Leaching of Chalcopyrite in Acidified Nitrate Using Seawater-Based Media
Abstract
1. Introduction
2. Materials and Methods
2.1. Ore Sample
2.2. Reagents
2.3. Experimental Procedure
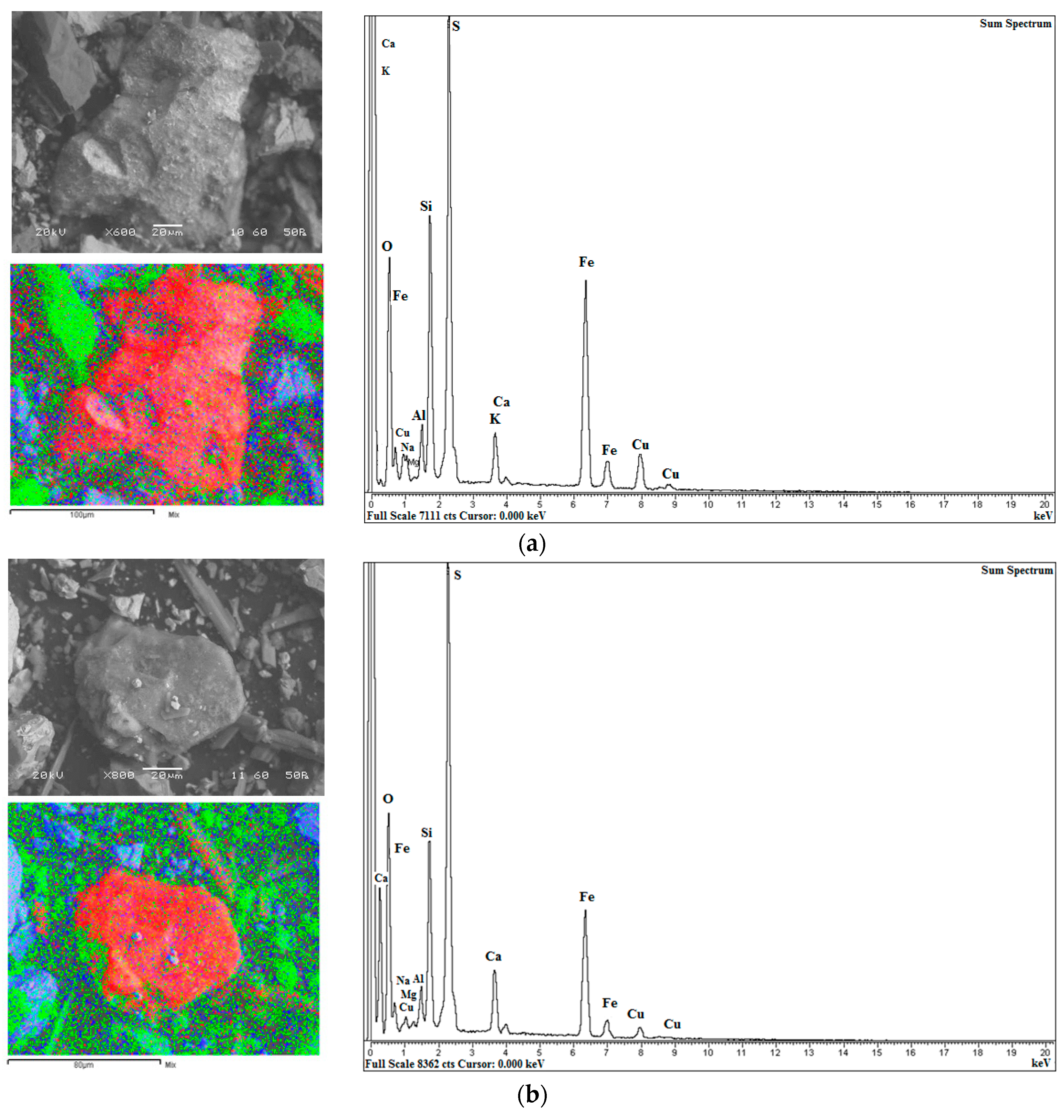
2.4. Characterization of Ore Residues
3. Results and Discussion
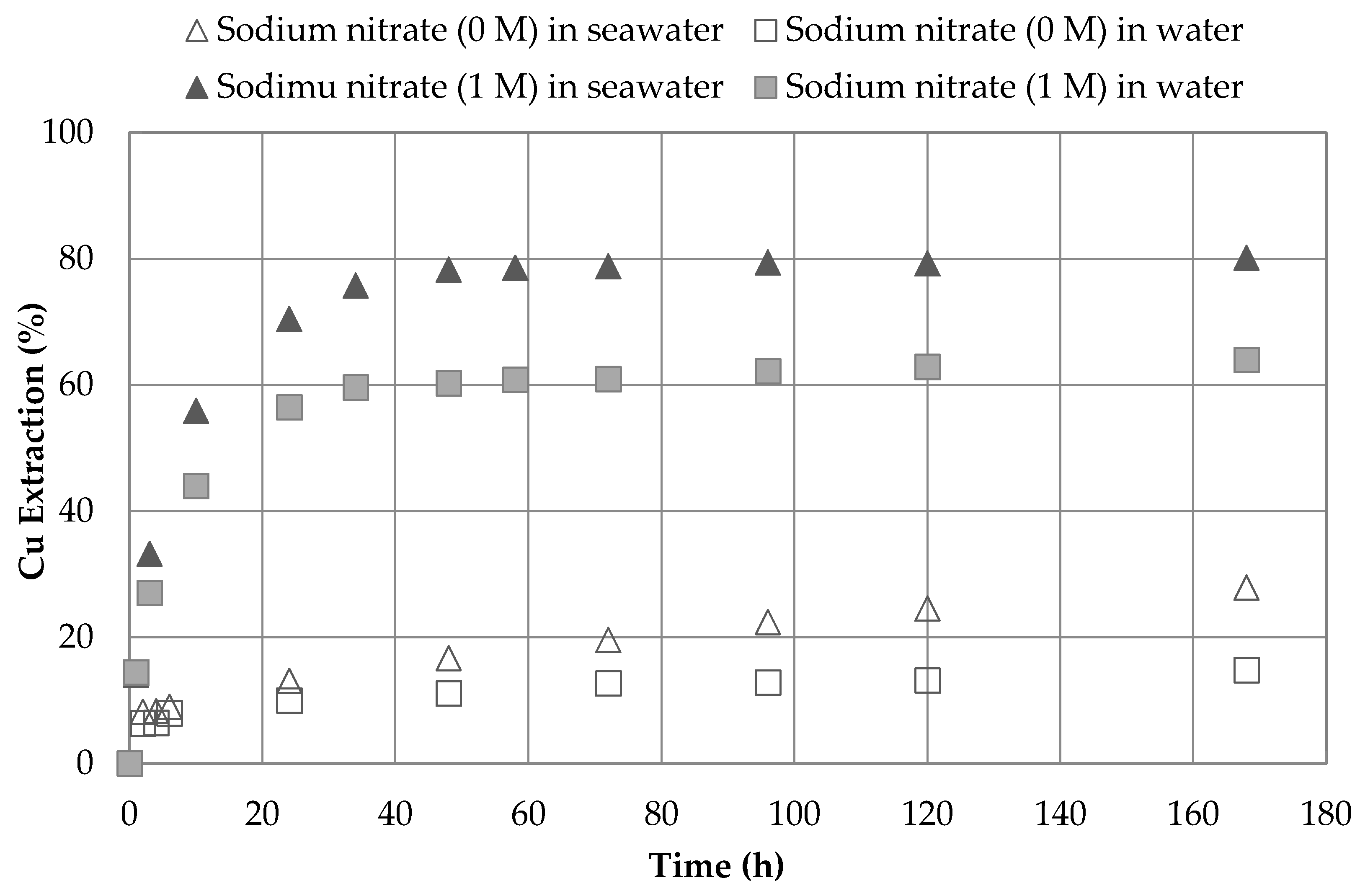
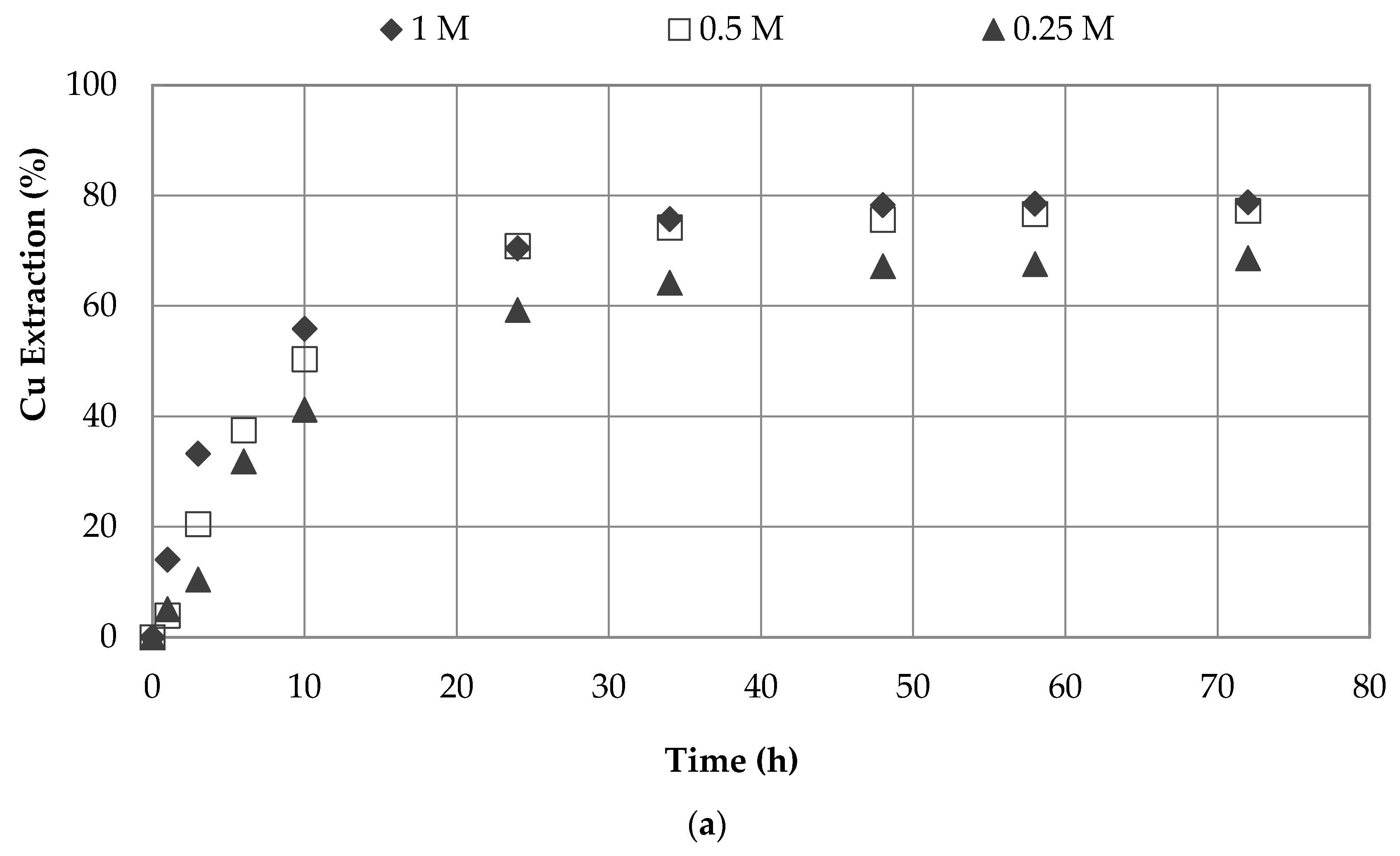
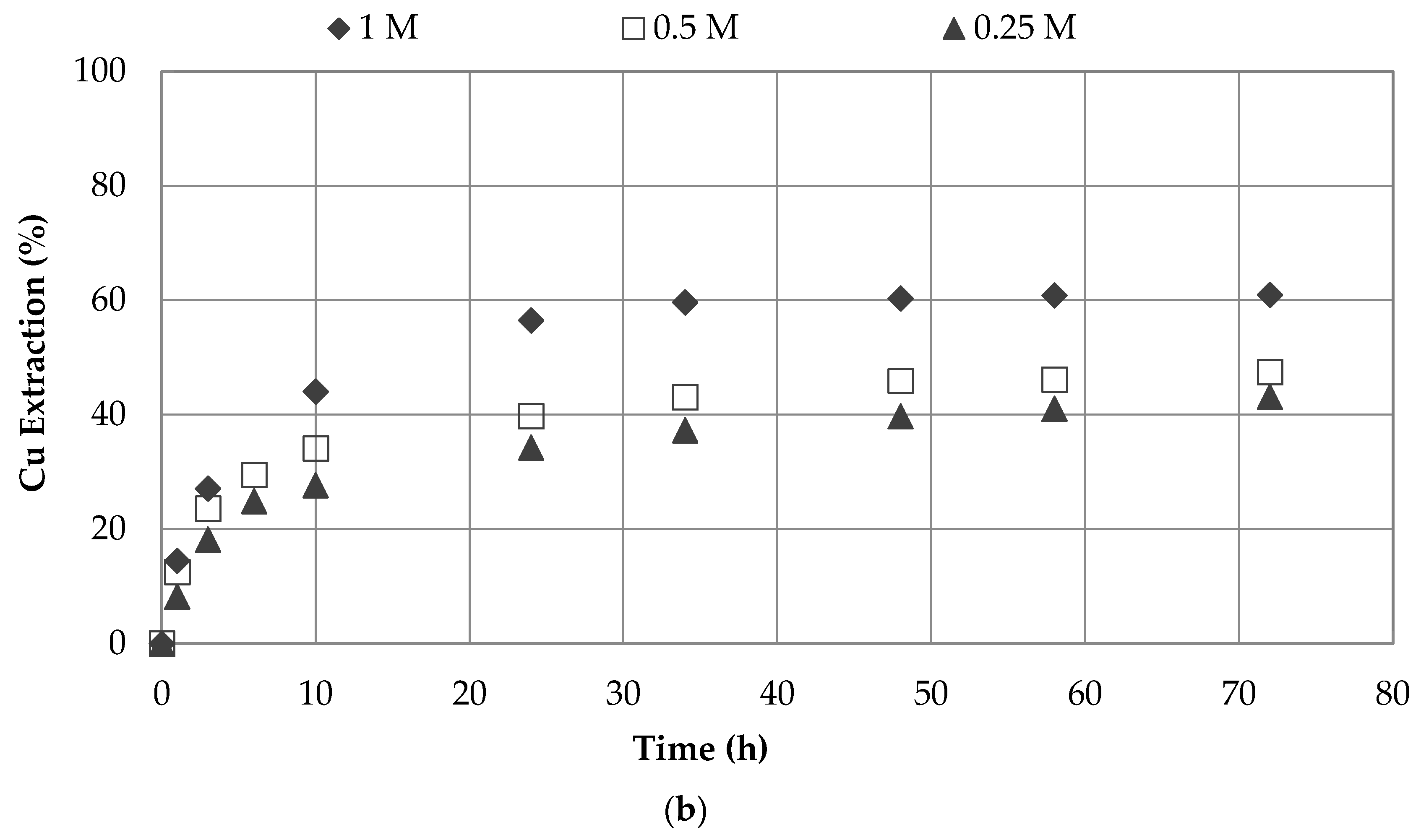
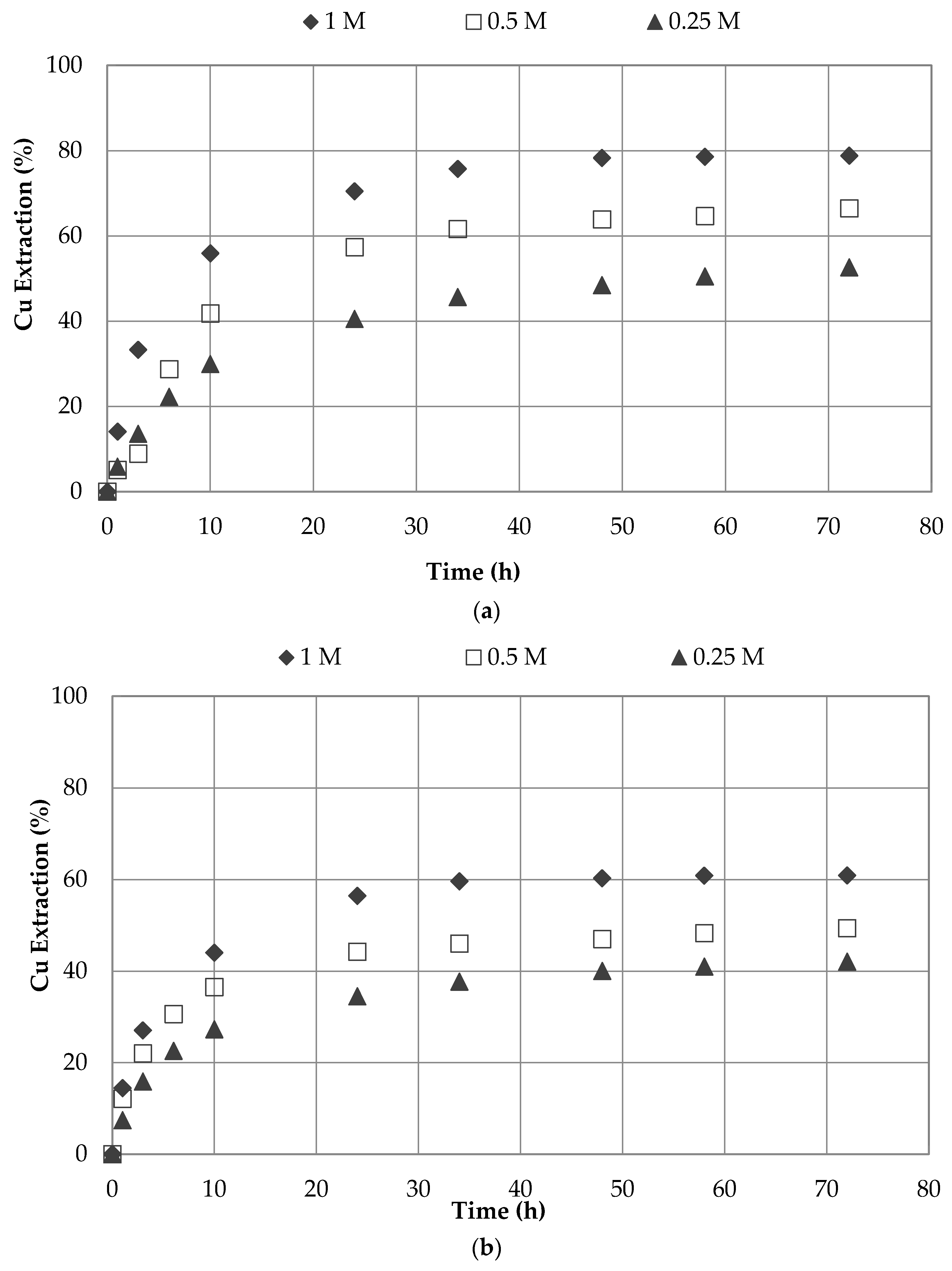
3.1. Effect of Sodium Nitrate and Concentrations
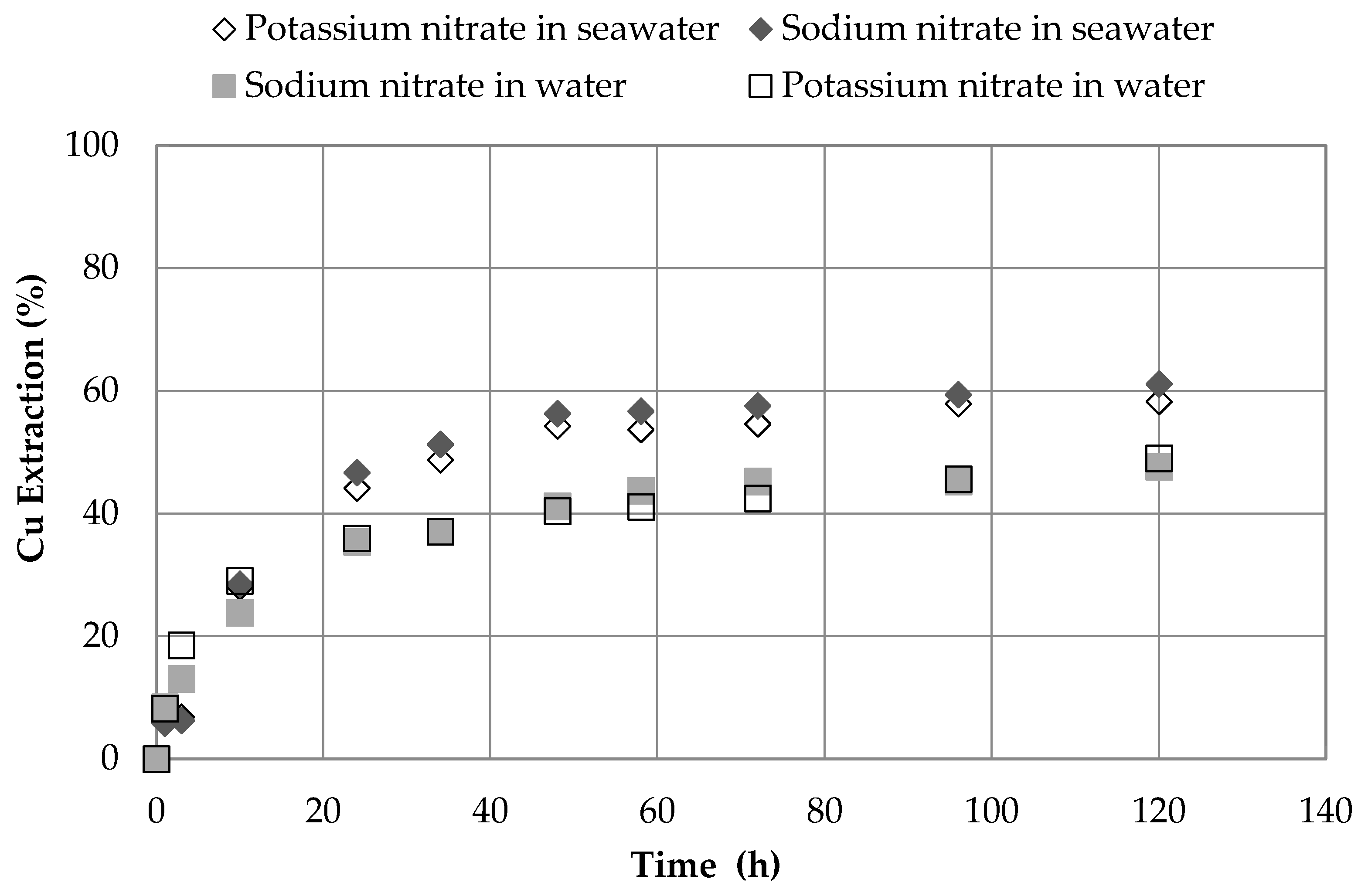
3.2. Effect of the Nitrate Source
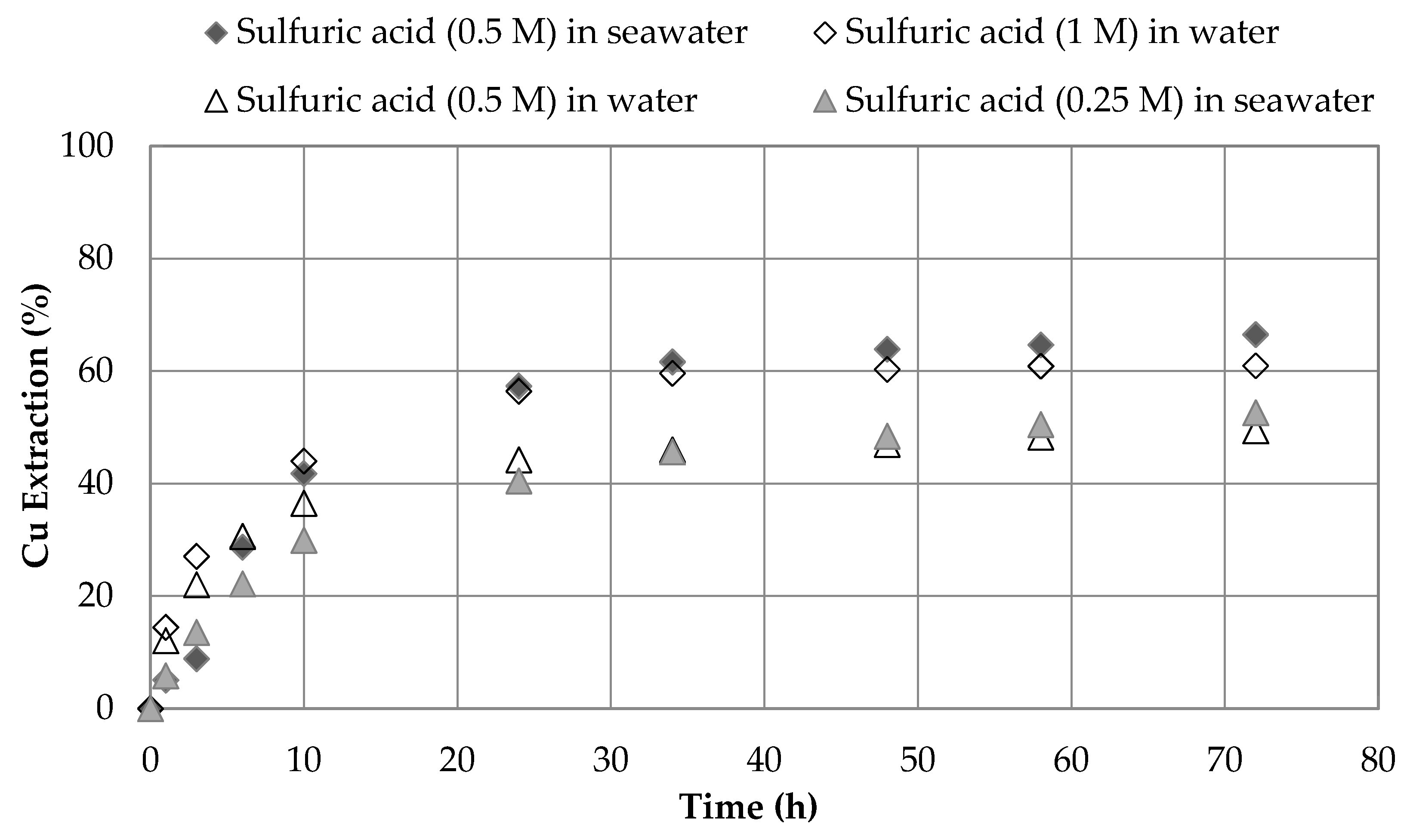
3.3. Effect of Sulfuric Acid Concentration
3.4. Redox Potential and pH
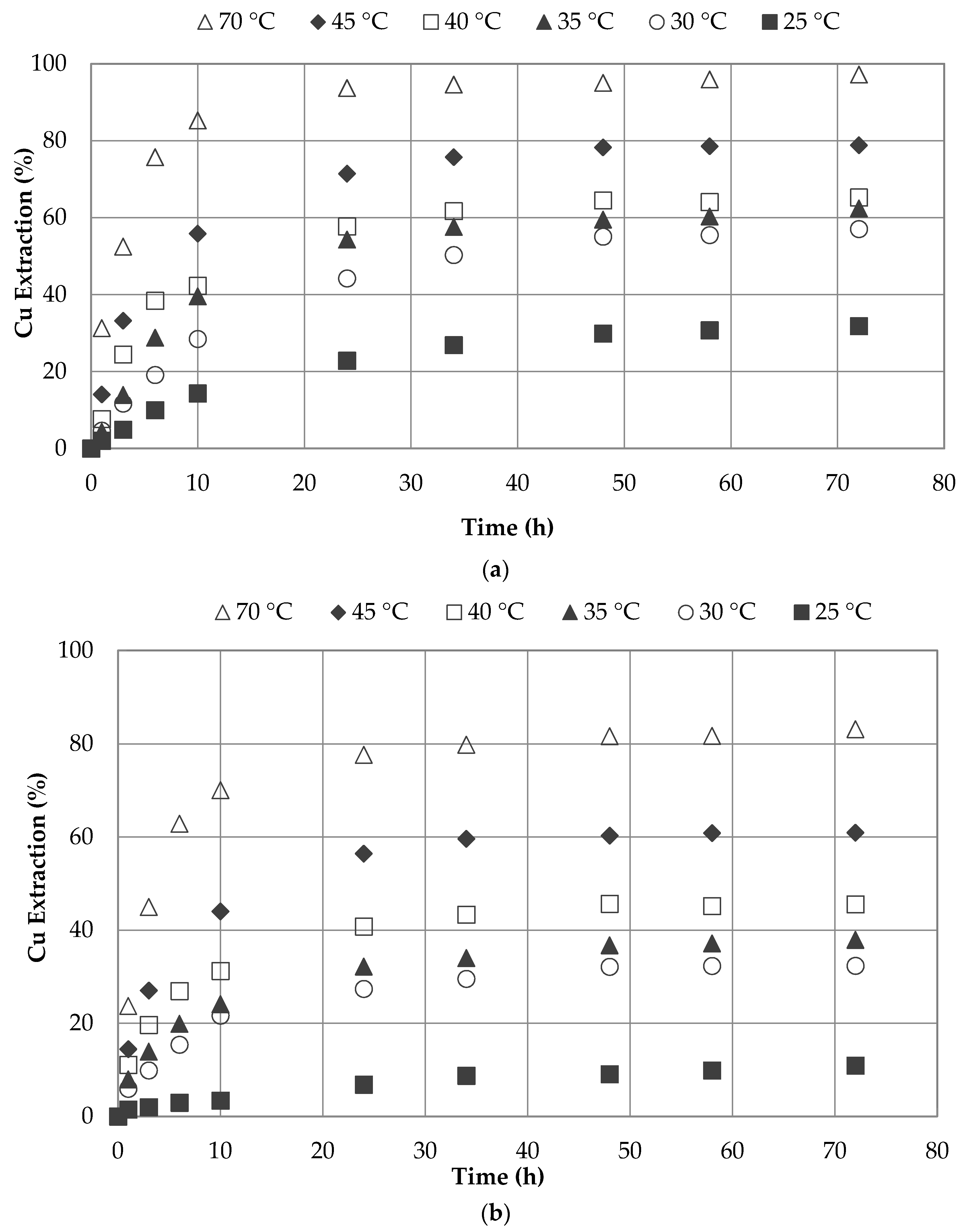
3.5. Effect of Temperature
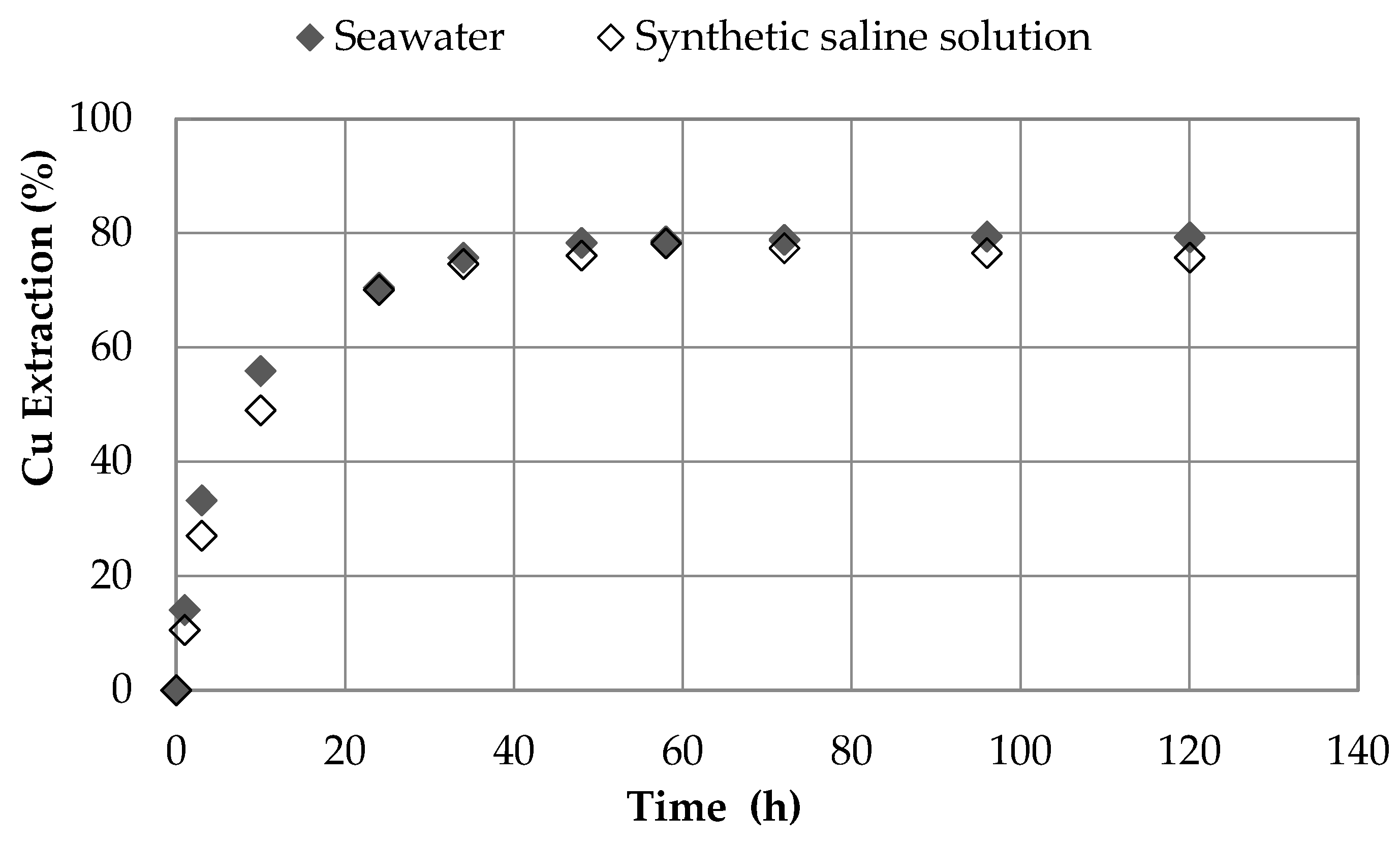
3.6. Effect of Synthetic Saline Solution
3.7. Characterization of Ore and Residue Samples
3.8. Thermodynamic and Chemical Reactions
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Liddicoat, J.; Dreisinger, D. Chloride leaching of chalcopyrite. Hydrometallurgy 2007, 89, 323–331. [Google Scholar] [CrossRef]
- Lu, Z.Y.; Jeffrey, M.I.; Lawson, F. The effect of chloride ions on the dissolution of chalcopyrite in acidic solutions. Hydrometallurgy 2000, 56, 189–202. [Google Scholar] [CrossRef]
- Velásquez-Yévenes, L.; Nicol, M.; Miki, H. The dissolution of chalcopyrite in chloride solutions: Part 1. The effect of solution potential. Hydrometallurgy 2010, 103, 108–113. [Google Scholar] [CrossRef]
- Herreros, O.; Viñals, J. Leaching of sulfide copper ore in a NaCl-H2SO4-O2 media with acid pre-treatment. Hydrometallurgy 2007, 89, 260–268. [Google Scholar] [CrossRef]
- Ibáñez, T.; Velásquez, L. Lixiviación de la calcopirita en medios clorurados. Rev. Metal. 2013, 49, 131–144. [Google Scholar] [CrossRef]
- Yoo, K.; Kim, S.-K.; Lee, J.-C.; Ito, M. Tsunekawa, and N. Hiroyoshi. Effect of chloride ions on leaching rate of chalcopyrite. Miner. Eng. 2010, 23, 471–477. [Google Scholar] [CrossRef]
- Watling, H. Chalcopyrite hydrometallurgy at atmospheric pressure: 2. Review of acidic chloride process options. Hydrometallurgy 2014, 146, 96–110. [Google Scholar] [CrossRef]
- Winand, R. Chloride hydrometallurgy. Hydrometallurgy 1991, 27, 285–316. [Google Scholar] [CrossRef]
- Senanayake, G. A review of chloride assisted copper sulfide leaching by oxygenated sulfuric acid and mechanistic considerations. Hydrometallurgy 2009, 98, 21–32. [Google Scholar] [CrossRef]
- Al-Harahsheh, M.; Kingman, S.; Al-Harahsheh, A. Ferric chloride leaching of chalcopyrite: Synergetic effect of CuCl2. Hydrometallurgy 2008, 91, 89–97. [Google Scholar] [CrossRef]
- Lundström, M.; Aromaa, J.; Forsén, O.; Hyvärinen, O.; Barker, M.H. Leaching of chalcopyrite in cupric chloride solution. Hydrometallurgy 2005, 77, 89–95. [Google Scholar] [CrossRef]
- Lundström, M.; Liipo, J.; Aromaa, J. Dissolution of copper and iron from sulfide concentrates in cupric chloride solution. Int. J. Miner. Process. 2012, 102–103, 13–18. [Google Scholar] [CrossRef]
- O’Malley, M.; Liddell, K. Leaching of CuFeS2 by aqueous FeCl3, HCl, and NaCl: Effects of solution composition and limited oxidant. Metall. Mater. Trans. B. 1987, 18, 505–510. [Google Scholar] [CrossRef]
- Moreno, P.; Aral, H.; Cuevas, J.; Monardes, A.; Adaro, M.; Norgate, T.; Bruckard, W. The use of seawater as process water at Las Luces copper–molybdenum beneficiation plant in Taltal (Chile). Miner. Eng. 2011, 24, 852–858. [Google Scholar] [CrossRef]
- Aroca, F.; Backit, A.; Jacob, J. CuproChlor®, a hydrometallurgical technology for mineral sulphides leaching. In Hydroprocess 2012, Proceedings of the 4th International Seminar on Process Hydrometallurgy; Casas, S.J.C., Ciminelli, V., Montes-Atenas, G., Stubina, N., Eds.; GECAMIN LTDA: Santiago, Chile, 2012; pp. 96–180. [Google Scholar]
- COCHILCO. Consumo de Agua en La minería del Cobre al 2016; Gobierno de Chile, Ministerio de Minería, Ed.; Registro Propiedad Intelectual: Santiago, Chile, 2017.
- Torres, C.; Taboada, M.; Graber, T.; Herreros, O.; Ghorbani, Y.; Watling, H. The effect of seawater based media on copper dissolution from low-grade copper ore. Miner. Eng. 2015, 71, 139–145. [Google Scholar] [CrossRef]
- Hernández, P.; Taboada, M.; Herreros, O.; Torres, C.; Ghorbani, Y. Chalcopyrite dissolution using seawater-based acidic media in the presence of oxidants. Hydrometallurgy 2015, 157, 325–332. [Google Scholar] [CrossRef]
- Taboada, M.; Hernández, P.; Galleguillos, H.; Flores, E.; Graber, T. Behavior of sodium nitrate and caliche mineral in seawater: Solubility and physicochemical properties at different temperatures and concentrations. Hydrometallurgy 2012, 113, 160–166. [Google Scholar] [CrossRef]
- Valencia, J.A.; Méndez, D.A.; Cueto, J.Y.; Cisternas, L.A. Saltpeter extraction and modelling of caliche mineral heap leaching. Hydrometallurgy 2008, 90, 103–114. [Google Scholar] [CrossRef]
- Ordóñez, J.I.; Moreno, L.; Gálvez, E.D.; Cisternas, L.A. Seawater leaching of caliche mineral in column experiments. Hydrometallurgy 2013, 139, 79–87. [Google Scholar] [CrossRef]
- Torres, M.A.; Meruane, G.E.; Graber, T.A.; Gutiérrez, P.C.; Taboada, M.E. Recovery of nitrates from leaching solutions using seawater. Hydrometallurgy 2013, 133, 100–105. [Google Scholar] [CrossRef]
- Habashi, F. Nitric Acid in the Hydrometallurgy of Sulfides. In EPD Congress 1999; TMS: Baltimore, MD, USA, 1999. [Google Scholar]
- Peters, E. Hydrometallurgical process innovation. Hydrometallurgy 1992, 29, 431–459. [Google Scholar] [CrossRef]
- Prasad, S.; Pandey, B. Alternative processes for treatment of chalcopyrite—A review. Miner. Eng. 1998, 11, 763–781. [Google Scholar] [CrossRef]
- Kazakov, A.; Rubtsov, Y.I.; Andrienko, L.; Manelis, G. Kinetics and mechanism of thermal decomposition of nitric acid in sulfuric acid solutions. Bull. Acad. Sci. USSR Div. Chem. Sci. 1987, 36, 1999–2002. [Google Scholar] [CrossRef]
- Baldwin, S.A.; van Weert, G. On the catalysis of ferrous sulphate oxidation in autoclaves by nitrates and nitrites. Hydrometallurgy 1996, 42, 209–219. [Google Scholar] [CrossRef]
- Sokić, M.D.; Marković, B.; Živković, D. Kinetics of chalcopyrite leaching by sodium nitrate in sulphuric acid. Hydrometallurgy 2009, 95, 273–279. [Google Scholar] [CrossRef]
- Gok, O.; Anderson, C.G. Dissolution of low-grade chalcopyrite concentrate in acidified nitrite electrolyte. Hydrometallurgy 2013, 134–135, 40–46. [Google Scholar] [CrossRef]
- Vračar, R.Ž.; Vučković, N.; Kamberović, Ž. Leaching of copper(I) sulphide by sulphuric acid solution with addition of sodium nitrate. Hydrometallurgy 2003, 70, 143–151. [Google Scholar] [CrossRef]
- Watling, H. Chalcopyrite hydrometallurgy at atmospheric pressure: 1. Review of acidic sulfate, sulfate-chloride and sulfate-nitrate process options. Hydrometallurgy 2013, 140, 163–180. [Google Scholar] [CrossRef]
- Anderson, C.G. Treatment of copper ores and concentrates with industrial nitrogen species catalyzed pressure leaching and non-cyanide precious metals recovery. JOM 2003, 55, 32–36. [Google Scholar] [CrossRef]
- Tsogtkhangai, D.; Mamyachenkov, S.V.; Anisimova, O.S.; Naboichenko, S.S. Kinetics of leaching of copper concentrates by nitric acid. Russ. J. Non Ferr. Met. 2011, 52, 469–472. [Google Scholar] [CrossRef]
- Tsogtkhangai, D.; Mamyachenkov, S.V.; Anisimova, O.S.; Naboichenko, S.S. Thermodynamics of reactions during nitric acid leaching of minerals of a copper concentrate. Russ. J. Non Ferr. Met. 2011, 52, 135–139. [Google Scholar] [CrossRef]
- Arias, J.A. Heap leaching copper ore using sodium nitrate. U.S. Patent US6,569,391B1, 27 May 2003. [Google Scholar]
- Queneau, P.; Prater, J. Nitric acid process for recovering metal values from sulfide ore materials containing iron sulfides. U.S. Patent US3,793,429, 19 Februar 1974. [Google Scholar]
- Antivachis, D.N.; Chatzitheodoridis, E.; Skarpelis, N.; Komnitsas, K. Secondary sulphate minerals in a cyprus-type ore deposit, Apliki, Cyprus: mineralogy and its implications regarding the chemistry of Pit Lake waters. Mine Water Environ. 2017, 36, 226–238. [Google Scholar] [CrossRef]
- Shiers, D.; Collinson, D.; Kelly, N.; Watling, H. Copper extraction from chalcopyrite: Comparison of three non-sulfate oxidants, hypochlorous acid, sodium chlorate and potassium nitrate, with ferric sulfate. Miner. Eng. 2016, 85, 55–65. [Google Scholar] [CrossRef]
- Alguacil, F.J.; Cobo, A.; Alonso, M. Copper separation from nitrate/nitric acid media using Acorga M5640 extractant: Part I: solvent extraction study. Chem. Eng. J. 2002, 85, 259–263. [Google Scholar] [CrossRef]
- Eyzaguirre, D. Efecto del nitrato en la extracción por solventes de Cía. Minera Lomas Bayas. In Proceedings of the VI Exposición Mundial Para la Minería Latinoamericana EXPOMIN, Seminario Innovación Tecnológica en Minería; Instituto de Ingenieros de Minas de Chile: Santiago, Chile, 2000. [Google Scholar]
- Yáñez, H.; Soto, A.; Soderstrom, M.; Bednarski, T. Nitration in copper SX? Cytec Acorga provides a new reagent. In Hydrocopper 2009; Domic, E., Casas, J., Eds.; GECAMIN LTDA: Antofagasta, Chile, 2009; pp. 332–341. [Google Scholar]
- Zambra, R.; Quilodrán, A.; Rivera, G.; Castro, O. Use of NR® reagents in presence of nitrate ion in SX: A revision of the present moment. In Hydroprocess 2013; GECAMIN LTDA: Santiago, Chile, 2013. [Google Scholar]
- Hamzah, B.; Jalaluddin, N.; Wahab, A.W.; Upe, A. Copper (II) Extraction from Nitric Acid Solution with 1-Phenyl-3-methyl-4-benzoyl-5-pyrazolone as a Cation Carrier by Liquid Membrane Emulsion. J. Chem. 2010, 7, 239–245. [Google Scholar] [CrossRef]
- Virnig, M.J.; Mattison, P.L.; Hein, H.C. Processes for the recovery of copper from aqueous solutions containing nitrate ions. U.S. Patent 6,702,872, 9 March 2004. [Google Scholar]
- Puvvada, G.V.K.; Sridhar, R.; Lakshmanan, V. Chloride metallurgy: PGM recovery and titanium dioxide production. J. Miner. Met. Mater. Soc. 2003, 55, 38–41. [Google Scholar] [CrossRef]
- Senanayake, G. Review of theory and practice of measuring proton activity and pH in concentrated chloride solutions and application to oxide leaching. Miner. Eng. 2007, 20, 634–645. [Google Scholar] [CrossRef]
- Narangarav, T.; Nyamdelger, S.; Ariunaa, G.; Azzaya, T.; Burmaa, G. Dissolution behavior of copper concentrate in acidic media using nitrate ions. Mong. J. Chem. 2014, 15, 79–84. [Google Scholar] [CrossRef]


| Mineralogy | Chemical Analysis | ||
|---|---|---|---|
| Minerals | (wt %) | Element | (wt %) |
| Magnetite (Fe3O4) | 36.7 | Iron (Fe) | 33.6 |
| Quartz (SiO2) | 17.1 | Silicon (Si) | 10.9 |
| Plagioclase ((Ca,Na)(Al,Si)AlSi2O8) | 9.1 | Calcium (Ca) | 4.1 |
| Pyrite (FeS2) | 8.1 | Sulphur (S) | 3.8 |
| Chalcopyrite (CuFeS2) | 4.8 | Aluminium (Al) | 2.8 |
| Calcite (CaCO3) | 4.6 | Sodium (Na) | 1.8 |
| Kaolinite (Al4(Si4O10)(OH)8) | 4.3 | Copper (Cu) * | 1.6 |
| Garnet (Ca3Fe2Si3O12) | 3.7 | Magnesium (Mg) | 0.9 |
| Actinolite (Ca2(Mg,Fe)2.5Si8O22(OH)2) | 3.5 | Manganese (Mn) | 0.6 |
| Amphibole (NaCa2(Mg,Fe,Al)5(Si,Al)8O22(OH)2) | 3.5 | Potassium (K) | 0.6 |
| Epidote (Ca2Al2FeSi3O12(OH)) | 2.6 | Cobalt (Co) | 0.4 |
| Sericite (KAl2(AlSi3O10)(OH)2) | ≤1.0 | Chromium (Cr) | 0.1 |
| Chlorite ((Mg,Fe)3(AlSi)4O10(OH)2(Mg,Fe)3(OH)6) | ≤1.0 | Nickel (Ni) | 0.1 |
| Zinc (Zn) | <0.1 | ||
| Molybdenum (Mo) | <0.1 | ||
| Silver (Ag) | <0.1 | ||
| Chemical Method | ICP-AES | AAS | Volumetric Analysis | Gravimetric Analysis | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Ionic Species | Na+ | Mg2+ | Ca2+ | K+ | B3+ | Cu2+ | NO3− | Cl− | HCO3− | SO42− |
| mg·L−1 | 9480 | 1190 | 386 | 374 | 4.6 | 0.07 | 2.05 | 18,765 | 142 | 2771 |
| No. | H2SO4 (M) | NaNO3 (M) | Water Type | T (°C) | t (Days) | Final Cu Extraction (wt %) |
|---|---|---|---|---|---|---|
| 1 | 1.0 | 1.0 | seawater | 45 | 7 | 80.2 |
| 1.0 | 1.0 | water | 45 | 7 | 63.9 | |
| 2 | 1.0 | 0.0 | seawater | 45 | 7 | 27.9 |
| 1.0 | 0.0 | water | 45 | 7 | 14.8 | |
| 3 | 1.0 | 1.0 | seawater | 45 | 3 | 78.8 |
| 1.0 | 1.0 | water | 45 | 3 | 61.0 | |
| 4 | 1.0 | 1.0 | seawater | 40 | 3 | 65.3 |
| 1.0 | 1.0 | water | 40 | 3 | 45.6 | |
| 5 | 1.0 | 1.0 | seawater | 35 | 3 | 62.4 |
| 1.0 | 1.0 | water | 35 | 3 | 37.2 | |
| 6 | 1.0 | 1.0 | seawater | 30 | 3 | 57.1 |
| 1.0 | 1.0 | water | 30 | 3 | 35.9 | |
| 7 | 0.5 | 1.0 | seawater | 45 | 3 | 66.5 |
| 0.5 | 1.0 | water | 45 | 3 | 49.4 | |
| 8 | 0.25 | 1.0 | seawater | 45 | 3 | 52.6 |
| 0.25 | 1.0 | water | 45 | 3 | 42.1 | |
| 9 | 1.0 | 0.5 | seawater | 45 | 3 | 77.3 |
| 1.0 | 0.5 | water | 45 | 3 | 47.4 | |
| 10 | 1.0 | 0.25 | seawater | 45 | 3 | 68.6 |
| 1.0 | 0.25 | water | 45 | 3 | 43.1 | |
| 11 | 1.0 | 1.0 | seawater | 45 | 5 | 79.3 |
| 1.0 | 1.0 | water | 45 | 5 | 62.9 | |
| 12 | 0.5 | 0.5 | seawater | 45 | 5 | 61.2 |
| 0.5 | 0.5 | water | 45 | 5 | 47.7 | |
| 13 | 0.5 | 0.5 * | seawater | 45 | 5 | 58.3 |
| 0.5 | 0.5 * | water | 45 | 5 | 49.1 | |
| 14 | 1.0 | 1.0 | saline solution | 45 | 5 | 75.7 |
| 15 | 1.0 | 1.0 | seawater | 70 | 3 | 97.2 |
| 1.0 | 1.0 | water | 70 | 3 | 83.2 | |
| 16 | 1.0 | 1.0 | seawater | 25 | 3 | 31.8 |
| 1.0 | 1.0 | water | 25 | 3 | 11.0 |
| No. | ΔG25 °C (kJ·mol−1) | ΔG70 °C (kJ·mol−1) |
|---|---|---|
| (7) | −1195.8 | −1205.8 |
| (8) | −1351.5 | −1338.8 |
| (9) | −340.0 | −413.6 |
| (10) | −417.9 | −480.2 |
| (11) | −245.7 | −274.8 |
| (12) | −28.5 | −38.2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hernández, P.C.; Taboada, M.E.; Herreros, O.O.; Graber, T.A.; Ghorbani, Y. Leaching of Chalcopyrite in Acidified Nitrate Using Seawater-Based Media. Minerals 2018, 8, 238. https://doi.org/10.3390/min8060238
Hernández PC, Taboada ME, Herreros OO, Graber TA, Ghorbani Y. Leaching of Chalcopyrite in Acidified Nitrate Using Seawater-Based Media. Minerals. 2018; 8(6):238. https://doi.org/10.3390/min8060238
Chicago/Turabian StyleHernández, Pía C., María E. Taboada, Osvaldo O. Herreros, Teófilo A. Graber, and Yousef Ghorbani. 2018. "Leaching of Chalcopyrite in Acidified Nitrate Using Seawater-Based Media" Minerals 8, no. 6: 238. https://doi.org/10.3390/min8060238
APA StyleHernández, P. C., Taboada, M. E., Herreros, O. O., Graber, T. A., & Ghorbani, Y. (2018). Leaching of Chalcopyrite in Acidified Nitrate Using Seawater-Based Media. Minerals, 8(6), 238. https://doi.org/10.3390/min8060238







