An Index of Non-Linear HRV as a Proxy of the Aerobic Threshold Based on Blood Lactate Concentration in Elite Triathletes
Abstract
:1. Introduction
2. Methods
2.1. Participants
2.2. Exercise Testing Protocol
2.3. Lactate Testing
2.4. RR Measurements and Calculation of DFA a1-Derived HRVT
2.5. Statistics
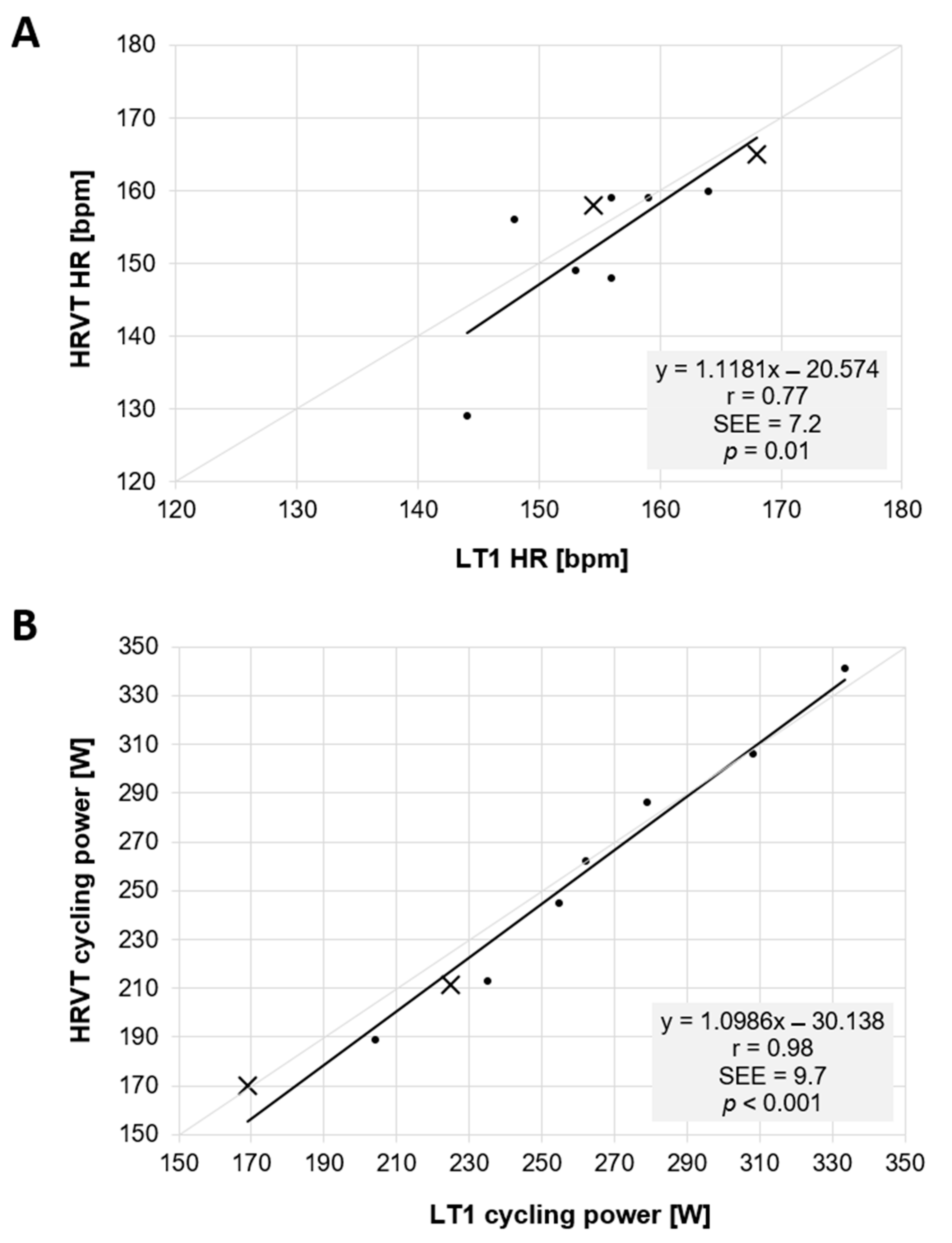
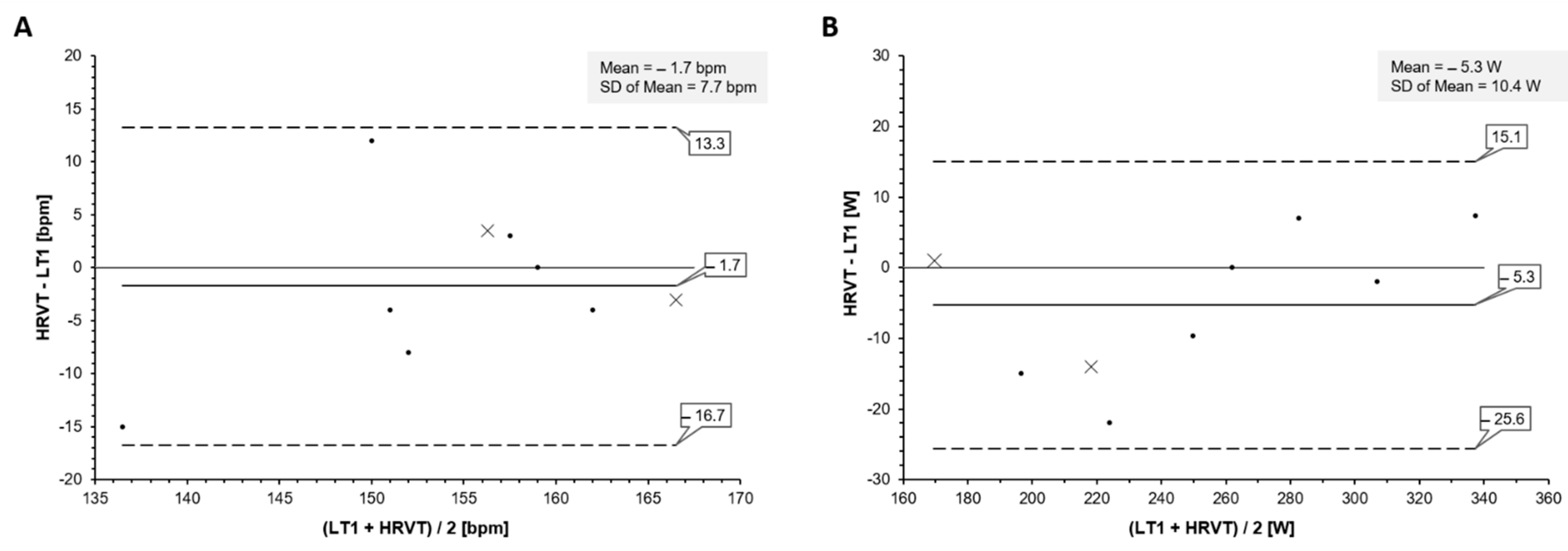
3. Results
Lactate Threshold and HRVT Comparison
4. Discussion
5. Limitations and Future Directions
6. Practical Applications
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Stöggl, T.L.; Sperlich, B. Editorial: Training Intensity, Volume and Recovery Distribution Among Elite and Recreational Endurance Athletes. Front. Physiol. 2019, 10, 592. [Google Scholar] [CrossRef] [PubMed]
- Mann, T.; Lamberts, R.P.; Lambert, M.I. Methods of prescribing relative exercise intensity: Physiological and practical considerations. Sports Med. 2013, 43, 613–625. [Google Scholar] [CrossRef] [PubMed]
- Faude, O.; Kindermann, W.; Meyer, T. Lactate threshold concepts. Sports Med. 2009, 39, 469–490. [Google Scholar] [CrossRef] [PubMed]
- Newell, J.; Higgins, D.; Madden, N.; Cruickshank, J.; Einbeck, J.; McMillan, K.; McDonald, R. Software for calculating blood lactate endurance markers. J. Sports Sci. 2007, 25, 1403–1409. [Google Scholar] [CrossRef] [PubMed]
- Tulppo, M.P.; Mäkikallio, T.H.; Takala, T.E.; Seppänen, T.; Huikuri, H.V. Quantitative beat-to-beat analysis of heart rate dynamics during exercise. Am. J. Physiol. 1996, 271, H244–H252. [Google Scholar] [CrossRef] [PubMed]
- Cottin, F.; Médigue, C.; Lopes, P.; Leprêtre, P.M.; Heubert, R.; Billat, V. Ventilatory thresholds assessment from heart rate variability during an incremental exhaustive running test. Int. J. Sports Med. 2007, 28, 287–294. [Google Scholar] [CrossRef]
- Karapetian, G.K.; Engels, H.J.; Gretebeck, R.J. Use of heart rate variability to estimate LT and VT. Int. J. Sports Med. 2008, 29, 652–657. [Google Scholar] [CrossRef]
- Gronwald, T.; Rogers, B.; Hoos, O. Fractal Correlation Properties of Heart Rate Variability: A New Biomarker for Intensity Distribution in Endurance Exercise and Training Prescription? Front. Physiol. 2020, 11, 1152. [Google Scholar] [CrossRef]
- Rogers, B.; Giles, D.; Draper, N.; Hoos, O.; Gronwald, T. A New Detection Method Defining the Aerobic Threshold for Endurance Exercise and Training Prescription Based on Fractal Correlation Properties of Heart Rate Variability. Front. Physiol. 2021, 11, 596567. [Google Scholar] [CrossRef]
- Casado, A.; Hanley, B.; Santos-Concejero, J.; Ruiz-Pérez, L.M. World-Class Long-Distance Running Performances Are Best Predicted by Volume of Easy Runs and Deliberate Practice of Short-Interval and Tempo Runs. J. Strength Cond. Res. 2019, 35, 2525–2531. [Google Scholar] [CrossRef]
- Fokkema, T.; van Damme, A.A.; Fornerod, M.W.; de Vos, R.J.; Bierma-Zeinstra, S.M.; van Middelkoop, M. Training for a (half-)marathon: Training volume and longest endurance run related to performance and running injuries. Scand. J. Med. Sci. Sports 2020, 30, 1692–1704. [Google Scholar] [CrossRef] [PubMed]
- Tarvainen, M.P.; Niskanen, J.P.; Lipponen, J.A.; Ranta-Aho, P.O.; Karjalainen, P.A. Kubios HRV–heart rate variability analysis software. Comput. Methods Programs Biomed. 2014, 113, 210–220. [Google Scholar] [CrossRef] [PubMed]
- Rogers, B.; Giles, D.; Draper, N.; Mourot, L.; Gronwald, T. Influence of Artefact Correction and Recording Device Type on the Practical Application of a Non-Linear Heart Rate Variability Biomarker for Aerobic Threshold Determination. Sensors 2021, 21, 821. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Ivanov, P.C.; Hu, K.; Stanley, H.E. Effect of nonstationarities on detrended fluctuation analysis. Phys. Rev. E 2002, 65, 041107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bland, J.M.; Altman, D.G. Measuring agreement in method comparison studies. Stat. Methods Med. Res. 1999, 8, 135–160. [Google Scholar] [CrossRef]
- Chan, Y.H. Biostatistics 104: Correlational analysis. Singap. Med. J. 2003, 44, 614–619. [Google Scholar]
- Stöggl, T.L.; Sperlich, B. The training intensity distribution among well-trained and elite endurance athletes. Front. Physiol. 2015, 6, 295. [Google Scholar] [CrossRef] [Green Version]
- White, D.W.; Raven, P.B. Autonomic neural control of heart rate during dynamic exercise: Revisited. J. Physiol. 2014, 592, 2491–2500. [Google Scholar] [CrossRef]
- Persson, P.B. Modulation of cardiovascular control mechanisms and their interaction. Physiol. Rev. 1996, 76, 193–244. [Google Scholar] [CrossRef]
- Naranjo-Orellana, J.; Nieto-Jiménez, C.; Ruso-Álvarez, J. Non-linear heart rate dynamics during and after three controlled exercise intensities in healthy men. Physiol. Int. 2021, 107, 501–512. [Google Scholar] [CrossRef]
- Rogers, B.; Mourot, L.; Gronwald, T. Aerobic Threshold Identification in a Cardiac Disease Population Based on Correlation Properties of Heart Rate Variability. J. Clin. Med. 2021, 10, 4075. [Google Scholar] [CrossRef] [PubMed]
- Brown, B.; Millar, L.; Somauroo, J.; George, K.; Sharma, S.; La Gerche, A.; Forsythe, L.; Oxborough, D. Left ventricular remodeling in elite and sub-elite road cyclists. Scand. J. Med. Sci. Sports 2020, 30, 1132–1139. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- La Gerche, A.; Heidbüchel, H.; Burns, A.T.; Mooney, D.J.; Taylor, A.J.; Pfluger, H.B.; Inder, W.J.; Macisaac, A.I.; Prior, D.L. Disproportionate exercise load and remodeling of the athlete’s right ventricle. Med. Sci. Sports Exerc. 2011, 43, 974–981. [Google Scholar] [CrossRef] [PubMed]
- Coote, J.H.; White, M.J. CrossTalk proposal: Bradycardia in the trained athlete is attributable to high vagal tone. J. Physiol. 2015, 593, 1745–1747. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jamnick, N.A.; Botella, J.; Pyne, D.B.; Bishop, D.J. Manipulating graded exercise test variables affects the validity of the lactate threshold and VO2peak. PLoS ONE 2018, 13, e0199794. [Google Scholar] [CrossRef] [Green Version]
- Pallarés, J.G.; Morán-Navarro, R.; Ortega, J.F.; Fernández-Elías, V.E.; Mora-Rodriguez, R. Validity and Reliability of Ventilatory and Blood Lactate Thresholds in Well-Trained Cyclists. PLoS ONE 2016, 11, e0163389. [Google Scholar] [CrossRef]
- Goodwin, M.L.; Harris, J.E.; Hernández, A.; Gladden, L.B. Blood lactate measurements and analysis during exercise: A guide for clinicians. J. Diabetes Sci. Technol. 2007, 1, 558–569. [Google Scholar] [CrossRef] [Green Version]
- Hartikainen, S.; Lipponen, J.A.; Hiltunen, P.; Rissanen, T.T.; Kolk, I.; Tarvainen, M.P.; Martikainen, T.J.; Castren, M.; Väliaho, E.S.; Jäntti, H. Effectiveness of the Chest Strap Electrocardiogram to Detect Atrial Fibrillation. Am. J. Cardiol. 2019, 123, 1643–1648. [Google Scholar] [CrossRef] [Green Version]
- Gronwald, T.; Berk, S.; Altini, M.; Mourot, L.; Hoos, O.; Rogers, B. Real-Time Estimation of Aerobic Threshold and Exercise Intensity Distribution Using Fractal Correlation Properties of Heart Rate Variability: A Single-Case Field Application in a Former Olympic Triathlete. Front Sports Act. Living 2021, 28, 668812. [Google Scholar] [CrossRef]
- Rogers, B.; Mourot, L.; Doucende, G.; Gronwald, T. Fractal correlation properties of heart rate variability as a biomarker of endurance exercise fatigue in ultramarathon runners. Physiol. Rep. 2021, 9, e14956. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rogers, B.; Berk, S.; Gronwald, T. An Index of Non-Linear HRV as a Proxy of the Aerobic Threshold Based on Blood Lactate Concentration in Elite Triathletes. Sports 2022, 10, 25. https://doi.org/10.3390/sports10020025
Rogers B, Berk S, Gronwald T. An Index of Non-Linear HRV as a Proxy of the Aerobic Threshold Based on Blood Lactate Concentration in Elite Triathletes. Sports. 2022; 10(2):25. https://doi.org/10.3390/sports10020025
Chicago/Turabian StyleRogers, Bruce, Sander Berk, and Thomas Gronwald. 2022. "An Index of Non-Linear HRV as a Proxy of the Aerobic Threshold Based on Blood Lactate Concentration in Elite Triathletes" Sports 10, no. 2: 25. https://doi.org/10.3390/sports10020025
APA StyleRogers, B., Berk, S., & Gronwald, T. (2022). An Index of Non-Linear HRV as a Proxy of the Aerobic Threshold Based on Blood Lactate Concentration in Elite Triathletes. Sports, 10(2), 25. https://doi.org/10.3390/sports10020025






