Abstract
The flotation separation of enargite from copper sulfide minerals is difficult owing to the similar floatability and reagent adsorption characteristics of these minerals. In this study, the effect of oxidation treatment using NaClO flow with FeCl3 on the flotation separation of covellite and enargite was systematically investigated. Micro-flotation tests and contact angle measurements indicated that the addition of NaClO and FeCl3 increased the hydrophobicity difference between covellite and enargite. The bench-scale flotation test results show that the bulk copper concentrate could be separated into two products: a low-arsenic-containing (0.46%) and a high-arsenic-containing (5.18%) copper concentrate. X-ray photoelectron spectroscopy and scanning electron microscopy revealed that the oxidization treatment of NaClO caused the accumulation of oxides on the covellite surface, but not on the enargite surface. The varying precipitation of ferric hydroxide on the surfaces of covellite and enargite further exacerbated the difference in the hydrophilicity of these minerals. Thus, a possible method for separating enargite from covellite was obtained through oxidation treatment using NaClO and FeCl3.
1. Introduction
With the increasing global demand for copper metal, it has become necessary for mineral processing industries to process low-grade or arsenic-bearing copper deposits [1]. The presence of arsenic in copper concentrates reduces the product quality, such as in terms of the electrical conductivity and ductility, and causes serious environmental problems during smelting. Therefore, nations have mandated that smelters keep arsenic levels in copper concentrates at or below 0.5% [2,3,4].
Enargite, a major arsenic-bearing mineral, is often associated with copper sulfide minerals such as chalcocite, covellite, chalcopyrite, bornite, and tennantite, in copper deposits [5,6]. Selective flotation is considered an ideal low-pollution method for separating enargite from copper sulfide minerals. However, the similar surface properties and reagent adsorption characteristics of these minerals pose a significant challenge for their flotation separation [7]. Therefore, several methods have been proposed to solve this problem.
Castro et al., reported the separation of enargite from chalcopyrite using a mixture of magnesium-ammonium (MAA). A limited depressant effect was observed, with the best results reported at pH 10 and an MAA dosage of 2.5 kg/t, resulting in an arsenic recovery of approximately 55%. This was attributed to the formation of MgNH4AsO4·6H2O on the enargite surface [8]. Menacho et al., proposed controlled pulp potential (Eh) flotation as a prospective approach to promote the separation of arsenic–copper minerals from other copper sulfides, owing to the strong Eh dependence of arsenic mineral flotation [9]. Later investigations of this phenomenon found that the separation mechanism involved the oxidation of minerals and collectors. Several studies have focused on the selective oxidation of enargite and copper sulfide minerals [10,11,12,13,14]. Suyantara et al., demonstrated the separation of copper sulfide and arsenic-bearing copper minerals through an oxidation treatment using H2O2, where the hydrophilic species formed on the mineral surfaces included CuO, Cu(OH)2, FeOOH, and Fe2(SO4)3. They found that the combination of H2O2 treatment and potassium amyl xanthate (PAX) addition produced the lowest As grade (0.15%) in the tailing after 30 min of flotation compared to that of other methods [15]. Furthermore, Suyantara et al., reported that the addition of PAX followed by Na2SO3 treatment could separate enargite from chalcopyrite. In the presence of 10 mM Na2SO3 and 0.1 mM PAX, the recovery of enargite was more than 80%, while that of covellite was less than 20% [16]. Although significant progress has been made regarding techniques for extracting enargite from copper sulfide minerals, an efficient flotation technique that can specifically target arsenic minerals for industrial use has not yet been developed. Moreover, the separation of enargite and covellite has not been investigated [17].
Several studies have demonstrated that strong oxidants can inhibit the flotation of copper sulfide minerals [18,19]. However, the literature data and our experimental results noted that an extremely large amount of oxidant is needed to suppress minerals with good natural floatability, such as enargite and covellite. Therefore, this study investigated differences in the oxidation characteristics of covellite and enargite. In addition, their wettability and flotation behavior under oxidation conditions were analyzed.
2. Materials and Methods
2.1. Materials and Reagents
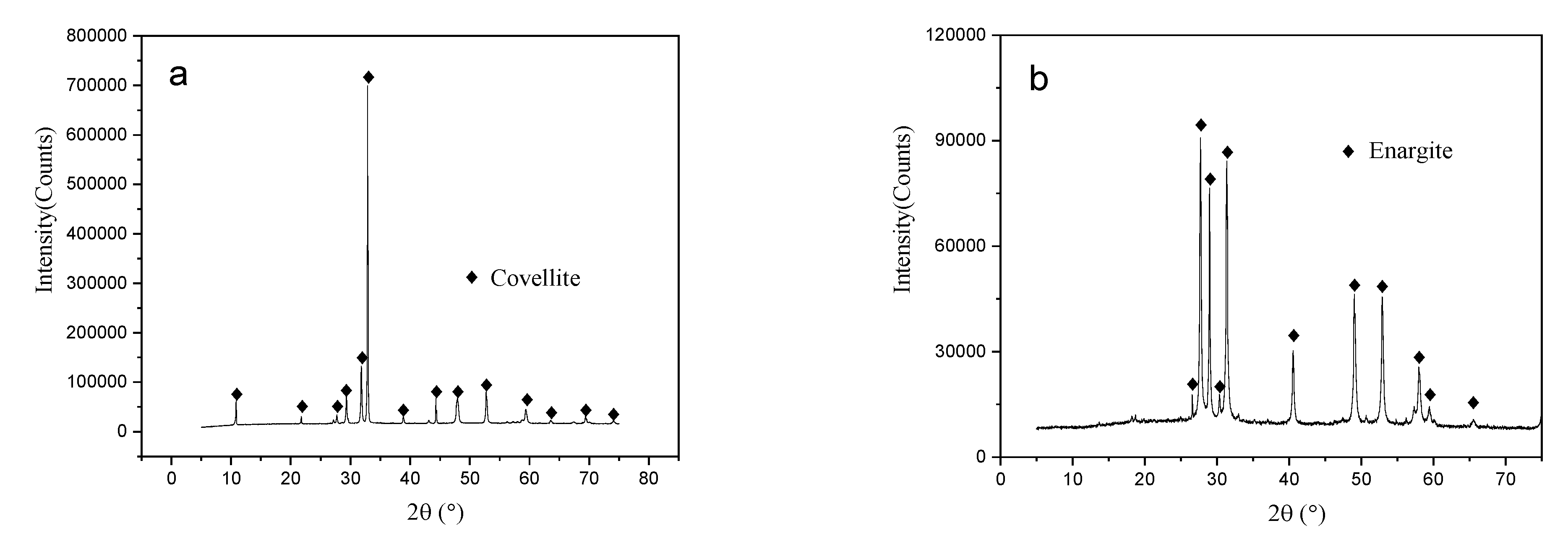
High-purity enargite and covellite specimens were hand-picked from the Timok mine in Serbia. The specimens were washed with distilled water and dried before further treatment. The samples were then manually ground using an agate mortar to obtain the desired particle size fractions. All samples were ultrasonically cleaned and vacuum-dried to avoid oxidation during crushing and grinding. X-ray diffraction (XRD) analysis indicated that the samples corresponded to covellite (PDF card #98-000-0176, Figure 1a) and enargite (PDF card #98-000-0205, Figure 1b) standards. Chemical composition analysis of the enargite and covellite samples was performed using inductively coupled plasma mass spectroscopy (ICP-MS). The elemental contents of copper and arsenic in the enargite sample were 47.2% and 18.6%, respectively, whereas those in the covellite sample were 61.4% and 32.1%, respectively. All specimens had purities of ≥94%.
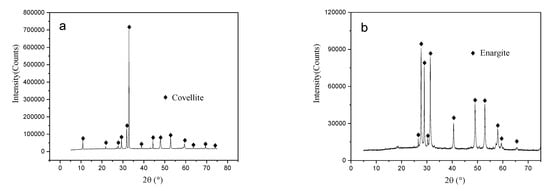
Figure 1.
XRD patterns of the covellite and enargite samples: (a) covellite, (b) enargite.
A bulk concentrate with a copper grade of 23.82% and an arsenic grade of 1.43% was used for the bench-scale flotation tests. Mineral liberation analysis of the bulk concentrate showed that the major copper sulfide mineral in the concentrate was covellite, and the main arsenic-bearing copper mineral was enargite. Pyrite and other gangue minerals accounted for 28.6% and 5.4%, respectively. Wet sieving analysis revealed that the particle size was 74 μm for 71% of the bulk concentrate.
Ammonium dibutyl dithiophosphate (ADD), methyl isobutyl carbinol (MIBC) hydrochloric acid, and CaO were purchased from Sinopharm Chemical Reagent Co. Ltd., Shanghai, China. NaClO and FeCl3 are common copper sulfide mineral depressants and, hence, were also used in this study. Deionized water was used throughout, and all experiments were conducted at 26 ± 1 °C.
2.2. Methods
2.2.1. Micro-Flotation Experiments
Micro-flotation tests of the individual minerals were carried out in a 40 mL flotation cell with a mechanical agitator at a rotational speed of 1992 rpm. Particle size fractions of −74–+38 μm were used for the flotation experiments. A mineral sample (2.0 g) was transferred to a 40 mL mechanical flotation cell to form a flotation pulp with 30 mL deionized water. After 1 min of conditioning, lime was added to regulate the pulp pH. Subsequently, as adjustors, freshly prepared solutions of NaClO followed by FeCl3 in specific concentrations were added. The collector (ADD, 1 × 10−5 mol/L) and frother (MIBC, 1 × 10−5 mol/L) were then sequentially added, and the mixture was conditioned for 2 and 1 min, respectively. Finally, the flotation froth was collected over 5 min. After filtration and drying, the concentrates and tailings were weighed, and the recovery of each mineral was calculated.
2.2.2. X-ray Photoelectron Spectroscopy
The enargite and covellite specimens obtained after different treatments were analyzed by X-ray photoelectron spectroscopy (XPS, ESCALAB 250, VG Systems, Waltham, MA, USA). The specimens were pulped at pH 10. NaClO and FeCl3 solutions were added at 2 and 0.5 g/L, respectively, with conditioning times of 30 and 10 min. The siphon technique was then used to remove the supernatant. Subsequently, the solid particles were washed, filtered, dried under vacuum, and stored in a plastic container under vacuum to prevent oxidation. Spectral data analysis and peak fitting were conducted using Thermo Avantage software. The binding energies were calibrated using C1s, which has a binding energy of 284.8 eV, as the reference.
2.2.3. Scanning Electron Microscopy
The surface morphologies of covellite and enargite before and after depressant treatment were measured using scanning electron microscopy (SEM, JSM 5600LV, Tokyo, Japan). The preparation process of the SEM samples was the same as that of the XPS samples.
2.2.4. Contact Angle Measurements
Contact angle measurements were carried out on the covellite and enargite specimens using a DSA255 goniometer (DSA25S, KRŰSS, Hamburg, Germany) via the sessile drop method. Fresh specimens were cut into a size of 10 × 10 × 5 mm3, using a slicer, and sequentially abraded on 400#, 600#, and 1200# mesh silicon carbide sandpapers. Hydrochloric acid diluted with deionized water was applied to clean the abraded specimens three times after vacuum drying at 40 °C. The method of specimen treatment by reagents was the same as that of the micro-flotation. Each specimen was measured three times, and the results were averaged to ensure accuracy.
2.2.5. Bench-Scale Flotation
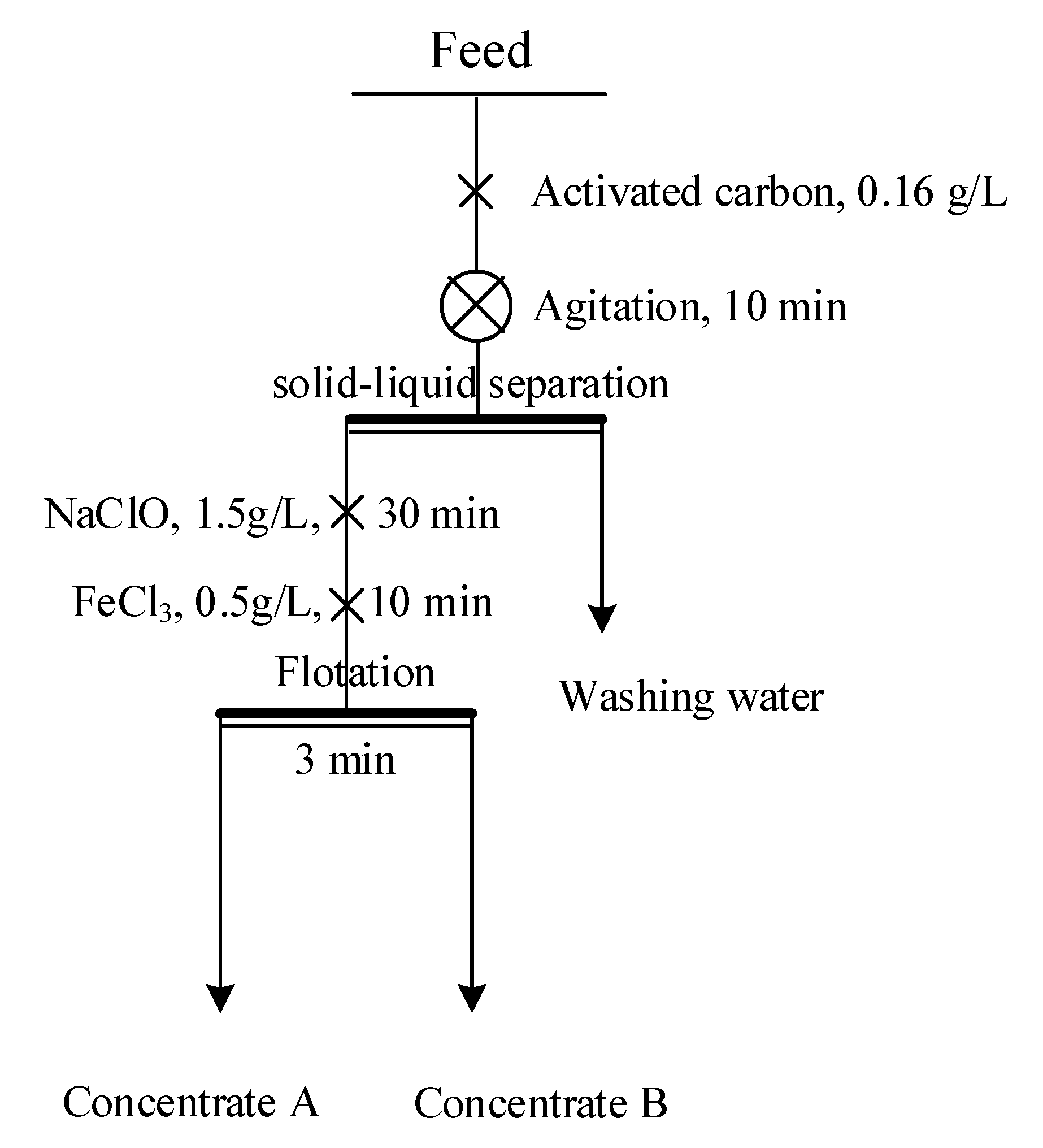
The bulk concentrate was first agitated in tap water for 10 min with the addition of 500 g/t activated carbon at a pulp density of 35% and a rotation speed of 1800 rpm. Subsequently, the pulp was left to stand for 1 h, and the supernatant was decanted. After the reagent was removed, the pulp was diluted to a pulp density of 20% and used for flotation tests. Bench-scale flotation tests were conducted in a 1.5 L XFD-type flotation machine (Changchun, China). The flotation tests were carried out following the flowchart shown in Figure 2. After flotation, the froth products were collected, filtered, dried in an oven for 24 h at 80 °C, weighed, and analyzed. Each flotation test was repeated twice, and the standard deviations were calculated.
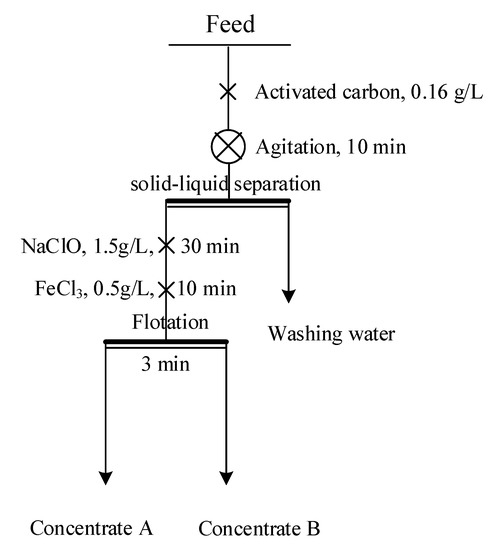
Figure 2.
Schematic of the bench-scale flotation test.
3. Results
3.1. Micro-Flotation of the Pure Mineral Sample
3.1.1. Flotation Performance of Covellite and Enargite
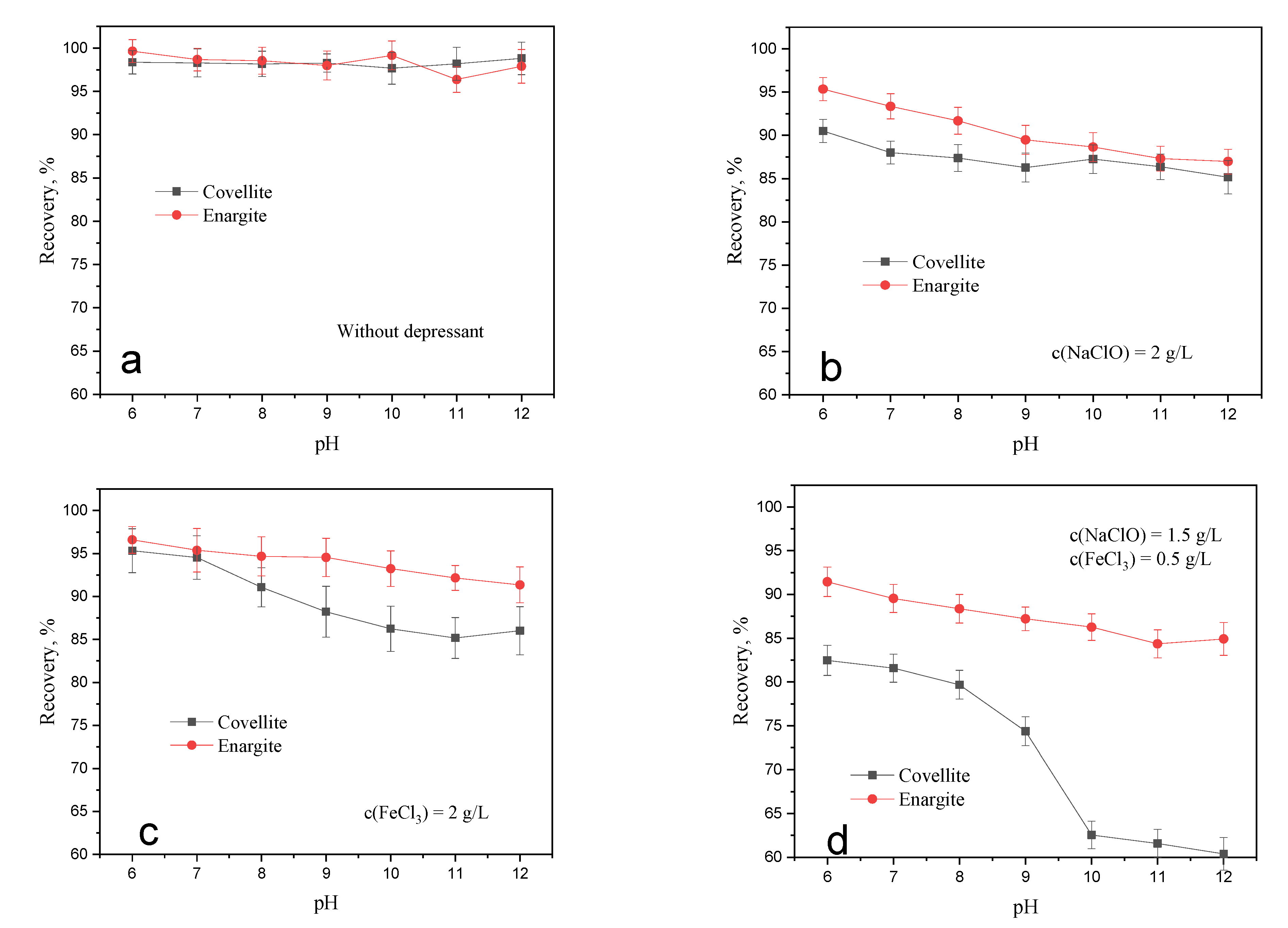
The floatability of sulfide minerals is affected by pH. The flotation tests of fresh covellite and enargite were performed at different pH levels. In these experiments, the ADD dosage was 1 × 10−5 mol/L, and the conditioning times for NaClO and FeCl3 were 20 and 5 min, respectively. Figure 3a shows the comparable high natural floatability of covellite and enargite in the evaluated pH range, which suggests their difficult separation. In the presence of 2 g/L NaClO, the recoveries of covellite and enargite both gradually decrease with increasing pH. Thus, the weakly acidic environment is more conducive to the separation of covellite and enargite minerals. As shown in Figure 3c, FeCl3 exhibits a similar inhibitory effect to that of NaClO. In addition, covellite is inhibited to a greater extent than enargite in alkaline environments. In the presence of both NaClO and FeCl3, the separation efficiency is enhanced compared to that when either is used alone, especially in alkaline environments. Therefore, the separation of covellite and enargite can be realized when NaClO and FeCl3 are used at pH levels greater than 10.
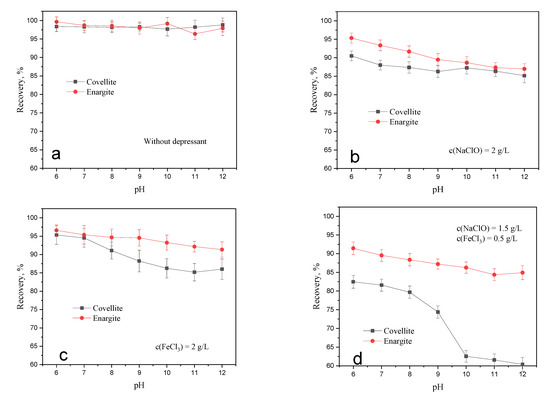
Figure 3.
Effect of oxidation treatment on the recovery of covellite and enargite at different pH levels: (a) without depressant, (b) NaClO, (c) FeCl3, (d) NaClO + FeCl3.
3.1.2. Effect of Depressant Dosage Ratio on the Separation of Covellite and Enargite
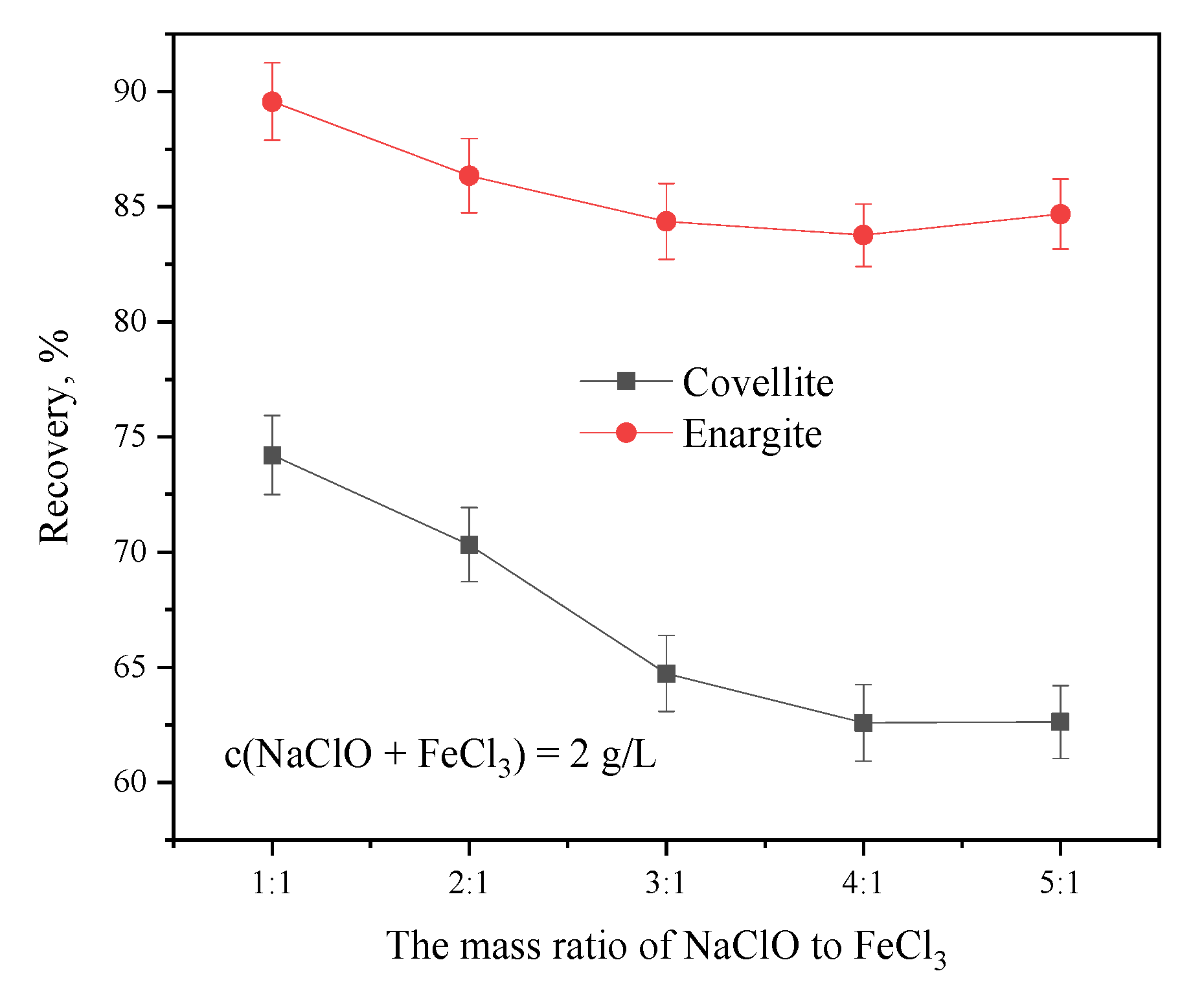
The combination of agents and their ratios play an important role in flotation [20]. Figure 4 shows the considerable effect of the NaClO-to-FeCl3 mass ratio on the floatability of covellite and enargite. For covellite, as the NaClO-to-FeCl3 ratio increases from 1:5, the recovery gradually decreases, reaching the lowest value of 63.67% at a ratio of 4:1. Similarly, the recovery of enargite decreases as the ratio increases; however, it is less affected than that of covellite. Thus, the optimum dosage ratio of NaClO to FeCl3 for the flotation separation of covellite and enargite was 4:1.
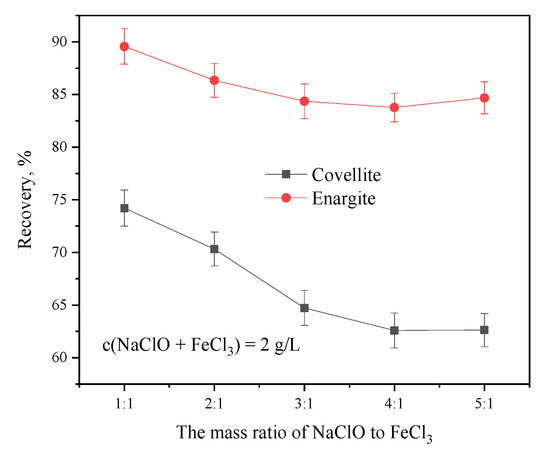
Figure 4.
Separation efficiency as a function of the mass ratios of NaClO to FeCl3 at pH = 10.
3.1.3. Effect of the Depressant Dosage on the Flotation Separation of Covellite and Enargite
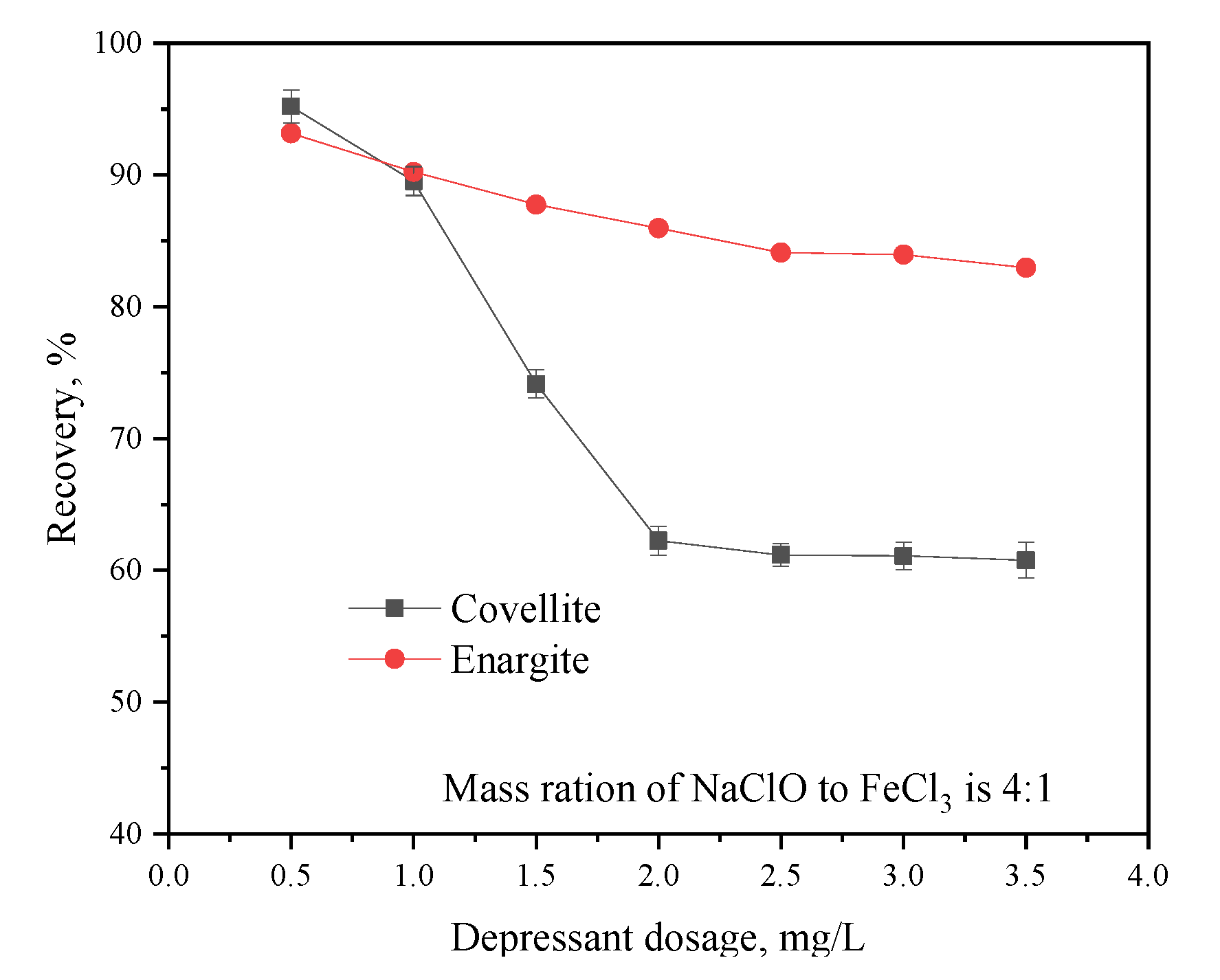
The effect of the depressant dosage on the flotation separation of covellite and enargite is illustrated in Figure 5. The recovery of enargite is significantly affected by the depressant dosage; as the dosage increases from 0.5 to 2 g/L, the recovery of covellite decreases sharply, while it decreases slowly with higher depressant dosages. However, the depressant dosage has no distinct effect on the recovery of enargite. In particular, when the dosage is increased from 0.5 to 3.5 g/L, the enargite recovery slowly decreases from 93.63% to 82.97%. When the depressant dosage exceeds 2 g/L, the recoveries of the two minerals show negligible changes. Therefore, the optimum depressant dosage for covellite and enargite separation was 2 g/L.
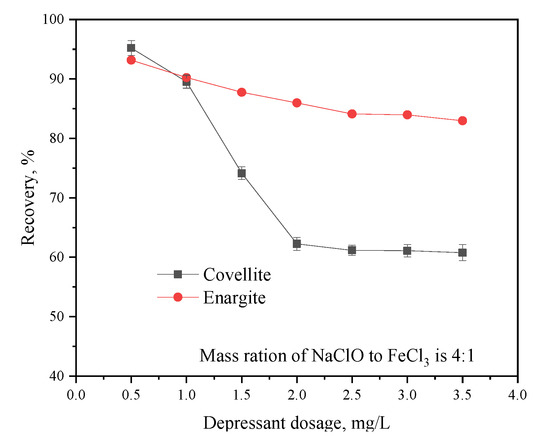
Figure 5.
Effect of the depressant dosage on the flotation behavior of covellite and enargite at pH = 10.
3.1.4. Effects of the Conditioning Times of Depressants on the Flotation Separation of Covellite and Enargite
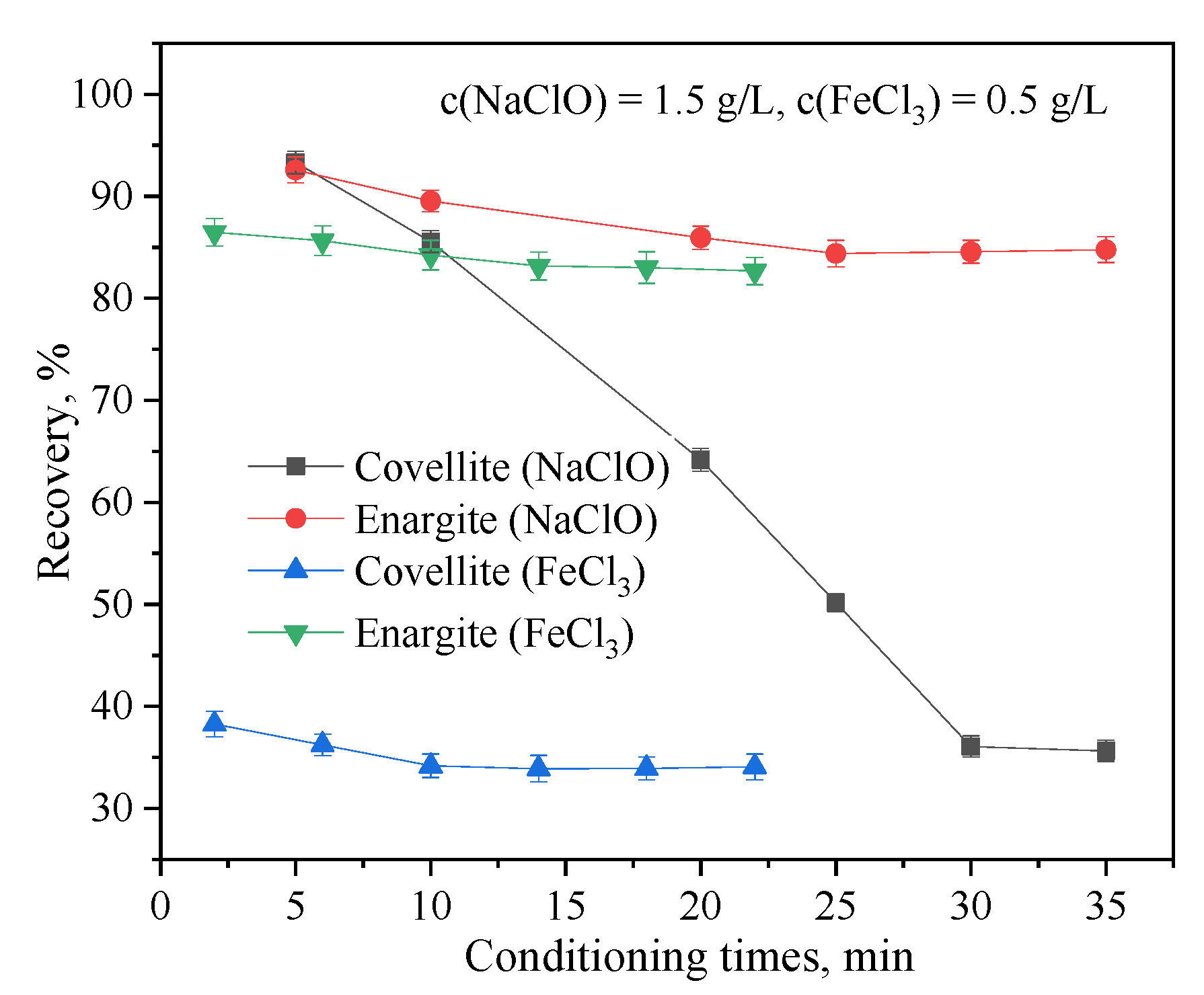
The effects of the NaClO and FeCl3 conditioning times on the flotation separation of covellite and enargite are shown in Figure 6. The conditioning time of NaClO has a significant impact on the separation. In particular, as the conditioning time of NaClO is increased, the recovery of covellite rapidly decreases, whereas that of enargite decreases minimally. Moreover, the conditioning time of FeCl3 had a lesser effect on the separation of covellite and enargite. The best separation results were obtained with NaClO and FeCl3 conditioning times of 30 and 10 min, respectively.
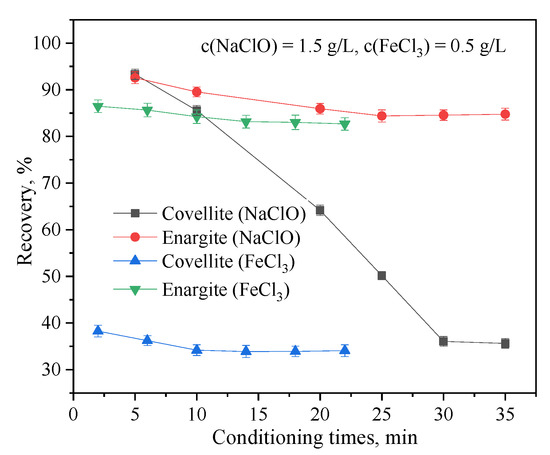
Figure 6.
Effect of the depressant conditioning time on the flotation behavior of covellite and enargite at pH = 10.
3.2. Separation Mechanism of Depressants
3.2.1. XPS Analysis
The elemental composition and chemical state are important indices for evaluating the surface modification of minerals by oxidizers [21]. In this study, the oxidation product species formed on the covellite and enargite surfaces using different depressants were analyzed by XPS. The atomic concentrations and species distributions were calculated through peak fitting and deconvolution. The assignments of Cu, As, O, and S were performed according to references. Table 1 shows the atomic concentrations of the covellite and enargite surfaces.

Table 1.
Atomic concentrations (at%) of the covellite and enargite surfaces with different depressants.
As shown in Table 1, in the absence of depressant treatment (denoted by Fresh), the atomic concentrations on the covellite surface of O, S, Cu, and As are 25.23, 52.46, 22.31, and 0 at%, respectively. In contrast, the atomic concentrations on the enargite surface of O, S, Cu, and As are 11.87, 42.18, 28.14, and 17.81 at%, respectively. For the samples treated with NaClO alone, much more O is detected on the covellite and enargite surfaces. After the oxidation of the sulfide minerals, metal oxides are formed on the mineral surface, which can be used to evaluate their degree of oxidation and hydrophilicity. The O content on the covellite surface is higher than that on the enargite surface, indicating the presence of more hydrophilic species on covellite, which is consistent with the flotation results [22]. When FeCl3 and NaClO are used as the depressants, the O content on covellite further increases to 35.89 at%, whereas the Cu and S contents decrease. Moreover, Fe with a concentration of 7.29 at% is detected on the covellite surface. After the addition of FeCl3, the O concentration on the enargite surface increases from 12.76 to 18.8 at%. Although Fe is detected on the enargite surface, its concentration is considerably lower than that on the covellite surface.
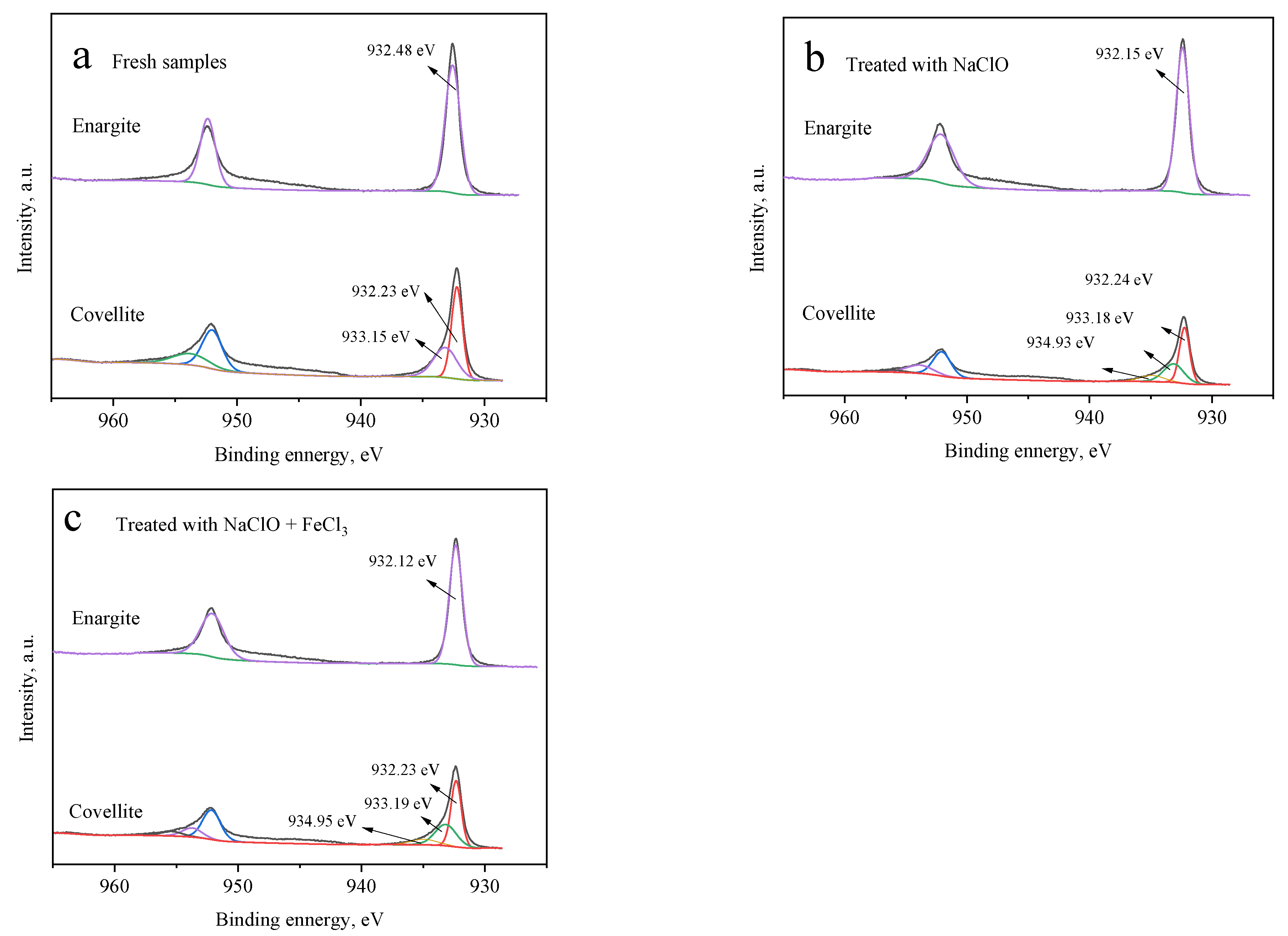
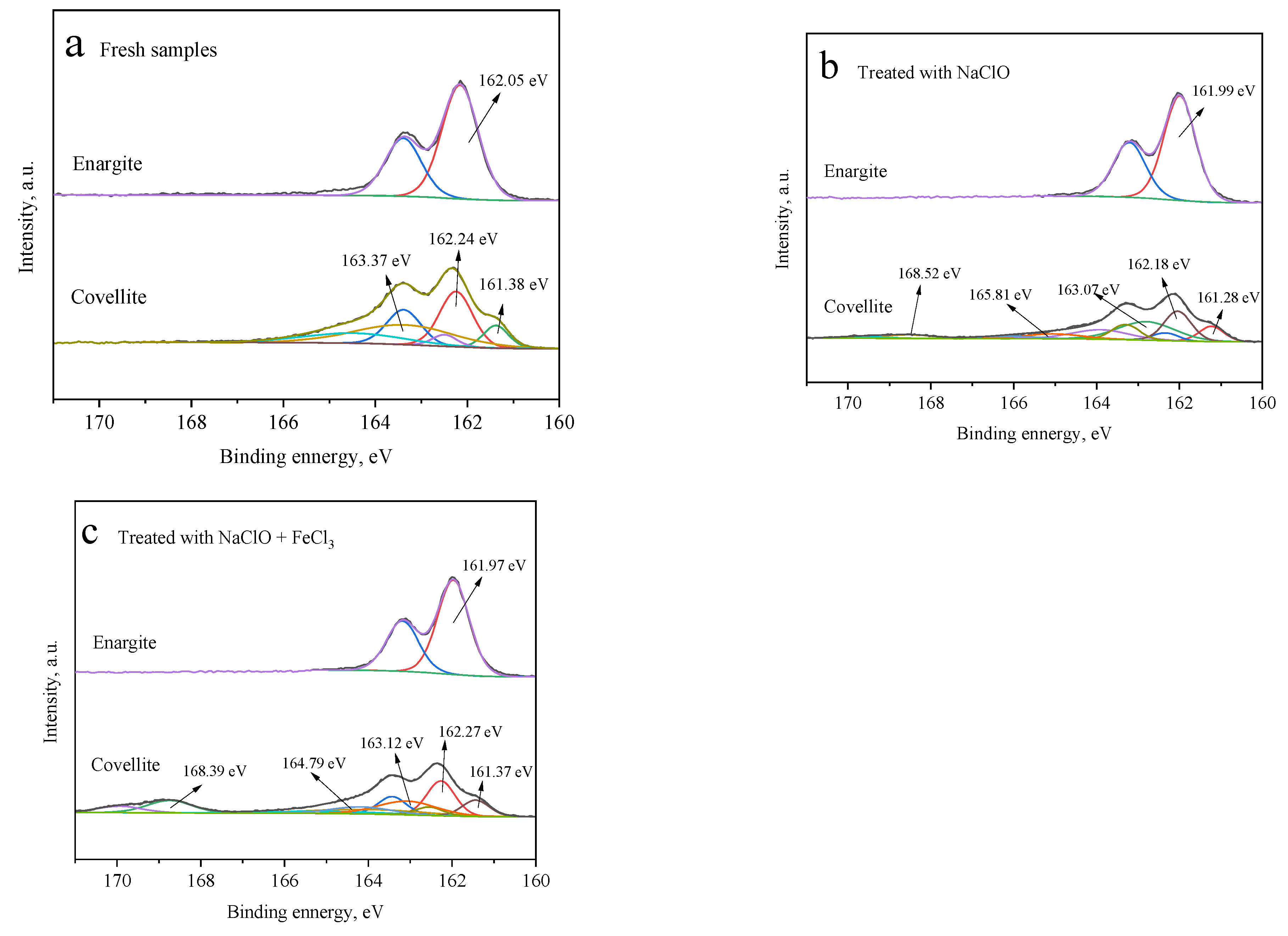
Figure 7 presents the Cu 2p3/2 XPS profiles of the covellite and enargite samples with and without depressant treatment. For fresh enargite (Figure 7a), a pair of peaks is noted at 932.48 and 954.62 eV, which are assigned to copper sulfide in enargite [15]. In the presence of NaClO (Figure 7b) alone or NaClO + FeCl3 (Figure 7c), the spectrum changes negligibly and no other peaks appear, indicating that there are no other copper species on the enargite surface. This is attributed to the copper species produced by the oxidation being released into the solution, which was also reported by Yu et al. [23]. For covellite, in addition to the peaks of the main mineral, other peaks are noted, indicating that the mineral surfaces are covered by other copper species. The small peaks at 934.93 eV (Figure 7b) and 934.95 eV (Figure 7c) after treatment with NaClO alone and NaClO + FeCl3, respectively, can be attributed to the copper oxides of CuO, CuSO4, and Cu(OH)2 [24]. In the presence of FeCl3, the components of the curve for covellite hardly change compared with those processed with NaClO alone.
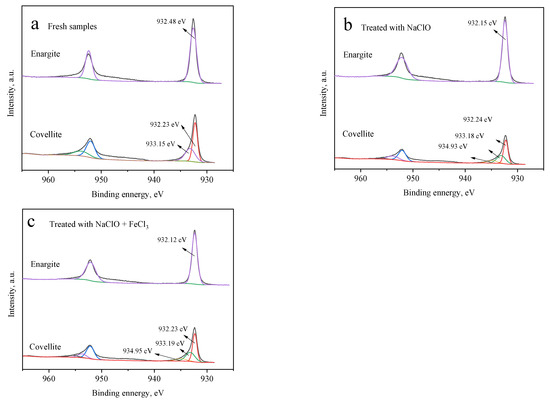
Figure 7.
Cu 2p3/2 spectra of the covellite and enargite samples treated with different depressants: (a) fresh samples, (b) NaClO, and (c) NaClO + FeCl3.
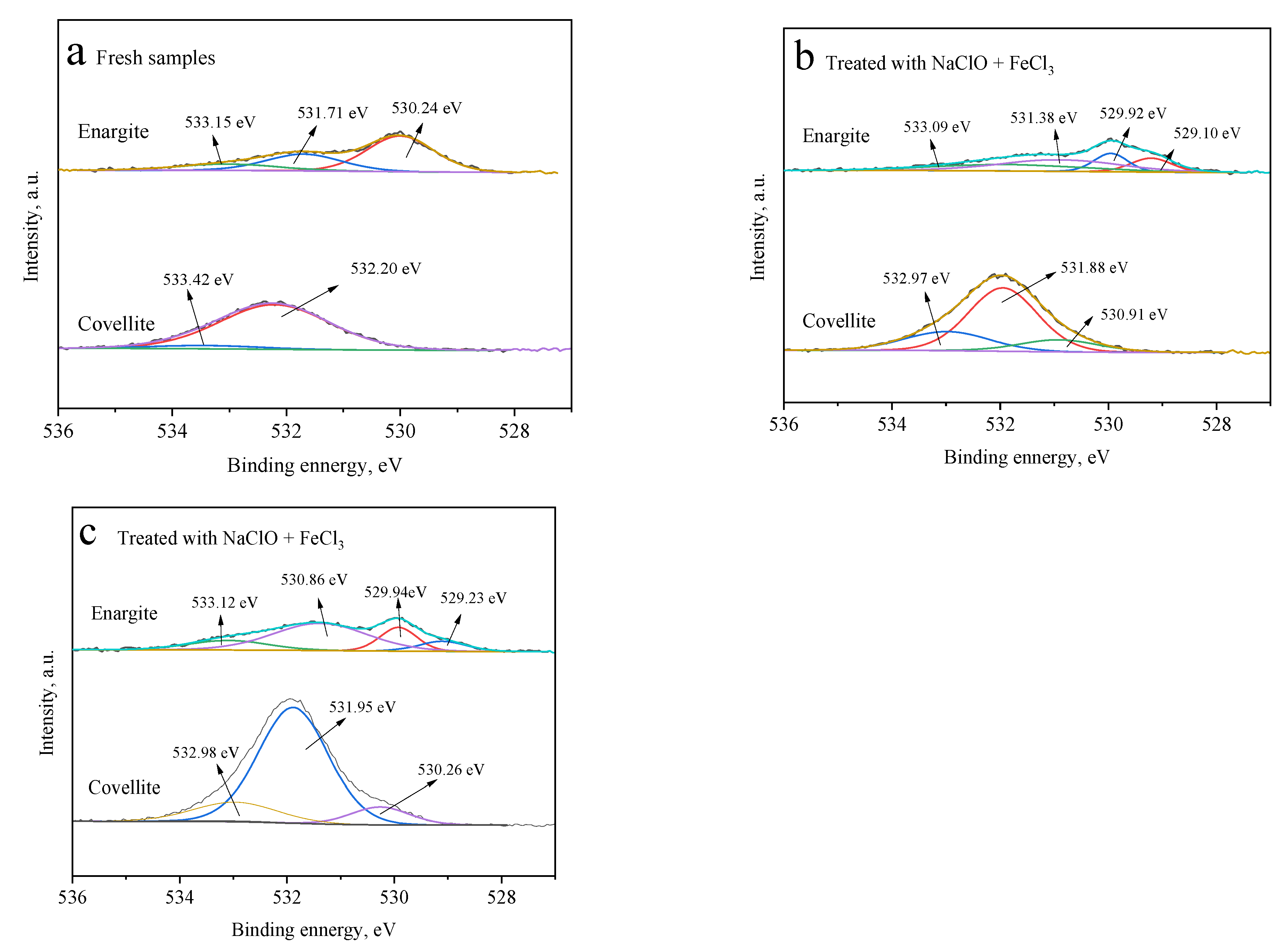
Figure 8 presents the different O species on the surfaces of covellite and enargite. After treatment with NaClO, three types of O species are observed on the covellite surface, namely, oxygen from carbon–oxygen contaminants, in the attached water, and from the oxide species of CuO or Cu(OH)2, which are located at 532.97, 531.88, and 530.91 eV, respectively [25,26]. Compared with fresh covellite, it can be inferred that these copper oxide species are produced by the oxidation of NaClO. In the presence of FeCl3, the amount of O in the form of CuO or Cu(OH)2 further increases. These results indicate that NaClO and FeCl3 have a strong oxidizing effect on the covellite surface, thereby decreasing the floatability.
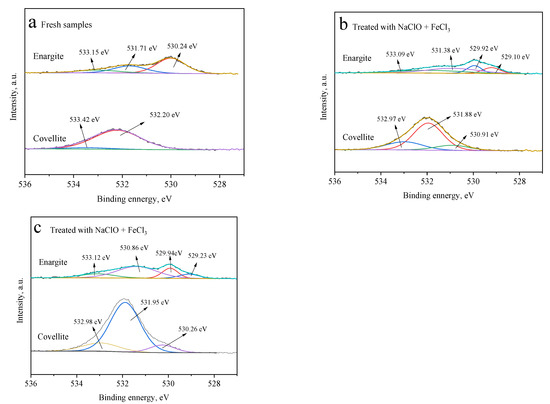
Figure 8.
O 1s spectra of the covellite and enargite samples treated with different depressants: (a) fresh samples, (b) NaClO, and (c) NaClO + FeCl3.
The O 1s spectrum of fresh enargite (Figure 8a) exhibits three well-separated peaks at 533.15, 531.78, and 530.24 eV. After treatment with NaClO alone, a new peak appears at 529.1 eV. After the treatment with NaClO + FeCl3, the oxide species on the enargite surface become more complex than those on the covellite. The binding energies at 528–531 and 531–532 eV are assigned to O in metal oxides and hydroxides, respectively [27,28]. However, these oxides do not significantly increase the hydrophobicity of enargite owing to the low atomic concentrations of O on the enargite surface.
With or without depressants, only one pair of characteristic peaks is noted in the S 2p3/2 spectra of enargite (Figure 9), indicating the absence of sulfur oxidation products on the surface. However, five pairs of peaks are noted at 161.28 ± 0.2, 162.1 ± 0.2, 163 ± 0.2, 165 ± 0.2, and 168 ± 0.2 eV on the covellite surface, which are assigned to S2−, S22−, S0/Sn2−, SO32−, and SO42−, respectively [15,29]. Hydrophilic sulfides of SO32− and SO42− are important for the hydrophilicity of covellite surfaces. After the treatment with NaClO alone, the sulfur oxides account for 46.13 at% of the total sulfur content. When FeCl3 is added, this proportion further increases to 54.35 at%.
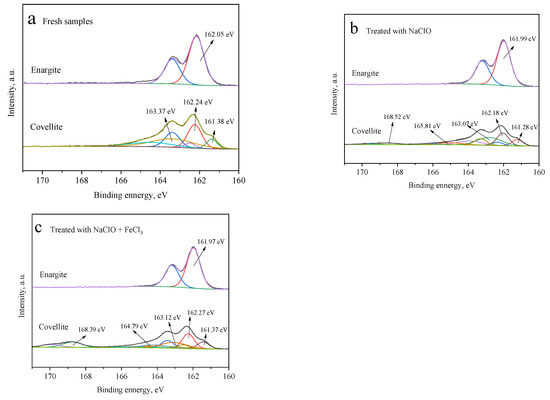
Figure 9.
S 2p3/2 spectra of the covellite and enargite samples treated with different depressants: (a) fresh samples, (b) NaClO, and (c) NaClO + FeCl3.
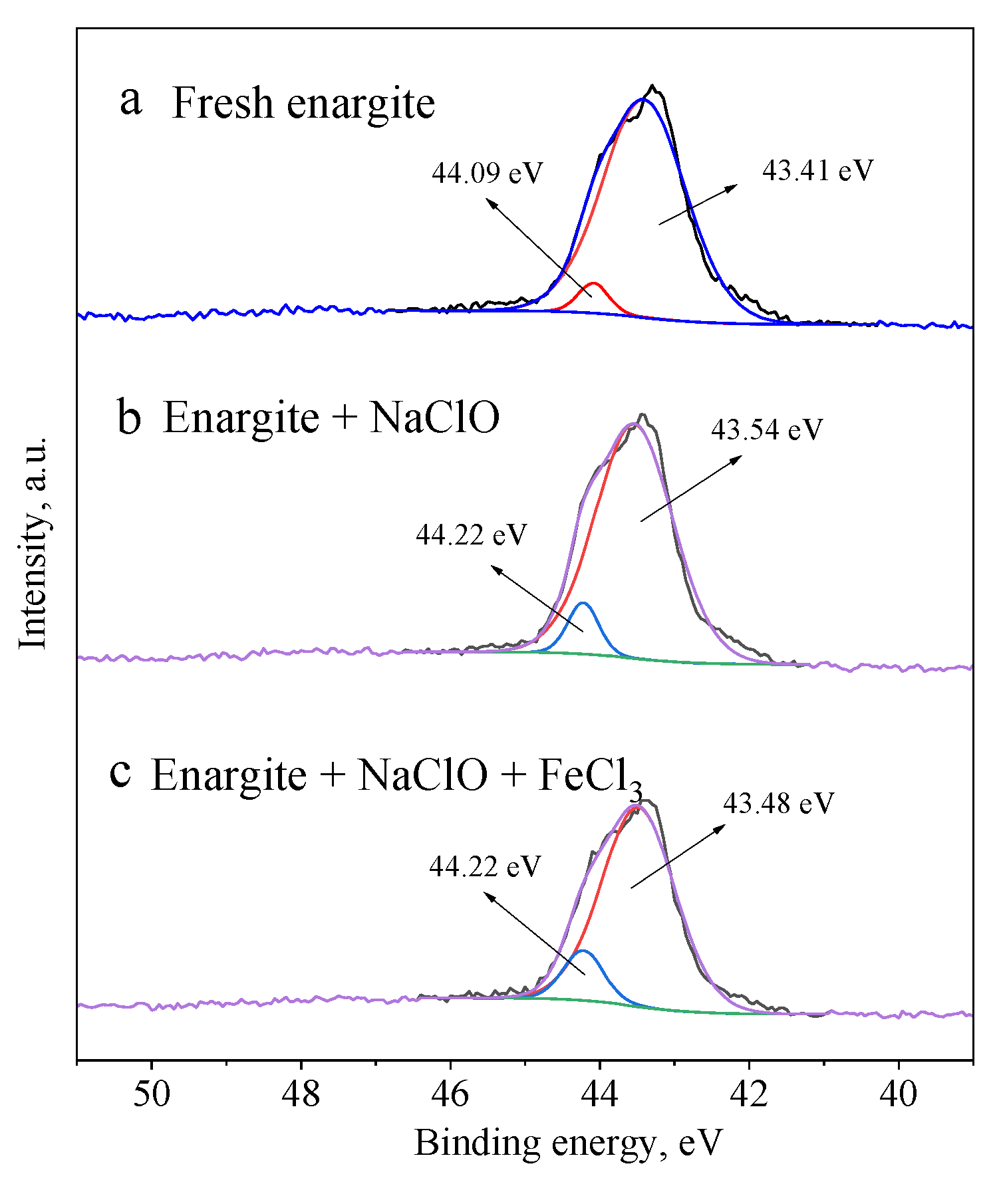
Figure 10 shows the As 3d spectra of enargite with different depressants. The peak at 43.5 ± 0.2 eV corresponds to the As in the bulk enargite, whereas the peak at 44.15 ± 0.1 eV is assigned to arsenic oxide [30,31]. Despite the production of arsenic oxides on the mineral surface, the arsenic oxides account for only 10.1 at% of the total As content in the presence of NaClO and FeCl3.
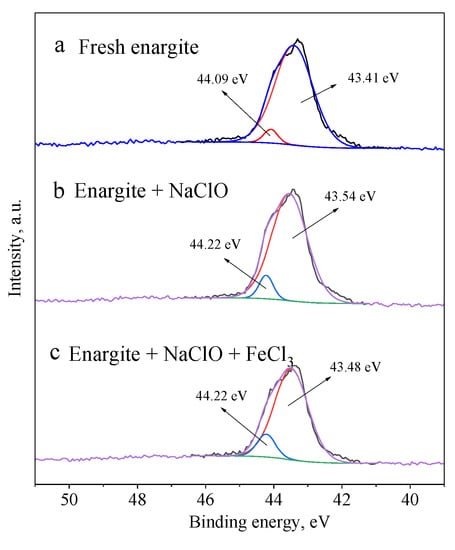
Figure 10.
As 3d spectra of the enargite sample treated with different depressants: (a) fresh sample, (b) NaClO, and (c) NaClO + FeCl3.
3.2.2. SEM Analysis
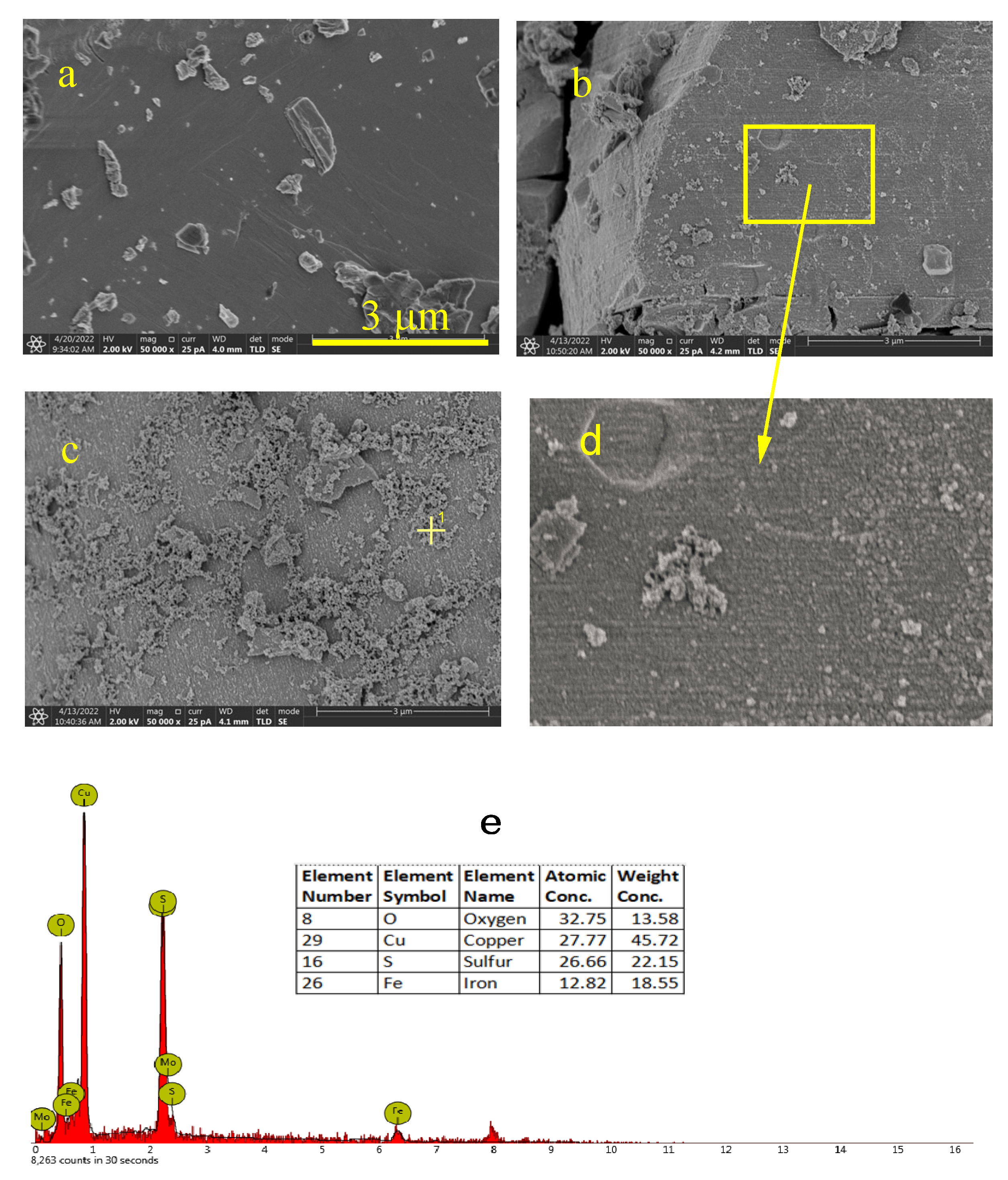
The surface morphologies of the covellite samples after treatment with different depressants were observed using SEM; the results are shown in Figure 11. Before the depressant treatment, fresh covellite exhibits a relatively uniform surface, except for small debris adhering to the surface. In the presence of NaClO, the surface becomes rough, and finely divided kidney-like substances appear, which can be ascribed to recrystallized oxidation products, indicating the occurrence of chemical attack. After the addition of FeCl3, the mineral surface is covered by a large amount of flocs. The EDS results (Figure 11e) indicate that these flocs contain iron and oxygen, and it can be inferred that these flocs are iron hydroxides generated from iron ions in an alkaline solution.
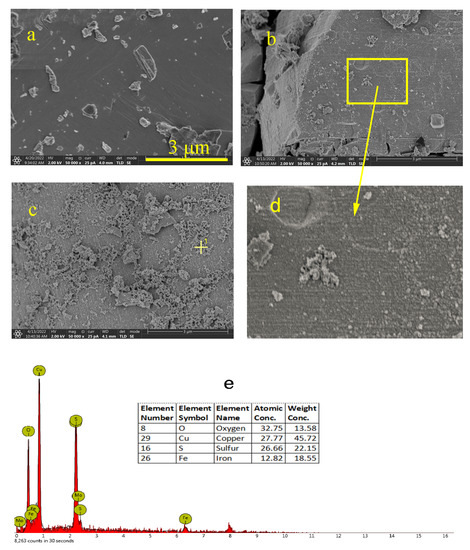
Figure 11.
SEM images of the covellite sample: (a) fresh sample, (b) sample treated with NaClO, (c) sample treated with NaClO and FeCl3, (d) Partial distribution of (b), (e) EDS result.
Figure 12 shows the surface morphologies of the enargite samples after treatment with different depressants. Similar to covellite, fresh enargite has a smooth surface. After reacting with NaClO, small caves appear owing to chemical attack. In contrast to covellite, finely divided kidney-like substances are not observed on the enargite surface in the presence of NaClO, which is consistent with the larger number of oxides on the covellite surface than on the enargite surface, as indicated by XPS. Moreover, the amount of iron hydroxide on the enargite surface is considerably lower than that on the covellite surface. These conclusions explain the higher susceptibility to the inhibition of covellite than to that of enargite under treatment with depressants.

Figure 12.
SEM images of the enargite samples: (a) fresh sample, (b) sample treated with NaClO, (c) sample treated with NaClO and FeCl3, (d) Partial distribution of (b), (e) EDS result.
3.2.3. Contact Angle Measurements
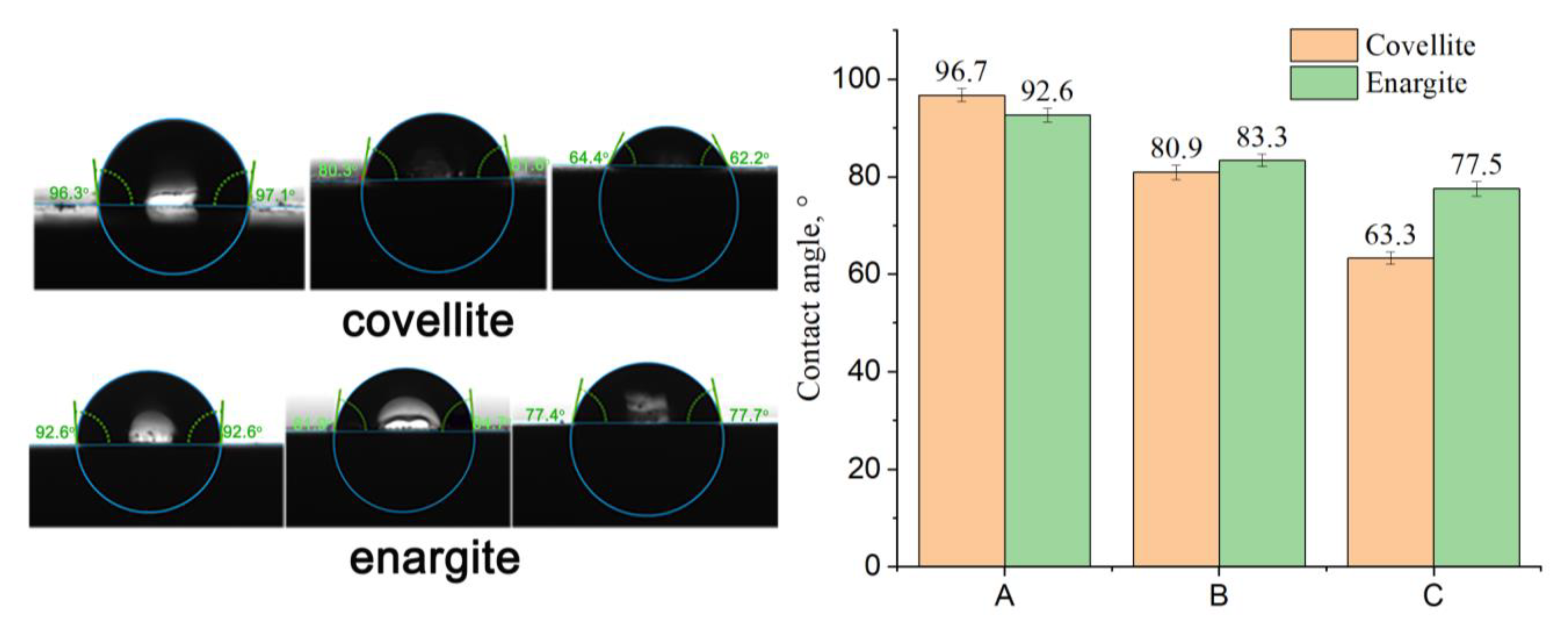
The contact angle was measured to evaluate the hydrophobicity of the mineral surface. As shown in Figure 13, the contact angles of the covellite and enargite samples are 96.7° and 92.6° after treatment with ADD, respectively, indicating good floatability. After the addition of NaClO, the contact angles of covellite and enargite decrease minimally. In the presence of FeCl3, the contact angles further decrease, and the difference between the contact angles of covellite and enargite increases from +4.1 to −14.2°. This demonstrates that the addition of depressants increases the difference in the hydrophobicity of the covellite and enargite surfaces, which is consistent with the flotation results.
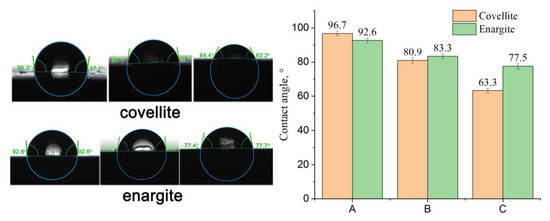
Figure 13.
Effect of the different reagents added on the contact angles of covellite and enargite (A: sample + 1 × 10−5 mol/L ADD; B: sample + 2 g/L NaClO + 1 × 10−5 mol/L ADD; and C: sample + 1.5 g/L NaClO + 0.5 g/L FeCl3 + 1 × 10−5 mol/L ADD).
3.2.4. Bench-Scale Flotation Tests
A copper bulk concentrate containing enargite was obtained by flotation using ADD as the collector. The flowsheet of the bench-scale flotation of the copper bulk concentrate tests is shown in Figure 2, and the results are presented in Table 2. Single-factor optimizing tests, including agitation times and reagent consumption, were performed before the open-circuit flotation experiments. Table 2 shows that the copper bulk concentrate is successfully separated into two products: low-arsenic-containing (As grade of 0.46%) and high-arsenic-containing (As grade of 5.18%) copper concentrates. This demonstrates the successful separation of copper sulfide minerals and arsenic-bearing copper minerals using oxidation treatment.

Table 2.
Results of open-circuit flotation separation of the bulk copper concentrate.
3.3. Discussion
Oxidation is one of the main methods used to inhibit copper sulfide minerals. The main inhibition mechanism is the formation of metal oxides on the mineral surface, which can reduce their floatability [32,33]. Although NaClO and FeCl3 are strong oxidants that can react with both covellite and enargite surfaces and increase their O species, more O species were generated on the covellite surface than on the enargite surface, resulting in the different floatability of these minerals. These O species are mainly composed of CuO and Cu(OH)2. SEM analysis revealed that finely divided kidney-like substances appeared on the covellite surface after the addition of NaClO, but not on the enargite surface. This is because most of the generated O species on the enargite surface dissolved, while only a small part of the oxide on the surface of covellite dissolved. In addition, because FeCl3 is not only an oxidant but also a flocculator, hydrophilic ferric hydroxide colloids are readily formed in solution [34]. The selective adsorption of ferric hydroxide on the covellite and enargite surfaces further exacerbated the difference in the floatability of the two minerals, thereby allowing their separation. Bench-scale flotation tests further showed that oxidation treatment is feasible to reduce the arsenic content in copper concentrates. However, at present, more research is needed on how to reduce the agent cost.
4. Conclusions
Herein, the effect of oxidation treatment using NaClO flow with FeCl3 on the flotation separation of covellite and enargite was systematically investigated. Micro-flotation tests, contact angle measurements, bench-scale flotation tests, XPS, and SEM were used to characterize the separation of the minerals. The results demonstrate the successful separation of enargite from covellite through oxidation treatment using NaClO and FeCl3. The main conclusions of this study are as follows:
- Micro-flotation and contact angle measurements indicated that the addition of NaClO followed by FeCl3 increased the hydrophobicity difference between covellite and enargite.
- Bench-scale flotation tests indicated that the bulk copper concentrate could be separated into two products: low- (As grade of 0.46%) and high-arsenic-containing (As grade of 5.18%) copper concentrates.
- XPS and SEM analyses established that the oxidization treatment of NaClO could lead to the accumulation of oxides on the covellite surface, but not on the enargite surface.
- The presence of FeCl3 caused varying precipitation of ferric hydroxide on the surfaces of covellite and enargite, which exacerbated the difference in the hydrophilicity of these minerals.
Author Contributions
Q.L. conceived and designed the experiments; Y.G. performed the experiments, analyzed the data, and wrote the paper; R.D. contributed reagents/materials/analysis tools. All authors have read and agreed to the published version of the manuscript.
Funding
This work was financially supported by the National Natural Science Foundation of China (Grant No. 51804080).
Acknowledgments
The authors would like to thank the Testing Center of Fuzhou University for the testing support.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Safarzadeh, M.S.; Moats, M.S.; Miller, J.D. Recent Trends in the Processing of Enargite Concentrates. Miner. Process. Extr. Metall. Rev. 2014, 35, 283–367. [Google Scholar] [CrossRef]
- Yu, L.; Liu, Q.J.; Li, S.M.; Deng, J.S.; Luo, B.; Lai, H. Depression mechanism involving Fe3+ during arsenopyrite flotation. Sep. Purif. Technol. 2019, 222, 109–116. [Google Scholar] [CrossRef]
- Plackowski, C.; Nguyen, A.V.; Bruckard, W.J. A critical review of surface properties and selective flotation of enargite in sulphide systems. Miner. Eng. 2012, 30, 1–11. [Google Scholar] [CrossRef]
- Tayebi-Khorami, M.; Manlapig, E.; Forbes, E.; Bradshaw, D.; Edraki, M. Selective flotation of enargite from copper sulphides in Tampakan deposit. Miner. Eng. 2017, 112, 1–10. [Google Scholar] [CrossRef]
- Fornasiero, D.; Fullston, D.; Li, C.; Ralston, J. Separation of enargite and tennantite from non-arsenic copper sulfide minerals by selective oxidation or dissolution. Int. J. Miner. Process. 2001, 61, 109–119. [Google Scholar] [CrossRef]
- Long, G.; Peng, Y.; Bradshaw, D. A review of copper–arsenic mineral removal from copper concentrates. Miner. Eng. 2012, 36, 179–186. [Google Scholar] [CrossRef]
- García-Garnica, R.; Castillo-Magallanes, N.; Rodríguez, I.; Cruz, R.; Lázaro, I. Electrochemical study of enargite within the mixed potential zone attained with different oxidizing reagents in an alkaline medium. Electrochim. Acta 2022, 425, 140719. [Google Scholar] [CrossRef]
- Castro, S.H.; Baltierra, L.; Munoz, P. Depression of enargite by magnesiumammonium mixtures. In Proceedings of the Copper 2003–Cobre 2003 the 5th International Conference vol. III–Mineral Processing, Santiago, Chile, 30 November–3 December 2003; pp. 257–269. [Google Scholar]
- Menacho, J.M.; Aliaga, W.; Valenzuela, R.; Ramos, V.; Olivares, I. Selective flotation of enargite and chalcopyrite. Minerals 1993, 48, 33–39. [Google Scholar]
- Pauporté, T.; Schuhmann, D. An electrochemical study of natural enargite under conditions relating to those used in flflotation of sulphide minerals. Colloids Surf. A Physicochem. Eng. Asp. 1996, 111, 1–19. [Google Scholar] [CrossRef]
- Asbjornsson, J.; Kelsall, G.H.; Pattrick, R.A.D.; Vaughan, D.J.; Wincott, P.L.; Hope, G.A. Electrochemical and surface analytical studies of enargite in acid solution. J. Electrochem. Soc. 2004, 151, 250–256. [Google Scholar] [CrossRef]
- Ma, Y.I.; Yang, Y.; Skinner, W.; Chen, M. Electrochemical and spectroscopic analysis of enargite (Cu3AsS4) dissolution mechanism in sulfuric acid solution. Hydrometallurgy 2020, 194, 105346. [Google Scholar] [CrossRef]
- Guo, H.; Yen, W.T. Electrochemical study of synthetic and natural enargites. Proc. Int. Miner. Process. Congr. 2008, 24, 1138–1145. [Google Scholar]
- Yepsen, R.; Gutierrez, L. Effect of Eh and pH on the flotation of enargite using seawater. Miner. Eng. 2020, 159, 106612. [Google Scholar] [CrossRef]
- Suyantara, G.; Hirajima, T.; Miki, H.; Sasaki, K.; Kuroiwa, S.; Aoki, Y. Effect of H2O2 and potassium amyl xanthate on separation of enargite and tennantite from chalcopyrite and bornite using flotation. Miner. Eng. 2020, 152, 106371. [Google Scholar] [CrossRef]
- Suyantara, G.P.W.; Hirajima, T.; Miki, H.; Sasaki, K.; Kuroiwa, S.; Aoki, Y. Effect of Na2SO3 on the floatability of chalcopyrite and enargite. Miner. Eng. 2021, 173, 107222. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhao, H.B.; Abashina, T.; Vainshtein, M. Review on arsenic removal from sulfide minerals: An emphasis on enargite and arsenopyrite. Miner. Eng. 2021, 172, 107113. [Google Scholar] [CrossRef]
- Sasaki, K.; Takatsugi, K.; Ishikura, K.; Hirajima, T. Spectroscopic study on oxidative dissolution of chalcopyrite, enargite and tennantite at different pH values. Hydrometallurgy 2010, 100, 144–151. [Google Scholar] [CrossRef]
- Tayebi-Khorami, M.; Manlapig, E.; Forbes, E.; Edraki, M.; Bradshaw, D. Effect of surface oxidation on the flotation response of enargite in a complex ore system. Miner. Eng. 2018, 119, 149–155. [Google Scholar] [CrossRef]
- Deng, R.D.; Yang, X.F.; Hu, Y.; Ku, J.G.; Zuo, W.R.; Ma, Y.Q. Effect of Fe(II) as assistant depressant on flotation separation of scheelite from calcite. Miner. Eng. 2018, 118, 133–140. [Google Scholar] [CrossRef]
- Nowak, P.; Laajalehto, K. Oxidation of galena surface–an XPS study of the formation of sulfoxy species. Appl. Surf. Sci. 2000, 157, 101–111. [Google Scholar] [CrossRef]
- Wu, D.D.; Wen, S.M.; Deng, J.S.; Liu, J.; Mao, Y.B. Study on the sulfidation behavior of smithsonite. Appl. Surf. Sci. 2015, 329, 315–320. [Google Scholar] [CrossRef]
- Yu, J.; Huang, W.L.; Wang, B.; Yuan, Y.; Fang, Z.; Cui, Y. Arsenic Removal and Solidification from Enargite. Chin. J. Rare Met. 2018, 42, 1093–1102. [Google Scholar]
- Hirajima, T.; Miki, H.; Suyantara, G.P.W.; Matsuoka, H.; Elmahdy, A.M.; SasakI, K.; Imaizumi, Y.; Kuroiwa, S. Selective flotation of chalcopyrite and molybdenite with H2O2 oxidation. Miner. Eng. 2017, 100, 83–92. [Google Scholar] [CrossRef]
- McIntyre, N.S.; Zetaruk, D.G. X-ray photoelectron spectroscopic studies of iron oxides. Anal. Chem. 1977, 49, 1521–1529. [Google Scholar] [CrossRef]
- Xiong, X.L.; Hua, X.M.; Zheng, Y.F.; Lu, X.G.; Li, S.G.; Cheng, H.W.; Xu, Q. Oxidation mechanism of chalcopyrite revealed by X-ray photoelectron spectroscopy and first principles studies. Appl. Surf. Sci. 2018, 427, 233–241. [Google Scholar] [CrossRef]
- Tan, B.J.; Klabunde, K.J.; Sherwood, P.M.A. X-ray photoelectron spectroscopy studies of solvated metal atom dispersed catalysts. Monometallic iron and bimetallic iron-cobalt particles on alumina. Chem. Mater. 1990, 2, 186–191. [Google Scholar] [CrossRef]
- Nesbitt, H.W.; Muir, I.J.; Prarr, A.R. Oxidation of arsenopyrite by air and air-saturated, distilled water, and implications for mechanism of oxidation. Geochim. Cosmochim. Acta 1995, 59, 1773–1786. [Google Scholar] [CrossRef]
- Iranmahboob, J.; Gardner, S.D.; Toghiani, H.; Hill, D.O. XPS study of molybdenum sulfide catalyst exposed to CO and H2. J. Colloid Interface Sci. 2004, 270, 123–126. [Google Scholar] [CrossRef]
- Parada, F.; Jeffrey, M.I.; Asselin, E. Leaching kinetics of enargite in alkaline sodium sulphide solutions. Hydrometallurgy 2014, 146, 48–58. [Google Scholar] [CrossRef]
- Rivera-Vasquez, B.F.; Dixon, D. Rapid atmospheric leaching of enargite in acidic ferric sulfate media. Hydrometallurgy 2015, 152, 149–158. [Google Scholar] [CrossRef]
- Nicol, M.J.; Tjandrawan, V.; Zhang, S.C. Cathodic reduction of iron(III) and copper(II) on various sulfide minerals in chloride solutions. Hydrometallurgy 2016, 166, 113–122. [Google Scholar] [CrossRef]
- Yin, Q.; Vaughan, D.J.; England, K.E.R.; Kelsall, G.H. Electrochemical Oxidation of Covellite (CuS) in Alkaline Solution. J. Colloid Interface Sci. 1994, 166, 133–142. [Google Scholar] [CrossRef]
- Deng, R.D.; Hu, Y.; Ku, J.G.; Zuo, W.R.; Yang, Z.G. Adsorption of Fe(III) on smithsonite surfaces and implications for flotation. Colloids Surf. A Physicochem. Eng. Asp. 2017, 533, 308–315. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).