Results of Numerical Modeling of Blood Flow in the Internal Jugular Vein Exhibiting Different Types of Strictures
Abstract
1. Introduction
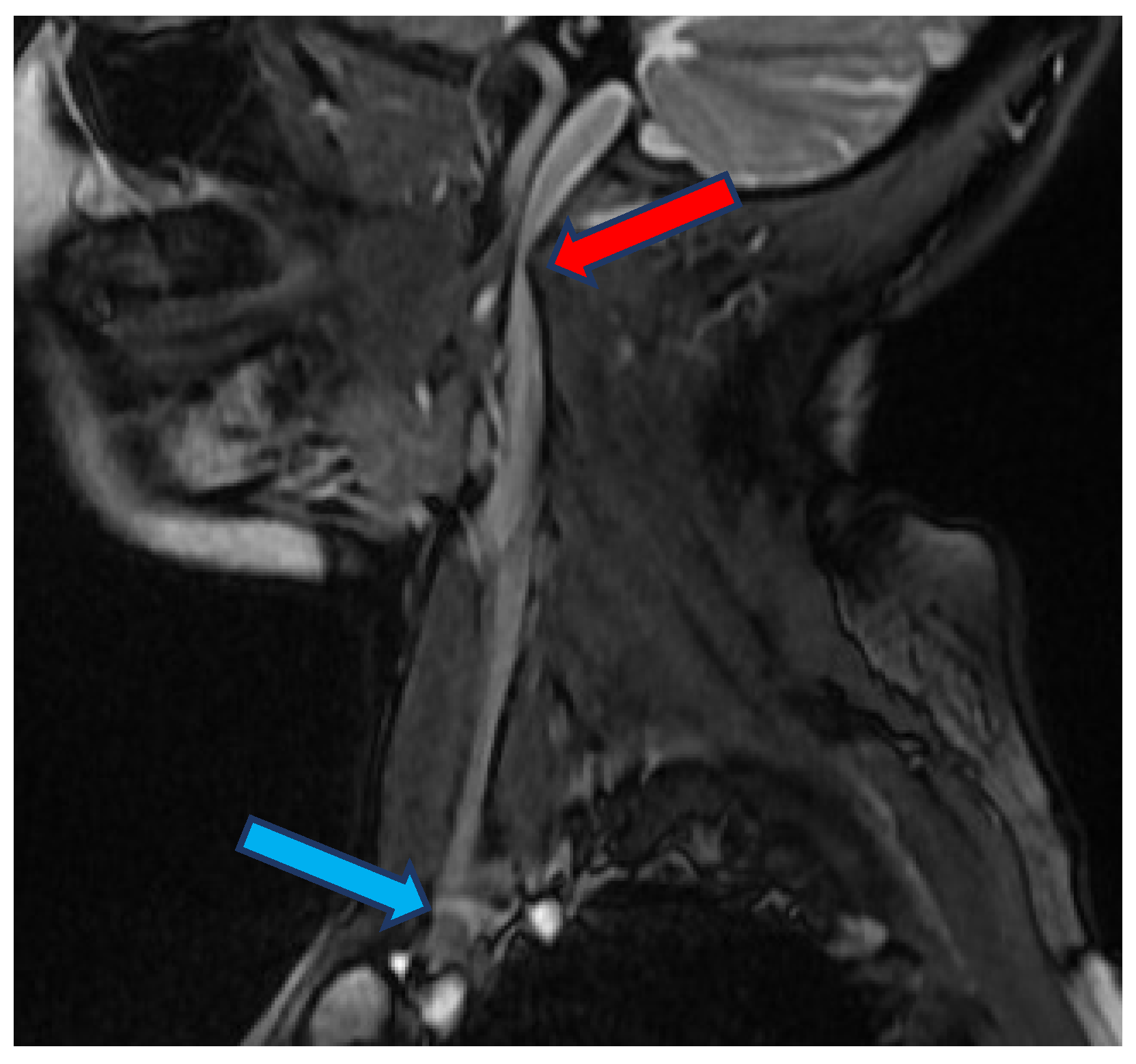
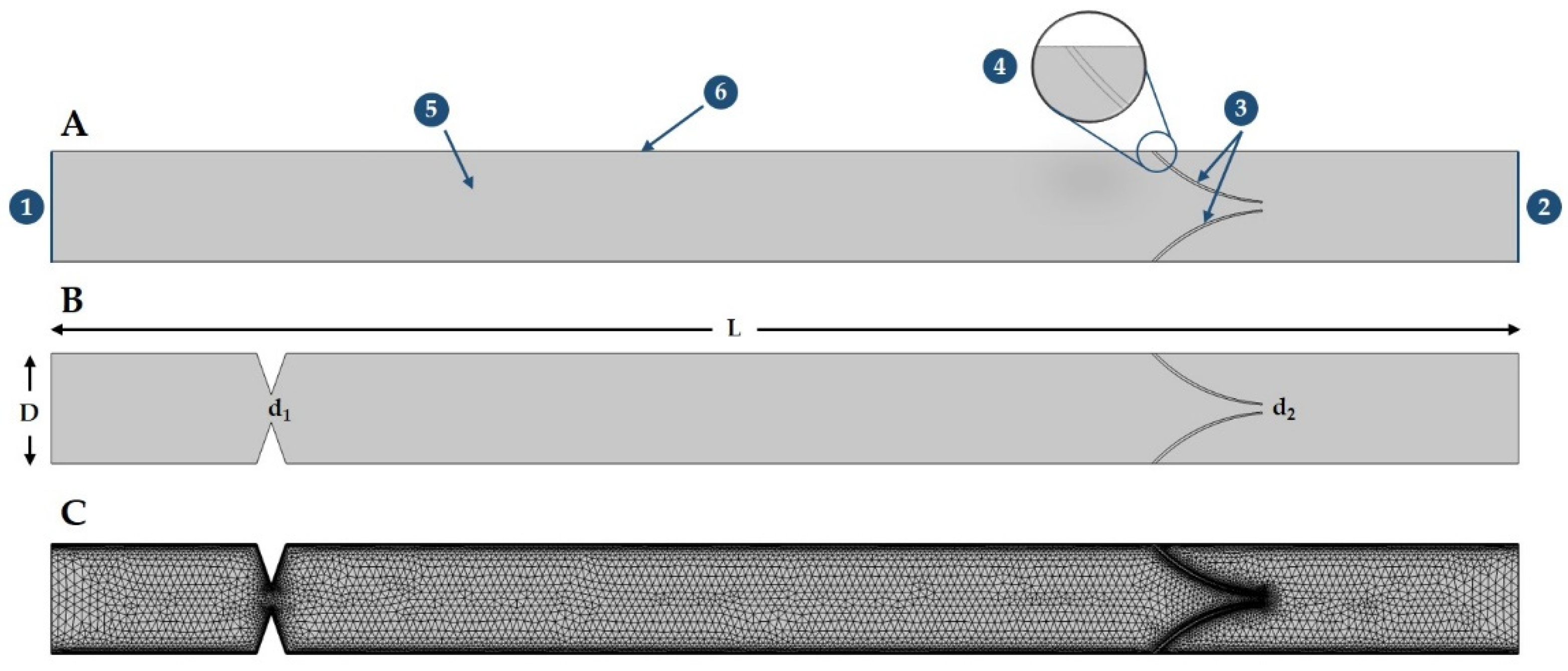
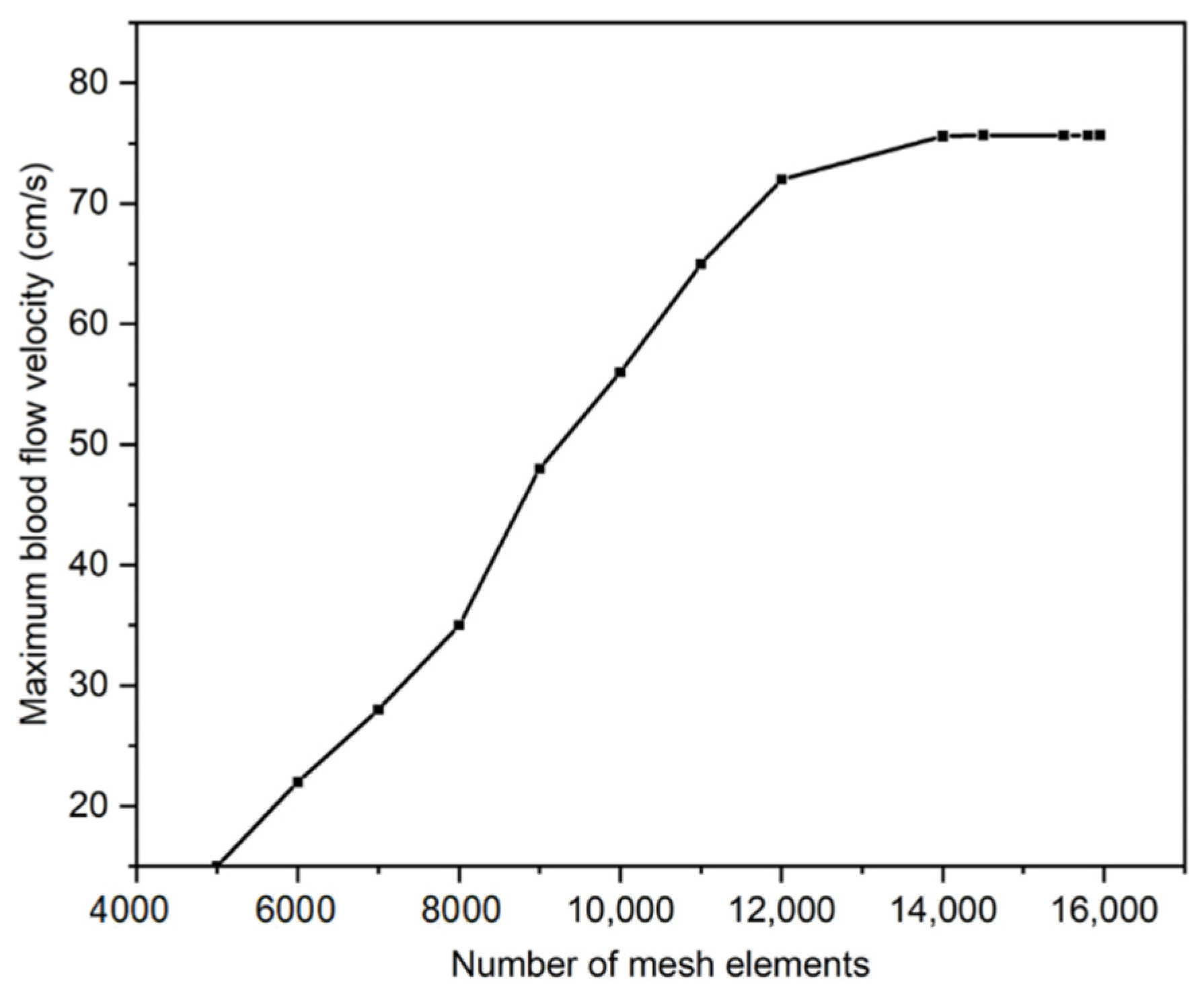
2. Materials and Methods
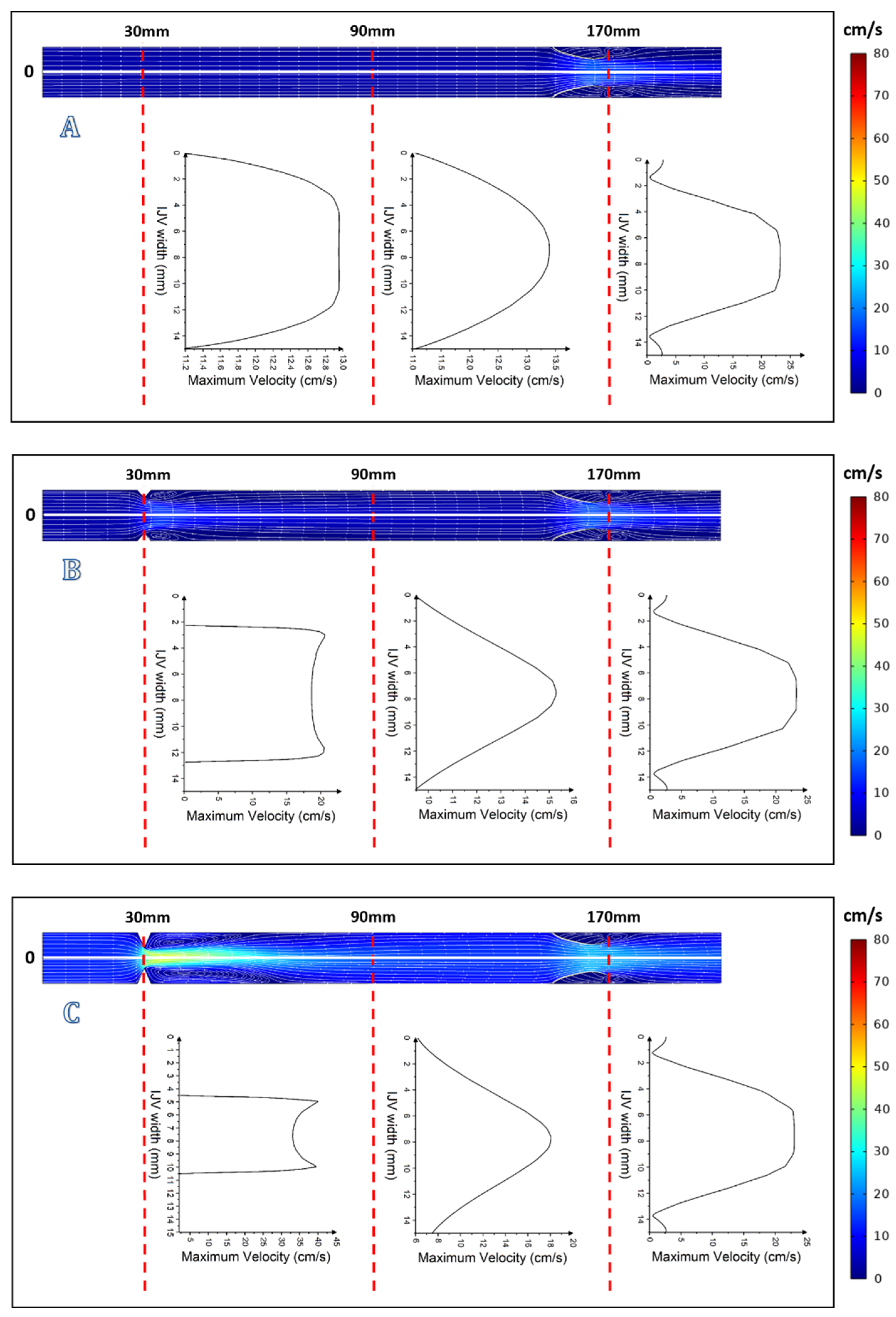
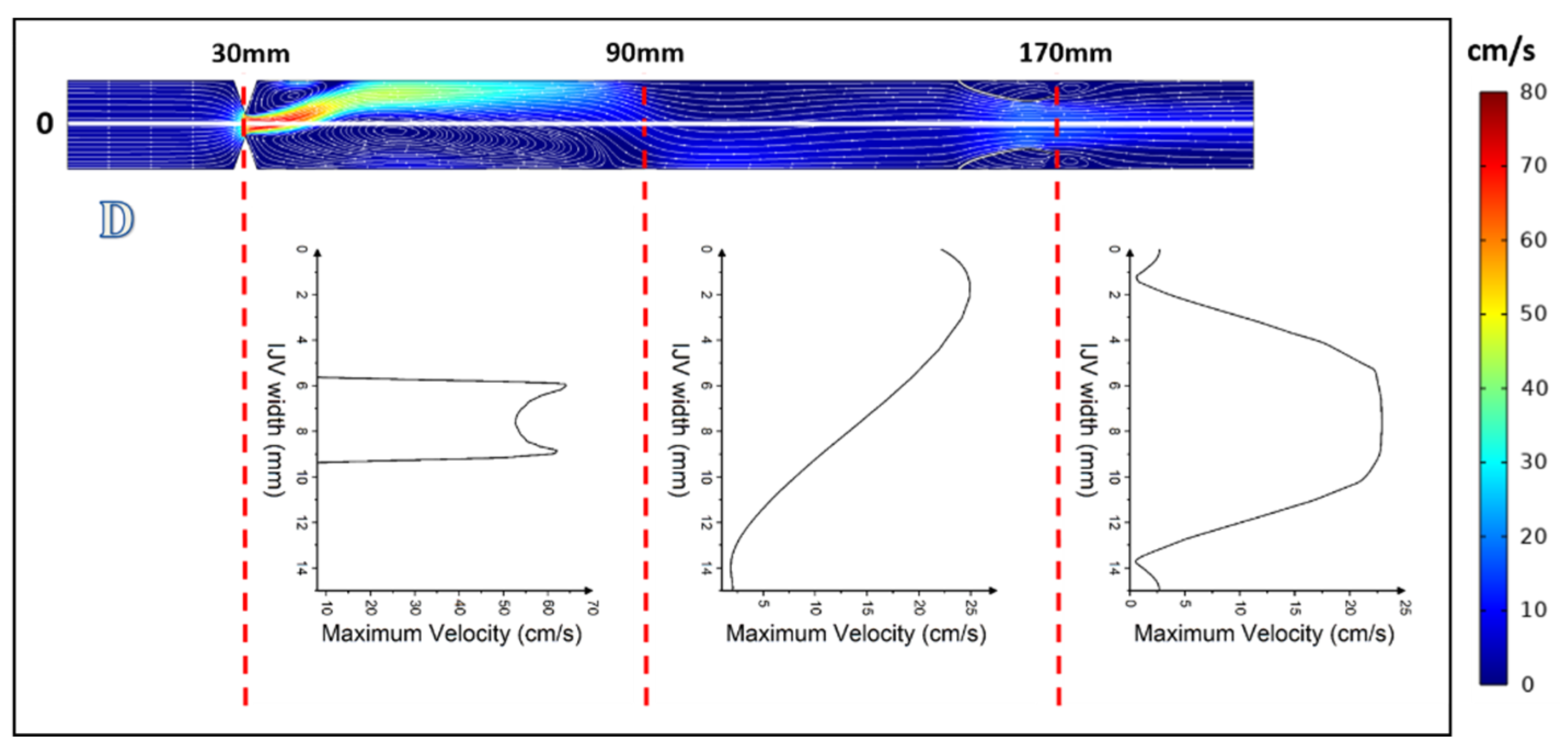
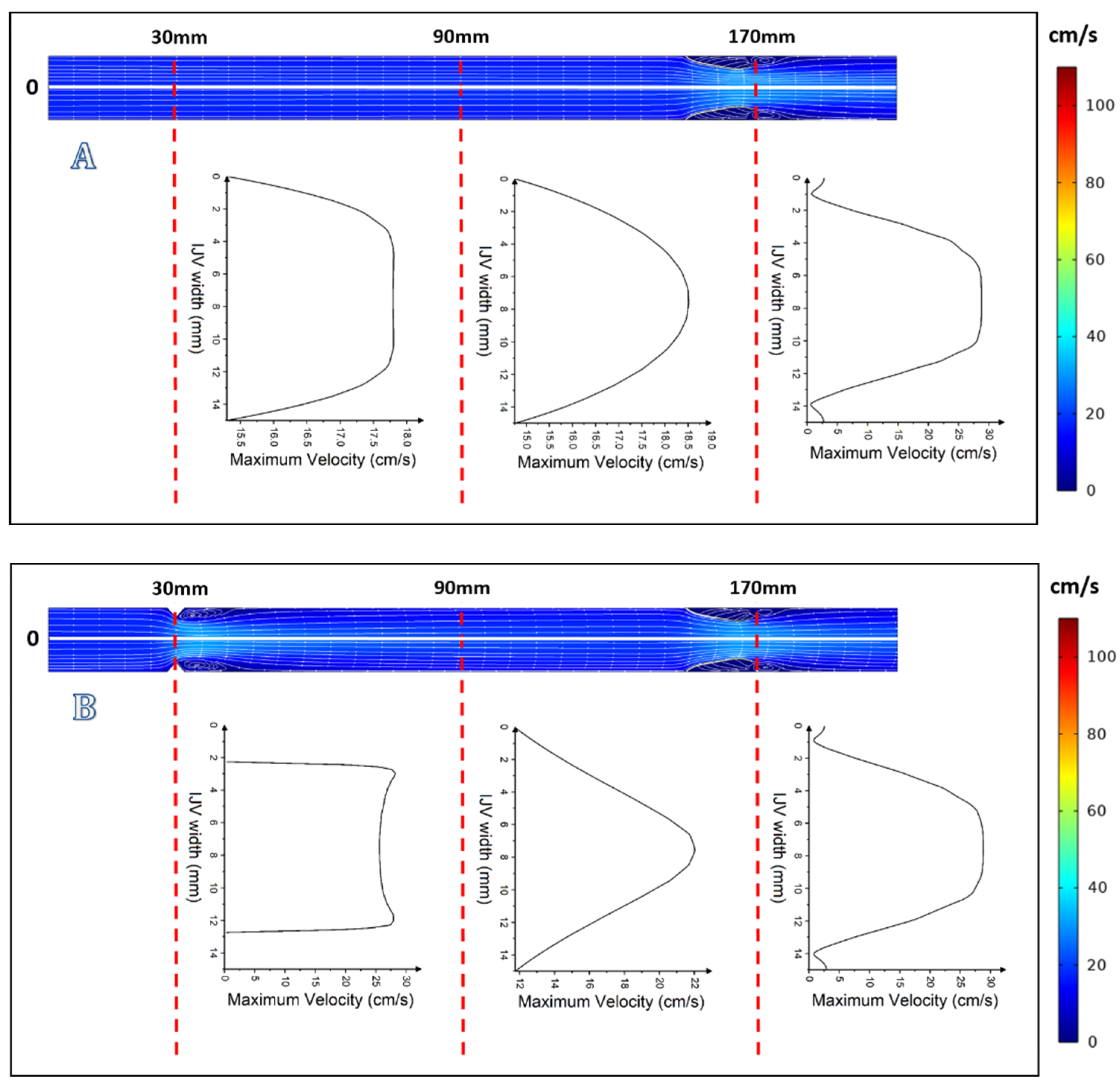
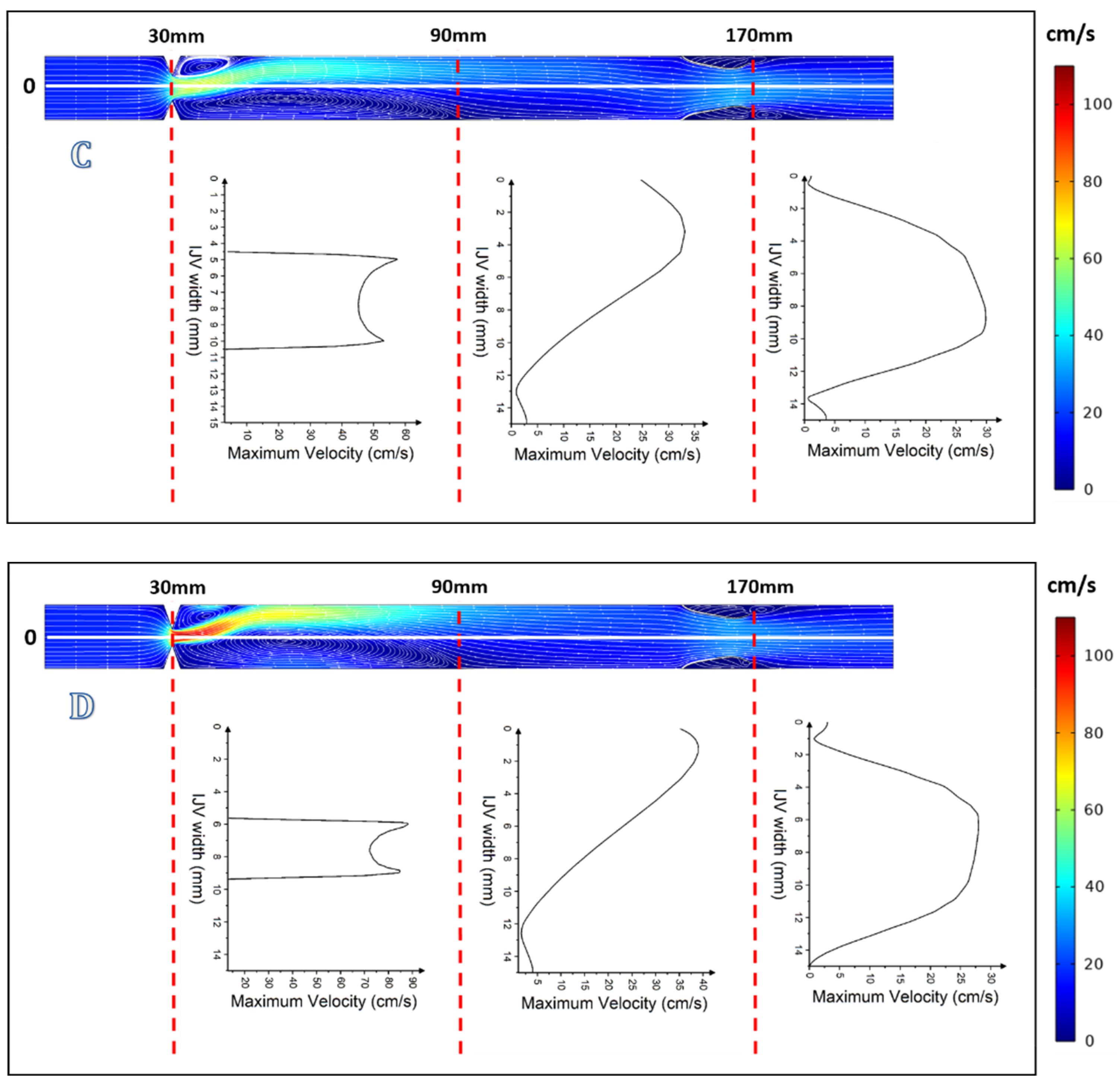
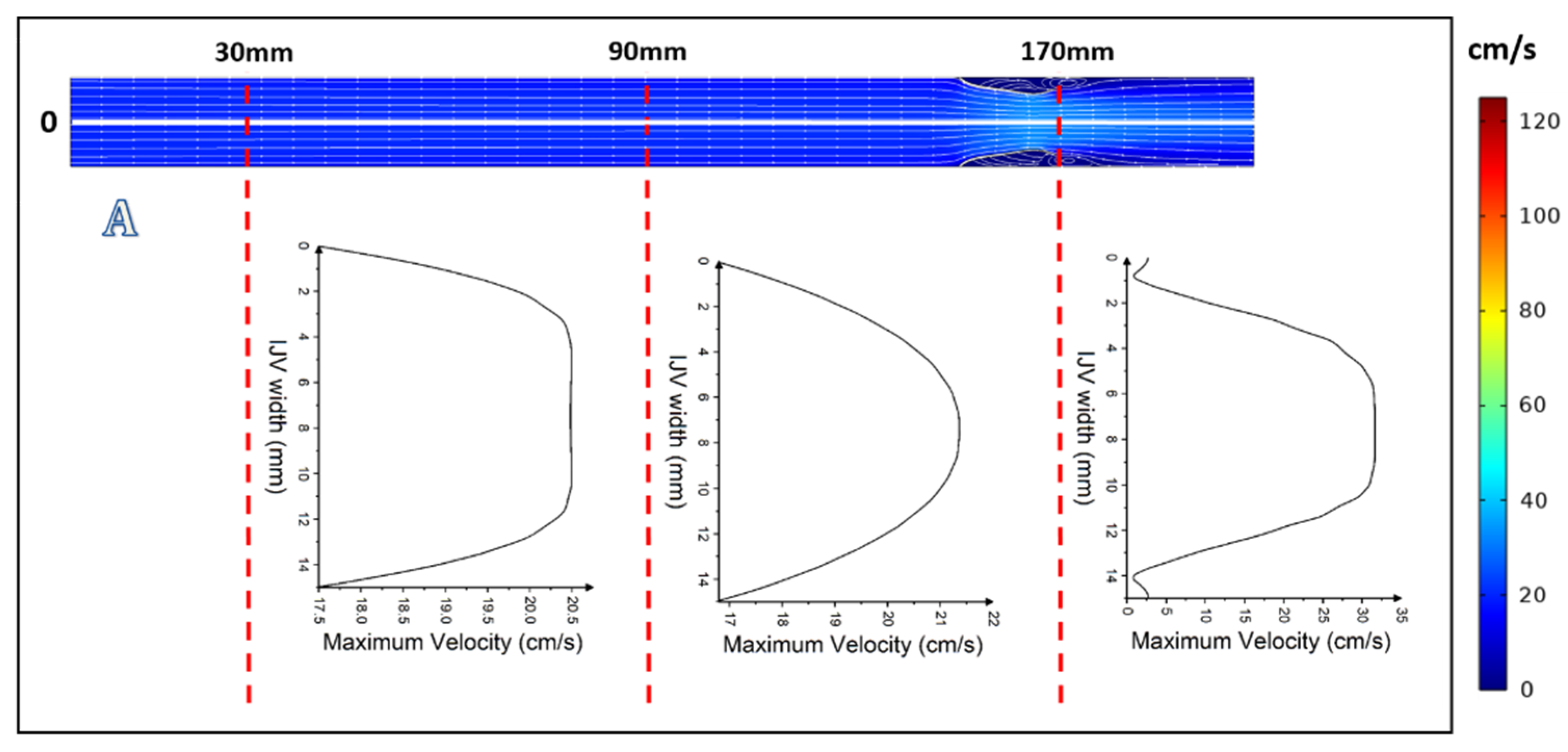
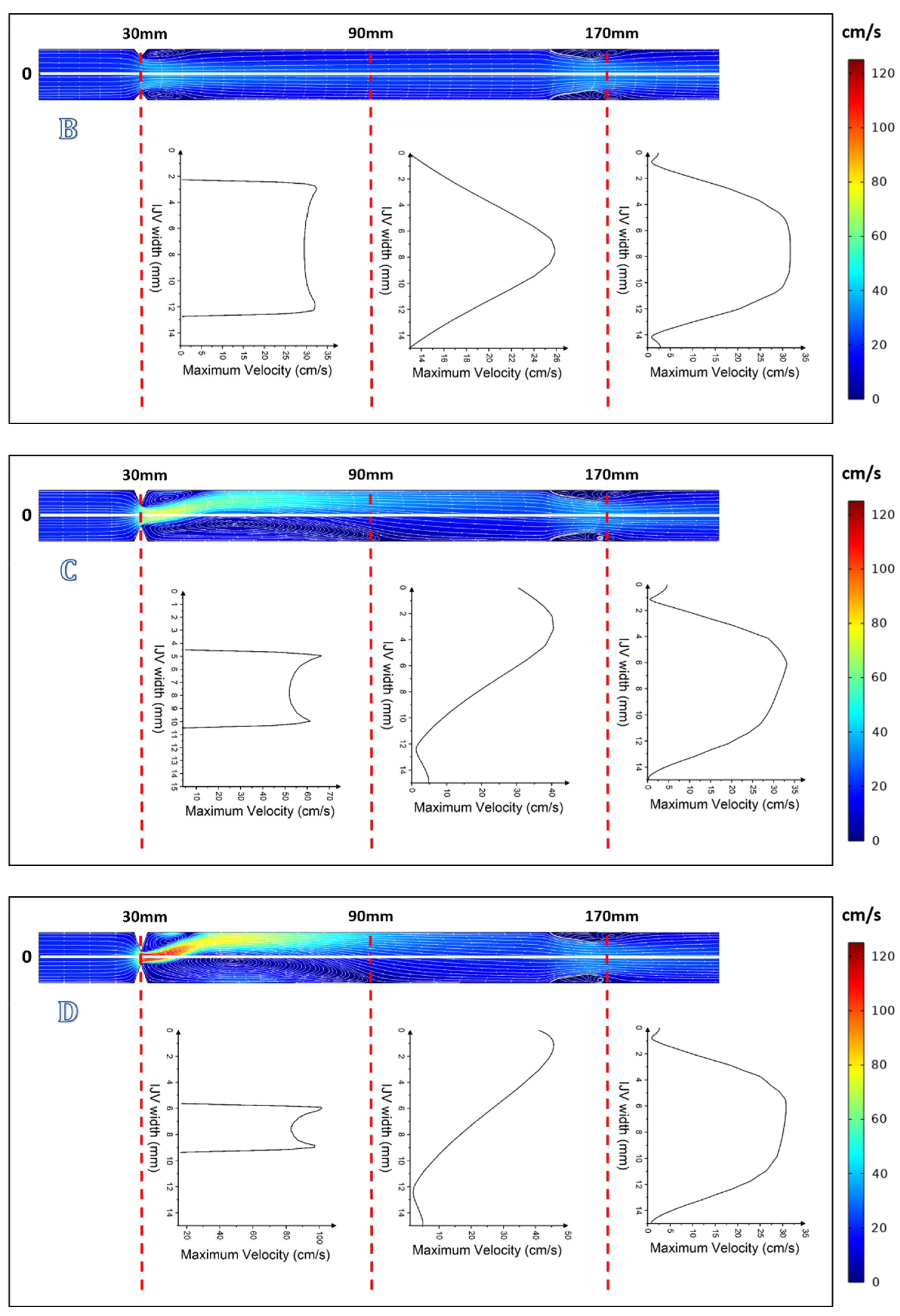
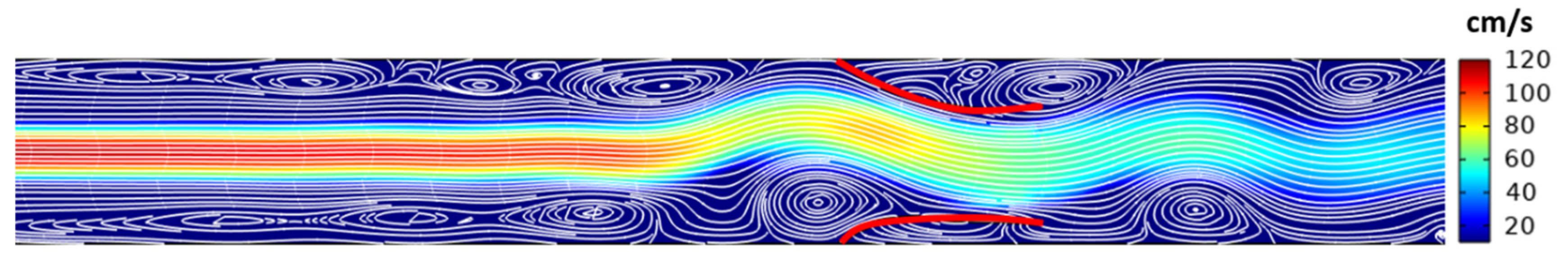
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zivadinov, R.; Chung, C.P. Potential Involvement of the Extracranial Venous System in Central Nervous System Disorders and Aging. BMC Med. 2013, 11, 260. [Google Scholar] [CrossRef] [PubMed]
- Beggs, C.; Chung, C.P.; Bergsland, N.; Wang, P.N.; Shepherd, S.; Cheng, C.Y.; Dwyer, M.G.; Hu, H.H.; Zivadinov, R. Jugular Venous Reflux and Brain Parenchyma Volumes in Elderly Patients with Mild Cognitive Impairment and Alzheimer’s Disease. BMC Neurol. 2013, 13, 157. [Google Scholar] [CrossRef] [PubMed]
- Chung, C.P.; Beggs, C.; Wang, P.N.; Bergsland, N.; Shepherd, S.; Cheng, C.Y.; Ramasamy, D.P.; Dwyer, M.G.; Hu, H.H.; Wang, P.N. Jugular Venous Reflux and White Matter Abnormalities in Alzheimer’s Disease: A Pilot Study. J. Alzheimers Dis. 2014, 39, 601–609. [Google Scholar] [CrossRef] [PubMed]
- Filipo, R.; Ciciarello, F.; Attanasio, G.; Mancini, P.; Covelli, E.; Agati, L.; Fedele, F.; Viccaro, M. Chronic Cerebrospinal Venous Insufficiency in Patients with Ménière’s Disease. Eur. Arch. Otorhinolaryngol. 2015, 272, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Alpini, D.C.; Bavera, P.M.; Hahn, A.; Mattei, V. Chronic Cerebrospinal Venous Insufficiency (CCSVI) in Meniere Disease. Case or Cause? Sci. Med. 2013, 4, 9–15. [Google Scholar]
- Li, M.; Sun, Y.; Chan, C.C.; Fan, C.; Ji, X.; Meng, R. Internal Jugular Vein Stenosis Associated with Elongated Styloid Process: Five Case Reports and Literature Review. BMC Neurol. 2019, 19, 112. [Google Scholar] [CrossRef]
- Feng, W.; Utriainen, D.; Trifan, G.; Elias, S.; Sethi, S.; Hewett, J.; Haacke, E.M. Characteristics of Flow through the Internal Jugular Veins at Cervical C2/C3 and C5/C6 Levels for Multiple Sclerosis Patients Using MR Phase Contrast Imaging. Neurol. Res. 2012, 34, 802–809. [Google Scholar] [CrossRef]
- Haacke, E.M.; Feng, W.; Utriainen, D.; Trifan, G.; Wu, Z.; Latif, Z.; Katkuri, Y.; Hewett, J.; Hubbard, D. Patients with Multiple Sclerosis with Structural Venous Abnormalities on MR Imaging Exhibit an Abnormal Flow Distribution of the Internal Jugular Veins. J. Vasc. Interv. Radiol. 2012, 23, 60–68. [Google Scholar] [CrossRef]
- Zivadinov, R.; Bastianello, S.; Dake, M.D.; Ferral, H.; Haacke, E.M.; Haskal, Z.J.; Hubbard, D.; Liasis, N.; Mandato, K.; Sclafani, S.; et al. Recommendations for Multimodal Noninvasive and Invasive Screening for Detection of Extracranial Venous Abnormalities Indicative of Chronic Cerebrospinal Venous Insufficiency: A Position Statement of the International Society for Neurovascular Disease. J. Vasc. Interv. Radiol. 2014, 25, 1785–1794. [Google Scholar] [CrossRef]
- Zhou, D.; Ding, J.Y.; Ya, J.Y.; Pan, L.Q.; Yan, F.; Yang, Q.; Ding, Y.C.; Ji, X.M.; Meng, R. Understanding jugular venous outflow disturbance. CNS Neurosci. Ther. 2018, 24, 473. [Google Scholar] [CrossRef]
- Veroux, P.; Giaquinta, A.; Perricone, D.; Lupo, L.; Gentile, F.; Virgilio, C.; Carbonaro, A.; De Pasquale, C.; Veroux, M. Internal Jugular Veins Out Flow in Patients with Multiple Sclerosis: A Catheter Venography Study. J. Vasc. Interv. Radiol. 2013, 24, 1790–1797. [Google Scholar] [CrossRef] [PubMed]
- Simka, M.; Latacz, P.; Ludyga, T.; Kazibudzki, M.; Świerad, M.; Janas, P.; Piegza, J. Prevalence of Extracranial Venous Abnormalities: Results from a Sample of 586 Multiple Sclerosis Patients. Funct. Neurol. 2011, 26, 197–203. [Google Scholar] [PubMed]
- Plog, B.A.; Nedergaard, M. The Glymphatic System in Central Nervous System Health and Disease: Past, Present, and Future. Annu. Rev. Pathol. 2018, 13, 379–394. [Google Scholar] [CrossRef] [PubMed]
- Fultz, N.E.; Bonmassar, G.; Setsompop, K.; Stickgold, R.A.; Rosen, B.R.; Polimeni, J.R.; Lewis, L.D. Coupled Electrophysiological, Hemodynamic, and Cerebrospinal Fluid Oscillations in Human Sleep. Science 2019, 366, 628–631. [Google Scholar] [CrossRef]
- Beggs, C.B.; Magnano, C.; Shepherd, S.J.; Marr, K.; Valnarov, V.; Hojnacki, D.; Bergsland, N.; Belov, P.; Grisafi, S.; Dwyer, M.G.; et al. Aqueductal Cerebrospinal Fluid Pulsatility in Healthy Individuals is Affected by Impaired Cerebral Venous Outflow. J. Magn. Reson. Imaging 2014, 40, 1215–1222. [Google Scholar] [CrossRef]
- Beggs, C.B.; Magnano, C.; Belov, P.; Krawiecki, J.; Ramasamy, D.P.; Hagemeier, J.; Zivadinov, R. Internal Jugular Vein Cross-Sectional Area and Cerebrospinal Fluid Pulsatility in the Aqueduct of Sylvius: A Comparative Study between Healthy Subjects and Multiple Sclerosis Patients. PLoS ONE 2016, 11, e0153960. [Google Scholar] [CrossRef]
- Beggs, C.B.; Shepherd, S.J.; Cecconi, P.; Lagana, M.M. Predicting the Aqueductal Cerebrospinal Fluid Pulse: A Statistical Approach. Appl. Sci. 2019, 9, 2131. [Google Scholar] [CrossRef]
- Zamboni, P.; Galeotti, R.; Menegatti, E.; Malagoni, A.M.; Gianesini, S.; Bartolomei, I.; Mascoli, F.; Salvi, F. A Prospective Open-Label Study of Endovascular Treatment of Chronic Cerebrospinal Venous Insufficiency. J. Vasc. Surg. 2009, 50, 1348–1358. [Google Scholar] [CrossRef]
- Siddiqui, A.H.; Zivadinov, R.; Benedict, R.H.B.; Karmon, Y.; Yu, J.; Hartney, M.L.; Marr, K.L.; Valnarov, V.; Kennedy, C.L.; Ramanathan, M.; et al. Prospective Randomized Trial of Venous Angioplasty in MS (PREMiSe). Neurology 2014, 83, 441–449. [Google Scholar] [CrossRef]
- Traboulsee, A.L.; Machan, L.; Girard, J.M.; Raymond, J.; Vosoughi, R.; Hardy, B.W.; Emond, F.; Gariepy, J.L.; Bone, J.N.; Siskin, G.; et al. Safety and Efficacy of Venoplasty in MS: A Randomized, Double-Blind, Sham-Controlled Phase II Trial. Neurology 2018, 91, e1660–e1668. [Google Scholar] [CrossRef]
- Zamboni, P.; Tesio, L.; Galimberti, S.; Massacesi, L.; Salvi, F.; D’Alessandro, R.; Cenni, P.; Galeotti, R.; Papini, D.; D’Amico, R.; et al. Efficacy and Safety of Extracranial Vein Angioplasty in Multiple Sclerosis: A Randomized Clinical Trial. JAMA Neurol. 2018, 75, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Napoli, V.; Berchiolli, R.; Carboncini, M.C.; Sartucci, F.; Marconi, M.; Bocci, T.; Perrone, O.; Mannoni, N.; Congestrì, C.; Benedetti, R.; et al. Percutaneous Venous Angioplasty in Patients with Multiple Sclerosis and Chronic Cerebrospinal Venous Insufficiency: A Randomized Wait List Control Study. Ann. Vasc. Surg. 2020, 62, 275–286. [Google Scholar] [CrossRef] [PubMed]
- Simka, M. An Overview of Randomized Controlled Trials on Endovascular Treatment for Chronic Cerebrospinal Venous Insufficiency in Multiple Sclerosis Patients. Phlebologie 2021, 50, 76–80. [Google Scholar] [CrossRef]
- Zamboni, P.; Galeotti, R.; Salvi, F.; Giaquinta, A.; Setacci, C.; Alborino, S.; Guzzardi, G.; Sclafani, S.J.; Maietti, E.; Veroux, P. Brave Dreams Research Group. Effects of Venous Angioplasty on Cerebral Lesions in Multiple Sclerosis: Expanded Analysis of the Brave Dreams Double-Blind, Sham-Controlled Randomized Trial. J. Endovasc. Ther. 2020, 27, 1526602819890110. [Google Scholar] [CrossRef] [PubMed]
- Simka, M.; Latacz, P. Numerical Modeling of Blood Flow in the Internal Jugular Vein with the Use of Computational Fluid Mechanics Software. Phlebology 2021, 36, 541–548. [Google Scholar] [CrossRef] [PubMed]
- Ding, J.Y.; Zhou, D.; Pan, L.Q.; Ya, J.Y.; Liu, C.; Yan, F.; Fan, C.Q.; Ding, Y.C.; Ji, X.M.; Meng, R. Cervical Spondylotic Internal Jugular Venous Compression Syndrome. CNS Neurosci. Ther. 2020, 26, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Scerrati, A.; Norri, N.; Mongardi, L.; Dones, F.; Ricciardi, L.; Trevisi, G.; Menegatti, E.; Zamboni, P.; Cavallo, M.A.; De Bonis, P. Styloidogenic-cervical spondylotic internal jugular venous compression, a vascular disease related to several clinical neurological manifestations: Diagnosis and treatment-a comprehensive literature review. Ann. Transl. Med. 2021, 9, 718. [Google Scholar] [CrossRef]
- De Bonis, P.; Menegatti, E.; Cavallo, M.A.; Sisini, F.; Trapella, G.; Scerrati, A.; Zamboni, P. JEDI (Jugular Entrapment, Dilated Ventricles, Intracranial Hypertension) Syndrome: A New Clinical Entity? A Case Report. Acta Neurochir. 2019, 161, 1367–1370. [Google Scholar] [CrossRef]
- Jeon, J.C.; Choi, W.I.; Lee, J.H.; Lee, S.H. Anatomical Morphology Analysis of Internal Jugular Veins and Factors Affecting Internal Jugular Vein Size. Medicina 2020, 56, 135. [Google Scholar] [CrossRef]
- Gataulin, Y.A.; Yukhnev, A.D.; Smirnov, S.I.; Rosukhovskiy, D.A. Numerical analysis of the leaflet elasticity effect on the flow in the model of a venous valve. J. Phys. Conf. Ser. 2019, 1359, 012010. [Google Scholar] [CrossRef]
- Phillips, R.A.; Van Slyke, D.D.; Hamilton, P.B.; Dole, V.P.; Emerson, K., Jr.; Archibald, R.M. Meaurement of specific gravities of whole blood and plasma by standard copper sulfate solutions. J. Biol. Chem. 1950, 183, 305–330. [Google Scholar] [CrossRef]
- Trudnowski, R.J.; Rico, R.C. Specific Gravity of Blood and Plasma at 4 and 37 °C. Clin. Chem. 1974, 20, 615–616. [Google Scholar] [CrossRef]
- Ciuti, G.; Righi, D.; Forzoni, L.; Fabbri, A.; Pignone, A.M. Differences Between Internal Jugular Vein and Vertebral Vein Flow Examined in Real Time with the Use of Multigate Ultrasound Color Doppler. Am. J. Neuroradiol. 2013, 34, 2000–2004. [Google Scholar] [CrossRef] [PubMed]
- Tikhomolova, L.G.; Gataulin, Y.A.; Yukhnev, A.D.; Rosukhovskiy, D.A. Fluid–Structure Interaction Modelling of the Venous Valve with Elastic Leaflets. In Proceedings of the Journal of Physics: Conference Series; IOP Publishing: Bristol, UK, 2020; Volume 1697, pp. 1–6. [Google Scholar]
- Lurie, F.; Kistner, R.L.; Eklof, B.; Kessler, D. Mechanism of Venous Valve Closure and Role of the Valve in Circulation: A New Concept. J. Vasc. Surg. 2003, 38, 955–961. [Google Scholar] [CrossRef]
- Zivadinov, R.; Marr, K.; Cutter, G.; Ramanathan, M.; Benedict, R.H.B.; Kennedy, C.; Elfadil, M.; Yeh, A.E.; Reuther, J.; Brooks, C.; et al. Prevalence, Sensitivity, and Specificity of Chronic Cerebrospinal Venous Insufficiency in MS. Neurology 2011, 77, 138–144. [Google Scholar] [CrossRef]
- Simka, M.; Hubbard, D.; Siddiqui, A.H.; Dake, M.D.; Sclafani, S.J.A.; Al-Omari, M.; Eisele, C.G.; Haskal, Z.J.; Ludyga, T.; Miloševič, Z.V.; et al. Catheter Venography for the Assessment of Internal Jugular Veins and Azygous Vein: Position Statement by Expert Panel of the International Society for Neurovascular Disease. Vasa 2013, 42, 168–176. [Google Scholar] [CrossRef]
- Mancini, M.; Lanzillo, R.; Liuzzi, R.; Di Donato, O.; Ragucci, M.; Monti, S.; Salvatore, E.; Morra, V.B.; Salvatore, M. Internal Jugular Vein Blood Flow in Multiple Sclerosis Patients and Matched Controls. PLoS ONE 2014, 9, e92730. [Google Scholar] [CrossRef]
- Simka, M.; Ludyga, T.; Latacz, P.; Kazibudzki, M.; Majewski, E.; Zaniewski, M. Chronic Cerebrospinal Venous Insufficiency is Unlikely to be a Direct Trigger of Multiple Sclerosis. Mult. Scler. Relat. Disord. 2013, 2, 334–339. [Google Scholar] [CrossRef]
- Simka, M.; Ludyga, T.; Kazibudzki, M.; Latacz, P.; Świerad, M. Multiple Sclerosis, An Unlikely Cause of Chronic Cerebrospinal Venous Insufficiency: Retrospective Analysis of Catheter Venography. JRSM Short Rep. 2012, 3, 1–6. [Google Scholar] [CrossRef]
- Lee, B.B.; Baumgartner, I.; Berlien, P.; Bianchini, G.; Burrows, P.; Gloviczki, P.; Huang, Y.; Laredo, J.; Loose, D.A.; Markovic, J.; et al. Diagnosis and Treatment of Venous Malformations. Consensus Document of the International Union of Phlebology (IUP): Updated 2013. Int. Angiol. 2015, 34, 97–149. [Google Scholar]
- Zamboni, P.; Tisato, V.; Menegatti, E.; Mascoli, F.; Gianesini, S.; Salvi, F.; Secchiero, P. Ultrastructure of Internal Jugular Vein Defective Valves. Phlebology 2015, 30, 644–647. [Google Scholar] [CrossRef] [PubMed]
- Coen, M.; Menegatti, E.; Salvi, F.; Mascoli, F.; Zamboni, P.; Gabbiani, G.; Bochaton-Piallat, M.L. Altered Collagen Expression in Jugular Veins in Multiple Sclerosis. Cardiovasc. Pathol. 2013, 22, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Pérez, M.G.; Vakkilainen, E. A comparison of turbulence models and two and three dimensional meshes for unsteady CFD ash deposition tools. Fuel 2019, 237, 806–811. [Google Scholar] [CrossRef]
- Abdi, R.; Krzaczek, M.; Tejchman, J. Comparative study of high-pressure fluid flow in densely packed granules using a 3D CFD model in a continuous medium and a simplified 2D DEM-CFD approach. Granul. Matter 2022, 24, 15. [Google Scholar] [CrossRef]
- Trozzo, R.; Boedec, G.; Leonetti, M.; Jaeger, M. Axisymmetric boundary element method for vesicles in a capillary. J. Comput. Phys. 2015, 289, 62–82. [Google Scholar] [CrossRef]
- Susan-Resiga, R.F.; Muntean, S.; Tănasă, C. Three-dimensional versus two-dimensional axisymmetric analysis for decelerated swirling flows. Conference on Modelling Fluid Flow (CMFF’09). In Proceedings of the 14th International Conference on Fluid Flow Technologies, Budapest, Hungary, 9–12 September 2009. [Google Scholar]

| Quantity (Symbol) | Unit | Quantity (Symbol) | Unit |
|---|---|---|---|
| Density of blood (ρ) | kg m−3 | Partial derivative | – |
| Time (t) | s | Fluid velocity (u) | m s−1 |
| Fluid pressure (p) | Pa | Turbulent intensity (I) | – |
| Dynamic viscosity (μ) | Pa s | External forces (F) | N |
| Turbulent kinetic energy (k) | J | Turbulent viscosity (μT) | kg2 s m−3 |
| Specific dissipation rate (ω) | J kg−1 s−1 | Mean rotation-rate tensor (Ωij) | – |
| Mean strain-rate tensor (Sij) | – | Venous valve density () | kg m−3 |
| Displacement vector (ud) | m | Deformation gradient (F) | – |
| Stress tensor (S, σ) | N m−2 | Volume force (FV) | N m−3 |
| Elastic volume ratio (Jel) | – | Identity tensor (I) | – |
| External stress tensor (Sext) | N m−2 | Strain energy density (Ws) | J |
| Lamé’s second parameter (μ) | MPa | Lamé’s first parameter (λ) | MPa |
| Modulus of elasticity (E) | MPa | Poisson’s ratio (ν) | – |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rashid, A.; Iqrar, S.A.; Rashid, A.; Simka, M. Results of Numerical Modeling of Blood Flow in the Internal Jugular Vein Exhibiting Different Types of Strictures. Diagnostics 2022, 12, 2862. https://doi.org/10.3390/diagnostics12112862
Rashid A, Iqrar SA, Rashid A, Simka M. Results of Numerical Modeling of Blood Flow in the Internal Jugular Vein Exhibiting Different Types of Strictures. Diagnostics. 2022; 12(11):2862. https://doi.org/10.3390/diagnostics12112862
Chicago/Turabian StyleRashid, Anas, Syed Atif Iqrar, Aiman Rashid, and Marian Simka. 2022. "Results of Numerical Modeling of Blood Flow in the Internal Jugular Vein Exhibiting Different Types of Strictures" Diagnostics 12, no. 11: 2862. https://doi.org/10.3390/diagnostics12112862
APA StyleRashid, A., Iqrar, S. A., Rashid, A., & Simka, M. (2022). Results of Numerical Modeling of Blood Flow in the Internal Jugular Vein Exhibiting Different Types of Strictures. Diagnostics, 12(11), 2862. https://doi.org/10.3390/diagnostics12112862









