Human Co-Infections between Borrelia burgdorferi s.l. and Other Ixodes-Borne Microorganisms: A Systematic Review
Abstract
1. Introduction
2. Results
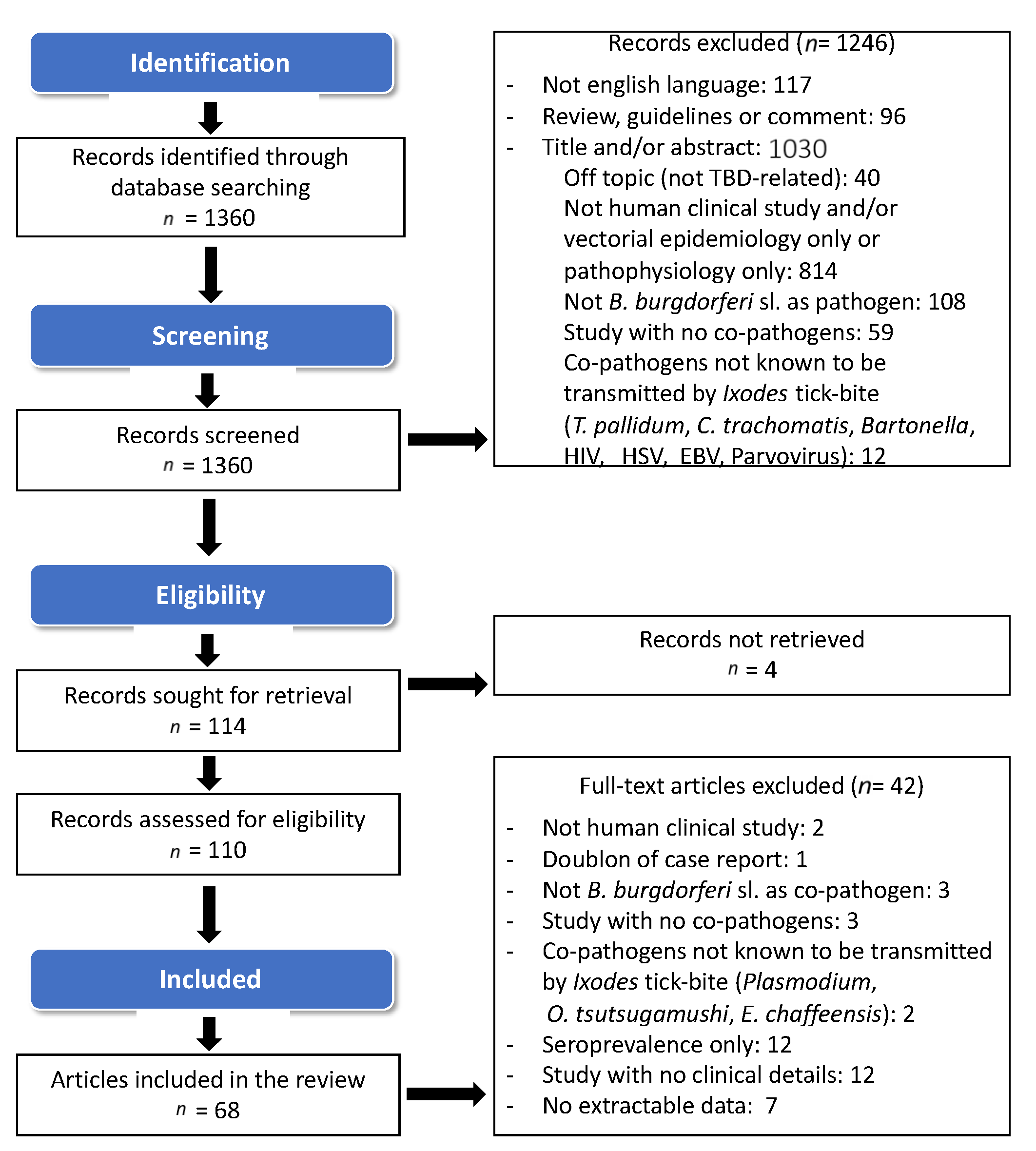
2.1. Study Selection and Search Results
2.2. Frequency of Co-Infections
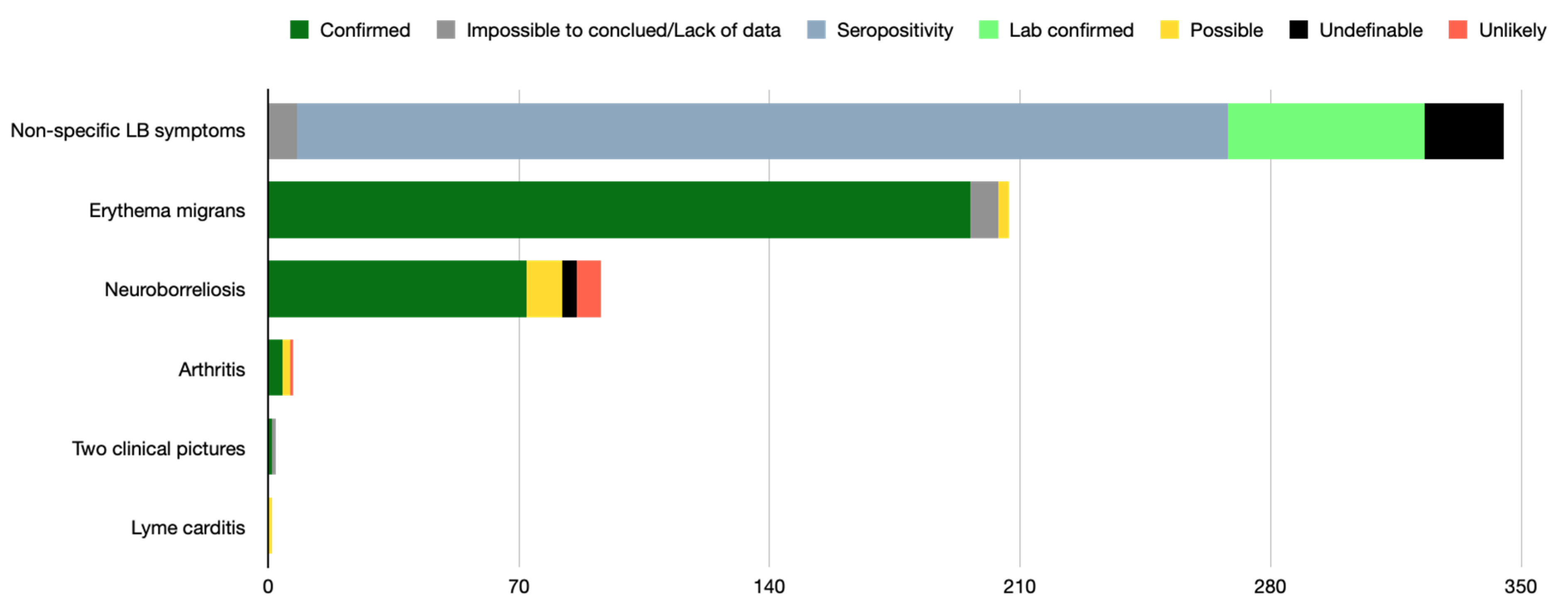
2.3. Clinical Picture of LB Observed in Patients Deemed Co-Infected
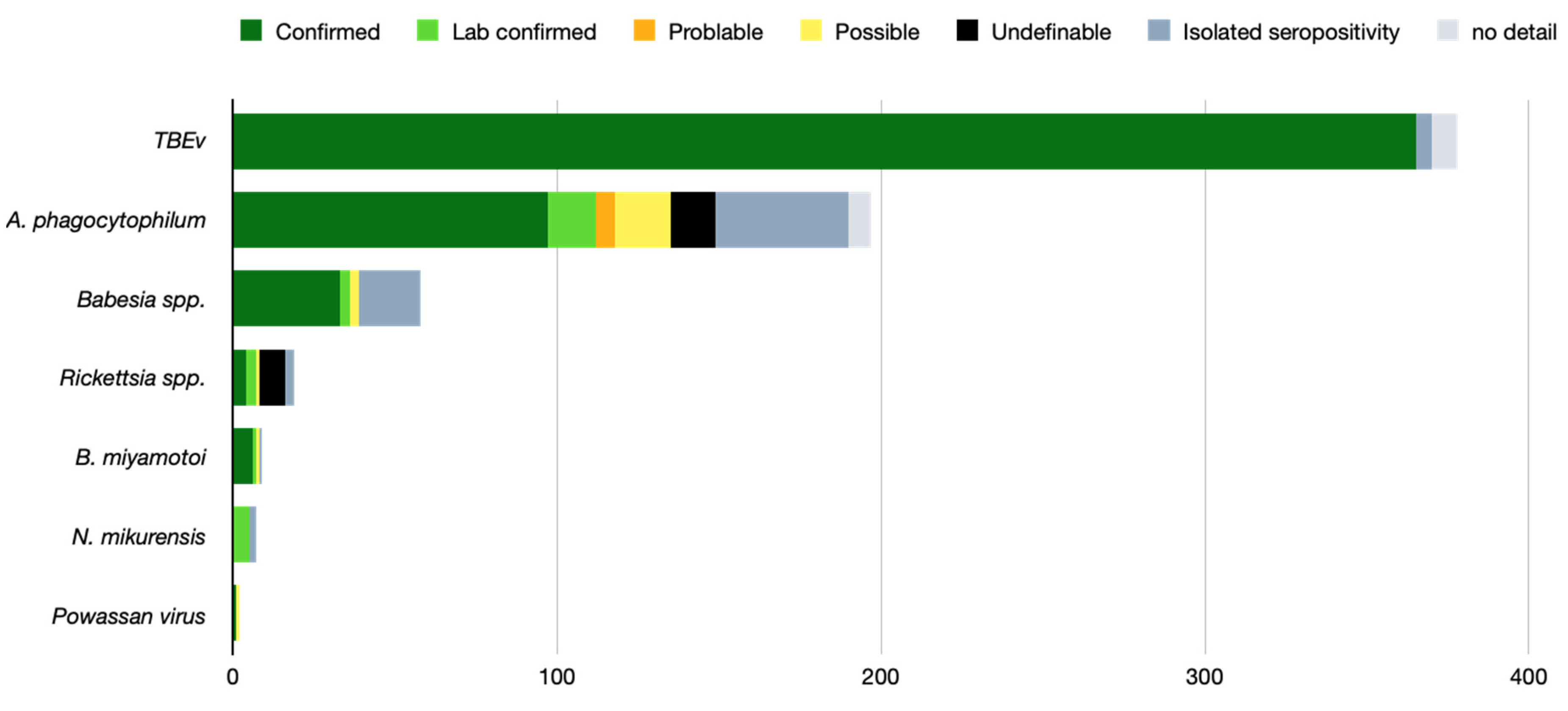
2.4. TBMs Associated with B. burgdorferi s.l.
2.5. Association between LB Clinical Presentation and Its Co-Pathogens
2.6. Impact of Co-Infection
2.7. Treatment
2.8. Outcome
3. Discussion
4. Materials and Methods
4.1. Search Strategy and Selection Criteria
4.2. Inclusion Criteria
4.3. Exclusion Criteria
4.4. Studies’ Analysis and Data Collection
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dantas-Torres, F.; Chomel, B.B.; Otranto, D. Ticks and Tick-Borne Diseases: A One Health Perspective. Trends Parasitol. 2012, 28, 437–446. [Google Scholar] [CrossRef]
- Keirans, J.E.; Needham, G.R.; Oliver, J.H., Jr. The Ixodes Ricinus Complex Worldwide: Diagnosis of the Species in the Complex, Hosts and Distribution. Acarol. IX 1999, 2, 341–347. [Google Scholar]
- Moutailler, S.; Valiente Moro, C.; Vaumourin, E.; Michelet, L.; Tran, F.H.; Devillers, E.; Cosson, J.-F.; Gasqui, P.; Van, V.T.; Mavingui, P.; et al. Co-Infection of Ticks: The Rule Rather than the Exception. PLOS Negl. Trop. Dis. 2016, 10, e0004539. [Google Scholar] [CrossRef]
- Ginsberg, H.S. Potential Effects of Mixed Infections in Ticks on Transmission Dynamics of Pathogens: Comparative Analysis of Published Records. Exp. Appl. Acarol. 2008, 46, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Bertola, M.; Montarsi, F.; Obber, F.; da Rold, G.; Carlin, S.; Toniolo, F.; Porcellato, E.; Falcaro, C.; Mondardini, V.; Ormelli, S.; et al. Occurrence and Identification of Ixodes Ricinus Borne Pathogens in Northeastern Italy. Pathogens 2021, 10, 1181. [Google Scholar] [CrossRef] [PubMed]
- Moustafa, M.A.M.; Taylor, K.; Nakao, R.; Shimozuru, M.; Sashika, M.; Rosà, R.; Thu, M.J.; Rizzoli, A.; Tsubota, T. Dynamics, Co-Infections and Characteristics of Zoonotic Tick-Borne Pathogens in Hokkaido Small Mammals, Japan. Ticks Tick-Borne Dis. 2016, 7, 922–928. [Google Scholar] [CrossRef] [PubMed]
- Egizi, A.; Roegner, V.; Faraji, A.; Healy, S.P.; Schulze, T.L.; Jordan, R.A. A Historical Snapshot of Ixodes Scapularis-Borne Pathogens in New Jersey Ticks Reflects a Changing Disease Landscape. Ticks Tick-Borne Dis. 2018, 9, 418–426. [Google Scholar] [CrossRef] [PubMed]
- Rigaud, E.; Jaulhac, B.; Garcia-Bonnet, N.; Hunfeld, K.-P.; Femenia, F.; Huet, D.; Goulvestre, C.; Vaillant, V.; Deffontaines, G.; Abadia-Benoist, G. Seroprevalence of Seven Pathogens Transmitted by the Ixodes Ricinus Tick in Forestry Workers in France. Clin. Microbiol. Infect. 2016, 22, 735.e1–735.e9. [Google Scholar] [CrossRef]
- Wormser, G.P.; Horowitz, H.W.; Nowakowski, J.; Mckenna, D.; Stephen Dumler, J.; Varde, S.; Schwartz, I.; Carbonaro, C.; Aguero-Rosenfeld, M. Positive Lyme Disease Serology in Patients With Clinical and Laboratory Evidence of Human Granulocytic Ehrlichiosis. Am. J. Clin. Pathol. 1997, 107, 142–147. [Google Scholar] [CrossRef]
- Jahfari, S.; Hofhuis, A.; Fonville, M.; van der Giessen, J.; van Pelt, W.; Sprong, H. Molecular Detection of Tick-Borne Pathogens in Humans with Tick Bites and Erythema Migrans, in the Netherlands. PLoS Negl. Trop. Dis. 2016, 10, e0005042. [Google Scholar] [CrossRef]
- Moniuszko-Malinowska, A.; Dunaj, J.; Andersson, M.O.; Chmielewski, T.; Czupryna, P.; Groth, M.; Grygorczuk, S.; Zajkowska, J.; Kondrusik, M.; Kruszewska, E.; et al. Anaplasmosis in Poland—Analysis of 120 Patients. Ticks Tick-Borne Dis. 2021, 12, 101763. [Google Scholar] [CrossRef]
- Grant, L.; Mohamedy, I.; Loertscher, L. One Man, Three Tick-Borne Illnesses. BMJ Case Rep. 2021, 14, e241004. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.M.; Shahzad, S.R.; Ashraf, M.F.; Naseer, U. Powassan Virus Encephalitis, Severe Babesiosis and Lyme Carditis in a Single Patient. BMJ Case Rep. 2019, 12, e231645. [Google Scholar] [CrossRef] [PubMed]
- Moniuszko, A.; Dunaj, J.; Swięcicka, I.; Zambrowski, G.; Chmielewska-Badora, J.; Zukiewicz-Sobczak, W.; Zajkowska, J.; Czupryna, P.; Kondrusik, M.; Grygorczuk, S.; et al. Co-Infections with Borrelia Species, Anaplasma Phagocytophilum and Babesia Spp. in Patients with Tick-Borne Encephalitis. Eur. J. Clin. Microbiol. Infect. Dis. Off. Publ. Eur. Soc. Clin. Microbiol. 2014, 33, 1835–1841. [Google Scholar] [CrossRef]
- Molloy, P.J.; Telford, S.R.; Chowdri, H.R.; Lepore, T.J.; Gugliotta, J.L.; Weeks, K.E.; Hewins, M.E.; Goethert, H.K.; Berardi, V.P. Borrelia Miyamotoi Disease in the Northeastern United States: A Case Series. Ann. Intern. Med. 2015, 163, 91–98. [Google Scholar] [CrossRef]
- Lindblom, A.; Wallménius, K.; Sjöwall, J.; Fryland, L.; Wilhelmsson, P.; Lindgren, P.-E.; Forsberg, P.; Nilsson, K. Prevalence of Rickettsia spp. in Ticks and Serological and Clinical Outcomes in Tick-Bitten Individuals in Sweden and on the Åland Islands. PLoS ONE 2016, 11, e0166653. [Google Scholar] [CrossRef] [PubMed]
- Ocias, L.F.; Wilhelmsson, P.; Sjöwall, J.; Henningsson, A.J.; Nordberg, M.; Jørgensen, C.S.; Krogfelt, K.A.; Forsberg, P.; Lindgren, P.-E. Emerging Tick-Borne Pathogens in the Nordic Countries: A Clinical and Laboratory Follow-up Study of High-Risk Tick-Bitten Individuals. Ticks Tick-Borne Dis. 2020, 11, 101303. [Google Scholar] [CrossRef] [PubMed]
- Beltrame, A.; Ruscio, M.; Arzese, A.; Rorato, G.; Negri, C.; Londero, A.; Crapis, M.; Scudeller, L.; Viale, P. Human Granulocytic Anaplasmosis in Northeastern Italy. Ann. N. Y. Acad. Sci. 2006, 1078, 106–109. [Google Scholar] [CrossRef]
- Grankvist, A.; Sandelin, L.L.; Andersson, J.; Fryland, L.; Wilhelmsson, P.; Lindgren, P.-E.; Forsberg, P.; Wennerås, C. Infections with Candidatus Neoehrlichia Mikurensis and Cytokine Responses in 2 Persons Bitten by Ticks, Sweden. Emerg. Infect. Dis. 2015, 21, 1462–1465. [Google Scholar] [CrossRef]
- Kalinova, Z.; Halanova, M.; Cislakova, L.; Sulinova, Z.; Jarcuska, P. Occurrence of IgG Antibodies to Anaplasma Phagocytophilum in Humans Suspected of Lyme Borreliosis in Eastern Slovakia. Ann. Agric. Environ. Med. AAEM 2009, 16, 285–288. [Google Scholar]
- Welc-Falęciak, R.; Hildebrandt, A.; Siński, E. Co-Infection with Borrelia Species and Other Tick-Borne Pathogens in Humans: Two Cases from Poland. Ann. Agric. Environ. Med. AAEM 2010, 17, 309–313. [Google Scholar] [PubMed]
- Tijsse-Klasen, E.; Sprong, H.; Pandak, N. Co-Infection of Borrelia Burgdorferi Sensu Lato and Rickettsia Species in Ticks and in an Erythema Migrans Patient. Parasit. Vectors 2013, 6, 347. [Google Scholar] [CrossRef] [PubMed]
- Steere, A.C.; McHugh, G.; Suarez, C.; Hoitt, J.; Damle, N.; Sikand, V.K. Prospective Study of Coinfection in Patients with Erythema Migrans. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2003, 36, 1078–1081. [Google Scholar] [CrossRef] [PubMed]
- Ocias, L.F.; Dessau, R.B.; Jørgensen, C.S.; Krogfelt, K.A.; Ornstein, K. More than Just Borrelia? A Study of Co-Infection and Etiology in Erythema Migrans Patients from Southernmost Sweden. Infect. Dis. 2019, 51, 618–621. [Google Scholar] [CrossRef] [PubMed]
- Hulínská, D.; Votýpka, J.; Vanousová, D.; Hercogová, J.; Hulínský, V.; Drevová, H.; Kurzová, Z.; Uherková, L. Identification of Anaplasma Phagocytophilum and Borrelia Burgdorferi Sensu Lato in Patients with Erythema Migrans. Folia Microbiol. 2009, 54, 246–256. [Google Scholar] [CrossRef]
- Moniuszko-Malinowska, A.; Dunaj, J.; Andersson, M.O.; Czupryna, P.; Zajkowska, J.; Guziejko, K.; Garkowski, A.; Grygorczuk, S.; Kondrusik, M.; Pancewicz, S. Assessment of Anaplasma Phagocytophilum Presence in Early Lyme Borreliosis Manifested by Erythema Migrans Skin Lesions. Travel Med. Infect. Dis. 2020, 36, 101648. [Google Scholar] [CrossRef]
- Wormser, G.P.; McKenna, D.; Scavarda, C.; Cooper, D.; El Khoury, M.Y.; Nowakowski, J.; Sudhindra, P.; Ladenheim, A.; Wang, G.; Karmen, C.L.; et al. Co-Infections in Persons with Early Lyme Disease, New York, USA. Emerg. Infect. Dis. 2019, 25, 748–752. [Google Scholar] [CrossRef]
- Christova, I.S.; Dumler, J.S. Human Granulocytic Ehrlichiosis in Bulgaria. Am. J. Trop. Med. Hyg. 1999, 60, 58–61. [Google Scholar] [CrossRef][Green Version]
- Horowitz, H.W.; Aguero-Rosenfeld, M.E.; Holmgren, D.; McKenna, D.; Schwartz, I.; Cox, M.E.; Wormser, G.P. Lyme Disease and Human Granulocytic Anaplasmosis Coinfection: Impact of Case Definition on Coinfection Rates and Illness Severity. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2013, 56, 93–99. [Google Scholar] [CrossRef]
- Welc-Falęciak, R.; Kowalec, M.; Zajkowska, J.; Pancewicz, S.A.; Siński, E. Clinical and Molecular Features of One Case of Human Infection with Anaplasma Phagocytophilum from Podlaskie Province in Eastern Poland. Ann. Agric. Environ. Med. AAEM 2015, 22, 414–417. [Google Scholar] [CrossRef]
- Gyllemark, P.; Wilhelmsson, P.; Elm, C.; Hoornstra, D.; Hovius, J.W.; Johansson, M.; Tjernberg, I.; Lindgren, P.-E.; Henningsson, A.J.; Sjöwall, J. Are Other Tick-Borne Infections Overlooked in Patients Investigated for Lyme Neuroborreliosis? A Large Retrospective Study from South-Eastern Sweden. Ticks Tick-Borne Dis. 2021, 12, 101759. [Google Scholar] [CrossRef] [PubMed]
- Benach, J.L.; Coleman, J.L.; Habicht, G.S.; MacDonald, A.; Grunwaldt, E.; Giron, J.A. Serological Evidence for Simultaneous Occurrences of Lyme Disease and Babesiosis. J. Infect. Dis. 1985, 152, 473–477. [Google Scholar] [CrossRef] [PubMed]
- Cimperman, J.; Maraspin, V.; Lotric-Furlan, S.; Ruzić-Sabljić, E.; Avsic-Zupanc, T.; Picken, R.N.; Strle, F. Concomitant Infection with Tick-Borne Encephalitis Virus and Borrelia Burgdorferi Sensu Lato in Patients with Acute Meningitis or Meningoencephalitis. Infection 1998, 26, 160–164. [Google Scholar] [CrossRef] [PubMed]
- Ocias, L.F.; Dessau, R.B.; Lebech, A.-M.; Jørgensen, C.S.; Petersen, R.F.; Krogfelt, K.A. Evidence of Rickettsiae in Danish Patients Tested for Lyme Neuroborreliosis: A Retrospective Study of Archival Samples. BMC Infect. Dis. 2018, 18, 325. [Google Scholar] [CrossRef]
- Tomazic, J.; Ihan, A.; Strle, F.; Cimperman, J.; Stare, J.; Maticic, M.; Zakotnik, B. Immunological Differentiation between Tickborne Encephalitis with and without Concomitant Neuroborreliosis. Eur. J. Clin. Microbiol. Infect. Dis. Off. Publ. Eur. Soc. Clin. Microbiol. 1997, 16, 920–923. [Google Scholar] [CrossRef]
- Krbková, L.; Homola, L.; Hlaváčová, A.; Mikolášek, P.; Bednářová, J.; Čermáková, Z. Assessment of Antibodies against Surface and Outer Membrane Proteins of Anaplasma Phagocytophilum in Lyme Borreliosis and Tick-Borne Encephalitis Paediatric Patients. Epidemiol. Infect. 2016, 144, 2597–2604. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Velušček, M.; Blagus, R.; Cerar Kišek, T.; Ružić-Sabljić, E.; Avšič-Županc, T.; F Bajrović, F.; Stupica, D. Antibiotic Use and Long-Term Outcome in Patients with Tick-Borne Encephalitis and Co-Infection with Borrelia Burgdorferi Sensu Lato in Central Europe. A Retrospective Cohort Study. J. Clin. Med. 2019, 8, E1740. [Google Scholar] [CrossRef]
- Lotric-Furlan, S.; Strle, F. Peripheral Facial Palsy in Patients with Tick-Borne Encephalitis. Clin. Microbiol. Infect. Off. Publ. Eur. Soc. Clin. Microbiol. Infect. Dis. 2012, 18, 1027–1032. [Google Scholar] [CrossRef][Green Version]
- Belongia, E.A.; Reed, K.D.; Mitchell, P.D.; Mueller-Rizner, N.; Vandermause, M.; Finkel, M.F.; Kazmierczak, J.J. Tickborne Infections as a Cause of Nonspecific Febrile Illness in Wisconsin. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2001, 32, 1434–1439. [Google Scholar] [CrossRef]
- Dunaj, J.; Moniuszko-Malinowska, A.; Swiecicka, I.; Andersson, M.; Czupryna, P.; Rutkowski, K.; Zambrowski, G.; Zajkowska, J.; Grygorczuk, S.; Kondrusik, M.; et al. Tick-Borne Infections and Co-Infections in Patients with Non-Specific Symptoms in Poland. Adv. Med. Sci. 2018, 63, 167–172. [Google Scholar] [CrossRef]
- Liu, H.-B.; Wei, R.; Ni, X.-B.; Zheng, Y.-C.; Huo, Q.-B.; Jiang, B.-G.; Ma, L.; Jiang, R.-R.; Lv, J.; Liu, Y.-X.; et al. The Prevalence and Clinical Characteristics of Tick-Borne Diseases at One Sentinel Hospital in Northeastern China. Parasitology 2019, 146, 161–167. [Google Scholar] [CrossRef]
- Bjöersdorff, A.; Wittesjö, B.; Berglund, J.; Massung, R.F.; Eliasson, I. Human Granulocytic Ehrlichiosis as a Common Cause of Tick-Associated Fever in Southeast Sweden: Report from a Prospective Clinical Study. Scand. J. Infect. Dis. 2002, 34, 187–191. [Google Scholar] [CrossRef]
- Mitchell, P.D.; Reed, K.D.; Hofkes, J.M. Immunoserologic Evidence of Coinfection with Borrelia Burgdorferi, Babesia Microti, and Human Granulocytic Ehrlichia Species in Residents of Wisconsin and Minnesota. J. Clin. Microbiol. 1996, 34, 724–727. [Google Scholar] [CrossRef]
- Belongia, E.A.; Reed, K.D.; Mitchell, P.D.; Chyou, P.-H.; Mueller-Rizner, N.; Finkel, M.F.; Schriefer, M.E. Clinical and Epidemiological Features of Early Lyme Disease and Human Granulocytic Ehrlichiosis in Wisconsin. Clin. Infect. Dis. 1999, 29, 1472–1477. [Google Scholar] [CrossRef] [PubMed]
- Taba, P.; Schmutzhard, E.; Forsberg, P.; Lutsar, I.; Ljøstad, U.; Mygland, Å.; Levchenko, I.; Strle, F.; Steiner, I. EAN Consensus Review on Prevention, Diagnosis and Management of Tick-Borne Encephalitis. Eur. J. Neurol. 2017, 24, 1214-e61. [Google Scholar] [CrossRef]
- Meer-Scherrer, L.; Adelson, M.; Mordechai, E.; Lottaz, B.; Tilton, R. Babesia Microti Infection in Europe. Curr. Microbiol. 2004, 48, 435–437. [Google Scholar] [CrossRef] [PubMed]
- Duffy, J.; Pittlekow, M.R.; Kolbert, C.P.; Rutledge, B.J.; Persing, D.H. Coinfection with Borrelia Burgdorferi and the Agent of Human Granulocytic Ehrlichiosis. Lancet 1997, 349, 399. [Google Scholar] [CrossRef]
- Nadelman, R.B.; Horowitz, H.W.; Hsieh, T.; Wu, J.M.; Aguero-Rosenfeld, M.E.; Schwartz, I.; Nowakowski, J.; Varde, S.; Wormser, G.P. Simultaneous Human Granulocytic Ehrlichiosis and Lyme Borreliosis. N. Engl. J. Med. 1997, 337, 27–30. [Google Scholar] [CrossRef]
- Hulinska, D.; Votypka, J.; Plch, J.; Vlcek, E.; Valesová, M.; Bojar, M.; Hulinsky, V.; Smetana, K. Molecular and Microscopical Evidence of Ehrlichia Spp. and Borrelia Burgdorferi Sensu Lato in Patients, Animals and Ticks in the Czech Republic. New Microbiol. 2002, 25, 437–448. [Google Scholar]
- Hoversten, K.; Bartlett, M.A. Diagnosis of a Tick-Borne Coinfection in a Patient with Persistent Symptoms Following Treatment for Lyme Disease. BMJ Case Rep. 2018, 2018, bcr-2018-225342. [Google Scholar] [CrossRef]
- Saetre, K.; Godhwani, N.; Maria, M.; Patel, D.; Wang, G.; Li, K.I.; Wormser, G.P.; Nolan, S.M. Congenital Babesiosis After Maternal Infection With Borrelia Burgdorferi and Babesia Microti. J. Pediatr. Infect. Dis. Soc. 2018, 7, e1–e5. [Google Scholar] [CrossRef]
- Martínez-Balzano, C.; Hess, M.; Malhotra, A.; Lenox, R. Severe Babesiosis and Borrelia Burgdorferi Co-Infection. QJM Int. J. Med. 2015, 108, 141–143. [Google Scholar] [CrossRef]
- Oksi, J.; Viljanen, M.K.; Kalimo, H.; Peltonen, R.; Marttila, R.; Salomaa, P.; Nikoskelainen, J.; Budka, H.; Halonen, P. Fatal Encephalitis Caused by Concomitant Infection with Tick-Borne Encephalitis Virus and Borrelia Burgdorferi. Clin. Infect. Dis. 1993, 16, 392–396. [Google Scholar] [CrossRef] [PubMed]
- Henningsson, A.J.; Aase, A.; Bavelaar, H.; Flottorp, S.; Forsberg, P.; Kirkehei, I.; Lövmar, M.; Nilsson, K.; Nyman, D.; Ornstein, K.; et al. Laboratory Methods for Detection of Infectious Agents and Serological Response in Humans With Tick-Borne Infections: A Systematic Review of Evaluations Based on Clinical Patient Samples. Front. Public Health 2021, 9, 580102. [Google Scholar] [CrossRef]
- Dumic, I.; Glomski, B.; Patel, J.; Nordin, T.; Nordstrom, C.W.; Sprecher, L.J.; Niendorf, E.; Singh, A.; Simeunovic, K.; Subramanian, A.; et al. “Double Trouble”: Severe Meningoencephalitis Due to Borrelia Burgdorferi and Powassan Virus Co-Infection Successfully Treated with Intravenous Immunoglobulin. Am. J. Case Rep. 2021, 22, e929952. [Google Scholar] [CrossRef] [PubMed]
- Dessau, R.B.; van Dam, A.P.; Fingerle, V.; Gray, J.; Hovius, J.W.; Hunfeld, K.-P.; Jaulhac, B.; Kahl, O.; Kristoferitsch, W.; Lindgren, P.-E.; et al. To Test or Not to Test? Laboratory Support for the Diagnosis of Lyme Borreliosis: A Position Paper of ESGBOR, the ESCMID Study Group for Lyme Borreliosis. Clin. Microbiol. Infect. 2018, 24, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Wormser, G.P. Documentation of a False Positive Lyme Disease Serologic Test in a Patient with Untreated Babesia Microti Infection Carries Implications for Accurately Determining the Frequency of Lyme Disease Coinfections. Diagn. Microbiol. Infect. Dis. 2021, 101, 115429. [Google Scholar] [CrossRef]
- Strle, F.; Stanek, G. Clinical Manifestations and Diagnosis of Lyme Borreliosis. In Lyme Borreliosis: Biological and Clinical Aspects; Karger: Basel, Switzerland, 2009; pp. 51–110. [Google Scholar]
- Lantos, P.M.; Rumbaugh, J.; Bockenstedt, L.K.; Falck-Ytter, Y.T.; Aguero-Rosenfeld, M.E.; Auwaerter, P.G.; Baldwin, K.; Bannuru, R.R.; Belani, K.K.; Bowie, W.R.; et al. Clinical Practice Guidelines by the Infectious Diseases Society of America (IDSA), American Academy of Neurology (AAN), and American College of Rheumatology (ACR): 2020 Guidelines for the Prevention, Diagnosis and Treatment of Lyme Disease. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2021, 72, e1–e48. [Google Scholar] [CrossRef]
- Krause, P.J.; Auwaerter, P.G.; Bannuru, R.R.; Branda, J.A.; Falck-Ytter, Y.T.; Lantos, P.M.; Lavergne, V.; Meissner, H.C.; Osani, M.C.; Rips, J.G.; et al. Clinical Practice Guidelines by the Infectious Diseases Society of America (IDSA): 2020 Guideline on Diagnosis and Management of Babesiosis. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2021, 72, e49–e64. [Google Scholar] [CrossRef]
- Biggs, H.M.; Behravesh, C.B.; Bradley, K.K.; Dahlgren, F.S.; Drexler, N.A.; Dumler, J.S.; Folk, S.M.; Kato, C.Y.; Lash, R.R.; Levin, M.L.; et al. Diagnosis and Management of Tickborne Rickettsial Diseases: Rocky Mountain Spotted Fever and Other Spotted Fever Group Rickettsioses, Ehrlichioses, and Anaplasmosis—United States. MMWR Recomm. Rep. Morb. Mortal. Wkly. Rep. Recomm. Rep. 2016, 65, 1–44. [Google Scholar] [CrossRef]
- Wormser, G.P.; McKenna, D.; Nowakowski, J. Management Approaches for Suspected and Established Lyme Disease Used at the Lyme Disease Diagnostic Center. Wien. Klin. Wochenschr. 2018, 130, 463–467. [Google Scholar] [CrossRef]
- Logina, I.; Krumina, A.; Karelis, G.; Elsone, L.; Viksna, L.; Rozentale, B.; Donaghy, M. Clinical Features of Double Infection with Tick-Borne Encephalitis and Lyme Borreliosis Transmitted by Tick Bite. J. Neurol. Neurosurg. Psychiatry 2006, 77, 1350–1353. [Google Scholar] [CrossRef][Green Version]
- Lou, Y.; Liu, L.; Gao, D. Modeling Co-Infection of Ixodes Tick-Borne Pathogens. Math. Biosci. Eng. MBE 2017, 14, 1301–1316. [Google Scholar] [CrossRef] [PubMed]
- Hersh, M.H.; Ostfeld, R.S.; McHenry, D.J.; Tibbetts, M.; Brunner, J.L.; Killilea, M.E.; LoGiudice, K.; Schmidt, K.A.; Keesing, F. Co-Infection of Blacklegged Ticks with Babesia Microti and Borrelia Burgdorferi Is Higher than Expected and Acquired from Small Mammal Hosts. PLoS ONE 2014, 9, e99348. [Google Scholar] [CrossRef] [PubMed]
- Hermance, M.E.; Thangamani, S. Tick–Virus–Host Interactions at the Cutaneous Interface: The Nidus of Flavivirus Transmission. Viruses 2018, 10, 362. [Google Scholar] [CrossRef]
- Djokic, V.; Akoolo, L.; Primus, S.; Schlachter, S.; Kelly, K.; Bhanot, P.; Parveen, N. Protozoan Parasite Babesia Microti Subverts Adaptive Immunity and Enhances Lyme Disease Severity. Front. Microbiol. 2019, 10, 1596. [Google Scholar] [CrossRef]
- Bhanot, P.; Parveen, N. Investigating Disease Severity in an Animal Model of Concurrent Babesiosis and Lyme Disease. Int. J. Parasitol. 2019, 49, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Krause, P.J.; Telford, S.R.; Spielman, A.; Sikand, V.; Ryan, R.; Christianson, D.; Burke, G.; Brassard, P.; Pollack, R.; Peck, J.; et al. Concurrent Lyme Disease and Babesiosis. Evidence for Increased Severity and Duration of Illness. JAMA 1996, 275, 1657–1660. [Google Scholar] [CrossRef]
- Thomas, V.; Anguita, J.; Barthold, S.W.; Fikrig, E. Coinfection with Borrelia Burgdorferi and the Agent of Human Granulocytic Ehrlichiosis Alters Murine Immune Responses, Pathogen Burden, and Severity of Lyme Arthritis. Infect. Immun. 2001, 69, 3359–3371. [Google Scholar] [CrossRef]
- Grab, D.J.; Nyarko, E.; Barat, N.C.; Nikolskaia, O.V.; Dumler, J.S. Anaplasma Phagocytophilum-Borrelia Burgdorferi Coinfection Enhances Chemokine, Cytokine, and Matrix Metalloprotease Expression by Human Brain Microvascular Endothelial Cells. Clin. Vaccine Immunol. CVI 2007, 14, 1420–1424. [Google Scholar] [CrossRef]
- Růžek, D.; Salát, J.; Singh, S.K.; Kopecký, J. Breakdown of the Blood-Brain Barrier during Tick-Borne Encephalitis in Mice Is Not Dependent on CD8+ T-Cells. PLoS ONE 2011, 6, e20472. [Google Scholar] [CrossRef]
| Type of Cohort | Geographical Area | No of Patients Explored | Frequency of Co-Infection | References |
|---|---|---|---|---|
| Tick bitten people | Europe | 495 | 1.0% | [16,17,18,19] |
| Patients with LB suspicion | Europe | 214 | 0.9% | [20] |
| Patient with LB | Europe | 24 | 4.2% | [21] |
| Patients with EM | Europe & US | 1309 | 5.9% | [10,22,23,24,25,26,27,28,29,30] |
| Neuroborreliosis suspicions | Europe | 1333 | 2.7% | [31,32,33,34,35] |
| Lyme arthritis suspicions | Europe | 146 | 0.7% | [36] |
| TBE patients | Europe | 805 | 41.6% | [14,37,38] |
| Patients with post-tick bite fever | Europe & US & China | 416 | 4.3% | [39,40,41,42] |
| Patients with Babesiosis | US | 41 | 22.0% | [32] |
| Patients with BMD | US | 51 | 11.7% | [15] |
| Patients with HGA or HGA suspicion | US & Europe | 496 | 9.7% | [9,11,43,44] |
| LB Clinical Picture | Co-Infection Agent | No. of Patients | Reference |
|---|---|---|---|
| Erythema migrans | A. phagocytophilum | 63 | [11,23,25,29,30,40,42,47,48,49] |
| TBEv | 15 | [48] | |
| Babesia spp. | 8 | [23,27,50,51,52] | |
| A. phagocytophilum & TBEv | 5 | [11] | |
| Rickettsia spp. | 2 | [41] | |
| B. miyamotoi | 1 | [15] | |
| Neuroborreliosis | TBEv | 62 | [33,35,37,38,53] |
| Powassan virus | 1 | [54] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boyer, P.H.; Lenormand, C.; Jaulhac, B.; Talagrand-Reboul, E. Human Co-Infections between Borrelia burgdorferi s.l. and Other Ixodes-Borne Microorganisms: A Systematic Review. Pathogens 2022, 11, 282. https://doi.org/10.3390/pathogens11030282
Boyer PH, Lenormand C, Jaulhac B, Talagrand-Reboul E. Human Co-Infections between Borrelia burgdorferi s.l. and Other Ixodes-Borne Microorganisms: A Systematic Review. Pathogens. 2022; 11(3):282. https://doi.org/10.3390/pathogens11030282
Chicago/Turabian StyleBoyer, Pierre H., Cédric Lenormand, Benoît Jaulhac, and Emilie Talagrand-Reboul. 2022. "Human Co-Infections between Borrelia burgdorferi s.l. and Other Ixodes-Borne Microorganisms: A Systematic Review" Pathogens 11, no. 3: 282. https://doi.org/10.3390/pathogens11030282
APA StyleBoyer, P. H., Lenormand, C., Jaulhac, B., & Talagrand-Reboul, E. (2022). Human Co-Infections between Borrelia burgdorferi s.l. and Other Ixodes-Borne Microorganisms: A Systematic Review. Pathogens, 11(3), 282. https://doi.org/10.3390/pathogens11030282





