Post-Subduction Granite Magmatism and Gold-Sulfide Mineralization in the Abu Zawal (Fatira) Area, Eastern Desert, Egypt
Abstract
1. Introduction
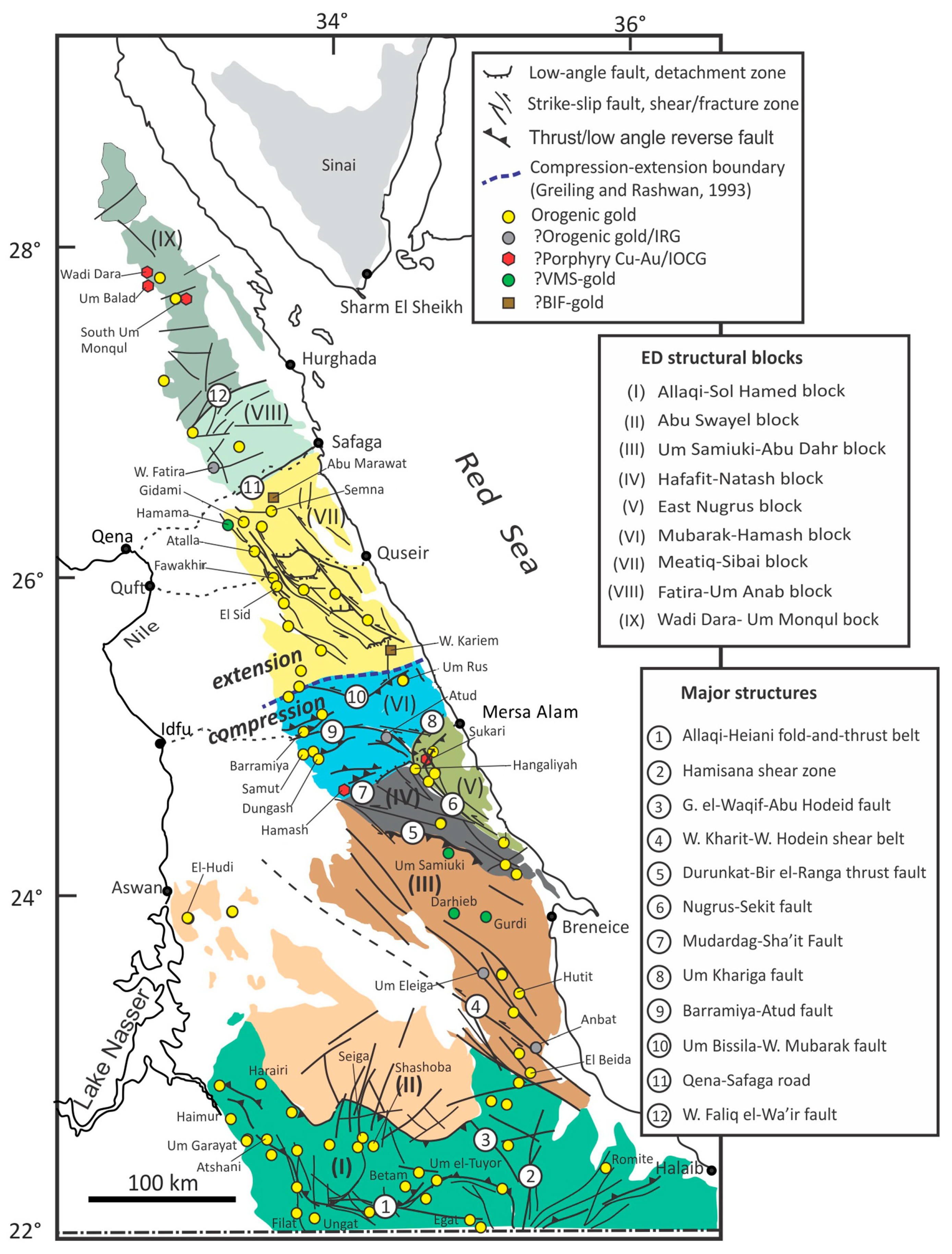
2. Regional and Local Geology
2.1. The Geology of the Egyptian Nubian Shield
2.2. Local Geologic Setting of the Abu Zawal Area
3. Gold Mineralization at Abu Zawal (Fatira) Area
4. Materials and Methods
5. Results
5.1. Petrography and XRD of Hydrothermal Alteration
5.2. Ore Mineralogy and Paragenesis
5.3. Mineral Chemistry of Sulfide Minerals
5.4. Geochemistry
5.4.1. Geochemistry of Host Rocks

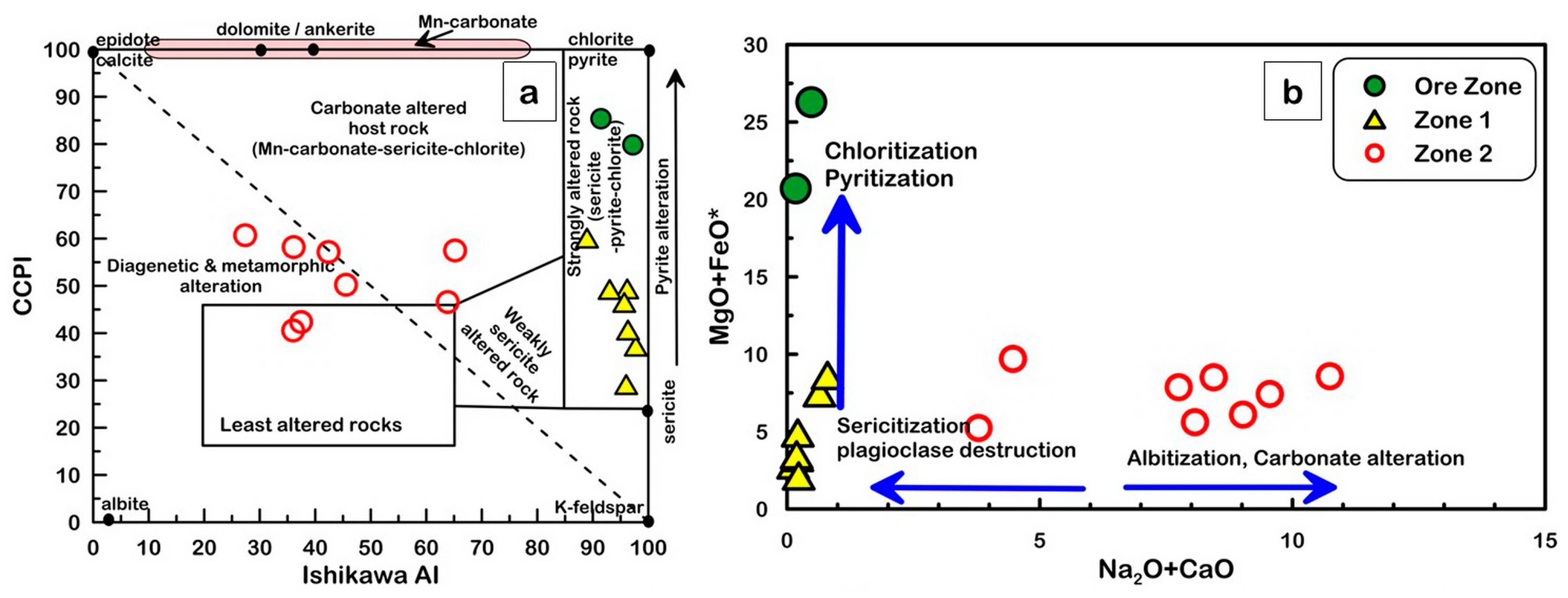
5.4.2. Alteration Geochemistry
6. Discussion
6.1. Mass Balance Calculations
6.2. Behavior of Large Ion Lithophile Elements (LILE) and High Field Strength Elements (HFSE)
6.3. REE Behavior during the Alteration Process and Gold Deposition
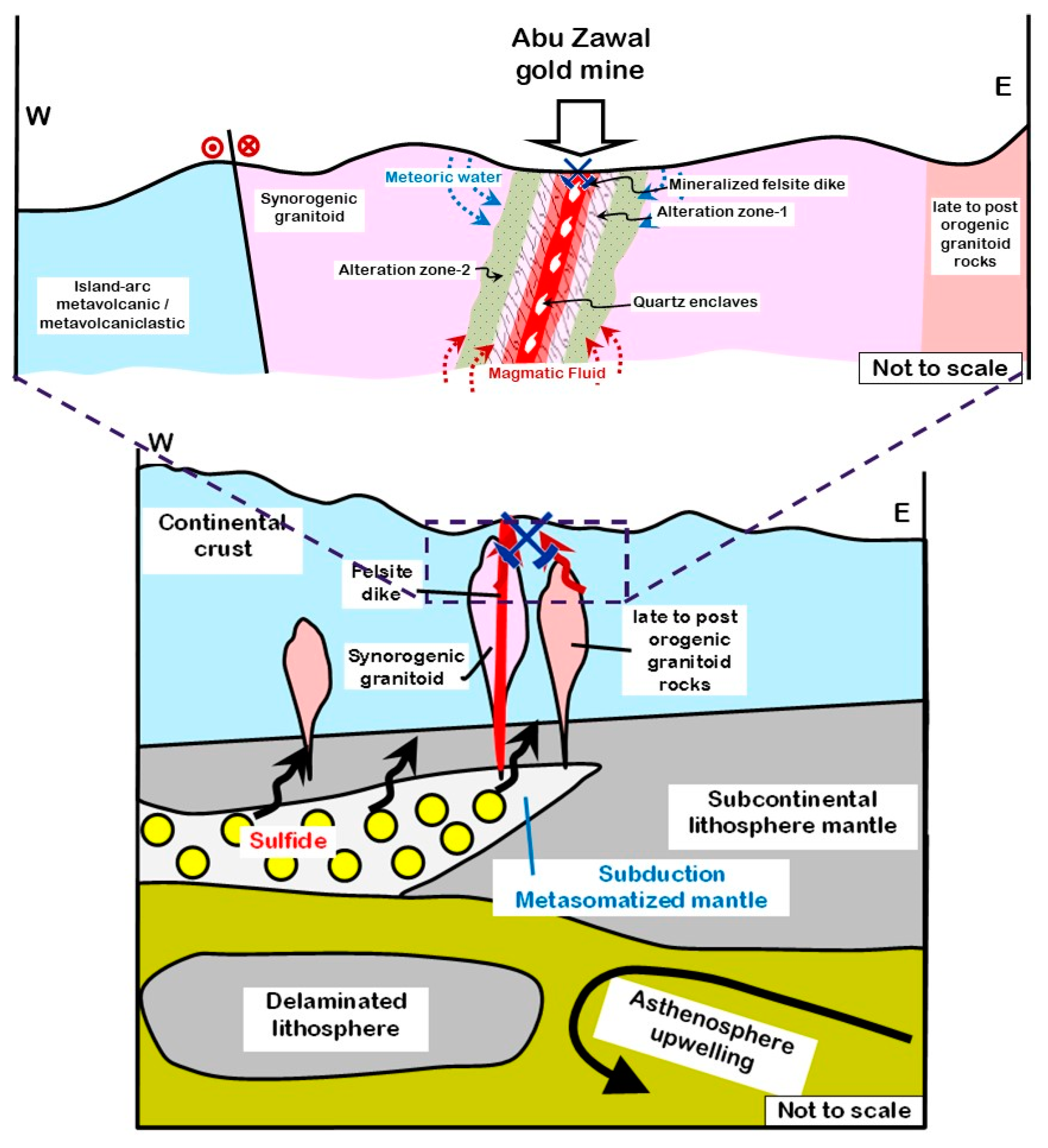
6.4. Tectono-Magmatic Controls of Post-Subduction Gold Mineralization at Abu Zawal Area
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Richards, J.P. Postsubduction porphyry Cu-Au and epithermal Au deposits: Products of remelting of subduction-modified lithosphere. Geology 2009, 37, 247–250. [Google Scholar] [CrossRef]
- Richards, J.P. Magmatic to hydrothermal metal fluxes in convergent and collided margins. Ore Geol. Rev. 2011, 40, 1–26. [Google Scholar] [CrossRef]
- Locmelis, M.; Fiorentini, M.L.; Rushmer, T.; Arevalo, R., Jr.; Adam, J.; Denyszyn, S.W. Sulfur and metal fertilization of the lower continental crust. Lithos 2016, 244, 74–93. [Google Scholar] [CrossRef]
- Fiorentini, M.L.; LaFlamme, C.; Denyszyn, S.; Mole, D.; Maas, R.; Locmelis, M.; Caruso, S.; Bui, T.-H. Post-collisional alkaline magmatism as gateway for metal and sulfur enrichment of the continental lower crust. Geochim. Cosmochim. Acta 2018, 223, 175–197. [Google Scholar] [CrossRef]
- Tomkins, A.G.; Rebryna, K.C.; Weinberg, R.F.; Schaefer, B.F. Magmatic sulfide formation by reduction of oxidized arc basalt. J. Petrol. 2012, 53, 1537–1567. [Google Scholar] [CrossRef]
- Hedenquist, J.W.; Lowenstern, J.B. The role of magmas in the formation of hydrothermal ore deposits. Nature 1994, 370, 519–527. [Google Scholar] [CrossRef]
- Johnson, P. The Civilization of Ancient Egypt; Harper Collins: New York, NY, USA, 1999. [Google Scholar]
- Botros, N.S. A new classification of the gold deposits of Egypt. Ore Geol. Rev. 2004, 25, 1–37. [Google Scholar] [CrossRef]
- Zoheir, B.A.; Johnson, P.R.; Goldfarb, R.J.; Klemm, D.D. Orogenic gold in the Egyptian Eastern Desert: Widespread gold mineralization in the late stages of Neoproterozoic orogeny. Gondwana Res. 2019, 75, 184–217. [Google Scholar] [CrossRef]
- El Aref, M.; Abd El-Rahman, Y.; Zoheir, B.; Surour, A.; Helmy, H.M.; Abdelnasser, A.; Ahmed, A.H.; El-Ahmadi Ibrahim, M. Mineral Resources in Egypt (I): Metallic Ores. In The Geology of Egypt; Hamimi, Z., El-Barkooky, A., Martínez Frías, J., Fritz, H., Abd El-Rahman, Y., Eds.; Springer International Publishing: Cham, Germany, 2020; pp. 521–587. [Google Scholar]
- Bakhit, F.S. Statistical analysis of structural data of Fatira Area, Central Eastern Desert, Egypt. Ann. Geol. Surv. Egypt 1986, 16, 315–319. [Google Scholar]
- Dourgham, A.; El Zalaky, M.; Neigm, S. Gold-sulfides hosted in fractured felsite dykes, Fatiri deposit, North Eastern Desert, Egypt. Ann. Geol. Surv. Egypt 2009, V.XXXI, 247–266. [Google Scholar]
- Abu-Alam, T.; Abd El Monsef, M.; Grosch, E. Shear-zone hosted gold mineralization of the Arabian–Nubian Shield: Devolatilization processes across the greenschist–amphibolite-facies transition. Geol. Soc. Lond. Spec. Publ. 2019, 478, 287–313. [Google Scholar] [CrossRef]
- Abd El Monsef, M.; Slobodník, M.; Salem, I.A. Characteristics and nature of gold-bearing fluids in Fatira area, North Eastern Desert of Egypt: Possible transition from intrusion-related to orogenic deposits. Arab. J. Geosci. 2020, 13, 1034. [Google Scholar] [CrossRef]
- Stoeser, D.B.; Camp, V.E. Pan-African microplate accretion of the Arabian Shield. Geol. Soc. Am. Bull. 1985, 96, 817–826. [Google Scholar] [CrossRef]
- Abdelsalam, M.G.; Stern, R.J. Sutures and shear zones in the Arabian-Nubian Shield. J. Afr. Earth Sci. 1996, 23, 289–310. [Google Scholar] [CrossRef]
- Abdelsalam, M.G.; Abdeen, M.M.; Dowaidar, H.M.; Stern, R.J.; Abdelghaffar, A.A. Structural evolution of the Neoproterozoic western Allaqi–Heiani suture, southeastern Egypt. Precambrian Res. 2003, 124, 87–104. [Google Scholar] [CrossRef]
- Johnson, P.; Andresen, A.; Collins, A.; Fowler, A.; Fritz, H.; Ghebreab, W.; Kusky, T.; Stern, R. Late Cryogenian–Ediacaran history of the Arabian–Nubian Shield: A review of depositional, plutonic, structural, and tectonic events in the closing stages of the northern East African Orogen. J. Afr. Earth Sci. 2011, 61, 167–232. [Google Scholar] [CrossRef]
- Kröner, A.; Grieling, R.; Reischmann, T.; Hussein, I.; Stern, R.; Dürr, S.; Krüger, J.; Zimmer, M. Pan-African crustal evolution in the Nubian segment of northeast Africa. Proterozic Lithospheric Evol. 1987, 17, 235–257. [Google Scholar]
- Abd El-Rahman, Y.; Polat, A.; Dilek, Y.; Fryer, B.J.; El-Sharkawy, M.; Sakran, S. Geochemistry and tectonic evolution of the Neoproterozoic incipient arc–forearc crust in the Fawakhir area, Central Eastern Desert of Egypt. Precambrian Res. 2009, 175, 116–134. [Google Scholar] [CrossRef]
- Akaad, M.K.; Abu El Ela, A. Geology of the Basement Rocks in the Eastern Half of the Belt between Latitudes 25°30′ and 26°30′ N, Central Eastern Desert, Egypt: Covering Parts of Sheets NG 36 K2, K3, L1 and NG 36 G5, G6, H4; Arab Republic of Egypt, Ministry of Industry and Technology, the Geological Survey of Egypt: Cairo, Egypt, 2002. [Google Scholar]
- Stern, R.J. Arc-assembly and continental collision in the Neoproterozoic African orogen: Implications for the consolidation of Gondwanaland. Annu. Rev. Earth Planet. Sci. 1994, 22, 319–351. [Google Scholar] [CrossRef]
- Eliwa, H.; Kimura, J.-I.; Itaya, T. Late Neoproterozoic Dokhan Volcanics, North Eastern Desert, Egypt: Geochemistry and petrogenesis. Precambrian Res. 2006, 151, 31–52. [Google Scholar] [CrossRef]
- Zoheir, B.; Zeh, A.; El-Bialy, M.; Ragab, A.; Deshesh, F.; Steele-MacInnis, M. Hybrid granite magmatism during orogenic collapse in the Eastern Desert of Egypt: Inferences from whole-rock geochemistry and zircon U–Pb–Hf isotopes. Precambrian Res. 2021, 354, 106044. [Google Scholar] [CrossRef]
- Stern, R.J.; Hedge, C.E. Geochronologic and isotopic constraints on late Precambrian crustal evolution in the Eastern Desert of Egypt. Am. J. Sci. 1985, 285, 97–127. [Google Scholar] [CrossRef]
- El-Gaby, S.; List, F.; Tehrani, R. Geology, evolution and metallogenesis of the Pan-African Belt in Egypt. In The Pan-African Belt of Northeast Africa and Adjacent Areas: Tectonic Evolution and Economic Aspects of a Late Proterozoic Orogen; El-Gaby, S., Greiling, R., Eds.; Friedr. Vieweg & Sohn Verlagskatalog: Braunschweig/Wiesbaden, Germany, 1988; pp. 17–68. [Google Scholar]
- El-Gaby, S.; List, F.; Tehrani, R. The basement complex of the Eastern Desert and Sinai. In Geology of Egypt; Said, R., Ed.; Routledge: London, UK, 1990; pp. 175–184. [Google Scholar]
- Abd El-Wahed, M.; Hamimi, Z. The Egyptian Nubian Shield Within the Frame of the Arabian–Nubian Shield. In The Geology of the Egyptian Nubian Shield; Hamimi, Z., Arai, S., Fowler, A.-R., El-Bialy, M.Z., Eds.; Springer International Publishing: Cham, Switzerland, 2021; pp. 15–51. [Google Scholar]
- Johnson, P.R.; Woldehaimanot, B. Development of the Arabian-Nubian Shield: Perspectives on accretion and deformation in the northern East African Orogen and the assembly of Gondwana. Geol. Soc. Lond. Spec. Publ. 2003, 206, 289–325. [Google Scholar] [CrossRef]
- Hargrove, U.; Stern, R.; Kimura, J.-I.; Manton, W.; Johnson, P. How juvenile is the Arabian–Nubian Shield? Evidence from Nd isotopes and pre-Neoproterozoic inherited zircon in the Bi’r Umq suture zone, Saudi Arabia. Earth Planet. Sci. Lett. 2006, 252, 308–326. [Google Scholar] [CrossRef]
- Klemm, R.; Klemm, D. Gold and Gold Mining in Ancient Egypt and Nubia: Geoarchaeology of the Ancient Gold Mining Sites in the Egyptian and Sudanese Eastern Deserts; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- Abu El-Leil, I.; Sweifi, B.; El Gammal, A. Petrography and geochemistry of some metavolcanics, Dokhan volcanics and younger volcanics along wadi Fatira El Zarqa, Central Eastern Desert, Egypt. Al-AZhar Bull. Sci. 1991, 2, 167–192. [Google Scholar]
- Khalaf, E.E.D.A.H. Stratigraphy, facies architecture, and palaeoenvironment of Neoproterozoic volcanics and volcaniclastic deposits in Fatira area, Central Eastern Desert, Egypt. J. Afr. Earth Sci. 2010, 58, 405–426. [Google Scholar] [CrossRef]
- Abu El-Leil, I.; Bekhit, M.H.; Tolba, A.S.; Moharem, A.F.; Shahin, T.M. Geological, Structural and Petrotectonical Aspectable Features of Neoproterozoic Rocks, Gabal El Dob Area, Northeastern Desert, Egypt. Int. J. Sci. Eng. Appl. Sci. 2015, 1, 332–350. [Google Scholar]
- Marten, B. Reconnaissance of the gold deposits of the Eastern Desert of Egypt; Parts I and II; Geological Survey of Egypt: Cairo, Egypt, 1986; pp. 1–44. [Google Scholar]
- Murr, A. Genesis of gold mineralization of Fatria, Gidami, Atalla and Hangaliya, Eastern Desert of Egypt. Münchner Geol. Hefte A 1999, 27, 202. [Google Scholar]
- Abd El Nabi, A.; Prokhorov, D.; Yakoop, F. Results of prospecting for nonferrous metals and gold in the area of Wadis, El Atrsh, El Ghozah and Fatira El Beida, North Eastern Desert of Egypt. In Internal Report; No. 511978; The geological Survey of Egypt: Cairo, Egypt, 1978; 145p. [Google Scholar]
- Carr, M. Igpet 2007 for Windows XP or Vista; Terra Softa Inc.: Somerset, NJ, USA, 2007. [Google Scholar]
- Coelho, J. GEOISO—A Windows™ program to calculate and plot mass balances and volume changes occurring in a wide variety of geologic processes. Comput. Geosci. 2006, 32, 1523–1528. [Google Scholar] [CrossRef]
- Gresens, R.L. Composition-volume relationships of metasomatism. Chem. Geol. 1967, 2, 47–65. [Google Scholar] [CrossRef]
- Grant, J.A. Isocon analysis: A brief review of the method and applications. Phys. Chem. Earth Parts A/B/C 2005, 30, 997–1004. [Google Scholar] [CrossRef]
- Armstrong, J.T. Citzaf-a package of correction programs for the quantitative Electron Microbeam X-Ray-Analysis of thick polished materials, thin-films, and particles. Microbeam Anal. 1995, 4, 177–200. [Google Scholar]
- Le Maitre, R.W.B.; Dudek, P.; Keller, A.; Lameyre, J.; Le Bas, J.; Sabine, M.; Schmid, P.; Sorensen, R.; Streckeisen, H.; Woolley, A. A classification of igneous rocks and glossary of terms: Recommendations of the International Union of Geological Sciences, Subcommission on the Systematics of Igneous Rocks; International Union of Geological Sciences, Blackwell: Hoboken, NJ, USA, 1989. [Google Scholar]
- Moghazi, A. Geochemistry and petrogenesis of a high-K calc-alkaline Dokhan Volcanic suite, South Safaga area, Egypt: The role of late Neoproterozoic crustal extension. Precambrian Res. 2003, 125, 161–178. [Google Scholar] [CrossRef]
- Wood, D.A. The application of a Th-Hf-Ta diagram to problems of tectonomagmatic classification and to establishing the nature of crustal contamination of basaltic lavas of the British Tertiary Volcanic Province. Earth Planet. Sci. Lett. 1980, 50, 11–30. [Google Scholar] [CrossRef]
- Sun, S.-S.; McDonough, W. Chemical and isotopic systematics of oceanic basalts: Implications for mantle composition and processes. Geol. Soc. Lond. Spec. Publ. 1989, 42, 313–345. [Google Scholar] [CrossRef]
- Miyashiro, A.; Shido, F. Tholeiitic and calc-alkalic series in relation to the behaviors of titanium, vanadium, chromium, and nickel. Am. J. Sci. 1975, 275, 265–277. [Google Scholar] [CrossRef]
- Irvine, T.; Baragar, W. A guide to the chemical classification of the common volcanic rocks. Can. J. Earth Sci. 1971, 8, 523–548. [Google Scholar] [CrossRef]
- Stern, R.J. Petrogenesis and tectonic setting of Late Precambrian ensimatic volcanic rocks, Central Eastern Desert of Egypt. Precambrian Res. 1981, 16, 195–230. [Google Scholar] [CrossRef]
- Kuno, H. Differentiation of basalt magmas. In Basalts: The Poldervaart treatise on rocks of basaltic composition; Interscience Publishers: New York, NY, USA, 1968; Volume 2, pp. 623–688. [Google Scholar]
- Petro, W.L.; Vogel, T.A.; Wilband, J.T. Major-element chemistry of plutonic rock suites from compressional and extensional plate boundaries. Chem. Geol. 1979, 26, 217–235. [Google Scholar] [CrossRef]
- Shand, S. On the relations between silica, alumina, and the bases in eruptive rocks, considered as a means of classification. Geol. Mag. 1927, 64, 446–449. [Google Scholar] [CrossRef]
- Chappell, B.W.; White, A. I-and S-type granites in the Lachlan Fold Belt. Earth Environ. Sci. Trans. R. Soc. Edinb. 1992, 83, 1–26. [Google Scholar]
- El Bouseily, A.; El Sokkary, A. The relation between Rb, Ba and Sr in granitic rocks. Chem. Geol. 1975, 16, 207–219. [Google Scholar] [CrossRef]
- Ishihara, S. The magnetite-series and ilmenite-series granitic rocks. Min. Geol. 1977, 27, 293–305. [Google Scholar]
- Pearce, J.A.; Harris, N.B.; Tindle, A.G. Trace element discrimination diagrams for the tectonic interpretation of granitic rocks. J. Petrol. 1984, 25, 956–983. [Google Scholar] [CrossRef]
- Taylor, S.R.; McLennan, S.M. The Continental Crust: Its Composition and Evolution; Blackwell: Oxford, UK, 1985; p. 349. [Google Scholar]
- Frost, B.R.; Barnes, C.G.; Collins, W.J.; Arculus, R.J.; Ellis, D.J.; Frost, C.D. A geochemical classification for granitic rocks. J. Petrol. 2001, 42, 2033–2048. [Google Scholar] [CrossRef]
- Large, R.R.; Gemmell, J.B.; Paulick, H.; Huston, D.L. The alteration box plot: A simple approach to understanding the relationship between alteration mineralogy and lithogeochemistry associated with volcanic-hosted massive sulfide deposits. Econ. Geol. 2001, 96, 957–971. [Google Scholar] [CrossRef]
- Ishikawa, Y.; Sawaguchi, T.; Iwaya, S.; Horiuchi, M. Delineation of prospecting targets for Kuroko deposits based on modes of volcanism of underlying dacite and alteration halos. Min. Geol. 1976, 26, 105–117. [Google Scholar]
- Paulick, H.; Herrmann, W.; Gemmell, J.B. Alteration of felsic volcanics hosting the Thalanga massive sulfide deposit (Northern Queensland, Australia) and geochemical proximity indicators to ore. Econ. Geol. 2001, 96, 1175–1200. [Google Scholar] [CrossRef]
- Grant, J.A. The isocon diagram; a simple solution to Gresens’ equation for metasomatic alteration. Econ. Geol. 1986, 81, 1976–1982. [Google Scholar] [CrossRef]
- Barton, M.D.; Staude, J.-M.; Snow, E.A.; Johnson, D.A. Aureole systematics. Rev. Mineral. Geochem. 1991, 26, 723–847. [Google Scholar]
- Hanson, G.N. The application of trace elements to the petrogenesis of igneous rocks of granitic composition. Earth Planet. Sci. Lett. 1978, 38, 26–43. [Google Scholar] [CrossRef]
- Arculus, R.J. Aspects of magma genesis in arcs. Lithos 1994, 33, 189–208. [Google Scholar] [CrossRef]
- Ringwood, A.E. Slab-mantle interactions: 3. Petrogenesis of intraplate magmas and structure of the upper mantle. Chem. Geol. 1990, 82, 187–207. [Google Scholar] [CrossRef]
- McCulloch, M.T.; Gamble, J.A. Geochemical and geodynamical constraints on subduction zone magmatism. Earth Planet. Sci. Lett. 1991, 102, 358–374. [Google Scholar] [CrossRef]
- Fitton, J.; James, D.; Kempton, P.; Ormerod, D.; Leeman, W. The role of lithospheric mantle in the generation of late Cenozoic basic magmas in the western United States. J. Petrol. 1988, 331–349. [Google Scholar] [CrossRef]
- Pearce, J.A.; Bender, J.F.; De Long, S.E.; Kidd, W.S.F.; Low, P.J.; Güner, Y.; Saroglu, F.; Yilmaz, Y.; Moorbath, S.; Mitchell, J.G. Genesis of collision volcanism in Eastern Anatolia, Turkey. J. Volcanol. Geotherm. Res. 1990, 44, 189–229. [Google Scholar] [CrossRef]
- Kempton, P.; Fitton, J.; Hawkesworth, C.; Ormerod, D. Isotopic and trace element constraints on the composition and evolution of the lithosphere beneath the southwestern United States. J. Geophys. Res. Solid Earth 1991, 96, 13713–13735. [Google Scholar] [CrossRef]
- Varol, E.; Temel, A.; Yürür, T.; Gourgaud, A.; Bellon, H. Petrogenesis of the Neogene bimodal magmatism of the Galatean Volcanic Province, Central Anatolia, Turkey. J. Volcanol. Geotherm. Res. 2014, 280, 14–29. [Google Scholar] [CrossRef]
- Michard, A. Rare earth element systematics in hydrothermal fluids. Geochim. Cosmochim. Acta 1989, 53, 745–750. [Google Scholar] [CrossRef]
- Williams-Jones, A.E.; Migdisov, A.A.; Samson, I.M. Hydrothermal mobilisation of the rare earth elements–a tale of “ceria” and “yttria”. Elements 2012, 8, 355–360. [Google Scholar] [CrossRef]
- Paoli, G.; Dini, A.; Rocchi, S. Footprints of element mobility during metasomatism linked to a late Miocene peraluminous granite intruding a carbonate host (Campiglia Marittima, Tuscany). Int. J. Earth Sci. 2019, 108, 1617–1641. [Google Scholar] [CrossRef]
- Lottermoser, B. Rare earth elements and hydrothermal ore formation processes. Ore Geol. Rev. 1992, 7, 25–41. [Google Scholar] [CrossRef]
- Zou, H.; Zindler, A. Constraints on the degree of dynamic partial melting and source composition using concentration ratios in magmas. Geochim. Cosmochim. Acta 1996, 60, 711–717. [Google Scholar] [CrossRef]
- Zou, H. Trace element fractionation during modal and nonmodal dynamic melting and open-system melting: A mathematical treatment. Geochim. Cosmochim. Acta 1998, 62, 1937–1945. [Google Scholar] [CrossRef]
- Zou, H. Modeling of trace element fractionation during non-modal dynamic melting with linear variations in mineral/melt distribution coefficients. Geochim. Cosmochim. Acta 2000, 64, 1095–1102. [Google Scholar] [CrossRef]
- Takahashi, Y.; Yoshida, H.; Sato, N.; Hama, K.; Yusa, Y.; Shimizu, H. W-and M-type tetrad effects in REE patterns for water–rock systems in the Tono uranium deposit, central Japan. Chem. Geol. 2002, 184, 311–335. [Google Scholar] [CrossRef]
- Humphris, S.E. The mobility of the rare earth elements in the crust. In Developments in Geochemistry; Elsevier: Amsterdam, The Netherlands, 1984; Volume 2, pp. 317–342. [Google Scholar]
- Jie, Z.; Da-Long, T.; Tun-Wen-Meng, Y.-Z.-Q.; Yang, L. Typomorphic Characteristics of Arsenopyrite in the Bake Gold Deposit, Eastern Guizhou Province. Geoscience 2011, 25, 750. [Google Scholar]
- Rice, S.; Langmuir, C.; Bender, J.; Hanson, G.; Bence, A.; Taylor, S. Basalts from Deep Sea Drilling Project holes 417A and 417D, fractionated melts of a light rare-earth depleted source. Initial Rep. Deep Sea Drill. Proj 1980, 51, 52–53. [Google Scholar]
- Pearce, J.A.; Peate, D.W. Tectonic implications of the composition of volcanic arc magmas. Annu. Rev. Earth Planet. Sci. 1995, 23, 251–285. [Google Scholar] [CrossRef]
- Wilson, M. Igneous Petrogenesis: A Global Tectonic Approach: London; Unwin Hyman: London, UK, 1989; 466p. [Google Scholar] [CrossRef]
- Shikazono, N.; Ogawa, Y.; Utada, M.; Ishiyama, D.; Mizuta, T.; Ishikawa, N.; Kubota, Y. Geochemical behavior of rare earth elements in hydrothermally altered rocks of the Kuroko mining area, Japan. J. Geochem. Explor. 2008, 98, 65–79. [Google Scholar] [CrossRef]
- Oreskes, N.; Einaudi, M.T. Origin of rare earth element-enriched hematite breccias at the Olympic Dam Cu-U-Au-Ag deposit, Roxby Downs, South Australia. Econ. Geol. 1990, 85, 1–28. [Google Scholar] [CrossRef]
- Parsapoor, A.; Khalili, M.; Mackizadeh, M. The behaviour of trace and rare earth elements (REE) during hydrothermal alteration in the Rangan area (Central Iran). J. Asian Earth Sci. 2009, 34, 123–134. [Google Scholar] [CrossRef]
- Abdelnasser, A. Genesis of the Gold Mineralization at Atud Area, Central Eastern Desert of Egypt: Geological, Ore Mineralogical and Geochemical Approaches. Ph.D. Thesis, Istanbul Technical University, Istanbul, Turkey, 2016. [Google Scholar]
- Abdelnasser, A.; Kumral, M.; Zoheir, B.; Karaman, M.; Weihed, P. REE geochemical characteristics and satellite-based mapping of hydrothermal alteration in Atud gold deposit, Egypt. J. Afr. Earth Sci. 2018, 145, 317–330. [Google Scholar] [CrossRef]
- Parr, J.M. Rare-earth element distribution in exhalites associated with Broken Hill-type mineralisation at the Pinnacles deposit, New South Wales, Australia. Chem. Geol. 1992, 100, 73–91. [Google Scholar] [CrossRef]
- Torres-Alvarado, I.S. Chemical equilibrium in hydrothermal systems: The case of Los Azufres geothermal field, Mexico. Int. Geol. Rev. 2002, 44, 639–652. [Google Scholar] [CrossRef]
- Jiang, N.; Sun, S.; Chu, X.; Mizuta, T.; Ishiyama, D. Mobilization and enrichment of high-field strength elements during late-and post-magmatic processes in the Shuiquangou syenitic complex, Northern China. Chem. Geol. 2003, 200, 117–128. [Google Scholar] [CrossRef]
- Hou, Z.; Yang, Z.; Lu, Y.; Kemp, A.; Zheng, Y.; Li, Q.; Tang, J.; Yang, Z.; Duan, L. A genetic linkage between subduction-and collision-related porphyry Cu deposits in continental collision zones. Geology 2015, 43, 247–250. [Google Scholar] [CrossRef]
- Hou, Z.; Zhou, Y.; Wang, R.; Zheng, Y.; He, W.; Zhao, M.; Evans, N.J.; Weinberg, R.F. Recycling of metal-fertilized lower continental crust: Origin of non-arc Au-rich porphyry deposits at cratonic edges. Geology 2017, 45, 563–566. [Google Scholar] [CrossRef]
- Rice, S.; Cuthbert, S.J.; Hursthouse, A. Tectono-magmatic controls of post-subduction gold mineralisation during late Caledonian soft continental collision in the Southern Uplands-Down-Longford Terrane, Britain and Ireland: A review. Ore Geol. Rev. 2018, 101, 74–104. [Google Scholar] [CrossRef]
- McCuaig, T.C.; Hronsky, J.M. The mineral system concept: The key to exploration targeting. Appl. Earth Sci. 2017, 126, 77–78. [Google Scholar] [CrossRef]
- Botros, N.S. Alteration zones: Are they a good target for gold deposits in Egypt. Arab Gulf J. Sci. Res. 2002, 20, 209–218. [Google Scholar]
- Zoheir, B.; Akawy, A.; Hassan, I. Role of fluid mixing and wallrock sulfidation in gold mineralization at the Semna mine area, central Eastern Desert of Egypt: Evidence from hydrothermal alteration, fluid inclusions and stable isotope data. Ore Geol. Rev. 2008, 34, 580–596. [Google Scholar] [CrossRef]
- Reyes, A.G. Petrology of Philippine geothermal systems and the application of alteration mineralogy to their assessment. J. Volcanol. Geotherm. Res. 1990, 43, 279–309. [Google Scholar] [CrossRef]
- Sillitoe, R. The influence of magmatic-hydrothermal models on exploration strategies for volcano-plutonic areas. Magmas Fluids Ore Depos. 1995, 266–272. [Google Scholar]
- Eliwa, H.; Breitkreuz, C.; Murata, M.; Khalaf, I.; Bühler, B.; Itaya, T.; Takahashi, T.; Hirahara, Y.; Miyazaki, T.; Kimura, J. SIMS zircon U–Pb and mica K–Ar geochronology, and Sr–Nd isotope geochemistry of Neoproterozoic granitoids and their bearing on the evolution of the north Eastern Desert, Egypt. Gondwana Res. 2014, 25, 1570–1598. [Google Scholar] [CrossRef]












| Chalcopyrite (n = 10) | Pyrite (n = 11) | |||||
|---|---|---|---|---|---|---|
| Dataset/Point | Min | Max | Av. | Min | Max | Av. |
| S | 32.11 | 34.72 | 33.56 | 49.95 | 52.84 | 51.87 |
| Fe | 32.05 | 36.07 | 32.99 | 46.22 | 47.46 | 47.04 |
| Te | 0.01 | 0.02 | 0.01 | 0.01 | 0.05 | 0.02 |
| Cu | 30.44 | 33.55 | 32.71 | 0.01 | 0.16 | 0.05 |
| Au | 0.02 | 0.04 | 0.03 | 0.01 | 0.05 | 0.03 |
| As | 0.03 | 0.14 | 0.06 | 0.03 | 3.58 | 0.80 |
| Ni | 0.01 | 0.03 | 0.02 | 0.01 | 0.01 | 0.01 |
| Ag | 0.01 | 0.08 | 0.03 | 0.02 | 0.05 | 0.03 |
| Sb | 0.51 | 0.51 | 0.51 | 0.06 | 0.06 | 0.06 |
| Zn | 0.01 | 0.04 | 0.02 | 0.01 | 0.02 | 0.02 |
| Total | 98.69 | 100.32 | 99.42 | 99.05 | 100.13 | 99.52 |
| atom % | ||||||
| S | 47.20 | 49.57 | 48.59 | 63.98 | 66.36 | 65.55 |
| Fe | 26.73 | 29.75 | 27.43 | 33.60 | 34.56 | 34.13 |
| Te | 0.01 | 0.01 | 0.01 | 0.02 | 0.02 | 0.02 |
| Cu | 22.06 | 24.55 | 23.91 | 0.01 | 0.10 | 0.03 |
| Au | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 |
| As | 0.02 | 0.09 | 0.04 | 0.02 | 1.96 | 0.43 |
| Ni | 0.01 | 0.02 | 0.02 | 0.01 | 0.01 | 0.01 |
| Ag | 0.01 | 0.03 | 0.01 | 0.01 | 0.02 | 0.01 |
| Sb | 0.20 | 0.20 | 0.20 | 0.02 | 0.02 | 0.02 |
| Zn | 0.01 | 0.03 | 0.02 | 0.01 | 0.01 | 0.01 |
| Sum | 100 | 100 | 100 | 100 | 100 | 100 |
| Formula | Cu0.96Fe1.1S1.94 | Fe1.02As0.01S1.97 | ||||
| S | 1.89 | 1.98 | 1.94 | 1.92 | 1.99 | 1.97 |
| Fe | 1.07 | 1.19 | 1.10 | 1.01 | 1.04 | 1.02 |
| Cu | 0.88 | 0.98 | 0.96 | |||
| As | 0.01 | 0.06 | 0.02 | |||
| Sum | 4.00 | 4.00 | 4.00 | 3.00 | 3.00 | 3.00 |
| Z-2 | Z-106 | Z-107 | Z-107a | Z-107b | Z-107c | Z-107d | |
|---|---|---|---|---|---|---|---|
| SiO2 | 75.28 | 75.35 | 73.60 | 74.71 | 74.62 | 74.46 | 74.63 |
| Al2O3 | 13.63 | 12.77 | 13.28 | 13.20 | 13.17 | 13.20 | 13.20 |
| Fe2O3 | 1.38 | 1.07 | 1.44 | 1.29 | 1.29 | 1.32 | 1.30 |
| MgO | 0.28 | 0.14 | 0.30 | 0.24 | 0.24 | 0.25 | 0.24 |
| CaO | 0.27 | 0.54 | 1.07 | 0.65 | 0.70 | 0.75 | 0.68 |
| Na2O | 4.66 | 3.83 | 4.10 | 4.17 | 4.13 | 4.14 | 4.16 |
| K2O | 3.40 | 5.41 | 5.34 | 4.80 | 4.92 | 4.96 | 4.86 |
| TiO2 | 0.23 | 0.14 | 0.22 | 0.19 | 0.19 | 0.20 | 0.20 |
| P2O5 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 |
| MnO | 0.02 | 0.02 | 0.02 | 0.02 | 0.02 | 0.02 | 0.02 |
| Cr2O3 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 |
| LOI | 0.70 | 0.55 | 0.39 | 0.54 | 0.52 | 0.50 | 0.52 |
| Sum | 99.87 | 99.84 | 99.78 | 99.83 | 99.82 | 99.82 | 99.83 |
| Trace and rare earth elements (REE) | |||||||
| As | 28.0 | 10.0 | 10.0 | 15.2 | 14.1 | 13.7 | 14.7 |
| Ba | 818.0 | 985.2 | 985.2 | 936.9 | 947.2 | 950.5 | 941.6 |
| Cd | 8.5 | 0.0 | 0.8 | 2.7 | 2.3 | 2.2 | 2.5 |
| Co | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Cs | 1.1 | 1.4 | 1.5 | 1.3 | 1.4 | 1.4 | 1.4 |
| Cu | 16.0 | 3.0 | 4.0 | 7.1 | 6.4 | 6.2 | 6.8 |
| Ga | 19.0 | 17.0 | 19.0 | 18.3 | 18.3 | 18.4 | 18.3 |
| Hf | 3.2 | 3.2 | 3.5 | 3.3 | 3.4 | 3.4 | 3.4 |
| Mo | 3.0 | 2.0 | 1.0 | 1.9 | 1.8 | 1.7 | 1.9 |
| Nb | 16.0 | 18.0 | 8.0 | 13.9 | 13.5 | 12.6 | 13.4 |
| Ni | 3.0 | 2.0 | 3.0 | 2.6 | 2.6 | 2.7 | 2.7 |
| Pb | 24.0 | 12.0 | 1.0 | 11.6 | 10.2 | 9.0 | 10.7 |
| Pd | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| Pt | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| Rb | 63.6 | 97.4 | 179.4 | 116.8 | 123.2 | 131.4 | 121.7 |
| Rh | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| Ru | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| S | 80.1 | 120.1 | 320.4 | 179.8 | 192.8 | 211.8 | 190.3 |
| Sb | 0.4 | 0.6 | 0.7 | 0.6 | 0.6 | 0.6 | 0.6 |
| Sn | 0.6 | 0.1 | 0.1 | 0.2 | 0.2 | 0.2 | 0.2 |
| Sr | 124.0 | 86.0 | 222.0 | 145.3 | 150.2 | 161.8 | 150.7 |
| Te | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| Tl | 0.4 | 0.6 | 0.8 | 0.6 | 0.6 | 0.7 | 0.6 |
| U | 5.6 | 5.4 | 8.3 | 6.4 | 6.6 | 6.8 | 6.6 |
| V | 7.0 | 4.0 | 8.0 | 6.3 | 6.3 | 6.6 | 6.4 |
| Zn | 18.0 | 24.0 | 27.0 | 23.3 | 23.9 | 24.3 | 23.6 |
| Zr | 99.0 | 96.0 | 124.0 | 106.8 | 108.1 | 110.6 | 108.1 |
| Sc | 1.0 | 3.0 | 1.0 | 1.7 | 1.7 | 1.6 | 1.7 |
| Y | 17.2 | 13.4 | 11.1 | 13.7 | 13.3 | 13.0 | 13.5 |
| La | 24.2 | 17.0 | 39.3 | 27.0 | 27.7 | 29.6 | 27.9 |
| Ce | 46.5 | 37.5 | 79.1 | 54.9 | 56.6 | 60.2 | 56.6 |
| Pr | 5.4 | 4.1 | 8.4 | 6.0 | 6.1 | 6.5 | 6.1 |
| Nd | 18.0 | 14.7 | 29.2 | 20.8 | 21.4 | 22.6 | 21.4 |
| Sm | 3.4 | 3.3 | 5.5 | 4.1 | 4.2 | 4.4 | 4.2 |
| Eu | 0.7 | 0.6 | 1.5 | 0.9 | 1.0 | 1.1 | 1.0 |
| Gd | 3.1 | 2.7 | 3.5 | 3.1 | 3.1 | 3.2 | 3.1 |
| Tb | 0.6 | 0.4 | 0.5 | 0.5 | 0.5 | 0.5 | 0.5 |
| Dy | 2.9 | 2.2 | 2.1 | 2.4 | 2.3 | 2.3 | 2.4 |
| Ho | 0.6 | 0.5 | 0.4 | 0.5 | 0.5 | 0.4 | 0.5 |
| Er | 1.7 | 1.2 | 1.0 | 1.3 | 1.2 | 1.2 | 1.3 |
| Tm | 0.3 | 0.3 | 0.1 | 0.2 | 0.2 | 0.2 | 0.2 |
| Yb | 1.6 | 1.5 | 0.9 | 1.3 | 1.3 | 1.2 | 1.3 |
| Lu | 0.2 | 0.3 | 0.2 | 0.2 | 0.2 | 0.2 | 0.2 |
| Th | 9.6 | 8.3 | 19.5 | 12.7 | 13.2 | 14.2 | 13.2 |
| Parameters | |||||||
| FeO* (FeO total) | 1.24 | 0.96 | 1.30 | 1.16 | 1.16 | 1.19 | 1.17 |
| FeO*/MgO | 4.44 | 6.88 | 4.32 | 4.90 | 4.90 | 4.77 | 4.85 |
| Na2O + K2O | 8.06 | 9.24 | 9.44 | 8.97 | 9.05 | 9.09 | 9.01 |
| ΣREE | 109.2 | 86.1 | 171.8 | 123.3 | 126.4 | 133.7 | 126.7 |
| ΣLREE | 98.2 | 77.2 | 163.0 | 113.8 | 117.0 | 124.4 | 117.3 |
| ΣHREE | 10.9 | 8.9 | 8.8 | 9.5 | 9.3 | 9.3 | 9.4 |
| (La/Yb)N | 10.2 | 7.8 | 27.6 | 13.7 | 14.5 | 16.1 | 14.4 |
| (La/Sm)N | 4.3 | 3.2 | 4.4 | 4.0 | 4.0 | 4.1 | 4.1 |
| (Gd/Yb)N | 1.5 | 1.5 | 2.9 | 1.9 | 1.9 | 2.0 | 1.9 |
| (Tb/Yb)N | 1.63 | 1.35 | 2.63 | 1.78 | 1.82 | 1.92 | 1.82 |
| Eu/Eu* | 0.6 | 0.6 | 1.1 | 0.8 | 0.8 | 0.9 | 0.8 |
| Synorogenic Granitoid Rocks | Late Orogenic Granitoid Rocks | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Z-10 | Z-44 | Z-103 | Z-33 | Z-38 | Z-46 | Z-98 | Z-43 | Z-104 | |
| SiO2 | 60.59 | 62.9 | 66.04 | 69.86 | 74.08 | 71.52 | 71.63 | 71.49 | 72.2 |
| Al2O3 | 16.73 | 15.64 | 15.75 | 13.86 | 13.05 | 14.46 | 13.93 | 14.13 | 14.3 |
| Fe2O3 | 4.37 | 4.07 | 2.99 | 1.86 | 1.36 | 1.23 | 1.39 | 1.31 | 1.09 |
| MgO | 2.14 | 2.01 | 1.53 | 0.97 | 0.19 | 0.38 | 0.64 | 0.66 | 0.34 |
| CaO | 4.69 | 4.38 | 3.71 | 1.85 | 0.81 | 1.06 | 1.2 | 0.96 | 1.01 |
| Na2O | 4.8 | 4.27 | 5.14 | 3.89 | 4.12 | 4.58 | 4.97 | 4.43 | 5.19 |
| K2O | 3.81 | 3.92 | 3.01 | 5.98 | 5.5 | 5.81 | 4.97 | 5.98 | 5.09 |
| TiO2 | 0.89 | 1.03 | 0.57 | 0.42 | 0.19 | 0.3 | 0.31 | 0.29 | 0.17 |
| P2O5 | 0.5 | 0.47 | 0.33 | 0.09 | 0.05 | 0.07 | 0.13 | 0.08 | 0.07 |
| MnO | 0.07 | 0.08 | 0.07 | 0.05 | 0.01 | 0.03 | 0.05 | 0.03 | 0.03 |
| Cr2O3 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 |
| Cl | 0.03 | 0.05 | 0.04 | 0.03 | 0.01 | 0.02 | 0.02 | 0.02 | 0.01 |
| LOI | 1.12 | 0.91 | 0.57 | 0.72 | 0.46 | 0.36 | 0.6 | 0.45 | 0.35 |
| Sum | 99.75 | 99.74 | 99.76 | 99.59 | 99.84 | 99.83 | 99.85 | 99.84 | 99.86 |
| Trace and rare earth elements (REE) | |||||||||
| As | 9 | 10 | 10 | 7 | 3 | 7 | 6 | 6 | 9 |
| Ba | 985.2 | 895.7 | 895.7 | 985.2 | 502 | 1164.4 | 825 | 895.7 | 895.7 |
| Cd | 3 | 0.5 | 0 | 1.6 | 1.1 | 0.2 | 0 | 3.7 | 0.4 |
| Co | 7 | 6 | 4 | 1 | 1 | 1 | 1 | 1 | 1 |
| Cs | 1.4 | 2.3 | 1.3 | 1.8 | 0.6 | 1.3 | 0.9 | 1.4 | 0.7 |
| Cu | 12 | 10 | 7 | 1 | 6 | 5 | 1 | 3 | 5 |
| Ga | 23 | 21 | 19 | 17 | 19 | 17 | 19 | 18 | 20 |
| Hf | 0.9 | 1.5 | 2 | 2.2 | 3.2 | 2.4 | 1.3 | 2.8 | 0.8 |
| Mo | 3 | 1 | 4 | 1 | 3 | 1 | 2 | 1 | 1 |
| Nb | 9 | 11 | 9 | 13 | 13 | 13 | 10 | 14 | 7 |
| Ni | 13 | 11 | 11 | 2 | 1 | 3 | 4 | 3 | 2 |
| Pb | 1 | 1 | 1 | 3 | 1 | 1 | 1 | 3 | 3 |
| Rb | 72.8 | 65.9 | 49.4 | 104.8 | 70.5 | 85 | 69.3 | 114.1 | 67.7 |
| S | 40 | 80.1 | 80.1 | 1001.1 | 360.4 | 40 | 120.1 | 80.1 | 40 |
| Sb | 0.2 | 0.1 | 0.1 | 0.1 | 0.2 | 0.1 | 0.1 | 0.1 | 0.1 |
| Sn | 0.1 | 0.5 | 0.1 | 0.7 | 0.1 | 0.3 | 0.1 | 0.8 | 0.1 |
| Sr | 845.6 | 845.6 | 748 | 130 | 149 | 137 | 201 | 114 | 237 |
| Tl | 0.3 | 0.4 | 0.3 | 0.5 | 0.4 | 0.4 | 0.2 | 0.4 | 0.3 |
| U | 1.2 | 2.5 | 2.1 | 2.2 | 5.6 | 1.8 | 1.5 | 4.3 | 0.9 |
| V | 41 | 45 | 28 | 9 | 6 | 7 | 11 | 8 | 9 |
| Zn | 68 | 65 | 44 | 36 | 12 | 17 | 36 | 22 | 33 |
| Zr | 214 | 237 | 166 | 194 | 89 | 178 | 142 | 183 | 155 |
| Sc | 7 | 8 | 3 | 3 | 1 | 2 | 4 | 1 | 1 |
| Y | 17 | 20.2 | 13 | 17.7 | 5.5 | 17.7 | 10.9 | 17.6 | 6.3 |
| La | 27 | 32 | 21.9 | 31.3 | 23 | 27.8 | 22.3 | 32.7 | 15.6 |
| Ce | 57.9 | 69.1 | 48.1 | 65.9 | 40.2 | 61 | 48.2 | 67.1 | 30.6 |
| Pr | 7 | 8.1 | 5.6 | 7.6 | 3.7 | 7.3 | 5.7 | 7.6 | 3.4 |
| Nd | 28.6 | 33.2 | 20.8 | 27.2 | 11.1 | 27.5 | 21.9 | 26.8 | 13 |
| Sm | 5.7 | 6.7 | 4.6 | 5.1 | 1.6 | 5.4 | 4 | 5.3 | 2.4 |
| Eu | 1.8 | 1.7 | 1.3 | 0.8 | 0.4 | 1 | 0.8 | 1.1 | 0.6 |
| Gd | 4.8 | 5.6 | 3.7 | 4.1 | 1.3 | 3.9 | 3.1 | 4 | 1.5 |
| Tb | 0.7 | 0.8 | 0.5 | 0.6 | 0.3 | 0.6 | 0.4 | 0.6 | 0.2 |
| Dy | 3.6 | 4.4 | 2.6 | 3.4 | 1 | 3.1 | 2.2 | 3.4 | 1.4 |
| Ho | 0.6 | 0.7 | 0.5 | 0.7 | 0.2 | 0.6 | 0.4 | 0.6 | 0.2 |
| Er | 1.7 | 2.3 | 1.4 | 1.8 | 0.6 | 1.7 | 1.1 | 2.1 | 0.7 |
| Tm | 0.2 | 0.3 | 0.2 | 0.2 | 0 | 0.2 | 0.1 | 0.3 | 0.1 |
| Yb | 1.7 | 2 | 1.3 | 1.9 | 0.7 | 1.8 | 1 | 1.8 | 0.6 |
| Lu | 0.3 | 0.3 | 0.2 | 0.3 | 0.1 | 0.3 | 0.1 | 0.3 | 0.1 |
| Th | 3.7 | 5.6 | 6.4 | 8.1 | 11.7 | 6.2 | 4.6 | 14.5 | 2.7 |
| Parameters | |||||||||
| FeO* (FeO total) | 3.93 | 3.66 | 2.69 | 1.67 | 1.22 | 1.11 | 1.25 | 1.18 | 0.98 |
| Na2O + K2O | 8.61 | 8.19 | 8.15 | 9.87 | 9.62 | 10.39 | 9.94 | 10.41 | 10.28 |
| Fe2O3 + TiO2 | 5.26 | 5.1 | 3.56 | 2.28 | 1.55 | 1.53 | 1.7 | 1.6 | 1.26 |
| FeO* + MnO | 4 | 3.74 | 2.76 | 1.72 | 1.23 | 1.14 | 1.3 | 1.21 | 1.01 |
| FeO*/(FeO* + MgO) | 0.65 | 0.65 | 0.64 | 0.63 | 0.87 | 0.74 | 0.66 | 0.64 | 0.74 |
| Y + Nb | 26.04 | 31.16 | 22.01 | 30.72 | 18.51 | 30.66 | 20.92 | 31.6 | 13.27 |
| Ore Zone | Alteration Zone 1 | Alteration Zone 2 | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Z12D | Z15 | Z12C | Z18D | Z18D1 | Z18E | Z24C | Z24D | Z25D | Z12A | Z12B | Z18A | Z24A | Z24B | Z24E | Z24F | Z24G | |
| SiO2 | 37.62 | 46.35 | 74.71 | 75.25 | 70.86 | 77.10 | 55.80 | 62.96 | 73.65 | 50.00 | 61.67 | 61.86 | 59.36 | 57.42 | 65.39 | 53.47 | 50.84 |
| Al2O3 | 18.78 | 17.48 | 14.25 | 13.55 | 14.89 | 11.93 | 22.03 | 16.53 | 15.86 | 17.89 | 16.38 | 15.26 | 15.22 | 15.66 | 16.05 | 16.32 | 15.81 |
| Fe2O3 | 28.27 | 22.13 | 2.61 | 3.12 | 4.98 | 3.57 | 7.26 | 8.66 | 1.85 | 5.68 | 4.29 | 4.80 | 6.64 | 8.41 | 4.17 | 7.24 | 6.32 |
| MgO | 0.84 | 0.79 | 0.50 | 0.52 | 0.44 | 0.29 | 0.98 | 0.84 | 0.48 | 2.76 | 1.75 | 1.78 | 1.46 | 2.13 | 1.48 | 1.99 | 2.89 |
| CaO | 0.27 | 0.05 | 0.02 | 0.06 | 0.04 | 0.06 | 0.45 | 0.67 | 0.06 | 3.67 | 3.51 | 3.38 | 6.88 | 3.54 | 3.01 | 6.29 | 7.76 |
| Na2O | 0.21 | 0.12 | 0.10 | 0.14 | 0.17 | 0.13 | 0.19 | 0.13 | 0.17 | 4.08 | 4.56 | 5.64 | 2.67 | 0.93 | 0.78 | 2.15 | 2.98 |
| K2O | 4.30 | 5.11 | 4.70 | 4.72 | 4.88 | 3.90 | 7.56 | 5.60 | 5.05 | 3.72 | 3.08 | 3.30 | 2.14 | 6.24 | 5.22 | 4.22 | 3.18 |
| TiO2 | 1.42 | 1.52 | 0.12 | 0.11 | 0.12 | 0.12 | 0.85 | 0.62 | 0.09 | 0.93 | 0.84 | 0.88 | 0.67 | 0.82 | 0.36 | 1.30 | 1.25 |
| P2O5 | 0.30 | 0.29 | 0.02 | 0.02 | 0.03 | 0.02 | 0.29 | 0.20 | 0.01 | 0.31 | 0.11 | 0.37 | 0.24 | 0.26 | 0.11 | 0.49 | 0.49 |
| MnO | 0.01 | 0.01 | 0.01 | 0.01 | 0.02 | 0.01 | 0.26 | 0.21 | 0.14 | 0.38 | 0.03 | 0.08 | 0.14 | 0.35 | 0.16 | 0.13 | 0.10 |
| Cr2O3 | 0.01 | 0.01 | 0.01 | 0.01 | 0.42 | 0.29 | 0.38 | 0.01 | 0.32 | 0.01 | 0.01 | 0.01 | 0.01 | 0.25 | 0.01 | 0.31 | 0.02 |
| Cl | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.02 | 0.01 | 0.01 | 0.02 | 0.02 | 0.02 | 0.03 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 |
| LOI | 6.86 | 5.55 | 2.78 | 2.27 | 2.56 | 2.34 | 3.72 | 3.31 | 2.14 | 5.02 | 3.26 | 2.07 | 4.34 | 3.73 | 3.14 | 5.88 | 7.91 |
| Sum | 98.9 | 99.42 | 99.84 | 99.79 | 99.42 | 99.78 | 99.78 | 99.75 | 99.84 | 94.47 | 99.51 | 99.46 | 99.78 | 99.75 | 99.89 | 99.8 | 99.56 |
| Trace and rare earth elements (REE) | |||||||||||||||||
| As | 24.0 | 56.0 | 23.0 | 20.0 | 37.0 | 31.0 | 306.0 | 472.0 | 25.0 | 26.0 | 20.0 | 17.0 | 17.0 | 182.0 | 75.0 | 121.0 | 19.0 |
| Au | 0.0 | 1.3 | 0.5 | 0.4 | 0.0 | 2.6 | 0.0 | 0.7 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
| Ba | 628.0 | 960.1 | 561.0 | 419.0 | 389.0 | 351.0 | 898.3 | 912.7 | 241.0 | 878.6 | 859.8 | 1067.6 | 897.4 | 1008.5 | 398.0 | 424.0 | 1569.2 |
| Cd | 0.0 | 0.0 | 0.0 | 0.4 | 0.0 | 0.0 | 1.4 | 0.6 | 2.8 | 6.4 | 0.8 | 0.0 | 0.0 | 2.3 | 0.8 | 0.0 | 0.0 |
| Co | 23.0 | 41.0 | 20.0 | 24.0 | 33.0 | 49.0 | 27.0 | 67.0 | 44.0 | 87.0 | 34.0 | 48.0 | 32.0 | 36.0 | 23.0 | 41.0 | 28.0 |
| Cs | 2.3 | 1.3 | 1.0 | 0.9 | 0.8 | 0.7 | 14.0 | 6.3 | 2.5 | 3.8 | 1.2 | 0.8 | 1.2 | 4.6 | 3.4 | 3.9 | 3.0 |
| Cu | 1156.7 | 442.0 | 55.0 | 12.0 | 20.0 | 27.0 | 22.0 | 13.0 | 63.0 | 39,068.5 | 1791.8 | 16.0 | 13.0 | 99.0 | 111.0 | 15.0 | 26.0 |
| Ga | 33.0 | 28.0 | 21.0 | 18.0 | 20.0 | 19.0 | 21.0 | 16.0 | 18.0 | 27.0 | 20.0 | 1.0 | 15.0 | 19.0 | 16.0 | 21.0 | 19.0 |
| Hf | 3.4 | 3.5 | 2.8 | 2.5 | 2.0 | 1.4 | 0.9 | 0.3 | 1.4 | 1.0 | 0.0 | 0.0 | 1.4 | 0.3 | 2.3 | 2.0 | 2.9 |
| Mo | 11.0 | 19.0 | 3.0 | 4.0 | 25.0 | 4.0 | 2.0 | 2.0 | 1.0 | 2.0 | 2.0 | 2.0 | 2.0 | 2.0 | 1.0 | 1.0 | 2.0 |
| Nb | 13.0 | 11.0 | 18.0 | 16.0 | 16.0 | 17.0 | 5.0 | 5.0 | 20.0 | 10.0 | 10.0 | 9.0 | 6.0 | 5.0 | 12.0 | 8.0 | 9.0 |
| Ni | 82.0 | 50.0 | 6.0 | 7.0 | 6.0 | 5.0 | 16.0 | 24.0 | 7.0 | 25.0 | 14.0 | 10.0 | 17.0 | 16.0 | 8.0 | 63.0 | 72.0 |
| Pb | 1.0 | 1.0 | 1.0 | 1.0 | 25.0 | 1.0 | 73.0 | 68.0 | 42.0 | 1.0 | 1.0 | 1.0 | 5.0 | 1.0 | 19.0 | 1.0 | 1.0 |
| Rb | 103.8 | 111.5 | 109.6 | 101.7 | 80.6 | 86.7 | 266.0 | 168.2 | 125.0 | 108.1 | 53.4 | 63.0 | 51.4 | 165.2 | 127.1 | 110.3 | 77.0 |
| S | 3067.9 | 1383.1 | 294.0 | 599.1 | 2017.9 | 589.1 | 113.4 | 221.7 | 219.3 | 161.7 | 260.2 | 1293.4 | 233.5 | 146.1 | 40.0 | 284.4 | 596.7 |
| Sb | 1.3 | 1.4 | 0.4 | 0.7 | 0.4 | 0.3 | 3.4 | 3.0 | 0.8 | 1.0 | 0.6 | 0.1 | 1.2 | 3.3 | 1.6 | 1.4 | 2.2 |
| Sn | 0.1 | 0.3 | 0.6 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 |
| Sr | 470.0 | 335.0 | 63.0 | 32.0 | 47.0 | 29.0 | 35.0 | 39.0 | 17.0 | 180.0 | 658.0 | 580.0 | 321.0 | 80.0 | 38.0 | 214.0 | 422.0 |
| Tl | 0.5 | 0.6 | 0.5 | 0.4 | 0.4 | 0.4 | 1.8 | 1.0 | 0.7 | 0.7 | 0.3 | 0.3 | 0.4 | 0.9 | 0.7 | 0.6 | 0.5 |
| U | 4.5 | 3.7 | 4.6 | 4.2 | 3.9 | 4.3 | 0.8 | 0.7 | 3.6 | 5.4 | 2.5 | 1.2 | 0.8 | 0.8 | 1.6 | 0.8 | 0.6 |
| V | 195.0 | 120.0 | 12.0 | 11.0 | 9.0 | 9.0 | 113.0 | 87.0 | 4.0 | 75.0 | 54.0 | 58.0 | 69.0 | 111.0 | 42.0 | 91.0 | 85.0 |
| Zn | 51.0 | 29.0 | 15.0 | 14.0 | 13.0 | 12.0 | 152.0 | 63.0 | 481.0 | 3473.7 | 88.0 | 76.0 | 90.0 | 204.0 | 182.0 | 102.0 | 122.0 |
| Zr | 174.0 | 172.0 | 107.0 | 99.0 | 98.0 | 101.0 | 124.0 | 123.0 | 93.0 | 242.0 | 199.0 | 234.0 | 160.0 | 100.0 | 120.0 | 187.0 | 178.0 |
| Sc | 10.0 | 12.0 | 1.0 | 3.0 | 1.0 | 1.0 | 22.0 | 15.0 | 1.0 | 11.0 | 7.0 | 5.0 | 16.0 | 21.0 | 9.0 | 9.0 | 8.0 |
| Y | 12.2 | 14.1 | 11.9 | 11.0 | 8.0 | 9.4 | 23.3 | 19.5 | 9.7 | 19.8 | 21.3 | 18.9 | 24.1 | 24.0 | 12.4 | 11.3 | 10.2 |
| La | 18.9 | 22.6 | 28.6 | 22.0 | 18.7 | 24.1 | 12.7 | 10.6 | 20.0 | 28.7 | 14.4 | 25.4 | 13.2 | 11.2 | 14.7 | 23.5 | 22.5 |
| Ce | 39.3 | 45.4 | 59.6 | 47.3 | 40.2 | 48.7 | 27.3 | 23.9 | 39.0 | 66.7 | 33.7 | 58.4 | 31.8 | 25.4 | 30.1 | 53.1 | 52.0 |
| Pr | 4.8 | 5.5 | 7.0 | 5.6 | 4.5 | 5.4 | 4.1 | 3.3 | 3.7 | 8.1 | 4.2 | 7.2 | 4.4 | 3.5 | 3.4 | 6.6 | 6.6 |
| Nd | 18.1 | 21.0 | 27.1 | 17.9 | 15.3 | 19.8 | 17.2 | 15.8 | 9.8 | 32.8 | 19.5 | 30.6 | 19.0 | 15.8 | 12.1 | 29.4 | 27.6 |
| Sm | 3.7 | 4.1 | 4.2 | 3.8 | 2.4 | 3.5 | 4.5 | 3.8 | 1.6 | 6.5 | 5.2 | 5.9 | 4.9 | 4.6 | 2.6 | 5.9 | 6.0 |
| Eu | 1.0 | 0.8 | 0.8 | 0.6 | 0.4 | 0.5 | 1.4 | 1.1 | 0.5 | 2.0 | 1.6 | 2.0 | 1.3 | 1.6 | 0.6 | 1.8 | 2.1 |
| Gd | 2.8 | 3.5 | 3.2 | 2.7 | 1.7 | 2.1 | 4.8 | 3.7 | 1.5 | 5.6 | 4.6 | 5.4 | 4.8 | 4.0 | 2.4 | 4.6 | 4.4 |
| Tb | 0.3 | 0.5 | 0.4 | 0.3 | 0.3 | 0.3 | 0.7 | 0.6 | 0.3 | 0.8 | 0.7 | 0.8 | 0.8 | 0.6 | 0.4 | 0.6 | 0.6 |
| Dy | 2.1 | 3.0 | 2.3 | 1.8 | 1.4 | 1.7 | 4.0 | 2.8 | 1.3 | 3.7 | 3.9 | 3.7 | 4.7 | 3.7 | 2.1 | 2.5 | 2.6 |
| Ho | 0.4 | 0.6 | 0.3 | 0.3 | 0.2 | 0.3 | 0.8 | 0.6 | 0.3 | 0.6 | 0.7 | 0.7 | 0.9 | 0.8 | 0.4 | 0.4 | 0.4 |
| Er | 1.1 | 1.6 | 1.2 | 1.3 | 1.0 | 1.0 | 2.7 | 2.0 | 1.2 | 2.1 | 1.7 | 2.0 | 2.7 | 2.3 | 1.6 | 1.2 | 1.1 |
| Tm | 0.1 | 0.2 | 0.2 | 0.3 | 0.1 | 0.2 | 0.4 | 0.3 | 0.1 | 0.2 | 0.3 | 0.2 | 0.3 | 0.4 | 0.2 | 0.1 | 0.1 |
| Yb | 1.3 | 1.7 | 1.2 | 1.4 | 1.2 | 1.4 | 2.4 | 2.0 | 1.0 | 1.6 | 1.8 | 1.5 | 2.6 | 3.2 | 1.8 | 1.2 | 0.9 |
| Lu | 0.2 | 0.3 | 0.2 | 0.2 | 0.1 | 0.2 | 0.4 | 0.3 | 0.2 | 0.2 | 0.3 | 0.2 | 0.5 | 0.4 | 0.3 | 0.1 | 0.1 |
| Th | 2.6 | 2.6 | 8.0 | 7.7 | 7.6 | 7.0 | 1.6 | 1.4 | 11.4 | 4.4 | 3.4 | 3.3 | 2.2 | 1.2 | 6.4 | 1.7 | 1.7 |
| Parameters | |||||||||||||||||
| FeO* (FeO total) | 25.44 | 19.92 | 2.35 | 2.81 | 4.48 | 3.21 | 6.53 | 7.79 | 1.67 | 5.11 | 3.86 | 4.32 | 5.98 | 7.57 | 3.75 | 6.52 | 5.69 |
| CCPI | 85.35 | 79.84 | 37.25 | 40.64 | 49.36 | 46.50 | 49.23 | 60.11 | 29.12 | 50.23 | 42.34 | 40.56 | 60.72 | 57.50 | 46.59 | 57.18 | 58.20 |
| Ishikawa AI | 91.46 | 97.20 | 97.74 | 96.32 | 96.20 | 95.66 | 93.03 | 88.95 | 96.01 | 45.54 | 37.44 | 36.03 | 27.38 | 65.19 | 63.87 | 42.39 | 36.11 |
| Na2O + CaO | 0.48 | 0.17 | 0.12 | 0.20 | 0.21 | 0.19 | 0.64 | 0.80 | 0.23 | 7.75 | 8.07 | 9.02 | 9.55 | 4.47 | 3.79 | 8.44 | 10.74 |
| MgO + FeO* | 26.28 | 20.71 | 2.85 | 3.33 | 4.92 | 3.50 | 7.51 | 8.63 | 2.15 | 7.87 | 5.61 | 6.10 | 7.44 | 9.70 | 5.23 | 8.51 | 8.58 |
| ΣREE | 94.03 | 110.61 | 136.39 | 105.57 | 87.48 | 109.25 | 83.33 | 70.85 | 80.52 | 159.62 | 92.39 | 143.99 | 91.80 | 77.53 | 72.60 | 130.98 | 127.14 |
| ΣLREE | 85.72 | 99.31 | 127.30 | 97.27 | 81.50 | 102.08 | 67.17 | 58.54 | 74.62 | 144.82 | 78.55 | 129.47 | 74.59 | 62.04 | 63.43 | 120.28 | 116.88 |
| ΣHREE | 8.31 | 11.29 | 9.09 | 8.30 | 5.98 | 7.17 | 16.16 | 12.31 | 5.90 | 14.80 | 13.84 | 14.51 | 17.21 | 15.48 | 9.17 | 10.70 | 10.26 |
| ΣHREE/ΣREE | 0.09 | 0.10 | 0.07 | 0.08 | 0.07 | 0.07 | 0.19 | 0.17 | 0.07 | 0.09 | 0.15 | 0.10 | 0.19 | 0.20 | 0.13 | 0.08 | 0.08 |
| ΣLREE/ΣREE | 0.91 | 0.90 | 0.93 | 0.92 | 0.93 | 0.93 | 0.81 | 0.83 | 0.93 | 0.91 | 0.85 | 0.90 | 0.81 | 0.80 | 0.87 | 0.92 | 0.92 |
| Eu/Eu* | 0.91 | 0.61 | 0.66 | 0.62 | 0.67 | 0.60 | 0.93 | 0.90 | 0.95 | 1.03 | 1.03 | 1.07 | 0.83 | 1.11 | 0.72 | 1.06 | 1.28 |
| K2O Index (K.I.) | 76.51 | 84.18 | 88.35 | 86.76 | 88.25 | 89.04 | 82.35 | 77.35 | 87.67 | 26.14 | 23.88 | 23.40 | 16.27 | 48.60 | 49.76 | 28.81 | 18.92 |
| MgO Index (M.I.) | 14.95 | 13.01 | 9.40 | 9.56 | 7.96 | 6.62 | 10.68 | 11.60 | 8.33 | 19.40 | 13.57 | 12.62 | 11.10 | 16.59 | 14.11 | 13.58 | 17.19 |
| Ore Zone | Zone-1 | Zone-2 | ||||
|---|---|---|---|---|---|---|
| (Mfi − Moi)/Mo | (Mfi − Moi)/Moi | (Mfi − Moi)/Mo | (Mfi − Moi)/Moi | (Mfi − Moi)/Mo | (Mfi − Moi)/Moi | |
| SiO2 | −26.03 | −0.41 | 8.94 | 0.14 | −5.79 | −0.09 |
| Al2O3 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Fe2O3 | 0.93 | 4.87 | 0.05 | 0.25 | 0.11 | 0.58 |
| FeO | 15.80 | 4.85 | 0.77 | 0.24 | 1.81 | 0.56 |
| MgO | −1.17 | −0.62 | −1.29 | −0.68 | 0.14 | 0.07 |
| CaO | −4.12 | −0.97 | −4.06 | −0.95 | 0.49 | 0.12 |
| Na2O | −4.59 | −0.97 | −4.59 | −0.97 | −1.78 | −0.38 |
| K2O | 0.59 | 0.16 | 1.77 | 0.50 | 0.30 | 0.09 |
| TiO2 | 0.47 | 0.57 | −0.53 | −0.64 | 0.05 | 0.06 |
| P2O5 | −0.17 | −0.38 | −0.35 | −0.81 | −0.13 | −0.30 |
| MnO | −0.06 | −0.87 | 0.02 | 0.32 | 0.10 | 1.42 |
| Cr2O3 | 0.00 | −0.12 | 0.21 | 20.62 | 0.07 | 6.99 |
| LOI | 4.62 | 5.32 | 1.94 | 2.23 | 3.54 | 4.07 |
| As | 25.72 | 2.66 | 124.76 | 12.90 | 49.85 | 5.16 |
| Au | 0.58 | 29.08 | 0.61 | 30.40 | ||
| Ba | −222.98 | −0.24 | −370.74 | −0.40 | −39.27 | −0.04 |
| Cd | −1.16 | −1.00 | −0.38 | −0.33 | 0.13 | 0.11 |
| Co | 22.64 | 3.99 | 33.15 | 5.85 | 35.38 | 6.24 |
| Cu | 697.55 | 72.14 | 21.51 | 2.23 | 5123.28 | 529.81 |
| Ga | 5.98 | 0.29 | −1.44 | −0.07 | −3.78 | −0.18 |
| Hf | 1.61 | 1.09 | 0.18 | 0.12 | −0.24 | −0.16 |
| Mo | 10.60 | 3.97 | 3.36 | 1.26 | −0.92 | −0.35 |
| Nb | 0.95 | 0.10 | 4.60 | 0.48 | −1.06 | −0.11 |
| Ni | 46.72 | 4.00 | −1.23 | −0.11 | 16.41 | 1.41 |
| Pb | −0.12 | −0.12 | 30.03 | 30.03 | 2.74 | 2.74 |
| Rb | 32.53 | 0.52 | 75.25 | 1.20 | 31.55 | 0.50 |
| S | 1902.22 | 28.50 | 529.55 | 7.94 | 309.56 | 4.64 |
| Sb | 1.06 | 7.08 | 1.18 | 7.85 | 1.27 | 8.45 |
| Sn | −0.02 | −0.12 | −0.07 | −0.35 | −0.14 | −0.74 |
| Sr | −456.96 | −0.56 | −774.53 | −0.95 | −502.01 | −0.62 |
| U | 1.64 | 0.84 | 1.29 | 0.66 | −0.24 | −0.13 |
| V | 101.34 | 2.67 | −1.97 | −0.05 | 34.99 | 0.92 |
| Zn | −23.61 | −0.40 | 51.30 | 0.87 | 482.20 | 8.17 |
| Zr | −52.61 | −0.26 | −96.10 | −0.47 | −28.50 | −0.14 |
| Mass Change | MC = −11.528 | MC = 2.953 | MC = −0.187 | |||
| Volume Change | VC = −26.318 | VC = 0.746 | VC = −1.623 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
El-Wardany, R.M.; Jiao, J.; Zoheir, B.; Kumral, M.; Kaya, M.; Abdelnasser, A. Post-Subduction Granite Magmatism and Gold-Sulfide Mineralization in the Abu Zawal (Fatira) Area, Eastern Desert, Egypt. Minerals 2023, 13, 489. https://doi.org/10.3390/min13040489
El-Wardany RM, Jiao J, Zoheir B, Kumral M, Kaya M, Abdelnasser A. Post-Subduction Granite Magmatism and Gold-Sulfide Mineralization in the Abu Zawal (Fatira) Area, Eastern Desert, Egypt. Minerals. 2023; 13(4):489. https://doi.org/10.3390/min13040489
Chicago/Turabian StyleEl-Wardany, Refaey M., Jiangang Jiao, Basem Zoheir, Mustafa Kumral, Mustafa Kaya, and Amr Abdelnasser. 2023. "Post-Subduction Granite Magmatism and Gold-Sulfide Mineralization in the Abu Zawal (Fatira) Area, Eastern Desert, Egypt" Minerals 13, no. 4: 489. https://doi.org/10.3390/min13040489
APA StyleEl-Wardany, R. M., Jiao, J., Zoheir, B., Kumral, M., Kaya, M., & Abdelnasser, A. (2023). Post-Subduction Granite Magmatism and Gold-Sulfide Mineralization in the Abu Zawal (Fatira) Area, Eastern Desert, Egypt. Minerals, 13(4), 489. https://doi.org/10.3390/min13040489













