Behavioral Methods for Severity Assessment
Simple Summary
Abstract
1. Introduction
2. Including the Animal’s Behavior
3. Preference Tests
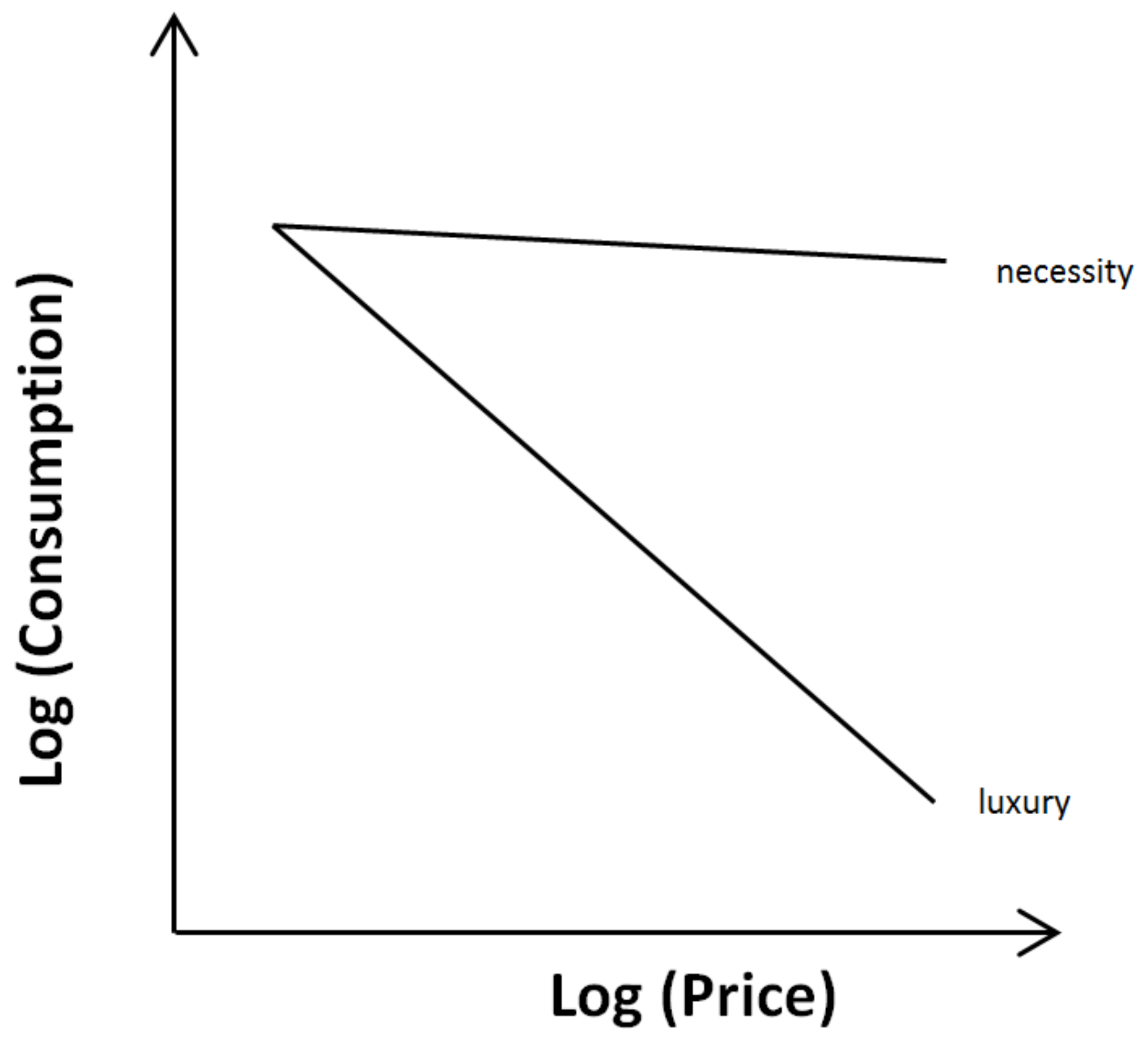
4. Consumer Demand
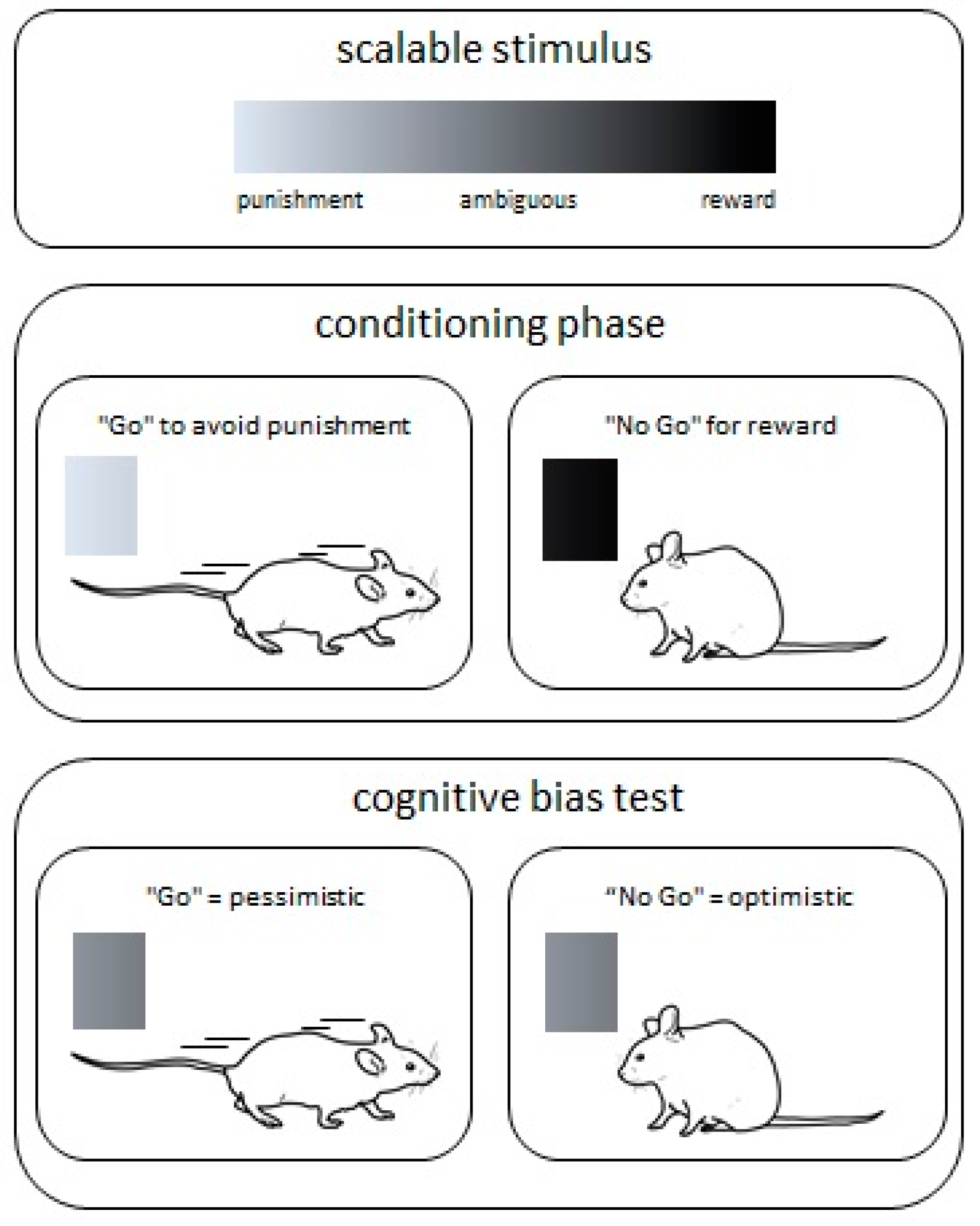
5. Cognitive Bias Test
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- European Commission. Report on the Statistics on the Use of Animals for Scientific Purposes in the Member States of the European Union in 2015–2017. Available online: https://ec.europa.eu/transparency/regdoc/rep/1/2020/EN/COM-2020-16-F1-EN-MAIN-PART-1.PDF (accessed on 27 March 2020).
- Russell, W.M.S.; Burch, R.L. The Principles of Humane Experimental Technique; Methuen: London, UK, 1959. [Google Scholar]
- Lewejohann, L.; Schwabe, K.; Häger, C.; Jirkof, P. Impulse for animal welfare outside the experiment. Lab. Anim. 2020, 54, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Fraser, D.; Weary, D.M.; Pajor, E.A.; Milligan, B.N. A scientific conception of animal welfare that reflects ethical concerns. Anim. Welf. 1997, 6, 187–205. [Google Scholar]
- Broom, D.M. The use of the concept animal welfare in European conventions, regulations and directives. Food Chain 2001, 2001, 148–151. [Google Scholar]
- Appleby, M.C.; Sandøe, P. Philosophical debate on the nature of well-being: Implications for animal welfare. Anim. Welf. 2002, 11, 283–294. [Google Scholar]
- Broom, D.M. Behaviour and welfare in relation to pathology. Appl. Anim. Behav. Sci. 2006, 97, 71–83. [Google Scholar] [CrossRef]
- Broom, D.M. Animal welfare: Concepts and measurement. J. Anim Sci. 1991, 69, 4167–4175. [Google Scholar] [CrossRef]
- Broom, D.M. Animal welfare defined in terms of attempts to cope with the environment. Acta Agric. Scand. Sec. A Anim. Sci. Suppl. 1996, 27, 22–28. [Google Scholar]
- Jirkof, P.; Rudeck, J.; Lewejohann, L. Assessing affective state in laboratory rodents to promote animal welfare—What is the progress in applied refinement research? Animals 2019, 9, 1026. [Google Scholar] [CrossRef]
- European Parliament. EU Directive 2010/63/EU of the European Parliament and of the Council of 22 September 2010 on the Protection of Animals Used for Scientific Purposes. Available online: https://www.bfr.bund.de/cm/343/5_Beratung_Anlage%203_2010-63-EU.pdf (accessed on 27 March 2020).
- Bugnon, P.; Heimann, M.; Thallmair, M. What the literature tells us about score sheet design. Lab. Anim. 2016, 50, 414–417. [Google Scholar] [CrossRef]
- Ullmann, K.; Jourdan, T.; Kock, M.; Unger, J.; Schulz, A.; Thöne-Reineke, C.; Abramjuk, C. Recommendations for the development and use of Score Sheets as a tool for applied refinement. Berl. Münch. Tierärztl. Wochenschr. 2018. [Google Scholar] [CrossRef]
- Van de Meer, M.; Rolls, A.; Baumans, V.; Olivier, B.; van Zutphen, L.F.M. Use of score sheets for welfare assessment of transgenic mice. Lab. Anim. 2001, 35, 379–389. [Google Scholar] [CrossRef]
- Lang, A.; Schulz, A.; Ellinghaus, A.; Schmidt-Bleek, K. Osteotomy models—The current status on pain scoring and management in small rodents. Lab. Anim. 2016, 50, 433–441. [Google Scholar] [CrossRef]
- Rix, A.; Drude, N.; Mrugalla, A.; Mottaghy, F.M.; Tolba, R.H.; Kiessling, F. Performance of severity parameters to detect chemotherapy-induced pain and distress in mice. Lab. Anim. 2019, 0, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Rybkin, I.I.; Zhou, Y.; Volaufova, J.; Smagin, G.N.; Ryan, D.H.; Harris, R.B.S. Effect of restraint stress on food intake and body weight is determined by time of day. Am. J. Physiol. 1997, 273, R1612–R1622. [Google Scholar] [CrossRef] [PubMed]
- Harris, R.B.S.; Zhou, J.; Youngblood, B.D.; Rybkin, I.I.; Smagin, G.N.; Ryan, D.H. Effect of repeated stress on body weight and body composition of rats fed low- and high-fat diets. Am. J. Physiol. 1998, 275, R1928–R1938. [Google Scholar] [CrossRef] [PubMed]
- Häger, C.; Keuler, L.M.; Talbot, S.R.; Biernot, S.; Weegh, N.; Buchheister, S.; Buettner, M.; Glage, S.; Bleich, A. Running in the wheel: Defining individual severity levels in mice. PLoS Biol. 2018, 16, e2006159. [Google Scholar] [CrossRef] [PubMed]
- Ullman-Cullere, M.H.; Foltz, C.J. Body Condition Scoring: A rapid and accurate method for assessing health status in mice. Lab. Anim. Sci. 1999, 49, 319–323. [Google Scholar] [PubMed]
- Dallman, M.F.; Akana, S.F.; Scribner, K.A.; Bradbury, M.J.; Walker, C.D.; Strack, A.M.; Cascio, C.S. Stress, feedback and facilitation in the hypothalamo-pituitary-adrenal axis. J. Neuroendocrinol. 1992, 4, 517–526. [Google Scholar] [CrossRef] [PubMed]
- Tsai, P.; Schlichtig, A.; Ziegler, E.; Ernst, H.; Haberstroh, J.; Stelzer, H.D.; Hackbarth, H. Effects of different blood collection methods on indicators of welfare in mice. Lab. Anim. 2015, 44, 301–310. [Google Scholar] [CrossRef]
- Touma, C.; Palme, R.; Sachser, N. Analysing corticosterone metabolites in fecal samples of mice: A noninvasive technique to monitor stress hormones. Horm. Behav. 2004, 45, 10–22. [Google Scholar] [CrossRef]
- Hohlbaum, K.; Bert, B.; Dietze, S.; Palme, R.; Fink, H.; Thöne-Reineke, C. Severity classification of repeated isoflurane anesthesia in C57BL/6JRj mice—Assessing the degree of distress. PLoS ONE 2017, 12, e0179588. [Google Scholar] [CrossRef] [PubMed]
- Leenaars, C.H.C.; van der Mierden, S.; Durst, M.; Goerlich-Jansson, V.C.; Ripoli, F.L.; Keubler, L.M.; Talbot, S.R.; Boyle, E.; Habedank, A.; Jirkof, P.; et al. Measurement of corticosterone in mice: A protocol for a mapping review. Lab. Anim. 2020, 54, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Dawkins, M.S. From an animal’s point of view: Motivation, fitness, and animal welfare. Behav. Brain Sci. 2009, 13, 1–9. [Google Scholar] [CrossRef]
- Lister, R.G. The use of a plus-maze to measure anxiety in the mouse. Psychopharmacology 1987, 92, 180–185. [Google Scholar] [CrossRef] [PubMed]
- Weigle, D.S.; Bukowski, T.R.; Foster, D.C.; Holderman, S.; Kramer, J.M.; Lasser, G.; Lofton-Day, C.E.; Prunkard, D.E.; Raymond, C.; Kuijper, J.L. Recombination of protein reduces feeding and body weight in the ob/ob mouse. J. Clin. Investig. 1995, 96, 2065–2070. [Google Scholar] [CrossRef] [PubMed]
- Baumans, V.; Bouwknecht, J.A.; Boere, H.; Kramer, K.; van Lith, H.A.; van de Weerd, H.A.; van Herck, H. Intra-abdominal transmitter implantation in mice: Effects on behaviour and body weight. Anim. Welf. 2001, 10, 291–302. [Google Scholar]
- Miller, A.L.; Miller, A.L.; Wright-Williams, S.L.; Flecknell, P.A.; Roughan, J.V. A comparison of abdominal and scrotal approach methods of vasectomy and the influence of analgesic treatments in laboratory mice. Lab. Anim. 2012, 46, 304–310. [Google Scholar] [CrossRef]
- Sliepen, S.H.J.; Diaz-Delcastillo, M.; Korioth, J.; Olsen, R.B.; Appel, A.K.; Christoph, T.; Heegaard, A.; Rutten, K. Cancer-induced bone pain impairs burrowing behaviour in mice. In Vivo 2019, 33, 1125–1132. [Google Scholar] [CrossRef]
- Gjendal, K.; Ottesen, J.L.; Olsson, A.S.; Sørensen, D.B. Burrowing and nest building activity in mice after exposure to grid floor, isoflurane or ip injetions. Physiol. Behav. 2019, 206, 59–66. [Google Scholar] [CrossRef]
- Roughan, J.V.; Flecknell, P.A. Evaluation of short duration behaviour-based post-operative pain scoring system in rats. Eur. J. Pain 2012, 7, 397–406. [Google Scholar] [CrossRef]
- Leach, M.; Klaus, K.; Miller, A.; Scotto di Perrotolo, M. The assessment of post-vasectomy pain in mice using behaviour and the mouse grimace scale. PLoS ONE 2012, 7, e35656. [Google Scholar] [CrossRef]
- Roughan, J.V.; Wright-Williams, S.L.; Flecknell, P.A. Automated analysis of postoperative behaviour: Assessment of HomeCageScan as a novel method to rapidly identify pain and analgesic effects in mice. Lab. Anim. 2009, 43, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Langford, D.J.; Bailey, A.L.; Chanda, M.L.; Clarke, S.E.; Drummond, T.E.; Echols, S.; Glick, S.; Ingrao, J.; Klassen-Ross, T.; La Croix-Fralish, M.; et al. Coding of facial expressions of pain in the laboratory mouse. Nat. Methods 2010, 7, 447–449. [Google Scholar] [CrossRef]
- Hohlbaum, K.; Bert, B.; Dietze, S.; Palme, R.; Fink, H.; Thöne-Reineke, C. Impact of repeated anesthesia with ketamine and xylazine on well-being of C57BL/6JRj mice. PLoS ONE 2018, 13, e0203559. [Google Scholar] [CrossRef] [PubMed]
- Andresen, N.; Wöllhaf, M.; Hohlbaum, K.; Lewejohann, L.; Hellwich, O.; Thöne-Reineke, C.; Belik, V. Toward a fully automated surveillance of well-being status in laboratory mice using deep learning. PLoS ONE 2020, 15, e0228059. [Google Scholar] [CrossRef] [PubMed]
- Ernst, L.; Kopaczka, M.; Schulz, M.; Talbot, S.R.; Zieglowski, L.; Meyer, M.; Bruch, S.; Merhof, D.; Tolba, R.H. Improvement of the mouse grimace scale set-up for implementing a semi-automated mouse grimace scale scoring (Part 1). Lab. Anim. 2020, 54, 83–91. [Google Scholar] [CrossRef]
- Ernst, L.; Kopaczka, M.; Schulz, M.; Talbot, S.R.; Struve, B.; Häger, C.; Bleich, A.; Durst, M.; Jirkof, P.; Arras, M.; et al. Semi-automated generation of pictures for the mouse grimace scale: A multi-laboratory analysis (Part 2). Lab. Anim. 2020, 54, 92–98. [Google Scholar] [CrossRef]
- Garner, J.P. Stereotypies and other abnormal repetitive behaviors: Potential impact on validity, reliability, and replicability of scientific outcomes. ILAR J. 2005, 46, 106–117. [Google Scholar] [CrossRef]
- Mason, G.J. Stereotypies and suffering. Behav. Process. 1991, 25, 103–115. [Google Scholar] [CrossRef]
- Powell, S.B.; Newman, H.A.; Pendergast, J.F.; Lewis, M.H. A rodent model of spontaneous stereotypy: Initial characterization of developmental, environmental, and neurobiological factors. Physiol. Behav. 1998, 66, 355–363. [Google Scholar] [CrossRef]
- Mason, G.J. Stereotypies: A critical review. Anim. Behav. 1991, 41, 1015–1037. [Google Scholar] [CrossRef]
- Habedank, A.; Kahnau, P.; Diederich, K.; Lewejohann, L. Severity assessment from an animal’s point of view. Berl. Münch. Tierärztl. Wochenschr. 2018. [Google Scholar] [CrossRef]
- Dawkins, M.S. Do hens suffer in battery cages? Environmental preferences and welfare. Anim. Behav. 1977, 25, 1034–1046. [Google Scholar] [CrossRef]
- Bloom, H.J.M.; Vanvorstenbosch, C.J.A.H.V.; Baumans, V.; Hoogervorst, M.J.C.; Beynen, A.C.; Vanzutphen, L.F.M. Description and validation of a preference test system to evaluate housing conditions for laboratory mice. Appl. Anim. Behav. Sci. 1992, 35, 67–82. [Google Scholar] [CrossRef]
- Dawkins, M.S. Battery hens name their price: Consumer demand theory and the measurement of ethological ‘needs’. Anim. Behav. 1983, 312, 1195–1205. [Google Scholar] [CrossRef]
- Freymann, J.; Tsai, P.; Stelzer, H.; Hackbarth, H. The impact of bedding volumes on laboratory mice. Appl. Anim. Behav. Sci. 2016, 186, 72–79. [Google Scholar] [CrossRef]
- Lewejohann, L.; Sachser, N. Evaluation of different housing conditions for male laboratory mice by means of preference tests. KTBL Schrift. 2000, 391, 170–177. [Google Scholar]
- Banjanin, S.; Mrosovsky, N. Preferences of mice, Mus musculus, for different types of running wheel. Lab. Anim. UK 2000, 34, 313–318. [Google Scholar] [CrossRef]
- Tzschentke, T.M. Measuring reward with the conditioned place preference paradigm: A comprehensive review of drug effects, recent progress and new issues. Prog. Neurobiol. 1998, 56, 613–672. [Google Scholar] [CrossRef]
- Schechter, M.D.; Calcagnetti, D.J. Trends in place preference conditioning with a cross-indexed bibliography; 1957–1991. Neurosci. Biobehav. Rev. 1993, 17, 21–41. [Google Scholar] [CrossRef]
- Cunningham, C.L.; Gremel, C.M.; Groblewski, P.A. Drug-induced conditioned place preference and aversion in mice. Nat. Protoc. 2006, 1, 1662–1670. [Google Scholar] [CrossRef] [PubMed]
- King, T.; Vera-Portocarrero, L.; Gutierrez, T.; Vanderah, T.W.; Dussor, G.; Lai, J.; Fields, H.L.; Porreca, F. Unmasking the tonic-aversive state in neuropathic pain. Nat. Neurosci. 2009, 12, 1364–1366. [Google Scholar] [CrossRef] [PubMed]
- Roughan, J.V.; Coulter, C.A.; Flecknell, P.A.; Thomas, H.D.; Sufka, K.J. The conditioned place preference test for assessing welfare consequences and potential refinements in a mouse bladder cancer model. PLoS ONE 2014, 9, e103362. [Google Scholar] [CrossRef] [PubMed]
- Millot, S.; Cerqueira, M.; Casatnheira, M.F.; Øverli, Ø.; Martins, C.I.M.; Oliveira, R.F. Use of conditioned place preference/avoidance tests to assess affective states in fish. Appl. Anim. Behav. Sci. 2014, 154, 104–111. [Google Scholar] [CrossRef]
- Panksepp, J.B.; Lahvis, G.P. Social reward among juvenile mice. Genes Bain Behav. 2007, 6, 661–671. [Google Scholar] [CrossRef]
- Dixon, L.M.; Sandilands, V.; Bateson, M.; Brocklehurst, S.; Tolkamp, B.J.; D’Eath, R.B. Conditioned place preference or aversion as animal welfare assessment tools: Limitations in their application. Appl. Anim. Behav. Sci. 2013, 148, 164–176. [Google Scholar] [CrossRef]
- Dawkins, M.S.; Beardsley, T. Reinforcing properties of access to litter in hens. Appl. Anim. Behav. Sci. 1986, 15, 351–365. [Google Scholar] [CrossRef]
- Sherwin, C.M. The motivation of group-housed laboratory mice, Mus musculus, for additional space. Anim. Behav. 2004, 67, 711–717. [Google Scholar] [CrossRef]
- Sherwin, C.M.; Nicol, C.J. Changes in meal patterning by mice measure the cost imposed by natural obstacles. Appl. Anim. Behav. Sci. 1995, 43, 291–300. [Google Scholar] [CrossRef]
- Manser, C.E.; Elliot, H.; Morris, T.H.; Broom, D.M. The use of a novel operant test to determine the strength of preferences for flooring in laboratory rats. Lab. Anim. 1996, 30, 1–6. [Google Scholar] [CrossRef]
- Dawkins, M.S. Behavioural Deprivation: A central problem in animal welfare. Appl. Anim. Behav. Sci. 1988, 20, 209–225. [Google Scholar] [CrossRef]
- Sherwin, C.M. The use and perceived importance of three resources which provide cage laboratory mice the opportunity for extended locomotion. Appl. Anim. Behav. Sci. 1998, 55, 353–367. [Google Scholar] [CrossRef]
- Mendl, M.; Burman, O.H.P.; Parker, R.M.A.; Paul, E.S. Cognitive bias as an indicator of animal emotion and welfare: Emerging evidence and underlying mechanisms. Appl. Anim. Behav. Sci. 2009, 118, 161–181. [Google Scholar] [CrossRef]
- Herrmann, E.; Call, J. Are there geniuses among the apes? Philos. Trans. R. Soc. Lond. B Biol. Sci. 2012, 367, 2753–2761. [Google Scholar] [CrossRef] [PubMed]
- Harding, E.J.; Paul, E.S.; Mendl, M. Cognitive bias and affective state. Nature 2004, 427, 2006159. [Google Scholar] [CrossRef] [PubMed]
- Richter, S.H.; Schick, A.; Hoyer, C.; Lankisch, K.; Gass, P.; Vollmayr, B. A glass full of optimism: Enrichment effects on cognitive bias in a rat model of depression. Cogn. Affect. Behav. Neurosci. 2012, 12, 527–542. [Google Scholar] [CrossRef]
- Boleij, H.; Klooster, J.; Lavrijsen, M.; Kirchhoff, S.; Arndt, S.S.; Ohl, F. A test to identify judgement bias in mice. Behav. Brain Res. 2012, 233, 45–54. [Google Scholar] [CrossRef][Green Version]
- Hintze, S.; Melotti, L.; Colosio, S.; Bailoo, J.D.; Boada-Sana, M.; Würbel, H.; Murphy, E. A cross-species judgement bias task: Integrating active trial initiation into a spatial go/no-go task. Sci. Rep. 2018, 8, 5104. [Google Scholar] [CrossRef]
- Verbeek, E.; Ferguson, D.; Lee, C. Are hungry sheep more pessimistic? The effects of food restriction on cognitive bias and the involvement of ghrelin in its regulation. Physiol. Behav. 2014, 123, 67–75. [Google Scholar] [CrossRef]
- Schlüns, H.; Welling, H.; Federici, J.R.; Lewejohann, L. The glass is not yet half empty: Agitation but not verroa treatment causes cognitive bias in honey bees. Anim. Cogn. 2017, 20, 233–241. [Google Scholar] [CrossRef]
- Jones, S.; Paul, E.S.; Dayan, P.; Robinson, E.S.J.; Mendl, M. Pavlovian influences on learning differ between rats and mice in a counter-balanced Go/NoGo judgement bias task. Behav. Brain Res. 2017, 331, 214–224. [Google Scholar] [CrossRef]
- Matheson, S.M.; Asher, L.; Bateson, M. Larger, enriched cages are associated with ’optimistic’ response biases in captive European starlings (Sturnus vulgaris). Appl. Anim. Behav. Sci. 2008, 109, 374–383. [Google Scholar] [CrossRef]
- Enkel, T.; Gholizadeh, D.; von Bohlen und Halbach, O.; Sanchis-Segura, C.; Hurlemann, R.; Spanagel, R.; Gass, P.; Vollmayr, B. Ambiguous-Cue interpretation is biased under stress and depression like states in rats. Neuropsychopharmacology 2010, 35, 1008–1015. [Google Scholar] [CrossRef]
- Gygax, L. The A to Z of statistics for testing cognitive judgement bias. Anim. Behav. 2014, 95, 59–69. [Google Scholar] [CrossRef]
- Bethell, E.J. A “How-To” guide for designing judgment bias studies to assess captive animal welfare. Appl. Anim. Welf. Sci. 2015, 1, 18–42. [Google Scholar] [CrossRef] [PubMed]
- Roelofs, S.; Boleij, H.; Nordquist, R.E.; van der Staay, F.J. Making decisions under ambiguity: Judgment bias tasks for assessing emotional state in animals. Front. Behav. Neurosci. 2016, 10, 119. [Google Scholar] [CrossRef]
- Brydges, N.M.; Leach, M.; Nicol, K.; Wright, R.; Bateson, M. Environmental enrichment induces optimistic cognitive bias in rats. Anim. Behav. 2011, 81, 169–175. [Google Scholar] [CrossRef]
- Kloke, V.; Schreiber, R.S.R.S.; Bodden, C.; Möllers, J.; Ruhmann, H.; Kaiser, S.; Lesch, K.P.K.; Sachser, N.; Lewejohann, L. Hope for the best or prepare for the worst? Towards a spatial cognitive bias test for mice. PLoS ONE 2014, 9, e105431. [Google Scholar] [CrossRef]
- Krakenberg, V.; Woigk, I.; Garcia Rodriguez, L.; Kästner, N.; Kaiser, S.; Sachser, N.; Richter, S.H. Technology or ecology? New tools to assess cognitive judgement bias in mice. Behav. Brain Res. 2019, 362, 279–287. [Google Scholar] [CrossRef]
- Doyle, R.E.; Vidal, S.; Hinch, G.N.; Fischer, A.D.; Boissy, A.; Lee, C. The effect of repeated testing on judgement biases in sheep. Behav. Process. 2010, 83, 349–352. [Google Scholar] [CrossRef]
- Jones, S.; Neville, V.; Higgs, L.; Paul, E.S.; Dayan, P.; Robinson, E.S.J.; Mendl, M. Assessing animal affect: An automated and self-initiated judgement bias task based on natural investigative behaviour. Sci. Rep. 2018, 812400. [Google Scholar] [CrossRef]
- Lewejohann, L.; Grune, B.; Schönfelder, G.; Bert, B. Cut back on surplus laboratory animals [All labortatory animals count]. Nature 2020, 578, 515. [Google Scholar] [CrossRef] [PubMed]
- Sandøe, P.; Simonsen, H.B. Assessing animal welfare: Where does science end and philosophy begin? Anim. Welf. 1992, 1, 257–267. [Google Scholar]
- Pool, T. Happy animals make good science. Lab. Anim. 1997, 31, 116–124. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kahnau, P.; Habedank, A.; Diederich, K.; Lewejohann, L. Behavioral Methods for Severity Assessment. Animals 2020, 10, 1136. https://doi.org/10.3390/ani10071136
Kahnau P, Habedank A, Diederich K, Lewejohann L. Behavioral Methods for Severity Assessment. Animals. 2020; 10(7):1136. https://doi.org/10.3390/ani10071136
Chicago/Turabian StyleKahnau, Pia, Anne Habedank, Kai Diederich, and Lars Lewejohann. 2020. "Behavioral Methods for Severity Assessment" Animals 10, no. 7: 1136. https://doi.org/10.3390/ani10071136
APA StyleKahnau, P., Habedank, A., Diederich, K., & Lewejohann, L. (2020). Behavioral Methods for Severity Assessment. Animals, 10(7), 1136. https://doi.org/10.3390/ani10071136





