± 0.023 | 0.49 ± 0.06 | 135.40 ± 2.65 |
| DOX | 190 ± 8.1 | 189.14 ± 8.36 * | 1.36 ± 0.047 * | 0.88 ± 0.11 * | 210.87 ± 5.26 * |
| DOX + DPPE | 192 ± 3.6 | 204.43 ± 2.72 | 1.04 ± 0.061 † | 0.63 ± 0.09 | 154.94 ± 2.06 † |
| Parameters | CTR | DPPE | DOX | DPPE + DOX |
|---|---|---|---|---|
| ALT (U/L) | 69.26 ± 1.98 | 66.39 ± 4.25 | 105.09 ± 3.71 * | 83.71 ± 3.04 † |
| AST (U/L) | 143.99 ± 4.74 | 140.25 ± 5.32 | 203.97 ± 6.98 * | 175.36 ± 6.27 † |
| LDH (U/L) | 496.99 ± 18.28 | 485.76 ± 31.16 | 1237.62 ± 65.72 * | 908.19 ± 9.61 † |
| CK (U/L) | 218.63 ± 17.16 | 203.71 ± 5.81 | 612.92 ± 7.97 * | 431.50 ± 17.14 † |
| CKMP (U/L) | 458.99 ± 24.39 | 440.86 ± 32.52 | 934.56 ± 41.55 * | 682.14 ± 37.44 † |
| CTnI (pg/mL) | 0.37 ± 0.05 | 0.30 ± 0.05 | 1.85 ± 0.38 * | 1.07 ± 0.13 † |
| CTnT (pg/mL) | 0.91 ± 0.018 | 0.87 ± 0.03 | 3.15 ± 0.17 * | 1.69 ± 0.15 † |
| NT- ProBNP (pg/mL) | 63.45 ± 3.73 | 57.31 ± 2.089 | 151.17 ± 3.44 * | 96.55 ± 4.02 † |
| Ca+2 (μg/g tissue) | 32.80 ± 3.60 | 31.81 ± 2.95 | 77.35 ± 5.54 * | 54.03 ± 5.17 † |
| Parameters | CTR | DPPE | DOX | DPPE + DOX |
|---|---|---|---|---|
| NO (μmol/g tissue) | 4.84 ± 0.93 | 3.32 ± 1.09 | 17.49 ± 2.45 * | 10.25 ± 1.07 † |
| MDA (nmol/g tissue) | 37.35 ± 2.41 | 34.48 ± 2.73 | 69.52 ± 3.52 * | 48.3726 ± 2.29 † |
| GSH (mmol/g tissue) | 25.27 ± 2.96 | 28.71 ± 1.65 | 11.73 ± 1.55 * | 19.63 ± 0.85 † |
| GPx (U/g tissue) | 17.23 ± 1.48 | 19.08 ± 2.21 | 5.42 ± 1.26 * | 11.84 ± 1.48 † |
| CAT (U/g tissue) | 34.61 ± 2.19 | 37.64 ± 2.38 | 18.92 ± 2.44 * | 26.75 ± 2.70 † |
| SOD (U/g tissue) | 13.71 ± 1.318 | 15.45 ± 1.21 | 4.31 ± 1.22 * | 9.85 ± 1.51 † |
| Parameters | CTR | DPPE | DOX | DPPE + DOX |
|---|---|---|---|---|
| NF-κB p65 (ng/g tissue) | 90.23 ± 3.5 | 83.65 ± 3.8 | 238.09 ± 22.5 * | 172.11 ± 14.8 † |
| TNF-α (pg/g tissue) | 34.29 ± 1.65 | 32.89 ± 1.89 | 80.75 ± 5.87 * | 50.67 ± 4.81 † |
| IL-1β (pg/g tissue) | 79.81 ± 1.6 | 81.54 ± 1.7 | 138.85 ± 3.23 * | 114.24 ± 3.65 † |
| IL-6 (pg/g tissue) | 47.20 ± 2.66 | 44.13 ± 1.71 | 87.63 ± 2.93 * | 64.64 ± 2.21 † |
| Hydroxyproline (μg/g tissue) | 22.67 ± 2.03 | 20.61 ± 1.92 | 46.33 ± 2.21 * | 38.3 ± 2.87 † |
| Groups (n = 7) | Lesion Severity | Loss of Muscular Striations | Myocardial Vacuolation | Myocardial Necrosis | Myofibrillar Loss | Interstitial Inflammatory Cells Infiltrations | Hyperemic Blood Vessels | Interstitial Edema | Interfibrillar Hemorrhage | Myocardial Fibrosis |
|---|---|---|---|---|---|---|---|---|---|---|
| CTR | None | 6 | 7 | 7 | 7 | 7 | 7 | 7 | 7 | 7 |
| (−) | −85.71% | −100% | −100% | −100% | −100% | −100% | −100% | −100% | −100% | |
| Mild | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| (+) | −14.28% | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% | |
| Moderate | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| (++) | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% | |
| Severe | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| (+++) | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% | |
| DPPE | None | 7 | 7 | 7 | 7 | 7 | 7 | 7 | 7 | 7 |
| (−) | −100% | −100% | −100% | −100% | −100% | −100% | −100% | −100% | −100% | |
| Mild | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| (+) | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% | |
| Moderate | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| (++) | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% | |
| Severe | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| (+++) | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% | |
| DOX | None | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| (−) | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% | |
| Mild | 0 | 0 | 0 | 1 | 0 | 0 | 1 | 1 | 1 | |
| (+) | 0% | 0% | 0% | −14.28% | 0% | 0% | −14.28% | −14.28% | −14.28% | |
| Moderate | 2 | 6 | 5 | 6 | 7 | 5 | 6 | 5 | 6 | |
| (++) | −28.57% | −85.71% | −71.42% | −85.71% | −100% | −71.42% | −85.71 | −71.42% | −85.71% | |
| Severe | 5 | 1 | 2 | 0 | 0 | 2 | 0 | 1 | 0 | |
| (+++) | (71.42%) | −14.28% | −28.57% | 0% | 0% | −28.57% | 0% | −14.28% | 0% | |
| DPPE + DOX | None | 0 | 0 | 0 | 1 | 0 | 0 | 1 | 2 | 0 |
| (−) | 0% | 0% | 0% | −14.28% | 0% | 0% | −14.28% | −28.57% | 0% | |
| Mild | 5 | 5 | 4 | 4 | 2 | 5 | 5 | 3 | 5 | |
| (+) | (71.42%) | −71.42% | −57.14% | −57.14% | −28.57% | (71.42%) | −71.42% | −42.85% | −71.42% | |
| Moderate | 2 | 2 | 3 | 2 | 5 | 2 | 1 | 2 | 2 | |
| (++) | −28.57% | −28.57% | −42.85% | −28.57% | −71.42% | −28.57% | −14.28% | −28.57% | −28.57% | |
| Severe | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| (+++) | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% | 0% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elblehi, S.S.; El-Sayed, Y.S.; Soliman, M.M.; Shukry, M. Date Palm Pollen Extract Avert Doxorubicin-Induced Cardiomyopathy Fibrosis and Associated Oxidative/Nitrosative Stress, Inflammatory Cascade, and Apoptosis-Targeting Bax/Bcl-2 and Caspase-3 Signaling Pathways. Animals 2021, 11, 886. https://doi.org/10.3390/ani11030886
Elblehi SS, El-Sayed YS, Soliman MM, Shukry M. Date Palm Pollen Extract Avert Doxorubicin-Induced Cardiomyopathy Fibrosis and Associated Oxidative/Nitrosative Stress, Inflammatory Cascade, and Apoptosis-Targeting Bax/Bcl-2 and Caspase-3 Signaling Pathways. Animals. 2021; 11(3):886. https://doi.org/10.3390/ani11030886
Chicago/Turabian StyleElblehi, Samar S., Yasser S. El-Sayed, Mohamed Mohamed Soliman, and Mustafa Shukry. 2021. "Date Palm Pollen Extract Avert Doxorubicin-Induced Cardiomyopathy Fibrosis and Associated Oxidative/Nitrosative Stress, Inflammatory Cascade, and Apoptosis-Targeting Bax/Bcl-2 and Caspase-3 Signaling Pathways" Animals 11, no. 3: 886. https://doi.org/10.3390/ani11030886
APA StyleElblehi, S. S., El-Sayed, Y. S., Soliman, M. M., & Shukry, M. (2021). Date Palm Pollen Extract Avert Doxorubicin-Induced Cardiomyopathy Fibrosis and Associated Oxidative/Nitrosative Stress, Inflammatory Cascade, and Apoptosis-Targeting Bax/Bcl-2 and Caspase-3 Signaling Pathways. Animals, 11(3), 886. https://doi.org/10.3390/ani11030886









