MRI-Based Assessment of Etiology-Specific Sarcopenia Phenotypes in Chronic Liver Disease: A Comparative Study of MASH and Viral Hepatitis
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.2. MRI Acquisition and Analysis
2.3. Clinical Data Collection
2.4. Statistical Analysis
- -
- Model 1 included the main effects of age, BMI, PDFF, liver stiffness, AST, ALT, and disease etiology.
- -
- Model 2 extended Model 1 by including interaction terms between disease etiology and each continuous predictor (age, BMI, PDFF, stiffness).
- -
- Model 3 and Model 4 were stratified logistic regression models applied separately to MASH and Virus subgroups, respectively, to explore etiology-specific associations. Interaction terms were assessed for statistical significance to determine effect modification by etiology. Model fit was evaluated using the Hosmer–Lemeshow test and pseudo R2. Two-sided p-values < 0.05 were considered statistically significant. Analyses were conducted using Stata version 18 (StataCorp, College Station, TX, USA).
3. Results
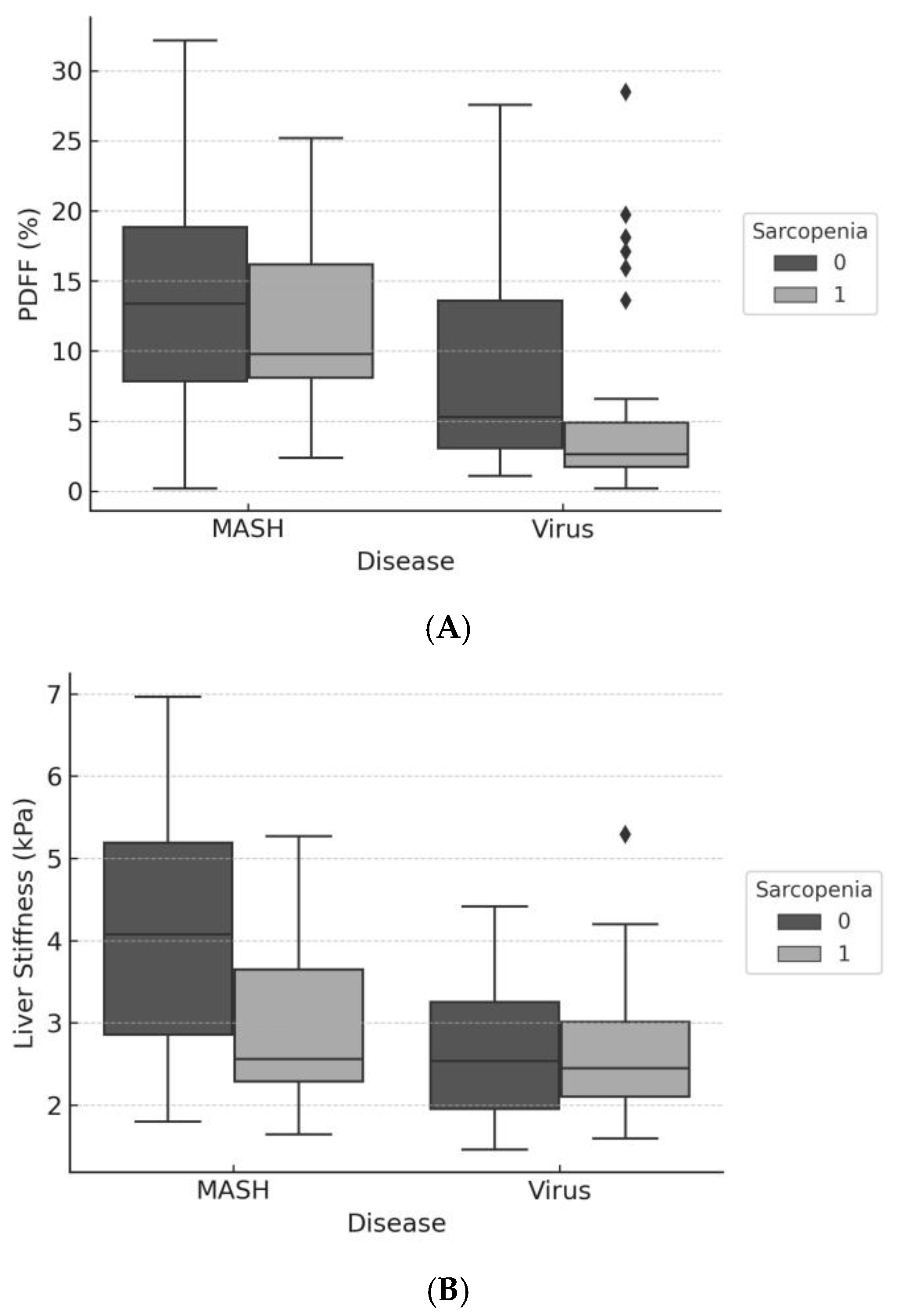
3.1. Baseline Characteristics
3.2. Multivariable Regression by Etiology
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyère, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European Consensus on Definition and Diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef]
- Dasarathy, S. Consilience in Sarcopenia of Cirrhosis. J. Cachexia Sarcopenia Muscle 2012, 3, 225–237. [Google Scholar] [CrossRef] [PubMed]
- Caussy, C.; Reeder, S.B.; Sirlin, C.B.; Loomba, R. Non-invasive, Quantitative Assessment of Liver Fat by Mri-Pdff as an Endpoint in Nash Trials. Hepatology 2018, 68, 763–772. [Google Scholar] [CrossRef] [PubMed]
- Imajo, K.; Kessoku, T.; Honda, Y.; Tomeno, W.; Ogawa, Y.; Mawatari, H.; Fujita, K.; Yoneda, M.; Taguri, M.; Hyogo, H.; et al. Magnetic Resonance Imaging More Accurately Classifies Steatosis and Fibrosis in Patients with Nonalcoholic Fatty Liver Disease Than Transient Elastography. Gastroenterology 2016, 150, 626–637.e7. [Google Scholar] [CrossRef] [PubMed]
- Jayakumar, S.; Middleton, M.S.; Lawitz, E.J.; Mantry, P.S.; Caldwell, S.H.; Arnold, H.; Diehl, A.M.; Ghalib, R.; Elkhashab, M.; Abdelmalek, M.F.; et al. Longitudinal Correlations between Mre, Mri-Pdff, and Liver Histology in Patients with Non-Alcoholic Steatohepatitis: Analysis of Data from a Phase Ii Trial of Selonsertib. J. Hepatol. 2019, 70, 133–141. [Google Scholar] [CrossRef]
- Yin, M.; Glaser, K.J.; Talwalkar, J.A.; Chen, J.; Manduca, A.; Ehman, R.L. Hepatic Mr Elastography: Clinical Performance in a Series of 1377 Consecutive Examinations. Radiology 2016, 278, 114–124. [Google Scholar] [CrossRef]
- Loomba, R.; Wolfson, T.; Ang, B.; Hooker, J.; Behling, C.; Peterson, M.; Valasek, M.; Lin, G.; Brenner, D.; Gamst, A.; et al. Magnetic Resonance Elastography Predicts Advanced Fibrosis in Patients with Nonalcoholic Fatty Liver Disease: A Prospective Study. Hepatology 2014, 60, 1920–1928. [Google Scholar] [CrossRef]
- Bohte, A.E.; de Niet, A.; Jansen, L.; Bipat, S.; Nederveen, A.J.; Verheij, J.; Terpstra, V.; Sinkus, R.; van Nieuwkerk, K.M.J.; de Knegt, R.J.; et al. Non-Invasive Evaluation of Liver Fibrosis: A Comparison of Ultrasound-Based Transient Elastography and MR Elastography in Patients with Viral Hepatitis B and C. Eur. Radiol. 2013, 24, 638–648. [Google Scholar]
- Wang, S.; Xie, H.; Gong, Y.; Kuang, J.; Yan, L.; Ruan, G.; Gao, F.; Gan, J. The Value of L3 Skeletal Muscle Index in Evaluating Preoperative Nutritional Risk and Long-Term Prognosis in Colorectal Cancer Patients. Sci. Rep. 2020, 10, 8153. [Google Scholar] [CrossRef]
- Nishikawa, H.; Shiraki, M.; Hiramatsu, A.; Moriya, K.; Hino, K.; Nishiguchi, S. Japan Society of Hepatology Guidelines for Sarcopenia in Liver Disease (1st Edition): Recommendation from the Working Group for Creation of Sarcopenia Assessment Criteria. Hepatol. Res. 2016, 46, 951–963. [Google Scholar] [CrossRef]
- Vallet-Pichard, A.; Mallet, V.; Nalpas, B.; Verkarre, V.; Nalpas, A.; Dhalluin-Venier, V.; Fontaine, H.; Pol, S. Fib-4: An Inexpensive and Accurate Marker of Fibrosis in HCV Infection. Comparison with Liver Biopsy and Fibrotest. Hepatology 2007, 46, 32–36. [Google Scholar] [CrossRef] [PubMed]
- Hiraoka, A.; Kumada, T.; Kudo, M.; Hirooka, M.; Tsuji, K.; Itobayashi, E.; Kariyama, K.; Ishikawa, T.; Tajiri, K.; Ochi, H.; et al. Albumin-Bilirubin (Albi) Grade as Part of the Evidence-Based Clinical Practice Guideline for HCC of the Japan Society of Hepatology: A Comparison with the Liver Damage and Child-Pugh Classifications. Liver Cancer 2017, 6, 204–215. [Google Scholar] [CrossRef] [PubMed]
- Consultation, W.H.O.E. Appropriate Body-Mass Index for Asian Populations and Its Implications for Policy and Intervention Strategies. Lancet 2004, 363, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Coelho, M.P.P.; de Castro, P.; de Vries, T.P.; Colosimo, E.A.; Bezerra, J.M.T.; Rocha, G.A.; Silva, L.D. Sarcopenia in Chronic Viral Hepatitis: From Concept to Clinical Relevance. World J. Hepatol. 2023, 15, 649–665. [Google Scholar] [CrossRef]
- Nakamura, A.; Yoshimura, T.; Ichikawa, T. Liver Disease-Related Sarcopenia: A Predictor of Poor Prognosis by Accelerating Hepatic Decompensation in Advanced Chronic Liver Disease. Cureus 2023, 15, e49078. [Google Scholar] [CrossRef]
- Zheng, Y.; Wang, S.; Wu, J.; Wang, Y. Mitochondrial Metabolic Dysfunction and Non-Alcoholic Fatty Liver Disease: New Insights from Pathogenic Mechanisms to Clinically Targeted Therapy. J. Transl. Med. 2023, 21, 510. [Google Scholar] [CrossRef]
- Losasso, M.R.; Parussolo, M.L.C.; Oliveira Silva, A.; Direito, R.; Quesada, K.; Penteado Detregiachi, C.R.; Bechara, M.D.; Méndez-Sánchez, N.; Abenavoli, L.; Araújo, A.C.; et al. Unraveling the Metabolic Pathways between Metabolic-Associated Fatty Liver Disease (Mafld) and Sarcopenia. Int. J. Mol. Sci. 2025, 26, 4673. [Google Scholar] [CrossRef]
- Coen, P.M.; Musci, R.V.; Hinkley, J.M.; Miller, B.F. Mitochondria as a Target for Mitigating Sarcopenia. Front. Physiol. 2018, 9, 1883. [Google Scholar] [CrossRef]
- Hadjispyrou, S.; Giannopoulos, A.; Philippou, A.; Theos, A. Mitochondrial Dysfunction and Sarcopenic Obesity: The Role of Exercise. J. Clin. Med. 2023, 12, 5628. [Google Scholar] [CrossRef]
- Hsu, W.H.; Wang, S.Y.; Chao, Y.M.; Chang, K.V.; Han, D.S.; Lin, Y.L. Novel Metabolic and Lipidomic Biomarkers of Sarcopenia. J. Cachexia Sarcopenia Muscle 2024, 15, 2175–2186. [Google Scholar] [CrossRef]
- Chen, N.; He, X.; Feng, Y.; Ainsworth, B.E.; Liu, Y. Effects of Resistance Training in Healthy Older People with Sarcopenia: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Eur. Rev. Aging Phys. Act. 2021, 18, 23. [Google Scholar] [CrossRef]
- Ismaiel, A.; Bucsa, C.; Farcas, A.; Leucuta, D.C.; Popa, S.L.; Dumitrascu, D.L. Effects of Branched-Chain Amino Acids on Parameters Evaluating Sarcopenia in Liver Cirrhosis: Systematic Review and Meta-Analysis. Front. Nutr. 2022, 9, 749969. [Google Scholar] [CrossRef]



| Variable | No Sarcopenia | Sarcopenia | p-Value |
| Age | 54.05 | 59.70 | 0.02 * |
| PLT | 20.91 | 21.76 | 0.52 |
| ALB | 4.34 | 4.42 | 0.24 |
| AST | 46.46 | 38.44 | 0.08 |
| ALT | 59.95 | 47.98 | 0.11 |
| TBIL | 0.75 | 0.68 | 0.23 |
| Creatinine | 0.89 | 0.73 | 0.22 |
| ALBI_score | −2.98 | −3.07 | 0.15 |
| FIB4_index | 1.90 | 1.90 | 0.98 |
| BMI | 27.69 | 23.63 | 0.00 * |
| PDFF | 12.75 | 8.68 | 0.005 * |
| Stiffness | 3.65 | 2.85 | 0.00 * |
| Variable | β | p-Value | OR | 95% CI | |
| Model 1: Overall | Age | 0.04 | 0.01 | 1.05 | 1.01–1.08 |
| BMI | −0.11 | 0.07 | 0.89 | 0.80–1.01 | |
| PDFF | −0.07 | 0.03 | 0.93 | 0.87–0.99 | |
| Stiffness | −0.68 | 0.006 | 0.51 | 0.31–0.82 | |
| AST | −0.002 | 0.92 | 1.0 | 0.96–1.03 | |
| ALT | 0.01 | 0.26 | 1.01 | 0.99–1.03 | |
| Disease | −0.005 | 0.99 | 0.99 | 0.37–2.68 | |
| Model 3: MASH | Age | 0.07 | 0.006 | 1.07 | 1.02–1.13 |
| BMI | −0.01 | 0.87 | 0.99 | 0.86–1.14 | |
| PDFF | −0.10 | 0.03 | 0.91 | 0.83–0.99 | |
| Stiffness | −1.19 | 0.0009 | 0.3 | 0.15–0.61 | |
| AST | 0.007 | 0.71 | 1.01 | 0.97–1.05 | |
| ALT | 0.01 | 0.23 | 1.01 | 0.99–1.04 | |
| Model 4: Virus | Age | 0.02 | 0.62 | 1.02 | 0.96–1.08 |
| BMI | −0.48 | 0.003 | 0.62 | 0.45–0.85 | |
| PDFF | 0.04 | 0.57 | 1.04 | 0.91–1.18 | |
| Stiffness | 0.36 | 0.53 | 1.44 | 0.47–4.40 | |
| AST | −0.07 | 0.27 | 0.93 | 0.83–1.05 | |
| ALT | 0.01 | 0.72 | 1.01 | 0.94–1.10 |
| Interaction Term | β | p-Value | OR | 95% CI |
| Age: Disease | −0.04 | 0.25 | 0.96 | 0.89–1.03 |
| BMI: Disease | −0.39 | 0.02 | 0.68 | 0.49–0.93 |
| PDFF: Disease | 0.07 | 0.31 | 1.07 | 0.94–1.22 |
| Stiffness: Disease | 0.89 | 0.10 | 2.42 | 0.84–7.01 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Yasutomi, M.; Saito, K.; Araki, Y.; Sugimoto, K.; Yoshimaru, D.; Shibukawa, S.; Ishida, M. MRI-Based Assessment of Etiology-Specific Sarcopenia Phenotypes in Chronic Liver Disease: A Comparative Study of MASH and Viral Hepatitis. Diagnostics 2026, 16, 306. https://doi.org/10.3390/diagnostics16020306
Yasutomi M, Saito K, Araki Y, Sugimoto K, Yoshimaru D, Shibukawa S, Ishida M. MRI-Based Assessment of Etiology-Specific Sarcopenia Phenotypes in Chronic Liver Disease: A Comparative Study of MASH and Viral Hepatitis. Diagnostics. 2026; 16(2):306. https://doi.org/10.3390/diagnostics16020306
Chicago/Turabian StyleYasutomi, Mika, Kazuhiro Saito, Yoichi Araki, Katsutoshi Sugimoto, Daisuke Yoshimaru, Shuhei Shibukawa, and Masanori Ishida. 2026. "MRI-Based Assessment of Etiology-Specific Sarcopenia Phenotypes in Chronic Liver Disease: A Comparative Study of MASH and Viral Hepatitis" Diagnostics 16, no. 2: 306. https://doi.org/10.3390/diagnostics16020306
APA StyleYasutomi, M., Saito, K., Araki, Y., Sugimoto, K., Yoshimaru, D., Shibukawa, S., & Ishida, M. (2026). MRI-Based Assessment of Etiology-Specific Sarcopenia Phenotypes in Chronic Liver Disease: A Comparative Study of MASH and Viral Hepatitis. Diagnostics, 16(2), 306. https://doi.org/10.3390/diagnostics16020306






