Use of a Tri-Axial Accelerometer Can Reliably Detect Play Behaviour in Newborn Calves
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Calf Recruitment and Data Collection
2.2. Extraction of IceTag Accelerometer Data
2.3. Correlation between Motion Index and Behavioural Observations
2.3.1. Behavioural Analysis
2.3.2. Statistical Analysis
2.4. Calculation of Sensitivity and Specificity and Comparison with One-Zero Sampling
2.4.1. Behavioural Analysis
2.4.2. Statistical Analysis
Calculation of Sensitivity and Specificity of MI to Detect Play Behaviour
Comparison between Motion Index and Visual One-Zero Sampling
3. Results
3.1. Correlation between Motion Index and Behavioural Observations
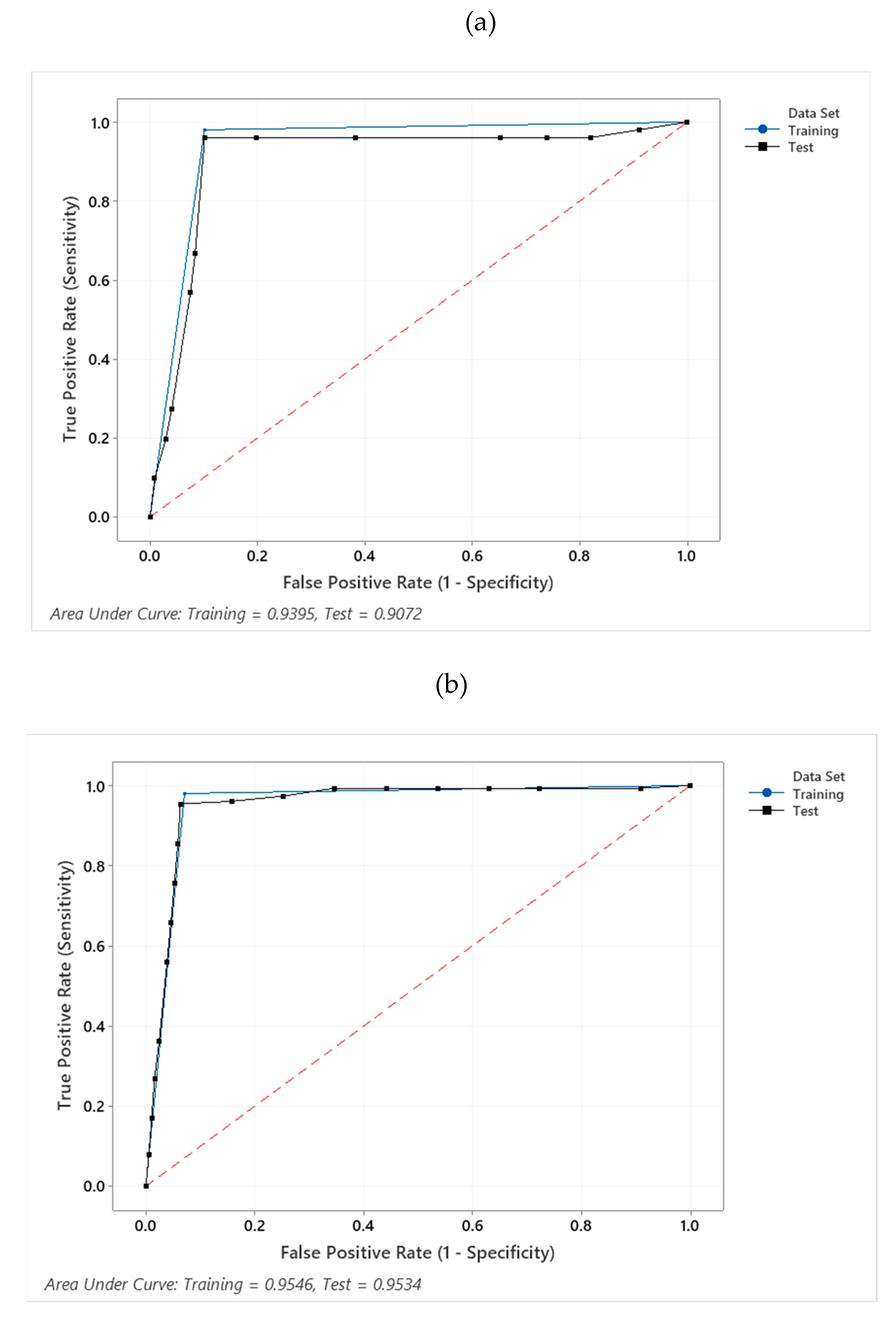
3.2. Sensitivity, Specificity and Comparison with One-Zero Sampling
3.2.1. Sensitivity and Specificity
3.2.2. Comparison between Motion Index and Visual One-Zero Sampling
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ortega, D.L.; Wolf, C.A. Demand for farm animal welfare and producer implications: Results from a field experiment in Michigan. Food Policy 2018, 74, 74–81. [Google Scholar] [CrossRef]
- von Keyserlingk, M.A.G.; Weary, D.M. A 100-Year Review: Animal welfare in the Journal of Dairy Science—The first 100 years. J. Dairy Sci. 2017, 100, 10432–10444. [Google Scholar] [CrossRef] [PubMed]
- Main, D.C.J.; Webster, A.J.F.; Green, L.E. Animal Welfare Assessment in Farm Assurance Schemes. Acta Agric. Scand. Sect. A Anim. Sci. 2001, 51, 108–113. [Google Scholar] [CrossRef]
- Main, D.C.J.; Mullan, S.; Atkinson, C.; Bond, A.; Cooper, M.; Fraser, A.; Browne, W.J. Welfare outcomes assessment in laying hen farm assurance schemes. Anim. Welf. 2012, 21, 389–396. [Google Scholar] [CrossRef]
- Main, D.C.J.; Whay, H.R.; Green, L.E.; Webster, A.J.F. Effect of the RSPCA Freedom Food scheme on the welfare of dairy cattle. Vet. Rec. 2003, 153, 227–231. [Google Scholar] [CrossRef] [PubMed]
- Veissier, I.; Butterworth, A.; Bock, B.; Roe, E. European approaches to ensure good animal welfare. Appl. Anim. Behav. Sci. 2008, 113, 279–297. [Google Scholar] [CrossRef]
- Farm Animal Welfare Council Report on the Welfare Implications of Farm Assurance Schemes. 2005. Available online: https://www.gov.uk/government/publications/fawc-report-on-the-welfare-implications-of-farm-assurance-schemes (accessed on 8 May 2019).
- Webster, A.J.F.; Main, D.C.J.; Whay, H.R. Welfare assessment: Indices from clinical observation. Anim. Welf. 2004, 13, 93–98. [Google Scholar]
- Lawrence, A.B.; Vigors, B.; Sandøe, P. What is so positive about positive animal welfare?—A critical review of the literature. Animals 2019, 9, 783. [Google Scholar] [CrossRef]
- Mellor, D.J. Positive animal welfare states and encouraging environment-focused and animal-to-animal interactive behaviours. N. Z. Vet. J. 2015, 63, 9–16. [Google Scholar] [CrossRef]
- Napolitano, F.; Knierim, U.; Grass, F.; De Rosa, G. Positive indicators of cattle welfare and their applicability to on-farm protocols. Ital. J. Anim. Sci. 2009, 8, 355–365. [Google Scholar] [CrossRef]
- Mellor, D.J. Animal emotions, behaviour and the promotion of positive welfare states. N. Z. Vet. J. 2012, 60, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Barrell, G.K. An Appraisal of Methods for Measuring Welfare of Grazing Ruminants. Front. Vet. Sci. 2019, 6, 289. [Google Scholar] [CrossRef] [PubMed]
- Martin, P.; Bateson, P. Recording Methods. In Measuring Behaviour An Introductory Guide; Cambridge University Press: Cambridge, UK, 2009; pp. 48–61. [Google Scholar]
- Altmann, J. Observational Study of Behavior: Sampling Methods. Behaviour 1974, 49, 227–267. [Google Scholar] [CrossRef] [PubMed]
- Mitlöhner, F.M.; Morrow-Tesch, J.L.; Wilson, S.C.; Dailey, J.W.; McGlone, J.J. Behavioral sampling techniques for feedlot cattle. J. Anim. Sci. 2001, 79, 1189–1193. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.M.; Schütz, K.E.; Tucker, C.B. Technical note: Comparison of instantaneous sampling and continuous observation of dairy cattle behavior in freestall housing. J. Dairy Sci. 2016, 99, 8341–8346. [Google Scholar] [CrossRef]
- Hämäläinen, W.; Ruuska, S.; Kokkonen, T.; Orkola, S.; Mononen, J. Measuring behaviour accurately with instantaneous sampling: A new tool for selecting appropriate sampling intervals. Appl. Anim. Behav. Sci. 2016, 180, 166–173. [Google Scholar] [CrossRef]
- Brown, D.D.; Kays, R.; Wikelski, M.; Wilson, R.; Klimley, A.P. Observing the unwatchable through acceleration logging of animal behavior. Anim. Biotelemetry 2013, 1. [Google Scholar] [CrossRef]
- Rushen, J.; Chapinal, N.; De Passillé, A.M. Automated monitoring of behavioural-based animal welfare indicators. Anim. Welf. 2012, 21, 339–350. [Google Scholar] [CrossRef]
- Vasseur, E.; Rushen, J.; Haley, D.B.; de Passillé, A.M. Sampling cows to assess lying time for on-farm animal welfare assessment. J. Dairy Sci. 2012, 95, 4968–4977. [Google Scholar] [CrossRef]
- Ungar, E.D.; Nevo, Y.; Baram, H.; Arieli, A. Evaluation of the IceTag leg sensor and its derivative models to predict behaviour, using beef cattle on rangeland. J. Neurosci. Methods 2018, 300, 127–137. [Google Scholar] [CrossRef]
- Müller, R.; Schrader, L. A new method to measure behavioural activity levels in dairy cows. Appl. Anim. Behav. Sci. 2003, 83, 247–258. [Google Scholar] [CrossRef]
- Neethirajan, S. Recent advances in wearable sensors for animal health management. Sens. Bio-Sens. Res. 2017, 12, 15–29. [Google Scholar] [CrossRef]
- Studd, E.K.; Boudreau, M.R.; Majchrzak, Y.N.; Menzies, A.K.; Peers, M.J.L.; Seguin, J.L.; Lavergne, S.G.; Boonstra, R.; Murray, D.L.; Boutin, S.; et al. Use of Acceleration and Acoustics to Classify Behavior, Generate Time Budgets, and Evaluate Responses to Moonlight in Free-Ranging Snowshoe Hares. Front. Ecol. Evol. 2019, 7. [Google Scholar] [CrossRef]
- Barwick, J.; Lamb, D.W.; Dobos, R.; Welch, M.; Trotter, M. Categorising sheep activity using a tri-axial accelerometer. Comput. Electron. Agric. 2018, 145, 289–297. [Google Scholar] [CrossRef]
- Alvarenga, F.A.P.; Borges, I.; Palkovič, L.; Rodina, J.; Oddy, V.H.; Dobos, R.C. Using a three-axis accelerometer to identify and classify sheep behaviour at pasture. Appl. Anim. Behav. Sci. 2016, 181, 91–99. [Google Scholar] [CrossRef]
- Zobel, G.; Weary, D.M.; Leslie, K.; Chapinal, N.; von Keyserlingk, M.A.G. Technical note: Validation of data loggers for recording lying behavior in dairy goats. J. Dairy Sci. 2014, 98, 1082–1089. [Google Scholar] [CrossRef]
- Hammond, T.T.; Springthorpe, D.; Walsh, R.E.; Berg-Kirkpatrick, T. Using accelerometers to remotely and automatically characterize behavior in small animals. J. Exp. Biol. 2016, 219, 1618–1624. [Google Scholar] [CrossRef]
- Finney, G.; Gordon, A.; Scoley, G.; Morrison, S.J. Validating the IceRobotics IceQube tri-axial accelerometer for measuring daily lying duration in dairy calves. Livest. Sci. 2018, 214, 83–87. [Google Scholar] [CrossRef]
- Swartz, T.H.; McGilliard, M.L.; Petersson-Wolfe, C.S. Technical note: The use of an accelerometer for measuring step activity and lying behaviors in dairy calves. J. Dairy Sci. 2016, 99, 9109–9113. [Google Scholar] [CrossRef]
- Kok, A.; van Knegsel, A.T.M.; van Middelaar, C.E.; Hogeveen, H.; Kemp, B.; de Boer, I.J.M. Technical note: Validation of sensor-recorded lying bouts in lactating dairy cows using a 2-sensor approach. J. Dairy Sci. 2015, 98, 7911–7916. [Google Scholar] [CrossRef]
- Trénel, P.; Jensen, M.B.; Decker, E.L.; Skjøth, F. Technical note: Quantifying and characterizing behavior in dairy calves using the IceTag automatic recording device. J. Dairy Sci. 2009, 92, 3397–3401. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, P.P.; Fontana, I.; Sloth, K.H.; Guarino, M.; Blokhuis, H. Technical note: Validation and comparison of 2 commercially available activity loggers. J. Dairy Sci. 2018, 101, 5449–5453. [Google Scholar] [CrossRef] [PubMed]
- Vázquez Diosdado, J.A.; Barker, Z.E.; Hodges, H.R.; Amory, J.R.; Croft, D.P.; Bell, N.J.; Codling, E.A. Classification of behaviour in housed dairy cows using an accelerometer-based activity monitoring system. Anim. Biotelemetry 2015, 3, 15. [Google Scholar] [CrossRef]
- Alsaaod, M.; Kredel, R.; Hofer, B.; Steiner, A. Technical note: Validation of a semi-automated software tool to determine gait-cycle variables in dairy cows. J. Dairy Sci. 2017, 100, 4897–4902. [Google Scholar] [CrossRef] [PubMed]
- de Passillé, A.M.; Jensen, M.B.; Chapinal, N.; Rushen, J. Technical note: Use of accelerometers to describe gait patterns in dairy calves. J. Dairy Sci. 2010, 93, 3287–3293. [Google Scholar] [CrossRef] [PubMed]
- Kour, H.; Patison, K.P.; Corbet, N.J.; Swain, D.L. Validation of accelerometer use to measure suckling behaviour in Northern Australian beef calves. Appl. Anim. Behav. Sci. 2018, 202, 1–6. [Google Scholar] [CrossRef]
- Roland, L.; Schweinzer, V.; Kanz, P.; Sattlecker, G.; Kickinger, F.; Lidauer, L.; Berger, A.; Auer, W.; Mayer, J.; Sturm, V.; et al. Technical note: Evaluation of a triaxial accelerometer for monitoring selected behaviors in dairy calves. J. Dairy Sci. 2018, 101, 10421–10427. [Google Scholar] [CrossRef]
- Williams, L.R.; Moore, S.T.; Bishop-Hurley, G.J.; Swain, D.L. A sensor-based solution to monitor grazing cattle drinking behaviour and water intake. Comput. Electron. Agric. 2020, 168, 105141. [Google Scholar] [CrossRef]
- Luu, J.; Johnsen, J.F.; de Passillé, A.M.; Rushen, J. Which measures of acceleration best estimate the duration of locomotor play by dairy calves? Appl. Anim. Behav. Sci. 2013, 148, 21–27. [Google Scholar] [CrossRef]
- Größbacher, V.; Bučková, K.; Lawrence, A.B.; Špinka, M.; Winckler, C. Discriminating spontaneous locomotor play of dairy calves using accelerometers. J. Dairy Sci. 2020, 103, 1866–1873. [Google Scholar] [CrossRef]
- Poulopoulou, I.; Lambertz, C.; Gauly, M. Are automated sensors a reliable tool to estimate behavioural activities in grazing beef cattle? Appl. Anim. Behav. Sci. 2019, 216, 1–5. [Google Scholar] [CrossRef]
- Ledgerwood, D.N.; Winckler, C.; Tucker, C.B. Evaluation of data loggers, sampling intervals, and editing techniques for measuring the lying behavior of dairy cattle. J. Dairy Sci. 2010, 93, 5129–5139. [Google Scholar] [CrossRef] [PubMed]
- Bonk, S.; Burfeind, O.; Suthar, V.S.; Heuwieser, W. Technical note: Evaluation of data loggers for measuring lying behavior in dairy calves. J. Dairy Sci. 2013, 96, 3265–3271. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, M.A.; Worth, G.M.; Cameron, C.; Ross, C.M.; Rapp, D. Health, physiology, and behavior of dairy calves reared on 4 different substrates. J. Dairy Sci. 2017, 100, 2148–2156. [Google Scholar] [CrossRef]
- Rushen, J.; de Passillé, A.M. Automated measurement of acceleration can detect effects of age, dehorning and weaning on locomotor play of calves. Appl. Anim. Behav. Sci. 2012, 139, 169–174. [Google Scholar] [CrossRef]
- Held, S.D.E.; Špinka, M. Animal play and animal welfare. Anim. Behav. 2011, 81, 891–899. [Google Scholar] [CrossRef]
- Jensen, M.B.; Vestergaard, K.S.; Krohn, C.C. Play behaviour in dairy calves kept in pens: The effect of social contact and space allowance. Appl. Anim. Behav. Sci. 1998, 56, 97–108. [Google Scholar] [CrossRef]
- Boissy, A.; Manteuffel, G.; Jensen, M.B.; Moe, R.O.; Spruijt, B.; Keeling, L.J.; Winckler, C.; Forkman, B.; Dimitrov, I.; Langbein, J.; et al. Assessment of positive emotions in animals to improve their welfare. Physiol. Behav. 2007, 92, 375–397. [Google Scholar] [CrossRef]
- Brown, S.M.; Klaffenböck, M.; Nevison, I.M.; Lawrence, A.B. Evidence for litter differences in play behaviour in pre-weaned pigs. Appl. Anim. Behav. Sci. 2015, 172, 17–25. [Google Scholar] [CrossRef]
- Marcet Rius, M.; Cozzi, A.; Bienboire-Frosini, C.; Teruel, E.; Chabaud, C.; Monneret, P.; Leclercq, J.; Lafont-Lecuelle, C.; Pageat, P. Selection of putative indicators of positive emotions triggered by object and social play in mini-pigs. Appl. Anim. Behav. Sci. 2018, 202, 13–19. [Google Scholar] [CrossRef]
- Dohoo, I.; Martin, W.; Stryhn, H. Screening and diagnostic tests. In Veterinary Epidemiologic Research 2nd Edition; VER Inc.: Charlottetown, PE, Canada, 2009; pp. 91–134. [Google Scholar]
- Lehner, P.N. Sampling methods in behavior research. Poult. Sci. 1992, 71, 643–649. [Google Scholar] [CrossRef] [PubMed]
- Gladden, N.; Ellis, K.; Martin, J.; Viora, L.; McKeegan, D. A single dose of ketoprofen in the immediate postpartum period has the potential to improve dairy calf welfare in the first 48 h of life. Appl. Anim. Behav. Sci. 2019, 212, 19–29. [Google Scholar] [CrossRef]
- Gladden, N.; McKeegan, D.; Viora, L.; Ellis, K. Postpartum ketoprofen treatment does not alter stress biomarkers in cows and calves experiencing assisted and unassisted parturition: A randomised controlled trial. Vet. Rec. 2018, 183, 414. [Google Scholar] [CrossRef] [PubMed]
- IceRobotics. Available online: https://www.icerobotics.com/researchers/#research-software (accessed on 8 May 2019).
- Friard, O.; Gamba, M. BORIS: A free, versatile open-source event-logging software for video/audio coding and live observations. Methods Ecol. Evol. 2016, 7, 1325–1330. [Google Scholar] [CrossRef]
- Brodersen, K.H.; Ong, C.S.; Stephan, K.E.; Buhmann, J.M. The balanced accuracy and its posterior distribution. Proc. Int. Conf. Pattern Recognit. 2010, 3121–3124. [Google Scholar] [CrossRef]
- Breiman, L.; Friedman, J.H.; Olshen, R.A.; Stone, C.J. Classification and Regression Trees; Wadsworth International Group: Belmont, CA, USA, 1984. [Google Scholar]
- Loh, W.Y. Fifty years of classification and regression trees. Int. Stat. Rev. 2014, 82, 329–348. [Google Scholar] [CrossRef]
- Burman, P. A Comparative Study of Ordinary Cross-Validation, v-Fold Cross-Validation and the Repeated Learning-Testing Methods. Biometrika 1989, 76, 503–514. [Google Scholar] [CrossRef]
- James, G.; Witten, D.; Hastie, T.; Tibshirani, R. Cross-validation. In An Introduction to Statistical Learning with Applications in R; Springer: New York, NY, USA, 2013; pp. 176–186. [Google Scholar]
- Diamond, J.; Bond, A.B. Social Play in Kaka (Nestor meridionalis) with Comparisons to Kea (Nestor notabilis). Behaviour 2004, 141, 777–798. [Google Scholar]
- Chua, B.; Coenen, E.; van Delen, J.; Weary, D.M. Effects of Pair Versus Individual Housing on the Behavior and Performance of Dairy Calves. J. Dairy Sci. 2002, 85, 360–364. [Google Scholar] [CrossRef]
- Hill, T.M.; Bateman, H.G.; Aldrich, J.M.; Quigley, J.D.; Schlotterbeck, R.L. Short communication: Intensive measurements of standing time of dairy calves housed in individual pens within a naturally ventilated, unheated nursery over different periods of the year. J. Dairy Sci. 2013, 96, 1811–1814. [Google Scholar] [CrossRef]
- De’Ath, G.; Fabricius, K.E. Classification and regression trees: A powerful yet simple technique for ecological data analysis. Ecology 2000, 81, 3178–3192. [Google Scholar] [CrossRef]
- Sutherland, M.; Worth, G.; Cameron, C.; Verbeek, E. Effect of morphine administration on social and non-social play behaviour in calves. Animals 2019, 9, 56. [Google Scholar] [CrossRef] [PubMed]
| Behaviour | Behavioural Description | Key | Category |
|---|---|---|---|
| Lying | The calf is in a lying position. This includes both sternal and lateral recumbency. The head may be either elevated in an alert position or rested on the ground or any part of the body. | L | Resting |
| Standing | The calf is in an upright standing position, all legs are extended beneath the body, and all four feet are on the ground. The calf may be still or concurrently engaged in other active behaviours. | S | Active |
| Posture change | The calf is moving from standing to lying or lying to standing. Both hindlimbs are extended with feet on the ground, and one or both forelimbs are flexed at the carpus with the antebrachium in contact with the ground. | PC | |
| Step | Step activity associated with movement of the right rear leg. | ST | |
| Feeding | The calf is drinking milk. | F | |
| Head shake | The head is shaken, rotated or tossed (HS). Further defined by recording of concurrent forelimb (HFFM) or hindlimb (HRFM) movement if observed. | HS/HFFM/HRFM | Play |
| Hop | Upward movement of either the two forelimbs (HPF) or the two hindlimbs (HPR) in a vertical direction simultaneously. | HPF/HPR | |
| Leap forward | Both forelimbs are simultaneously lifted from the ground and stretched forward, causing the forequarters of the body to be lifted and the calf to move in a forward direction. | LF | |
| Leap sideward | All four limbs are elevated off the ground, and the calf moves in a lateral direction. All four feet land on the ground simultaneously. | LS | |
| Turn | Both forelimbs are lifted from the ground and stretched forward and laterally. The forequarters of the body are lifted, and the calf turns to one side. The direction of movement is upward, lateral and forward. | T | |
| Reverse | The calf moves in a backwards direction. | RV | |
| Running | Gait that is faster than a walk and contains a brief period of suspension. | R | |
| Buck low | Both hindfeet are simultaneously elevated to a level below the tarsus whilst both forelimbs remain in contact with the ground. The body lifts from front to back, and the head is lowered. | BL | |
| Buck high | Both hindfeet are simultaneously elevated to a level above the tarsus whilst both forelimbs remain in contact with the ground. The body lifts from front to back, and the head is lowered. | BH | |
| Buck kick | Both hindfeet are simultaneously elevated to a level equal to, or above, the tarsus, and one or both hindlimbs are kicked away from the body in a caudal or lateral direction. The body lifts from front to back, and the head is lowered. | BK | |
| Kick | One rear leg is kicked away from the body in a caudal or lateral direction. The other three limbs remain in contact with the ground. | K | |
| Management practice | Management practices performed by the farm staff that influence the calf’s behaviour and movement. | M | N/A |
| Out of View | The calf cannot be observed on the video footage. | O |
| Behaviour | Behavioural Description | Key | Behavioural Category |
|---|---|---|---|
| Play | Calf engages in locomotor play—defined as running (moving at a faster pace than walking with a period of elevation), jumping (all four limbs are lifted away from the ground at the same time—the calf may remain in the same position in space or may move forwards, backwards or laterally during the jump), bucking (both hindlimbs are elevated at the same time and kicked away from the body, either caudally or laterally—the calf may remain in the same plane of motion or may twist the body during the buck), kicking (one or both hindlimbs is elevated to a height above the tarsus and kicked out caudally or laterally from the body—the calf may be in motion or may remain still), hopping (the calf lifts both forelimbs away from the ground at the same time—the calf may remain in the same position in space, or the forequarters may move forwards, backwards or laterally during the hop), and spinning (calf lifts both hindlimbs at the same time and moves both around the central axis in either a clockwise or anticlockwise direction; the front of the body turns but remains in the same place in space, and both forelimbs remain on the ground). | p | Play |
| Lying | The calf is lying on the ground in any position. The whole body is in contact with the ground. The head may or may not be in contact with the ground. | l | Lying |
| Posture change | The calf is transitioning either from a lying to standing position or from a standing to lying position. The forelimbs are bent, and the antebrachium is in contact with the ground. The hindlimbs are straightening, and only the feet are in contact with the ground. The chest/sternum may be in contact with the ground. The abdomen and/or hindquarters do not contact the ground. The head may or may not be in contact with the ground. | s | Active (excl. play) |
| Standing | The calf is standing still with all four feet on the ground and all four limbs straight. No other part of the body is in contact with the ground. | t | Active (excl. play) |
| Walking | The calf takes two or more steps in a forward or backward direction. Three limbs are in contact with the ground at any one time and no part of the body is in contact with the ground. | w | Active (excl. play) |
| Sidestepping | The calf steps or stumbles one or both hind limbs laterally without moving the front limbs, or the calf moves the hindlimbs individually around the forelimb axis without moving its position in space | f | Active (excl. play) |
| Behaviour | Observation Period | ||||
|---|---|---|---|---|---|
| 0 to 12 h | 12 to 24 h | 24 to 36 h | 36 to 48 h | Combined 48 h | |
| Lying (no. bouts) | 0.277 | 0.317 | 0.782 | 0.448 | 0.607 |
| 0.384 | 0.342 | 0.008 | 0.226 | 0.048 | |
| Lying (duration (s)) | −0.035 | 0.027 | −0.031 | −0.309 | 0.264 |
| 0.914 | 0.937 | 0.931 | 0.418 | 0.433 | |
| Play (no. bouts) | 0.562 | 0.811 | 0.926 | 0.871 | 0.922 |
| 0.057 | 0.002 | <0.001 | 0.002 | <0.001 | |
| Play (duration (s)) | 0.388 | 0.829 | 0.918 | 0.937 | 0.773 |
| 0.213 | 0.002 | <0.001 | <0.001 | 0.005 | |
| Active excl. play (no. bouts) | 0.250 | 0.175 | 0.780 | 0.215 | 0.552 |
| 0.433 | 0.607 | 0.008 | 0.579 | 0.078 | |
| Active excl. play (duration (s)) | 0.124 | 0.273 | 0.879 | 0.726 | 0.386 |
| 0.701 | 0.417 | 0.001 | 0.027 | 0.241 | |
| Active incl. play (no. bouts) | 0.481 | 0.726 | 0.995 | 0.835 | 0.552 |
| 0.114 | 0.011 | <0.001 | 0.005 | 0.078 | |
| Active incl. play (duration (s)) | 0.144 | 0.273 | 0.887 | 0.749 | 0.386 |
| 0.656 | 0.417 | 0.001 | 0.020 | 0.241 | |
| Motion Index * (15 min) | Sensitivity | Specificity | Balanced Accuracy |
|---|---|---|---|
| 300 | 19.6% | 100% | 59.8% |
| 200 | 29.4% | 99.7% | 64.6% |
| 100 | 62.8% | 98.6% | 80.7% |
| 50 | 84.3% | 95.9% | 90.1% |
| 45 | 90.2% | 95.4% | 92.8% |
| 40 | 90.2% | 92.2% | 91.2% |
| 35 | 94.1% | 92.2% | 93.2% |
| 30 | 96.1% | 91.0% | 93.6% |
| 28 | 96.1% | 90.4% | 93.3% |
| 27 | 96.1% | 90.1% | 93.1% |
| 26 | 96.1% | 90.1% | 93.1% |
| 25 | 98.0% | 89.9% | 93.6% |
| 20 | 98.0% | 87.8% | 92.9% |
| Motion Index * (1 min) | Sensitivity | Specificity | Balanced Accuracy |
|---|---|---|---|
| 60 | 21.7% | 99.8% | 60.8% |
| 50 | 30.3% | 99.7% | 65.0% |
| 40 | 38.8% | 99.5% | 69.2% |
| 30 | 54.0% | 99.2% | 76.6% |
| 20 | 69.1% | 98.5% | 83.8% |
| 10 | 90.1% | 96.3% | 93.2% |
| 5 | 96.1% | 94.3% | 95.2% |
| 3 | 98.0% | 92.9% | 95.5% |
| Calculation | Sample Interval Duration | Analysis | Sampling Method | Number of Positive Sample Intervals 1 | Total Number of Sample Points | One-Zero Score 2 |
|---|---|---|---|---|---|---|
| Initial calculation | 1 min | CART | Visual observations | 152 | 5938 | 0.026 |
| MI ≥ 2.5 | 560 | 5938 | 0.094 | |||
| Manual calculation | Visual observations | 74 | 3501 | 0.021 | ||
| MI ≥ 23 | 76 | 3501 | 0.022 | |||
| 15 min | CART | Visual observations | 51 | 396 | 0.129 | |
| MI ≥ 24.5 | 85 | 396 | 0.215 | |||
| Manual calculation | Visual observations | 27 | 234 | 0.115 | ||
| MI ≥ 62 | 27 | 234 | 0.115 | |||
| Results when applied to Dataset 2 * | 1 min | Manual calculation | Visual observations | 78 | 2437 | 0.032 |
| MI ≥ 23 | 95 | 2437 | 0.039 | |||
| 15 min | Manual calculation | Visual observations | 24 | 162 | 0.148 | |
| MI ≥ 62 | 23 | 162 | 0.142 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gladden, N.; Cuthbert, E.; Ellis, K.; McKeegan, D. Use of a Tri-Axial Accelerometer Can Reliably Detect Play Behaviour in Newborn Calves. Animals 2020, 10, 1137. https://doi.org/10.3390/ani10071137
Gladden N, Cuthbert E, Ellis K, McKeegan D. Use of a Tri-Axial Accelerometer Can Reliably Detect Play Behaviour in Newborn Calves. Animals. 2020; 10(7):1137. https://doi.org/10.3390/ani10071137
Chicago/Turabian StyleGladden, Nicola, Erin Cuthbert, Kathryn Ellis, and Dorothy McKeegan. 2020. "Use of a Tri-Axial Accelerometer Can Reliably Detect Play Behaviour in Newborn Calves" Animals 10, no. 7: 1137. https://doi.org/10.3390/ani10071137
APA StyleGladden, N., Cuthbert, E., Ellis, K., & McKeegan, D. (2020). Use of a Tri-Axial Accelerometer Can Reliably Detect Play Behaviour in Newborn Calves. Animals, 10(7), 1137. https://doi.org/10.3390/ani10071137





