Advances towards a Clean Hydrometallurgical Process for Chromite
Abstract
:1. Introduction
2. Chromium Salt Preparation Technology
3. Leaching Process of Chromite in Sulfuric Acid Solution
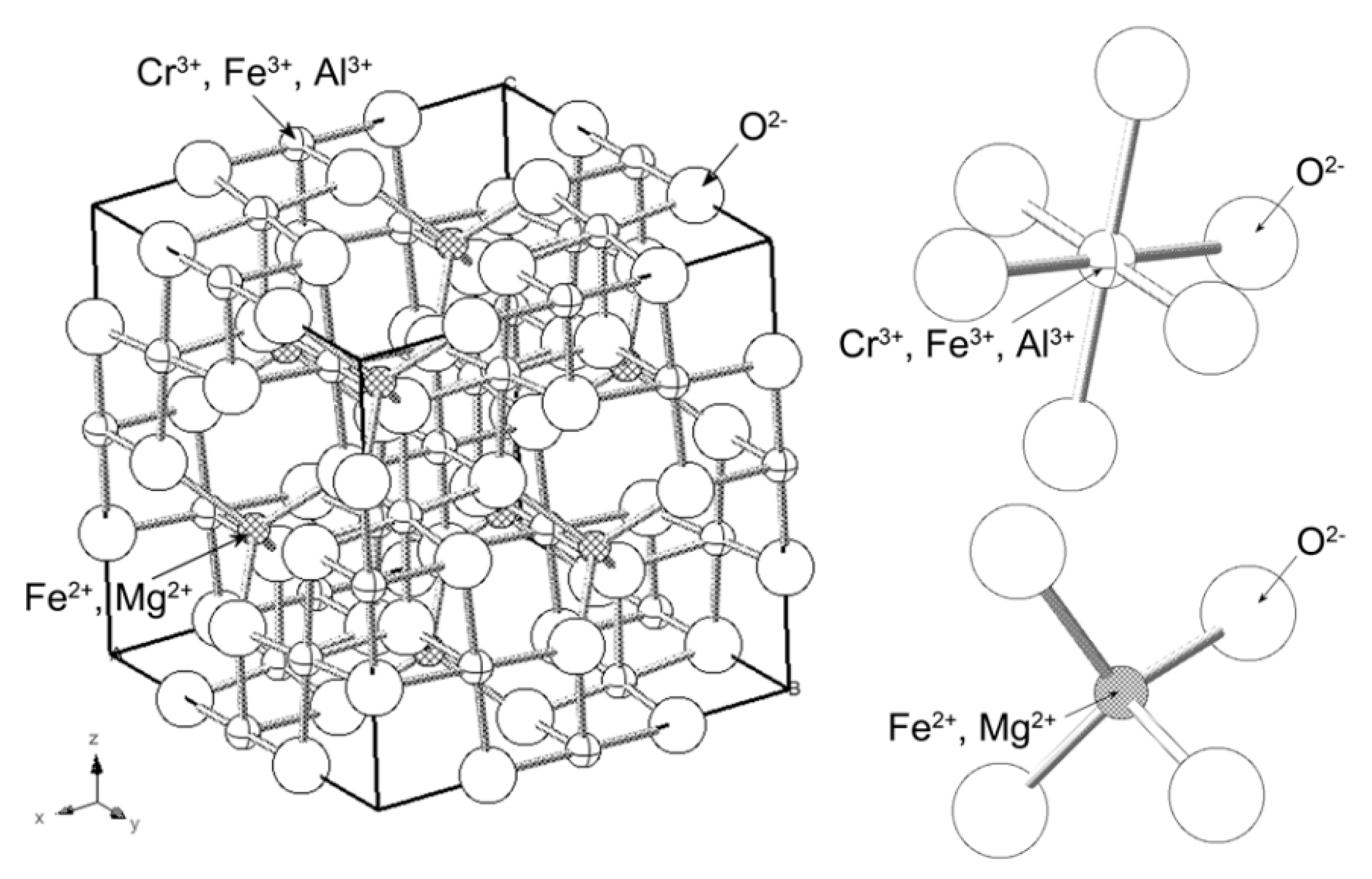
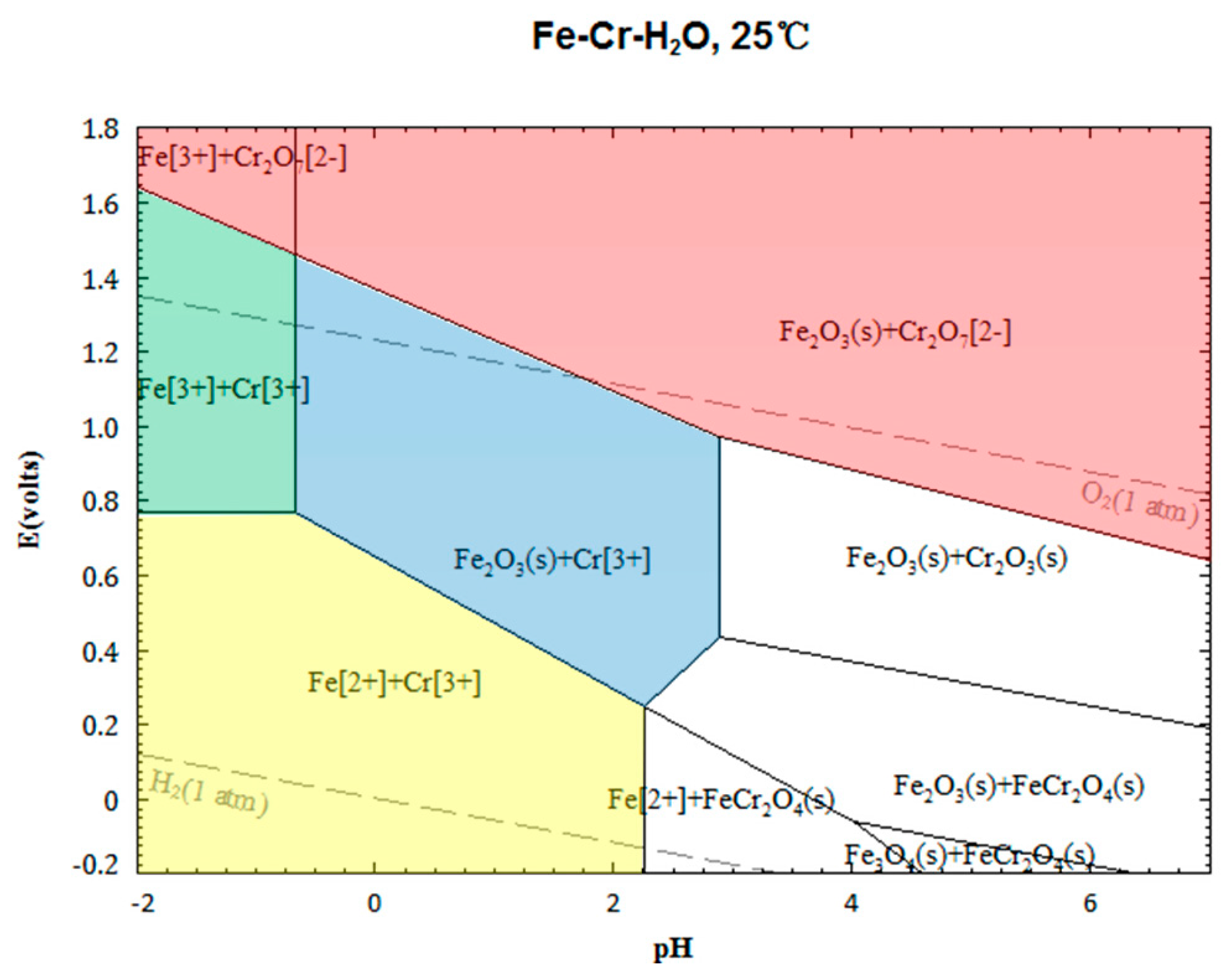
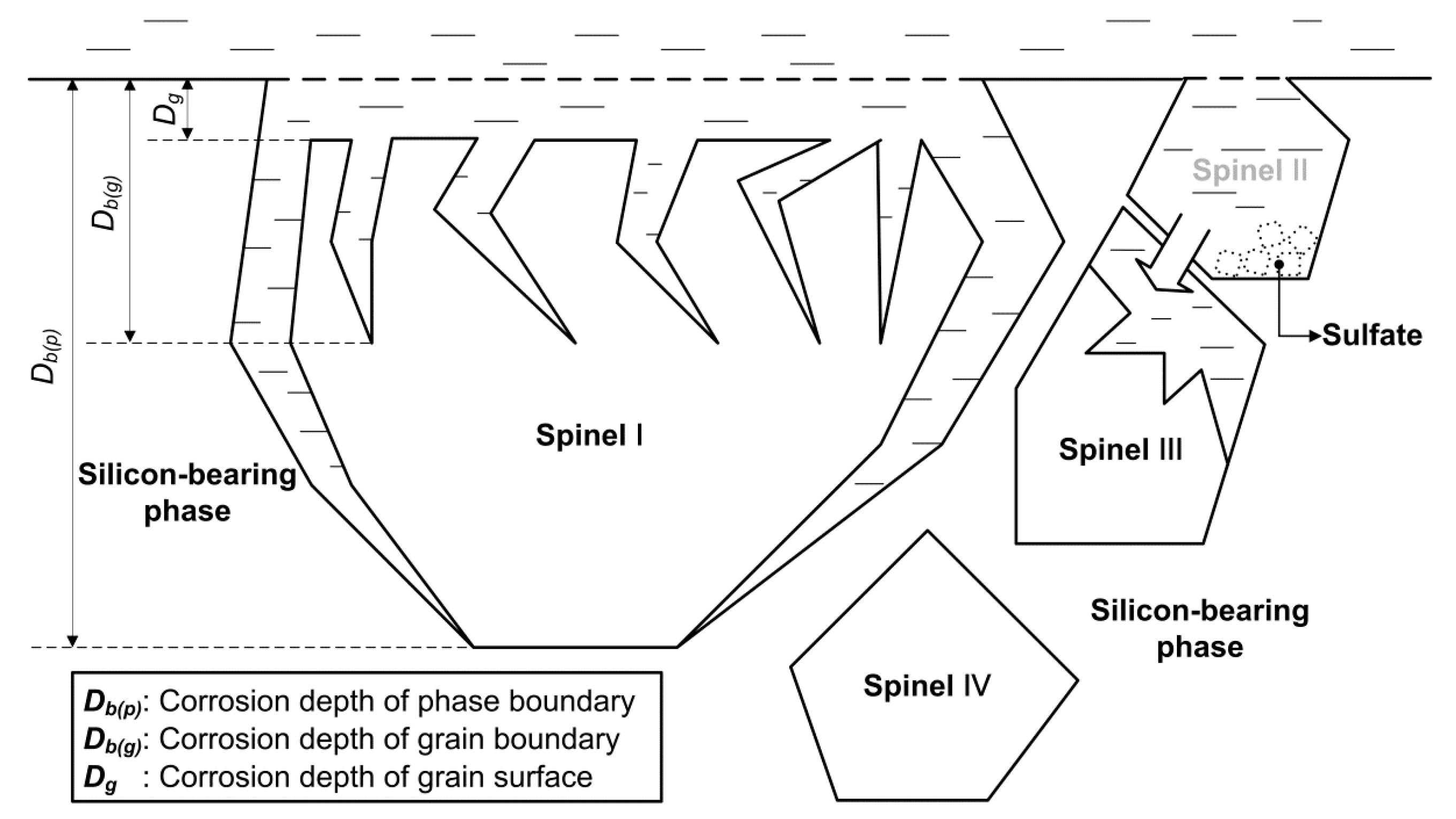
4. Separation of Cr3+ and Fe3+ in Sulfuric Acid Leaching Solution
4.1. Separation by Solvent Extraction
4.1.1. Liquid-Liquid Extraction of Chromium
4.1.2. Liquid-Liquid Extraction of Iron
4.2. Recovery of Chromium by Salting-Out
4.3. Removal of Iron by Precipitation Methods
4.3.1. Iron Hydrolysis
4.3.2. Jarosite Process
4.3.3. Formation of Mohr’s Salt
4.3.4. Ferrous Oxalate Precipitation
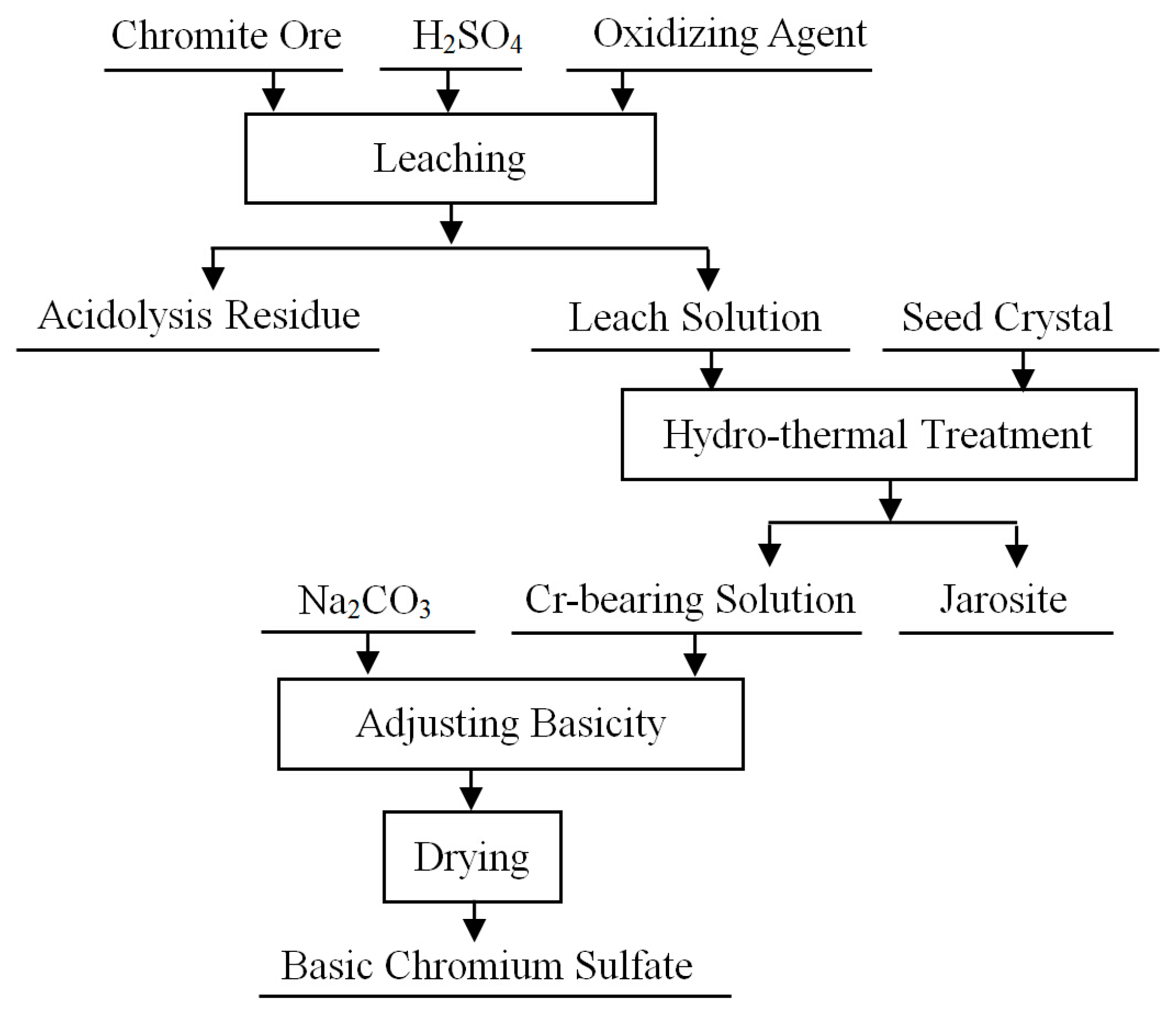
5. Conclusions and Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Yan, J.X.; Chen, J.X.; Hu, L. Metallurgy of Chromium; Metallurgical Industry Press: Beijing, China, 2007. (In Chinese) [Google Scholar]
- Li, Z.Y. The current status and development suggestion of chromium salt industry. Inorg. Salt Ind. 2004, 38, 1–5. (In Chinese) [Google Scholar]
- Meegoda, J.N.; Partymiller, K.; Richards, M.K.; Kamolpornwijit, W.; Librizzi, W.; Noval, B.A.; Mueller, R.T.; Santora, S. Remediation of chromium-contaminated soils: Pilot-scale investigation. Pract. Period. Hazard. Toxic Radioact. Waste Manag. 2000, 4, 7–15. [Google Scholar] [CrossRef]
- Wang, T.G.; He, M.L.; Pan, Q. A new method for the treatment of chromite ore processing residues. J. Hazard. Mater. 2007, 149, 440–447. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, Z.H.; Wang, Z.K.; Chen, J.Y. Green chemistry and chromium salt industry a new generation of industrial revolution. Chem. Ind. Eng. Prog. 1986, 10, 172–178. (In Chinese) [Google Scholar]
- Tinjum, J.M.; Benson, C.H.; Edil, T.B. Mobilization of Cr(VI) from chromite ore processing residue through acid treatment. Sci. Total Environ. 2008, 391, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Wazne, M.; Jagupilla, S.C.; Moon, D.H.; Christodoulatos, C.; Koutsospyros, A. Leaching mechanisms of Cr(VI) from chromite ore processing residue. J. Environ. Qual. 2008, 37, 2125–2134. [Google Scholar] [CrossRef] [PubMed]
- Meegoda, J.N.; Kamolpornwijit, W.; Vaccari, D.A.; Ezeldin, A.S.; Noval, B.A.; Mueller, R.T.; Santora, S. Remediation of chromium-contaminated soils: Bench-scale investigation. Pract. Period. Hazard. Toxic Radioact. Waste Manag. 1999, 3, 124–131. [Google Scholar] [CrossRef]
- Dean, J.A. Lange’s Chemistry Handbook Version; Science Press: Beijing, China, 1991. (In Chinese) [Google Scholar]
- Zheng, S.; Zhang, Y.; Li, Z.; Qi, T.; Li, H.; Xu, H. Green metallurgical processing of chromite. Hydrometallurgy 2006, 82, 157–163. [Google Scholar] [CrossRef]
- Amer, A.M.; Ibrahim, I.A. Leaching of a low grade Egyptian chromite ore. Hydrometallurgy 1996, 43, 307–316. [Google Scholar] [CrossRef]
- Amadta, U.; Batza, M.; Bellinghaousena, R. Method for manufacturing alkali chromates from chromics ore. Miner. Eng. 1996, 9, 1183. [Google Scholar]
- Burke, T.; Fagliano, J.; Goldoft, M.; Hazen, R.E.; Tglewicz, R.; Mckee, T. Chromite ore processing residue in Hudson County, New Jersey. Environ. Health Perspect. 1991, 92, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Katz, S.A.; Salem, H. The Biological and Environmental Chemistry of Chromium; VCH Publishers: New York, NY, USA, 2000. [Google Scholar]
- Wilbur, S.B. Toxicological Profile for Chromium; Department of Health and Human Services: Washington, DC, USA, 2000.
- Cheng, S.W.; Ding, Y.; Yang, C.R. Chromate Production Process; Chemical Industry Press: Beijing, China, 1987. (In Chinese) [Google Scholar]
- Weber, R.; Rosenow, B.; Block, H.D. Process for the Preparation of Sodium Dichromate. U.S. Patent 5273735, 28 December 1993. [Google Scholar]
- Kapland, M.A.; Annapolis, M.D.; Robinson, M.W. Process for Treating and Stabilizing Chromium Ore Waste. U.S. Patent 4504321, 12 March 1985. [Google Scholar]
- Zhu, H.S.; Xie, G. The study of the behavior of chromium ore in roasting process. Min. Metall. Eng. 2006, 26, 57–60. (In Chinese) [Google Scholar]
- Ji, Z. Overview of non-calcium roasting technology for chromite. Chromium Salt Ind. 1996, 2, 12–19. (In Chinese) [Google Scholar]
- Korallus, U. The significance of ascorbic acid and glutathione for chromate metabolism in man. Toxicol. Environ. Chem. 1989, 12, 332–340. [Google Scholar]
- Zhang, D.W.; Li, X.; Ji, Z. Study on parameter control of chromite roasting with non-calcium process. Inorg. Chem. Ind. 2012, 6, 37–39. (In Chinese) [Google Scholar]
- Ji, Z.; Tian, Q.; Zhao, Q.Z. The phase composition of chromite calcium-free roasting clinker and slag. Inorg. Chem. Ind. 1984, 6, 31–32. (In Chinese) [Google Scholar]
- William, W.L.; Douglas, G.F.; Alan, B.G. Method for the Conversion of Chromite Ore to Sodium Chromate. U.S. Patent 3816094, 11 June 1974. [Google Scholar]
- Charles, P.B.; William, W.L.; Edmund, W.S. Method for Recovering Chromite Values from Chromite Ore. U.S. Patent 3816095, 11 June 1974. [Google Scholar]
- Charles, P.B.; Douglas, G.F. Production of Sodium Chromate. G.B. Patent 1454125, 30 June 1976. [Google Scholar]
- Wolfgang, B.; Hans, G.N.; Hans, N. Alkaline Disintegration of Chromites. U.S. Patent 4066734, 3 January 1978. [Google Scholar]
- Somanahalli, N.S.; Thomas, R.M.; Douglas, G.F. Method for Production of Alkali Metal Chromates from Chrome Ores. U.S. Patent 4244925, 13 January 1981. [Google Scholar]
- Heinrich, S. Alkali Treatment of Chromium Ores. U.S. Patent 3510256, 5 May 1970. [Google Scholar]
- Johann, N.M.; Hans, N.; Hans, G.N. Disintegration of Chromates. U.S. Patent 4500350, 19 February 1979. [Google Scholar]
- Cooper, D.; Hugh, S.; Rand, E.; Henry, J. Process for Producing Alkali Metal Chromates and Dichromates. U.S. Patent 3932598, 13 January 1976. [Google Scholar]
- Ji, Z. Reaction mechanism of calcium-free roasting of chromite. Inorg. Chem. Ind. 1997, 1, 18–21. (In Chinese) [Google Scholar]
- Yu, K.P.; Zhang, H.L.; Chen, B.; Xu, H.B.; Zhang, Y. Investigation of reaction mechanism on the lime-free roasting of chromium-containing slag. Metal. Mater. Trans. B 2015, 46, 2553–2563. [Google Scholar] [CrossRef]
- Reid, R.D.; Schneidmiller, D. Resolution of the mixed waste issue for EDTA-based steam generator chemical cleaning waste solutions. Waste Manag. 1996, 16, 271–276. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, Z.H.; Wang, Z.K.; Chen, J.Y. Green chemistry and new revolution of chromic salts industry. Prog. Chem. 1998, 10, 172–178. (In Chinese) [Google Scholar]
- Sun, Z.; Zheng, S.L.; Zhang, Y. Thermodynamics study on the decomposition of chromite with KOH. Acta Metal. Sin. 2007, 20, 187–192. [Google Scholar] [CrossRef]
- Zheng, S.L.; Zhang, Y. Thermodynamic analysis on new reaction system of liquid phase oxidation of chromite in molten salt. Chin. J. Nonferr. Metals 1999, 9, 800–804. [Google Scholar]
- Sun, Z.; Zheng, S.L.; Xu, H.B.; Zhang, Y. Oxidation decomposition of chromite ore in molten potassium hydroxide. Int. J. Miner. Process. 2007, 83, 60–67. [Google Scholar] [CrossRef]
- Zhang, Y.; Zheng, S.L.; Xu, H.B.; Du, H.; Zhang, Y. Decomposition of chromite ore by oxygen in molten NaOH-NaNO3. Int. J. Miner. Process. 2010, 95, 10–17. [Google Scholar] [CrossRef]
- Xu, H.B.; Zheng, S.L.; Zhang, Y.; Li, Z.H.; Wang, Z.K. Oxidative leaching of a vietnamese chromite ore in highly concentrated potassium hydroxide aqueous solution at 300 °C and atmospheric pressure. Miner. Eng. 2005, 18, 527–535. [Google Scholar] [CrossRef]
- Xu, H.B.; Zhang, Y.; Li, Z.H. A Pressure Leaching of Chromite Sodium Chromate Cleaner Production Methods. China Patent 101817561A, 1 September 2010. [Google Scholar]
- Wang, Z.H.; Du, H.; Wang, S.N.; Zheng, S.L.; Zhang, Y.; Hwang, S.; Kim, N.S.; Jeong, T.E. Electrochemical enhanced oxidative decomposition of chromite ore in highly concentrated KOH solution. Miner. Eng. 2014, 57, 16–24. [Google Scholar] [CrossRef]
- Kamolpornwijit, W.; Meegoda, J.N.; Hu, Z. Characterization of chromite ore processing residue. Pract. Period. Hazard. Toxic Radioact. Waste Manag. 2007, 11, 234–239. [Google Scholar] [CrossRef]
- Ji, Z. Preparation of trivalent chromium compounds from chromite by acid-leaching technique. Inorg. Chem. Ind. 2012, 44, 1–5. (In Chinese) [Google Scholar]
- Zhao, Q. Fundamental Research on the Cleaner Preparation Process of Basic Chromium Sulfate. Ph.D. Thesis, Northeastern University, Shenyang, China, 2015. [Google Scholar]
- Kanari, N.; Gaballah, I.; Allain, E. A study of chromite carbochlorination kinetics. Metal. Mater. Trans. B 1999, 30, 577–587. [Google Scholar] [CrossRef]
- Hussein, M.K.; Winterhager, H.; Kammel, R. Chlorination behavior of the main oxide components chromite ores. Trans. Inst. Min. Metal. Sect. C 1974, 83, 154–160. [Google Scholar]
- Bale, C.W.; Chartrand, P.; Decterov, S.A. FactSage thermochemical software and databases. Calphad 2002, 62, 189–228. [Google Scholar] [CrossRef]
- Biermann, W.J.; Heinrichs, M. The attack of chromite by sulphuric acid. Can. J. Chem. 1960, 38, 1449–1454. [Google Scholar] [CrossRef]
- Geveci, A.; Topkaya, Y.; Ayhan, E. Sulfuric acid leaching of Turkish chromite concentrate. Miner. Eng. 2002, 15, 885–888. [Google Scholar] [CrossRef]
- Vardar, E; Eric, R.H.; Letowski, F.K. Acid leaching of chromite. Miner. Eng. 1994, 7, 605–617. [Google Scholar]
- Shi, P.Y.; Jiang, M.F.; Liu, C.J.; Liu, S.L. A Method for Preparing Basic Chrome Sulphate. China Patent 1264755C, 19 July 2006. [Google Scholar]
- Shi, P.Y.; Liu, S.L. Experimental study on sulphuric acid leaching of chromite. J. Chin. Rare Earth Soc. 2002, 20, 472–474. (In Chinese) [Google Scholar]
- Liu, C.J.; Shi, P.Y.; Jiang, M.F. A Method of Sulfuric acid Leaching to Process Chromite. China Patent 101979679A, 23 February 2011. [Google Scholar]
- Liu, C.J.; Sun, L.F.; Shi, P.Y.; Jiang, M.F. Study on sulfuric acid leaching technology from chromite. J. Ecotechnol. Res. 2006, 12, 177–180. [Google Scholar]
- Liu, C.J.; Qi, J.; Jiang, M.F. Experimental study on sulfuric acid leaching behavior of chromite with different temperature. Adv. Mater. Res. 2012, 361, 628–631. [Google Scholar] [CrossRef]
- Zhao, Q.; Liu, C.J.; Shi, P.Y.; Zhang, B.; Jiang, M.F.; Zhang, Q.S.; Zevenhoven, H.S.R. Sulfuric acid leaching of South African chromite. Part 1: Study on leaching behavior. Int. J. Miner. Process. 2014, 130, 95–101. [Google Scholar] [CrossRef]
- Jiang, M.F.; Zhao, Q.; Liu, C.J.; Shi, P.Y.; Zhang, B.; Yang, D.P.; Saxén, H.; Zevenhoven, R. Sulfuric acid leaching of South African chromite. Part 2: Optimization of leaching conditions. Int. J. Miner. Process. 2014, 130, 102–107. [Google Scholar] [CrossRef]
- Liu, S.Y. Industry standard profile of chromium oxides. Chem. Stand. Qual. Surveill. 1997, 1, 7–9. (In Chinese) [Google Scholar]
- Cheng, J.Y. Hydrometallurgy Manual; Metallurgical Industry Press: Beijing, China, 2005. (In Chinese) [Google Scholar]
- Stauter, J.C.; Richard, T. Recovery of Chromium Values. U.S. Patent 4029734, 14 June 1977. [Google Scholar]
- Akash, D.; Paulo, F.M.; Jorge, M.R. Selective recoveries of Fe(III) and Cr(III) from a tannery filtrate using Cyanex 923. Anal. Chim. Acta 2006, 558, 254–260. [Google Scholar]
- Shi, P.Y.; Liu, C.J.; Jiang, M.F. One Method to Separate Iron and Chromium from Multi-Component Solution. China Patent 101974688A, 16 February 2011. [Google Scholar]
- Ma, H.R.; Li, D.X.; Shi, X.F. Solvent extraction of iron and chromium from bio-leachate derived from tannery sludge. Environ. Chem. 2007, 26, 508–511. (In Chinese) [Google Scholar]
- Wei, Y.Z.; Kumaya, M.; Takashima, Y. A Separation and Recovery Method of Chromium. Japan Patent 157219, 18 June 1996. [Google Scholar]
- Speight, J.G. Langec’s Handbook of Chemistry; Science Press: Beijing, China, 1999. (In Chinese) [Google Scholar]
- Souza, E.S.; Mello, N.T.; Menezes, D.M.; Monteneqro, M.C.; Araujo, A.N.; Barros, N.B.; Silva, A.L. Extraction and recovery of chromium from electroplating sludge. J. Hazard. Mater. 2006, 128, 39–43. [Google Scholar]
- Chen, J.Y.; Yu, S.Q.; Wu, Z.C. Separation and Utilization of Iron in Hydrometallurgy; Metallurgical Industry Press: Beijing, China, 1991. (In Chinese) [Google Scholar]
- Jiang, H.Y. Hydrometallurgy Process of Physical Chemistry; Metallurgical Industry Press: Beijing, China, 1984. (In Chinese) [Google Scholar]
- Chen, J.; Wang, Y.C.; Yang, H. Technical study on iron removal from basic chromium sulfate produced by patty recovered. Chem. Eng. 2009, 12, 60–71. (In Chinese) [Google Scholar]
- Hu, G.R.; Li, G.; Deng, X.R. Removal of iron sulfuric acid leaching solution of ferrochromium alloy by goethite. Hydrometal. China 2006, 25, 198–201. (In Chinese) [Google Scholar]
- Zhu, C.S.; Sun, Z.Y.; Gong, W.Q.; Chen, H.S. Study on treatment of chromium-containing wastewater with biomineralized goethite. Res. Environ. Sci. 2003, 16, 57–58. (In Chinese) [Google Scholar]
- Zhang, Q.S. Exploratory Study on the Separation of Chromium and Iron in Multicomponent Acid Solution System. Master’s Thesis, Northeastern University, Shenyang, China, 2014. [Google Scholar]
- Shi, P.Y. Study on Leaching Process of Preparation of Alkaline Chromium Sulphate by Sulphuric Acid Leaching. Master’s Thesis, Northeastern University, Shenyang, China, 2001. [Google Scholar]
- Wu, J.H.; Yang, L.Z.; Zhang, J. Research on separation of chromium and iron in waste ferrochromium alloy. Hydrometal. China 2011, 30, 51–56. (In Chinese) [Google Scholar]
- Fan, J.Y. The research of technology on producing chrome by electrolysis method. J. Kunming Metal. Coll. 2001, 17, 27–30. (In Chinese) [Google Scholar]
- Jiang, X.J. Separation of iron from chromium electrolyte with DDTC-Na. Ferro Alloys 2000, 5, 19–21. (In Chinese) [Google Scholar]
- Hu, G.H. One Method for the Production of Chromium Oxide and Ferrous Oxlalate from Carbon Ferrochrome. China Patent 101041466A, 26 September 2007. [Google Scholar]
- Wang, Q.M. Study on Effective Separation and Comprehensive Utilization of Elements from Carbon Ferrochromium. Master’s Thesis, Central South University, Changsha, China, 2011. [Google Scholar]
- Zhang, B.; Chi, W.H.; Shi, P.Y.; Liu, C.J.; Jiang, M.F. Study on the clean production process of basic chromium sulphate. Adv. Mater. Res. 2014, 888, 651–656. [Google Scholar] [CrossRef]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, B.; Shi, P.; Jiang, M. Advances towards a Clean Hydrometallurgical Process for Chromite. Minerals 2016, 6, 7. https://doi.org/10.3390/min6010007
Zhang B, Shi P, Jiang M. Advances towards a Clean Hydrometallurgical Process for Chromite. Minerals. 2016; 6(1):7. https://doi.org/10.3390/min6010007
Chicago/Turabian StyleZhang, Bo, Peiyang Shi, and Maofa Jiang. 2016. "Advances towards a Clean Hydrometallurgical Process for Chromite" Minerals 6, no. 1: 7. https://doi.org/10.3390/min6010007
APA StyleZhang, B., Shi, P., & Jiang, M. (2016). Advances towards a Clean Hydrometallurgical Process for Chromite. Minerals, 6(1), 7. https://doi.org/10.3390/min6010007





