High-Resolution Computed Tomography and Lung Ultrasound in Patients with Systemic Sclerosis: Which One to Choose?
Abstract
:1. Introduction
2. Systemic Sclerosis Associated Interstitial Lung Disease
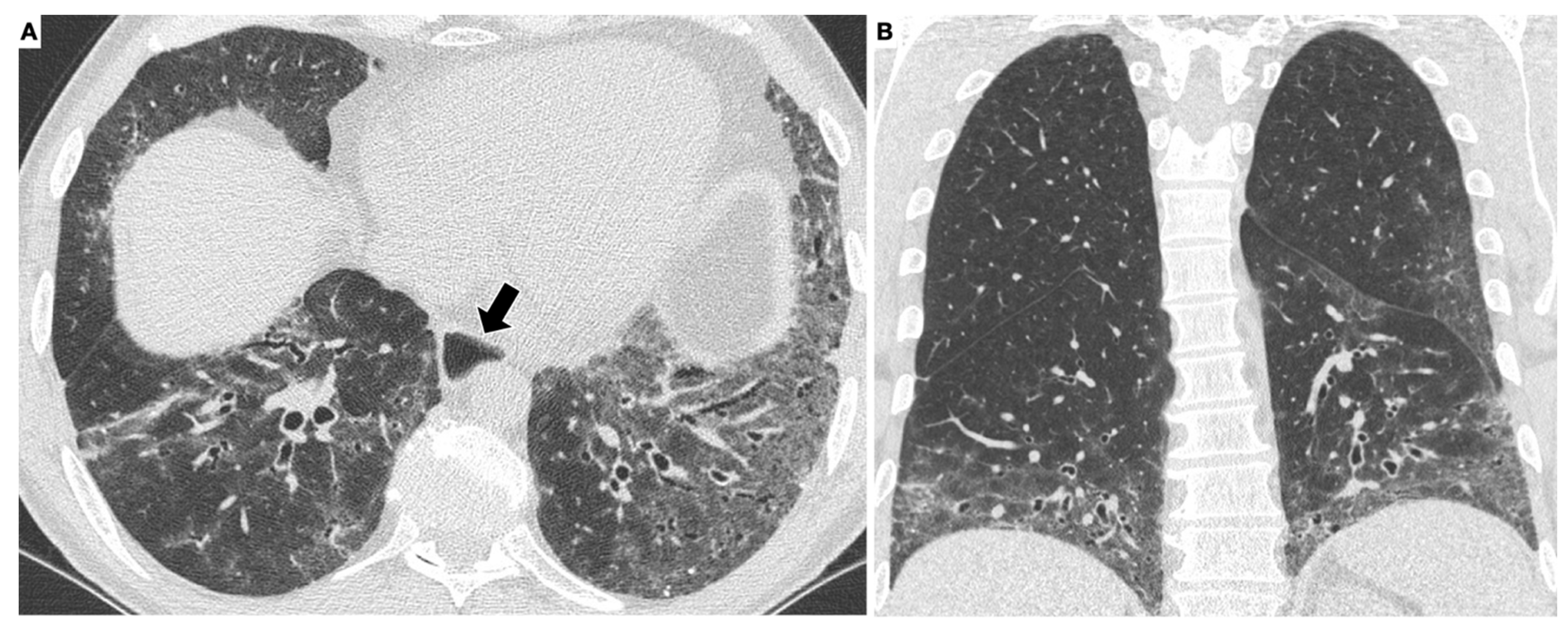
3. High-Resolution Computed Tomography in SSc-ILD
3.1. High-Resolution Computed Tomography in SSc-ILD: Qualitative Evaluation
3.2. High-Resolution Computed Tomography in SSc-ILD: Quantitative Evaluation
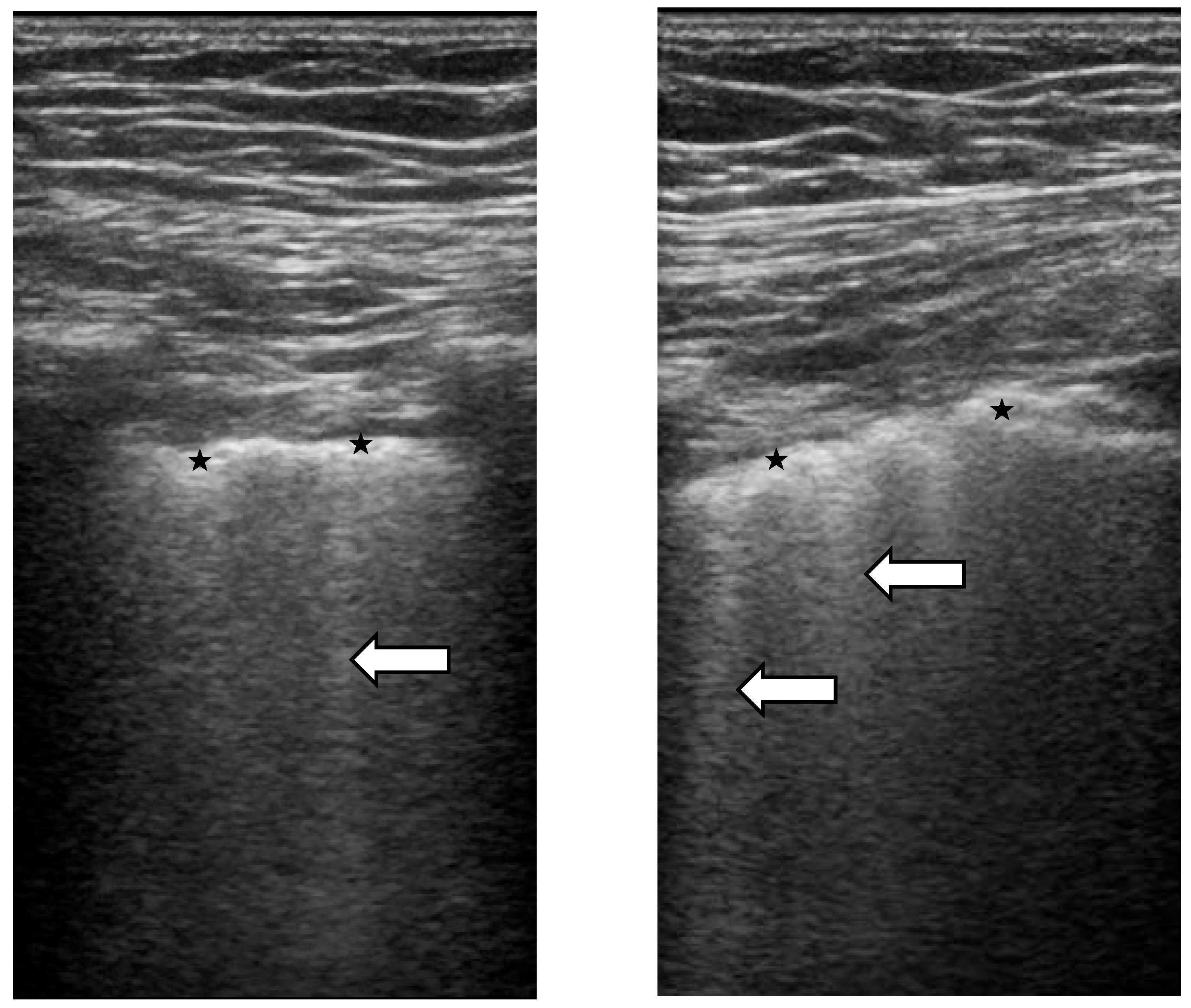
4. Lung Ultrasound
Lung Ultrasound in SSc-ILD: Qualitative Evaluation
5. The Correlation between HRTC and LUS
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Distler, O.; Assassi, S.; Cottin, V.; Cutolo, M.; Danoff, S.K.; Denton, C.P.; Distler, J.H.; Hoffmann-Vold, A.-M.; Johnson, S.R.; Ladner, U.M.; et al. Predictors of progression in systemic sclerosis patients with interstitial lung disease. Eur. Respir. J. 2020, 55, 1902026. [Google Scholar] [CrossRef] [Green Version]
- Elhai, M.; Meune, C.; Avouac, J.; Kahan, A.; Allanore, Y. Trends in mortality in patients with systemic sclerosis over 40 years: A systematic review and meta-analysis of cohort studies. Rheumatology 2012, 51, 1017–1026. [Google Scholar] [CrossRef] [Green Version]
- Tyndall, A.J.; Bannert, B.; Vonk, M.; Airò, P.; Cozzi, F.; Carreira, P.E.; Bancel, D.F.; Allanore, Y.; Müller-Ladner, U.; Distler, O.; et al. Causes and risk factors for death in systemic sclerosis: A study from the EULAR Scleroderma Trials and Research (EUSTAR) database. Ann. Rheum. Dis. 2010, 69, 1809–1815. [Google Scholar] [CrossRef] [Green Version]
- Volkmann, E.R.; Tashkin, D.P.; Sim, M.; Li, N.; Goldmuntz, E.; Keyes-Elstein, L.; Pinckney, A.; Furst, D.E.; Clements, P.J.; Khanna, D.; et al. Short-term progression of interstitial lung disease in systemic sclerosis predicts long-term survival in two independent clinical trial cohorts. Ann. Rheum. Dis. 2018, 78, 122–130. [Google Scholar] [CrossRef]
- Ruaro, B.; Sulli, A.; Smith, V.; Pizzorni, C.; Paolino, S.; Alessandri, E.; Trombetta, A.C.; Cutolo, M. Advances in nailfold capillaroscopic analysis in systemic sclerosis. J. Scleroderma Relat. Disord. 2018, 3, 122–131. [Google Scholar] [CrossRef] [Green Version]
- Trombetta, A.C.; Smith, V.; Pizzorni, C.; Meroni, M.; Paolino, S.; Cariti, C.; Ruaro, B.; Sulli, A.; Cutolo, M. Quantitative Alterations of Capillary Diameter Have a Predictive Value for Development of the Capillaroscopic Systemic Sclerosis Pattern. J. Rheumatol. 2016, 43, 599–606. [Google Scholar] [CrossRef]
- Fischer, A.; Patel, N.M.; Volkmann, E.R. Interstitial Lung Disease in Systemic Sclerosis: Focus on Early Detection and Intervention. Open Access Rheumatol. 2019, 11, 283–307. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murdaca, G.; Caprioli, S.; Tonacci, A.; Billeci, L.; Greco, M.; Negrini, S.; Cittadini, G.; Zentilin, P.; Ventura Spagnolo, E.; Gangemi, S. A Machine Learning Application to Predict Early Lung Involvement in Scleroderma: A Feasibility Evaluation. Diagnostics 2021, 11, 1880. [Google Scholar] [CrossRef] [PubMed]
- Khanna, D.; Tashkin, D.P.; Denton, C.P.; Renzoni, E.A.; Desai, S.R.; Varga, J. Etiology, Risk Factors, and Biomarkers in Systemic Sclerosis with Interstitial Lung Disease. Am. J. Respir. Crit. Care Med. 2020, 201, 650–660. [Google Scholar] [CrossRef] [PubMed]
- Cappelli, S.; Bellando Randone, S.; Camiciottoli, G.; De Paulis, A.; Guiducci, S.; Matucci-Cerinic, M. Interstitial lung disease in systemic sclerosis: Where do we stand? Eur. Respir. Rev. 2015, 24, 411–419. [Google Scholar] [CrossRef]
- Demizio, D.J.; Bernstein, E. Detection and classification of systemic sclerosis-related interstitial lung disease: A review. Curr. Opin. Rheumatol. 2019, 31, 553–560. [Google Scholar] [CrossRef] [PubMed]
- Ruaro, B.; Casabella, A.; Paolino, S.; Pizzorni, C.; Alessandri, E.; Seriolo, C.; Botticella, G.; Molfetta, L.; Odetti, P.; Smith, V.; et al. Correlation between bone quality and microvascular damage in systemic sclerosis patients. Rheumatology 2018, 57, 1548–1554. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smith, V.; Scirè, C.A.; Talarico, R.; Airo, P.; Alexander, T.; Allanore, Y.; Bruni, C.; Codullo, V.; Dalm, V.; De Vries-Bouwstra, J.; et al. Systemic sclerosis: State of the art on clinical practice guidelines. RMD Open 2018, 4, e000782. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Orlandi, M.; Landini, N.; Sambataro, G.; Nardi, C.; Tofani, L.; Bruni, C.; Randone, S.B.; Blagojevic, J.; Melchiorre, D.; Hughes, M.; et al. The role of chest CT in deciphering interstitial lung involvement: Systemic sclerosis versus COVID-19. Rheumatology 2021, 28, 615. [Google Scholar] [CrossRef]
- Clukers, J.; Lukers, J.; Lanclus, M.; Belmans, D.; van Holsbeke, C.; de Backer, W.; Vummidi, D.; Cronin, P.; Lavon, B.R.; de Backer, J.; et al. Interstitial lung disease in systemic sclerosis quantification of disease classification and progression with high-resolution computed tomography: An observational study. J. Scleroderma Relat. Disord. 2021, 6, 154–164. [Google Scholar] [CrossRef]
- Ruaro, B.; Baratella, E.; Confalonieri, P.; Wade, B.; Marrocchio, C.; Geri, P.; Busca, A.; Pozzan, R.; Andrisano, A.G.; Cova, M.A.; et al. High-Resolution Computed Tomography: Lights and Shadows in Improving Care for SSc-ILD Patients. Diagnostics 2021, 11, 1960. [Google Scholar] [CrossRef] [PubMed]
- Desai, S.R.; Veeraraghavan, S.; Hansell, D.M.; Nikolakopolou, A.; Goh, N.S.; Nicholson, A.G.; Colby, T.V.; Denton, C.P.; Black, C.M.; Du Bois, R.M.; et al. CT features of lung disease in patients with systemic sclerosis: Comparison with idiopathic pulmonary fibrosis and non-specific interstitial pneumonia. Radiology 2004, 232, 560–567. [Google Scholar] [CrossRef] [PubMed]
- Launay, D.; Remy-Jardin, M.; Michon-Pasturel, U.; Mastora, I.; Hachulla, E.; Lambert, M.; Delannoy, V.; Queyrel, V.; Duhamel, A.; Matran, R.; et al. High resolution computed tomography in fibrosing alveolitis associated with systemic sclerosis. J. Rheumatol. 2006, 33, 1789–1801. [Google Scholar]
- Soldano, S.; Trombetta, A.C.; Contini, P.; Tomatis, V.; Ruaro, B.; Brizzolara, R.; Montagna, P.; Sulli, A.; Paolino, S.; Pizzorni, C.; et al. Increase in circulating cells coexpressing M1 and M2 macrophage surface markers in patients with systemic sclerosis. Ann. Rheum. Dis. 2018, 77, 1842–1845. [Google Scholar] [CrossRef]
- Ruaro, B.; Bruni, C.; Wade, B.; Baratella, E.; Confalonieri, P.; Antonaglia, C.; Geri, P.; Biolo, M.; Confalonieri, M.; Salton, F. Laser Speckle Contrast Analysis: Functional Evaluation of Microvascular Damage in Connective Tissue Diseases. Is There Evidence of Correlations with Organ Involvement, Such as Pulmonary Damage? Front. Physiol. 2021, 12, 710298. [Google Scholar] [CrossRef]
- Varga, J.; Abraham, D. Systemic sclerosis: A prototypic multisystem fibrotic disorder. J. Clin. Investig. 2007, 117, 557–567. [Google Scholar] [CrossRef] [PubMed]
- Delle Sedie, A.; Doveri, M.; Frassi, F.; Gargani, L.; D’Errico, G.; Pepe, P.; Bazzichi, L.; Riente, L.; Caramella, D.; Bombardieri, S. Ultrasound Lung Comets in Systemic Sclerosis: A Useful Tool to Detect Lung Interstitial Fibrosis. Clin. Exp. Rheumatol. 2010, 28, S54. [Google Scholar] [PubMed]
- Wang, Y.; Gargani, L.; Barskova, T.; Furst, D.E.; Cerinic, M.M. Usefulness of lung ultrasound B-lines in connective tissue disease-associated interstitial lung disease: A literature review. Arthritis Res. Ther. 2017, 19, 206. [Google Scholar] [CrossRef] [Green Version]
- Gargani, L.; Doveri, M.; D’Errico, L.; Frassi, F.; Bazzichi, M.L.; Delle Sedie, A.; Scali, M.C.; Monti, S.; Mondillo, S.; Bombardieri, S.; et al. Ultrasound Lung Comets in Systemic Sclerosis: A Chest Sonography Hallmark of Pulmonary Interstitial Fibrosis. Rheumatology 2009, 48, 1382–1387. [Google Scholar] [CrossRef] [Green Version]
- Vicente-Rabaneda, E.F.; Bong, D.A.; Castañeda, S.; Möller, I. Use of ultrasound to diagnose and monitor interstitial lung disease in rheumatic diseases. Clin. Rheumatol. 2021, 40, 3547–3564. [Google Scholar] [CrossRef] [PubMed]
- Fairchild, R.; Chung, M.; Yang, D.; Sharpless, L.; Li, S.; Chung, L. Development and Assessment of Novel Lung Ultrasound Interpretation Criteria for the Detection of Interstitial Lung Disease in Systemic Sclerosis. Arthritis Care Res. 2021, 73, 1338–1342. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann-Vold, A.M.; Molberg, Ø. Detection, screening, and classification of interstitial lung disease in patients with systemic sclerosis. Curr. Opin. Rheumatol. 2020, 32, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Trombetta, A.C.; Smith, V.; Gotelli, E.; Ghio, M.; Paolino, S.; Pizzorni, C.; Vanhaecke, A.; Ruaro, B.; Sulli, A.; Cutolo, M. Vitamin D deficiency and clinical correlations in systemic sclerosis patients: A retrospective analysis for possible future developments. PLoS ONE 2017, 12, e0179062. [Google Scholar] [CrossRef]
- Volpicelli, G. Lung Ultrasound B-Lines in Interstitial Lung Disease. Chest 2020, 158, 1323–1324. [Google Scholar] [CrossRef]
- Hughes, M.; Bruni, C.; Ruaro, B.; Confalonieri, M.; Matucci-Cerinic, M.; Bellando-Randone, S. Digital Ulcers in Systemic Sclerosis. Presse Med. 2021, 50, 104064. [Google Scholar] [CrossRef] [PubMed]
- Cutolo, M.; Vanhaecke, A.; Ruaro, B.; Deschepper, E.; Ickinger, C.; Melsens, K.; Piette, Y.; Trombetta, A.C.; De Keyser, F.; Smith, V.; et al. Is laser speckle contrast analysis (LASCA) the new kid on the block in systemic sclerosis? A systematic literature review and pilot study to evaluate reliability of LASCA to measure peripheral blood perfusion in scleroderma patients. Autoimmun. Rev. 2018, 17, 775–780. [Google Scholar] [CrossRef] [PubMed]
- Ruaro, B.; Confalonieri, M.; Salton, F.; Wade, B.; Baratella, E.; Geri, P.; Confalonieri, P.; Kodric, M.; Biolo, M.; Bruni, C. The Relationship between Pulmonary Damage and Peripheral Vascular Manifestations in Systemic Sclerosis Patients. Pharmaceuticals 2021, 14, 403. [Google Scholar] [CrossRef]
- Varga, J.; Trojanowska, M.; Kuwana, M. Pathogenesis of systemic sclerosis: Recent insights of molecular and cellular mechanisms and therapeutic opportunities. J. Scleroderma Relat. Disord. 2017, 2, 137–152. [Google Scholar] [CrossRef]
- Ruaro, B.; Soldano, S.; Smith, V.; Paolino, S.; Contini, P.; Montagna, P.; Pizzorni, C.; Casabella, A.; Tardito, S.; Sulli, A.; et al. Correlation between circulating fibrocytes and dermal thickness in limited cutaneous systemic sclerosis patients: A pilot study. Rheumatol. Int. 2019, 39, 1369–1376. [Google Scholar] [CrossRef] [PubMed]
- Bouros, D.; Wells, A.U.; Nicholson, A.G.; Colby, T.V.; Polychronopoulos, V.; Pantelidis, P.; Haslam, P.L.; Vassilakis, D.A.; Black, C.M.; Du Bois, R.M. Histopathologic subsets of fibrosing alveolitis in patients with systemic sclerosis and their relationship to outcome. Am. J. Respir. Crit. Care Med. 2002, 165, 1581–1586. [Google Scholar] [CrossRef] [PubMed]
- Highland, K.B.; Distler, O.; Kuwana, M.; Allanore, Y.; Assassi, S.; Azuma, A.; Bourdin, A.; Denton, C.P.; Distler, J.H.; Hoffmann-Vold, A.M.; et al. Efficacy and safety of nintedanib in patients with systemic sclerosis-associated interstitial lung disease treated with mycophenolate: A subgroup analysis of the SENSCIS trial. Lancet Respir. Med. 2021, 9, 96–106. [Google Scholar] [CrossRef]
- Denton, C.P.; Khanna, D. Systemic sclerosis. Lancet 2017, 390, 1685–1699. [Google Scholar] [CrossRef]
- Nihtyanova, S.I.; Schreiber, B.E.; Ong, V.H.; Rosenberg, D.; Moinzadeh, P.; Coghlan, J.G.; Wells, A.U.; Denton, C.P. Prediction of pulmonary complications and long-term survival in systemic sclerosis. Arthritis Rheumatol. 2014, 66, 1625–1635. [Google Scholar] [CrossRef]
- Steen, V.; Domsic, R.T.; Lucas, M.; Fertig, N.; Medsger, T.A. A clinical and serologic comparison of African-American and Caucasian patients with systemic sclerosis. Arthritis Rheum. 2012, 64, 2986–2994. [Google Scholar] [CrossRef]
- Jaeger, V.K.; Wirz, E.G.; Allanore, Y.; Rossbach, P.; Riemekasten, G.; Hachulla, E.; Distler, O.; Airò, P.; Carreira, P.E.; Balbir Gurman, A.; et al. Incidences and risk factors of organ manifestations in the early course of systemic sclerosis: A longitudinal EUSTAR study. PLoS ONE 2016, 11, e0163894. [Google Scholar] [CrossRef] [PubMed]
- van den Hoogen, F.; Khanna, D.; Fransen, J.; Johnson, S.R.; Baron, M.; Tyndall, A.; Matucci-Cerinic, M.; Naden, R.P.; Medsger, T.A., Jr.; Carreira, P.E.; et al. 2013 classification criteria for systemic sclerosis: An American college of rheumatology/European league against rheumatism collaborative initiative. Ann. Rheum. Dis. 2013, 72, 1747–1755. [Google Scholar] [CrossRef] [Green Version]
- Ho, K.T.; Reveille, J.D. The clinical relevance of autoantibodies in scleroderma. Arthritis Res. Ther. 2003, 5, 80–93. [Google Scholar] [PubMed] [Green Version]
- Sulli, A.; Ruaro, B.; Smith, V.; Pizzorni, C.; Zampogna, G.; Gallo, M.; Cutolo, M. Progression of Nailfold Microvascular Damage and Antinuclear Antibody Pattern in Systemic Sclerosis. J. Rheumatol. 2013, 40, 634–639. [Google Scholar] [CrossRef] [PubMed]
- Lynch, D.A.; Sverzellati, N.; Travis, W.D.; Brown, K.K.; Colby, T.V.; Galvin, J.R.; Goldin, J.G.; Hansell, D.M.; Inoue, Y.; Johkoh, T.; et al. Diagnostic criteria for idiopathic pulmonary fibrosis: A Fleischner Society White Paper. Lancet Respir. Med. 2018, 6, 138–153. [Google Scholar] [CrossRef]
- Baratella, E.; Ruaro, B.; Giudici, F.; Wade, B.; Santagiuliana, M.; Salton, F.; Confalonieri, P.; Simbolo, M.; Scarpa, A.; Tollot, S.; et al. Evaluation of Correlations between Genetic Variants and High-Resolution Computed Tomography Patterns in Idiopathic Pulmonary Fibrosis. Diagnostics 2021, 11, 762. [Google Scholar] [CrossRef]
- Hansell, D.M.; Bankier, A.A.; MacMahon, H.; McLoud, T.C.; Müller, N.L.; Remy, J. Fleischner Society: Glossary of Terms for Thoracic Imaging. Radiology 2008, 246, 697–722. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baratella, E.; Marrocchio, C.; Cifaldi, R.; Santagiuliana, M.; Bozzato, A.M.; Crivelli, P.; Ruaro, B.; Salton, F.; Confalonieri, M.; Cova, M.A. Interstitial lung disease in patients with antisynthetase syndrome: A retrospective case series study. Jpn. J. Radiol. 2021, 39, 40–46. [Google Scholar] [CrossRef]
- Yoo, H.; Hino, T.; Han, J.; Franks, T.J.; Im, Y.; Hatabu, H.; Chung, M.P.; Lee, K.S. Connective tissue disease-related interstitial lung disease (CTD-ILD) and interstitial lung abnormality (ILA): Evolving concept of CT findings, pathology and management. Eur. J. Radiol. Open 2020, 8, 100311. [Google Scholar] [CrossRef]
- Remy-Jardin, M.; Giraud, F.; Remy, J.; Copin, M.C.; Gosselin, B.; Duhamel, A. Importance of ground-glass attenuation in chronic diffuse infiltrative lung disease: Pathologic-CT correlation. Radiology 1993, 189, 693–698. [Google Scholar] [CrossRef] [PubMed]
- Shah, R.M.; Jimenez, S.; Wechsler, R. Significance of Ground-glass Opacity on HRCT in Long-term Follow-up of Patients With Systemic Sclerosis. J. Thorac. Imaging 2007, 22, 120–124. [Google Scholar] [CrossRef]
- Cogliati, C.; Antivalle, M.; Torzillo, D.; Birocchi, S.; Norsa, A.; Bianco, R.; Costantino, G.; Ditto, M.C.; Battellino, M.; Sarzi Puttini, P.C.; et al. Standard and pocket-size lung ultrasound devices can detect interstitial lung disease in rheumatoid arthritis patients. Rheumatology 2014, 53, 1497–1503. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kang, E.H.; Song, Y.W. Pharmacological Interventions for Pulmonary Involvement in Rheumatic Diseases. Pharmaceuticals 2021, 14, 251. [Google Scholar] [CrossRef] [PubMed]
- Bruni, C.; Mattolini, L.; Tofani, L.; Gargani, L.; Landini, N.; Lepri, G.; Orlandi, M.; Guiducci, S.; Randone, S.B.; Matucci-Cerinic, M. Lung ultrasound to assess the severity of interstitial lung disease in systemic sclerosis. Ann. Rheum. Dis. 2021, 80, 675. [Google Scholar] [CrossRef]
- Rotondo, C.; Chiala, A.; Nivuori, M.; Coladonato, L.; Giannini, M.; Anelli, M.G.; Righetti, G.; Scioscia, C.; Fiorentini, C.; Lopalco, G.; et al. Chest Ultrasound Signs of Interstitial Lung Disease in Systemic Sclerosis Patients: A Comparison between High Resolution Chest Computed Tomography Findings. Ann. Rheum. Dis. 2016, 75, 747. [Google Scholar] [CrossRef]
- Goh, N.S.; Desai, S.R.; Veeraraghavan, S.; Hansell, D.M.; Copley, S.J.; Maher, T.M.; Corte, T.J.; Sander, C.R.; Ratoff, J.; Devaraj, A.; et al. Interstitial lung disease in systemic sclerosis: A simple staging system. Am. J. Respir. Crit. Care Med. 2008, 177, 1248–1254. [Google Scholar] [CrossRef]
- Goh, N.S.; Hoyles, R.K.; Denton, C.P.; Hansell, D.M.; Renzoni, E.; Maher, T.M.; Nicholson, A.G.; Wells, A.U. Short-Term Pulmonary Function Trends Are Predictive of Mortality in Interstitial Lung Disease Associated with Systemic Sclerosis. Arthritis Rheumatol. 2017, 69, 1670–1678. [Google Scholar] [CrossRef]
- Albera, C.; Verri, G.; Sciarrone, F.; Sitia, E.; Mangiapia, M.; Solidoro, P. Progressive Fibrosing Interstitial Lung Diseases: A Current Perspective. Biomedicines 2021, 9, 1237. [Google Scholar] [CrossRef]
- Yabuuchi, H.; Matsuo, Y.; Tsukamoto, H.; Horiuchi, T.; Sunami, S.; Kamitani, T.; Jinnouchi, M.; Nagao, M.; Akashi, K.; Honda, H. Evaluation of the extent of ground-glass opacity on high-resolution CT in patients with interstitial pneumonia associated with systemic sclerosis: Comparison between quantitative and qualitative analysis. Clin. Radiol. 2014, 69, 758–764. [Google Scholar] [CrossRef]
- Ninaber, M.K.; Stolk, J.; Smit, J.; Le Roy, E.J.; Kroft, L.J.; Bakker, M.E.; de Vries Bouwstra, J.K.; Schouffoer, A.A.; Staring, M.; Stoel, B.C. Lung structure and function relation in systemic sclerosis: Application of lung densitometry. Eur. J. Radiol. 2015, 84, 975–979. [Google Scholar] [CrossRef]
- Riente, L.; Delle Sedie, A.; Filippucci, E.; Iagnocco, A.; Meenagh, G.; Epis, O.; Grassi, W.; Valesini, G.; Montecucco, C.; Bombardieri, S. Ultrasound imaging for the rheumatologist XIV. Ultrasound imaging in connective tissue diseases. Clin. Exp. Rheumatol. 2008, 26, 230–233. [Google Scholar]
- Marten, K.; Dicken, V.; Kneitz, C.; Hoehmann, M.; Kenn, W.; Hahn, D.; Engelke, C. Computer-assisted quantification of interstitial lung disease associated with rheumatoid arthritis: Preliminary technical validation. Eur. J. Radiol. 2009, 72, 278–283. [Google Scholar] [CrossRef] [PubMed]
- Salaffi, F.; Carotti, M.; Bosello, S.; Ciapetti, A.; Gutierrez, M.; Bichisecchi, E.; Giuseppetti, G.; Ferraccioli, G. Computer-aided quantification of interstitial lung disease from high resolution computed tomography images in systemic sclerosis: Correlation with visual reader-based score and physiologic tests. Biomed. Res. Int. 2015, 15, 834262. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salaffi, F.; Carotti, M.; Di Donato, E.; Di Carlo, M.; Ceccarelli, L.; Giuseppetti, G. Computer-aided tomographic analysis of interstitial lung disease (ILD) in patients with systemic sclerosis (SSc). Correlation with pulmonary physiologic tests and patientcentred measures of perceived dyspnea and functional disability. PLoS ONE 2016, 11, e0149240. [Google Scholar] [CrossRef] [PubMed]
- Ariani, A.; Silva, M.; Seletti, V.; Bravi, E.; Saracco, M.; Parisi, S.; De Gennaro, F.; Idolazzi, L.; Caramaschi, P.; Benini, C.; et al. Quantitative chest computed tomography is associated with two prediction models of mortality in interstitial lung disease related to systemic sclerosis. Rheumatology 2017, 56, 922–927. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, H.J.; Brown, M.S.; Elashoff, R.; Li, G.; Gjertson, D.W.; Lynch, D.A.; Strollo, D.C.; Kleerup, E.; Chong, D.; Shah, S.K.; et al. Quantitative texturebased assessment of one-year changes in fibrotic reticular patterns on HRCT in scleroderma lung disease treated with oral cyclophosphamide. Eur. Radiol. 2011, 21, 2455–2465. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Tashkin, D.P.; Gjertson, D.W.; Brown, M.S.; Kleerup, E.; Chong, S.; Belperio, J.A.; Roth, M.D.; Abtin, F.; Elashoff, R.; et al. Transitions to different patterns of interstitial lung disease in scleroderma with and without treatment. Ann. Rheum. Dis. 2016, 75, 1367–1371. [Google Scholar] [CrossRef]
- Bocchino, M.; Bruzzese, D.; D’Alto, M.; Argiento, P.; Borgia, A.; Capaccio, A.; Romeo, E.; Russo, B.; Sanduzzi, A.; Valente, T.; et al. Performance of a new quantitative computed tomography index for interstitial lung disease assessment in systemic sclerosis. Sci. Rep. 2019, 9, 9468. [Google Scholar] [CrossRef]
- Sambataro, D.; Orlandi, M.; Colaci, M.; Malatino, L.; Sambataro, G.; Ariani, A. Correlation between quantitative computed tomography and disease activity in systemic sclerosis. Ann. Rheum. Dis. 2021, 80, 388. [Google Scholar] [CrossRef]
- Occhipinti, M.; Bruni, C.; Camiciottoli, G.; Bartolucci, M.; Bellando-Randone, S.; Bassetto, A.; Cuomo, G.; Giuggioli, D.; Ciardi, G.; Fabbrizzi, A.; et al. Quantitative analysis of pulmonary vasculature in systemic sclerosis at spirometry-gated chest CT. Ann. Rheum. Dis. 2020, 79, 1210–1217. [Google Scholar] [CrossRef]
- Ferro, F.; Delle Sedie, A. The use of ultrasound for assessing interstitial lung involvement in connective tissue diseases. Clin. Exp. Rheumatol. 2018, 36 (Suppl. 114), 165–170. [Google Scholar]
- Reyes-Long, S.; Gutierrez, M.; Clavijo-Cornejo, D.; Alfaro-Rodríguez, A.; González-Sámano, K.; Cortes-Altamirano, J.L.; Muñoz-Louis, R.; Cruz-Arenas, E.; Camargo, K.; Gonzalez, F.; et al. Subclinical Interstitial Lung Disease in Patients with Systemic Sclerosis. A Pilot Study on the Role of Ultrasound. Reumatol. Clin. 2019, 17, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Delle Sedie, A.; Carli, L.; Cioffi, E.; Bombardieri, S.; Riente, L. The promising role of lung ultrasound in systemic sclerosis. Clin. Rheumatol. 2012, 31, 1537–1541. [Google Scholar] [CrossRef]
- Gutierrez, M.; Salaffi, F.; Carotti, M.; Tardella, M.; Pineda, C.; Bertolazzi, C.; Bichisecchi, E.; Filippucci, E.; Grassi, W. Utility of a simplified ultrasound assessment to assess interstitial pulmonary fibrosis in connective tissue disorders--preliminary results. Arthritis Res. Ther. 2011, 13, R134. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Warnecke, K.; Galanski, M.; Peters, E.; Hansen, J. Pneumothorax: Evaluation by ultrasound Preliminary results. J. Thorac. Imaging 1987, 2, 76–78. [Google Scholar] [CrossRef]
- Lichtenstein, D.; Mezière, G.; Biderman, P.; Gepner, A. The comet-tail artifact: An ultrasound sign ruling out pneumothorax. Intensive Care Med. 1999, 25, 383–388. [Google Scholar] [CrossRef] [PubMed]
- Warrick, J.H.; Bhalla, M.; Schabel, S.I.; Silver, R.M. High resolution computed tomography in early scleroderma lung disease. J. Rheumatol. 1991, 18, 1520–1528. [Google Scholar] [PubMed]
- Landis, J.R.; Koch, G.G. The measurement of observer agreement for categorical data. Biometrics 1977, 33, 159–174. [Google Scholar] [CrossRef] [Green Version]
- Salaffi, F.; Manganelli, P.; Carotti, M.; Baldelli, S. The differing patterns of subclinical pulmonary involvement in connective tissue diseases as shown by application of factor analysis. Clin. Rheumatol. 2000, 19, 35–41. [Google Scholar]
- Wells, A.U.; Rubens, M.B.; Du Bois, R.M.; Hansell, D.M. Functional impairment in fibrosing alveolitis: Relationship to reversible disease on thin section computed tomography. Eur. Respir. J. 1997, 10, 280–285. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salaffi, F.; Carotti, M.; Baldelli, S. Subclinical interstitial lung involvement in rheumatic diseases. Correlation of high resolution computerized tomography and functional and cytologic findings. Radiol. Med. 1999, 97, 33–41. [Google Scholar] [PubMed]
- Manolescu, D.; Oancea, C.; Timar, B.; Traila, D.; Malita, D.; Birsasteanu, F.; Tudorache, V. Ultrasound mapping of lung changes in idiopathic pulmonary fibrosis. Clin. Respir. J. 2019, 14, 54–63. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, S.; Lin, J.; Xie, X.; Hu, S.; Lin, Q.; Zheng, K.; Du, G.; Huang, X.; Zhang, G.; et al. Lung ultrasound B-lines and serum KL-6 correlate with the severity of idiopathic inflammatory myositis-associated interstitial lung disease. Rheumatology 2020, 59, 2024–2029. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Srivastava, G.N.; Chokhani, A.; Verma, A.; Siddiqui, Z. Transthoracic ultrasonography in patients with interstitial lung disease. Lung India Off. Organ Indian Chest Soc. 2020, 37, 400–406. [Google Scholar] [CrossRef]
- Singh, A.K.; Mayo, P.H.; Koenig, S.; Talwar, A.; Narasimhan, M. The Use of M-Mode Ultrasonography to Differentiate the Causes of B Lines. Chest 2018, 153, 689–696. [Google Scholar] [CrossRef] [PubMed]
- Soldati, G.; Demi, M.; Inchingolo, R.; Smargiassi, A.; Demi, L. On the Physical Basis of Pulmonary Sonographic Interstitial Syndrome. J. Ultrasound Med. 2016, 35, 2075–2086. [Google Scholar] [CrossRef]
- Buda, N.; Kosiak, W. Is a linear probe helpful in diagnosing diseases of pulmonary interstitial spaces? J. Ultrason. 2017, 17, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Buda, N.; Skoczylas, A.; Demi, M.; Wojteczek, A.; Cylwik, J.; Soldati, G. Clinical Impact of Vertical Artifacts Changing with Frequency in Lung Ultrasound. Diagnostics 2021, 11, 401. [Google Scholar] [CrossRef] [PubMed]
- Barskova, T.; Gargani, L.; Guiducci, S.; Randone, S.B.; Bruni, C.; Carnesecchi, G.; Conforti, M.L.; Porta, F.; Pignone, A.; Caramella, D.; et al. Lung ultrasound for the screening of interstitial lung disease in very early systemic sclerosis. Ann. Rheum. Dis. 2013, 72, 390–395. [Google Scholar] [CrossRef]
- Xie, H.Q.; Zhang, W.W.; Sun, D.S.; Chen, X.M.; Yuan, S.F.; Gong, Z.H.; Liu, L. A simplified lung ultrasound for the diagnosis of interstitial lung disease in connective tissue disease: A meta-analysis. Arthritis Res. 2019, 21, 93. [Google Scholar] [CrossRef] [Green Version]
- Song, G.G.; Bae, S.-C.; Lee, Y.H. Diagnostic accuracy of lung ultrasound for interstitial lung disease in patients with connective tissue diseases: A meta-analysis. Clin. Exp. Rheumatol. 2016, 34, 11–16. [Google Scholar]
- Gasperini, M.L.; Gigante, A.; Iacolare, A.; Pellicano, C.; Lucci, S.; Rosato, E. The predictive role of lung ultrasound in progression of scleroderma interstitial lung disease. Clin. Rheumatol. 2020, 39, 119–123. [Google Scholar] [CrossRef] [PubMed]
- Gargani, L.; Bruni, C.; Romei, C.; Frumento, P.; Moreo, A.; Agoston, G.; Guiducci, S.; Bellando-Randone, S.; Lepri, G.; Belloli, L.; et al. Prognostic Value of Lung Ultrasound B-Lines in Systemic Sclerosis. Chest 2020, 158, 1515–1525. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, M.; Soto-Fajardo, C.; Pineda, C.; Alfaro-Rodriguez, A.; Terslev, L.; Bruyn, G.; Iagnocco, A.; Bertolazzi, C.; D’Agostino, M.A.; Delle Sedie, A. Ultrasound in the Assessment of Interstitial Lung Disease in Systemic Sclerosis: A Systematic Literature Review by the OMERACT Ultrasound Group. J. Rheumatol. 2020, 47, 991–1000. [Google Scholar] [CrossRef] [PubMed]
- Gigante, A.; Fanelli, F.R.; Lucci, S.; Barilaro, G.; Quarta, S.; Barbano, B.; Giovannetti, A.; Amoroso, A.; Rosato, E. Lung ultrasound in systemic sclerosis: Correlation with high-resolution computed tomography, pulmonary function tests and clinical variables of disease. Intern. Emerg. Med. 2016, 11, 213–217. [Google Scholar] [CrossRef] [Green Version]
- Moazedi-Fuerst, F.C.; Zechner, P.M.; Tripolt, N.J.; Kielhauser, S.M.; Brickmann, K.; Scheidl, S.; Lutfi, A.; Graninger, W.G. Pulmonary echography in systemic sclerosis. Clin. Rheumatol. 2012, 31, 1621–1625. [Google Scholar] [CrossRef] [PubMed]
- Moazedi-Fuerst, F.C.; Kielhauser, S.; Brickmann, K.; Tripolt, N.; Meilinger, M.; Lufti, A.; Graninger, W. Sonographic assessment of interstitial lung disease in patients with rheumatoid arthritis, systemic sclerosis and systemic lupus erythematosus. Clin. Exp. Rheumatol. 2015, 33 (Suppl. 91), S87–S91. [Google Scholar] [PubMed]
- Sperandeo, M.; De Cata, A.; Molinaro, F.; Trovato, F.M.; Catalano, D.; Simeone, A.; Varriale, A.; Martines, G.F.; Trovato, G. Ultrasound signs of pulmonary fibrosis in systemic sclerosis as timely indicators for chest computed tomography. Scand. J. Rheumatol. 2015, 44, 389–398. [Google Scholar] [CrossRef]
- Pinal-Fernandez, I.; Pallisa-Nuñez, E.; Selva-O’Callaghan, A.; Castella-Fierro, E.; Simeon-Aznar, C.P.; Fonollosa-Pla, V.; Vilardell-Tarres, M. Pleural irregularity, a new ultrasound sign for the study of interstitial lung disease in systemic sclerosis and antisynthetase syndrome. Clin. Exp. Rheumatol. 2015, 33 (Suppl. 91), S136–S141. [Google Scholar]
- Çakir Edis, E.; Hatipoğlu, O.N.; Pamuk, Ö.N.; Mutlucan Eraslan, R.; Aktöz, M.; Tuncel, S.A. Effectiveness of Thoracic Ultrasonography in the Evaluation of the Severity of Pulmonary Involvement in Patients with Systemic Sclerosis. Arch. Rheumatol. 2016, 31, 364–370. [Google Scholar] [CrossRef] [Green Version]
- Buda, N.; Piskunowicz, M.; Porzezińska, M.; Kosiak, W.; Zdrojewski, Z. Lung Ultrasonography in the Evaluation of Interstitial Lung Disease in Systemic Connective Tissue Diseases: Criteria and Severity of Pulmonary Fibrosis—Analysis of 52 Patients. Ultraschall Med. 2016, 37, 379–385. [Google Scholar] [CrossRef]
- Tardella, M.; Di Carlo, M.; Carotti, M.; Filippucci, E.; Grassi, W.; Salaffi, F. Ultrasound B-lines in the evaluation of interstitial lung disease in patients with systemic sclerosis: Cut-off point definition for the presence of significant pulmonary fibrosis. Medicine 2018, 97, e0566. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruaro, B.; Baratella, E.; Confalonieri, P.; Confalonieri, M.; Vassallo, F.G.; Wade, B.; Geri, P.; Pozzan, R.; Caforio, G.; Marrocchio, C.; et al. High-Resolution Computed Tomography and Lung Ultrasound in Patients with Systemic Sclerosis: Which One to Choose? Diagnostics 2021, 11, 2293. https://doi.org/10.3390/diagnostics11122293
Ruaro B, Baratella E, Confalonieri P, Confalonieri M, Vassallo FG, Wade B, Geri P, Pozzan R, Caforio G, Marrocchio C, et al. High-Resolution Computed Tomography and Lung Ultrasound in Patients with Systemic Sclerosis: Which One to Choose? Diagnostics. 2021; 11(12):2293. https://doi.org/10.3390/diagnostics11122293
Chicago/Turabian StyleRuaro, Barbara, Elisa Baratella, Paola Confalonieri, Marco Confalonieri, Fabio Giuseppe Vassallo, Barbara Wade, Pietro Geri, Riccardo Pozzan, Gaetano Caforio, Cristina Marrocchio, and et al. 2021. "High-Resolution Computed Tomography and Lung Ultrasound in Patients with Systemic Sclerosis: Which One to Choose?" Diagnostics 11, no. 12: 2293. https://doi.org/10.3390/diagnostics11122293
APA StyleRuaro, B., Baratella, E., Confalonieri, P., Confalonieri, M., Vassallo, F. G., Wade, B., Geri, P., Pozzan, R., Caforio, G., Marrocchio, C., Cova, M. A., & Salton, F. (2021). High-Resolution Computed Tomography and Lung Ultrasound in Patients with Systemic Sclerosis: Which One to Choose? Diagnostics, 11(12), 2293. https://doi.org/10.3390/diagnostics11122293







