Molecular Diagnostic Review of Diffuse Large B-Cell Lymphoma and Its Tumor Microenvironment
Abstract
1. Introduction
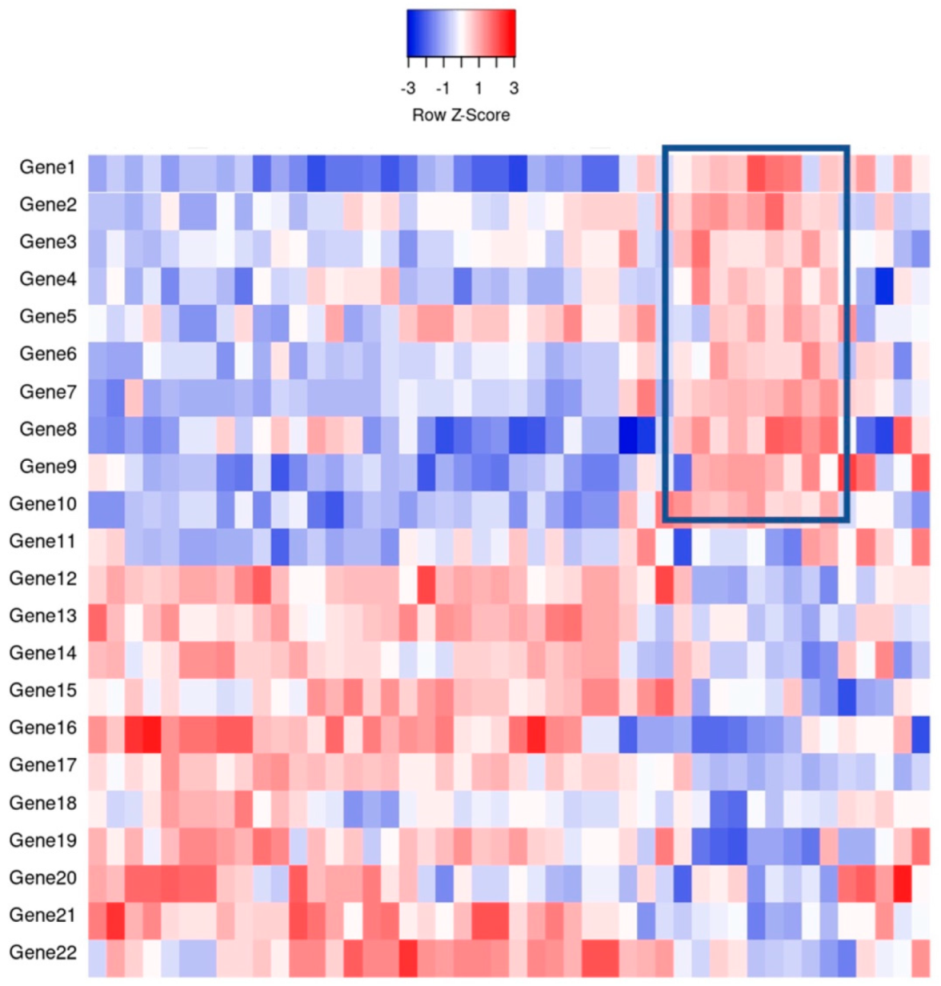
2. Gene Expression Profiling
3. Immunohistochemistry
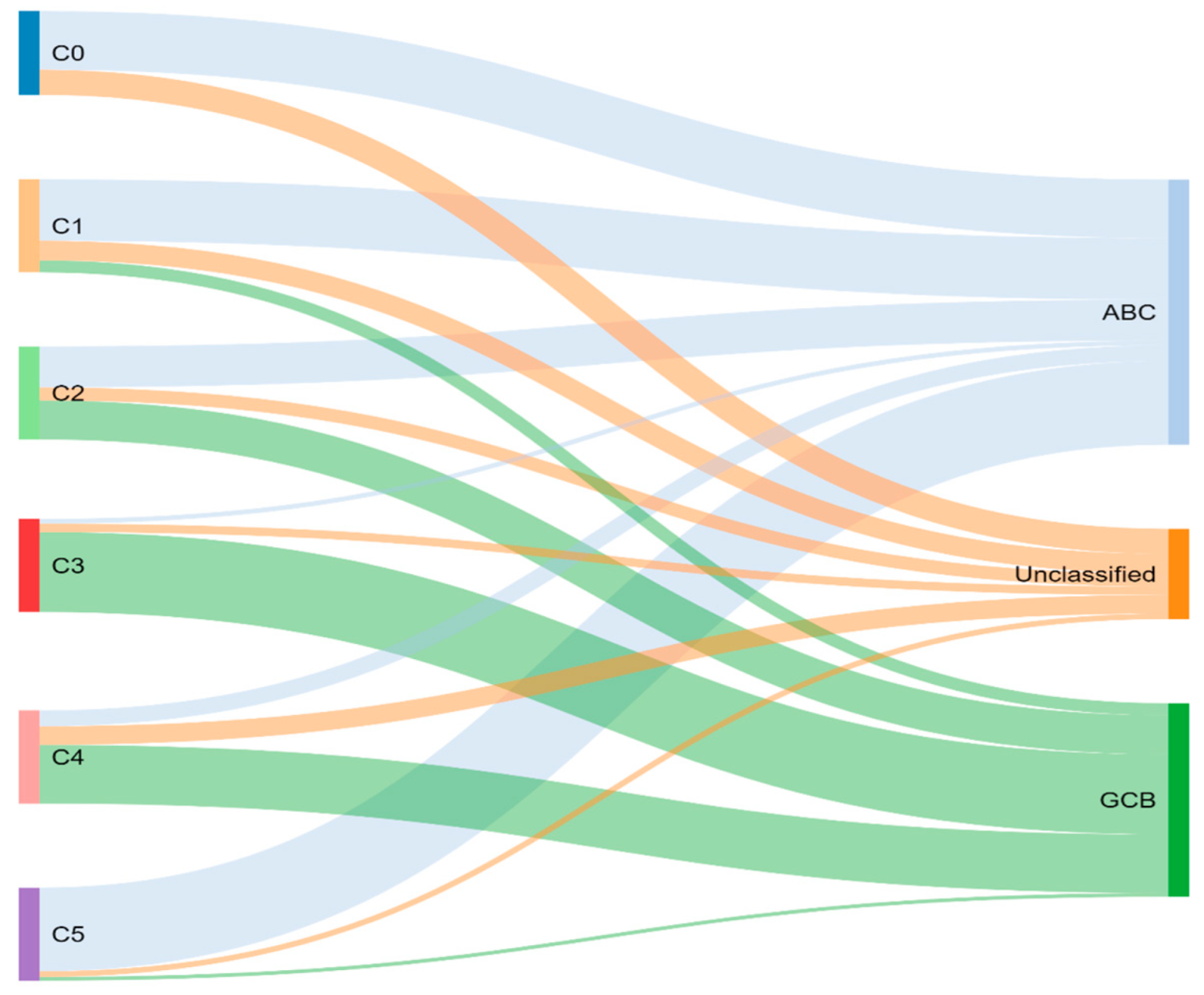
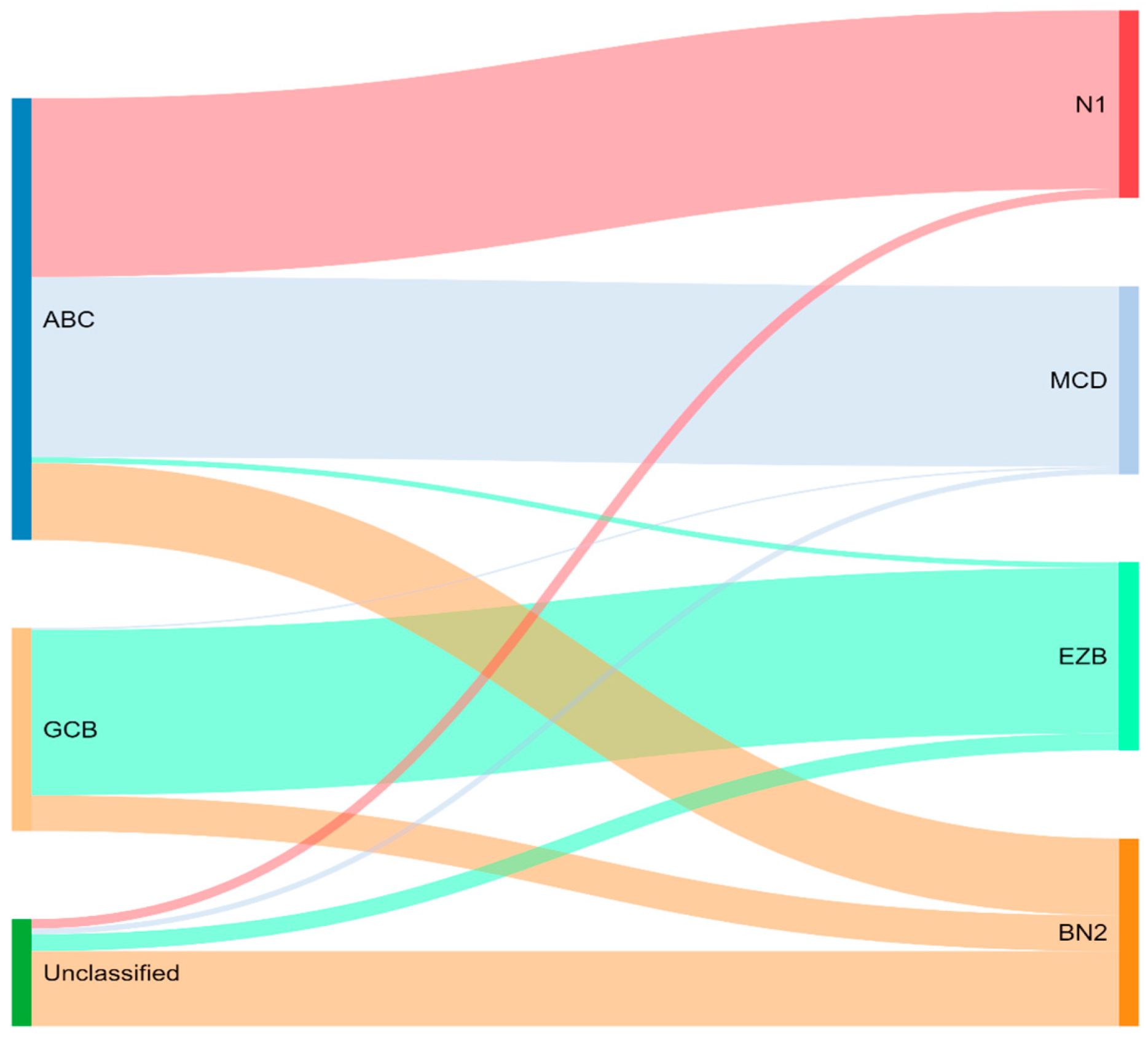
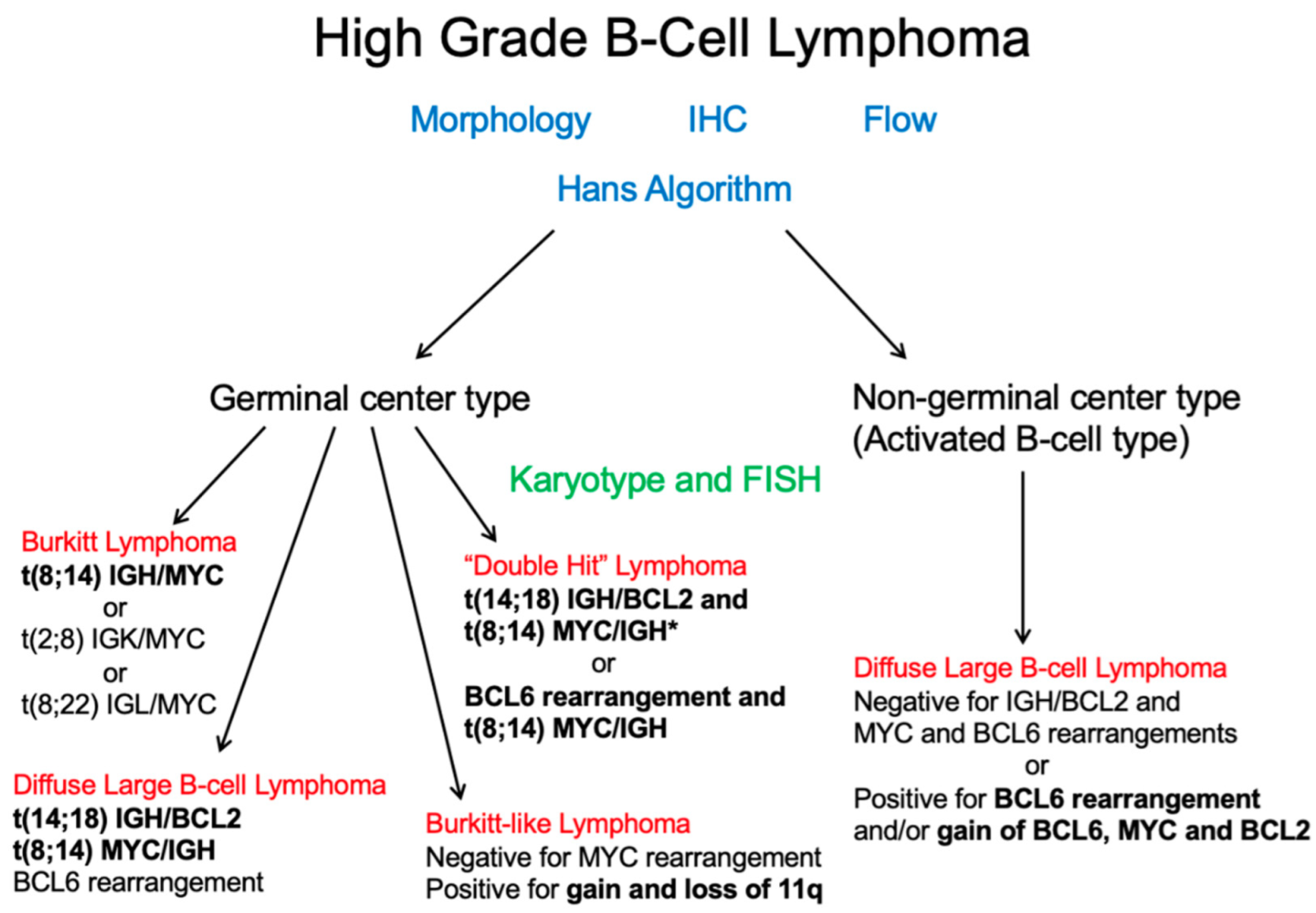
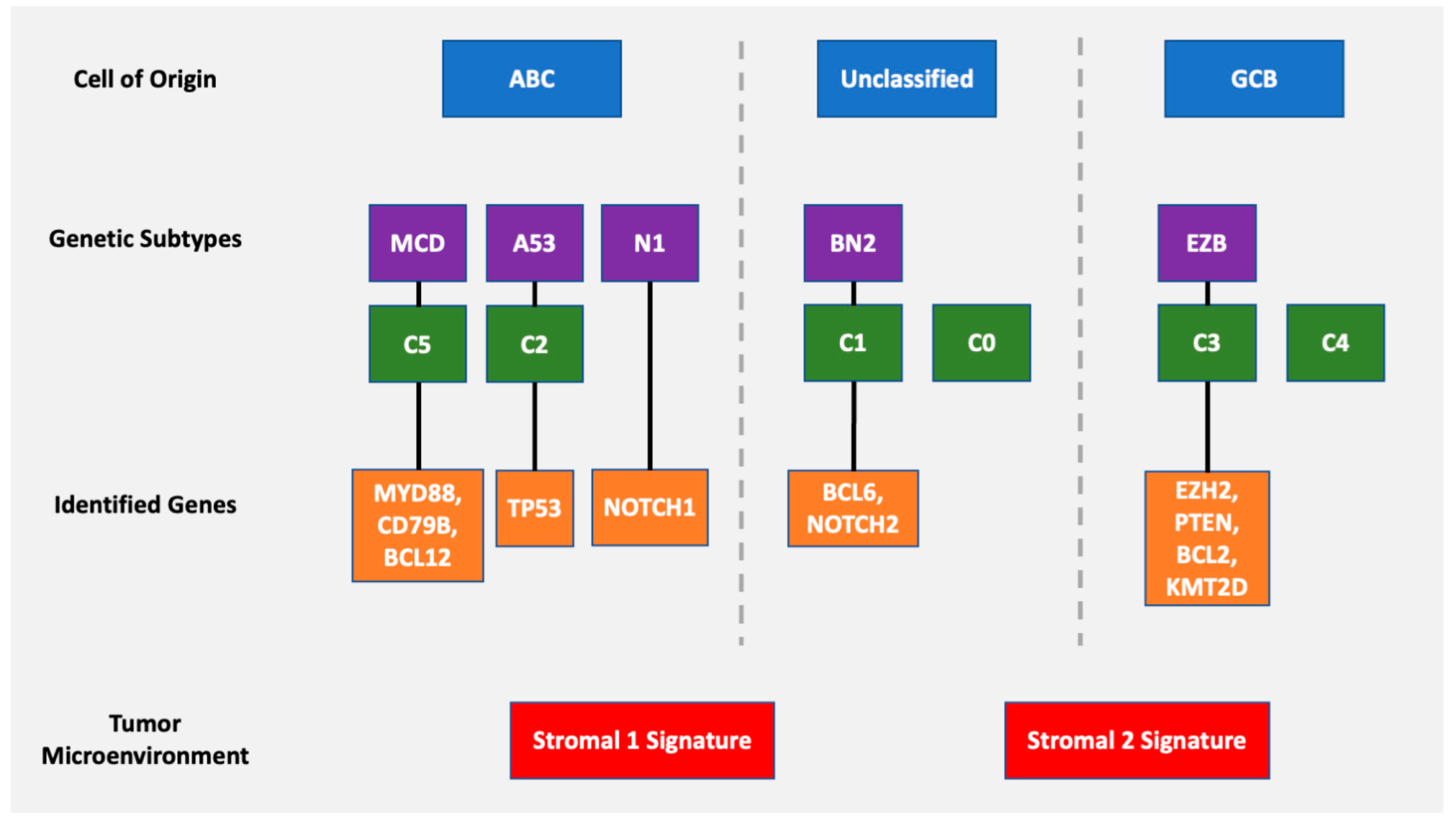
4. Genetic Classification
Cytogenetics and/or Fluorescence In-Situ Hybridization
5. Microenvironment
6. Minimal Diagnostic Criteria
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Golub, T.R.; Slonim, D.K.; Tamayo, P.; Huard, C.; Gaasenbeek, M.; Mesirov, J.P.; Coller, H.; Loh, M.L.; Downing, J.R.; Caligiuri, M.A.; et al. Molecular Classification of Cancer: Class Discovery and Class Prediction by Gene Expression Monitoring. Science 1999, 286, 531–537. [Google Scholar] [CrossRef] [PubMed]
- Alizadeh, A.A.; Eisen, M.; Davis, R.; Ma, C.; Sabet, H.; Tran, T.; Powell, J.I.; Yang, L.; Marti, G.; Moore, D.T.; et al. The Lymphochip: A Specialized cDNA Microarray for the Genomic-scale Analysis of Gene Expression in Normal and Malignant Lymphocytes. Cold Spring Harb. Symp. Quant. Biol. 1999, 64, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Alizadeh, A.A.; Eisen, M.B.; Davis, R.E.; Ma, C.; Lossos, I.S.; Rosenwald, A.; Boldrick, J.C.; Sabet, H.; Tran, T.; Yu, X.; et al. Distinct types of diffuse large B-cell lymphoma identified by gene expression profiling. Nature 2000, 403, 503–511. [Google Scholar] [CrossRef]
- Swerdlow, S.H.; Campo, E.; Harris, N.L.; Jaffe, E.S.; Pileri, S.A.; Stein, H.; Thiele, J. (Eds.) WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues, Revised 4th ed.; IARC: Lyon, France, 2017.
- Shipp, M.A.; Ross, K.N.; Tamayo, P.; Weng, A.; Kutok, J.L.; Aguiar, R.C.; Gaasenbeek, M.; Angelo, M.; Reich, M.; Pinkus, G.S.; et al. Diffuse large B-cell lymphoma outcome prediction by gene-expression profiling and supervised machine learning. Nat. Med. 2002, 8, 68–74. [Google Scholar] [CrossRef]
- Rosenwald, A.; Wright, G.; Chan, W.C.; Connors, J.M.; Campo, E.; Fisher, R.I.; Gascoyne, R.D.; Muller-Hermelink, H.K.; Smeland, E.B.; Giltnane, J.M.; et al. The Use of Molecular Profiling to Predict Survival after Chemotherapy for Diffuse Large-B-Cell Lymphoma. N. Engl. J. Med. 2002, 346, 1937–1947. [Google Scholar] [CrossRef]
- Lenz, G.; Wright, G.; Dave, S.S.; Xiao, W.; Powell, J.; Zhao, H.; Xu, W.; Tan, B.; Goldschmidt, N.; Iqbal, J.; et al. Stromal Gene Signatures in Large-B-Cell Lymphomas. N. Engl. J. Med. 2008, 359, 2313–2323. [Google Scholar] [CrossRef]
- Scott, D.W.; Wright, G.W.; Williams, P.M.; Lih, C.-J.; Walsh, W.; Jaffe, E.; Rosenwald, A.; Campo, E.; Chan, W.C.; Connors, J.M.; et al. Determining cell-of-origin subtypes of diffuse large B-cell lymphoma using gene expression in formalin-fixed paraffin-embedded tissue. Blood 2014, 123, 1214–1217. [Google Scholar] [CrossRef]
- Balasubramanian, S.; Wang, S.; Major, C.; Hodkinson, B.; Schaffer, M.; Sehn, L.H.; Johnson, P.; Zinzani, P.L.; Carey, J.; Shreeve, S.M.; et al. Comparison of immunohistochemistry and gene expression profiling subtyping for diffuse large B-cell lymphoma in the phase III clinical trial of R-CHOP ± ibrutinib. Br. J. Haematol. 2021, 194, 83–91. [Google Scholar] [CrossRef]
- Ta, R.; Santini, C.; Gou, P.; Lee, G.; Tai, Y.C.; O’Brien, C.; Fontecha, M.; Grant, C.; Bacon, L.; Finn, S.; et al. Molecular Subtyping of Diffuse Large B-Cell Lymphoma Using a Novel Quantitative RT-PCR Assay. J. Mol. Diagn. 2020, 23, 323–340. [Google Scholar] [CrossRef]
- Reddy, A.; Zhang, J.; Davis, N.S.; Moffitt, A.; Love, C.L.; Waldrop, A.; Leppä, S.; Pasanen, A.; Meriranta, L.; Karjalainen-Lindsberg, M.-L.; et al. Genetic and Functional Drivers of Diffuse Large B Cell Lymphoma. Cell 2017, 171, 481–494.e15. [Google Scholar] [CrossRef] [PubMed]
- Knittel, G.; Liedgens, P.; Korovkina, D.; Seeger, J.M.; Al-Baldawi, Y.; Al-Maarri, M.; Fritz, C.; Vlantis, K.; Bezhanova, S.; Scheel, A.H.; et al. B-cell–specific conditional expression of Myd88p.L252P leads to the development of diffuse large B-cell lymphoma in mice. Blood 2016, 127, 2732–2741. [Google Scholar] [CrossRef] [PubMed]
- Souroullas, G.P.; Jeck, W.R.; Parker, J.S.; Simon, J.M.; Liu, J.-Y.; Paulk, J.; Xiong, J.; Clark, K.S.; Fedoriw, Y.; Qi, J.; et al. An oncogenic Ezh2 mutation induces tumors through global redistribution of histone 3 lysine 27 trimethylation. Nat. Med. 2016, 22, 632–640. [Google Scholar] [CrossRef] [PubMed]
- Chapuy, B.; Stewart, C.; Dunford, A.J.; Kim, J.; Kamburov, A.; Redd, R.A.; Lawrence, M.S.; Roemer, M.G.M.; Li, A.J.; Ziepert, M.; et al. Molecular subtypes of diffuse large B cell lymphoma are associated with distinct pathogenic mechanisms and outcomes. Nat. Med. 2018, 24, 679–690. [Google Scholar] [CrossRef]
- Schmitz, R.; Wright, G.W.; Huang, D.W.; Johnson, C.A.; Phelan, J.D.; Wang, J.Q.; Roulland, S.; Kasbekar, M.; Young, R.M.; Shaffer, A.L.; et al. Genetics and Pathogenesis of Diffuse Large B-Cell Lymphoma. N. Engl. J. Med. 2018, 378, 1396–1407. [Google Scholar] [CrossRef]
- Nowakowski, G.S.; Chiappella, A.; Witzig, T.E.; Spina, M.; Gascoyne, R.D.; Zhang, L.; Flament, J.; Repici, J.; Vitolo, U. ROBUST: Lenalidomide-R-CHOP versus placebo-R-CHOP in previously untreated ABC-type diffuse large B-cell lymphoma. Futur. Oncol. 2016, 12, 1553–1563. [Google Scholar] [CrossRef]
- Wilson, W.H.; Young, R.M.; Schmitz, R.; Yang, Y.; Pittaluga, S.; Wright, G.; Lih, C.-J.; Williams, P.M.; Shaffer, A.L.; Gerecitano, J.; et al. Targeting B cell receptor signaling with ibrutinib in diffuse large B cell lymphoma. Nat. Med. 2015, 21, 922–926. [Google Scholar] [CrossRef]
- Younes, A.; Sehn, L.H.; Johnson, P.; Zinzani, P.L.; Hong, X.; Zhu, J.; Patti, C.; Belada, D.; Samoilova, O.; Suh, C.; et al. Randomized Phase III Trial of Ibrutinib and Rituximab Plus Cyclophosphamide, Doxorubicin, Vincristine, and Prednisone in Non-Germinal Center B-Cell Diffuse Large B-Cell Lymphoma. J. Clin. Oncol. 2019, 37, 1285–1295. [Google Scholar] [CrossRef]
- Wilson, W.H.; Wright, G.W.; Huang, D.W.; Hodkinson, B.; Balasubramanian, S.; Fan, Y.; Vermeulen, J.; Shreeve, M.; Staudt, L.M. Effect of ibrutinib with R-CHOP chemotherapy in genetic subtypes of DLBCL. Cancer Cell 2021, 39, 1643–1653.e3. [Google Scholar] [CrossRef]
- Landsburg, D.J.; Hughes, M.E.; Koike, A.; Bond, D.; Maddocks, K.J.; Guo, L.; Winter, A.M.; Hill, B.T.; Ondrejka, S.L.; Hsi, E.D.; et al. Outcomes of patients with relapsed/refractory double-expressor B-cell lymphoma treated with ibrutinib monotherapy. Blood Adv. 2019, 3, 132–135. [Google Scholar] [CrossRef]
- Davies, A.; Cummin, T.E.; Barrans, S.; Maishman, T.; Mamot, C.; Novak, U.; Caddy, J.; Stanton, L.; Kazmi-Stokes, S.; McMillan, A.; et al. Gene-expression profiling of bortezomib added to standard chemoimmunotherapy for diffuse large B-cell lymphoma (REMoDL-B): An open-label, randomised, phase 3 trial. Lancet Oncol. 2019, 20, 649–662. [Google Scholar] [CrossRef]
- Hans, C.P.; Weisenburger, D.D.; Greiner, T.C.; Gascoyne, R.D.; Delabie, J.; Ott, G.; Müller-Hermelink, H.K.; Campo, E.; Braziel, R.M.; Jaffe, E.S.; et al. Confirmation of the molecular classification of diffuse large B-cell lymphoma by immunohistochemistry using a tissue microarray. Blood 2004, 103, 275–282. [Google Scholar] [CrossRef]
- Choi, W.W.L.; Weisenburger, D.D.; Greiner, T.C.; Piris, M.A.; Banham, A.H.; Delabie, J.; Braziel, R.M.; Geng, H.; Iqbal, J.; Lenz, G.; et al. A New Immunostain Algorithm Classifies Diffuse Large B-Cell Lymphoma into Molecular Subtypes with High Accuracy. Clin. Cancer Res. 2009, 15, 5494–5502. [Google Scholar] [CrossRef]
- Muris, J.J.F.; Meijer, C.J.L.M.; Vos, W.; Van Krieken, J.H.J.M.; Jiwa, N.M.; Ossenkoppele, G.J.; Oudejans, J.J. Immunohistochemical profiling based on Bcl-2, CD10 and MUM1 expression improves risk stratification in patients with primary nodal diffuse large B cell lymphoma. J. Pathol. 2006, 208, 714–723. [Google Scholar] [CrossRef]
- Nyman, H.; Jerkeman, M.; Karjalainen-Lindsberg, M.-L.; Banham, A.; Leppä, S. Prognostic impact of activated B-cell focused classification in diffuse large B-cell lymphoma patients treated with R-CHOP. Mod. Pathol. 2009, 22, 1094–1101. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Opinto, G.; Vegliante, M.C.; Negri, A.; Skrypets, T.; Loseto, G.; Pileri, S.A.; Guarini, A.; Ciavarella, S. The Tumor Microenvironment of DLBCL in the Computational Era. Front. Oncol. 2020, 10, 351. [Google Scholar] [CrossRef] [PubMed]
- Keane, C.; Gill, D.; Vari, F.; Cross, D.; Griffiths, L.; Gandhi, M. CD4+ Tumor infiltrating lymphocytes are prognostic and independent of R-IPI in patients with DLBCL receiving R-CHOP chemo-immunotherapy. Am. J. Hematol. 2013, 88, 273–276. [Google Scholar] [CrossRef]
- Meyer, P.N.; Fu, K.; Greiner, T.; Smith, L.; Delabie, J.; Gascoyne, R.; Ott, G.; Rosenwald, A.; Braziel, R.; Campo, E.; et al. The Stromal Cell Marker SPARC Predicts for Survival in Patients With Diffuse Large B-Cell Lymphoma Treated With Rituximab. Am. J. Clin. Pathol. 2011, 135, 54–61. [Google Scholar] [CrossRef]
- Niitsu, N.; Tamaru, J.-I.; Yoshino, T.; Nakamura, N.; Nakamura, S.; Ohshima, K.; Nakamine, H.; Okamoto, M. A study on nm23-H1 expression in diffuse large B-cell lymphoma that was treated with CyclOBEAP plus rituximab therapy. Ann. Hematol. 2011, 90, 185–192. [Google Scholar] [CrossRef]
- Croci, G.; Au-Yeung, R.; Reinke, S.; Staiger, A.; Koch, K.; Oschlies, I.; Richter, J.; Poeschel, V.; Held, G.; Loeffler, M.; et al. SPARC-positive macrophages are the superior prognostic factor in the microenvironment of diffuse large B-cell lymphoma and independent of MYC rearrangement and double-/triple-hit status. Ann. Oncol. 2021, 32, 1400–1409. [Google Scholar] [CrossRef]
- Keane, C.; Law, S.C.; Gould, C.; Birch, S.; Sabdia, M.B.; De Long, L.M.; Thillaiyampalam, G.; Abro, E.; Tobin, J.W.; Tan, X.; et al. LAG3: A novel immune checkpoint expressed by multiple lymphocyte subsets in diffuse large B-cell lymphoma. Blood Adv. 2020, 4, 1367–1377. [Google Scholar] [CrossRef]
- Green, T.M.; Young, K.H.; Visco, C.; Xu-Monette, Z.Y.; Orazi, A.; Go, R.S.; Nielsen, O.; Gadeberg, O.V.; Mourits-Andersen, T.; Frederiksen, M.; et al. Immunohistochemical Double-Hit Score Is a Strong Predictor of Outcome in Patients With Diffuse Large B-Cell Lymphoma Treated With Rituximab Plus Cyclophosphamide, Doxorubicin, Vincristine, and Prednisone. J. Clin. Oncol. 2012, 30, 3460–3467. [Google Scholar] [CrossRef] [PubMed]
- Johnson, N.A.; Slack, G.W.; Savage, K.J.; Connors, J.M.; Ben-Neriah, S.; Rogic, S.; Scott, D.W.; Tan, K.L.; Steidl, C.; Sehn, L.H.; et al. Concurrent Expression of MYC and BCL2 in Diffuse Large B-Cell Lymphoma Treated With Rituximab Plus Cyclophosphamide, Doxorubicin, Vincristine, and Prednisone. J. Clin. Oncol. 2012, 30, 3452–3459. [Google Scholar] [CrossRef]
- Hu, S.; Xu-Monette, Z.Y.; Tzankov, A.; Green, T.; Wu, L.; Balasubramanyam, A.; Liu, W.-M.; Visco, C.; Li, Y.; Miranda, R.; et al. MYC/BCL2 protein coexpression contributes to the inferior survival of activated B-cell subtype of diffuse large B-cell lymphoma and demonstrates high-risk gene expression signatures: A report from The International DLBCL Rituximab-CHOP Consortium Program. Blood 2013, 121, 4021–4031. [Google Scholar] [CrossRef] [PubMed]
- Staiger, A.M.; Ziepert, M.; Horn, H.; Scott, D.W.; Barth, T.F.; Bernd, H.-W.; Feller, A.C.; Klapper, W.; Szczepanowski, M.; Hummel, M.; et al. Clinical Impact of the Cell-of-Origin Classification and the MYC/BCL2 Dual Expresser Status in Diffuse Large B-Cell Lymphoma Treated Within Prospective Clinical Trials of the German High-Grade Non-Hodgkin’s Lymphoma Study Group. J. Clin. Oncol. 2017, 35, 2515–2526. [Google Scholar] [CrossRef]
- Ennishi, D.; Jiang, A.; Boyle, M.; Collinge, B.; Grande, B.M.; Ben-Neriah, S.; Rushton, C.; Tang, J.; Thomas, N.; Slack, G.W.; et al. Double-Hit Gene Expression Signature Defines a Distinct Subgroup of Germinal Center B-Cell-Like Diffuse Large B-Cell Lymphoma. J. Clin. Oncol. 2019, 37, 190–201. [Google Scholar] [CrossRef] [PubMed]
- Sha, C.; Barrans, S.; Cucco, F.; Bentley, M.A.; Care, M.A.; Cummin, T.; Kennedy, H.; Thompson, J.S.; Uddin, R.; Worrillow, L.; et al. Molecular High-Grade B-Cell Lymphoma: Defining a Poor-Risk Group That Requires Different Approaches to Therapy. J. Clin. Oncol. 2019, 37, 202–212. [Google Scholar] [CrossRef]
- Ennishi, D.; Hsi, E.D.; Steidl, C.; Scott, D.W. Toward a New Molecular Taxonomy of Diffuse Large B-cell Lymphoma. Cancer Discov. 2020, 10, 1267–1281. [Google Scholar] [CrossRef]
- Wright, G.W.; Huang, D.W.; Phelan, J.D.; Coulibaly, Z.A.; Roulland, S.; Young, R.M.; Wang, J.Q.; Schmitz, R.; Morin, R.; Tang, J.; et al. A Probabilistic Classification Tool for Genetic Subtypes of Diffuse Large B Cell Lymphoma with Therapeutic Implications. Cancer Cell 2020, 37, 551–568.e14. [Google Scholar] [CrossRef]
- Matsushita, H.; Vesely, M.D.; Koboldt, D.C.; Rickert, C.G.; Uppaluri, R.; Magrini, V.J.; Arthur, C.D.; White, J.M.; Chen, Y.S.; Shea, L.K.; et al. Cancer exome analysis reveals a T-cell-dependent mechanism of cancer immunoediting. Nature 2012, 482, 400–404. [Google Scholar] [CrossRef]
- Küppers, R.; Dührsen, U.; Hansmann, M.-L. Pathogenesis, diagnosis, and treatment of composite lymphomas. Lancet Oncol. 2014, 15, e435–e446. [Google Scholar] [CrossRef]
- Green, M.R.; Gentles, A.J.; Nair, R.V.; Irish, J.; Kihira, S.; Liu, C.L.; Kela, I.; Hopmans, E.S.; Myklebust, J.H.; Ji, H.; et al. Hierarchy in somatic mutations arising during genomic evolution and progression of follicular lymphoma. Blood 2013, 121, 1604–1611. [Google Scholar] [CrossRef] [PubMed]
- González-Rincón, J.; Méndez, M.; Gómez, S.; García, J.F.; Martin-Acosta, P.; Bellas, C.; Pedrosa, L.; Rodríguez-Pinilla, S.M.; Camacho, F.I.; Quero, C.; et al. Unraveling transformation of follicular lymphoma to diffuse large B-cell lymphoma. PLoS ONE 2019, 14, e0212813. [Google Scholar] [CrossRef] [PubMed]
- Tzankov, A.; Xu-Monette, Z.Y.; Gerhard, M.; Visco, C.; Dirnhofer, S.; Gisin, N.; Dybkaer, K.; Orazi, A.; Bhagat, G.; Richards, K.L.; et al. Rearrangements of MYC gene facilitate risk stratification in diffuse large B-cell lymphoma patients treated with rituximab-CHOP. Mod. Pathol. 2013, 27, 958–971. [Google Scholar] [CrossRef] [PubMed]
- Dojcinov, D.S.; Wilkins, D.B.; Calaminici, D.M. Standards for Specialist Laboratory Integration and Dataset for the Histopathological Reporting of Lymphomas; Technical Report; The Royal College of Pathologists: London, UK, 2015. [Google Scholar]
- Duncavage, E.; Advani, R.H.; Agosti, S.; Foulis, P.; Gibson, C.; Kang, L.; Khoury, J.D.; Medeiros, L.J.; Ohgami, R.S.; O’Malley, D.P.; et al. Template for Reporting Results of Biomarker Testing of Specimens From Patients with Diffuse Large B-Cell Lymphoma, Not Otherwise Specified (NOS). College of American Pathologists: Northfield, IL, USA, 2014. Available online: https://documents.cap.org/protocols/cp-diffuse-large-b-cell-lymphoma-biomarker-17-1002.pdf (accessed on 28 February 2022).
- Scott, D.W.; King, R.L.; Staiger, A.M.; Ben-Neriah, S.; Jiang, A.; Horn, H.; Mottok, A.; Farinha, P.; Slack, G.W.; Ennishi, D.; et al. High-grade B-cell lymphoma with MYC and BCL2 and/or BCL6 rearrangements with diffuse large B-cell lymphoma morphology. Blood 2018, 131, 2060–2064. [Google Scholar] [CrossRef]
- Bergman, C.C.; Cuillière-Dartigues, P.; Baia, M.; Briere, J.; Delarue, R.; Canioni, D.; Salles, G.; Parrens, M.; Belhadj, K.; Fabiani, B.; et al. MYC-IG rearrangements are negative predictors of survival in DLBCL patients treated with immunochemotherapy: A GELA/LYSA study. Blood 2015, 126, 2466–2474. [Google Scholar] [CrossRef]
- Pillai, R.K.; Sathanoori, M.; Van Oss, S.B.; Swerdlow, S.H. Double-hit B-cell Lymphomas with BCL6 and MYC Translocations Are Aggressive, Frequently Extranodal Lymphomas Distinct From BCL2 Double-hit B-cell Lymphomas. Am. J. Surg. Pathol. 2013, 37, 323–332. [Google Scholar] [CrossRef]
- Hinshaw, D.C.; Shevde, L.A. The tumor microenvironment innately modulates cancer progression. Cancer Res. 2019, 79, 4557–4566. [Google Scholar] [CrossRef]
- Ingravallo, G.; Tamma, R.; Opinto, G.; Annese, T.; Gaudio, F.; Specchia, G.; Perrone, T.; Musto, P.; Cazzato, G.; Bellitti, E.; et al. The Effect of the Tumor Microenvironment on Lymphoid Neoplasms Derived from B Cells. Diagnostics 2022, 12, 573. [Google Scholar] [CrossRef]
- Höpken, U.E.; Rehm, A. Targeting the Tumor Microenvironment of Leukemia and Lymphoma. Trends Cancer 2019, 5, 351–364. [Google Scholar] [CrossRef]
- Kant, S.; Kumar, A.; Singh, S.M. Tumor growth retardation and chemosensitizing action of fatty acid synthase inhibitor orlistat on T cell lymphoma: Implication of reconstituted tumor microenvironment and multidrug resistance phenotype. Biochim. Biophys. Acta (BBA) Gen. Subj. 2014, 1840, 294–302. [Google Scholar] [CrossRef] [PubMed]
- Mourcin, F.; Pangault, C.; Amin-Ali, R.; Amé-Thomas, P.; Tarte, K. Stromal Cell Contribution to Human Follicular Lymphoma Pathogenesis. Front. Immunol. 2012, 3, 280. [Google Scholar] [CrossRef]
- Cayrol, F.; Sterle, H.A.; Diaz Flaqué, M.C.; Barreiro Arcos, M.L.; Cremaschi, G.A. Non-genomic Actions of Thyroid Hormones Regulate the Growth and Angiogenesis of T Cell Lymphomas. Front. Endocrinol. 2019, 10, 63. [Google Scholar] [CrossRef]
- Cioroianu, A.I.; Stinga, P.I.; Sticlaru, L.; Cioplea, M.D.; Nichita, L.; Popp, C.; Staniceanu, F. Tumor Microenvironment in Diffuse Large B-Cell Lymphoma: Role and Prognosis. Anal. Cell. Pathol. 2019, 2019, 8586354. [Google Scholar] [CrossRef] [PubMed]
- Brandt, S.; Montagna, C.; Georgis, A.; Schüffler, P.J.; Bühler, M.M.; Seifert, B.; Thiesler, T.; Curioni-Fontecedro, A.; Hegyi, I.; Dehler, S.; et al. The combined expression of the stromal markers fibronectin and SPARC improves the prediction of survival in diffuse large B-cell lymphoma. Exp. Hematol. Oncol. 2013, 2, 27. [Google Scholar] [CrossRef] [PubMed]
- Pileri, S.; Tripodo, C.; Melle, F.; Motta, G.; Tabanelli, V.; Fiori, S.; Vegliante, M.; Mazzara, S.; Ciavarella, S.; Derenzini, E. Predictive and Prognostic Molecular Factors in Diffuse Large B-Cell Lymphomas. Cells 2021, 10, 675. [Google Scholar] [CrossRef]
- Ciavarella, S.; Vegliante, M.; Fabbri, M.; De Summa, S.; Melle, F.; Motta, G.; De Iuliis, V.; Opinto, G.; Enjuanes, A.; Rega, S.; et al. Dissection of DLBCL microenvironment provides a gene expression-based predictor of survival applicable to formalin-fixed paraffin-embedded tissue. Ann. Oncol. 2018, 29, 2363–2370. [Google Scholar] [CrossRef]
- Steen, C.B.; Liu, C.L.; Alizadeh, A.A.; Newman, A.M. Profiling Cell Type Abundance and Expression in Bulk Tissues with CIBERSORTx. Antimicrob. Pept. 2020, 2117, 135–157. [Google Scholar] [CrossRef]
- Ghorab, D.S.-D.; Helaly, A.M.; El Mahdi, H.S.; Khatatbeh, M.; Ibrahiem, A.T. Prognostic Role of Tumor Microenvironment in DLBCL and Relation to Patients’ Clinical Outcome: A Clinical and Immunohistochemical Study. Anal. Cell. Pathol. 2022, 2022, 9993496. [Google Scholar] [CrossRef]
- Solimando, A.; Annese, T.; Tamma, R.; Ingravallo, G.; Maiorano, E.; Vacca, A.; Specchia, G.; Ribatti, D. New Insights into Diffuse Large B-Cell Lymphoma Pathobiology. Cancers 2020, 12, 1869. [Google Scholar] [CrossRef]
- Kotlov, N.; Bagaev, A.; Revuelta, M.V.; Phillip, J.M.; Cacciapuoti, M.T.; Antysheva, Z.; Svekolkin, V.; Tikhonova, E.; Miheecheva, N.; Kuzkina, N.; et al. Clinical and Biological Subtypes of B-cell Lymphoma Revealed by Microenvironmental Signatures. Cancer Discov. 2021, 11, 1468–1489. [Google Scholar] [CrossRef]
- Swerdlow, S.H.; Campo, E.; Pileri, S.A.; Harris, N.L.; Stein, H.; Siebert, R.; Advani, R.; Ghielmini, M.; Salles, G.A.; Zelenetz, A.D.; et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood 2016, 127, 2375–2390. [Google Scholar] [CrossRef] [PubMed]
- King, J.F.; Lam, J.T. A Practical Approach to Diagnosis of B-Cell Lymphomas with Diffuse Large Cell Morphology. Arch. Pathol. Lab. Med. 2020, 144, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Disanto, M.G.; Ambrosio, M.R.; Rocca, B.J.; Ibrahim, H.A.H.; Leoncini, L.; Naresh, K.N. Optimal Minimal Panels of Immunohistochemistry for Diagnosis of B-Cell Lymphoma for Application in Countries With Limited Resources and for Triaging Cases Before Referral to Specialist Centers. Am. J. Clin. Pathol. 2016, 145, 687–695. [Google Scholar] [CrossRef] [PubMed]
- Riedell, P.A.; Smith, S.M. Double hit and double expressors in lymphoma: Definition and treatment. Cancer 2018, 124, 4622–4632. [Google Scholar] [CrossRef]
- Dunleavy, K.; Fanale, M.A.; Abramson, J.S.; Noy, A.; Caimi, P.F.; Pittaluga, S.; Parekh, S.; Lacasce, A.; Hayslip, J.W.; Jagadeesh, D.; et al. Dose-adjusted EPOCH-R (etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin, and rituximab) in untreated aggressive diffuse large B-cell lymphoma with MYC rearrangement: A prospective, multicentre, single-arm phase 2 study. Lancet Haematol. 2018, 5, e609–e617. [Google Scholar] [CrossRef]
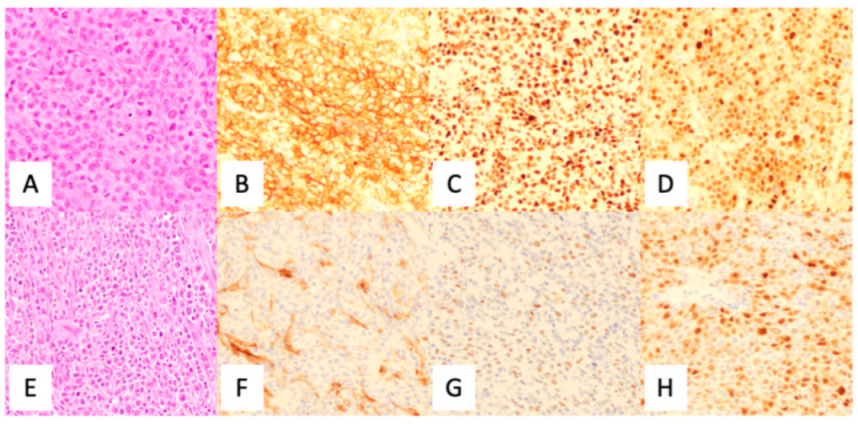

| Algorithm | Antibodies | Comments |
|---|---|---|
| Hans [23] | CD10, BCL6, MUM1 | ≥30% staining to be considered positive |
| Choi [24] | CD10, BCL6, MUM1, GCET, FOXP1 | ≥80% GCET, FOXP1, MUM1 ≥30% staining for CD10 and BCL6 |
| Muris [25] | BCL2, CD10, MUM1 | BCL2 ≥ 50% and CD10 or BCL6 ≥ 30% |
| Nyman [26] | MUM1, FOXP1 | ≥30% staining to be considered positive |
| Diagnostic Technology | Advantages | Disadvantages | Subclassification Methods/Key Studies |
|---|---|---|---|
| Immunohistochemistry |
|
|
|
| Gene expression profiling |
|
|
|
| Cytogenetics and fluorescence in-situ hybridization (FISH) |
|
|
|
| Next generation sequencing |
|
|
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ta, R.; Yang, D.; Hirt, C.; Drago, T.; Flavin, R. Molecular Diagnostic Review of Diffuse Large B-Cell Lymphoma and Its Tumor Microenvironment. Diagnostics 2022, 12, 1087. https://doi.org/10.3390/diagnostics12051087
Ta R, Yang D, Hirt C, Drago T, Flavin R. Molecular Diagnostic Review of Diffuse Large B-Cell Lymphoma and Its Tumor Microenvironment. Diagnostics. 2022; 12(5):1087. https://doi.org/10.3390/diagnostics12051087
Chicago/Turabian StyleTa, Robert, David Yang, Christian Hirt, Thomas Drago, and Richard Flavin. 2022. "Molecular Diagnostic Review of Diffuse Large B-Cell Lymphoma and Its Tumor Microenvironment" Diagnostics 12, no. 5: 1087. https://doi.org/10.3390/diagnostics12051087
APA StyleTa, R., Yang, D., Hirt, C., Drago, T., & Flavin, R. (2022). Molecular Diagnostic Review of Diffuse Large B-Cell Lymphoma and Its Tumor Microenvironment. Diagnostics, 12(5), 1087. https://doi.org/10.3390/diagnostics12051087







